Abstract
Clinical History and Gross Findings
A 4-year-old, spayed female ferret was referred to the Zoological Companion Animal Medicine service at Tufts University for a firm swelling on the right cranial elbow. The swelling was noticed by the owner 10 days prior to the first visit. The animal did not have systemic clinical signs or signs of lameness; however, she objected to extension of the affected elbow. Initial diagnostics included radiographs and cytology of the right elbow.
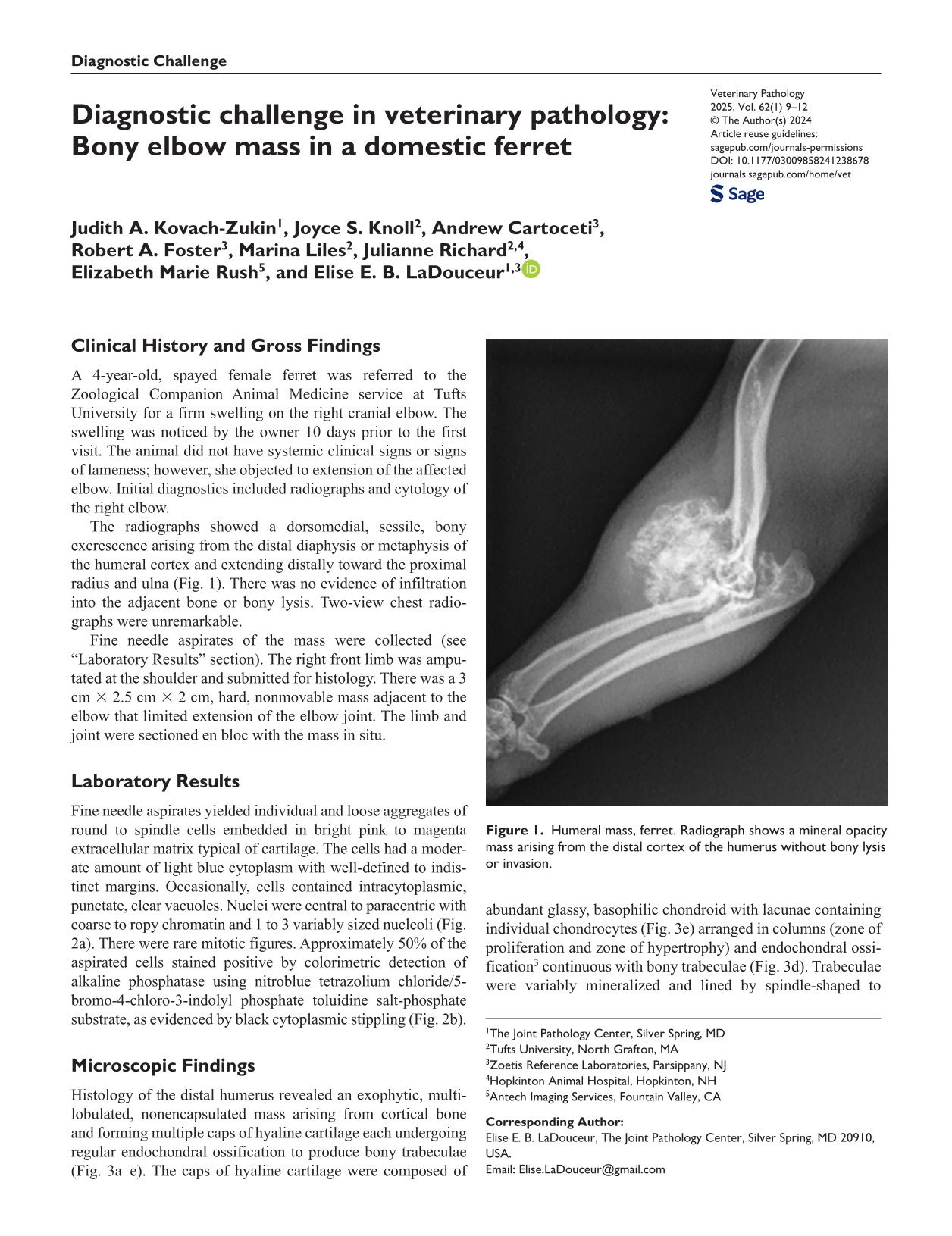
The radiographs showed a dorsomedial, sessile, bony excrescence arising from the distal diaphysis or metaphysis of the humeral cortex and extending distally toward the proximal radius and ulna (Fig. 1). There was no evidence of infiltration into the adjacent bone or bony lysis. Two-view chest radiographs were unremarkable.
Humeral mass, ferret. Radiograph shows a mineral opacity mass arising from the distal cortex of the humerus without bony lysis or invasion.
Fine needle aspirates of the mass were collected (see “Laboratory Results” section). The right front limb was amputated at the shoulder and submitted for histology. There was a 3 cm × 2.5 cm × 2 cm, hard, nonmovable mass adjacent to the elbow that limited extension of the elbow joint. The limb and joint were sectioned en bloc with the mass in situ.
Laboratory Results
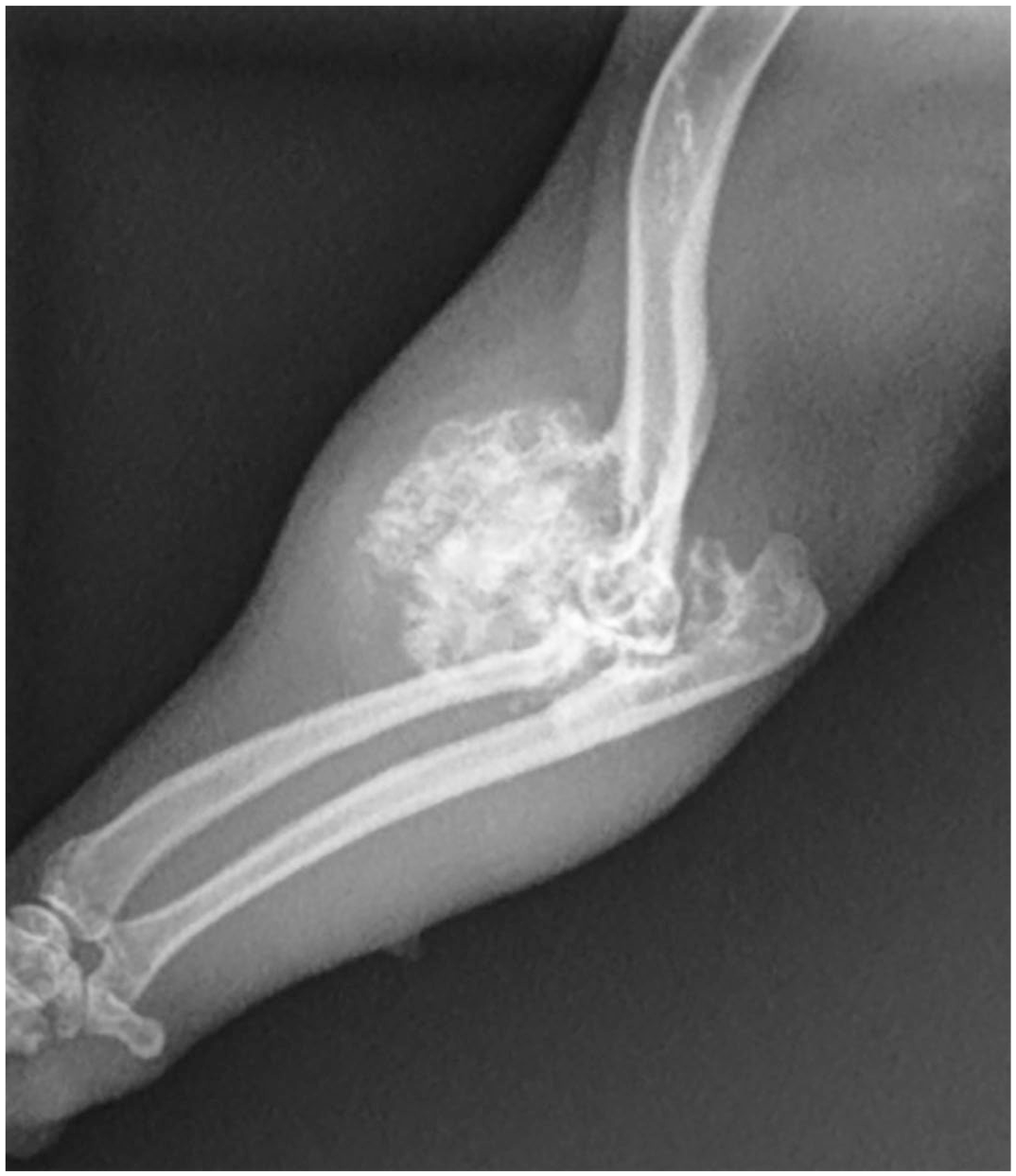
Fine needle aspirates yielded individual and loose aggregates of round to spindle cells embedded in bright pink to magenta extracellular matrix typical of cartilage. The cells had a moderate amount of light blue cytoplasm with well-defined to indistinct margins. Occasionally, cells contained intracytoplasmic, punctate, clear vacuoles. Nuclei were central to paracentric with coarse to ropy chromatin and 1 to 3 variably sized nucleoli (Fig. 2a). There were rare mitotic figures. Approximately 50% of the aspirated cells stained positive by colorimetric detection of alkaline phosphatase using nitroblue tetrazolium chloride/5-bromo-4-chloro-3-indolyl phosphate toluidine salt-phosphate substrate, as evidenced by black cytoplasmic stippling (Fig. 2b).
Humeral mass, ferret. (a) Fine needle aspirate of the mass reveals spindle cells with pleomorphism, prominent nucleoli, and a mitotic figure (arrow). Aqueous Romanowsky stain. (b) Colorimetric staining for alkaline phosphatase reveals positive staining (black, arrow) in the cytoplasm spindle cells. Nitroblue tetrazolium chloride/5-bromo-4-chloro-3-indolyl phosphate toluidine salt.
Microscopic Findings
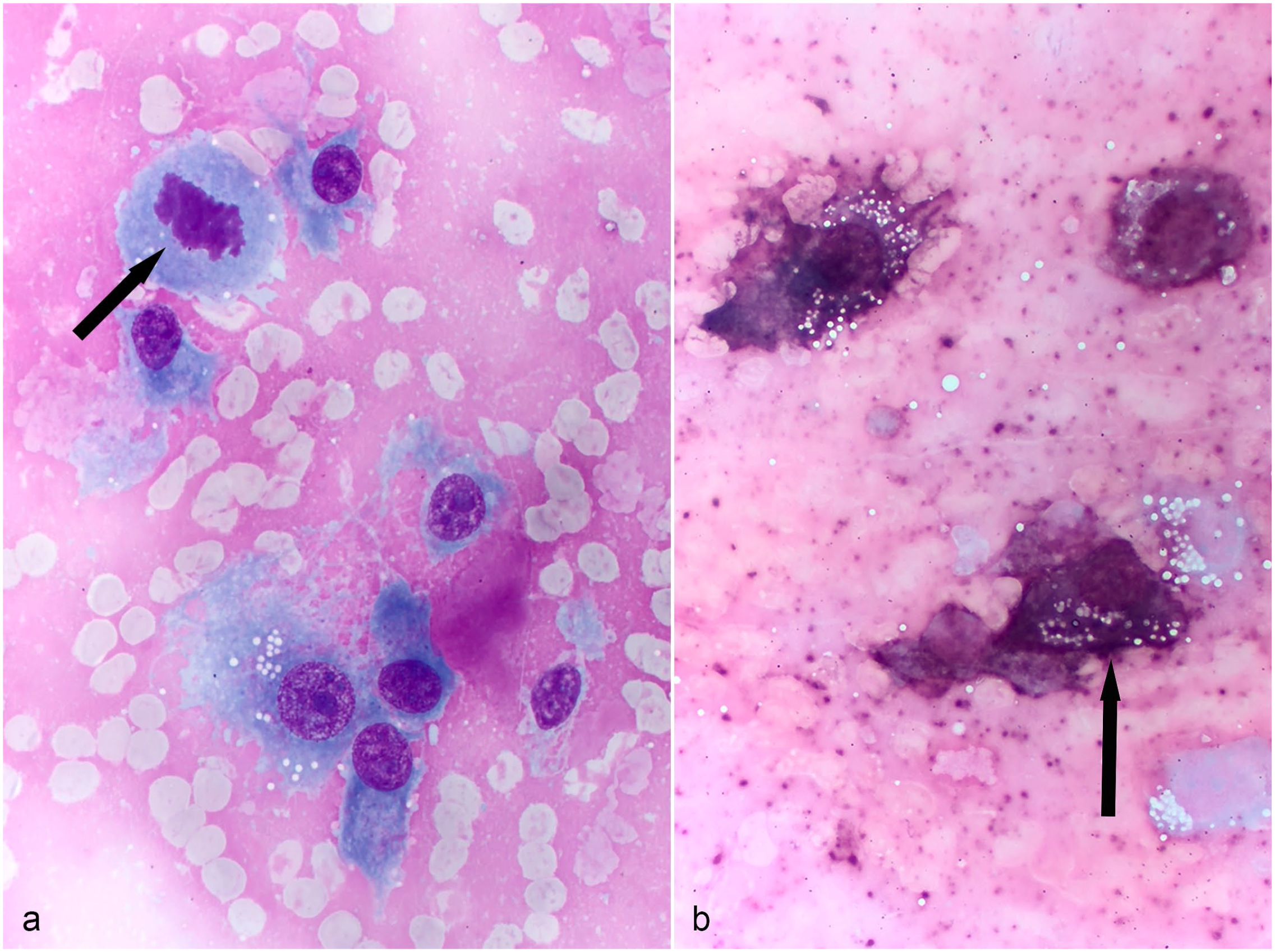
Histology of the distal humerus revealed an exophytic, multilobulated, nonencapsulated mass arising from cortical bone and forming multiple caps of hyaline cartilage each undergoing regular endochondral ossification to produce bony trabeculae (Fig. 3a–e). The caps of hyaline cartilage were composed of abundant glassy, basophilic chondroid with lacunae containing individual chondrocytes (Fig. 3e) arranged in columns (zone of proliferation and zone of hypertrophy) and endochondral ossification 3 continuous with bony trabeculae (Fig. 3d). Trabeculae were variably mineralized and lined by spindle-shaped to cuboidal osteoblasts (Fig. 3b) and fewer osteoclasts in erosion lacunae. Intertrabecular spaces contained loosely aggregated mesenchymal cells with fibrous tissue or adipocytes, hematopoietic progenitor cells, and small caliber blood vessels (bone marrow) (Fig. 3c). Mesenchymal cells were spindle-shaped with a moderate amount of eosinophilic cytoplasm and oval nuclei with prominent nucleoli and rare mitotic figures (1 per 2.37 mm2). The bony trabeculae were continuous with the pre-existing bone and communicated with the underlying medullary cavity. The mass expanded and compressed surrounding skeletal muscle and connective tissue.
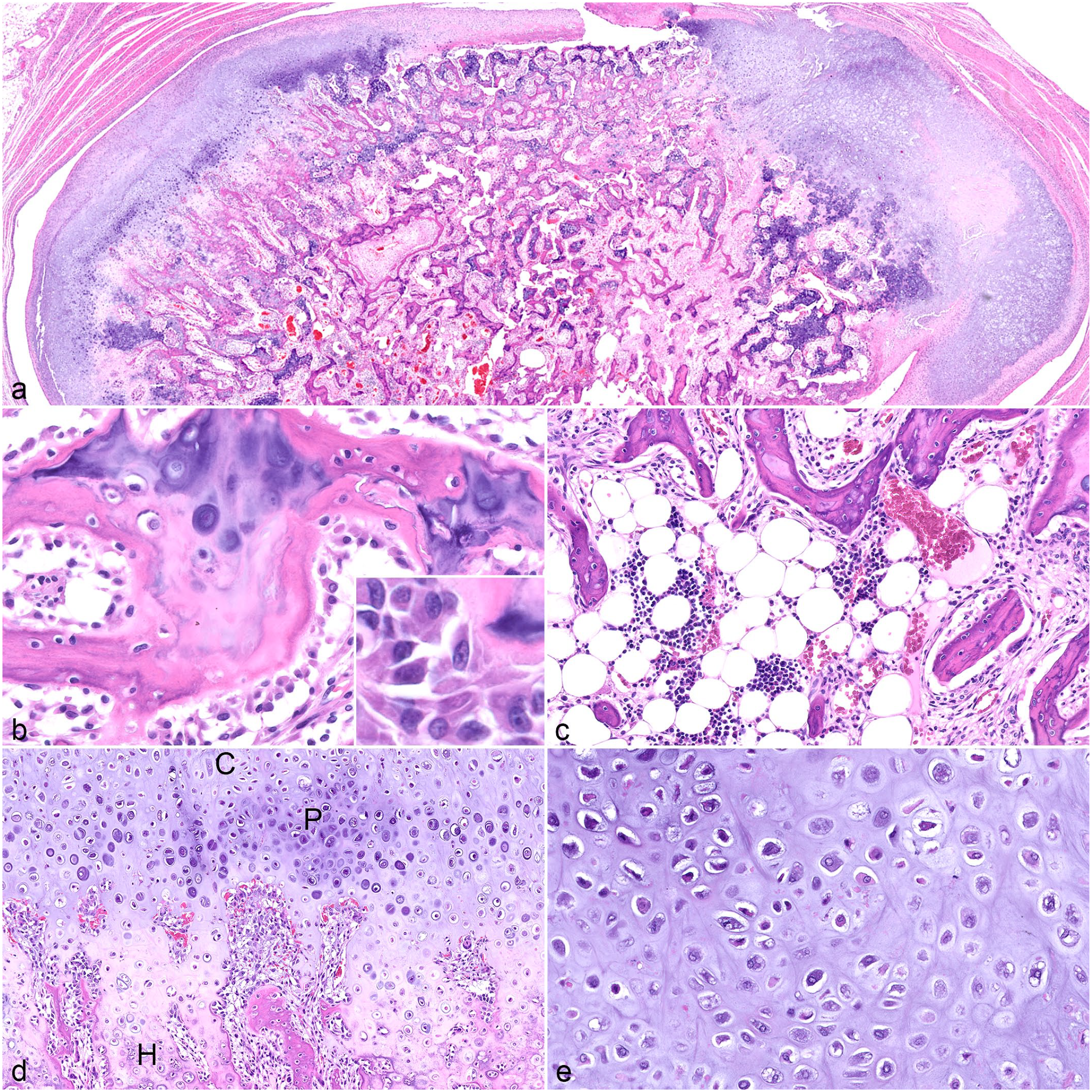
Humeral mass, ferret, hematoxylin and eosin. (a) The mass is made up of a cap of hyaline cartilage that arises from cortical bone and forms endochondral ossification. (b) Bony trabeculae are lined by osteoblasts. Inset: high magnification of osteoblasts. (c) Inter-trabecular spaces contain either fibrous tissue (right side of image) or adipocytes with hematopoietic precursors (bone marrow). (d) The cartilage cap (C) is subtended by endochondral ossification with prominent proliferative (P) and hypertrophic zones (H). (e) Cartilage is composed of basophilic matrix studded with lacunae encircling chondrocytes.
Differential Diagnoses
The primary cytologic differential diagnosis was osteosarcoma based on the predominantly spindle-shaped cells with few malignant morphologic features (ie, prominent nucleoli and rare mitotic figures) and positive alkaline phosphatase staining. Radiographically, the lesion was mineralized and arose from the underlying humeral cortex with no evidence of underlying bony lysis. The primary differential diagnoses for these radiographical findings were osteochondroma and parosteal or periosteal osteosarcoma. Osteomas in ferrets usually occur in flat bones, so osteoma was considered unlikely for this lesion. Histologic findings generated a definitive diagnosis of osteochondroma due to the characteristic microscopic features of cartilage caps, endochondral ossification, communication of the neoplasm with the underlying cortex and medullary cavity, and the absence of features of malignancy.
Discussion
Osteochondromas are cartilage-capped bony proliferations that arise from the surface of endochondral bones. They can be solitary (monostotic) or multiple (polyostotic); the latter is often hereditary and most often diagnosed in dogs and horses. The pathogenesis of solitary osteochondromas is not fully understood. They may represent true neoplasms or displaced physeal cartilage. Many osteochondromas occur near a physis in growing animals, and growth ceases after skeletal maturity. Osteochondromas also arise from trauma or degeneration of the cartilage. 7 Trauma was not in this animal’s history, and there was no evidence of cartilage degeneration or other degenerative joint disease radiographically or histologically. The lesion in this case arose in a skeletally mature animal (4 years old) and was noticed by the owner 10 days prior to initial presentation. Reports of osteochondromas arising in adult animals without trauma or degenerative joint disease are rare. 6 Osteochondromatosis has been described in cats but appears to represent a separate entity than that described above. Osteochondromatosis in cats arises most commonly from flat bones with intramembranous ossification and does not communicate with the underlying marrow cavity; viral induction has been suggested in cats. 3
Radiographically, the mass was an ossified, noninvasive lesion that arose from the cortex. There was no evidence of underlying bony lysis or proliferation in the adjacent bone to suggest central osteosarcoma. The prognostic divergence from the malignant morphology of the cytologic specimens demonstrates the importance of pairing diagnostic imaging with cytologic and histologic results when diagnosing bony lesions. 4 The cytologic specimen was likely composed of both osteoblasts, which were reactive to alkaline phosphatase, and mesenchymal cells in the intertrabecular spaces, which were consistent with fibroblasts and had morphologic features suggestive of malignancy such as mitotic figures. Osteochondromas arise from the bony cortex and do not invade underlying bone or cause bony lysis. The radiographs in this case were characteristic for osteochondroma and inconsistent/discordant with the typical radiographic findings for central osteosarcomas, which include variable combinations of proliferation and bony lysis. 8 Radiographic differential diagnoses for this case included osteoma, and parosteal or periosteal osteosarcoma. Parosteal osteosarcomas arise from the outer fibrous layer of the periosteum, and periosteal osteosarcomas arise from the cambium layer of the periosteum; both are types of peripheral osteosarcoma and rarely cause bone lysis. In contrast, a central osteosarcoma arises from the medulla of the bone and causes characteristic bony proliferation and lysis. 7 Osteomas, parosteal osteosarcomas, and osteochondromas originate from the cortex, do not invade underlying bone, and may contain cartilage that undergoes ossification. However, osteomas, parosteal osteosarcomas, and periosteal osteosarcomas are not continuous with the underlying medullary cavity. Osteomas are made up of dense columns of bone and seldom have cartilage. Parosteal osteosarcoma is separate from the periosteum, and periosteal osteosarcoma is contiguous with periosteal fibrous tissue. Therefore, these conditions must be differentiated based on diagnostic imaging and histology.
Key histologic features of an osteochondroma include a cartilage cap that undergoes orderly endochondral ossification forming trabeculae of bone that contain bone marrow and communicate with the underlying normal bone and medullary cavity.3,7 Therefore, adequate sectioning of the sample to provide both orientation and sufficient cross-sectional area are required to identify the key histologic features and avoid misdiagnosis of osteoma, chondroma, osteosarcoma, or bony callus. While there is minimal atypia and mitotic activity and no evidence of invasion, compression of adjacent soft tissues and joints may be present. Adequate sectioning of the neoplasm is also required to search for regions of malignant transformation. Neoplastic transformation of osteochondroma to chondrosarcoma and osteosarcoma is reported in dogs and occurs in approximately 5% to 25% of human cases.1,5 There was no evidence of malignant transformation in this case.
Primary bone tumors in ferrets are rare. Most reports are of osteomas of flat bones and less commonly osteosarcomas of long and flat bones. 9 This, to the authors’ knowledge, is the first report of an osteochondroma in a ferret. This case demonstrates the importance of a multimodal approach of diagnostic imaging, cytology, and histology when diagnosing bony lesions. Finally, osteochondroma should be considered as a differential diagnosis for cytologic specimens of bony lesions that are reactive for alkaline phosphatase. This is one of many bony lesions that contains osteoblasts, which are highlighted with the alkaline phosphatase stain. 2
Footnotes
Disclaimer
The views expressed in this manuscript are those of the authors and do not necessarily reflect the official policy of the Department of Defense or the U.S. Government.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
