Abstract
Multicentric cutaneous neuroendocrine (Merkel cell) carcinoma was diagnosed in a 5-year-old castrated male Keeshond dog with multiple firm nodular cutaneous masses. The neoplastic tissue locally effaced the periadnexal and deep dermis and consisted of densely cellular confluent clusters of round to polygonal cells supported by a delicate fibrovascular stroma. The cells were moderately immunoreactive with chromogranin A, synaptophysin, and cytokeratin. Ultrastructurally, the cells had characteristic membrane-bound dense-core neuroendocrine granules approximately 120 nm in diameter and randomly dispersed throughout the cytoplasm. Effacement of dermal structures and multicentric distribution suggested low-grade malignant phenotype. These findings contrast with the typical benign behavior of canine cutaneous neuroendocrine tumors.
Neuroendocrine (Merkel) cells are widely dispersed throughout the skin, oral cavity, and internal organs. They are considered to be a normal component of the dermis and follicular epithelium, residing at the bulge of the follicular isthmus, and they are believed to play a role in the slow-adapting sensory mechanoreceptor. 7,17 Cutaneous neuroendocrine tumors are relatively uncommon in humans and exceedingly rare in dogs and cats. 2,4,5,7,10 In humans, neuroendocrine carcinomas are regarded as relatively high-grade malignant tumors with frequent metastasis to regional lymph nodes. 11,13 A recent study reported that most human Merkel cell carcinomas contain DNA sequences from a newly identified Merkel cell polyomavirus. 9 Such etiologic associations have not been implicated in domestic animals. Comparatively, the majority of canine cutaneous neuroendocrine tumors are phenotypically benign, and complete surgical excision is generally curative. 17 Local recurrence is rare, and metastasis has been reported in only 1 dog. 2,3 Neuroendocrine tumors are typically reported in older dogs, ranging from 7 to 14 years; however, because of the small number of reported cases (n = 25), breed and sex predilections have not been identifed. 2,8,17 Of the 2 reported feline cases, 1 tumor was considered benign, and the other was biologically similar to human tumors with widespread pulmonary and mediastinal metastasis. 1,10
Neuroendocrine cells arise from embryonic epidermal progenitors and are likely components of the amine precursor uptake and decarboxylation system. 12,16 The cells contain characteristic dense-core neurosecretory granules that are often compartmentalized or irregularly dispersed throughout the cytoplasm. These granules express a variety of peptides and amines, including neuron-specific enolase, chromogranin A, synaptophysin, insulin, gastrin, substance P, and calcitonin—all of which are useful markers that aid in the diagnosis of these tumors. 6 Expression of specific peptides and amines varies among species and with anatomic location, although chromogranin A is always expressed in mammalian Merkel cells. 14 This report describes the histologic features, immunohistochemical profile, and ultrastructural findings of a multicentric cutaneous neuroendocrine carcinoma in a dog. The differential diagnosis is discussed.
Case Presentation
A 5-year-old castrated male Keeshond dog was referred to the Auburn University Small Animal Teaching Hospital for 6 firm nodular cutaneous masses (diameter, 0.5 to 2.5 cm) in the ventral aspect of the neck, dorsolateral thorax, and caudodorsal lumbar region and over both hocks. The dog had been previously diagnosed with sebaceous adenocarcinoma, but there was no evidence of recurrence. Several enlarged abdominal lymph nodes were seen with abdominal ultrasound; however, patterns or features consistent with metastasis were not identified upon thoracic radiography and abdominal echography.
Microscopic Examination
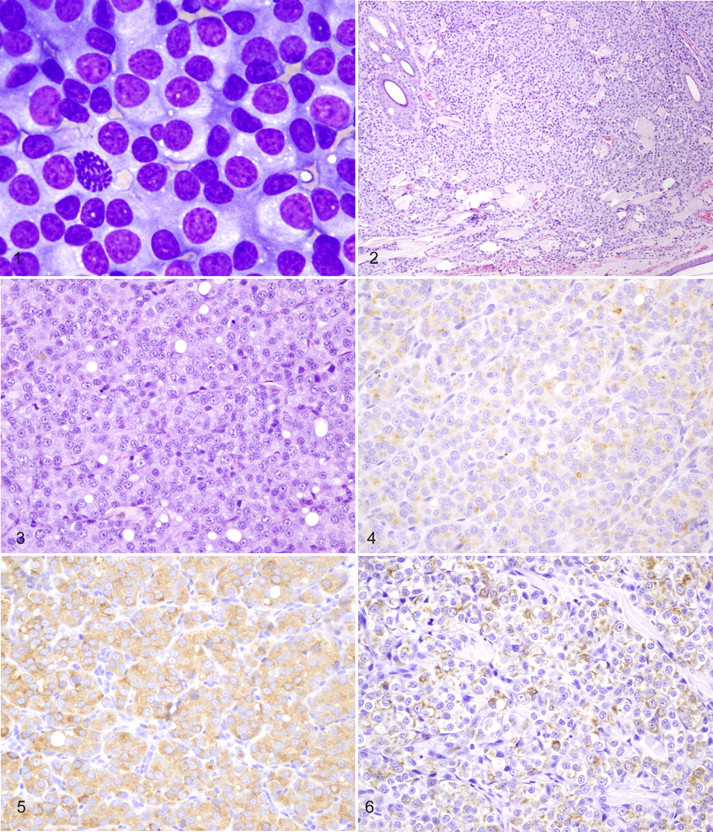
A monomorphic population of neoplastic round to polygonal cells was admixed with many bare nuclei from lysed cells and within a heavy background of proteinaceous material in modified Wright’s stain cytologic preparations from fine-needle aspirates of the cutaneous masses (Fig. 1). There was moderate anisocytosis and anisokaryosis. The cells had round to oval central or eccentric nuclei with coarse chromatin, prominent round nucleoli, moderate amounts of pale basophilic cytoplasm, and well-defined cytoplasmic margins. A moderate number of binucleated cells were seen; mitotic figures were rare.
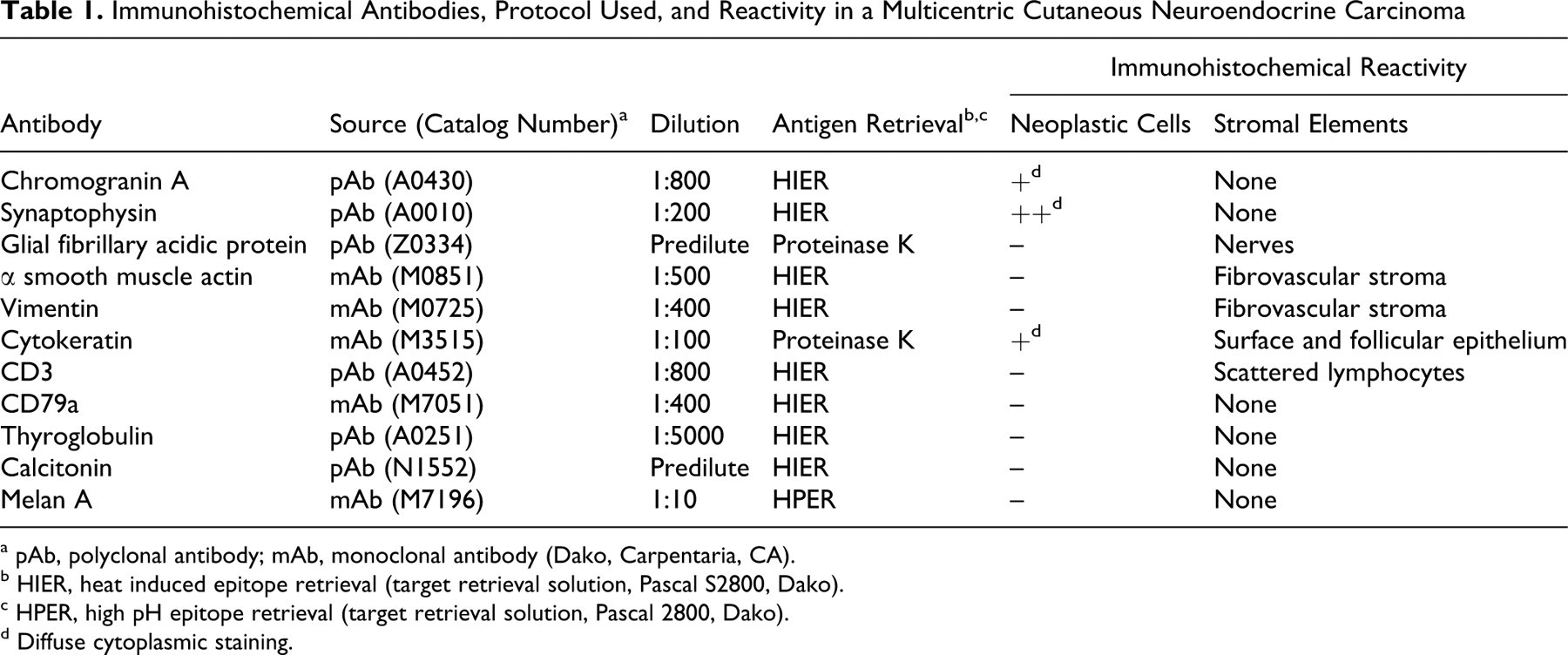
All the masses were excised en bloc, and samples from the masses were routinely processed for histopathology. Additional sections were examined immunohistochemically with a streptavidin–biotin kit (Dako, Carpentaria, CA) and commercially available antisera to chromogranin A, synaptophysin, glial fibrillary acidic protein, α-smooth muscle actin, vimentin, cytokeratin, CD3, CD79a, Melan A, thyroglobulin, and calcitonin, with appropriate control tissues for each antibody. Table 1 summarizes the immunohistochemical antibodies, protocol, and reactivity.
Immunohistochemical Antibodies, Protocol Used, and Reactivity in a Multicentric Cutaneous Neuroendocrine Carcinoma
a pAb, polyclonal antibody; mAb, monoclonal antibody (Dako, Carpentaria, CA).
b HIER, heat induced epitope retrieval (target retrieval solution, Pascal S2800, Dako).
c HPER, high pH epitope retrieval (target retrieval solution, Pascal 2800, Dako).
d Diffuse cytoplasmic staining.
Histologically, locally extensive regions of the periadnexal and deep dermis were effaced by a densely cellular and infiltrative neoplasm composed of solid sheets and cords of monomorphic round to polygonal cells subdivided into confluent packets and clusters by a delicate fibrovascular stroma (Fig. 2). Neoplastic cells had a moderate amount of brightly eosinophilic, granular cytoplasm with variably distinct cell margins (Fig. 3). Nuclei were round and centrally located, with coarsely clumped chromatin and single distinct nucleoli. Anisokaryosis was minimal, and mitotic figures ranged from 0 to 1 per 400× field. Many hair follicles were effaced; remaining follicles were surrounded by neoplastic cells.
Immunohistochemically, the neoplastic cells had diffuse, granular cytoplasmic and membrane reactivity with antisera to chromogranin A (Fig. 4), synaptophysin (Fig. 5), and cytokeratin (Fig. 6). Neoplastic cells did not react with antisera to glial fibrillary acidic protein, α-smooth muscle actin, vimentin, CD3, CD79a, Melan A, thyroglobulin, or calcitonin.
Ultrastructurally, dense sheets of closely appositioned cells had irregular plasma membrane interdigitations and few scattered desmosomes (Fig. 7). The cytoplasm contained scattered clusters of 110- to 165-nm membrane-bound dense-core neurosecretory granules, pleomorphic mitochondria, abundant rough endoplasmic reticulum, and a dilated cytocavitary network.
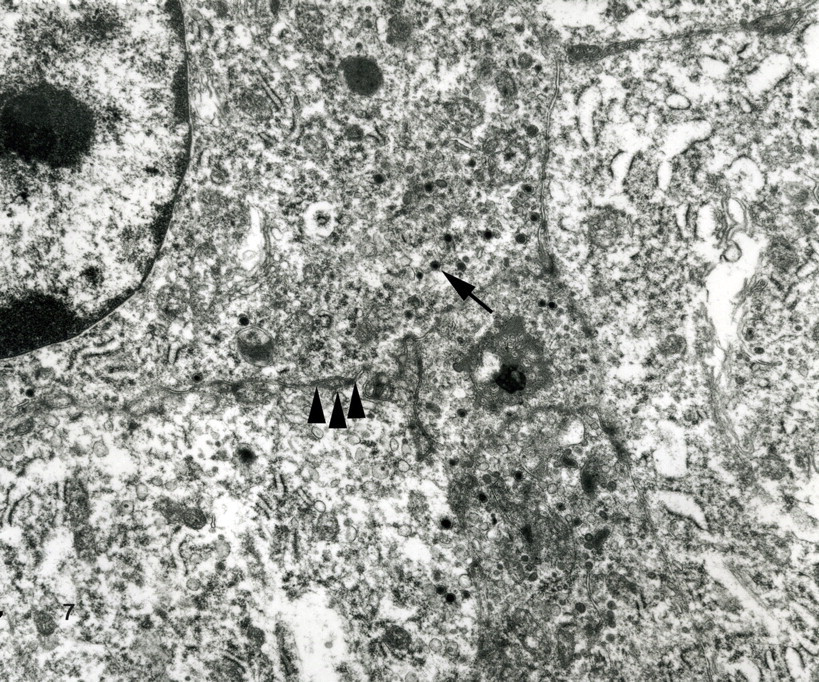
The typical neuroendocrine pattern, presence of intracytoplasmic dense-core neurosecretory granules, and immunohistochemical expression of chromogranin A, synaptophysin, and cytokeratin support a neuroendocrine cell of origin. The differential diagnosis included lymphoma, plasmacytoma, histiocytoma, melanoma, and thyroid follicular or C-cell carcinoma. Melanosomes were not seen with electron microscopy, and the cells were not reactive with antisera to Melan A, thus eliminating the possibility of a multicentric or metastatic melanoma. Other entities in the differential diagnosis were readily eliminated on the basis of histomorphologic features and lack of immunoreactivity with the appropriate antibodies, as summarized in Table 1. The diagnosis was multiple cutaneous neuroendocrine carcinoma.
Case Outcome
Because treatment of this neoplasm beyond surgical excision had not been reported in veterinary medicine, recommendations were based on the human medical literature. 15 The recommended treatment was an alternating course of carboplatin (300 mg/m2 intravenously) and doxorubicin/cyclophosphamide (30 mg/m2 intravenously and 100 mg/m2 per os, respectively) every 3 weeks. The dog received three doses of each drug; however, when it continued to develop disseminated and progressive cutaneous tumors, an antiangiogenic (metronomic) chemotherapy regimen was initiated: 5 mg/kg doxycycline per os twice daily, 25 mg/m2 cyclophosphamide per os every 2 days, and 0.3 mg/kg piroxicam per os once daily). The dog died because of progressive disease 8 months postdiagnosis. Necropsy was not permitted.
Discussion
Human cutaneous neuroendocrine carcinomas are subdivided into types based on histomorphologic features—namely, trabecular cell, intermediate cell, and small cell. 4 Treatment of human Merkel cell tumors may include surgical excision, radiation therapy, and/or various chemotherapeutics. The tumors are generally responsive to treatment, but recurrence is common. 15
Histologic subclassification of neuroendocrine tumors has not been established in companion animals, and the human criteria do not adapt well to the morphologic features of canine neuroendocrine tumors. 17 Based on the multicentric presentation and localized dermal effacement, the tumors in the present case were considered to be of low-grade malignancy, simultaneously originating from cutaneous neuroendocrine cells in multiple sites. Close association with remaining hair follicles and complete effacement of many hair follicles suggest that the neoplasm originated from the follicle-associated neuroendocrine cells.
The multicentric presentation of neuroendocrine carcinoma in this dog has not been reported for cutaneous neuroendocrine tumors without metastasis to regional lymph nodes and internal organs. In this case, there was no overt evidence of regional lymph node metastasis. Internal neoplasms rarely metastasize to the skin; however, postmortem evaluation was not possible, and a metastatic neuroendocrine carcinoma arising from internal organs cannot be completely ruled out.
Footnotes
Acknowledgement
We thank Dr Andy Wilks for initial diagnostic procedures and referral of this case, as well as case management during chemotherapy.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
