Abstract
In the summer of 2023, ingestion of Astylus atromaculatus (pollen beetle) was linked to spontaneous fatal disease in grazing cattle and sheep in Argentina and Uruguay. While the disease was experimentally reproduced in sheep and guinea pigs in the 1970’s, no experimental reproductions have been attempted in cattle, and controversy exists as to whether this insect is indeed noxious to cattle and at which dose. Here, we demonstrate that A. atromaculatus causes acute fatal disease in Hereford calves at single oral dosages of 2.5, 4.5, 10.0, and 15.0 g of insect/kg body weight. Death or severe disease necessitating euthanasia occurred at 38 to 48 hours postinoculation regardless of the dose, suggesting that the single fatal dosage is likely <2.5 g/kg body weight (this dose representing approximately 850 mL of intact beetles in a 100 kg calf). Clinically, the disease was characterized by acute anorexia, prolonged recumbency, reluctance to move, listlessness/apathy, depression, ruminal hypomotility and tympany, hypothermia, bruxism with frothing at the mouth, and mucoid diarrhea progressing to death. Hematologic and biochemical alterations included hemoconcentration, stress/acute inflammatory leukogram, negative energy balance, and ketosis. The pathological hallmark of this experimental disease is acute necrotizing omaso-reticulo-rumenitis, fibrinohemorrhagic enteritis, and exfoliative colitis with intralesional chitinous insect fragments. While A. atromaculatus might contain a gastrointestinal toxin or pathogen, extensive toxicological testing failed to identify a causative toxin. Other pathomechanisms such as direct physical damage caused by insect fragments on the alimentary tract seem plausible, although further studies are needed to elucidate the pathogenesis of A. atromaculatus-associated disease.
Astylus atromaculatus (phylum Arthropoda, order Coleoptera, family Melyridae), colloquially referred to as pollen beetle, spotted maize beetle, astylus beetle, siete de oro, astilo moteado (Spanish), and bontmieliekewer (Afrikaans), is an insect native to South America that was introduced to South Africa, presumably in the 1910’s. Its life cycle is annual with the adult stage occurring in the summer (January–March in the Southern Hemisphere). Adults have a lifespan of several months, feed on pollen and nectar of a wide variety of plants, and are important pollinators. 22 After mating, the females lay clusters of orange-colored eggs under dry leaves or grass. Once the larvae hatch, approximately 2 weeks later, they live underground and feed on decayed vegetable matter; however, they can also feed on germinating seeds, causing damage during establishment of cultivated crops. The larvae mature within 6 months and then hibernate in the soil. After a pupal period of approximately 2 weeks, the adults emerge and the cycle is reinitiated.3,26
The pollen beetle has been suspected to cause spontaneous outbreaks of intoxication in ruminants. In January 1970 in South Africa, 20 of 24 cows grazing on sunflowers developed diarrhea (83% morbidity) and 4 of the affected cows died (17% mortality, 20% lethality). During the outbreak, A. atromaculatus beetles were seen clustering on the sunflower heads. Based on the presumptive toxicity of the insect, the authors conducted experimental inoculations in sheep and guinea pigs and concluded that A. atromaculatus was lethal to both species. In sheep, the disease was characterized clinically by listlessness, anorexia, ruminal stasis with mild bloating, and mucoid diarrhea, and pathologically by catarrhal to pseudomembranous enteritis and typhlitis. In guinea pigs, anorexia and listlessness followed by death were the main clinical findings, while pathological examinations revealed acute hemorrhagic enteritis and typhlitis. 16
Given some parallelism between A. atromaculatus and the so-called blister beetles of the family Meloidae (Epicauta spp., Mylabris spp.), which are toxic due to the production of the terpenoid cantharidin, 15 and the somewhat overlapping clinical signs and gastrointestinal lesions caused by blister beetles and A. atromaculatus, Kellerman et al. hypothesized that pollen beetles might contain cantharidin. However, cantharidin was not detected in A. atromaculatus by toxicological testing, leading the authors to conclude that the toxic principle remained to be identified. 16 Unfortunately, no additional experimental studies aiming at identifying a putative toxic principle in A. atromaculatus or reproducing the disease have been published in the peer-reviewed scientific literature since the initial work by Kellerman et al. 16 in South Africa.
In January 2023, an unusually large population of A. atromaculatus beetles was reported in Argentina and Uruguay. Heavy infestations were documented in several weeds (Carduus acanthoides, Cirsium vulgare, and Amaranthus spp.) and crops (sorghum, maize, soybean, sunflower, grapevines, and common fig), including alfalfa (Medicago sativa) and other pastures used to feed ruminants (Fig. 1a–d). Outbreaks of mortality in beef cattle, sheep, and presumably horses in both countries were suspected to have been caused by ingestion of A. atromaculatus colonizing alfalfa. Consequently, warnings were issued to alert farmers and veterinarians about these episodes by the National Institute of Agricultural Technology in Argentina (https://www.argentina.gob.ar/noticias/siete-de-oro-que-es-y-que-hacer-con-el-ganado), and the Ministry of Livestock, Agriculture and Fisheries (https://www.gub.uy/ministerio-ganaderia-agricultura-pesca/comunicacion/noticias/escarabajo-siete-oro-uniradd-alerta-mortandad-rumiantes) and the National Institute of Agricultural Research (http://www.inia.uy/Paginas/siete-de-oro.aspx) in Uruguay.

(a) Adult Astylus atromaculatus forming clusters in intact alfalfa plants or (b) in the stems of alfalfa plants that had been recently defoliated by grazing livestock on a farm in Colonia, Uruguay. (c) Dorsal view of an adult A. atromaculatus. Note the yellow elytra with symmetrical black spots and setae (“hairs”) in the legs, head, thorax, and abdomen. (d) Frontal view of the specimen shown in (c) depicting the head (including the eyes, antennae, and buccal apparatus), thorax, and first pair of legs.
In these outbreaks, the suspicions were based on the presence of abundant pollen beetles in the forages being fed to the animals, the clinical signs (anorexia, lethargy, and diarrhea), and postmortem examinations of cattle and sheep, which revealed fibrinosuppurative and necrotizing gastrointestinal lesions.13,19 Ingestion was confirmed in all cases by finding numerous fragments of A. atromaculatus in the ruminal contents. However, controversy exists in the veterinary and scientific community as to whether the pollen beetle is indeed noxious to cattle and responsible for the reported spontaneous outbreaks of mortality in this species, and if so, what is the minimal quantity of beetles that cattle must ingest to develop the disease. The situation is further obscured by the fact that the clinical signs and lesions described in the affected animals are not exclusive of A. atromaculatus-associated disease and can be present in other conditions, including infectious and toxic diseases.24,25,31
Therefore, the aims of this study were to assess the clinical and pathological effects of single oral administration of different doses of A. atromaculatus in cattle and search for putative toxic compounds in this insect species. This information is of value to clinicians, pathologists, and researchers working in the fields of toxicology, entomology, and animal nutrition.
Materials and Methods
Two experiments (1 and 2) were conducted to assess the clinical and pathological effects of single oral administration of A. atromaculatus in calves. All procedures involving live calves, including housing, inoculation, clinical examinations, sampling, and euthanasia, were reviewed and approved by National Institute of Agricultural Research’s committee for the use of animals in experimentation (CEUA, protocol #2023.1).
Experiment 1
Astylus atromaculatus collection, isolation, and determination of the size, weight, volume, and % of dry matter
For this experiment, insects were collected from 2 rural properties in the department of Colonia, Uruguay, from a commercial plantation of grapevines and a nearby alfalfa pasture (property 1, February 10) and a backyard fig tree in a dairy farm (property 2, February 13–15) that were heavily colonized by A. atromaculatus adults. The insects were collected mostly by hand from the grapevines and fig tree and using entomological nets from the alfalfa pasture.
The collected material was placed into plastic containers and transported to the laboratory where live adult A. atromaculatus were isolated by separation from other arthropods and plant particles and subjected to morphologic entomological identification. 16 Approximately 1.2 and 1.8 kg of A. atromaculatus were obtained from properties 1 and 2, respectively. Isolated A. atromaculatus obtained from both properties were pooled and kept alive under refrigeration (4–6°C) until preparation of the inoculum.
The length from the distal end of the abdomen to the tip of the head and the width at the widest point of the abdomen of 20 individuals (10 males and 10 females determined by observation of copulating pairs) was obtained with a hand caliper. In addition, the average fresh weight of 50 live adult individuals was obtained with a calibrated precision digital scale (Pioneer PA 214; Ohaus, New Jersey, USA), and the volume of 250 g of adult A. atromaculatus was measured using a graduated vessel. For the assessment of the percentage of dry matter, 3 aliquots of 20.0 g each of intact A. atromaculatus were processed by heating in an oven at 60°C for 72 hours.
Calves and housing conditions, preparation of the inocula and inoculations
Four, approximately 2.5- to 3-month-old, weaned, neutered male, Hereford calves weighing 88.5 to 98.0 kg were housed in a common pen with continuous access to shade and fresh water. The calves were fed wheat haylage ad libitum and increasing amounts (from 0.5 to 1.75 kg/calf/day) of a pelleted ration formulated for weaned calves (Progenie Recría; PROLESA, Uruguay). Seven days before the inoculations, the calves were randomly assigned correlative numbers from 1 to 4 using an online randomizing tool (https://www.randomizer.org/).
On February 16, approximately 3.0 kg (wet weight) of pooled adult A. atromaculatus collected from properties 1 and 2 were removed from the refrigerator and left for 20 minutes at room temperature (aliquots of intact beetles were stored at –80°C for toxicological testing). After verifying their viability by observing their mass motility, the insects were ground with an electric stick hand blender (Futura FUT-BM 800, China). Immediately after processing, 3 separate aliquots of 400, 940, and 1470 g (wet weight) of ground beetles were suspended in distilled water within clean individual plastic containers, homogenized with an electric bench blender (Philips, Argentina), and transferred into new 4-liter Perfect Udder bags (Dairy Tech Inc, Colorado, USA) (inocula 1–3). A fourth bag was filled with 4 liters of distilled water with no insects (inoculum 4). The 4 bags were kept at room temperature until inoculation of calves 1–4, respectively, within approximately 2.5 hours of preparation of the inocula.
The inoculations were conducted by esophageal tubing. The 4-liter Perfect Udder bags containing the inocula were attached to the stainless-steel esophageal probe of Springer Magrath fluid feeders designed for calves using new hoses. Calves 1 to 3, of 88.5, 94.0, and 98.0 kg body weight (BW), respectively, were inoculated with inocula 1 to 3, so that they received dosages of 4.5, 10.0, or 15.0 g/kg BW (wet weight) of ground A. atromaculatus suspended in distilled water, respectively. Calf 4, weighing 95.0 kg, was administered inoculum 4 consisting of distilled water (control).
Clinical examinations, sampling of live calves, and end of the experiment
Before and after the inoculations, the calves were observed periodically during the daytime by a veterinarian to assess their general appearance and attitude, and, if seen defecating, record the fecal consistency score and the presence of mucus, fibrin, and/or blood in the feces. Six and two days before the inoculations (times -6 and -2 days), minutes before the inoculations (time 0 hours) and approximately 12, 24, and 48 hours postinoculation (p.i.), the calves were subjected to clinical examinations and sampling.
The clinical examination included evaluation of the fecal consistency score and the presence of mucus, fibrin, and/or blood in the feces; the rumen contraction rate (number of contractions in 3 minutes determined by auscultating the rumen with a stethoscope on the left paralumbar fossa); the rectal temperature (°C); the heart rate (beats/min) and the respiratory rate (breaths/min); the capillary refill time (seconds) assessed in the gingival mucosa; and evaluation of the color and moisture of the oral mucosa.
For fecal consistency, a 4-tier scoring system was used, with scores 0, 1, 2, and 3 corresponding to firm, soft, runny, and watery feces, 23 respectively, and scores 2 and 3 indicating diarrhea. 5 The rumen contraction rate was interpreted as follows: no contractions in 3 minutes = ruminal hypomotility, 1–3 contractions in 3 minutes = normal ruminal motility. The capillary refill time was considered normal at or below 2 seconds, and prolonged at or above 3 seconds. 17
Whole blood, serum, and/or fecal samples were collected at times -6 days, 0, 12, 24, and 48 hours p.i. and processed for different laboratory tests, including hematology, serum biochemistry, virology, parasitology, and bacteriology (see below). Blood was obtained by coccygeal venipuncture using vacutainer tubes with ethylenediaminetetraacetic acid (EDTA) for whole blood and clot activator for serum. After centrifugation, the serum was aliquoted and stored at –20°C until analyzed.
Calves showing severe illness were euthanized and subjected to postmortem pathological examination. Euthanasia was conducted by rapid intravenous injection of an overdose of barbiturate solution in the jugular vein, a method recommended in the American Veterinary Medical Association guidelines for the euthanasia of bovids. 2 The end of the experiment for calves not showing clinical signs was set at 7 days p.i.
Postmortem examination and sampling
Following death or euthanasia, the calves were subjected to complete necropsies and all major organs were carefully inspected for gross lesions. Fresh jejunal and ruminal contents were collected for virology and pH determination, respectively. In addition, tissue samples were collected for histopathology. These were immersion-fixed in 10% buffered formalin for greater than or equal to 72 hours and processed to produce 4-µm sections that were mounted on glass slides, stained with hematoxylin and eosin, and cover-slipped for histological examination under optic microscope (Axio Scope.A1; Carl Zeiss, Germany).
The examined tissues included tongue, esophagus, reticulum, omasum, rumen, abomasum, duodenum, jejunum, ileum, cecum, colon, rectum, liver, gallbladder, kidneys, adrenal gland, thyroid gland, salivary gland, urinary bladder, pancreas, spleen, mesenteric and ruminal lymph nodes, aorta, diaphragm, skeletal muscles, heart, trachea, lungs, thymus, spinal cord, and brain.
Experiment 2
Once experiment 1 was concluded, at 7 days p.i., the only unaffected calf (calf 4, uninoculated control) was dismissed from experiment 1 and enrolled in experiment 2. In this second experiment, the calf, weighing 100.5 kg BW (weight gain between experiments of ~0.8 kg/day), was dosed with an aqueous suspension of 252 g of ground A. atromaculatus, representing 2.5 g/kg BW on a wet weight basis. For this experiment, A. atromaculatus were collected from alfalfa pastures on 2 farms in the departments of Colonia (40 g, property 3, February 18, Fig. 1b) and Soriano (260 g, property 4, February 22), Uruguay. The inoculation took place on February 23.
All conditions including housing of the calf, insect handling, isolation and preparation of the inoculum, sampling of A. atromaculatus before processing for toxicological testing, inoculation, clinical examination, premortem sampling (conducted at times 0, 12, 24, and 47 hours p.i.), euthanasia, and postmortem pathological examination and sampling were the same as for experiment 1.
Hematology and serum biochemistry
Whole blood and serum obtained at times -6 days, 0, 12, 24, and/or 48 hours p.i. from calves in experiment 1, and at times 0, 12, 24, and 47 hours p.i. in the calf of experiment 2, were processed for hematology and serum biochemistry, respectively. Hematologic analyses were conducted with an automatic hematology analyzer (BC-5000 Vet; Mindray, China) and included white blood cell (WBC) count, neutrophil, lymphocyte, monocyte, eosinophil, and basophil counts; red blood cell count; hematocrit; and hemoglobin concentration.
Serum biochemistry analyses were conducted with an automated analyzer (BA200; Biosystems, Barcelona, Spain) and commercial reagents and controls, and included quantification of total proteins, albumin, the activity of aspartate aminotransferase and gamma-glutamyl transferase, cholesterol, calcium (Ca), phosphorous (P), magnesium (Mg), zinc (Zn), urea, nonesterified fatty acids (NEFA), beta-hydroxybutyrate (B-HB), and glucose. The interassay coefficients of variation for the commercial controls used were less than or equal to 10%. The results of the hemogram and serum biochemistry analyses were interpreted considering the relative individual variations before (-6 days and 0 hours) and after (times 12, 24 and 47–48 hours) the inoculations for each calf, and by comparison with reference values.27,33
Bacteriology, Parasitology, and Virology
Feces collected at -6 days from all 4 calves in experiment 1 were processed for selective bacterial culture for Salmonella spp., 8 and fecal flotation for quantification of nematode eggs and coccidia (Eimeria spp.) oocysts per gram of feces with a limit of detection of 40 eggs/oocysts. Whole blood obtained at -6 days was processed by reverse transcriptase quantitative polymerase chain reaction (RT-qPCR) for bovine viral diarrhea virus detection. 11
Laboratory Testing on Samples Collected From the Necropsied Calves
Fresh/frozen jejunal contents obtained postmortem from the 4 calves were processed and tested, as previously described, by RT-qPCR for the detection and/or quantification of bovine coronavirus, 9 group A rotavirus (RVA), 6 and bovine viral diarrhea virus. 11 The pH of the ruminal contents was assessed with a digital pH meter (ST2100; Ohaus).
Analysis for Cantharidin, Batrachotoxin, and Nontargeted Analysis
Aliquots of A. atromaculatus obtained before grinding for the preparation of the inocula in both experiments were processed for toxicological testing. Insect material was homogenized in 0.1% formic acid in water, and then extracted into 0.1% formic acid in acetonitrile. The resulting supernatant was cleaned up using QuEChERS, followed by lipid removal by dispersive solid phase extraction. It was split into 2 equal portions and evaporated under nitrogen, with one being redissolved for liquid chromatography-mass spectrometry (LC-MS/MS) analysis and the other for gas chromatography-mass spectrometry (GC-MS) analysis.
LC-MS/MS analysis was done using a Q Exactive Orbitrap mass spectrometer (ThermoFisher Scientific, Waltham, Massachusetts, USA) with electrospray ionization in positive mode. An Agilent Eclipse Plus C18 column (100 × 2.1 mm) (Agilent, Santa Clara, California) was used with 0.1% formic acid in water (mobile phase A) and 0.1% formic acid in acetonitrile (mobile phase B), and a gradient from 1% to 98% Mobile Phase B. Data were searched for any positive identifications versus an in-house library of over 200 compounds, including pesticides, environmental toxicants, drugs, and natural products. A separate LC-MS/MS run was also done using parallel-reaction monitoring for batrachotoxin, and comparisons made to a reference standard.
Targeted GC-MS analysis of cantharidin was done using an Agilent 7890 B GC with a 5977A MSD and DB-5MS column. Selected ion monitoring was used for cantharidin detection, and comparisons made to a reference standard.
An additional extraction was done for nontargeted GC-MS screening by adding ammonium hydroxide and extracting into ethyl acetate. The resulting supernatant was analyzed by GC-MS as described for cantharidin, with the exception that it was run scanning over the entire range of 40 to 700 m/z, rather than using selected ion monitoring. Data were searched for any positive identifications using a commercially available library (Wiley Science Solutions, Hoboken, New Jersey, USA).
Metals and Selenium Analysis
Representative portions of samples were taken and digested with nitric acid by hot block digestion. Following digestion, hydrochloric acid was added to samples, and they were brought to a final volume of 10 mL with 18 megohm water and filtered with a syringe filter. Filtered samples were run by inductively coupled plasma optical emission spectroscopy to measure the metals included in the heavy metal screen (arsenic, cadmium, copper, iron, lead, manganese, mercury, molybdenum, and zinc). For selenium analysis, a 250 µL portion of filtered digest was subsampled and diluted to 5 mL with diluent containing internal standards and run by inductively coupled plasma mass spectrometry. Method blank and certified reference materials were prepared with samples for quality control. A standard curve and blank were run at the beginning of the sample batch to calculate concentration and continuing calibration verification, and blanks were run every 10 samples to ensure drift was not more than 10% throughout the run.
Mycotoxin Analysis
Insect samples were rough chopped, and 1 g of sample was combined with 1 mL of water and homogenized. This was next extracted with 4 mL of acetonitrile:water (1:1). The resulting supernatant was analyzed by LC-MS/MS using an Agilent 1290 HPLC combined with a SciEx 7500 QTrap mass spectrometer. Data were collected using electrospray ionization in positive mode with multiple reaction monitoring. Identifications were made based on comparisons to standard reference compounds for aflatoxin B1, deoxynivalenol, T-2 toxin, ochratoxin A, fumonisin B1, and zearalenone.
Microscopic Examination of the Beetles
Intact beetles were fixed in 10% buffered formalin for 48 hours and then crushed and chopped with a scalpel to produce tiny fragments that were routinely processed histologically, stained with hematoxylin and eosin, and mounted for microscopic examination following the same procedure used for vertebrate tissues. In addition, the grounded insects of the inoculum used for experiment 1 were suspended in water, and this suspension was placed on glass slides, cover-slipped, and examined directly under the microscope (wet mount).
Results
Clinical Evaluations
In experiment 1, the calves were inspected twice daily for 7 days before the inoculations, when they were bright, alert, and responsive (BAR). There was no feed refusal on the 5 days prior to the inoculations, indicating a normal feed intake. On the clinical examinations conducted at times -6 days, -2 days, and 0 hours, all 4 calves were BAR and had heart rates, respiratory rates, rectal temperatures, capillary refill times, and color and moisture of the oral mucosa within reference ranges, with normal ruminal motility and fecal scores less than or equal to 1 with no mucus, fibrin, nor blood.
The clinical examinations conducted at approximately 12 hours p.i. revealed ruminal hypomotility in calves 1 and 3, with normal ruminal motility in calves 2 and 4. All calves were BAR and had heart rates, respiratory rates, rectal temperatures, capillary refill times, and color and moisture of the oral mucosa within normal limits, and fecal scores less than or equal to 1. At approximately 15 hours p.i., calf 3 was in sternal recumbency with drooling, ruminal distension (tympany), and reluctance to stand up, while calves 1, 2, and 4 were BAR.
At approximately 24 hours p.i., all 4 calves were in sternal recumbency. Calves 2 to 4 stood up and moved away when approached, but calf 1 remained in sternal recumbency, was reluctant to stand up and depressed, and had ruminal distension (tympany). When the calves were herded, calves 1 to 3 walked slowly and were listless (apathy). Upon clinical examination, calves 1 to 3 had ruminal hypomotility and low rectal temperature (36.5°C–37.1°C), while calf 4 (control) had normal ruminal motility and was normothermic (38.6°C). In addition, calves 2 and 3 had mild muscle fasciculations in the gluteal region. All calves had fecal scores of 0 with no mucus, fibrin, nor blood.
At approximately 33 hours p.i., there were leftovers of the pelleted ration in the feeder, indicating feed refusal. Calves 1 to 3 remained in sternal recumbency for long periods (Fig. 2a). They were alert but allowed to be touched and pushed, were reluctant to stand up, and had mild ruminal distension (tympany). At approximately 38 hours p.i., calf 2 showed bruxism with frothing at the mouth but remained alert.

Clinical signs. (a) Calf 3 at approximately 33 hours after oral inoculation with 15.0 g/kg of body weight (BW) of Astylus atromaculatus. The calf would spend prolonged periods in sternal recumbency with the neck extended, was conscious of the surroundings but listless, allowed to be approached, and was reluctant to stand up even when touched or pushed; however, at this time point he was able to get up if forced to and remain in standing position. (b) Watery feces with mucus (diarrhea) deposited on the ground of the pen by the only calf in experiment 2, approximately 42 hours after oral inoculation with 2.5 g/kg BW of A. atromaculatus.
At approximately 48 hours p.i., calves 1 and 2 were found dead (died overnight). Calf 3 was in lateral recumbency, unable to rise, had muscle fasciculations on the right front limb, and was euthanized. Calf 4 remained BAR and clinically normal until 7 days p.i., when he was dismissed from experiment 1 and enrolled in experiment 2. During this period, he was fed twice daily. There was no feed refusal, and the calf had a daily weight gain of approximately 0.8 kg, indicating normal feed intake.
In experiment 2, at 0 hours, the calf was BAR and had a heart rate, respiratory rate, rectal temperature, ruminal motility, fecal score, capillary refill time, and color and moisture of oral mucosa within normal limits. There was feed refusal starting at approximately 12 hours p.i. and the clinical examination conducted at this time revealed that the calf was BAR and had a slightly extended capillary refill time (3 seconds), with all the other assessed clinical parameters within normal limits.
At approximately 24 hours p.i., the calf passed green fluid feces (fecal score = 2, diarrhea) and had ruminal hypomotility with the other clinical parameters within normal limits. At 29 hours p.i., he was BAR and had a fecal score of 2 with mucus. Between 33 and 42 hours p.i., the calf spent prolonged periods in sternal recumbency, was bright and alert, got up when approached but had decreased response, walked with some weakness, and continued to have diarrhea (fecal score = 2–3) with mucus (Fig. 2b). At 47 hours p.i., the calf was in lateral recumbency; unable to stand; and had score 3 diarrhea, capillary refill time of 3 sec, rumen hypomotility, and tachypnea with shortness of breath; and was euthanized.
Hematology and Serum Biochemistry
The individual results of the hematologic and biochemical analyses are shown in Supplemental Table S1. In experiment 1, calves 1 and 3 had red blood cell counts, hematocrits, and hemoglobin concentrations within the reference ranges at times -6 days and 0 hours, while these variables increased above the reference ranges at 12 to 48 hours p.i. In calf 2, even though these parameters were above the reference ranges before the inoculations, they increased slightly and progressively at 12 and 24 hours p.i. In calf 4 (control), these parameters were within reference ranges at all sampling points during this experiment.
Calves 2 and 3 had increased WBC counts (leukocytosis) with increased neutrophil counts and percentages (neutrophilia) at 12 (calf 3) and 24 (calf 2) hours p.i. In calf 3, neutrophilia was followed by a progressive reduction in WBC at 24 and 48 hours p.i. with leukopenia at 48 hours p.i. due to progressive reductions in neutrophil and lymphocyte counts. Calves 1 and 4 (control) had WBC counts within the reference range, although the neutrophil counts were below the reference range at times 0 hours (calf 1) and -6 days, 0, 12, and 48 hours (calf 4).
In experiment 2, the calf had a red blood cell count, hematocrit, and hemoglobin concentration within the reference ranges at times 0, 12, and 24 hours p.i., but these variables increased above the reference range at 47 hours p.i. The WBC count was within the reference interval at 0, 12, and 24 hours p.i., but the calf had leukopenia at 47 hours p.i.
Regarding serum biochemistry, in experiment 1, calves 1 to 3 had markedly increased NEFA (≥1.05 mmol/L) at 12 (calves 1–3) and 24 (calf 2) hours p.i., with increased B-HB (≥1 mmol/L) at 12 (calves 1–3) and 24 (calves 1–2) hours p.i. Calf 4 (control) had mildly increased NEFA (0.85 mmol/L) at 48 hours p.i., but B-HB levels were within the reference range at all sampling times. Calf 3 had terminal elevations of P (7.1 mmol/L), Mg (2.4 mmol/L), and urea (7.5 mmol/L) at 48 hours p.i.
In experiment 2, the calf had mildly increased NEFA (0.81 mmol/L) at 24 hours p.i. with no elevations of B-HB at any sampling time point. At 47 hours p.i., this calf had terminal elevations of P (7.6 mmol/L), Mg (2.7 mmol/L), urea (10.3 mmol/L), and aspartate aminotransferase activity (289 IU), and was markedly hypoglycemic (0.2 mmol/L).
Calf 3 in experiment 1 and the calf in experiment 2, which were the only animals that remained alive until 47 to 48 hours p.i., had progressive relative elevations in the concentration of total proteins at all sampling points after the inoculations, reaching maximum levels of 17% to 20% above the preinoculation levels (time 0 hours) at 47 to 48 hours p.i. In calf 3, there was a concomitant 36% increase in the concentration of albumin, which at this time point was above the reference range (hyperalbuminemia).
Unless stated otherwise above, in both experiments the activities of gamma-glutamyl transferase and aspartate aminotransferase, the levels of albumin, total proteins, cholesterol, Ca, P, Mg, and Zn were unremarkable, although many animals had total proteins, cholesterol, and Zn below the reference ranges, and some had P and/or Mg levels slightly above the reference ranges at different sampling times both before and after the inoculations.
Bacteriology, Parasitology, and Virology Testing of Live Calves
All 4 calves were negative for Salmonella spp. culture in feces and bovine viral diarrhea virus RT-qPCR in whole blood collected 6 days before the inoculation (experiment 1). No nematode eggs or coccidia oocysts were seen on fecal flotation.
Necropsy Findings
In all 4 calves, the reticulum, omasum, and rumen had multifocally extensive areas of reddening of the mucosa (Fig. 3a–b), which was similar in extension and severity in all calves. In addition, in calf 4 (experiment 2), there was extensive edema in the wall and serosa of the rumen (Fig. 3b). In calves 1 to 3, there was regional edema in the mesentery/serosa of the small curvature of the abomasum at the level of the junction with the omasum (Fig. 3c), with corresponding edema in the wall of the underlying abomasal folds.
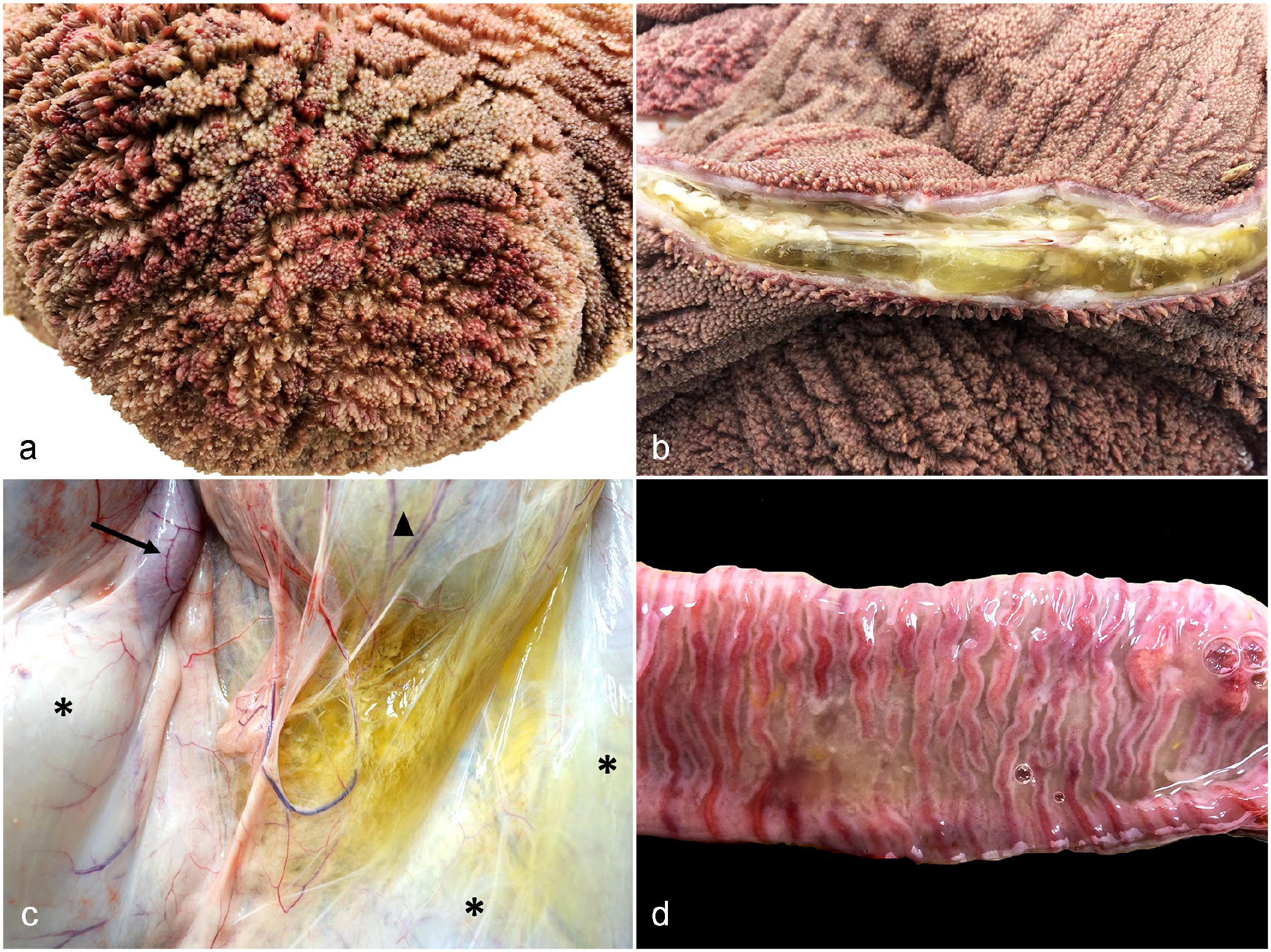
Necropsy findings in calves orally inoculated with Astylus atromaculatus. (a) Rumen, calf 3. There is extensive reddening of the mucosa overlying the papillae (rumenitis). (b) Rumen, calf 4. The wall is expanded by yellowish translucent gelatinous material (mural edema). (c) Calf 3. Serosal view of the abomasum (asterisks), omasum (arrowhead), and first segment of the duodenum (arrow). The mesentery and serosa at the level of the smaller curvature of the abomasum and its junction with the omasum is expanded by yellowish translucent gelatinous material (edema). (d) Jejunum, calf 3. The lumen is devoid of digesta and contains grayish fine mucoid material (catarrhal exudate) deposited over a diffusely reddened (congested) mucosa.
Segmentally in all calves, the small intestine had scant or no digesta but contained large amounts of mucus. In calf 3, the mucosa of the small intestine was diffusely reddened (Fig. 3d), while calf 2 had regional hyperemia with engorged capillaries and scattered petechiae in the jejunal mucosa. No lesions were found in the small intestinal mucosa of calves 1 and 4. The large intestines were grossly unremarkable in all calves.
In calves 1 and 2, which were found dead, the lungs had a dark pink to red parenchyma and a meaty texture (terminal pulmonary congestion/edema). In calves 3 and 4 that were euthanized, the lungs were unremarkable.
In all 4 calves, the urinary bladder contained clear amber urine, and there were no gross lesions in the urinary bladder, gallbladder, liver, pancreas, kidneys, adrenal gland, salivary gland, thyroid gland, thymus, oral mucosa, tongue, larynx, esophagus, mesenteric lymph nodes draining the intestines, spleen, myocardium, diaphragm, skeletal muscles, brain, or spinal cord. Ground fragments of the insects were recognized in the contents of the forestomachs and intestines in all calves.
Histopathology
In all 4 calves, the most significant microscopic lesions were diffuse necrotizing reticulo-omaso-rumenitis, characterized by extensive swelling, hydropic degeneration, and necrosis of keratinocytes with loss of intercellular cohesion and the formation of intraepithelial vesicles filled with proteinaceous fluid and/or neutrophils (Fig. 4a–c). Necrotic keratinocytes were shrunken and hypereosinophilic with pyknotic and hyperchromatic nucleus. Multifocally, the lamina propria at the tip of the papillae had pyknotic and karyorrhectic cellular debris, and/or was infiltrated by neutrophils, lymphocytes, and macrophages. These lesions were largely indistinguishable among calves; however, in calves 1, 2, and 4, they more often involved the basilar layer of keratinocytes resulting in multifocal superficial ulcers, which in calves 1 and 4 were accompanied by plasma and fibrin extravasation in the lamina propria/submucosa with scattered microhemorrhages, microthrombi, and pleocellular inflammatory infiltrates. In calf 1, golden-brown, black, or translucent, acicular, foreign bodies with sharp edges, interpreted as chitinous insect fragments, surrounded by severe inflammation were found embedded in the wall of the rumen (Fig. 4d–e).
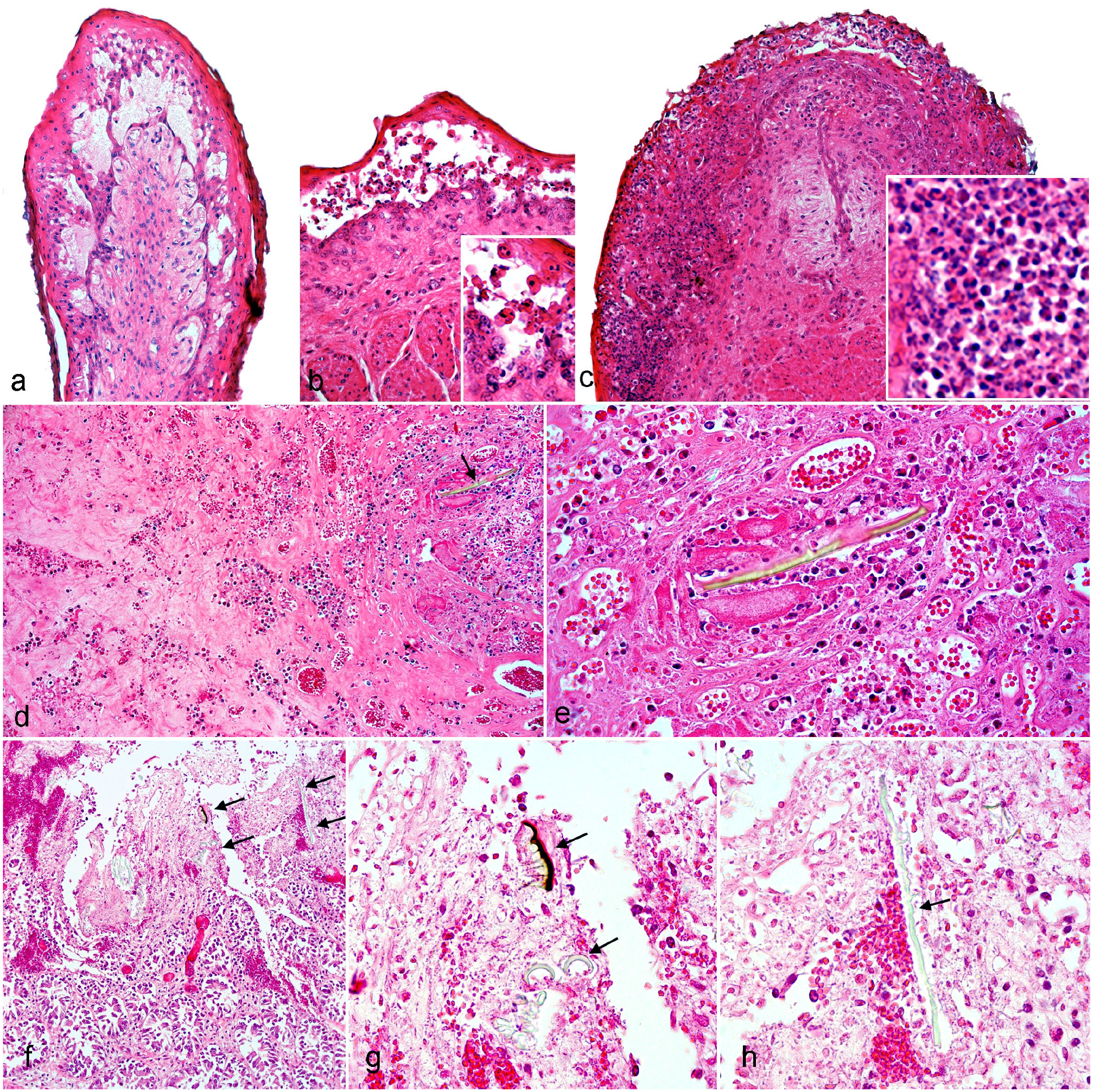
Histological lesions in calves inoculated with Astylus atromaculatus. Hematoxylin and eosin (HE). (a) Rumen, calf 3. There is intercellular edema in the epithelium of the tip of a ruminal papilla that results in the formation of intraepithelial vesicles filled with eosinophilic fluid. (b) Omasum, calf 3. In the lateral aspect of an omasal fold, there is loss of intercellular cohesion (acantholysis) between keratinocytes of the strata spinosum and granulosum, which results in detachment and the creation of empty intraepithelial spaces. Detached keratinocytes are necrotic (inset). The basilar keratinocytes are still attached to the basement membrane. (c) Omasum, calf 3. At the tip of an omasal fold, there is full-thickness epithelial necrosis and infiltration of the epithelium by abundant transmigrating neutrophils with the formation of pustules (inset). The lamina propria is infiltrated by mixed inflammatory cells. (d) Rumen, calf 1. The submucosa/muscularis is expanded by edema, extravasated fibrin, and erythrocytes (hemorrhage). There is an intralesional chitinous insect fragment (arrow) of approximately 140 µm in length. (e) Higher magnification of (d) showing the chitinous insect fragment (center) surrounded by fibrin, hemorrhage, and neutrophils. (f) Jejunum, calf 2. There is necrosis of the villi and hemorrhage expanding the lamina propria (hemorrhagic enteritis) with several chitinous insect fragments inserted into the damaged mucosa (arrows). (g) Higher magnification of (f) showing pigmented and nonpigmented chitinous insect fragments with sharp ends (arrows) embedded into the damaged mucosa. (h) Higher magnification of (f) depicting an approximately 130 by 3 to 4 µm, nonpigmented, acicular, chitinous insect fragment (arrow) embedded into the damaged mucosa.
In calves 1 to 3, there was transmural edema and fibrin extravasation in the wall of the abomasum and the corresponding omasal wall at the level of the omaso-abomasal junction. This same lesion was present in calf 4, affecting the ruminal wall instead of the omaso-abomasal junction. There was submucosal edema in the abomasum of all of the calves, but none of the calves had lesions in the abomasal mucosa.
In different segments of the small intestines, all 4 calves had multifocal villus blunting with mildly increased cellularity and pyknotic/karyorrhectic debris in the superficial lamina propria of affected villi. Multifocally, a few crypts were dilated and contained sloughed enterocytes with swollen or hypereosinophilic cytoplasm and pyknotic nuclei. In calves 3 and 4, there was multifocal crypt herniation into the submucosa and/or the lymphoid aggregates of the Peyer’s patches, with herniated crypts often containing sloughed necrotic enterocytes. In calf 3, there was infrequent multifocal necrosis of enterocytes at the tips of the villi. In calf 2, there was multifocal hemorrhage and fibrin extravasation in the mucosa of the jejunum (fibrinohemorrhagic enteritis), which colocalized with foreign bodies that were interpreted to be chitinous insect fragments that were embedded into the damaged mucosa (Fig. 4f–h) and located in between affected villi. In calf 4, there was superficial exfoliative colitis; sloughed enterocytes in the mucosa-lumen interface were occasionally admixed with mucus, cellular debris, and foreign bodies interpreted as chitinous insect fragments.
Calves 1 and 2 (found dead) had acute pulmonary lesions consisting of multifocal patchy congestion of alveolar capillaries with alveolar edema, which in calf 1 were accompanied by alveolar hemorrhage. No lung lesions were present in calves 3 and 4 (euthanized).
In calves 2 and 3, there was moderate Alzheimer type II astrocytosis throughout the cerebral cortex of the frontal, parietal, and/or occipital lobes. There were no lesions in the cerebral hemispheres in calves 1 and 4.
Laboratory Testing of Postmortem Samples
RVA was detected by RT-qPCR in the intestinal contents of calves 1, 3, and 4 at low viral loads of 7500, 9500, and 35,000 genome copies/mL, respectively (log10 viral load of 3.9, 4.0, and 4.5, respectively). Calf 2 was negative for RVA. The RT-qPCR assays for bovine coronavirus and bovine viral diarrhea virus were negative in the intestinal contents of all 4 calves. The pH of the ruminal contents was 5.8, 5.4, 6.6, and 6.2 in calves 1 to 4, respectively.
Astylus atromaculatus Size, Weight, Volume, and % of Dry Matter
The average length and width of 20 adult individuals was 10.38 × 3.99 mm, with males being slightly smaller (9.87 × 3.83 mm) than females (10.89 × 4.15 mm). The average fresh weight of adult individuals (n = 50) was 27.3 mg, and the dry matter content was 34.05%. The volume of 250 g of adult individuals was approximately 850 mL.
Toxicological Testing
Cantharidin, batrachotoxin, arsenic, cadmium, lead, mercury, molybdenum, and the mycotoxins aflatoxin B1, deoxynivalenol, T-2, ochratoxin A, fumonisin B1, and zearalenone were not detected on either sample at or above the reported limit of detection (Table 1). Concentrations of copper, iron, manganese, Zn, and selenium ranged between 13 and 14, 42 and 57, 11 and 12, 78 and 90, and 0.08 and 0.1 ppm (wet weight), respectively (Table 1).
Results of the analyses for the quantification of cantharidin, batrachotoxin, heavy metals, selenium, and mycotoxins in aliquots of Astylus atromaculatus used for the preparation of the inocula in experiments 1 and 2.
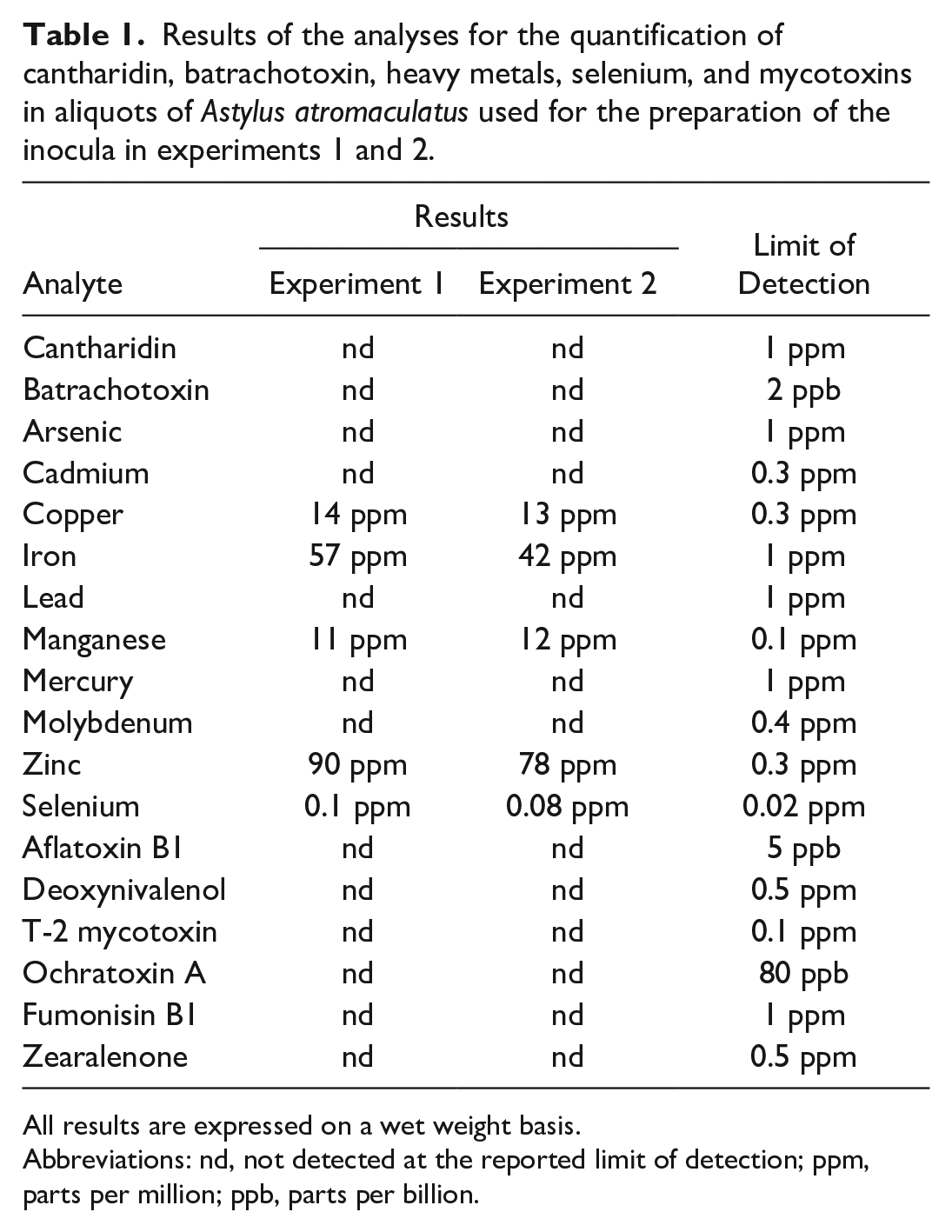
All results are expressed on a wet weight basis.
Abbreviations: nd, not detected at the reported limit of detection; ppm, parts per million; ppb, parts per billion.
The nontargeted LC-MS/MS screen revealed the pyrrolizidine alkaloid “intermedine” in the inoculum of experiment 1 and did not detect compounds that were likely to have toxicological significance in the inoculum of experiment 2. The nontargeted GC-MS screen did not detect toxic compounds on either sample.
Microscopic examination of the beetles
Numerous golden-brown, acicular, chitinous insect fragments with sharp ends measuring up to approximately 200 μm in length were observed both on hematoxylin and eosin-stained sections of the insects and on unstained wet mounts (Fig. 5a–c). Some of these were still attached to the insect cuticle, suggesting that they might be setae (Fig. 5a–b), while others formed clusters (Fig. 5c) or were individualized and not clearly associated with the cuticle, suggesting that they might have been part of internal structures of the insect. Some fragments morphologically resembled the ones found histologically in the gastrointestinal tissues of the inoculated calves.
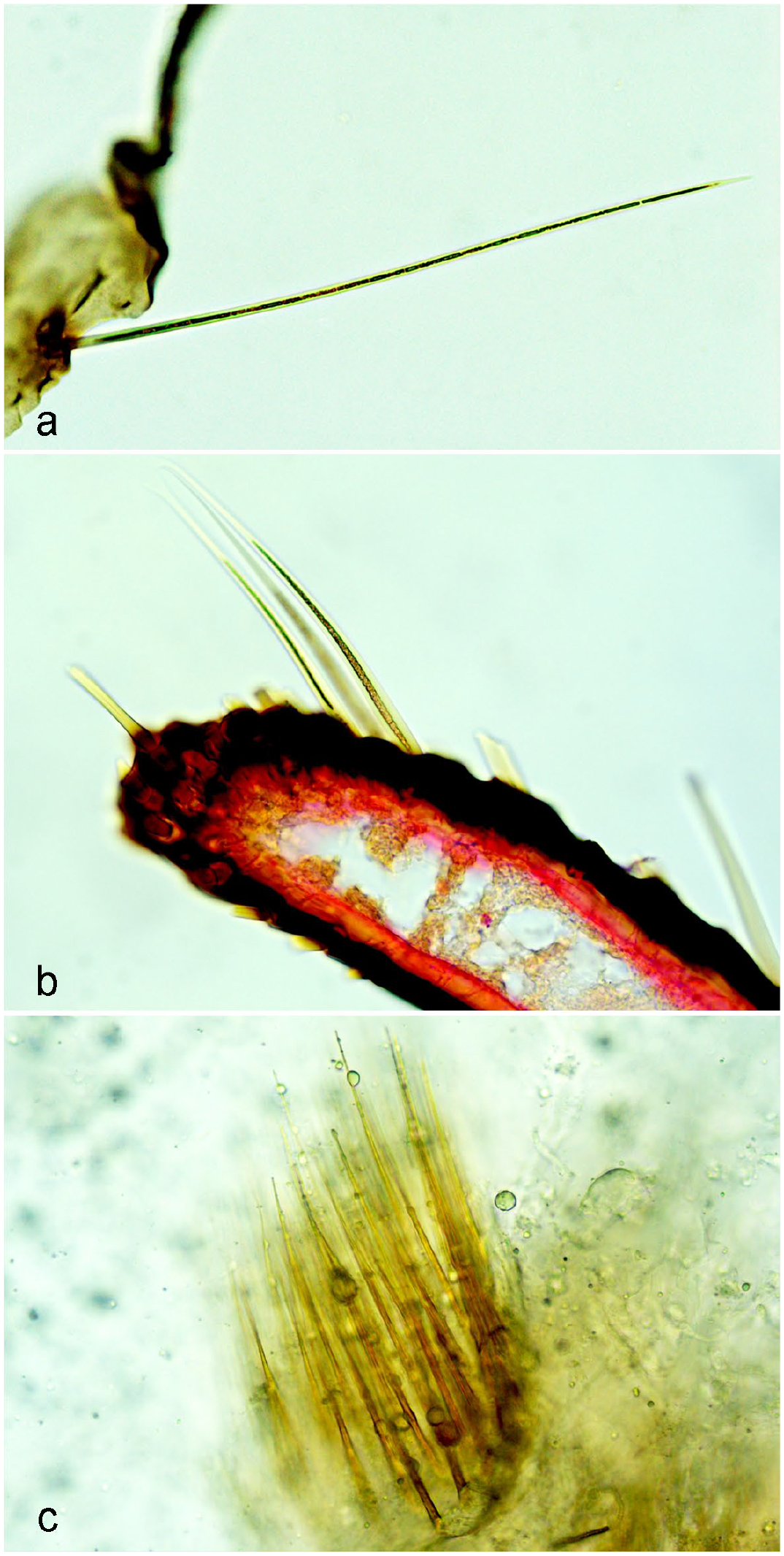
Microscopic examination of fragments of Astylus atromaculatus processed and stained with (a, b) hematoxylin and eosin or (c) on unstained wet mount. Chitinous, golden-brown sharp-ended setae inserted in the pigmented cuticle range approximately from (a) 200 to (b) 125 µm in length. (c) Cluster of acicular sharp-ended golden-brown structures measuring approximately 140 µm in length not clearly associated with the insect cuticle.
Discussion
In this study, we demonstrate that A. atromaculatus is lethal to cattle when administered at a single oral dose. The clinical and pathological outcomes were similar regardless of the dose. In experiment 1, calves 1 to 3 inoculated with 4.5, 10.0, and 15.0 g/kg BW, showed similar clinical signs, duration of the clinical course, clinical outcome, and postmortem lesions, except for calf 2, which was the only one with fibrinohemorrhagic enteritis. In the calf of experiment 2 that was inoculated with 2.5 g/kg BW, the clinical course, outcome, and the pathological findings were largely indistinguishable from those seen in calves of experiment 1, although this was the only calf that developed diarrhea and exfoliative colitis. Despite the limitations in our study due to the low number of inoculated calves, our results suggest that the single oral dose that is lethal to cattle is probably less than 2.5 g/kg BW.
Considering the average weight of 27.3 mg/beetle, and the total dose of approximately 250 g of beetles that was lethal to an approximately 100 kg calf, ingestion of 9158 beetles would be required to reach this lethal dose. Depending on factors such as forage availability and quality that influence the grazing behaviors, the biting rate of grazing cattle can range from 30 to 60 bites/min with grazing periods of 6 to 11 h/day. 4 Taking into account average values of 45 bites/min and a grazing period of 8.5 h/day (22,950 total bites), a 100 kg calf ingesting on average 0.4 beetles/bite (2 beetles every 5 bites) could theoretically ingest this dose in a day. This seems plausible in heavily invaded pastures with pollen beetles clustering on plant parts that are palatable such as flowers and leaves (Fig. 1), and even more so in confined cattle fed fresh chopped alfalfa mixed with beetles, as recorded in 2 of the outbreaks in Argentina. 13
In all calves in this study, the disease was acute with clinical signs becoming evident a few hours postinoculation, and rapid progression ending in death or euthanasia within 48 hours. In the experiment conducted in sheep in South Africa, 16 the interval between dosing and development of clinical signs was longer (2–4 days), and death occurred at 6 and 9 days p.i. In that study, one sheep received a single dose of 5 g/kg (similar to the dosage of 4.5 g/kg we administered to one of the calves), while 2 other sheep received repeated daily dosages of 3 g/kg over 3 days and 1.2 g/kg over 18 days (total dosages of 9.0 and 21.6 g/kg, respectively). Interestingly, the sheep receiving the highest total dose over the longest period developed clinical disease but recovered and survived. In spontaneous outbreaks of A. atromaculatus-associated disease in cattle ingesting unknown doses (but likely lower than the ones we used in experiment 1), clinical signs were first observed between 1 and 5 days after exposure to the beetles, and the clinical course ranged from 0 (sudden death) to 15 days. 13 Based on this limited information, it could be hypothesized that cattle might be more susceptible than sheep to A. atromaculatus, although this should be further explored in properly designed experiments using standardized inocula.
Clinically, the disease in experiment 1 was characterized by feed refusal, prolonged sternal recumbency with reluctance to stand up (starting at approximately 15 hours p.i.), apathy, depression, ruminal hypomotility, mild ruminal tympany, and hypothermia with an acute course progressing to lateral recumbency and death (calves 1–2) or severe disease necessitating euthanasia (calf 3) within 48 hours of inoculation. In addition, one calf (calf 2) showed bruxism with frothing at the mouth, and another (calf 3) had drooling. No diarrhea was seen in any of the calves in this experiment. In experiment 2, the clinical course was initially characterized by feed refusal at approximately 12 hours p.i., followed by diarrhea as early as 24 hours p.i., lasting until the end of the experiment. Other signs included ruminal hypomotility, prolonged recumbency with reluctance to stand up, and depression with rapid clinical deterioration necessitating euthanasia by 47 hours p.i. Thus, the main difference in the clinical signs in this calf inoculated with the lowest dose was the manifestation of diarrhea.
Overall, the clinical signs in the calves of our study overlap with those described in experimentally inoculated sheep, 16 and in spontaneous cases in cattle. 13 Interestingly, Kellerman et al described that lactating cows naturally exposed to A. atromaculatus dried off. While this is not unexpected in cattle with clinical gastrointestinal disease, the effects of A. atromaculatus ingestion on milk production should be further evaluated experimentally in lactating animals.
The elevations in the red blood cell counts, hematocrits, and hemoglobin concentrations observed in the calves inoculated with the beetles at 12–48 hours p.i. in both experiments were probably due to hemoconcentration because of increased vascular permeability. This was supported by the severe edema found in the rumen, reticulum, and omasum in all 4 calves. Increased vascular permeability could have occurred because of acute inflammation of the forestomachs induced by the beetles, and/or by translocation of bacterial endotoxins (endotoxemia) through the severely damaged mucosa. 27 Hemoconcentration could have been further aggravated by reduced water intake, considering that all the affected animals went off feed (and presumably water), and, in the calf of experiment 2, possibly by dehydration secondary to diarrhea.
Hemoconcentration due to dehydration alone is usually accompanied by hyperproteinemia. 27 While none of the calves in either experiment had total protein levels above the reference range, the only 2 calves that remained alive until 47–48 hours p.i (calf 3 in experiment 1 and the only calf in experiment 2) had progressive relative elevations in the concentration of total proteins at all sampling points after the inoculations, that reached maximum levels of 17% to 20% above the initial preinoculation levels at 47 to 48 hours p.i. In calf 3, this was accompanied by hyperalbuminemia. These results support that hemoconcentration could be partially attributed to mild dehydration due to reduced water intake after the inoculations in both calves, as well as diarrhea in the calf of experiment 2, but do not explain the hemoconcentration seen in all calves, which as discussed above most likely resulted from increased vascular permeability in the forestomachs.
Leukocytosis with neutrophilia at 12 to 24 hours p.i. followed by leukopenia (47–48 hours p.i.) due to reductions in neutrophil and lymphocyte counts were found in calves 2 and 3 of experiment 1 and in the calf of experiment 2. These results are consistent with an initial corticosteroid/stress leukogram followed by an acute inflammatory stimulus, 27 which also matches with the severe omaso-reticulo-rumenitis found postmortem.
The serum levels of NEFA were highest at 12 to 24 hours p.i. in calves 1 to 3, indicating negative energy balance. In calves 2 and 3, these NEFA elevations were accompanied by the highest levels of B-HB (ketosis). This is expected in animals going off feed as an early indicator of disease and is consistent with the clinical sign of ruminal hypomotility due to rumenitis in both calves. 27 Indirectly, these NEFA and B-HB elevations suggest that the calves had abnormal alimentary tract functions, resulting in an inability to obtain enough energy from the digestive tract and subsequent mobilization of fatty acids from fat storages followed by hepatic ketogenesis. 28 The ruminal hypomotility also explains the concomitant hypothermia found at 24 hours p.i. in calves 1 to 3, as normal digestive activity and ruminal movements are important sources of metabolic heat. Ruminal hypomotility would also explain the ruminal tympany observed at some clinical examinations, as normal ruminal motility is required for eructation, which prevents the excessive accumulation of gas in the dorsal sacs of the rumen.
Other serum biochemical abnormalities found in the A. atromaculatus-inoculated calves were less informative and likely result of terminal imbalances, particularly in samples obtained from severely affected animals immediately before euthanasia. These included terminal hyperphosphatemia, hypermagnesemia, and increased uremia in calf 3 of experiment 1 and the calf of experiment 2, with concomitant slightly increased aspartate aminotransferase activity and marked hypoglycemia in the latter.
The main pathological finding in all 4 calves was extensive, acute necro-suppurative omaso-reticulo-rumenitis (including the formation of vesicles and pustules), which explained the rapid clinical deterioration and/or death in all calves. The pathogenesis of these lesions in the context of A. atromaculatus-associated disease is uncertain at this stage. Similar lesions can result from a variety of causes, including (but not limited to) infectious agents (eg, bovine viral diarrhea virus, bluetongue virus) and toxic diseases (see below). Acute ruminal acidosis, caused by the ingestion of large amounts of highly fermentable carbohydrates resulting in a ruminal pH below 5, 21 is also characterized by similar lesions. We ruled out this condition because the calves were not being fed rations with high content of highly fermentable carbohydrates, and more importantly the pH of the rumen contents assessed postmortem was above the critical value in all calves.
Toxic diseases that are characterized by similar lesions in the forestomachs include intoxications with the plant Baccharis coridifolia, 25 to which the cattle in our study were not exposed, trichothecenes, 20 and lead arsenate. 24 For this reason, we tested the beetles for the trichothecene T-2 toxin and for heavy metals (including lead and arsenic), which were not detected. In addition, in these diseases there are usually concurrent lesions such as necrosis of lymphoid organs, liver, and renal tubules that were not seen in any of the calves in this study. Given that Zn toxicity is also characterized by severe gastrointestinal lesions, 1 we evaluated the concentrations of Zn in both the insects and the serum of the inoculated calves, which were at levels not considered problematic. In addition, pancreatic necrosis which is a hallmark lesion of Zn toxicity was not observed in the inoculated calves.
On the presumption that A. atromaculatus might contain toxic compounds, we conducted extensive targeted and nontargeted toxicological testing. The targeted analysis included toxins produced by beetles known to be harmful to other animals, such as cantharidin, produced by blister beetles (Epicauta spp., Mylabris spp.),7,15 and batrachotoxin, a cardio and neurotoxic alkaloid produced by beetles of the family Melyridae. 12 The targeted approach also included various mycotoxins, metals, and selenium. The nontargeted approaches included over 200 organic and inorganic compounds such as pesticides, drugs, environmental toxicants, alkaloids, and other natural products. Despite extensive testing, we were unable to identify a putative toxin responsible for the clinicopathological effects seen in the inoculated cattle.
The inoculum of experiment 1 tested positive for the pyrrolizidine alkaloid intermedine. This is a phytochemical that the insects might have obtained from the plants they were feeding on when collected. Pyrrolizidine alkaloids are not expected to cause the severe acute damage observed in the forestomachs of the inoculated calves. Because most pyrrolizidine alkaloids are metabolically activated in the liver, they mainly cause liver damage,10,32 which was not observed in the inoculated calves. Furthermore, the inoculum of experiment 2 did not contain intermedine nor other pyrrolizidine alkaloids and was equally fatal to the inoculated calf despite being administered at the lowest dose.
Severe fibrinous (pseudomembranous) enteritis was described in field cases of A. atromaculatus-associated disease in cattle, 13 and in experimental cases in sheep. 16 In these reports, the time elapsed between ingestion of the beetles and the onset of clinical signs and death ranged from several days to 2 weeks, as opposed to less than or equal to 2 days in our study. In this context of rapid disease progression, we did not find pseudomembranous enteritis upon necropsy of any of the calves, although calf 2 had grossly visible multifocal petechiae in the mucosa of the jejunum that was histologically consistent with peracute fibrinohemorrhagic enteritis, and calf 4 had superficial exfoliative colitis. Interestingly, in these calves, the histological examination revealed foreign bodies consistent with chitinous insect fragments embedded in the damaged mucosa of the jejunum (Fig. 4f–h) and colon. Based on this unusual finding, we speculate that sharp penetrating insect micro-fragments (Fig. 5a–c) might cause primary physical damage to the mucosa (ie, lining enterocytes and microvasculature of the underlying lamina propria), resulting in hemorrhage and fibrin extravasation. Expansion of this primary lesion could occur due to secondary complications such as bacterial invasion or exacerbated immune response to insect fragments, which in turn could result in severe fibrinosuppurative (pseudomembranous) enteritis in animals affected for several days, as described in cattle and sheep.13,16 In some of the spontaneous cases recently reported in cattle, similar insect micro-fragments were seen embedded in the mucosa of the intestine and surrounded by severe fibrinosuppurative inflammation (J.A. García, personal observation). A similar pathogenic mechanism mediated by physical damage has been proposed for crystal-containing drugs, with crystalline material possibly acting as a trigger of necro-inflammatory lesions in the intestinal tract of humans, 18 in cases of gastritis in cats, and gastroenteritis and endometritis in mares associated with penetrating chitinous setae (“hairs”) present in the exoskeleton of caterpillars,29,30 and in intoxications by plants containing insoluble (calcium) oxalate needle-like crystals (raphides). 14 Other mechanisms not considered here might be involved.
Histologically, the lesions in the small intestines of calves 1, 3, and 4 were mild and resembled those caused by gastroenteric pathogenic viruses. Some of them such as villus blunting and necrosis of enterocytes lining the tips of villi are compatible with those caused by RVA in neonate calves. 31 We cannot rule out a contribution of this virus to the mild infrequent small intestinal lesions in these 3 animals, although the quantified viral loads were very low and most likely subclinical in calves of this age. The log10 viral loads detected in calves 1, 3, and 4 (3.9–4.5) were below the log10 viral load cutoff (9.2) associated with RVA causality in diarrheic neonate calves. 6 In addition, the calf with more severe small intestinal lesions (calf 2) was negative for RVA, which further supports that RVA infection alone does not explain all the enteric lesions seen in these calves.
While the experiments we conducted provide a general framework of the clinical and pathological picture of A. atromaculatus-associated disease in cattle, they may not necessarily reflect the natural disease. For instance, under our experimental circumstances, the rapid administration of ground A. atromaculatus suspended in water by esophageal tubing probably resulted in quick availability of the causative agent in the ruminal content, which entered in contact with the mucosa of the forestomachs causing rapid damage. In grazing scenarios, the ingestion of beetles would occur at lower doses over a more prolonged period, with more delayed release of smaller amounts of the causative agent as mastication, rumination, and digestion of the beetles take place. These factors may help explain some of the variability seen in the clinical signs and lesions in spontaneous versus experimental cases, such as a more protracted clinical course, a higher frequency of diarrhea, and the development of grossly visible pseudomembranous enteritis. Additional studies are required to assess the effects of feeding doses less than 2.5 g/kg BW and multiple repeated versus single point exposures, including potential subclinical effects (ie, on weight gain and milk production).
We conclude that A. atromaculatus is lethal to calves at single oral doses of 2.5 to 15.0 g/kg BW, the minimum lethal dose probably being below 2.5 g/kg BW. The experimental disease is characterized clinically by acute anorexia, prolonged recumbency, reluctance to move, apathy, depression, ruminal hypomotility and tympany, hypothermia, bruxism with frothing at the mouth, and mucoid diarrhea progressing to death within 38 to 48 hours p.i. Severe acute vesiculopustular necrotizing omaso-reticulo-rumenitis, fibrinohemorrhagic enteritis, and exfoliative colitis are the main pathological findings. While A. atromaculatus might contain a gastrointestinal toxin, no causative chemicals have been identified by toxicological testing. Other mechanisms such as direct physical damage, or pathogen- or immune-mediated damage caused by insect fragments on the alimentary tract seem plausible, although further studies are needed to elucidate the mechanisms by which A. atromaculatus induces gastrointestinal disease.
Supplemental Material
sj-xlsx-1-vet-10.1177_03009858241231557 – Supplemental material for Experimental oral administration of pollen beetle (Astylus atromaculatus) to cattle results in an acute lethal gastrointestinal disease
Supplemental material, sj-xlsx-1-vet-10.1177_03009858241231557 for Experimental oral administration of pollen beetle (Astylus atromaculatus) to cattle results in an acute lethal gastrointestinal disease by Federico Giannitti, Mizael Machado, Caroline da Silva Silveira, Ximena Cibils-Stewart, Nicolás Baráibar, Cintia R. R. Queiroz-Machado, Robert H. Poppenga, Alejo Menchaca, Francisco A. Uzal, Juan A. García, Carolina Matto, Fernando Dutra, Gretel Ruprechter, Darío Caffarena and Anderson Saravia in Veterinary Pathology
Footnotes
Acknowledgements
We thank Juan Pedro Posse, Julio Frugoni and Franco Rodríguez from INIA Tacuarembó; Mabel Pessio, Pablo Calistro, Alejandro Mendoza, Matías Dorsch, Marcelo Pla, Eduardo Vidal, Santiago Fariña, Yisell Perdomo, Marina Berón and María Laura Casaux from INIA La Estanzuela; Guilherme Figuereido and Amanda de Moura from Universidade Federal da Bahia; Laura Nogueira and Karen Freitas from Univeridade Federal de Pelotas; Matías Castells and Leticia Maya from UdelaR; and Jim Langston from CAHFS. FG, MM, CSS, XCS, AM, CM, FD, GR, and DC are members of the “Sistema Nacional de Investigadores” (SNI) of the Uruguayan “Agencia Nacional de Investigación e Innovación” (ANII).
Author Contributions
F.G. conceived the study; F.G., M.M., C.d.S.S., A.M., and D.C. designed the experiments; F.G., C.d.S.S., D.C., and A.S. performed the experiments; F.G., M.M., X.C.-S., N.B., C.R.R.Q.-M. and A.S. collected the insects; X.C.-S. conducted the entomological identification; F.G., N.B., and A.S. obtained the photographs of the pastures and insects; G.R. and A.S. conducted biochemical and hematological analyses; F.G., M.M., C.R.R.Q.-M, D.C., and A.S. collected clinical data and performed the necropsies; F.G. performed the histopathologic examinations and obtained microphotographs; R.H.P. conducted the toxicological analyses and wrote corresponding materials and methods section; J.A.G., C.M., F.D., and F.A.U. helped with the interpretation of the pathological findings and provided valuable insights based on their experience with natural cases of this disease; F.G. wrote the first draft of the manuscript; all authors reviewed, edited, and approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by grant PL_27 from the “Instituto Nacional de Investigación Agropecuaria” (INIA), Uruguay.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
