Abstract

Clinical History
A 1-year-old Simmental cow was inspected during routine passive disease surveillance at the slaughterhouse due to the presence of a large mass in the right mandibular area that developed in about 15 days. This cow was born and reared on a 6 hectares organic milk farm in a lagoon area of Italy. On this farm, 2-month-old weaned calves were raised in outdoor paddocks with electrified fences and then were moved into sheds at 1 year of age. The vaccination plan included a commercial vaccine against Histophilus somni and Mannheimia haemolytica leukotoxin.
On clinical examination, the mass consisted of a rounded, 30 cm diameter, cold, solid, subcutaneous lesion. It extended from the base of the ear, laterally and ventro-medially to the mandible, up to the intermandibular space and contralateral mandible, and was covered by an intact and adherent skin (Fig. 1a). At the post-mortem examination, the mass weighed 14 kg and was easily dissected from the surrounding soft tissues with no involvement of the mandibular bone or the oral cavity. The mass and the mandible were submitted to the Istituto Zooprofilattico Sperimentale delle Venezie for further diagnostic investigations.
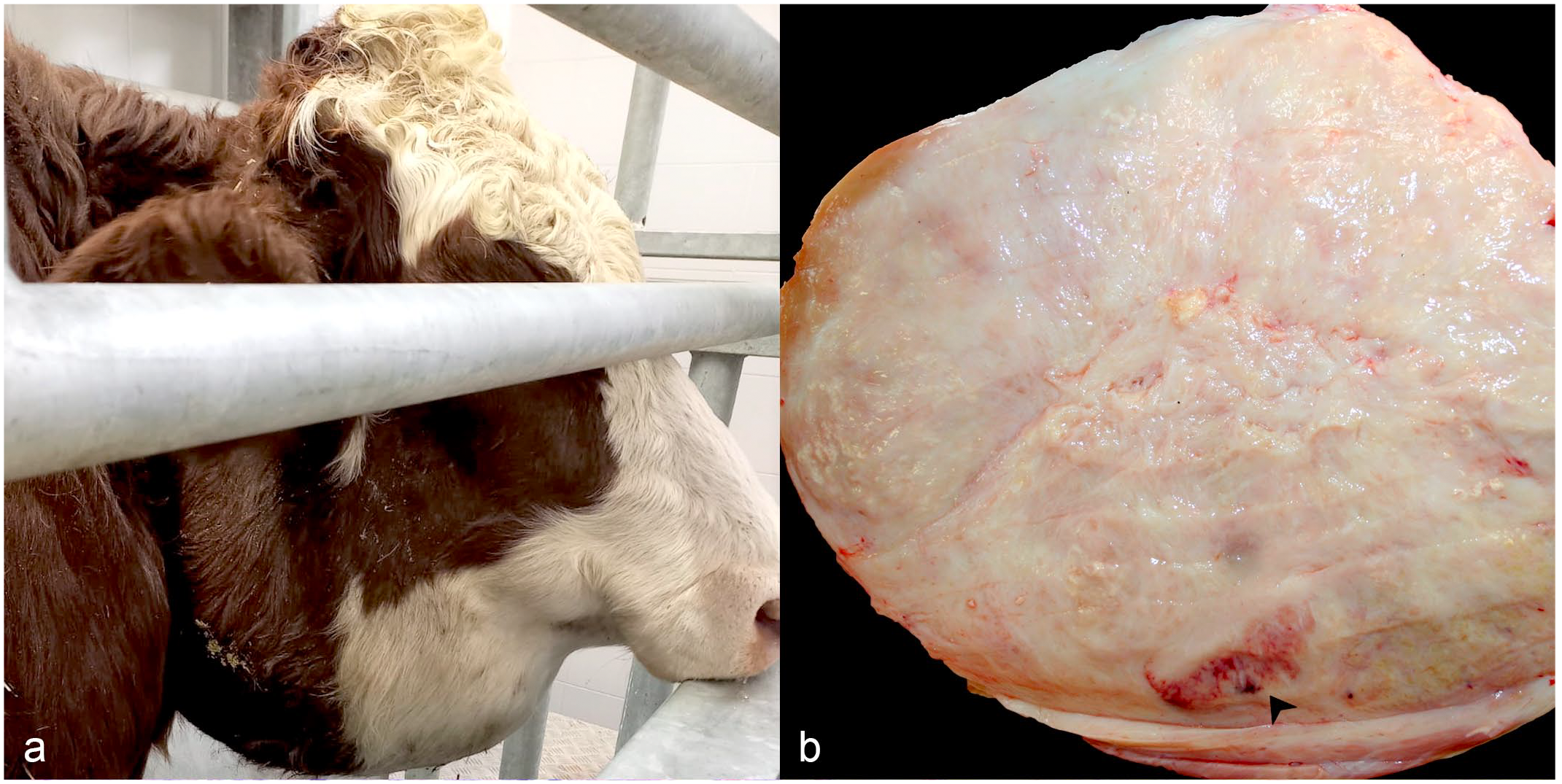
Mandibular mass, skin, cow. (a) Expansion of the right mandibular region by an approximately 30 cm diameter, round, ventrolateral mass. (b) Capsulated, homogenous, white, and firm mass with a maximum diameter of 24 cm. A hyperplastic/reactive lymph node is at the margin of the mass (arrowhead).
Gross Findings
On cross-section, the mass was entirely composed of firm to hard gray tissue intermingled with irregularly arranged fibrous bands and delineated by a thick peripheral fibrous capsule. Within the lesion, there were multifocal to coalescing, serpiginous, or irregularly circular, white bands of granular tissue, frequently associated with gritty consistency (calcium salts deposition). Multiple, swollen, hyperplastic/reactive lymph nodes were adjacent to the mass (Fig. 1b). The lateral, ventral, and medial aspects of the right mandibular corpus were homogenous. From the medial aspect of the corpus, residual portions of the mass were easily dissected from the underlying bone that was characterized by a smooth surface. Teeth displayed no alterations in position, number, or shape.
Differential Diagnoses
Based on the clinical and macroscopic findings, the lesion was suggestive of a chronic and fibrosing pyogranulomatous cellulitis/lymphadenitis. As the tongue was not involved, an atypical or cutaneous actinobacillosis was considered as a possible differential diagnosis. However, the absence of skin ulcers and lesions consistent with lymphangitis and the solid surface of the lesion were not suggestive of a form of atypical actinobacillosis. 5 Other bacteria-induced pyogranulomatous diseases, such as those caused by Trueperella pyogenes, Nocardia spp., or Rhodococcus equi, as well as zygomycotic lymphadenitis were considered as possible differentials; whereas, given the homogenous cut surface and the absence of grossly evident necrosis, foreign body reaction and necrobacillosis were considered less likely, respectively. 8 Finally, the location of the lesion could also be compatible with a sialocele, a periesophageal abscess/hematoma, and actinomycosis. Based on the solid consistency of the lesion, the fibrotic appearance of the cut surface, and because it spared the mandibular bone for the latter, these options were considered unlikely.
Microscopic Findings
On histologic examination using routine hematoxylin and eosin stain, the mass consisted of an abundant fibrosclerosing reaction that partially involved the peripheral lymph nodes, skeletal muscles fibers, and salivary glands. Sparsely distributed within the mass, there were numerous pyogranulomas (up to 300 µm in diameter) characterized by a central core mainly composed of degenerated neutrophils admixed with macrophages and eosinophils (Fig. 2a). The pyogranulomas contained occasional colonies of basophilic, 1 × 2 µm, gram-negative coccobacilli surrounded by eosinophilic, radiating, club-like material (Splendore-Hoeppli material) (Fig. 2a, b). Additional histochemical stains performed toward the detection of other possible infectious agents (Grocott methenamine silver stain for fungi and Ziehl-Neelsen stain for acid-fast bacilli) were negative. The surrounding tissue showed hypertrophied and hyperplastic fibroblasts with multifocal dystrophic mineralization of the collagen fibers and dystrophic osseous matrix deposition (Fig. 2a). Mild-to-moderate lymphoplasmacytic interstitial infiltration was also detected diffusely within the mass.
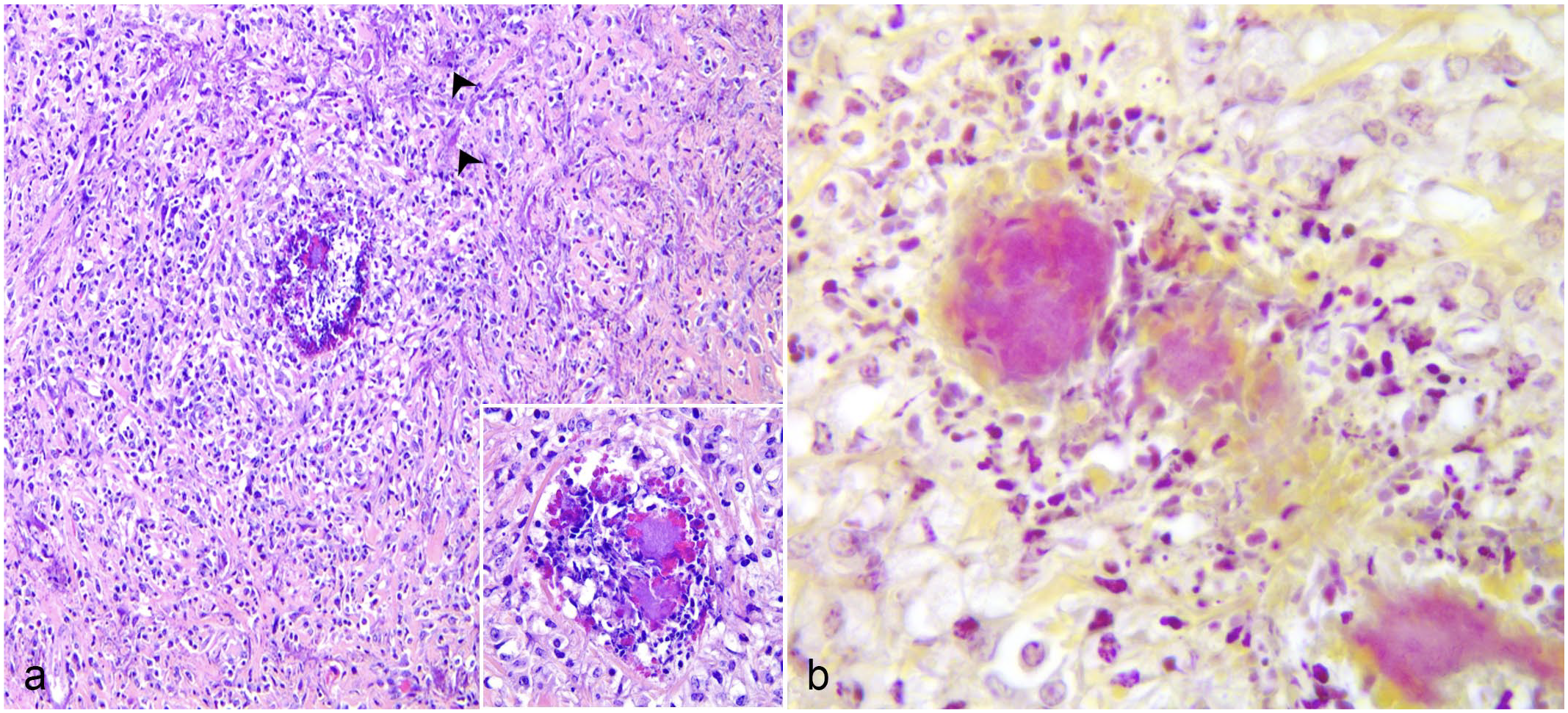
Mandibular mass, skin, cow. (a) Multifocal pyogranulomas with Splendore-Hoeppli material and intralesional coccobacillary colonies (inset) surrounded by fibrosis with multifocal dystrophic mineralization of the collagen fibers (arrowheads). Hematoxylin and eosin. (b) Gram-negative colonies of coccobacilli in pyogranulomas. Gram stain.
Peripheral lymph nodes exhibited medullary plasmacytosis, sinus histiocytosis, multifocal hemorrhages, and mild interstitial septa fibrosis.
Further Investigations and Diagnosis
Swabs from the inner aspect of the mass and the marginal and peripheral lymph nodes were submitted for routine aerobic, microaerophilic, and anaerobic bacteriological procedures. From the mass, a pure culture of small, gray, sticky, and viscous colonies with a strong mucinous odor and incomplete hemolysis were isolated on Columbia agar base with 5% sheep blood after 24 hours of incubation in aerobiosis. The colonies were composed of rod-shaped, gram-negative pleomorphic cells, occurring individually, in pairs, or short chains and were weakly positive on the oxidase test. A presumptive identification of Pasteurella/Mannheimia spp. was considered. The lymph nodes culture yielded no growth of Pasteurella/Mannheimia spp. colonies.
Bacterial identification was assigned by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI Microflex Biotyper LT; Bruker Daltonics GmbH, Bremen, Germany) with a score 2.23 for Mannheimia granulomatis and 2.03 for M. haemolytica (genus identification certain, species identification probable). The MALDI Biotyper library version 3.0 and software version 3.1 were used for bacterial identification.
Species taxonomy was further confirmed by 16S rRNA sequencing. Amplification of a specific segment of the bacterial 16S rRNA gene was performed with the following primers: 16SBAq F 5’-CAC GGY CCA GAC TCC TAC-3’ and 16SBAq R 5’-CGA TTA CTA GCG AYT CCR AC-3’ (Eurofins Genomics Europe Shared Services GmbH, Ebersberg, Germany). Sanger sequencing of the polymerase chain reaction (PCR) product revealed an identity of 99.41% with M. granulomatis (Genbank accession number: NR_115215.1).
Discussion
The location of the lesion and the incidence of the different pathological entities that could potentially affect the cervical-mandibular region in the bovine could lead to a presumptive diagnosis of actinomycosis or atypical/cutaneous actinobacillosis. A proper clinical examination in association with the macroscopic findings and the clinical history of a rapid development of the mass were fundamental to extend the potential list of differential diagnoses toward less common microorganisms causing chronic fibrosing pyogranulomas. Bacterial culture and identification of the causative organism together with the histological findings were therefore necessary to properly identify the etiologic agent of the lesion.
M. granulomatis (class Gammaproteobacteria, family Pasteurellaceae) is a gram-negative, nonmotile, facultative anaerobic rod that was first recognized as a new taxon in the early nineties and is able to cause disease in cattle, deer, and hares. 9 Traditionally, M. granulomatis is known as the cause of the so-called “lechiguana” or focal proliferative fibrogranulomatous panniculitis of cattle, a peculiar disease typically reported in Brazil, where a causative relationship with Dermatobia hominis warbles infection has been postulated. 10 Moreover, a similar lesion has been reported in a 4-month-old male Japanese black calf in Japan. 6 Other diseases associated with M. granulomatis are infrequent in cattle and include oral abscesses, suppurative bronchopneumonia, and mastitis.3,4
Similar to the case described here, focal proliferative fibrogranulomatous panniculitis is characterized by 1 or 2 hard subcutaneous swellings, involving mainly the prescapular region or, less frequently, the dorsolumbar/gluteal region or the abdominal wall. The overlying skin is usually not affected, whereas, regional lymphadenomegaly is a consistently associated finding. Lesions can grow rapidly and potentially lead to death if untreated. 10
The histopathological changes in this case (Fig. 2a, b) were compatible with the features of focal proliferative fibrogranulomatous panniculitis as they are reported to be characterized by a well-vascularized and frequently mineralized fibrous tissue embedding multiple eosinophilic microabscesses and granulomas with Splendore-Hoeppli phenomenon and intralesional gram-negative bacilli or coccobacilli. In addition, eosinophilic lymphangitis is frequently observed in these cases, especially in early lesions. Similar lesions can also occur in regional lymph nodes.1,2,10
Interestingly, a lesion reminiscent of focal proliferative fibrogranulomatous panniculitis has also been described sporadically in white-tailed deer (Odocoileus virginianus) and mule deer (Odocoileus hemionus) in the USA since 2002. Typically, the lesion causes a severe swelling of the muzzle and upper lips due to granulomatous cellulitis consistently associated with intralesional bacteria identified as M. granulomatis. Likewise, as in the present case, lymphangitis is not evident. 7 In Europe, M. granulomatis has been isolated in lesions in roe deer (Capreolus capreolus) including glossitis, stomatitis, and cervical phlegmon/abscess, and in an episode of glossitis in a fallow deer (Dama dama). 7 With regard to our case, given the location of the farm in a nature park, the proximity to wildlife might be considered as a possible source of exposure to M. granulomatis infection but cannot be confirmed.
In conclusion, M. granulomatis should be included in the list of differential diagnoses of the bacteria and fungi responsible of local pyogranulomatous cellulitis and regional lymphadenitis in cattle.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
