Abstract
Neurobrucellosis is a shared condition of cetaceans and humans. However, the pathogenesis and immune response in cetacean neurobrucellosis has not been extensively studied. In this multicentric investigation, 21 striped dolphin (Stenella coeruleoalba) neurobrucellosis (Brucella ceti) cases diagnosed over a 10-year period (2012-2022) were retrospectively evaluated. For each case, morphological changes were assessed by evaluating 21 histological parameters. Furthermore, the immunohistochemical expression of Brucella antigen, glial fibrillary acid protein (GFAP), and a selection of inflammatory cell (IBA-1, CD3, and CD20) and cytokine (tumor necrosis factor-alpha [TNF-α], interferon-gamma [IFN-γ], interleukin [IL]-1β, IL-2, and IL-6) markers were investigated. Inflammation of the leptomeninges, ependyma, and/or choroid plexus was lymphohistiocytic, containing macrophages/microglia (IBA-1+), T-cells (CD3+), and B-cells (CD20+) in equal proportion. B-cells occasionally formed tertiary follicles. GFAP expression showed astrocytosis in most cases. Expression of TNF-α, IFN-γ, and IL-2 indicated an intense proinflammatory response, stimulating both macrophages and T-cells. Our results showed that the inflammation and neuroinflammation in neurobrucellosis of striped dolphins mimic human neurobrucellosis and in vitro and in vivo studies in laboratory animals. Cetacean disease surveillance can be exploited to expand the knowledge of the pathogenesis and immunology of infectious diseases, particularly brucellosis, under a One Health approach.
Brucellosis is an important zoonosis endemic in many parts of the world, caused by some Brucella spp., most commonly Brucella melitensis, Brucella abortus, and Brucella suis (biovars 1 and 3). Other Brucella spp. can cause sporadic cases of brucellosis in humans, such as B. suis biovar 2 and 4, Brucella canis, Brucella neotomae, and marine mammal strains.46,57,58 Human infection with terrestrial Brucella mainly occurs via ingestion of contaminated unpasteurized dairy products, contact with infected animals, or inhalation of aerosols. 4
The multisystemic nature of brucellosis in humans is due to the involvement of the osteoarticular, lymphoid, hepatobiliary, and vascular systems. 4 Brucella can also affect the central nervous system (CNS),31,38 resulting in neurobrucellosis in up to 25% of brucellosis patients,24,47 especially immunocompromised individuals. 31 Neurological manifestations in neurobrucellosis may occur in both acute and chronic infections. 38
Neurobrucellosis in humans is clinically obscure. Brucella can affect any region of the CNS including the leptomeninges, cerebrum, cerebellum, brainstem, cranial nerves, spinal cord, and/or peripheral nerves, and this explains the broad spectrum of clinical presentations.14,31,35,55 In the postantibiotic era, mortality due to neurobrucellosis is around 5%, but permanent neurological deficits as sequel of infections are not infrequent.14,36
The clinical diagnosis of neurobrucellosis in humans is frequently achieved by exclusion. Neuroimaging supports clinical suspects, but neurobrucellosis confirmation requires ancillary techniques. Cerebrospinal fluid can be screened looking for cellular, protein, and/or glucose alterations. Brucella infection must be confirmed by means of bacterial isolation from cerebrospinal fluid or evidencing a systemic immune response against Brucella. 31 Novel techniques such as metabolomics may contribute to a rapid diagnosis. 63
Brucella reaches the CNS by hematogenous spread during early bacteremia or after reactivation in immunosuppressive states that facilitate Brucella immune evasion.31,38,43 The lesions induced during neurobrucellosis in humans comprise a direct effect of persisting Brucella, as well as indirect damage due to the host inflammatory response triggered by the bacteria or its endotoxins.31,42 Despite recent advances on the knowledge of neurobrucellosis in human beings, many aspects of the pathogenesis and inflammation in human neurobrucellosis remain elusive. This is partly because the lack of animal models mimicking the disease in humans. 43
Brucella ceti 16 is a phenotypically smooth strain causing cetacean brucellosis in mysticetes and odontocetes worldwide. 25 Brucellosis is regarded as endemic in many cetacean populations.9,40 Transmission may occur both horizontally (secretions, milk, and semen) and vertically.10,26,29,39 Some carnivorous species, such as killer whales (Orcinus orca), are thought to become infected by ingesting tissues of other cetaceans with brucellosis. 11 Pulmonary nematodes (Halocerus spp. and Pseudalius inflexus) harboring B. ceti are suspected to contribute as vectors to transmission in bottlenose dolphin (Tursiops truncatus) and harbor porpoise (Phocoena phocoena) based on bacteriology and immunohistochemical studies.11,29
Cetaceans from which B. ceti has been isolated are often found stranded, moribund, or dead. 46 Besides neurological disease, B. ceti causes reproductive (abortion, placentitis, endometritis, mastitis, and epididymo-orchitis), osteoarticular (arthritis and discospondylitis), cutaneous (blubber abscesses), and cardiac (myocarditis and endocarditis) lesions.21,22 The liver, kidney, spleen, lymph nodes, and lungs can also be affected.18,28,46
B. ceti involvement of the CNS (neurobrucellosis) has been particularly reported in the striped dolphin (Stenella coeruleoalba).9,12,18,23,26 Striped dolphins stranded alive affected from neurobrucellosis commonly show neurologic signs such as uncoordinated swimming, lethargy, circling, lateralization, inability to maintain buoyancy, opisthotonos, tremors, and/or tonic-clonic seizures.26,28 Gross findings include leptomeningeal clouding, acquired hydrocephalus, and turbid cerebrospinal fluid (CSF) in some cases.11,21
The hallmark histological lesion in the CNS is a nonsuppurative meningoencephalitis, periventriculitis, choroiditis, and/or radiculitis in cerebrum, cerebellum, brainstem, and/or spinal cord, along with occasional perivascular cuffing.1,9,18,20,21,23,26,28 Despite B. ceti seeming to be a major cause of morbidity and mortality in cetacean populations, 9 the pathogenesis and immunology of cetacean neurobrucellosis have not been extensively studied. 13 In this study, we aimed to describe the immunopathology of cetacean neurobrucellosis in the striped dolphin and to compare our results with previous studies in humans and in vitro and in vivo laboratory animal models of neurobrucellosis.
Materials and Methods
Retrospective Database Search
The databases of the World Organisation for Animal Health Collaborating Centre for the Health of Marine Mammals (Italian National Reference Centre for Diagnostic Activities in Stranded Marine Mammals, [C.Re.Di.Ma], Torino Italy) and of the Autonomous University of Barcelona (Spain) were searched for deceased striped dolphins (Stenella coeruleoalba) found stranded along the Italian or Catalonian coastlines, respectively, over a 10-year period (2012–2022). No ethical approval was required for this study because tissues from deceased wildlife animals submitted for routine diagnosis were used retrospectively.
Inclusion Criteria
Cases included (n = 21) had been submitted for autopsy and had available formalin-fixed, paraffin-embedded CNS samples. All cases were diagnosed with neurobrucellosis based on (1) Brucella isolation in the CNS and other organs (at least spleen, liver, and ovary/testis) using Brucella medium agar base, modified Thayer-Martin agar, CITA media, Farrell media, Columbia agar, and/or MacConkey agar;9,18,28 (2) B. ceti molecular confirmation (see below); and/or (3) positive serological results using a rapid serum agglutination test (Rose Bengal plate test) (distributed by the National Reference Laboratories for Brucellosis; reagent available only for official testing).9,23 For molecular confirmation in most cases, Brucella isolates were sub-cultured and subjected to DNA extraction using the Maxwell1 16 Tissue DNA Purification Kit and the Maxwell1 16 Instrument (Promega, Madison, Wisconsin) or the High Pure DNA Template Preparation kit (Roche Diagnostics, Mannheim, Germany) and were then identified as B. ceti using at least the PCR-RFLP (restriction fragment length polymorphism) method. 8 Case 12 and 20 were PCR positive (fresh tissue) in brain, lung, spleen, and/or blubber abscesses; seropositive; and showed Brucella-compatible gross and histological lesions, thus confirming a diagnosis of brucellosis. Data regarding postmortem condition code were recorded when available: code 1 (extremely fresh carcass, just dead), code 2 (fresh), code 3 (moderate decomposition), code 4 (advanced decomposition), or code 5 (mummified or skeletal remains) (Table 1).
Striped dolphin (Stenella coeruleoalba) cases with neurobrucellosis (Brucella ceti).
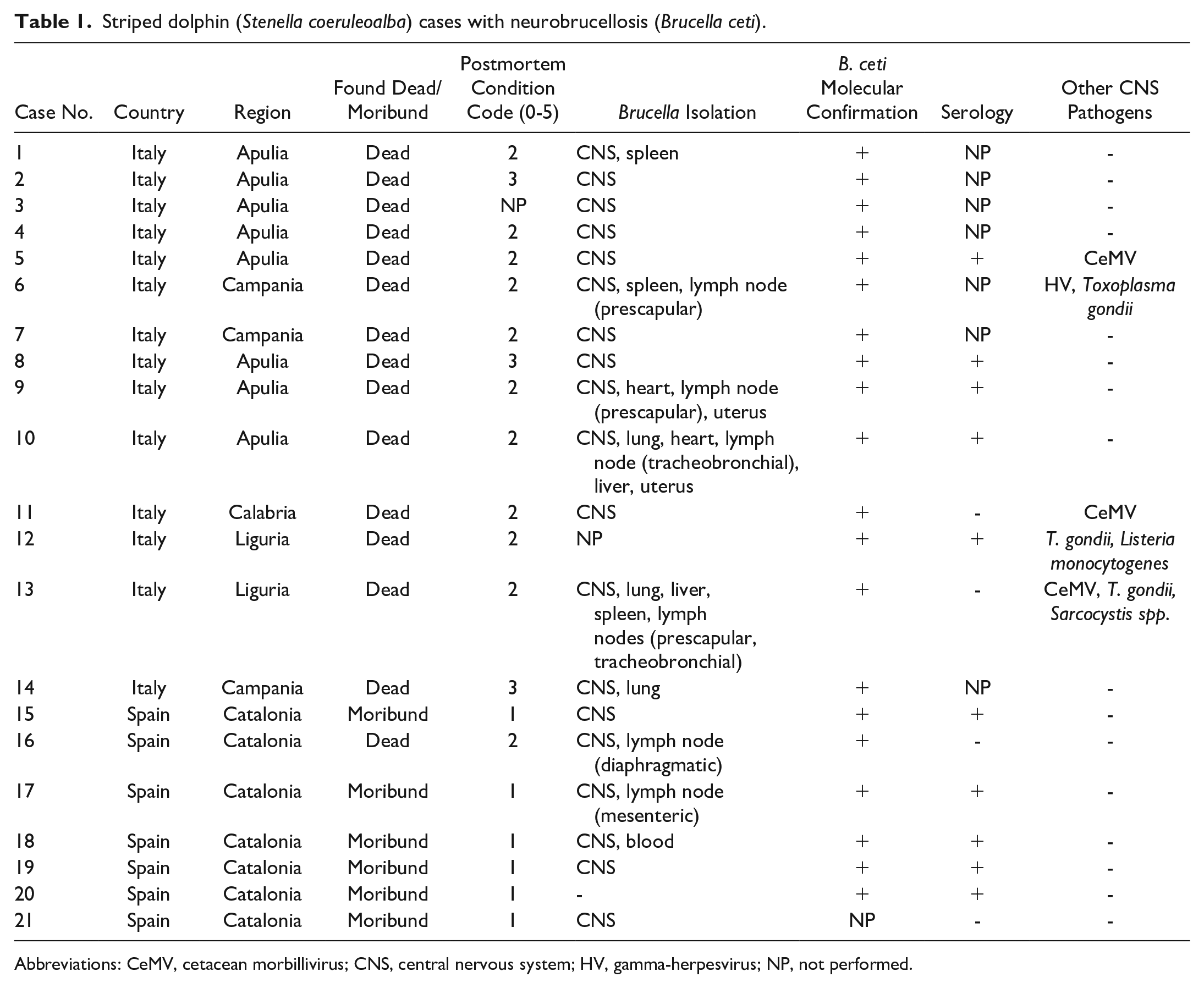
Abbreviations: CeMV, cetacean morbillivirus; CNS, central nervous system; HV, gamma-herpesvirus; NP, not performed.
Additional Testing
Additional testing results for other cetacean pathogens targeting the CNS had been performed in fresh tissues, including cetacean morbillivirus (PCR),32,60 gamma-herpesvirus (PCR), 59 Toxoplasma gondii (PCR), 61 and Listeria monocytogenes (bacterial culture) (Table 1). In cases found to be coinfected (cases 5, 6, 11, 12, and 13), sections included in this study were negative by immunohistochemistry (IHC) for cetacean morbillivirus, gamma-herpesvirus, and Toxoplasma gondii.9,23,52 L. monocytogenes and Sarcocystis spp. infection was excluded given the absence of characteristic histological lesions. 19
Histology
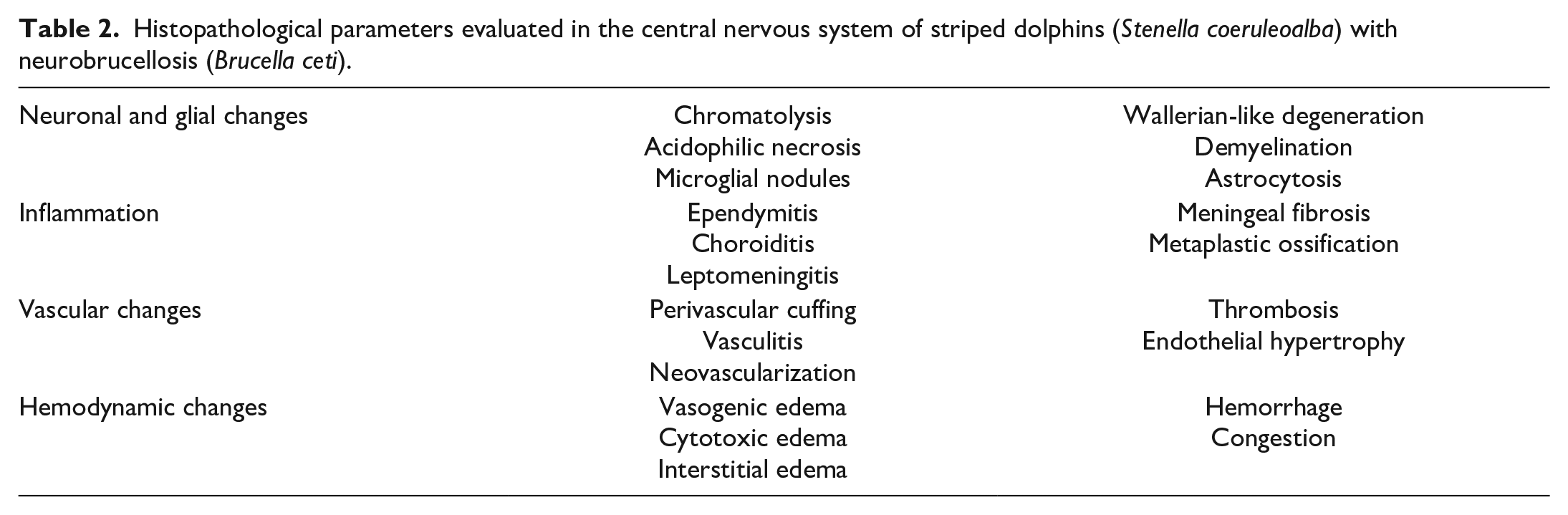
Necropsies had been performed, and a complete set of organs (CNS [cerebrum, cerebellum, brainstem, and spinal cord], lymph nodes, spleen, liver, pancreas, adrenal glands, kidneys, ovaries/testes, stomach, intestine, trachea, thyroid and parathyroid glands, thymus, lungs, heart, skin, blubber, and skeletal muscle) were collected and fixed by immersion in 10%, buffered, pH 7.2 formalin for 24 to 72 hours, before being routinely processed to produce 4-µm thick, hematoxylin and eosin stained sections. The Kluver-Barrera stain was performed when chromatolysis or demyelination was suspected. For each case, the 21 histological parameters detailed in Table 2 were semiquantitatively evaluated using a score from 0 to 3 (grade 0: absence of lesions; grade 1: focal lesions; grade 2: multifocal lesions; grade 3: diffuse lesions) in the hematoxylin and eosin section displaying the most severe inflammation (Table 1). Immunohistochemical expression of GFAP was used to assess astrocytosis (see below). The postmortem condition code did not affect the histological evaluation as for most animals the carcass was fresh.
Histopathological parameters evaluated in the central nervous system of striped dolphins (Stenella coeruleoalba) with neurobrucellosis (Brucella ceti).
Immunohistochemistry
For each case, consecutive paraffin sections scored histologically were placed on charged slides. Deparaffinization, rehydration, and heat-induced epitope retrieval was performed using a PT Lab Vision module (Thermo Fisher). Then, the endogenous peroxidase was quenched through immersion in a hydrogen peroxide solution in methanol (Panreac Química). The samples were incubated, first in horse serum and then in primary antibodies (Table 3).
Primary antibodies used for the immunohistochemical study of the central nervous system of striped dolphins (Stenella coeruleoalba) with neurobrucellosis (Brucella ceti).
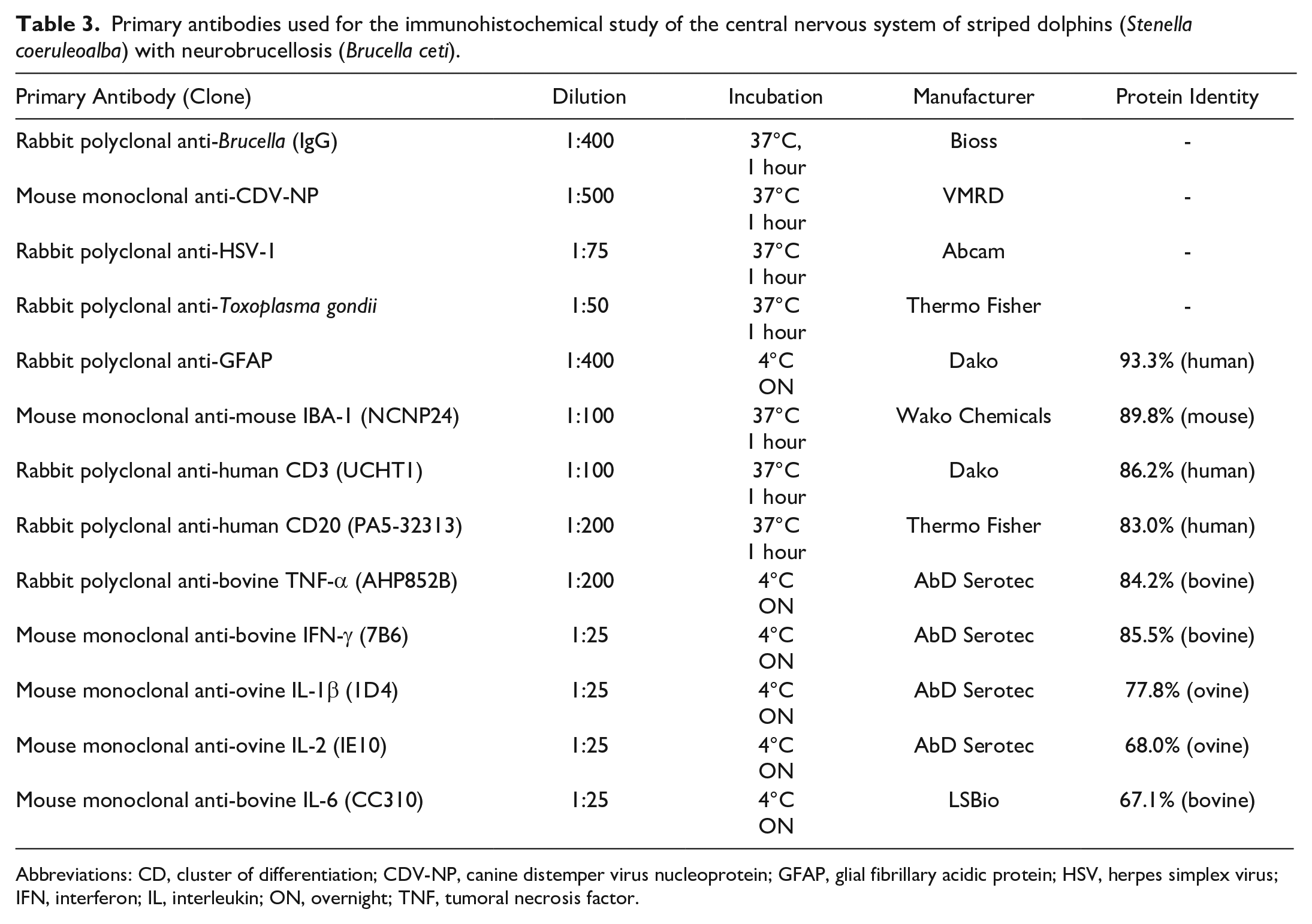
Abbreviations: CD, cluster of differentiation; CDV-NP, canine distemper virus nucleoprotein; GFAP, glial fibrillary acidic protein; HSV, herpes simplex virus; IFN, interferon; IL, interleukin; ON, overnight; TNF, tumoral necrosis factor.
Due to the high identity of cytokines between cetaceans and ruminants, 15 commercial antibodies for ruminant cytokines were used in this study. Antibody protein sequence identity between the bottlenose dolphin (Tursiops truncatus) and the target species of the antibody (human, mouse, bovine, or ovine) was assessed using pBLAST tool (National Center for Biotechnology Information, Bethesda, MA) (Table 3). The bottlenose dolphin was used as the cetacean reference because most protein sequences of striped dolphins are not characterized yet. Inflammatory cell and cytokine antibodies cross-reactivity (IBA-1, CD3, CD20, tumor necrosis factor-alpha [TNF-α], interferon-gamma [IFN-γ], interleukin [IL]-1β, IL-2, and IL-6), not specificity, was assessed in lymphoid tissues (spleen and lymph node) from the target species (normal tissue and a tuberculous granuloma), and bottlenose and striped dolphin (normal tissue and lymphadenitis or splenitis).
For all reactions except CD20, commercial reagents were used for the secondary antibody (ImmPRESS-VR horse anti-rabbit IgG polymer kit, Vector) and chromogen (ImmPACT NovaRED peroxidase substrate, Vector). Only for CD20, immunolabeling was performed on an autostainer (Autostainer Plus, Dako) using procedures, buffers, and solutions provided by the manufacturer; slides were incubated in DAB-chromogen-hydrogen peroxide solution (Dako, Denmark). In all cases, samples were counterstained with hematoxylin and coverslipped.
Formalin-fixed paraffin-embedded samples from the CNS and lymphoid organs from unrelated striped and bottlenose dolphins; from the target species of the antibody; or from animals infected by Brucella, cetacean morbillivirus, gamma-herpesvirus, or Toxoplasma gondii were used as positive controls when needed (depending on the primary antibody used). For negative controls, the primary antibody was replaced with a commercial universal negative control reagent.
Brucella immunolabeling was recorded as absent/present. Immunolabeling for the rest of antibodies was quantitatively evaluated in leptomeninges and/or choroid plexuses as follows: 100 cells were counted in 5 adjacent, non-overlapping fields under a high-power field (HPF) (400×, size = 0.237 mm2), and immunolabeled cells were set as a percentage of the cell count (0%-100%). A score from 0 to 3 was assigned to each sample for statistical purposes—grade 0, absence: 0% immunopositive cells; grade 1, low numbers (less than 30%) of immunopositive cells; grade 2, moderate numbers (30%-60%) of immunopositive cells; and grade 3, abundant (more than 60%) of immunopositive cells.
Statistics
To evaluate the relationship between the histopathological lesions observed and the immunohistochemical results, a Kendall tau-b correlation coefficient was calculated in R. 48
Results
Histopathology
Histologic lesions were observed in the cerebrum (19/21; 90%), cerebellum (20/21; 95%), brainstem (19/19; 100%), and spinal cord (16/16; 100%) (Table 4). Neuronal changes were characterized by minimal focal areas of chromatolysis (central) (mean score [MS] 0.76, 0.70 standard deviation [SD]) (Fig. 1g), microglial nodules (MS 0.38 ± 0.67 SD) (Fig. 1e), demyelination of the white matter (MS 0.38 ± 0.74 SD), and astrocytosis (GFAP immunolabeling) (MS 0.43 ± 0.75 SD) (Fig. 1f). Acidophilic necrosis and Wallerian-like degeneration were not found in any case. Inflammation (non-suppurative and lymphohistiocytic) was diffusely observed especially in the leptomeninges (MS 2.14 ± 0.96 SD) (Fig. 1a) compared with ependymal and subependymal areas (MS 0.52 ± 0.93 SD) (Fig. 1b), and the choroid plexus (MS 0.38 ± 0.86 SD) (Fig. 1c). Mild focal leptomeningeal fibrosis was observed in a few cases (MS 0.43 ± 0.75 SD). Metaplastic ossification of the leptomeninges was not observed.
Anatomic distribution of histologic lesions in the central nervous system of striped dolphins (Stenella coeruleoalba) with neurobrucellosis (Brucella ceti).
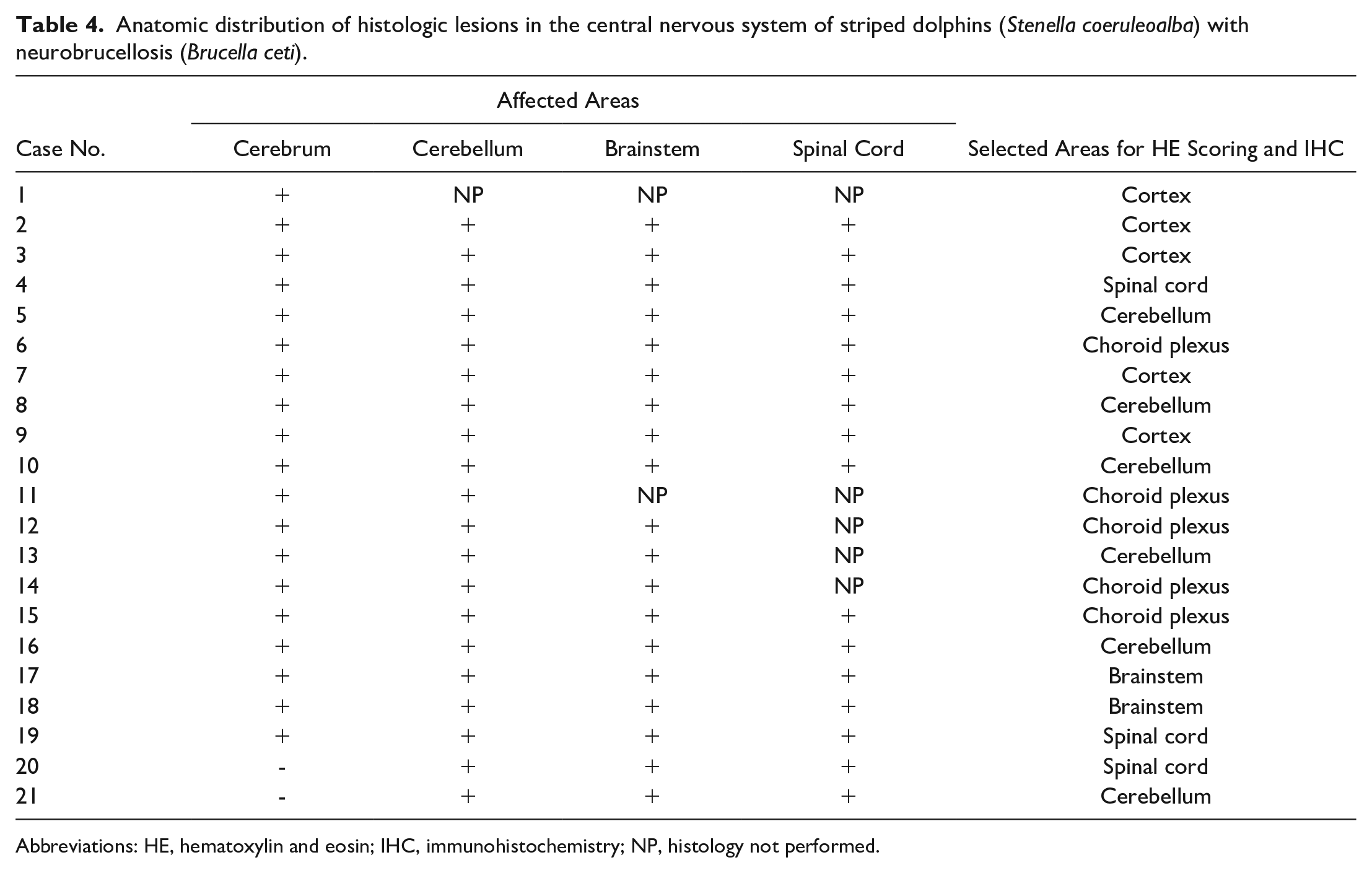
Abbreviations: HE, hematoxylin and eosin; IHC, immunohistochemistry; NP, histology not performed.
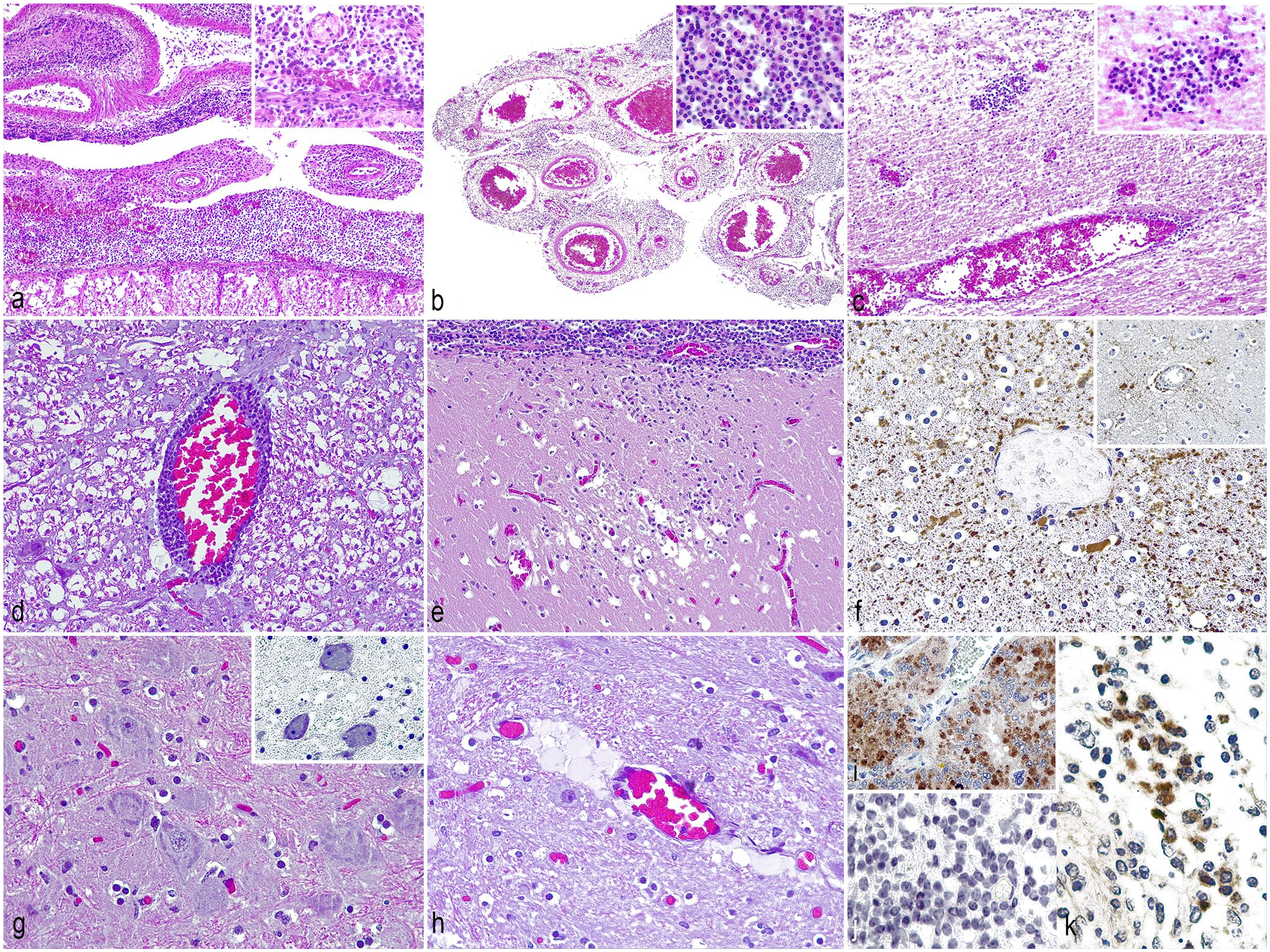
Histological and immunohistochemical findings in neurobrucellosis (Brucella ceti) of striped dolphins (Stenella coeruleoalba). (a) Spinal cord. Leptomeningitis, lymphohistiocytic, diffuse, chronic, severe. Case 4. Hematoxylin and eosin (HE). (b) Lateral ventricle. Choroid plexitis, lymphohistiocytic, diffuse, chronic, severe. Case 14. HE. (c) Cerebrum. Ependymitis and subependymal perivascular encephalitis, lymphohistiocytic, multifocal, chronic, and moderate. Case 3. HE. (d) Spinal cord. Blood vessels are surrounded by a non-suppurative inflammatory infiltrate (perivascular cuffing). Case 4. HE. (e) Cerebrum. Microglial nodule adjacent to an area of leptomeningitis. Case 3. HE. (f) Cerebrum. Astrocytosis assessed as an increased expression of glial fibrillary acidic protein (GFAP). Inset: GFAP expression in a negative control dolphin. Case 2. GFAP immunohistochemistry (IHC). (g) Cerebrum. Central chromatolysis. Case 3. HE. Inset: centrifugal distribution and condensation of Nissl granules and peripheral nuclear displacement. Kluver-Barrera. (H) Spinal cord. Vasogenic edema. Case 19. HE. (i) Positive control, presence of Brucella antigen in the placenta of a sheep experimentally infected with B. melitensis. Brucella IHC. (j) Negative control, absence of Brucella antigen in an area of leptomeningitis of a dolphin not infected by B. ceti. Brucella IHC. (k) Striped dolphin with neurobrucellosis. Presence of Brucella antigen in the cytoplasm of macrophages. Brucellai IHC.
Regarding vascular changes, multifocal perivascular cuffing (nonsuppurative, lymphohistiocytic) was the most common finding (MS 1.29 ± 0.78 SD) (Fig. 1d). Endothelial cells of leptomeninges and parenchymal blood vessels were occasionally hypertrophied (MS 0.86 ± 0.73 SD). Rarely, thrombosis was observed (MS 0.29 ± 0.46 SD). Vasculitis and neovascularization were not observed. Hemodynamic changes consisted of multifocal vasogenic (MS 0.86 ± 0.65 SD) (Fig. 1h) and cytotoxic (MS 0.29 ± 0.46 SD) edema, hemorrhage (MS 0.95 ± 0.74 SD), and congestion (MS 0.86 ± 0.65 SD). Interstitial edema was almost absent (MS 0.05 ± 0.22 SD).
Individual histological parameters are shown in Supplemental Table S1.
Immunohistochemistry
Brucella immunolabeling was observed intracellularly in scattered macrophages located in the areas of leptomeningitis, ependymitis, or choroiditis in all cases (Fig. 1k) and occasionally in inflammatory cells surrounding noninflamed blood vessels in the neuroparenchyma.
Inflammation consisted of IBA-1+ macrophages and microglia, CD3+ T-cells, and CD20+ B-cells. IBA-1+ cells (macrophages/microglia) were multifocally observed in the areas of inflammation in the leptomeninges, ependyma, or choroid plexus (Fig. 2a). IBA-1+ cells were concentrated mainly in the outer areas of perivascular inflammation in the leptomeninges, ependyma, and choroid plexus. IBA-1+ cells were in low (grade 1, 16/21; 76%) or moderate (grade 2, 5/21; 24%) numbers (MS 1.24 ± 0.44 SD). CD3+ cells (T-cells) were in the leptomeningeal, ependymal, or choroidal areas of inflammation in a random distribution (Fig. 2b), but also in areas of neuronophagia (microglial nodules). T-cells were in low numbers (grade 1, 14/21; 67%), moderate numbers (grade 2, 5/21; 24%), or abundant (grade 3, 2/21; 9%) (MS 1.43 ± 0.68 SD). Membranous expression of CD20 was seen in B-cells in the areas of inflammation. B-cells were found randomly but occasionally formed lymphoid follicles (6/21; 29%) (Fig. 2c). B-cells were absent (grade 0, 1/21; 5%), in low numbers (grade 1, 8/21; 38%), in moderate numbers (grade 2, 9/21; 43%), or abundant (grade 3, 3/21; 14%) (MS 1.67 ± 0.80 SD).
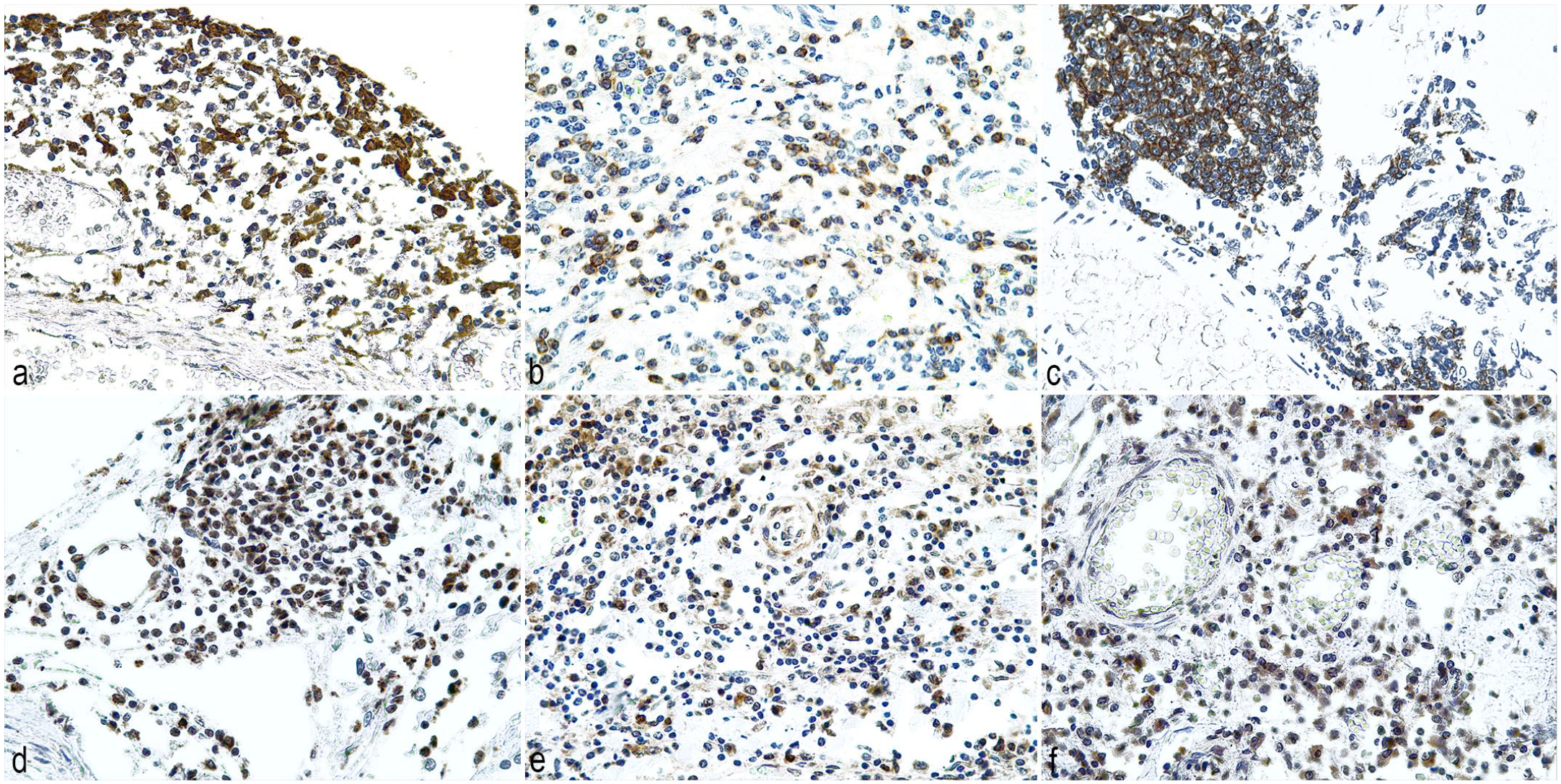
Immunohistochemical findings in neurobrucellosis (Brucella ceti) of striped dolphins (Stenella coeruleoalba). (a) Macrophages/microglia are located multifocally in areas of leptomeningitis. Case 5. IBA-1 immunohistochemistry (IHC). (b) T-cells are multifocally distributed in the inflamed leptomeninges. Case 17. CD3 IHC. (c) B-cells are multifocally distributed and occasionally forming lymphoid follicles in the leptomeninges. Case 15. CD20 IHC. (d) Tumor necrosis factor-alpha (TNF-α) is expressed in inflammatory cells in a multifocal distribution. Case 9. TNF-α IHC. (e) Interferon-gamma (IFN-γ)-expressing inflammatory cells are multifocally distributed in inflamed areas. Case 2. IFN-γ IHC. (f) Interleukin-2 (IL-2) is expressed in inflammatory cells distributed multifocally in the inflamed leptomeninges. Case 10. IL-2 IHC.
Cytokines evaluated included TNF-α, IFN-γ, IL-2, IL-1β, and IL-6. TNF-α was expressed in the cytoplasm of inflammatory cells (macrophages and lymphocytes based on cell morphology) (Fig. 2d), especially around blood vessels of inflamed leptomeninges, ependyma, and choroid plexuses. TNF-α-expressing cells were absent (grade 0, 1/21; 5%), in low numbers (grade 1, 14/21; 67%), in moderate numbers (grade 2, 5/21; 23%), or abundant (grade 3, 1/21; 5%) (MS 1.29 ± 0.64 SD). The expression of IFN-γ was observed in the cytoplasm of inflammatory cells (lymphocytes and macrophages based on cell morphology) and was distributed randomly through the areas of inflammation (Fig. 2e). IFN-γ-expressing cells were absent (grade 0, 2/21; 9%), in low numbers (grade 1, 11/21; 53%), in moderate numbers (grade 2, 7/21; 33%), or abundant (grade 3, 1/21; 5%) (MS 1.33 ± 0.73 SD). IL-2 expression occurred in the cytoplasm of inflammatory cells (lymphocytes and microglia based on cell morphology), randomly and eccentrically distributed with respect to blood vessels in inflamed areas (Fig. 2f). IL-2-expressing cells were absent (grade 0, 2/21; 9%), in low numbers (grade 1, 10/21; 48%), or in moderate numbers (grade 2, 9/21; 43%) (MS 1.33 ± 0.66 SD). Antibodies targeting IL-1β and IL-6 did not show immunoreactivity in dolphin tissues. IL-1β immunoreactivity was observed in the target species but not in dolphin tissues, and IL-6 immunoreactivity was not observed in either the target species or dolphins.
Individual immunohistochemical parameters and are shown in Supplemental Table S1. Individual immunohistochemical expression of IBA-1, CD3, CD20, TNF-α, IFN-γ, and IL-2 and distribution among histological grades is shown in Fig. 3.
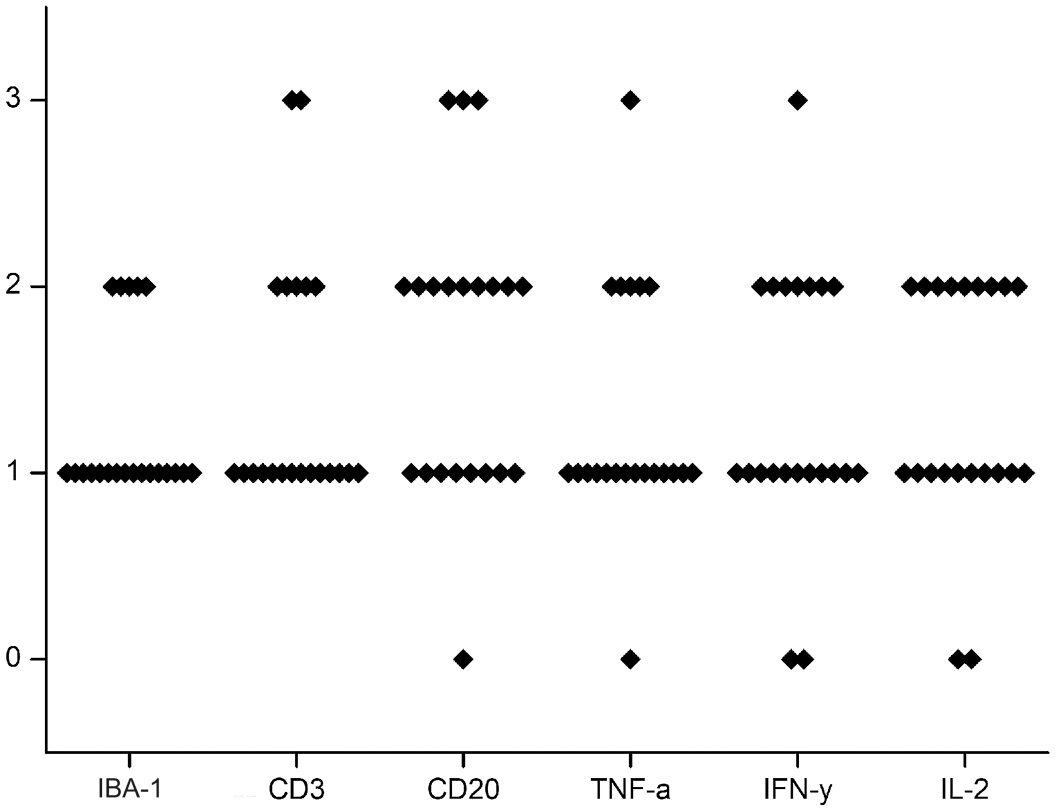
Immunohistochemical findings in striped dolphins (Stenella coeruleoalba) with neurobrucellosis. Semiquantitative results (0-3) of immunohistochemical expression of IBA-1, CD3, CD20, tumor necrosis factor-alpha (TNF-α), interferon-gamma (IFN-γ), and interleukin-2 (IL-2).
Statistics
Statistical correlations between immunohistochemical and histopathological score values showed a significant moderate positive correlation between IBA-1 expression and the degree of leptomeningitis (τB = 0.56 [95% confidence interval [CI] = 0.26–0.85]; P = .0078). Endothelial hypertrophy showed a significant weak correlation with CD20 expression (τB = 0.47 [95% CI = 0.23–0.71]; P = .019). Also, there was a significant moderate correlation between perivascular cuffing and TNF-α expression (τB = 0.50 [95% CI = 0.21–0.79]; P = .015). Astrocytosis (GFAP expression) and IL-2 expression showed a significant moderate correlation (τB = 0.50 [95% CI = 0.23–0.77]; P = .017). Finally, there was a significant weak correlation between thrombosis and IL-2 expression (τB = 0.49 [95% CI = 0.19–0.80]; P = .017). No other significant correlations were observed between immunohistochemical and histopathological score values (Supplemental Figure S1).
Discussion
The pathogenesis of neurobrucellosis is not completely understood. 43 Bacteremia during brucellosis facilitates Brucella invasion of the CNS in a phagocyte-facilitated manner. 14 In vitro investigations demonstrated that B. abortus transverses human brain microvascular endothelial cell monolayers using monocytes as a Trojan horse, 43 infecting endothelial cells, and trans-endothelially in inflammatory cells.34,42,51 Localization of Brucella in the CNS lesions of dolphins was observed intracellularly in macrophages surrounding vascular areas in most cases in our study, but the presence of Brucella antigens was lower than expected considering the severity of the lesions. This agrees with the reported fastidious histochemical and immunohistochemical detection of Brucella in neurobrucellosis in both humans and dolphins,21,25,38,54,56 supporting the hypothesis that the pathological consequences of neurobrucellosis are more related to the immune response elicited by the host than to the presence of intralesional bacteria.4,46
Wildlife often exhibit comorbidities; in this study, in cases where co-infections were observed, cetacean neuropathogens were not detected by IHC. No anatomical predisposition within CNS was observed in our study; notably, the cerebrum was not affected in 2 cases. The results obtained coincide with previous investigations that reported that dolphins suffering from neurobrucellosis develop inflammation in the leptomeninges, choroid plexuses, ependyma, and in a perivascular distribution within the neuroparenchyma.1,9,18,21,23,25,26,28,56 Leptomeninges are the preferred location for B. ceti to elicit an inflammatory response in dolphins. Concordantly, Brucella has a great affinity for leptomeninges according to reported biopsies or autopsies in human cases of neurobrucellosis.14,31,38
Lymphocytes stand out as the main cellular population in cetacean1,9,21,25,26,28,56 and human2,38,54 neurobrucellosis, even in the cerebrospinal fluid in humans.14,31,33,36,38,42 T-cells account for the most abundant lymphocyte population in human neurobrucellosis cases.24,54 T-cells are pivotal for controlling intracellular pathogens in the CNS. 54 However, autoreactive T-cells also facilitate tissue damage mediated by excessive inflammation. 49 B-cells have been reported in the leptomeninges in human neurobrucellosis patients in a lower proportion compared with T-cells. 49 Conversely, we observed a significant B-cell population in cetacean neurobrucellosis, occasionally forming tertiary follicles in the leptomeninges. Tertiary follicles contribute to inflammation in the CNS acting as antigen presenters, proinflammatory cytokine secretors, and antibody producers in persistent infections. 45 Therefore, B-cell tertiary follicles in cetacean neurobrucellosis may be indicators of disease chronicity or severity, mimicking their function in other organs, and in the CNS in some human diseases. 45
T-cell production of TNF-α and IFN-γ stimulate natural killer cells and macrophages to phagocytize and produce nitric oxide to control Brucella infection.5,27,30 Chronic brucellosis cannot be overcome by increasing the production of IFN-γ because Brucella inhibit the IFN-γ-mediated activation of macrophages and/or accelerates the inactivation of IFN-γ-activated macrophages.5,30 Therefore, the expression of IFN-γ would be inefficient to control cetacean neurobrucellosis based on the intralesional presence of Brucella antigens. Indeed, rats chronically infected (2 months) with B. melitensis do not show an induction of the IFN-γ mRNA levels in the brain. 41 The expression of IBA-1 was correlated with the rate of leptomeningitis, suggesting that macrophages/microglia contribute in a relevant manner to the severity of inflammation. Unlike cetaceans, some human neurobrucellosis cases report cerebral non-necrotizing granulomas or abscesses2,14,33,54,57 with occasional multinucleated giant cells 57 or nongranulomatous inflammation of the leptomeninges.38,54
Brucella infection of monocytes in vitro precedes the infection of astrocytes and microglia. 43 Microgliosis and astrocytosis occur during human neurobrucellosis,2,17,33,38,54,57 in macaques infected with B. melitensis, 34 and in the cetaceans studied here. B. abortus has been reported to target microglia in a preferential manner, 17 and most of the neuronal death observed in neurobrucellosis seem to occur due to the microglial release of nitric oxide. 50 Thus, microglial nodules and neuronal death observed in dolphins are likely consequences of Brucella infection of microglia rather than due to a direct neurotropic effect, as suggested in mice that showed absence of neuronal death after B. abortus infection if neurons were not cocultured with microglia. 50 Both astrocyte proliferation and apoptosis have been reported in neurobrucellosis. On one hand, astrocytes, but not microglia, are susceptible to B. abortus-induced apoptosis. 17 On the other hand, astrocytic proliferation in neurobrucellosis follows the secretion of proinflammatory cytokines by microglia. 17
The expression of IL-2 in striped dolphin neurobrucellosis cases suggests a role of this cytokine in differentiation, stimulation, and amplification of T-cells in response to brucellosis. 6 Microglia in cetacean neurobrucellosis may express IL-2 upon stimulation of Brucella lipopolysaccharide, as reported in mice, 53 to stimulate T-cell to produce TNF-α. 27 However, mice microglia and astrocytes can secrete TNF-α in vitro if stimulated by Brucella outer membrane proteins (Omp31 and Omp25),35,62 B. abortus Omp19,4,44 or B. abortus.17,37,42 TNF-α mRNA levels have been found to increase in the brain of rats chronically infected (1 month) with B. melitensis 48 and in a murine monocyte-macrophage cell line (Raw 264.7 cells) infected with B. suis S2. 64 Similarly, TNF-α was found to be overexpressed in the CNS of the B. ceti-infected dolphins studied here. TNF-α not only activates effector cells that limit Brucella survival, 6 as is suggested here based on the correlation between TNF-α and the severity of perivascular cuffing, but also activates brain endothelium increasing endothelial permeability of the blood-brain barrier in vitro. 42
A limitation of this study is that the specificity of antibodies could not be assessed without specific purified antigens. Despite several antibodies in ruminants (Ruminantiamorpha) recognize conserved epitopes in cetaceans (Cetancodontamorpha), 15 some ruminant-targeted antibodies used (IL-1β and IL-6) did not react in cetacean tissues despite their moderate to high protein sequence identity. The contribution of IL-1β and IL-6 in cetacean neurobrucellosis is warranted given that these cytokines are upregulated in vivo and in vitro in brucellosis in humans and mice according to genomic and serologic studies.33,37,42,55,63 –65
Stroke, aneurisms, and submeningeal hemorrhage have been attributed to a Brucella-induced vasculitis in some human neurobrucellosis cases based on magnetic resonance imaging,3,7,31,55 but these have not been reported in cetacean neurobrucellosis. However, excessive inflammation and neuroinflammation in the CNS lead to blood-brain barrier dysfunction manifesting as edema and hemorrhage. 49 Brucella is known to infect endothelial cells to traverse the blood-brain barrier.34,51 Furthermore, in vitro studies showed that B. abortus stimulates microglia to produce IL-1β, activating the brain microvasculature in vitro, 42 but also platelets that activate human brain microvascular endothelial cells and promote trans-endothelial migration of inflammatory cells.42,51 Both mechanisms explain the blood-brain barrier activation and dysfunction reported during neurobrucellosis, 42 as well as the hemodynamic changes of the dolphins here studied. Perivascular inflammation in cetacean neurobrucellosis probably occurs due to the trans-endothelial migration of inflammatory cells in neurobrucellosis. 51
Rarely, human neurobrucellosis can induce CNS demyelination;14,31 however, this finding is not considered an outstanding histological feature based on studies performing specific myelin stains (luxol fast blue). 54 Demyelination was not widespread in the CNS of dolphins in our study. However, peripheral nervous system demyelination can occur in human patients suffering from brucellosis because of physical compression of the spinal cord or a Guillain-Barré-like syndrome leading to neuropathies.4,14,31 Therefore, peripheral nervous system sampling and evaluation should be systematically performed in future cetacean neurobrucellosis cases to assess whether lesions also occur in the peripheral nervous system of this species.
To conclude, neurobrucellosis is a shared condition of cetaceans and humans. The immune response in neurobrucellosis of striped dolphins mimics in vitro and in vivo findings of neurobrucellosis in humans and animal models. Inflammation of the leptomeninges, ependyma, and/or choroid plexus contained macrophages/microglia, T-cells, and B-cells. B-cells occasionally formed tertiary follicles. GFAP expression demonstrated astrocytosis. Expression of TNF-α, IFN-γ, and IL-2 indicates an intense proinflammatory response. Cetacean disease surveillance is valuable not only for diagnosing threats to endangered species but can also be exploited to expand the knowledge of the pathogenesis and immunology of infectious diseases, particularly brucellosis, under a One Health approach.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241250336 – Supplemental material for Neurobrucellosis (Brucella ceti) in striped dolphins (Stenella coeruleoalba): Immunohistochemical studies on immune response and neuroinflammation
Supplemental material, sj-pdf-1-vet-10.1177_03009858241250336 for Neurobrucellosis (Brucella ceti) in striped dolphins (Stenella coeruleoalba): Immunohistochemical studies on immune response and neuroinflammation by Agustín Rebollada-Merino, Federica Giorda, Martí Pumarola, Laura Martino, Alberto Gomez-Buendia, Umberto Romani-Cremaschi, Cristina Casalone, Virginia Mattioda, Fabio Di Nocera, Giuseppe Lucifora, Antonio Petrella, Lucas Domínguez, Mariano Domingo, Carla Grattarola and Antonio Rodríguez-Bertos in Veterinary Pathology
Footnotes
Author Contributions
AR-M, MD, and AR-B designed and performed the experiments; FG, MP, LM, UR-C, CC, VM, FDN, GL, AP, LD, and CG contributed to the experimental design; AR-M, FG, LM, MD, and AR-B performed histologic evaluations; AG-B performed statistical analysis; the manuscript was written by AR-M with contribution from the other authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
