Abstract
AA-amyloidosis is frequent in shelter cats, and chronic kidney disease is the foremost cause of death. The aims were to describe kidney laboratory and microscopic findings in shelter cats with AA-amyloidosis. Cats were included if kidney specimens were collected post-mortem and laboratory data were available within 6 months before death. Renal lesions were evaluated with optical and electron microscopy. Mass spectrometry was used to characterize amyloid. Nine domestic short-hair cats were included; 4 females and 5 males with a median age of 8 years (range = 2–13). All cats had blood analyses and urinalyses available. Serum creatinine concentrations were increased in 6 cats and symmetric dimethylarginine was increased in all of the cats. All of the cats had proteinuria. Eight of 9 cats had amyloid in the medulla, and 9 had amyloid in the cortex (glomeruli). All cats had amyloid in the interstitium. Six cats had concurrent interstitial nephritis and 1 had membranoproliferative glomerulonephritis. All cats had extrarenal amyloid deposits. Amyloid was AA in each case. In conclusion, renal deposition of amyloid occurs in both cortex and medulla in shelter cats and is associated with azotemia and proteinuria. Renal involvement of systemic AA-amyloidosis should be considered in shelter cats with chronic kidney disease. The cat represents a natural model of renal AA-amyloidosis.
Introduction
AA-amyloidosis is the most common form of amyloidosis in companion animals. AA-amyloid fibrils derive from serum AA (SAA), a positive acute-phase protein primarily synthesized by hepatocytes. 43 Inflammatory and neoplastic conditions can trigger the synthesis of SAA, or it can be idiopathic. In case of recurrent or prolonged overproduction, or if proteolytic enzymes are defective, SAA tends to deposit as fibrils in organs, mainly the kidneys, liver, and spleen, inducing systemic AA-amyloidosis.24,28
Both light microscopy (LM) and transmission electron microscopy (TEM) allow for visualization of amyloid deposits. Using LM, AA-amyloid is identified by its red-orange appearance after Congo red staining, and yellow-green birefringence under polarized light. TEM shows nonbranching fibrils, consisting of twisted filaments, 7 to 10 nm in diameter and variable in length. Mass spectrometry or immunogold electron microscopy is used to identify the amyloid fibril protein. 11
Historically, AA-amyloidosis in cats has been mainly described in predisposed breeds such as the Abyssinian. In this breed, AA-amyloidosis occurs as a familial trait, primarily affecting the kidney with predominant medullary involvement and induces chronic kidney disease (CKD).6,10,29,41 A localized form of amyloidosis has been reported in the pancreas of cats as a consequence of islet amylin polypeptide deposition. 46 In domestic short-hair cats, AA-amyloidosis has rarely been reported, often in cats with concurrent spontaneous feline immunodeficiency virus infection, and has been associated with liver rupture, pulmonary hemorrhage, and proteinuria.1,3,5,20,25,32,34,45 Recently, a high prevalence of AA-amyloidosis, ranging from 52% to 73%, has been documented in cats housed in shelters; of note, 50.6% had renal involvement.13,35 These findings suggest that AA-amyloidosis is frequent in shelters cats and the kidney is a target organ. To date, no studies have been conducted to assess whether shelter cats with renal involvement of systemic AA-amyloidosis have CKD and whether amyloid deposition primarily affects the cortex or medulla.
Hence, aims of this study were to assess if renal involvement of systemic AA-amyloidosis is associated with clinicopathologic abnormalities consistent with CKD, and how amyloid deposits are distributed in the kidneys of cats housed in shelters.
Materials and Methods
Selection of Cases
The medical records of cats from a shelter located in northern Italy (close to Turin) were retrospectively reviewed. Cats were included if: (1) death had occurred between 2017 and 2019; (2) the latest available laboratory data were obtained within 6 months prior to death; (3) tissue sampling was performed within 6 hours after death; (4) kidney, hepatic, and splenic specimens were collected and stored for both histologic and ultrastructural examinations; and (5) amyloid deposits were identified. Ethical approval was not required, as the cats were studied after spontaneous death or after euthanasia performed for reasons unrelated to the investigation.
Clinical and Laboratory Data
Data retrieved from medical records included signalment (ie, breed, sex, and age), history, and results of available blood analyses, urinalyses, and abdominal ultrasound. Laboratory data regarding the kidney were creatinine, urea nitrogen, symmetric dimethylarginine (SDMA), and phosphate, as well as urine-specific gravity and urine protein-to-creatinine ratio; in addition, laboratory data regarding the liver were albumin, total protein, and bilirubin. All blood and urine analyses were conducted in the same laboratory (IDEXX Laboratories, Novara, Italy). Staging of CKD was performed according to the guidelines of the International Renal Interest Society (IRIS) in cats. 14 In brief, CKD stage I was determined by creatinine <1.6 mg/dl and SDMA <18 µg/dl, CKD stage II was determined by creatinine 1.6 to 2.8 mg/dl and SDMA 18 to 25, CKD stage III was determined by creatinine 2.9 to 5.0 mg/dl and SDMA 26 to 38 µg/dl, and CKD stage IV was determined by creatinine >5.0 mg/dl and SDMA >38 µg/dl.
Histopathologic and Ultrastructural Data
Kidney, liver, and spleen specimens were processed for LM examinations; in addition, kidney samples were processed for TEM examination. Specimens were fixed in 10% buffered-formalin, paraffin-embedded, and cut at 3 to 5 µm with an automatic microtome (Microm HM 325, Thermo Scientific, Milano, Italy). To characterize parenchymal damage, sections were routinely stained with hematoxylin and eosin using an automatic stainer (Autostainer XL, Leica, Milano, Italy) and manually stained with periodic acid-Schiff, Masson’s trichrome and periodic Schiff-methenamine silver. Congo red staining was performed to detect congophilia under polarized light. Slides were scanned using the D-sight image scope (Visia Imaging, San Giovanni Valdarno, Arezzo, Italy). For TEM examination, paraffin-embedded samples were hydrated after deparaffinization and washed in cacodylate buffer, post-fixed in 1% osmium tetroxide in 0.1 M sodium cacodylate buffer for 1 hour at 4°C, dehydrated in a graded ethanol series, and embedded in an epoxy resin (Sigma-Aldrich, Milan, Italy). Ultrathin sections (60–70 nm) were obtained with an ultramicrotome (Ultrotome V, LKB, Vienna, Austria), counterstained with uranyl acetate and lead citrate, and viewed with the TEM (Tecnai G 2 , FEI, Milan, Italy) operating at 100 kV. Images were captured with a digital camera (Veleta, Olympus Soft Imaging System, Segrate, Milan, Italy).
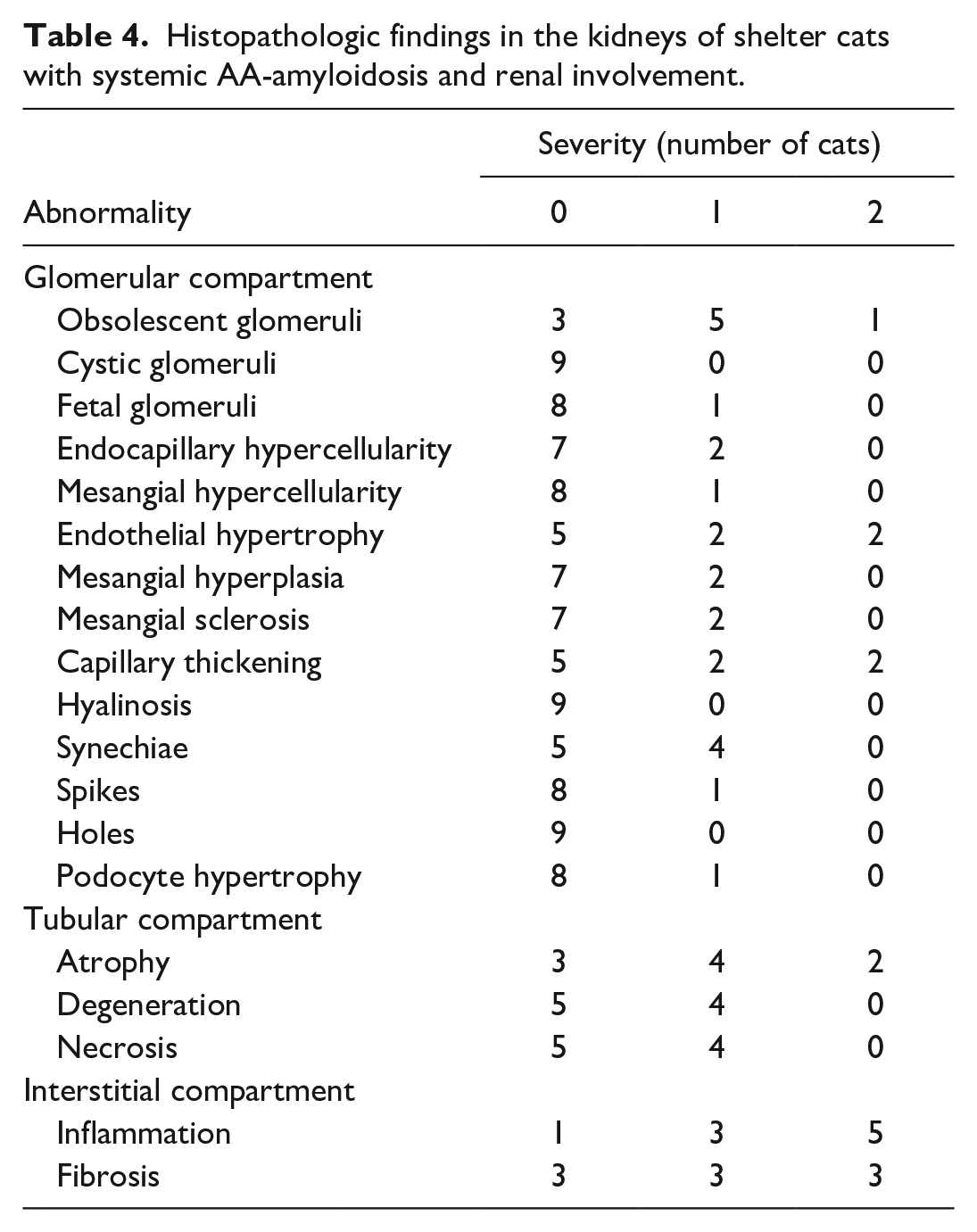
Using LM, the renal amyloid deposition was described as cortical and/or medullary; in the glomeruli, interstitium, tubules, and/or vessel walls. The amyloid density and the severity of any other histologic and ultrastructural abnormalities were assessed with a score from 0 to 2, as follows: 0 if absent, 1 if mild, and 2 if moderate to severe. 8 Other than amyloid deposition, the following histopathological lesions were recorded. In the glomerular compartment, the presence of obsolescent, cystic, and fetal glomeruli; endocapillary and mesangial hypercellularity; capillary thickening; sclero-hyalinosis; synechiae; spikes and holes in the glomerular basement membrane; and podocyte hypertrophy was recorded. In the tubulointerstitial compartment, the presence of tubular atrophy, degeneration and necrosis, inflammation, and fibrosis was recorded. Using TEM, endothelial hypertrophy, mesangial hyperplasia, and mesangial sclerosis were scored, and fusion of the foot processes of the podocytes and immune complex deposition were evaluated as present or absent.
By LM, the splenic amyloid deposition was described as vascular, interstitial, trabecular, and/or capsular, and the hepatic amyloid deposition as capsular, centrilobular, vascular, and/or as affecting the portal triad and/or the Disse’s space. The amyloid density was scored from 0 to 2, as follows: 0 if absent, 1 if mild, and 2 if moderate to severe. In both splenic and hepatic samples, any concurrent histopathologic alteration was recorded.
LM and TEM examination was performed by 3 pathologists (SF, SLB, and LA), first independently and then comparing and discussing the results together until consensus was reached.
Liquid Chromatography–Tandem Mass Spectrometry and Amyloid Typing
Amyloid fibrils were extracted from frozen kidney tissue using a water-based procedure, as previously described. 38 Samples were analyzed by sodium dodecyl–sulfate polyacrylamide gel electrophoresis (SDS-PAGE) under denaturing and reducing conditions; gels were stained with colloidal Coomassie dye G-250 (Thermo Fisher Scientific, Waltham, Massachusetts). 38 For protein quantification, the extract was incubated for 30 minutes with 8 M urea to dissolve protein aggregates and for 1 hour with 100 mM dithiothreitol to reduce disulfide bonds. Protein samples were quantified using the Bradford protein assay (BioRad, Segrate, Milano, Italy). For liquid chromatography–tandem mass spectrometry (LC-MS/MS) analyses, water extraction fraction #2 or #3 was used. Briefly, 40 μg of urea-dissolved proteins were subjected to disulfide bonds reduction (100 mM dithiothreitol for 1 hour at room temperature) and alkylation of thiol groups (150 mM iodoacetamide for 1 hour at room temperature in the dark), followed by dilution with 100 mM ammonium bicarbonate to bring the urea concentration to 1.3 M.
Proteins were trypsin-digested (trypsin:protein ratio 1:20 w/w) (Sequence grade modified trypsin, Promega, Madison, Wisconsin, USA) for 16 hours at 37°C, and the reaction was stopped by adding 1% trifluoroacetic acid. Peptides were purified using Pierce TipC18 (Thermo Fisher Scientific) and analyzed on a Dionex Ultimate 3000 UHPLC system coupled to a Q Exactive mass spectrometer (Thermo Fisher Scientific).18,21 In some cases, proteins present in Coomassie-stained SDS-PAGE gels were also identified by LC-MS/MS; procedures for in-gel digestion and processing were previously described. 38 Data were processed using Proteome Discoverer software, version 2.0 (Thermo Fisher Scientific), against the proteome of Felis catus (downloaded from Uniprot on September 2019, 34292 entries). For each identified protein, the peptide-spectrum match count was normalized for protein length in order to more accurately estimate the relative abundance of the various proteins in the sample.
Results
Signalment and History
Nine cats fulfilled the inclusion criteria; all were domestic short-hair, 5 were males (4 castrated and 1 intact) and 4 were females (3 spayed and 1 intact), and the median age at death was 8 years (range = 2–13). The median duration of stay in the shelter was 5.4 years (range = 7 months to 10 years); 8 cats entered as stray cats and 1 was an owner surrender. All had spent at least the last third of their life in the shelter.
All cats were unvaccinated. The medical history of 8 cats included one or more of the following diseases: gingivostomatitis, inflammatory bowel disease, pneumonia, eyelid squamous cell carcinoma, intestinal lymphoma, cutaneous abscess, splenic hematoma, feline panleukopenia, and feline leukemia virus (FeLV). Overall, 4 were positive for FeLV. In the 6 months prior to death, surgeries and medications were reported in the medical records of 1 and 3 cats, respectively; the former was a pulmonary lobectomy due to chronic pneumonia, the latter included amlodipine, colchicine, erythropoietin, phosphate binders, and telmisartan (all 3 cats treated for CKD). One cat had no disease, surgery, or medication listed in the medical record. Regarding the cause of death, CKD was reported in 7 cats, and eyelid squamous cell carcinoma and intestinal lymphoma were each reported in 1 cat.
Laboratory and Ultrasonographic Findings
All cats had blood analyses and urinalyses results available. Seven had the latest available laboratory data obtained within 2 weeks prior to death, laboratory from 1 cat was from 6 weeks before death, and the remaining cat’s laboratory data were from more than 2 months before death. Laboratory results are shown in Table 1.
Results of blood and urine analyses in shelter cats with systemic AA-amyloidosis and renal involvement.
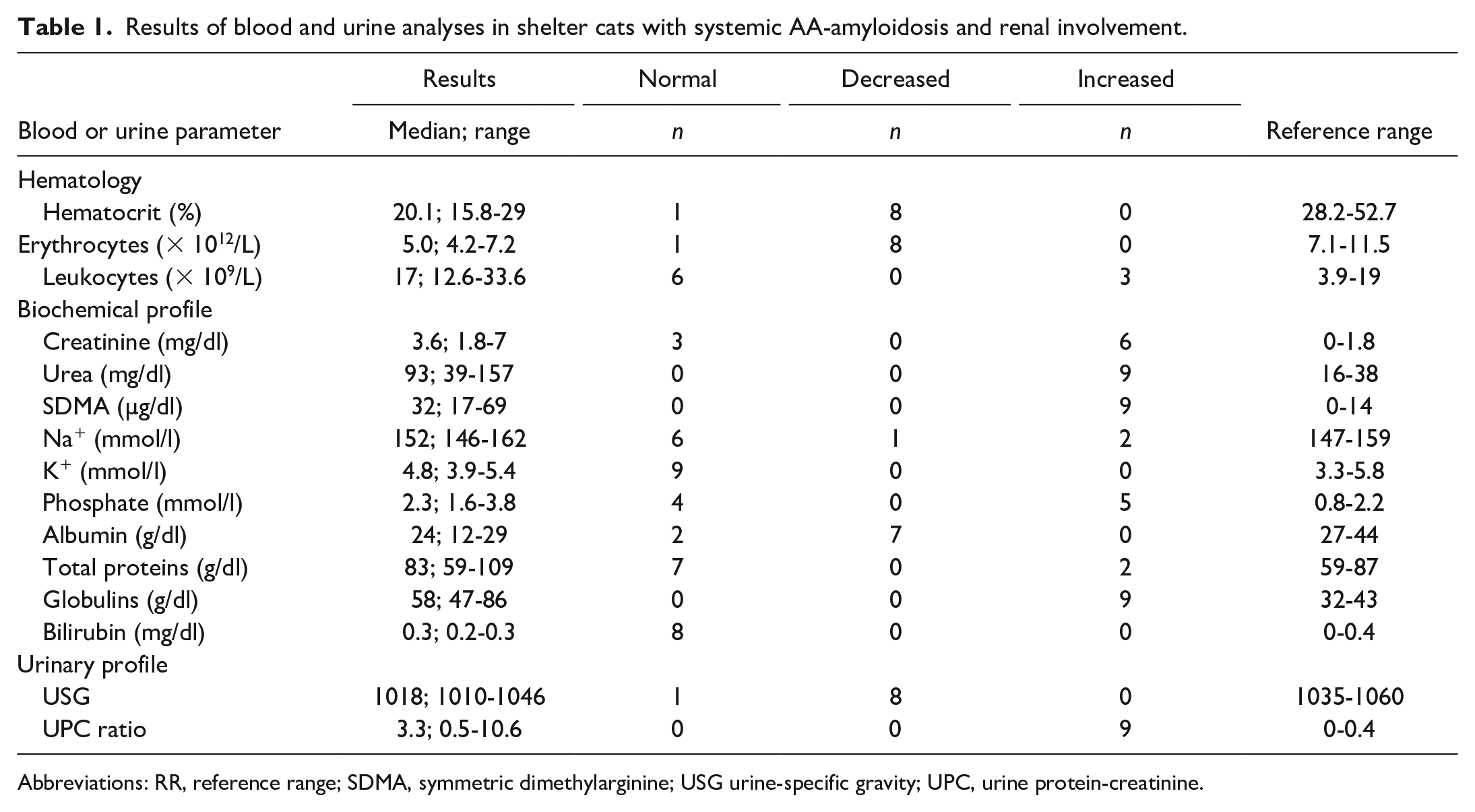
Abbreviations: RR, reference range; SDMA, symmetric dimethylarginine; USG urine-specific gravity; UPC, urine protein-creatinine.
All cats were affected by CKD; 4 in stage II, 3 in stage III, and 2 in stage IV. Overall, 6 cats had high creatinine, and all cats had high urea and SDMA. Serum potassium levels were normal in all cats. One cat had mild hyponatremia and 2 cats had mild hypernatremia; sodium levels were normal in the remaining cats. The 5 IRIS stages III and IV cats had hyperphosphatemia. Overall, 7 cats had hypoalbuminemia, and 9 had hyperglobulinemia. Serum total bilirubin concentrations were available in 8 cats, which was within the reference range in all cats. All cats had mild to moderate anemia. Regarding white blood cell counts, 3 cats had leukocytosis.
All 9 cats had proteinuria; 6 had a urine protein-creatinine ratio >2 (normal = <0.4). Eight cats had urine-specific gravities <1035 and 2 had urine specific gravities <1015 (normal: >1034). Regarding urine sediment, 1 cat had amorphous crystals and 1 had normoglycemic glycosuria on repeated tests.
Overall, 7 cats had abdominal ultrasound reports available. Ultrasonographic findings included multiple renal infarcts in 3 cats, diffusely increased renal cortical echogenicity in 4 cats, and a medullary rim sign (defined as a distinct, hyperechoic zone in the outer renal medulla) in 1 cat; 2 cats had no renal abnormalities on ultrasound. 12
Histopathologic and Ultrastructural Findings
Overall, renal amyloid deposition was mild in 1 cat and moderate to severe in 8 cats. Nine cats had amyloid deposition in the cortex (ie, in the glomeruli and/or interstitium) and 8 had amyloid in the medulla (ie, in or around the tubules and interstitium). Specifically, the amyloid deposits were both in the cortex and in the medulla in 8 cats and only in the cortex in 1 cat (Fig. 1a–l). Cortical amyloidosis was mild in 4 and moderate to severe in 5 cats, whereas medullary involvement was mild in 2 and moderate to severe in 6 cats.
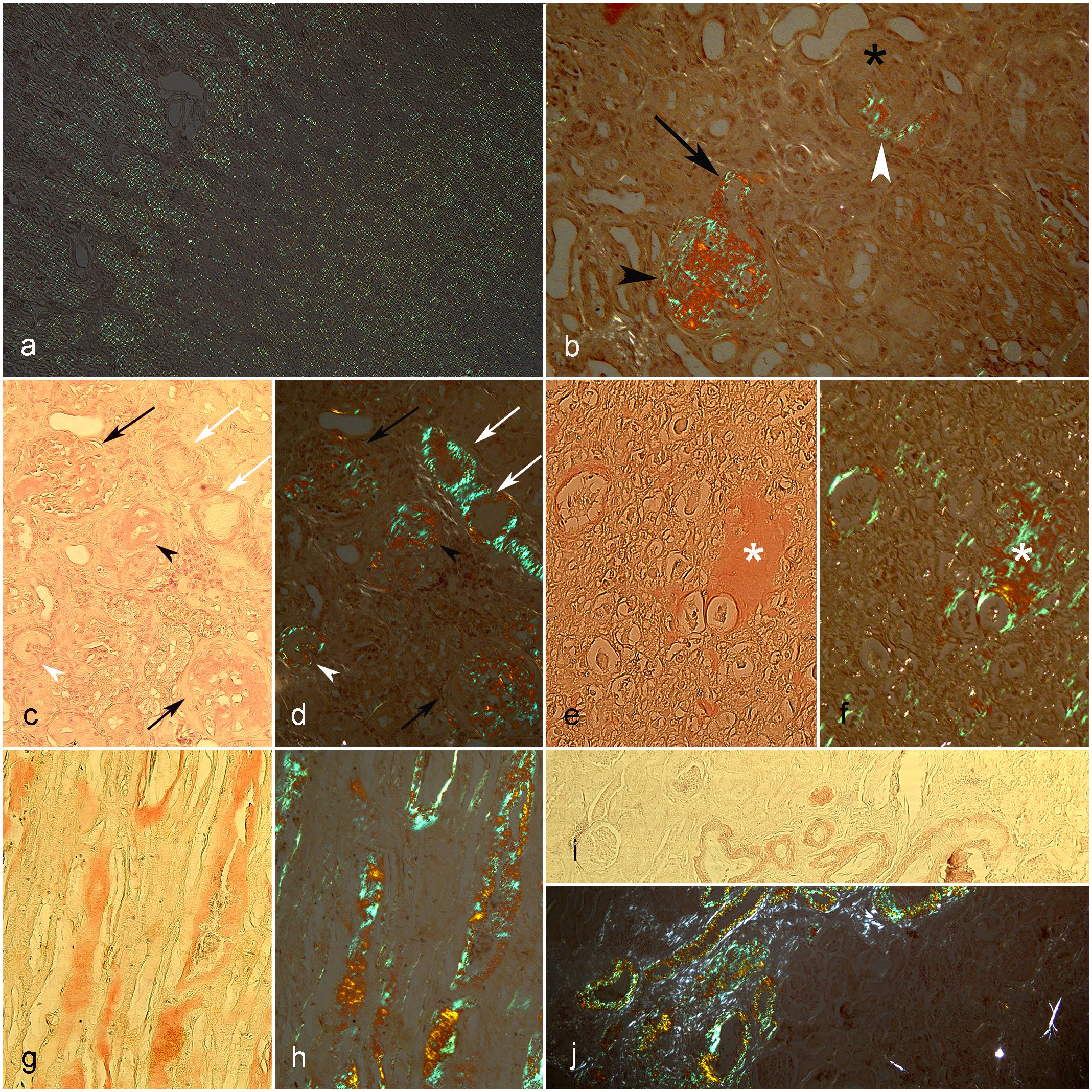
(a–j) Kidney, cat. Distribution of amyloid deposits in the renal parenchyma. (a, b, d, f, h, j) Congo red stain under polarized light. (c, e, g, i) Congo red stain. (a) Diffuse distribution of amyloid in the medullary (top left) and cortical (bottom right) compartments. Score 2. (b) Global (black arrowhead) and segmental (white arrow) glomerular amyloid deposits. One of the glomeruli is sclerotic (asterisk). Amyloid is also visible at the vascular pole (black arrow). (c, d) The amyloid is evident in the glomeruli (black arrows), vessel’s walls (white arrows), tubular wall (white arrowhead), and focally in the interstitial, probably around an atrophied tubule (black arrowhead). (e, f) Moderate interstitial amyloid deposit (asterisks). (g, h) Peritubular amyloid deposits associated with tubular atrophy. (i, j) Amyloid is evident only in the wall of the vessels.
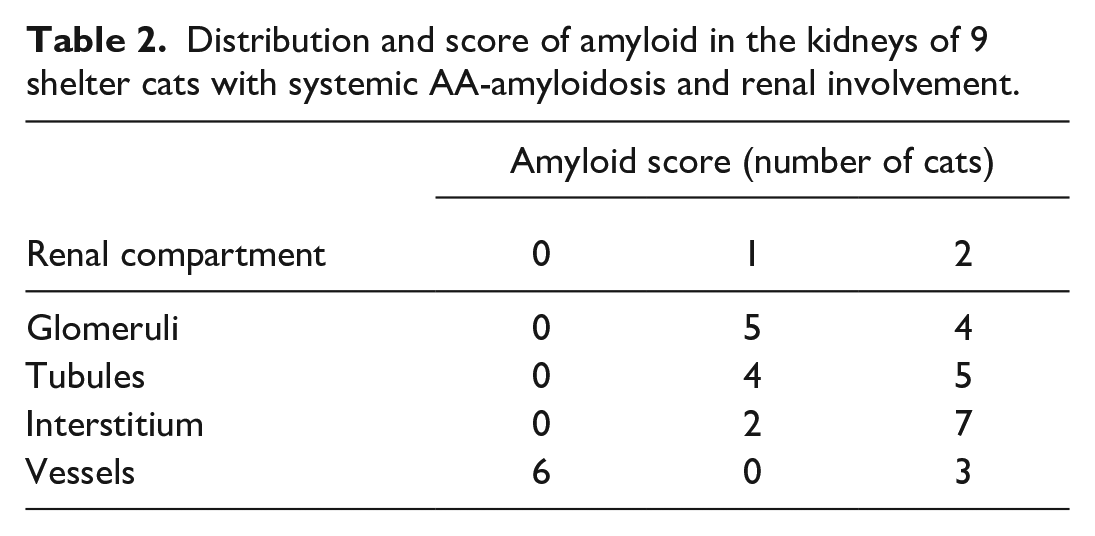
Glomerular amyloid deposits were present in all 9 cats. They all had intraglomerular deposition (ie, within the glomerular tufts), with distributions ranging from focal (ie, less than 50% of the glomeruli involved) to diffuse (ie, more than 50% of the glomeruli involved) and from segmental (ie, affecting portions of the glomerulus) to global (ie, affecting the entire glomerulus) (Fig. 1b–d). All cats had amyloid deposition in both the tubular and interstitial compartments. Of the cats with cortical amyloidosis, 4 had deposits in the cortical interstitium. The medullary interstitium showed deposits in all cats with medullary amyloidosis; in the medullary tubules, the deposits were peritubular in all cats and intratubular in 3 (Figs. 1d–h, 2a–d, Supplemental Fig. S1). Three cats had amyloid deposition involving the vascular compartment (Fig. 1c, d, i, j). The scores attributed to amyloid deposits for each renal compartment are reported in Table 2. The distribution pattern of amyloid deposition and morphological diagnosis in each cat are reported in Table 3.
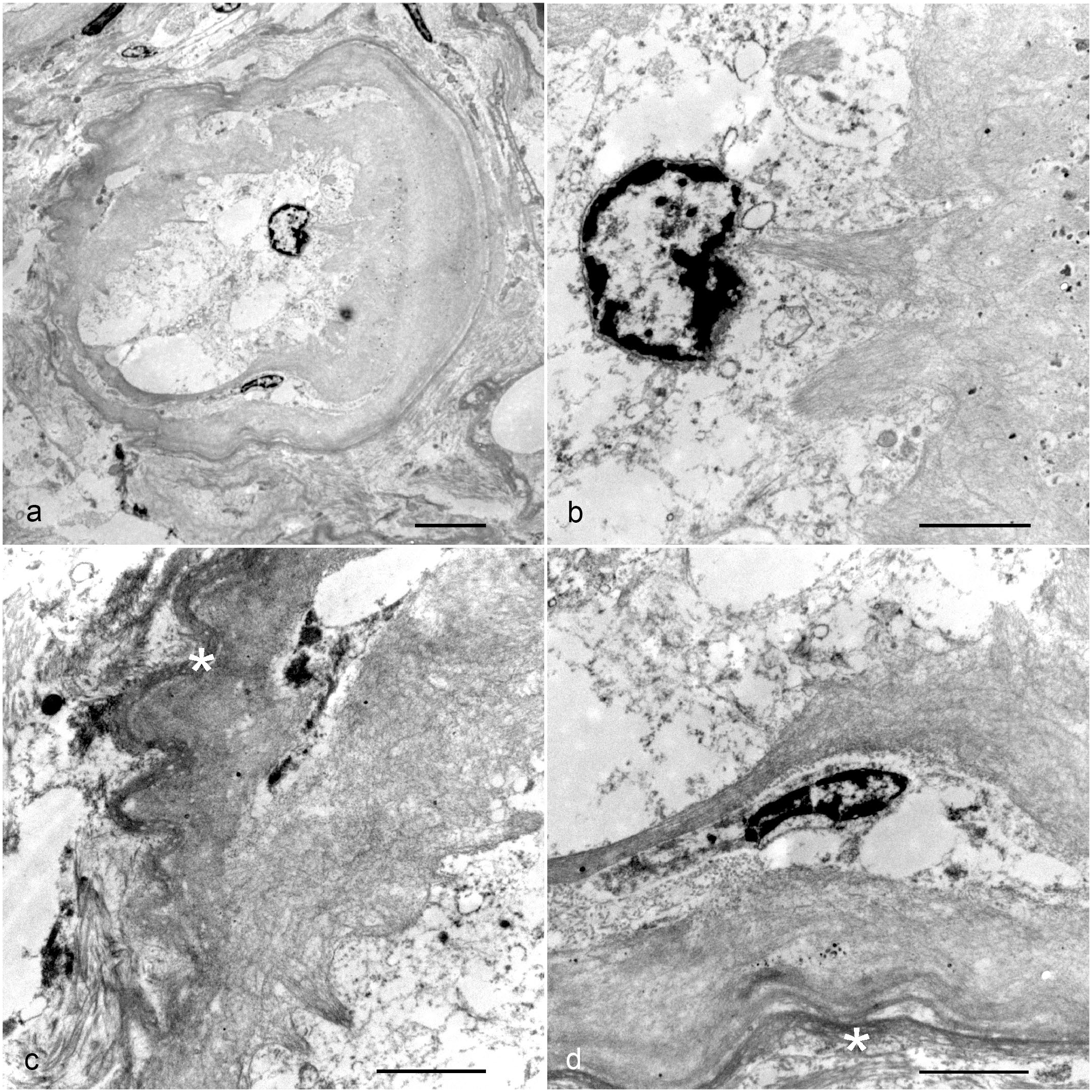
Kidney, renal tubule, cat, transmission electron microscopy. (a) Severe deposition of haphazardly arranged fibrils of 9 to 11 nm in diameter (amyloid) that atrophy and partially replace the epithelial cells. Bar: 5 µm. (b) Higher magnification of (a) showing amyloid and a residual degenerated epithelial cell. Bar: 2 µm. (c, d) Higher magnification of (a) showing amyloid that expands and effaces tubular basement membrane (asterisk). Bar: 2 µm.
Distribution and score of amyloid in the kidneys of 9 shelter cats with systemic AA-amyloidosis and renal involvement.
Amyloid distribution pattern and morphological diagnosis in the kidneys of 9 shelter cats with systemic AA-amyloidosis and renal involvement.
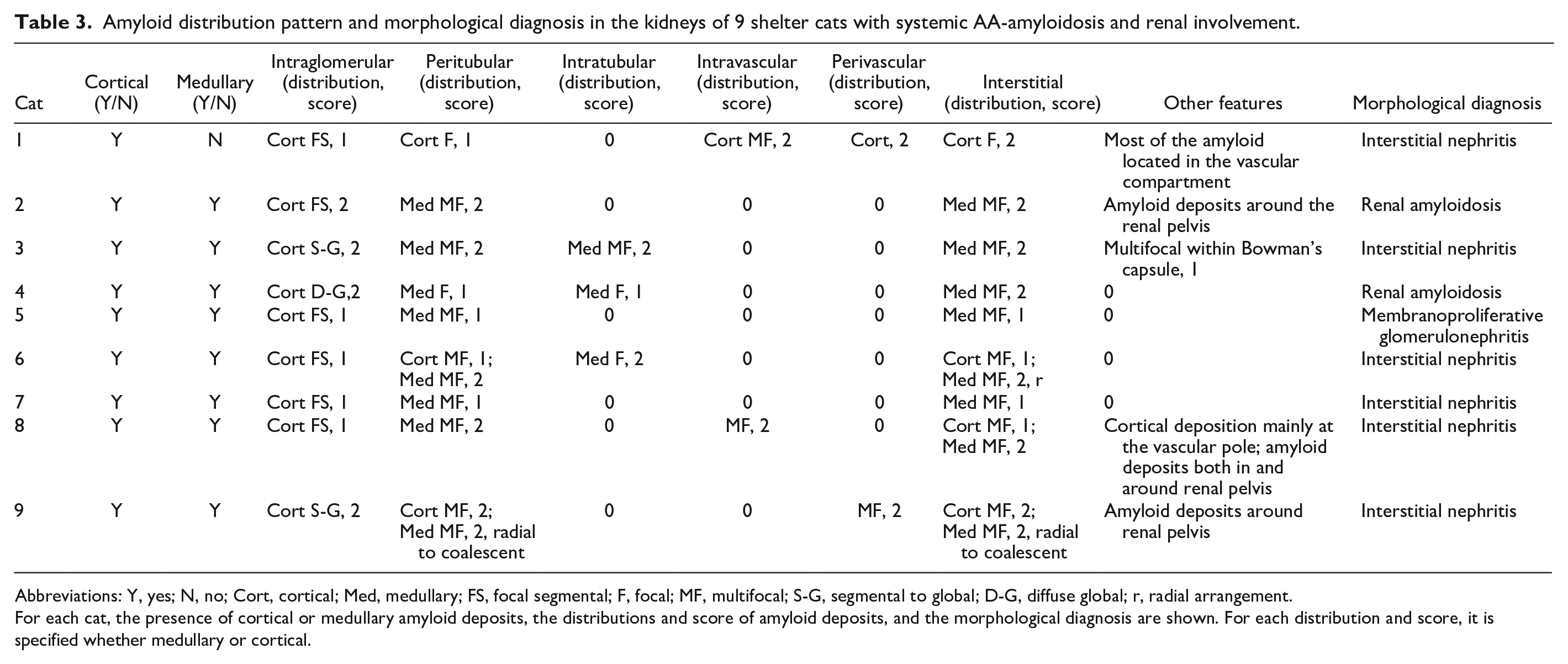
Abbreviations: Y, yes; N, no; Cort, cortical; Med, medullary; FS, focal segmental; F, focal; MF, multifocal; S-G, segmental to global; D-G, diffuse global; r, radial arrangement.
For each cat, the presence of cortical or medullary amyloid deposits, the distributions and score of amyloid deposits, and the morphological diagnosis are shown. For each distribution and score, it is specified whether medullary or cortical.
From the combination of LM and TEM, 6 cats had concurrent interstitial nephritis and 1 had membranoproliferative glomerulonephritis. In the latter cat, electron-dense deposits were observed in the intramembranous, subepithelial, and mesangial regions and were in IRIS stage II CKD. In addition, 2 cats in IRIS stage III with interstitial nephritis, moderate to severe interstitial amyloidosis, and mild glomerular amyloidosis had a urine protein-creatinine ratio >2.
On LM examination, there were obsolescent glomeruli regardless of the IRIS stage. The most common histologic findings in the glomerular compartment were obsolescence, capillary thickening, and synechiae, which were present in 6, 4, and 4 cats, respectively. Regarding the renal tubules, 3 cats had no lesions, 2 had only mild or moderate tubular atrophy, and 4 had mild tubular atrophy, degeneration, and necrosis. Cats with tubular abnormalities were in IRIS stages III and IV, except for the cat with only mild tubular atrophy, which was in IRIS stage II. Pathologic changes in the renal interstitium were found in all cats; specifically, 8 had mild to severe inflammation and 6 had fibrosis. The interstitial inflammatory cell infiltrate consisted predominantly of lymphocytes and plasma cells with occasional neutrophils and macrophages. Other common pathologic findings were multiple intratubular hyaline casts and crystals, multifocal to disseminated tubulointerstitial dystrophic mineralization, and focal hyperplasia of the tunica media of the arterioles. On TEM examination, 4 cats had fusion of the foot processes of the podocytes, and 4 had mild mesangial matrix expansion (Fig. 3a–d). Renal histologic findings scored 0 to 2 and ultrastructural abnormalities are shown in Tables 4 and 5, respectively.
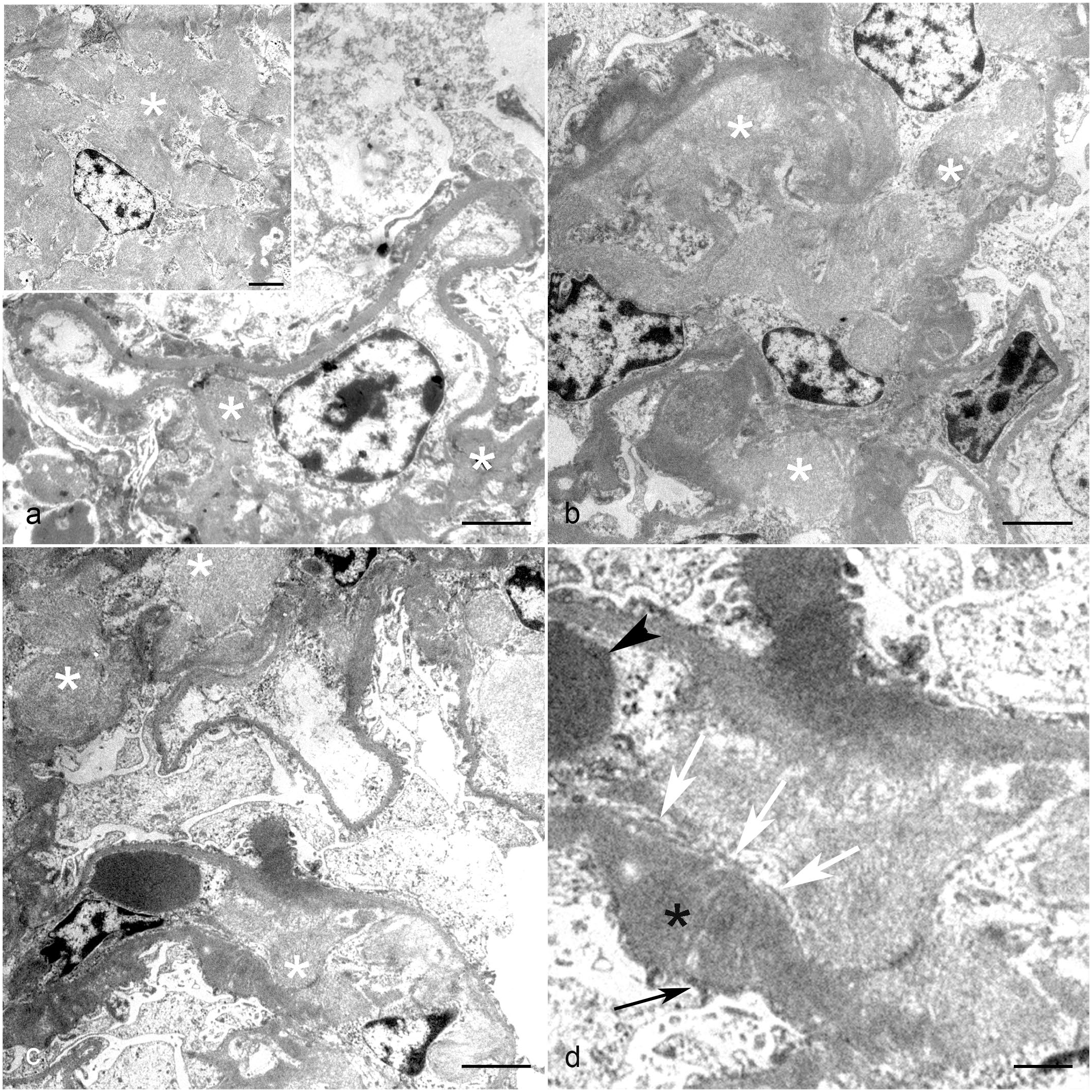
Kidney, glomerulus, cat, transmission electron microscopy. (a) Multifocal mild deposits of haphazardly arranged fibrils of 9 to 11 nm in diameter (amyloid, asterisks). Amyloid deposits are subendothelial and slightly expand the capillary loops. Inset: severe, diffuse, deposits of amyloid (asterisk) efface the tuft architecture. Bars: 2 µm. (b) Multifocal moderate deposition of amyloid (asterisks), which expands the subepithelial space and often effaces the capillary loop architecture. Bar: 2 µm. (c) Multifocal moderate deposition of amyloid (asterisks). Bar: 2 µm. (d) Higher magnification of (c) highlights the presence of amyloid filling the capillary lumen. White arrows indicate the fenestrated endothelium. Note the expansion of the glomerular basal membrane with loss of stratification (black asterisk) and associated fusion and effacement of foot processes of podocytes (black arrow). Arrowhead: red blood cell. Bar: 0.5 µm.
Histopathologic findings in the kidneys of shelter cats with systemic AA-amyloidosis and renal involvement.
Transmission electron microscopy findings in the kidneys of 9 shelter cats with systemic AA-amyloidosis and renal involvement.

All cats had extrarenal amyloid deposition; all cats had splenic amyloidosis and 8 had hepatic. In the spleen, amyloid deposition was mild in 1 and moderate to severe in 8 cats. Eight cats had amyloid depositions described as vascular, 6 as interstitial, 3 as trabecular, and 3 as capsular (Fig. 4a–c). In addition to amyloid deposits, lymphoid hyperplasia, plasma cell hyperplasia, and disseminated severe follicular hyperplasia were reported in the spleen of 1 cat each.
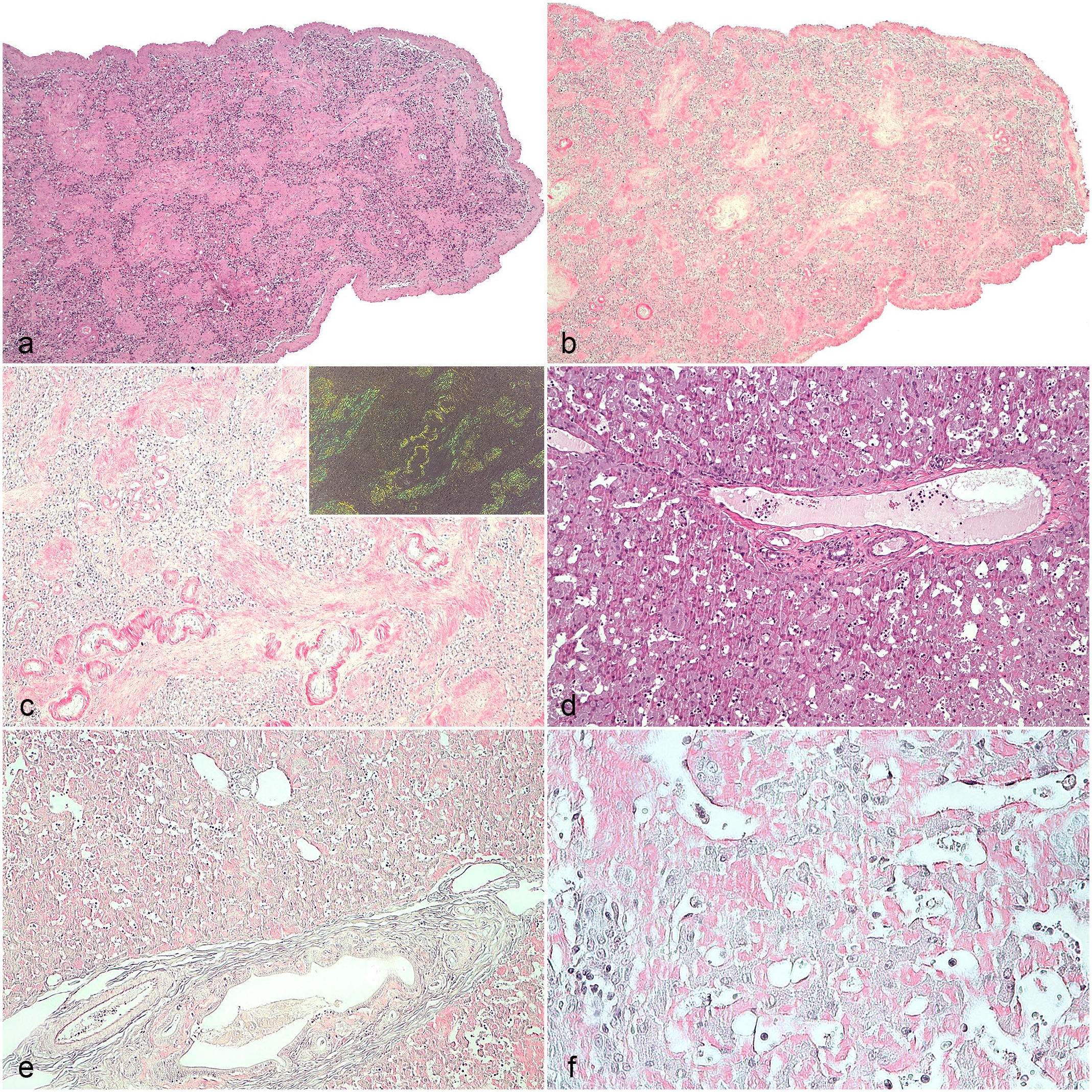
Histologic aspect of feline severe amyloidosis (score 2) in the (a–c) spleen and (d–f) liver. (a) Spleen. Abundant eosinophilic material expands trabeculae. Hematoxylin and eosin (HE). (b) Spleen. The eosinophilic material stains red with Congo red (CR). (c) Spleen. The amyloid stains red with CR and is localized in the vessel walls, the trabeculae, and multifocally in the parenchyma. Inset: amyloid is birefringent under polarized light. CR. (d) Liver. Diffuse deposition of amorphous eosinophilic material in the space of Disse. HE. (e) Liver. The eosinophilic material stains red with CR. (f) Liver. Abundant red stained amyloid in the space of Disse separates the hepatocytes, which appear atrophic. CR.
In the liver, amyloid deposition was mild in 5 cats and moderate to severe in 3. Amyloid depositions were described as capsular in 7 cats, centrilobular in 3, vascular in 3, affecting the portal triad in 3, and/or the space of Disse in 6 cats (Fig. 4d–f). In addition to amyloid deposits, multifocal nonspecific chronic inflammation was reported in the liver of 2 cats, hepatic lipidosis and mild diffuse cholangitis in the liver of 1 cat each.
Liquid Chromatography–Tandem Mass Spectrometry-Based Identification of Kidney Amyloid Fibrils
Fibrils were extracted from amyloid-positive kidneys of all 9 cats for which frozen organs were available. In all of these cats, SAA was identified as the most abundant amyloidogenic protein in the samples by LC-MS/MS (Supplemental Table S1). In all, a single prevalent band with a molecular weight of approximately 12 kDa was visible on SDS-PAGE and identified as SAA by mass spectrometry.
Discussion
Azotemia and proteinuria are the hallmarks of renal AA-amyloidosis in humans and dogs; similarly, CKD is observed in Abyssinian cats with familial amyloidosis.17,29,37 Recently, a high prevalence of systemic AA-amyloidosis has been found in shelter cats; the authors hypothesized that a horizontal transmission occurs, similarly to findings in captive cheetahs. 44 Multiple organs were reported to be concurrently affected. The disease was considered systemic and was suspected to cause CKD.13,35
This study shows that domestic short-hair cats with nonfamilial AA-amyloidosis housed in shelters have laboratory and histopathologic abnormalities consistent with CKD. In addition to renal deposits, all cats had concurrent amyloid fibrils in the liver and/or spleen; in these cats, CKD was therefore considered the renal manifestation of a systemic disease.
All affected cats had evidence of CKD. Serum SDMA concentrations were above the reference range in each cat, suggesting it may be a more sensitive marker of renal function than creatinine, which was increased in 6 of the 9 cats. This finding may be due to SDMA being less affected by loss of lean body mass. 14 In addition, all cats had increased serum urea and proteinuria. These clinical pathology findings are comparable to those reported in the literature for affected humans and dogs with renal amyloidosis. Indeed, proteinuria occurs in approximately 95% of people with renal AA-amyloidosis, of which nearly 50% reach end-stage renal disease.2,4,16,30 Similarly, increased urea and proteinuria were found in 91% and 97% of dogs with nonfamilial renal AA-amyloidosis, respectively. 37 Given that proteinuria was observed in all available urine samples, being marked in two thirds, and that inadequately concentrated urines were observed in almost all of the cats, azotemia was likely due to renal disease involving both the glomeruli and the tubules. 2 Interestingly, 1 cat had normoglycemic glycosuria on repeated tests. This finding might suggest tubular dysfunction, which has sometimes been described in humans with renal AA-amyloidosis. 23
Other complications commonly associated with CKD in addition to azotemia and proteinuria were hypoalbuminemia, hyperphosphatemia, and anemia. Notably, 7 of the 9 cats had hypoalbuminemia and 5 had hyperphosphatemia. Although a causal relationship cannot be demonstrated, both hypoalbuminemia and hyperphosphatemia were possibly due to kidney damage, the former as a consequence of persistent loss of albumin in the urine and the latter caused by the decrease in glomerular filtration rate.2,27 Interestingly, despite hypoalbuminemia in most cats, none had hypoproteinemia, but 2 did have hyperproteinemia; hyperglobulinemia was reported in all cats. As albumin is a negative acute-phase protein and cytokines are known to increase globulin synthesis, some of the concurrent inflammatory diseases of affected cats may also have contributed to hypoalbuminemia and hyperglobulinemia.27,39 As expected, hyperphosphatemia was associated with advanced CKD. Mild to moderate anemia was found in all cats, possibly due to azotemia and impaired renal synthesis of erythropoietin. 33 In contrast, none had hypokalemia, even if this is another common complication in feline CKD; this may be partially explained by treatment with angiotensin receptor blockers in 3 of the cats.
The above laboratory results need to be interpreted bearing in mind that 8 of the 9 cats had a history of inflammatory or neoplastic disease, and 8 had hepatic amyloidosis. Hepatic amyloidosis can lead to liver dysfunction; therefore, hypoalbuminemia may have been caused by both increased urine loss due to renal amyloidosis and decreased liver production due to hepatic amyloidosis. 37 Except for albumin and total bilirubin, the latter found to be within the reference range in all of the cats tested, the liver panel was not available. However, hypoalbuminemia has only rarely been reported in cats with hepatic amyloidosis. 3
Histopathologic investigations in humans and dogs with renal AA-amyloidosis showed deposition of amyloid mainly in the cortex (ie, in the glomeruli) and less frequently in the medulla, except in Chinese Shar-Pei dogs with familial amyloidosis in which both the cortex and the medulla were severely affected.9,37,42 In contrast, in Abyssinian and Siamese cats, as well as in nondomestic felids, amyloid deposits were found predominantly in the medulla.6,10,26,31,36,40,41 To date, no studies have been conducted in domestic short-hair cats with renal involvement of systemic AA-amyloidosis to assess whether cortex and medulla are differently involved. However, this is of great interest, as kidney biopsies in living cats are collected from the cortex, sparing the medulla; therefore, the deposition of amyloid in the medulla would not be identified, resulting in diagnostic errors. Moreover, as the distribution of glomerular amyloid is often focal, the sensitivity of detecting amyloid is strictly dependent on the number of glomeruli in the sample. Surprisingly, in this study, 8 of the 9 cats had amyloid deposits in both cortex and medulla, with the medulla being unaffected in one, and all in the interstitial compartment. Overall, the interstitium appeared to be more severely affected in the medulla than in the cortex.
Renal AA-amyloidosis is included among the diseases causing CKD in cats, but up to now has been considered rare in domestic short-hair breeds. Typical pathologic changes in renal structure associated with CKD are glomerulosclerosis, tubular atrophy, interstitial inflammation, and interstitial fibrosis. 7 In this study, 4 of the 9 cats were FeLV-positive; moreover, in addition to renal amyloidosis, 6 were diagnosed with interstitial nephritis and 1 had membranoproliferative glomerulonephritis. Of the 6 cats with tubular lesions, 5 were in IRIS stages III and IV; this is not surprising as tubular abnormalities are mostly associated with advanced CKD.15,22 Interestingly, 2 cats with interstitial nephritis, prominent interstitial amyloidosis, and only mild glomerular amyloidosis had urine protein-creatinine ratios >2; therefore, proteinuria can be severe even though glomerular involvement appears to be minor.
All cats had extrarenal amyloid deposits in the spleen and in the liver. Amyloid fibrils were most commonly identified in the vascular and interstitial compartments of the spleen and in the vascular compartment and space of Disse of the liver. In Siamese, Oriental and Abyssinian cats affected by familial amyloidosis, the spleen and the liver can be diffusely affected by amyloid deposits but the most common distribution of amyloid deposits in these organs is currently unknown.10,41 Similarly, nonpredisposed breed cats affected by hepatic and splenic amyloidosis have been sporadically reported but a score-deposition pattern of amyloid fibrils has never been reported.1,3,5,25,45
The high prevalence of systemic AA-amyloidosis recently described in shelter cats likely ruled out a familial predisposition to the disease. 13 These findings, along with the different distribution of AA-amyloid within the kidney, might suggest that systemic AA-amyloidosis represents a separate pathologic entity to familial amyloidosis.
This study is limited by its retrospective nature. No data related to disease progression of cats with CKD associated with renal involvement of systemic AA-amyloidosis were available; aside from kidney function, only minimal clinicolaboratory information was available. Furthermore, given the strict inclusion criteria, the number of cats included was low. In addition, 5 cats also had another disease known to contribute to CKD (ie, FeLV infection, membranoproliferative glomerulonephritis); therefore, in these cats, renal amyloidosis may not have been the only trigger of CKD. FeLV, in particular, is associated with immunocomplex glomerulonephritis, neoplasia, and infections; however, in the present investigation, only 1 cat had evidence of immunocomplex glomerulonephritis, and no cats showed evidence of renal neoplasia or infection.19,34 In the authors’ opinion, albeit other diseases had contributed to the observed clinicolaboratory alterations, these limitations do not overshadow the importance of the above findings, which could provide an explanation as to why at least some of the many cases of feline CKD occur in shelters and whose underlying cause is typically unknown. These data are expected to have a significant impact on shelter medicine, potentially opening a new avenue for the future management of multicat households.
In conclusion, renal involvement of systemic AA-amyloidosis is associated with CKD in cats housed in shelters. Unlike familial amyloidosis in predisposed feline breeds, AA-amyloidosis in shelter cats involves both the renal cortex and medulla. Severe proteinuria may also be observed in cats with prominent fibril deposition in the interstitium but only mild in the glomeruli. Renal involvement of systemic AA-amyloidosis should always be included in the differential diagnoses of shelter cats with CKD. Furthermore, given the similarities to humans revealed in this study, the cat may represent a new and spontaneous model of systemic AA-amyloidosis.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241257903 – Supplemental material for Renal alterations in cats (Felis catus) housed in shelters and affected by systemic AA-amyloidosis: Clinicopathological data, histopathology, and ultrastructural features
Supplemental material, sj-pdf-1-vet-10.1177_03009858241257903 for Renal alterations in cats (Felis catus) housed in shelters and affected by systemic AA-amyloidosis: Clinicopathological data, histopathology, and ultrastructural features by Filippo Ferri, Silvia Ferro, Silvia Lucia Benali, Luca Aresu, Lorenza Muscardin, Federico Porporato, Francesco Rossi, Chiara Guglielmetti, Enrico Gallo, Carlo Palizzotto, Carolina Callegari, Stefano Ricagno, Maria Mazza, Luigi Michele Coppola, Gabriele Gerardi, Francesca Lavatelli, Serena Caminito, Giulia Mazzini, Giovanni Palladini, Giampaolo Merlini and Eric Zini in Veterinary Pathology
Footnotes
Acknowledgements
We acknowledge Valter Fiore, Vittoria Franco, Tim Paul Schulte, and Sonia Iacoponi for technical support and DiBio Imaging Facility, Department of Biology, University of Padova for electron microscopy.
Authors’ Note
Meeting where the study was presented: 30th ECVIM-CA Congress Online, September 2020.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by BIRD 193233 University of Padova; 10/19 RC Istituto Zooprofilattico Sperimentale del Piemonte Liguria e Valle d’Aosta; and 2019 and 2020 Research Funds AniCura. This study was partially supported by Ricerca Corrente funding from Italian Ministry of Health to IRCCS Policlinico San Donato.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
