Abstract
Animals infected with Mycobacterium avium subspecies paratuberculosis show a variety of granulomatous lesions that range from focal forms, seen in the subclinical stages, to diffuse lesions associated with clinical signs. The aim of this study was to phenotypically characterize the macrophages present in the different lesion types using immunohistochemical methods. Lesions from a total of 23 animals with bovine paratuberculosis, natural and experimental, were examined by immunohistochemistry. Antibodies against inducible nitric oxide synthase (iNOS), tumor necrosis factor α (TNF-α), CD163, interleukin 10 (IL-10), transforming growth factor β (TGF-β), major histocompatibility complex (MHC) class II, natural resistance-associated macrophage protein 1 (Nramp-1), calprotectin, Ki-67, CD68, lysozyme, and ionized calcium-binding adaptor molecule 1 (Iba-1) molecules were employed. Samples were scored semiquantitatively using a complete histological score (H-score), reflecting the staining intensity and the percentage of immunolabeled macrophages. Differences in the H-score were seen depending on the lesion type. In focal lesions, with none or few acid-fast bacilli (AFB), macrophages were polarized toward M1 phenotype, with high H-scores for iNOS and TNF-α. Diffuse multibacillary lesions showed M2 differentiation, with high expression of CD163, IL-10, and TGF-β as well as Nramp-1 and MHC class II antigens. Macrophages in diffuse paucibacillary forms showed high H-scores for iNOS but low ones for TNF-α. Diffuse lesions, either multibacillary or paucibacillary, showed high calprotectin and low Ki-67 expression, suggesting a progressive character, while focal forms, with low H-scores for these antigens, would be consistent with latency. Lysozyme and CD68 expression were related to the amount of AFB. H-score for Iba-1 antibody was similar among all types. The findings of this study provide insights into the polarization status of macrophages and lesion development in bovine paratuberculosis.
Keywords
Paratuberculosis or Johne’s disease is a chronic infection of domestic and wild ruminants caused by Mycobacterium avium subspecies paratuberculosis (Map). It is distributed worldwide and causes important economic losses to domestic livestock associated with chronic diarrhea and progressive loss of body condition. 19 Despite its relevance, the pathogenesis of paratuberculosis is still poorly understood. Although transplacental infection or excretion of Map in the milk or colostrum has been demonstrated, the ingestion of Map-containing feces by young animals is the most important transmission route. 19 As a consequence, infected animals can show different outcomes such as resistance to infection, development of latent infections that can last for an entire lifetime, or clinical disease that will appear only in the adulthood. 19,58
In Map-infected animals, a variety of granulomatous lesions can be detected, which have been classified according to the intensity, location, cellular types, and number of acid-fast bacilli (AFB). 6,18,43 Although slight differences are observed between species, 4 main types of lesions have been recognized, which are associated with different stages of the disease: focal forms, composed of small, well-demarcated granulomas located exclusively in the intestinal lymphoid tissue or lymph nodes, have been related to the initial phases of infection or to latent stages in adult animals. 11,18 Multifocal forms, seen in subclinically infected animals, are characterized by the presence of small granulomas in the lymphoid tissue and also in the lamina propria either related to or not related to the Peyer’s patches; the normal histological architecture of the intestine is not substantially modified. 11,18 Animals with clinical signs usually show diffuse lesions, characterized by a widespread granulomatous enteritis, affecting both the lymphoid tissue and lamina propria causing the thickening of the intestinal wall. 6,18,43 According to the cell types and amount of AFB, they have been classified as multibacillary/histiocytic forms, where epithelioid cells harboring large amounts of AFB are the main component, and as paucibacillary/lymphocytic lesions that are characterized by a diffuse infiltrate of lymphocytes in the lamina propria, with scattered granulomas formed by macrophages and giant cells containing none or low numbers of AFB. 18,43
The factors responsible for the development of the different pathological responses remain unclear. A relation between the immune response developed by the host and the type of lesion has been reported. 44,56 Focal, multifocal, or diffuse paucibacillary lesions have been associated with high peripheral cellular immune responses, while in those animals with multibacillary lesions, a humoral response is predominant. However, in some cases, this polarization of forms is not so clearly defined. 14,56,58
Map infection occurs mainly through M cells of the Peyer’s patches and, to a lesser extent, through differentiated epithelial cells. 49 After penetrating the intestinal epithelial barrier, Map invades the subepithelial macrophages that play a crucial role in the host–pathogen interaction. 49 Granulomatous lesions, composed mainly of macrophages, are the hallmark of paratuberculosis, constituting a complex environment where mycobacteria can inhibit the maturation and acidification of phagosomes and transform the potentially hostile macrophages into protected havens assuring their replication. 49,55 There is still a lack of information regarding macrophage function or diversity within the granuloma in paratuberculosis. In addition, why some granulomas control bacterial growth while others permit it remains unknown. As a reflection of the complexity of these interactions, the existence of different myeloid cell populations that can interact with antigens and participate in the local immune response has been described in the intestine of cattle, with differences related to age or intestinal location. 17
A common feature across different species is that macrophages are characterized by a remarkable diversity and plasticity and can acquire distinct functional phenotypes depending on polarization status. 9,34,35,48 Traditionally, it has been considered that, in response to different signals, macrophages can be differentiated into 2 main types 34,48 : M1 or classically activated which are characterized by a proinflammatory response and promotion of Th1 responses, and M2 or alternatively activated which are considered to have immunoregulatory functions involved in parasite containment and promotion of tissue remodeling. M1 macrophages are mainly activated by interferon γ (IFN-γ), whereas M2 cells are activated by interleukin (IL) 4 or IL-10. They can be distinguished by the differential expression of diverse markers, such as inducible nitric oxide synthase (iNOS) or tumor necrosis factor α (TNF-α) for M1 macrophages or CD163, IL-10, or transforming growth factor β (TGF-β) for M2 macrophages. 3,32,34 Pathology of different diseases has been associated with dynamic changes in macrophage activation. 5,23,25,30,33,48 Most of the information regarding M1 and M2 polarization have been obtained from in vitro studies involving cultured macrophages 23,25,35 that probably do not correlate with the complexity of the immune responses and the environment that take place in the in vivo developed granulomas. 9,55 The study of macrophage polarization associated with lesion types has been assessed through the immunohistochemical labeling of different markers associated with M1 and M2 polarization in different diseases. 3,23,25,33
Concerning mycobacterial infections in human tuberculosis, it has been shown that M1 macrophages are associated with early stages of infection where they promote the formation of tuberculous granulomas and a protective response, while M2 macrophages are more typically associated with an advanced stage of the disease where host responses are less effective in controlling infection. 23,33 In paratuberculosis, a recent study on blood monocytes from subclinically infected cattle has detected a heterogeneous macrophage activation pattern characterized by both classical and alternative phenotypes. 54
The main objective of this study was to carry out the phenotypical characterization of the macrophages present within the different granulomatous lesions associated with Map infection through immunohistochemical labeling of different markers related to macrophage biology. Particularly, the existence of different polarization status among the macrophages in the lesions was also evaluated.
Materials and Methods
Animals
A total of 23 female cattle were used in the study. Eight of them were calves experimentally infected with Map for other purposes. 37 Briefly, 4-month-old calves were orally challenged with a total dose of 6 × 1010 bacilli obtained directly from the intestinal mucosa of a clinically affected cow, which were purified and administered in 6 different doses given every 2 days. Two calves were culled at 120 days postinfection (dpi), and the remaining 6 animals at 270 dpi. Two additional uninfected, control cattle culled at 270 dpi were also examined as negative controls. All the animals were sedated with xylazine (Rompun; Bayer, Mannheim, Germany) and immediately euthanized by an intravenous overdose of embutramide and mebezonium iodide (T61; Intervet, Salamanca, Spain). The remaining 15 animals were naturally Map-infected cows that had been killed in a slaughterhouse by standard authorized methods. They were unvaccinated Holstein cows coming from 2 different herds in whom a follow-up study on losses due to paratuberculosis was ongoing. Infection was confirmed by bacteriological culture of frozen tissues, 18 serum indirect enzyme-linked immunosorbent assay, 14,36 and IFN-γ release test, 14,36 following the methodology detailed in the referred articles. Complete information on the animals and the results of the different tests are provided in Supplemental Table 1. The experimental procedures carried out in this study were performed in accordance with Spanish Royal Decree 1201/2005 for the protection of animals used for experimental and other scientific purposes and were approved by the “Instituto de Ganadería de Montaña CSIC-ULE” Animal Ethics Committee (Ref LE-B-12).
Tissue Samples
After necropsy or at the slaughterhouse, at least one sample from different areas of the intestine (ileocecal valve; distal, middle, or proximal ileum; and jejunum [with and without Peyer’s patches]; duodenum; and large intestine—including cecum, colon, and rectum) and ileocecal, ileal, and jejunal mesenteric lymph nodes was taken from each animal for histopathological examination. All samples were fixed in 10% buffered formalin for less than 48 hours and subsequently dehydrated through a graded alcohol series before being embedded in paraffin wax. Sections, 3.5 µm thick, were stained with hematoxylin and eosin and Ziehl-Neelsen method for AFB identification. A subjective classification of the sections was made according to the amount of AFB present, from 0 (no detectable bacilli), 1 (scant AFB in the cytoplasm of macrophages), 2 (moderate, easily detectable bacilli), to 3 (high load of AFB). Detailed information on the lesion type and AFB category present in each examined sample are provided in Supplemental Table 1. Granulomatous lesions were detected in different small intestine and lymph node samples. Only in 1 case, granulomas were seen in the cecum (Supplemental Table 1). Control cattle did not show any lesion consistent with Map infection.
Classification of Lesions
Lesions associated with Map infection were classified into 4 categories, according to the guidelines provided by González et al 18 : focal, multifocal, diffuse multibacillary, and diffuse paucibacillary. Each animal was classified based on the most severe lesion found in the examined samples, bearing in mind that the type of lesion could vary among the tissue samples obtained from the same individual (Supplemental Table 1).
Focal lesions were characterized by small, well-demarcated granulomas composed of 5–50 macrophages located exclusively in the interfollicular areas of the intestinal lymphoid tissue of the ileocecal valve or the isolated jejunal Peyer’s patches. They were also detected in the paracortical area of the lymph nodes. Acid-fast bacilli were absent or detected in very low amounts in the cytoplasm of the macrophages (category 0 or 1).
Three experimentally infected calves and 2 naturally infected cows were classified as having focal lesions. In these animals, intestinal sections from 2 locations (ileocecal valve and jejunal Peyer’s patches) presented this type of lesion (Supplemental Table 1).
Sections containing multifocal lesions presented focal granulomas in the lymphoid tissue, as well as small and well-demarcated granulomas, formed by 5–25 macrophages and giant cells in the intestinal lamina propria related to the Peyer’s patches or in the lamina propria of villi not related to the lymphoid tissue. Granulomas were scanty and did not substantially modify the normal intestinal mucosa architecture. As in focal lesions, AFB were not detected or present in very low amounts (score 0 or 1).
A total of 6 animals (3 experimentally challenged calves and 3 adult cows) were classified under this category. In this group, this lesion was detected in 24 tissue sections of the intestine (Supplemental Table 1).
Diffuse lesions were characterized by the presence of a widespread granulomatous lymphadenitis and enteritis that altered significantly the normal intestinal architecture. The lesion was present in areas of the intestine both with and without lymphoid tissue. Large numbers of macrophages and occasional giant cells appeared in the interfollicular areas, infiltrating the lymphoid follicles. The lamina propria was markedly thickened and distended due to the presence of inflammatory cells. According to the cellular types and number of AFB, 2 types of diffuse forms were considered: diffuse multibacillary lesions, characterized by the predominance of macrophages morphologically consistent with epithelioid cells harboring large numbers of AFB (category 2 or more frequently 3) and diffuse paucibacillary lesions, wherein the infiltrate was composed mainly of lymphocytes, with some macrophages and giant cells, with occasional AFB (score 1) located among them.
Diffuse multibacillary lesions were identified in six animals (1 experimentally infected and 5 naturally infected) and diffuse paucibacillary forms in an other six (5 naturally and 1 experimentally infected cattle). Concerning tissue samples, diffuse multibacillary forms were identified in 30 intestine sections and diffuse paucibacillary forms in 29 samples (Supplemental Table 1). In all 12 cattle with diffuse lesions, grossly visible thickening of the intestinal wall of the ileum and jejunum and enlargement of the regional lymph nodes were seen in different degrees.
Immunohistochemistry
Immunohistochemical studies were performed in a total of 23 intestinal sections from different animals (Supplemental Table 1), showing the 4 main types of lesions considered (6 different samples from each lesion, except for the focal lesions where 5 samples were examined). Focal lesions were evaluated in sections obtained from the jejunal Peyer’s patches, whereas multifocal, diffuse multibacillary, and diffuse paucibacillary lesions were evaluated in tissue sections obtained from the middle ileum.
Different primary antibodies raised against antigens expressed by macrophages, including M1 and M2 subpopulations, and proliferation markers were used (Supplemental Table 2). Immunohistochemistry was performed using the Envision+ System (Dako, Agilent Technologies, Glostrup, Denmark) in 3.5-μm-thick tissue sections that were placed onto poly-
All the antibodies were tested in the 23 selected intestinal sections. A single antigen was examined per section. From each intestinal sample, serial sections were used for the sequential immunostaining of the antigens.
Evaluation of Immunostains
Some of the antibodies used (mainly those against TNF-α, IL-10, major histocompatibility complex [MHC] class II, and Ki-67 antigens) immunostained other cells apart from macrophages (mainly lymphocytes). Moreover, macrophages unrelated to the granulomas and present in the intestinal lamina propria were also positively immunolabeled with some of the antibodies. For this study, only those immunolabeled cells with a clear macrophage morphology (abundant cytoplasm and ovoid nucleus) that were forming part of the granulomas present in the different lesion types were considered and evaluated.
For staining comparison among tissues and types of lesion, samples were scored semiquantitatively using a complete H-score that considers both the staining intensity and the percentage of macrophages positively immunolabeled. 12 A total of 6 different slides (5 in the case of focal lesions) were evaluated for each type of lesion. In each slide, 5 randomly selected fields containing granulomatous lesions were selected and photographed at 400×. In each individual sample, granulomas were evaluated regardless of their location in the lamina propria or lymphoid tissue (for multifocal and diffuse lesions). Cell counting and image analysis were performed using the Image J processing and analysis software (US National Institutes of Health, Bethesda, Maryland).
For each type of lesion, a complete H-score was calculated by adding the products of the percentage of cells (0-100) labeled at a given staining intensity present in each selected field and the staining intensity score (0, none; 1, weak; 2, moderate; and 3, intense). The H-score obtained in each random field can be placed in a range of values between 0 and 300.
The final H-score for each antibody and type of lesion was obtained considering the average value for the 5 areas counted and the 6 (or 5 for focal forms) sections examined.
Evaluation of macrophage immunostaining was performed independently by 2 pathologists (M.F., V.P.), and discordant results were reviewed with a multiheaded microscope to reach consensus.
Statistical Analysis
Immunohistochemical data (H-score) were analyzed for normality of distribution using Shapiro-Wilk and Kolmogorov-Smirnov tests. As data were not normally distributed and logarithmic transformation failed to correct skewness, they were analyzed with nonparametric methods of statistical analysis. The Kruskal-Wallis test was used to compare groups of lesions (focal, multifocal, diffuse multibacillary, and diffuse paucibacillary) for each antibody. The Mann-Whitney test was applied for post hoc comparison when significant differences were observed. A P ≤ .05 was considered to indicate statistical significance. Correlation between AFB category and H-scores for the different antibodies was evaluated using Spearman test. All statistical analyses were performed with the use of statistical software SPSS version 21.0 (IBM Corp., Armonk, New York).
Results
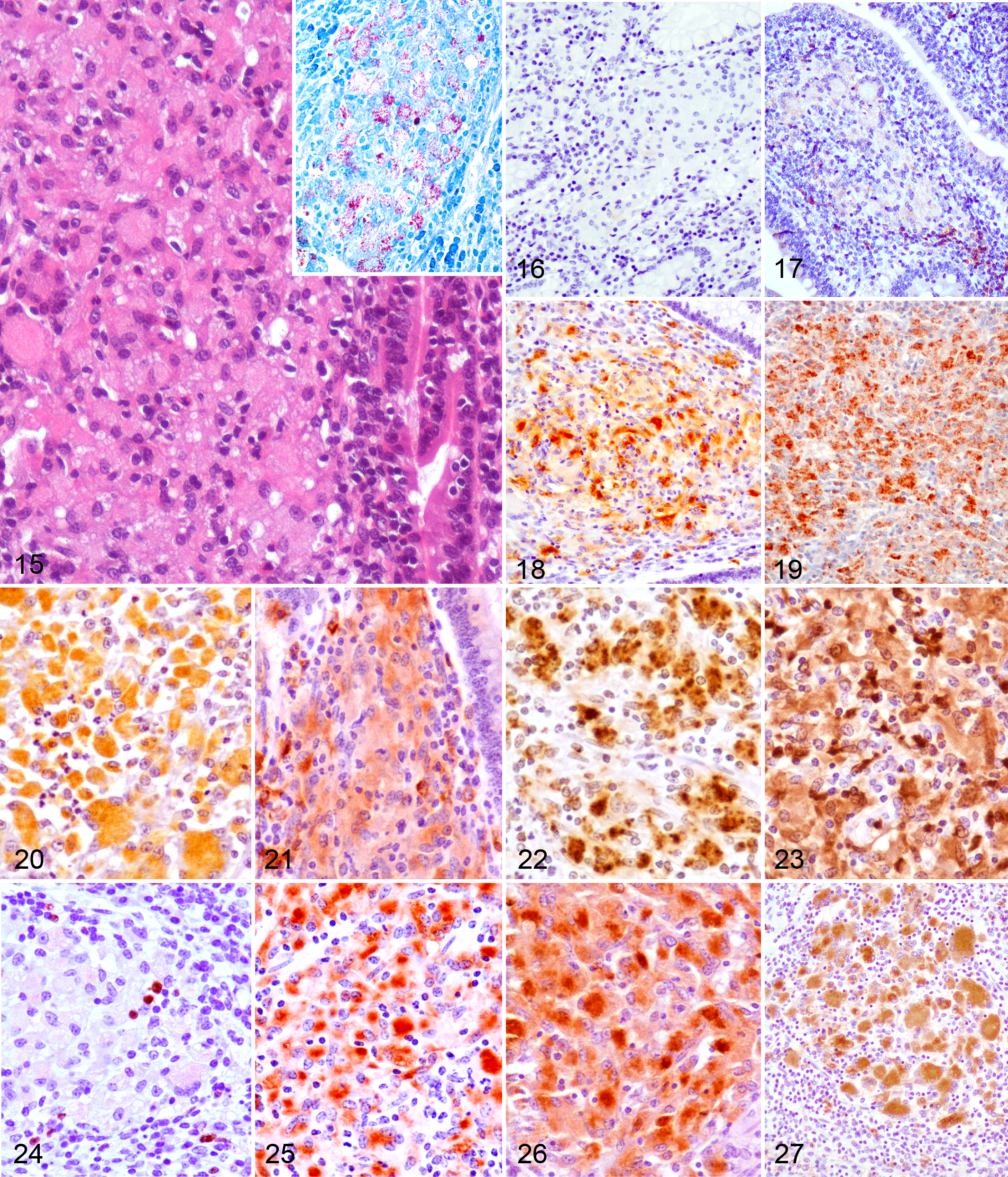
Macrophages present in the 4 types of lesions considered were immunolabeled with the 12 tested antibodies showing differences in the intensity of the signal and the number of immunolabeled cells. Figure 1 shows the mean of the H-score obtained for each antibody according to the lesion category. Images corresponding to the patterns of immunostaining for the different antibodies and the main types of lesion are shown in Figs. 2-14 (focal), Figs. 15-27 (diffuse multibacillary), and Supplemental Figs. 1-13 (diffuse paucibacillary).
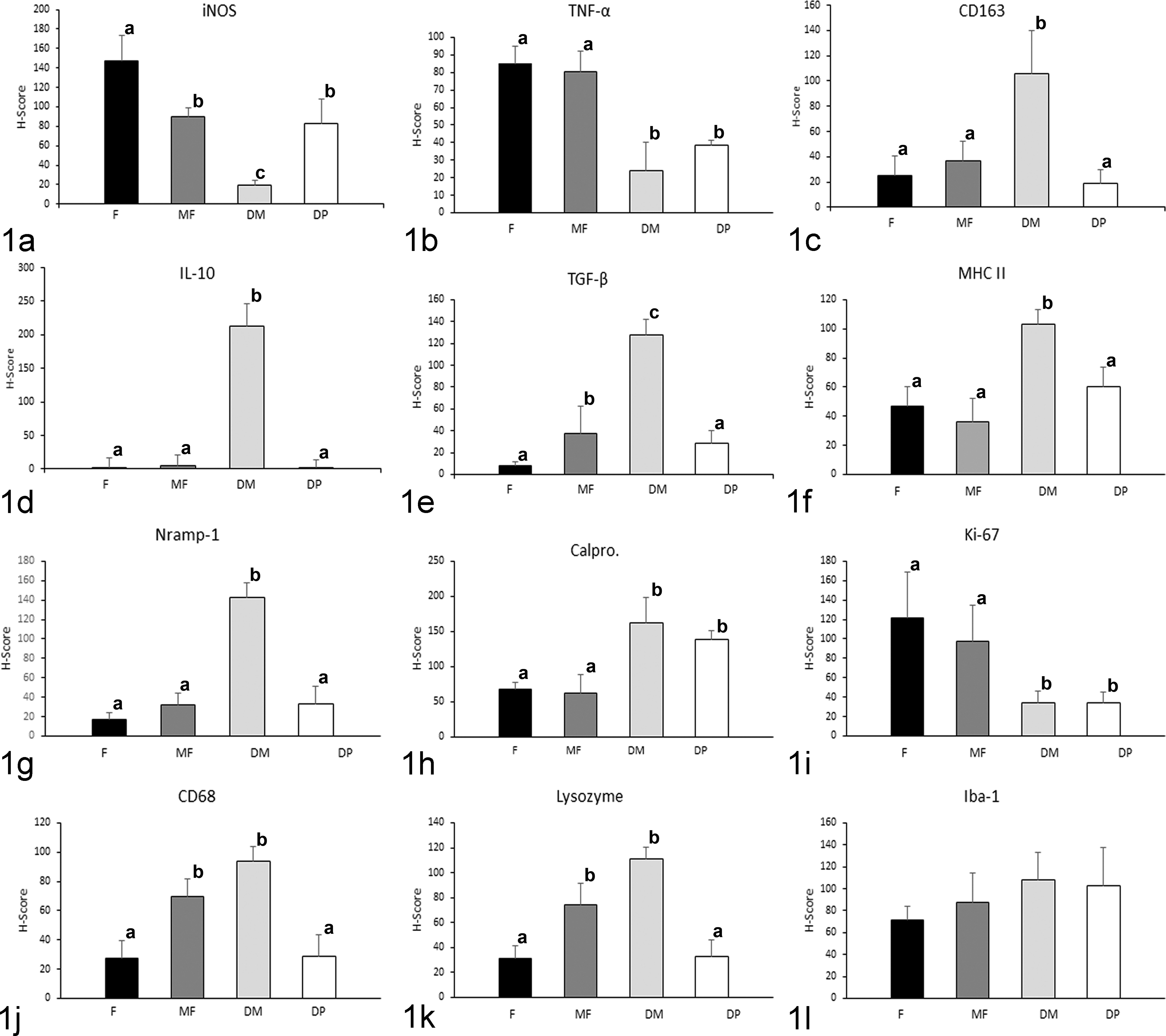
Mean histological score (H-score) for immunohistochemical staining according to the different lesion categories for (a) inducible oxide synthase (iNOS), (b) tumor necrosis factor-α (TNF-α), (c) CD163, (d) interleukin-10 (IL-10), (e) transforming growth factor-β (TGF-β), (f) MHC class II, (g) natural resistance macrophage protein 1 (Nramp-1), (h) calprotectin, (i) Ki-67, (j) CD68, (k) lysozyme, and (l) ionized calcium binding adaptor molecule 1 (Iba-1) antibodies. DM indicates diffuse multibacillary; DP, diffuse paucibacillary; F, focal; MF, multifocal. Different superscript letters following the values indicate statistical significance (P < .05). Error bars indicate standard error of the mean.
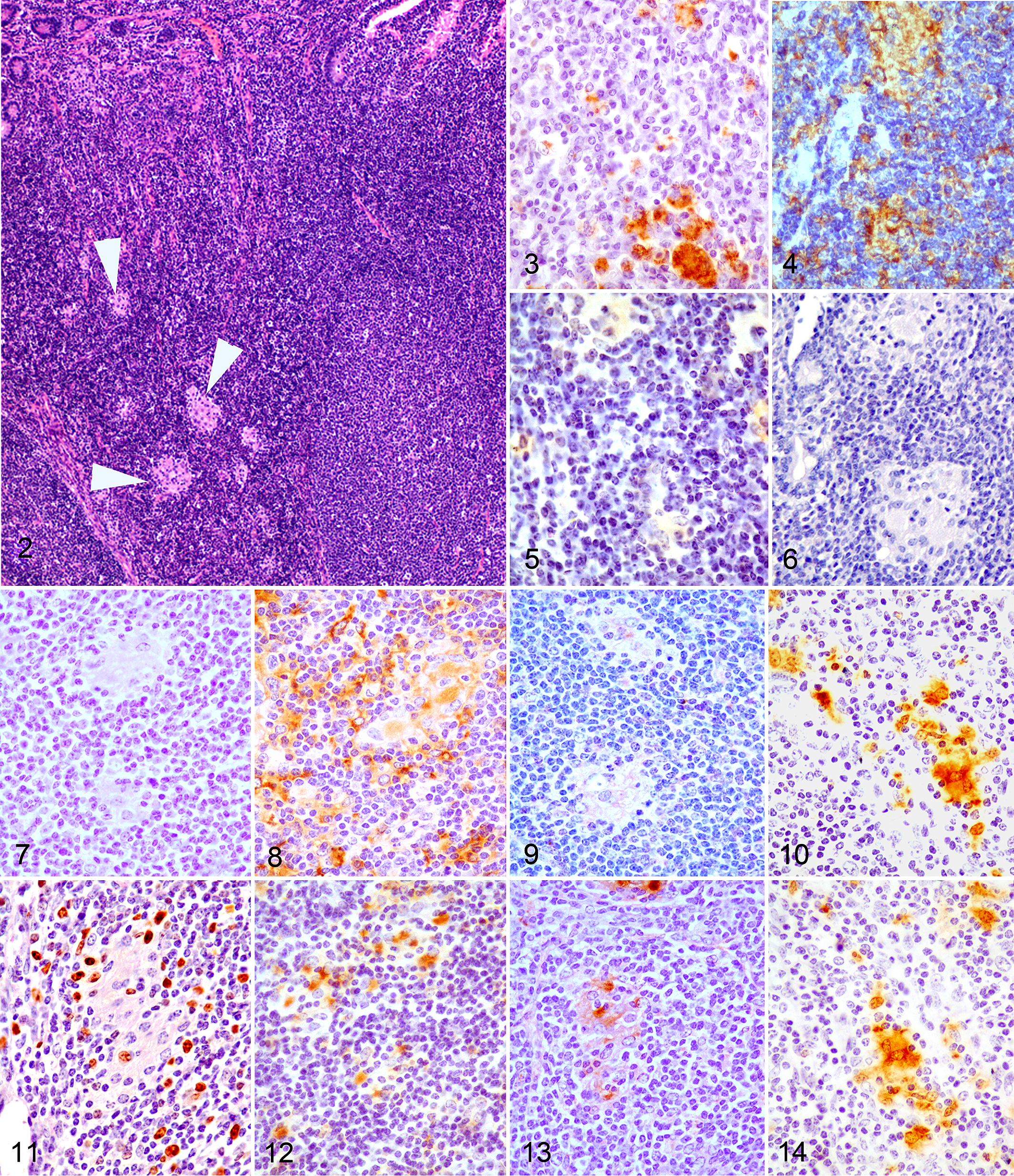
Histological score values for iNOS were significantly higher in focal lesions than in diffuse multibacillary (P < .001) or multifocal and diffuse paucibacillary forms (P < .05). No differences were observed between the last 2, both of them showing higher values than diffuse multibacillary forms. The lowest H-score values for this marker were found in diffuse multibacillary lesions. Expression of TNF-α was higher in focal and multifocal forms (P < .05) than in diffuse multibacillary or paucibacillary lesions, showing no differences between them.
The highest H-score values for CD163 and IL-10 antibodies were obtained in diffuse multibacillary lesions, with significant differences compared to the rest of the lesion types (P < .001). High H-scores were also obtained for TGF-β immunolabeling, although in this case differences were also observed between focal and multifocal and diffuse paucibacillary type lesions (P < .05).
Immunohistochemical expression of MHC class II antigen, associated with antigen-presenting cells, was significantly higher in diffuse multibacillary lesions than in the rest of the forms (P < .001), where significant differences were not observed. Expression of natural resistance–associated macrophage protein 1 (Nramp-1) was higher in diffuse multibacillary lesions than in the rest of forms (P < .001) exhibiting similar H-score values.
The intensity of immunostaining for calprotectin antibody was significantly lower in focal and multifocal forms than in diffuse lesions (P < .001), with no differences between both diffuse types. Histological scores for immunodetection of Ki-67 antigen, which marks nuclear proteins expressed in cells that are proliferating, were significantly lower in both types of diffuse lesions, that is, focal and multifocal forms (P < .001) that rendered the highest values.
CD68 and lysozyme antibody immunolabeling rated the highest H-score values in multifocal and diffuse multibacillary lesions in relation to focal and diffuse paucibacillary forms (P < .05) that showed similar levels. For ionized calcium-binding adaptor molecule 1 (Iba-1) antibody, no significant differences in the H-score values were observed among the different types of lesions.
Antibody Immunolabeling According to the Type of Lesion
Focal lesions showed high H-score values for iNOS, TNF-α, and Ki-67 antibodies, as observed in Figs. 1 and 2. A similar pattern was observed in multifocal lesions except for the immunolabeling of TGF-β, CD68, and lysozyme, which was higher. Diffuse multibacillary lesions showed high H-score values for CD163, IL-10, TGF-β, lysozyme, CD68, calprotectin, Nramp-1, and MHC class II antibodies, whereas iNOS and TNF-α immunolabeling was significantly lower. Diffuse paucibacillary lesion immunolabeling profiles were also similar to those of focal forms, except for the immunolabeling of TNF-α, calprotectin, and Ki-67 antibodies which showed a pattern similar to that presented by the diffuse multibacillary forms. High H-score values were observed for Iba-1 antibody in the 4 types of lesions, with no differences between them.
Correlation Between AFB Category and H-Scores for the Different Antibodies
The amount of AFB present in the lesions was directly correlated with the H-score for the different antibodies used, with a variable statistical significance (between P < .05 and P < .0001) depending on the marker used (Supplemental Fig. 14). Histological scores for iNOS, TNF-α, and Ki-67 were negatively correlated with the amount of AFB, whereas a positive correlation was confirmed with the H-scores for the rest of the markers.
Discussion
As a consequence of Map infection, animals can develop morphologically distinct granulomatous lesions, from focal forms seen in the subclinical phase of the disease to diffuse lesions usually related to clinical signs. 6,11,18 This study has shown that macrophages present in the granulomatous infiltrates show different immunohistochemical expression of the markers examined, as seen by the differences in the H-scores for several of these markers, depending on the lesion type. These differences can probably play different roles in their development. Considering this, the semiquantitative method used (H-score) has proven to be a useful tool to evaluate immunostaining, also enabling statistical comparison between these antibodies. Since it was initially proposed for the quantification of hormone receptors in neoplastic cells, 12 the H-score has been proven to be useful for the immunophenotypic characterization of different cell populations. 8 However, this method has evident disadvantages such as only permitting a semiquantitative evaluation of biomarker production that cannot completely correlate with the real expression and the difficulty in assessing the immunostaining when it is not present in a single cell population. 13
Depending on the polarization status, 2 different phenotypes of macrophages, named M1 and M2, have been recognized. Briefly, M1 or classically activated macrophages are involved in the response against intracellular pathogens throughout the secretion of proinflammatory cytokines and iNOS, whereas M2 or alternatively activated macrophages have immunoregulatory functions and play a role in tissue remodeling. 25,32,34,48 Several of the molecules whose expression has been evaluated in this study are considered to be M1 (iNOS, TNF-α) or M2 (CD163, IL-10, or TGF-β) markers and have shown differential expression. The results of this study suggest that after Map infection, there is a modulation in the macrophage polarization toward proinflammatory and anti-inflammatory forms in the different types of lesions in such a way that in focal lesions, macrophages express markers related to M1 status, whereas in diffuse multibacillary lesions, M2 macrophages would predominate. Similar findings have been reported in Mycobacterium tuberculosis infection. 23,33 In a recent study on paratuberculosis, a heterogeneous macrophage activation pattern was observed, characterized by both classical and alternative phenotypes 54 ; although in the same study, it was found that in animals with low peripheral IFN-γ responses, the proportion of monocytes expressing CD163 (an M2 marker) was higher, while they showed the lowest expression of iNOS (an M1 marker), in accordance with our results. In any case, that study was conducted in blood monocytes from subclinically affected calves, where the lesions were not determined.
Macrophages in focal lesions that have been related to the subclinical stages of paratuberculosis, either to initial phases of Map infection or to latent stages, 11,18,43 show an M1 phenotype associated with the absence or low numbers of AFB and the production of high levels of iNOS and TNF-α, a proinflammatory cytokine. 32,34 A similar finding has been observed in the initial granulomas in M. bovis infection 40,42 also with very low numbers or absence of bacilli and with a similar histological morphology of lesions. Moreover, IFN-γ and TNF-α are the main stimuli that will drive macrophages to an M1 phenotype. 32,34 In this sense, focal lesions are associated with high levels of plasma IFN-γ production after lymphocyte stimulation. 14,45,56 Tumor necrosis factor α has been shown previously to be produced by macrophages after infection with mycobacteria and it is considered, together with IFN-γ, as an endogenous cofactor in the induction of mycobacterial growth inhibition. 15 Inducible nitric oxide synthase induces the production of nitric oxide (NO) that has reported to play a role in mycobacterial infections by supporting killing mechanisms within macrophages 22,40 and has been related to the subclinical phases of paratuberculosis. 27 The expression of MHC class II, a molecule that plays a key role in antigen presentation, has also been previously observed in M1-polarized macrophages 5,32 and in granulomas found in the initial phases of paratuberculosis in goats, with weaker intensity of labeling in those macrophages harboring low amounts of mycobacteria, 29,55 as in this study. In this sense, in vitro studies have revealed that, shortly after mycobacterial infection, monocytes downregulate MHC class II expression. 57 Natural resistance–associated macrophage protein 1 has been linked to innate resistance to intracellular pathogens 4 by reducing the susceptibility to phagosome rupture in macrophages infected with M. tuberculosis. 50 Since it is almost undetected in nonactivated macrophages, 10 its presence in focal lesions could be linked to their ability to control Map multiplication, suggesting an activated state of macrophages in these lesions. All these findings suggest that focal lesions could be the reflection of highly protective stages that could last long periods of time (latency), where subclinically infected animals can control the growth of Map inside the M1-polarized macrophages, without developing a severe inflammatory response related to clinical signs.
Macrophages in diffuse multibacillary lesions exhibit an M2 profile, as seen by the high expression of CD163, IL-10, and TGF-β markers and the low levels of iNOS and TNF-α. CD163 has been widely considered an M2 marker, 3,23,48 but some studies have reported that it is not totally specific, and macrophage characterization should be done in combination with other markers. 3,25 Following these suggestions, we explored CD163, IL-10, and TGF-β immunostaining, whose high expression has been recognized to be linked to an M2 phenotype, 32,34 more precisely to the M2c category. In contrast to M2a macrophages, associated with allergy and killing and encapsulation of parasites, 32 or M2b macrophages, related to a Th2 activation induced by IL-1, 32 M2c macrophages induced by the presence of IL-10 and TGF-β 32 show a high immunoregulatory and anti-inflammatory activity 32,34 and predominate in the late stages of tuberculosis infection, posing a risk of exacerbating the disease. 30 These mechanisms are probably operating in Map infection, since diffuse multibacillary lesions are detected in the late stages of the disease and are characterized by harboring large amounts of AFB, low numbers of other inflammatory cells rather than macrophages, 6,18 and low expression of iNOS 22 as in our case. These immunoregulatory actions are related to the production of TGF-β and IL-10 by macrophages that have been demonstrated to play a role in the downregulation of NO production. 27,35 This scenario is consistent with the low levels of iNOS and TNF-α (a well-known proinflammatory cytokine) expression found in this type of lesions opposed to those found in focal forms. In paratuberculosis infection, upregulation of TGF-β and IL-10 has been reported in clinical cases during the late stages of the disease, 1,27 which are highly expressed in macrophages harboring high numbers of Map, 36,53 in agreement with our results. Expression of MHC class II was significantly higher in diffuse multibacillary lesions compared to the rest of the lesion types. In contrast to this finding, Navarro et al 38 did not find immunohistochemical expression of this marker in the cytoplasm of epithelioid cells associated with diffuse lesions and harboring AFB in goats. However, in agreement with the results of this study, Lybeck et al 31 and Krüger et al 29 have also reported positive immunolabeling of MHC class II in macrophages from diffuse lesions and showing AFB in their cytoplasm, also in goats and using antibodies different to the one used in this work. A high level of Nramp-1 expression was also observed in these lesions, in the presence of abundant AFB, as it was noted previously by Delgado et al 10 in paratuberculosis or by Pereira-Suárez et al 42 for M. bovis infection. For these authors, the expression was interpreted as a reflection of mycobacterial infection, since they only examined lesions with remarkable numbers of AFB. In our study, the differential expression of Nramp-1 is also strongly related to the high numbers of AFB as seen in the multibacillary forms. Since this protein is located on the membrane of the macrophage-containing phagosomes, 16 its higher level of expression would be linked to the higher number of these structures present in these cells as a consequence of harboring AFB. Its action in mycobacterial infections would be both to reduce phagosomal rupture 50 and to inhibit the capacity of mycobacteria to arrest phagosome maturation, 16 hampering bacterial growth. In diffuse multibacillary lesions, however, these mechanisms would not be efficiently activated, despite high level of Nramp-1 expression, permitting Map intracellular growth and whose nature deserves further investigation.
In comparison with diffuse multibacillary lesions, macrophages in paucibacillary lesions would be mostly polarized toward an M1 type, as shown by high H-scores for iNOS and low scores for CD163, IL-10, and TGF-β. 32,34 However, paucibacillary granulomas showed lower level of TNF-α expression compared to focal lesions. Similar findings have been reported previously, 51,53 and these are probably related to the severe T-lymphocyte infiltration observed in paucibacillary lesions, 38,51 which are not reported in focal forms. Supporting this, in human patients with Crohn’s disease, a disorder in which Map has been proposed to play an etiological role, and where lesions similar to the diffuse paucibacillary forms reported in this study have been observed, 20 a downregulation of TNF-α has been found. 7 It has already been pointed out that severe lesions of paratuberculosis are related to low TNF-α expression regardless of the amount of mycobacteria. 1 Further research is necessary to elucidate the mechanisms underlying these findings.
An interesting finding of this study is the difference between focal and diffuse lesions in the expression levels of calprotectin. Calprotectin is a major cytosolic protein complex present in monocytes that is expressed in tissue macrophages recently recruited from peripheral blood but whose expression is lost upon further differentiation. 52 This fact together with the differences observed in the H-score for Ki-67 protein—a cellular marker for cell proliferation 47 —leads to the hypothesis that a high number of macrophages forming granulomas in diffuse lesions would have been recently recruited from blood, while in focal forms, these would be multiplying in situ, supporting the latent character of this type of lesion and the active and progressive character of diffuse forms, seen in the advanced and uncontrolled cases of paratuberculosis. 6,18 Supporting this, Valheim et al 55 also found a high number of macrophages expressing Ki-67 in granulomas from subclinically paratuberculosis-affected goats, and high levels of calprotectin in plasma have been associated with severe and active tuberculosis in humans. 41 Opposed to this hypothesis, Jenkins et al 24 have pointed out that local macrophage proliferation is a signature of a Th2 inflammation. However, their study was conducted in pleural macrophages from rodents infected with a nematode, while Map is an intracellular pathogen affecting the intestine. Probably these facts together with the different methods used for macrophage evaluation in both studies could explain the different results. In any case, this hypothesis should be carefully considered, since lesions in paratuberculosis can show a dynamic character, 28 as seen in this study by the coexistence of multifocal and diffuse lesions in the same animal and that in this work only one single time point has been evaluated.
Multifocal lesions, with a majority of M1-polarized macrophages showing high H-scores for iNOS and TNF-α, 32,34 could represent transient lesions between focal and diffuse forms. This is supported by the simultaneous higher expression of TGF-β and lower expression of calprotectin and Ki-67 compared to focal lesions. Thus, although there is a reduced Map multiplication and low numbers of new macrophages recruited from the blood, the increased expression of TGF-β could indicate that they are starting to progress to more severe and uncontrolled lesions. 27,36
CD68 and lysozyme have been widely used as general macrophage markers, 2,29,43,46,55 which is in agreement with our findings, since they have been found in all the lesion types. However, their expression is shown to be positively correlated with the number of AFB, with the highest H-score levels observed in diffuse multibacillary lesions, as previously reported in paratuberculosis. 2,22 The fact that CD68 antigen is located in lysosomal membranes, especially in phagolysosomes, and that lysozyme is also produced in the lysosomes 26 would explain why its expression is increased when the number of intracellular bacteria is high. 21,46 A marked expression of Iba-1 has been found in all the lesion types with no differences in the H-score between them. This is a marker for a calcium-binding protein that has been considered to be specific for macrophages/microglia. 39 According to our results, it may be used as a general macrophage marker in granulomatous lesions.
According to the results of this study, the macrophages composing the granulomas associated with Map infection show differences in the expression of several proteins, reflecting changes in their functionality based on the type of lesion. M1-type macrophages will predominate in focal and multifocal forms, suggesting their latent character and the ability to control Map infection, and partially in diffuse paucibacillary lesions. M2 are the main type in diffuse multibacillary forms, with an immunoregulatory profile that would permit intracellular Map growth. The immunohistochemical analysis of macrophage subsets within Map infection–associated lesions has contributed to increased knowledge on the pathogenesis of this disease.
Footnotes
Acknowledgements
We acknowledge the excellent technical assistance of Gloria Belver, Carmen Espiniella, and María Jesús González. We also thank F. Javier Giráldez for his help with the statistical analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflict of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was financially supported by grant AGL2012-39818-C02-01 of the Spanish Ministry of Economy and Competitiveness. M. Fernández and P. Castaño are the recipients of a predoctoral contract from the “Junta de Castilla y León,” partially funded by the European Social Found (European Union). J. Benavides and M. Royo are supported by a “Ramón y Cajal” and a “Predoctoral” contract of the Spanish Ministry of Economy and Competitiveness, respectively.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
