Abstract
The reemergence of the highly pathogenic avian influenza virus (HPAIV) subtype H5N1 in the United Kingdom in 2021–2022 has caused unprecedented epizootic events in wild birds and poultry. During the summer of 2022, there was a shift in virus transmission dynamics resulting in increased HPAIV infection in seabirds, and consequently, a profound impact on seabird populations. To understand the pathological impact of HPAIV in seabirds, we evaluated the virus antigen distribution and associated pathological changes in the tissues of great skua (Stercorarius skua, n = 8), long-tailed skua (Stercorarius longicaudus, n = 1), European herring gull (Larus argentatus, n = 5), and black-headed gull (Chroicocephalus ridibundus, n = 4), which succumbed to natural infection of HPAIV during the summer of 2022. Cases were collected from Shetland, including Scatness (mainland), No Ness (mainland), Clumlie (mainland), Hermaness (island), Fair Isle (island), Noss (island), and the West Midlands, South East, and South West of England. Grossly, gizzard ulceration was observed in one great skua and pancreatic necrosis was observed in 4 herring gulls, with intralesional viral antigen detected subsequently. Microscopical analysis revealed neuro-, pneumo-, lymphoid-, and cardiomyotropism of HPAIV H5N1, with the most common virus-associated pathological changes being pancreatic and splenic necrosis. Examination of the reproductive tract of the great skua revealed HPAIV-associated oophoritis and salpingitis, and virus replication within the oviductal epithelium. The emergence of HPAIV in seabirds Stercorariidae and Laridae, particularly during summer 2022, has challenged the dogma of HPAIV dynamics, posing a significant threat to wild bird life with potential implications for the reproductive performance of seabirds of conservation importance.
Keywords
Highly pathogenic avian influenza virus (HPAIV) H5N1 clade 2.3.4.4b Goose/Guangdong (Gs/Gd) lineage has reemerged in Europe during the 2020–2021, 2021–2022, and 2022–2023 seasons (defined as start of each October) and has brought about a series of epizootic events in poultry and wild birds. The reemergence of HPAIV H5N1 clade 2.3.4.4.b in Europe and the United Kingdom during 2021–2022 has also contributed to the trans-Atlantic dissemination of virus into North America likely mediated though migratory wild birds. 13
Conventionally, it is understood that Anseriformes are the carrier for HPAIV during the winter period in Western Europe. However, during the 2021–2022 HPAIV season in the United Kingdom, there was a shift in infection from Anseriformes predominating in the colder months to a series of explosive outbreaks in seabird species across the northern coast of Scotland during the summer. 5 During the summer of 2021, infection with H5N1 was detected in great skuas (Stercorarius skua) 7 but those events, alongside sporadic small-scale outbreaks across northern Europe were the only cases of H5N1 reported during the summer months. In contrast, during the summer of 2022, infection in great skuas was detected several months earlier than during 2021 and was followed by extensive outbreaks in a number of shorebird species (Order Charadriiformes). 16 High mortality events in seabirds, including northern gannet, great skua, and several species of gulls, were observed.5,16 Seabirds from the Laridae family have been previously associated with infection with low pathogenic avian influenza virus (LPAIV).18,21,25,35,57 However, a recent experimental model demonstrated that previous exposure of the European herring gulls (Larus argentatus) with LPAIV H5N1 (non-Gs/Gd) or H13N6 only confers partial protection to a subsequent HPAIV H5N8 clade 2.3.4.4b challenge. 53
Prior to the unusual increase in cases during the summer of 2022, HPAIV-associated disease in the Laridae has been sporadically reported in East Asia and Europe, often in small numbers.1,2,13,15,37,39,41,43 More recently, there has been an increased detection of HPAI-positive seabirds or Charadriiformes16,30 and increased mortality events in seabirds associated with HPAIV infection reported in the United Kingdom, Europe, and North America.3,6,7,50 The data collected through the avian influenza (AI) wild bird passive surveillance scheme in Great Britain, whereby “found dead” wild birds are reported by the general public and submitted for testing, indicated a rise in HPAIV H5Nx positive birds within the Laridae family from 1.3% during the 2020–2021 season to 15% within the 2021–2022 season. Furthermore, the number of HPAIV-positive Laridae birds, particularly the black-headed gull (Chroicocephalus ridibundus), in the United Kingdom and Europe remained high over spring-summer of 2023 with reports of significant mortality events at breeding colonies. 3
One of the hypotheses for the enzootic transmission of HPAIV in wild birds in Europe is the maintenance in wild birds during summer in Northern Europe. 47 Previously, Anseriformes were thought to be responsible for virus transmission given potential virus adaptation in the host.11,12 However, the expanded susceptibility of avian taxa to HPAIV and increased incidence of disease, such as in the seabirds also challenges the status quo and introduces further uncertainty on the transmission dynamics at both the local and global levels.
The present report aims to investigate the gross and histological lesions and to evaluate the distribution of influenza A virus antigen in the tissues of seabirds in the Stercorariidae and Laridae families that died due to natural HPAIV H5N1 infection. The species examined include the great skua, long-tailed skua (Stercorarius longicaudus), European herring gull, and black-headed gull.
Materials and Methods
Postmortem Examination
Carcasses received at Scotland’s Rural College, NatureScot, or Animal and Plant Health Agency (APHA) regional laboratories were frozen for transport and thawed for necropsy at APHA Weybridge (Supplemental Table S1). Postmortem examinations were conducted by FZXL, NF, and AN in a microbiological safety cabinet within the high containment facility certified for level 4 specific animal pathogen order. The herring gulls were obtained from wildlife rehabilitation centers (East Sussex and Cornwall) and were submitted for investigation as case reports. The black-headed gull carcasses were recovered from Birmingham, and the great skuas carcasses originated from colonies on Shetland, including Scatness (mainland), No Ness (mainland), Hermaness (island), Fair Isle (island), and Noss (island). The long-tailed skua carcass originated from Clumlie (mainland). These specimens were collected as part of the UK AI wild bird passive surveillance scheme. Oropharyngeal and cloacal swabs and tissues were tested to confirm infection status with HPAIV H5N1 by standard virological tests, as described previously. 38 Major organs, including pectoral sternal skin, pectoral skeletal muscle, heart, brain, spleen, kidney, nasal turbinate, trachea, lung, proventriculus, gizzard, liver, pancreas, duodenum, ovary, oviduct, and testis were fixed in 10% neutral-buffered formalin for microscopic evaluation.
Virological Investigation
Oropharyngeal and cloacal swabs were collected from the great skuas, herring gulls, and black-headed gulls. Individual tissues were sampled from the great skuas and long-tailed skua, while pooled tissues were collected from the herring gulls.
Oropharyngeal or cloacal swabs were placed into a microcentrifuge tube containing 1 ml of Leibovitz’s L-15 Medium (ThermoFisher Scientific) with antibiotics. Each tube was gently mixed for a minimum of 5 seconds to elute the material from the swab before being allowed to stand at room temperature for a minimum of 2 minutes. Tissue suspensions (approximately 10% v/v) were prepared in phosphate-buffered saline containing antibiotics. Nucleic acid was extracted from each swab or tissue suspension sample by an automated process using the KingFisher Flex (ThermoFisher Scientific) with the reagents from the MagMAX CORE Nucleic Acid Purification Kit (ThermoFisher Scientific).
All nucleic acid extractions were tested by a suite of 3 avian influenza virus (AIV) real-time reverse transcription polymerase chain reaction (RRT-PCR assays) for generic influenza A virus detection (M gene), 45 for specific detection of HPAIV H5 AIV, 29 and by an N1-specific RRT-PCR to confirm the presence of H5N1 AIV according to the procedure described by Payungporn et al 46 and adapted to the RRT-PCR chemistry at APHA by Slomka et al. 51 Samples producing a threshold cycle (Cq) value less than or equal to 36.0 were considered positive. All PCR amplifications were carried out in an AriaMx qPCR System (Agilent).
Histopathology and Immunohistochemistry
Formalin-fixed tissues were processed using routine histological methods into paraffin blocks. Tissues were sectioned at a 4 μm thickness and stained with hematoxylin and eosin for histological evaluation and immunohistochemical labeling using a mouse monoclonal IgG1 antibody against the nucleoprotein of influenza A virus (Statens Serum Institute, Denmark; HYB 340-05) at 1 in 4000 dilution for the detection of influenza viral antigen, as described previously. 42 The tissues were assessed on conventional light microscope for histopathology and scored as absent (–), minimal (+), mild (++), moderate (+++), or severe (++++). The abundance of virus antigens was scored as absent (–), rare (+), scattered (++), confluent (+++), or abundant (++++). Microscopic evaluations were performed by FZXL. 7 Detailed microscopic criteria are outlined in Supplemental Tables S2 and S3.
Results
History and Clinical Findings
The captive herring gulls from 2 sites in England, East Sussex and Cornwall, each containing approximately 100 gulls, were reported to have exhibited clinical signs, such as cyanotic heads, gasping, muscle twitching, diarrhea, and sudden deaths with mortality rates ranging between 20% and 50% (Supplemental Table S4). The carcasses of non-captive wild birds, including great skuas, long-tailed skuas, and black-headed gulls were retrieved after being found dead in the environment (Shetland and Birmingham). No clinical signs were recorded due to the lack of live observation.
Influenza Virus Detection
All birds tested positive for HPAIV H5N1 infection by RRT-PCR on oropharyngeal and cloacal swabs or tissues targeting M, HP H5, and N1 genes. No viral RNA for avian avulavirus was detected using RRT-PCR for L gene. The oropharyngeal and cloacal swabs from great skuas yielded Cq values ranging from 20 to 28 and from 22 to 33, respectively (Supplemental Table S1). Meanwhile, oropharyngeal and cloacal swabs from herring gulls showed Cq values ranging from 21 to 29 and from 25 to 37, respectively. Black-headed gulls had Cq values ranging from 22 to 29 for oropharyngeal swabs and from 19 to 31 for cloacal swabs. In cases where tissues were tested, the brain consistently yielded high viral loads with low Cq values ranging from 14 to 25 for great skuas and from 15 to 20 for herring gulls (Supplemental Tables S1 and S5). Other tissues, including the trachea, lung, heart, liver, spleen, kidney, and intestine were positive on RRT-PCR. Where individual tissues were tested by RRT-PCR in the great skua, the respiratory tissues (trachea and lung) had lower Cq values compared to the intestines and kidneys (Supplemental Table S5). The oropharyngeal swabs also consistently yielded lower Cq values compared to the cloacal swabs. Similar trends were noted in the tissues and swabs from the herring gulls and the swabs from the black-headed gulls. Overall molecular virological testing confirmed virus shedding from the oropharyngeal and cloacal orifices, with viral RNA being more abundant from the respiratory tissues and demonstrated multisystemic dissemination of HPAIV within the birds.
Postmortem Findings
All submitted birds were in fair to good body condition. On necropsy, the great skuas (n = 8, 6 females and 2 males) were moderately autolysed. Only one of the birds had multifocal, approximately 1–2 mm in diameter, dark red ulcers on the gizzard close to the proventricular-gizzard junction (Fig. 1a). The long-tailed skua (n = 1, male) was moderately autolysed and otherwise unremarkable. For the herring gulls (n = 5, 2 males and 3 sex not determined) were mildly autolysed. Postmortem examination findings included multifocal, faint tan patches within the pancreatic parenchyma (n = 5; Fig. 1b), suggestive of necrosis; mild splenomegaly (n = 3); and intestinal nematodiasis (n = 1). Four black-headed gulls (2 males and 2 females) were examined, but due to severe autolysis, the gross interpretation was limited.
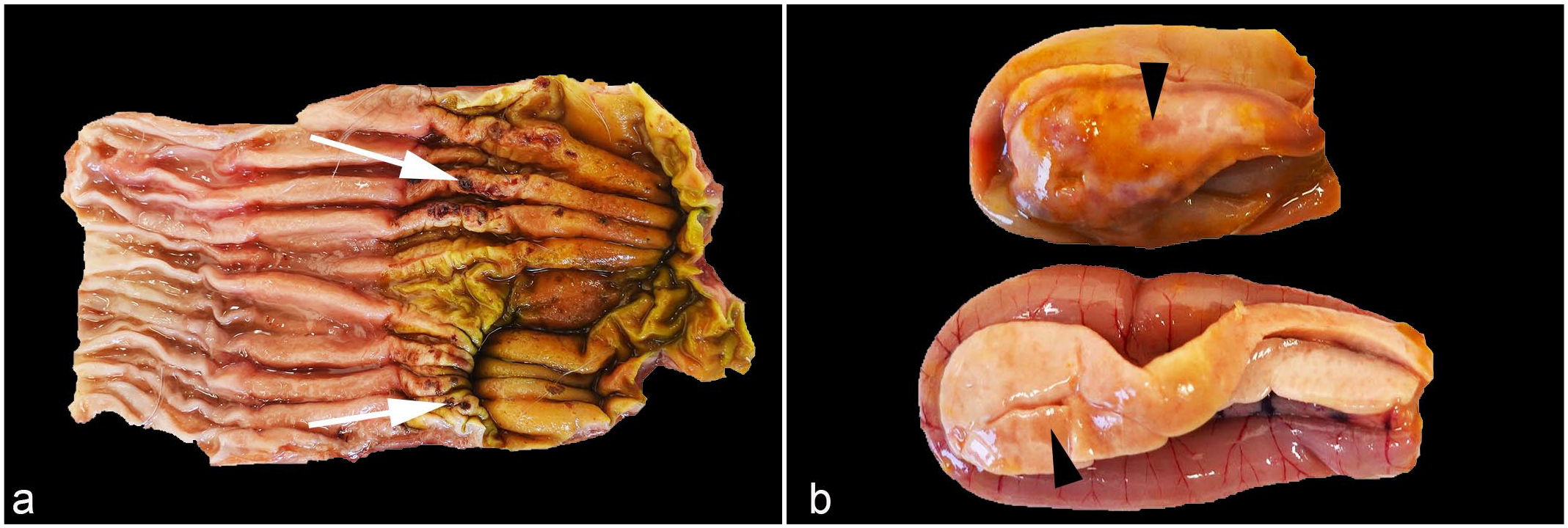
Gross lesions of highly pathogenic avian influenza virus H5N1 infected seabirds. (a) Gizzard, great skua (Stercorarius skua). Multifocal dark red depressions (white arrow) are present on the mucosa of the gizzard close to the proventricular-gizzard junction. (b) Pancreas, European herring gull (Larus argentatus). Multifocal to coalescing pale tan areas in the pancreas are consistent with necrosis (arrowheads).
Histopathology and Viral Immunohistochemistry
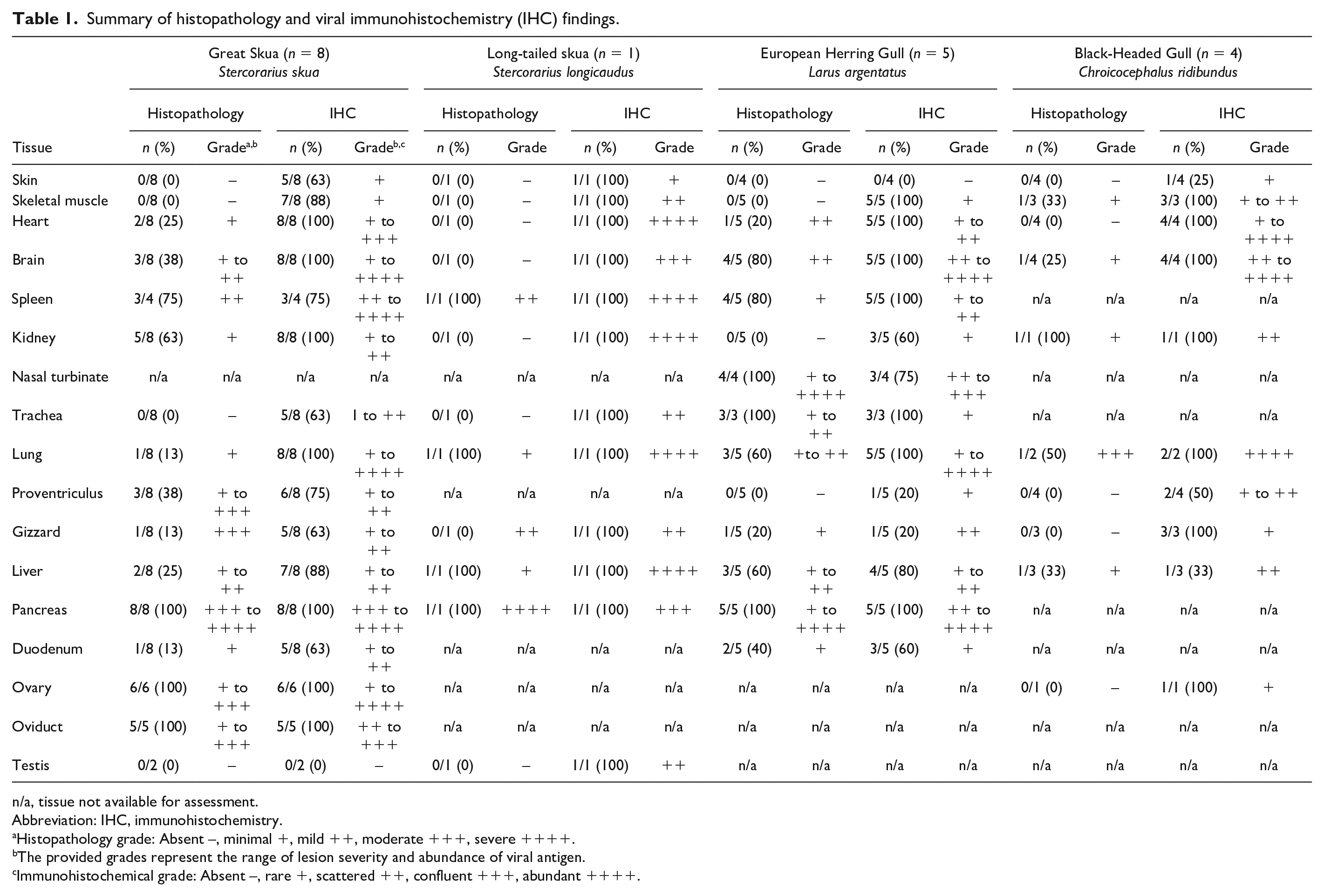
In the great skua, virus antigen was consistently detected in the heart, brain, kidney, lung, and pancreas of all birds examined (Table 1, Supplemental Table S6). The pancreas was particularly affected, with moderate to severe, multifocal to confluent areas of necrosis. These necrotic areas correlated with moderate to abundant distribution of virus antigens in all great skuas examined (Fig. 2a; n = 8/8). Correlative viral immunohistochemistry (IHC) and histology also revealed viral-associated myocardial necrosis (Fig. 2b, n = 2/8), splenic necrosis (Fig. 2c; n = 3/4), and renal tubular necrosis (Fig. 2d; n = 5/8), which were mild. In the gizzard of a great skua where ulceration was noted during the postmortem examination (Fig. 1a), histological and IHC examinations confirmed of the presence of viral protein in the mucosa (Fig. 3a) and glandular epithelium (Fig. 3b). The mucosal damage was extensive and was replaced with necrotic cellular debris, degenerated heterophils, and fibrin deposition (Fig. 3a, b). Similar proventricular mucosa damage was also observed histologically in other 2 great skuas where lesions were not observed grossly. Nevertheless, viral immunolabeling in the proventriculus (n = 6/8) and gizzard (n = 5/8) were more common than histopathological changes (n = 3/8, 1/8; respectively).
Summary of histopathology and viral immunohistochemistry (IHC) findings.
n/a, tissue not available for assessment.
Abbreviation: IHC, immunohistochemistry.
Histopathology grade: Absent –, minimal +, mild ++, moderate +++, severe ++++.
The provided grades represent the range of lesion severity and abundance of viral antigen.
Immunohistochemical grade: Absent –, rare +, scattered ++, confluent +++, abundant ++++.
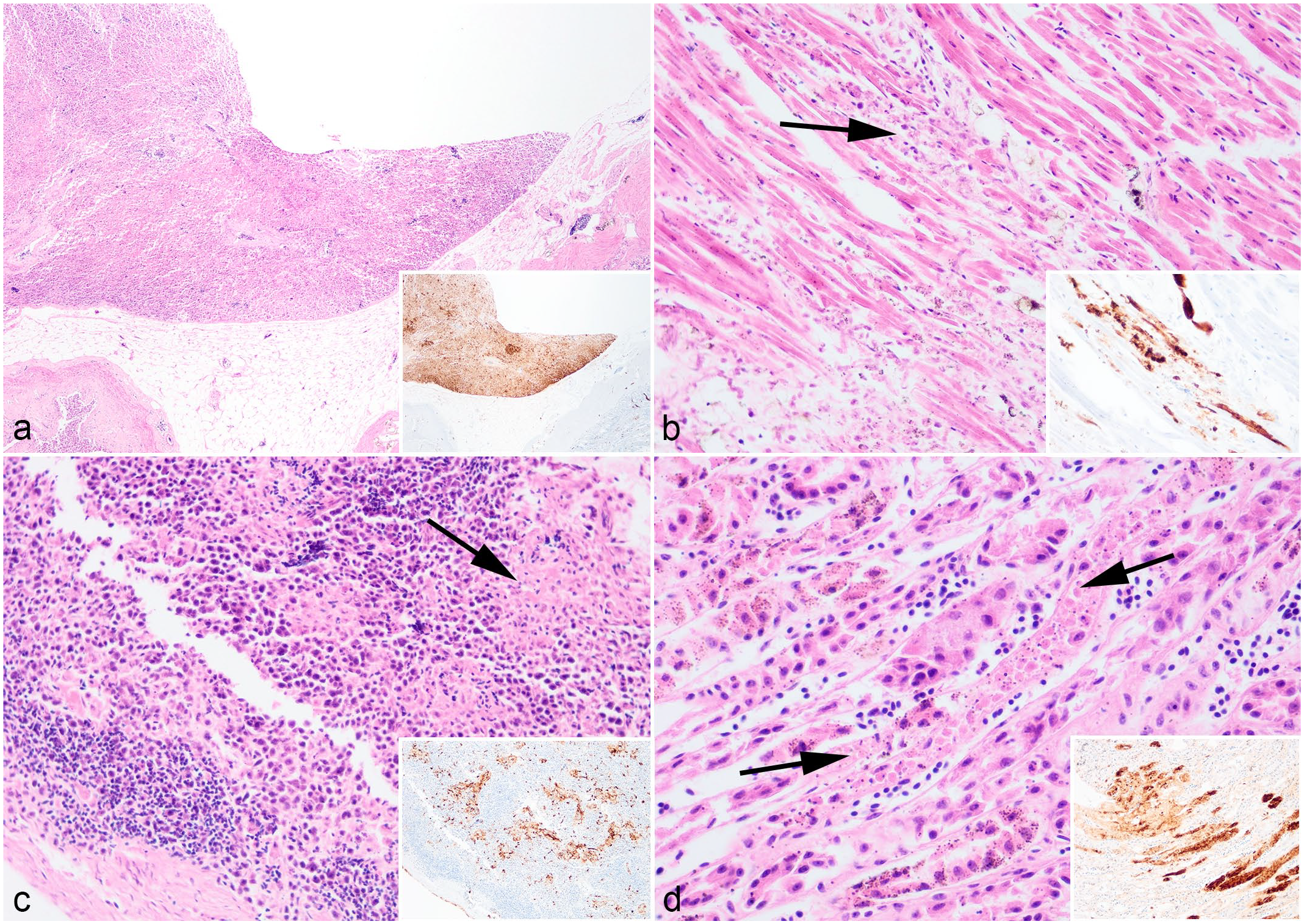
Microscopic findings of great skua (Stercorarius skua) infected with highly pathogenic avian influenza virus H5N1. Hematoxylin and eosin. (a) Pancreas. Severe, confluent, pancreatic necrosis. (b) Heart. Moderate, multifocal, myocardial necrosis. (c) Spleen. Mild, multifocal, splenic necrosis. (d) Kidney. Mild, multifocal, renal tubular necrosis. Arrows indicate area of necrosis. Insets (a–d): Colocalization of viral antigens with areas of necrosis. Influenza A nucleoprotein immunohistochemistry.
Microscopic findings of great skua (Stercorarius skua) infected with highly pathogenic avian influenza virus H5N1. Hematoxylin and eosin. (a and b) Gizzard. Moderate, focal, gizzard necrosis and fracturing of koilin (b, arrowhead), with evidence of mucosa epithelial degeneration (b, arrow), and deposition of cellular debris, degenerated heterophils, extravasated erythrocytes, and fibrin within the disrupted koilin layer. (c and d) Ovary. (c) Moderate, diffuse, oophoritis. (d) The theca interna and stroma are necrotic (arrow), and there is fibrin deposition within the mural typically around blood vessels (arrowhead). (e and f) Oviduct. (e) Moderate necrotizing salpingitis, with abundant intraluminal cellular debris. (f) The mucosa is eroded and infiltrated with heterophils and lymphocytes. Insets (a–f): Colocalization of viral antigens within areas of necrosis and intraluminal debris. Influenza A nucleoprotein immunohistochemistry.
Furthermore, virus antigens were detected in the ovaries (n = 6/6) and oviducts (n = 5/5) but not in the testis (n = 0/2). In the ovaries, there was confluent distribution of viral antigens (Fig. 3c), mainly in the theca interna and occasionally transmurally in the preovulatory follicles. Viral antigens were associated with necrosis within the tunica interna and blood vessels of the stroma, and the stromal wall was moderately to markedly expanded with lymphocytes, plasma cells, and fibrin deposits (Fig. 3d). In the oviduct, there was intraluminal cellular debris, mucosal ulceration, and heterophilic and lymphocytic infiltration of submucosa wall. Viral antigens were present in the intraluminal cellular debris (Fig. 3e), mucosa epithelium, and submucosal cells (Fig. 3f).
Only one long-tailed skua was examined, which had severe pancreatic necrosis, mild splenic and hepatic necrosis, and an abundance of viral antigens that were colocalized to these lesions (Table 1). Virus antigens were also detected in other organs where histopathological changes were absent, including the skin, skeletal muscle, heart, brain, kidney, trachea, gizzard, and testis.
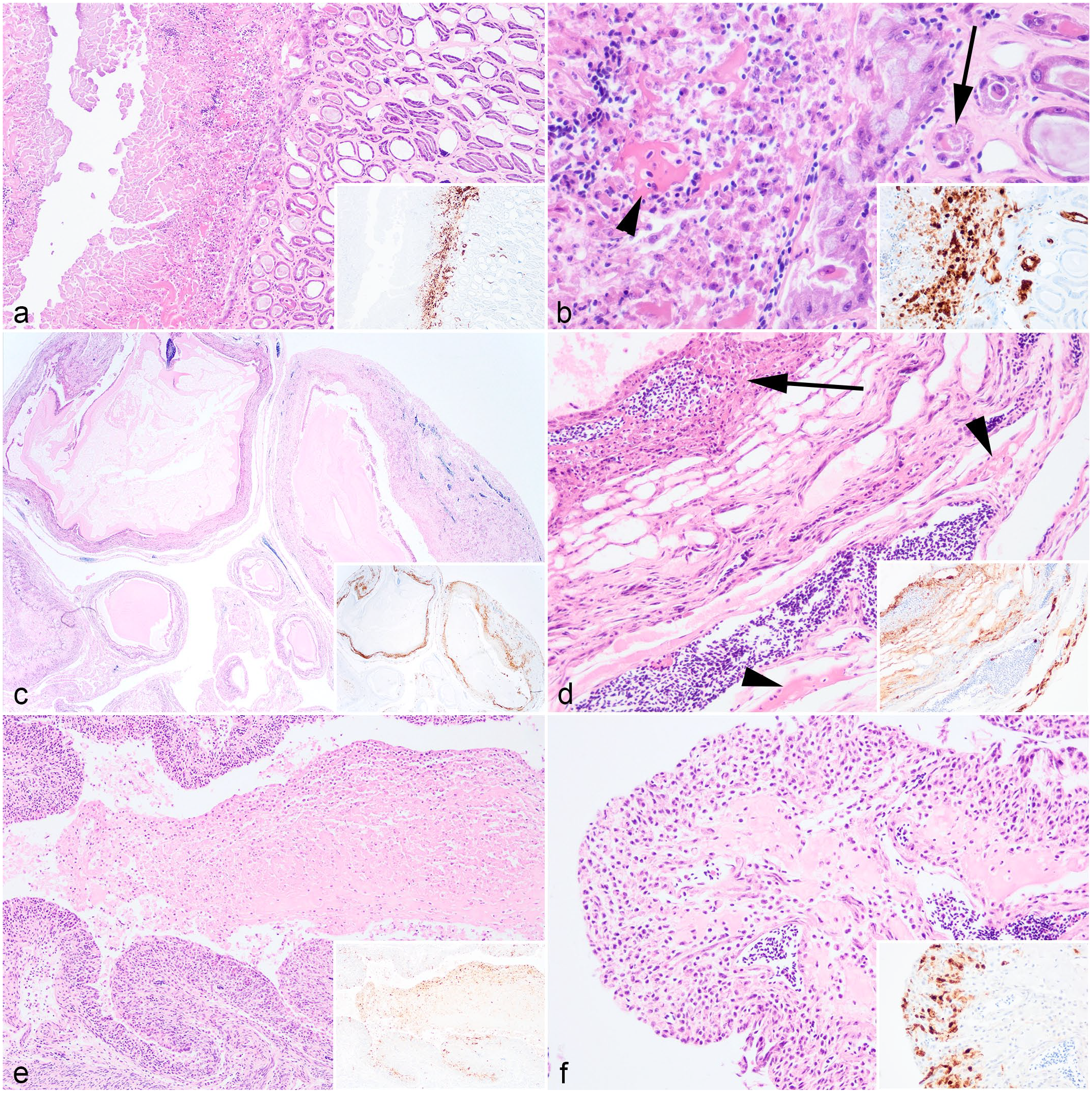
In the herring gulls, viral antigens were found in the pancreas (n = 5/5, Fig. 4a), spleen (n = 5/5, Fig. 4b), brain (n = 5/5, Fig. 4c), and lung (n = 5/5), and were consistently associated with histological lesions. Pancreatic necrosis was multifocal to confluent and with a range of minimal to severe changes (Fig. 4a). In the spleen, mild lymphoid depletion was associated with lymphoid necrosis (Fig. 4b). In the brain, there was mild neuronal necrosis and dispersed degenerated heterophils within the neuropil. In the cerebellum, there was occasional loss of Purkinje cells attributed to viral infection (Fig. 4c). Rhinitis ranged from mild changes, including scant heterophilic exudate with occasional intraepithelial heterophils, or in severe cases with abundant exudation, complete loss of mucosa with submucosa necrosis and fibrin deposition (Fig. 4d). In the lungs, there were mild to moderate air capillary necrosis (Fig. 4e) with occasional fibrin deposition in air capillary walls. A gizzard lesion was detected in one herring gull and was characterized by necrosis of the mucosa-submucosa and loss of koilin with virus antigens detected in the mucosa epithelium and lymphoid cells within the submucosa (Fig. 4f). Incidental findings included the presence of an intestinal cestode and proventricular nematodes in 2 herring gulls but were not associated with overt intestinal lesions.
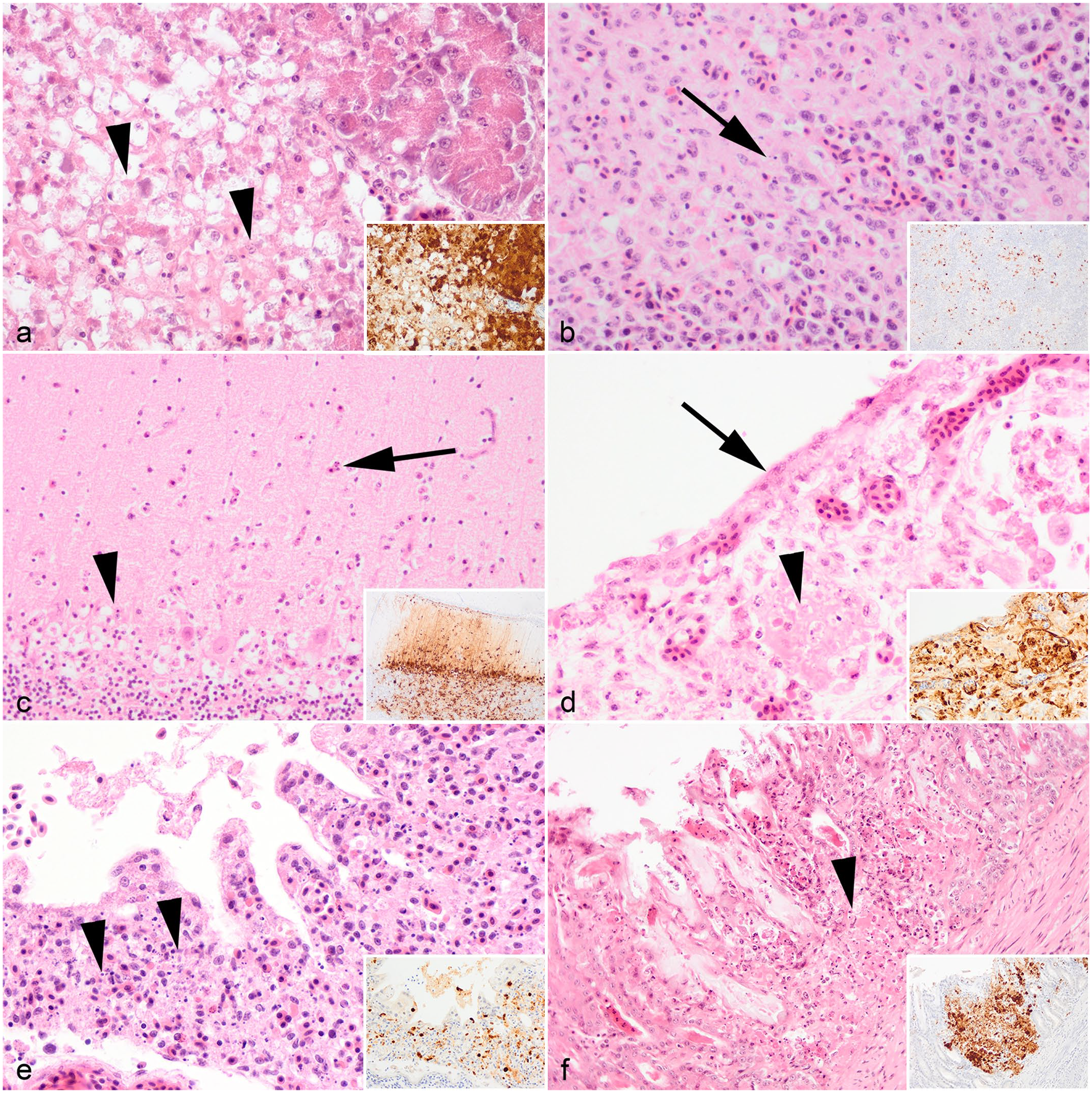
Microscopic findings of European herring gull (Larus argentatus) infected with highly pathogenic avian influenza virus H5N1. Hematoxylin and eosin. (a) Pancreas. Mild, multifocal, necrosis of the pancreatic acinar cells characterized by marked cytoplasmic swelling (arrowhead) and nuclear pyknosis. (b) Spleen. Mild, multifocal necrosis (arrow) of splenic white pulp. (c) Cerebellum. Minimal, multifocal, neuronal necrosis with scattered heterophilic infiltration of neuropil (arrow) and loss of Purkinje cells (arrowhead). (d) Nasal cavity. Severe, confluent, necrotizing rhinitis with extensive loss or attenuation of epithelial cells (arrow) and fibrin deposition within the submucosa (arrowhead). (e) Lung. Moderate, multifocal, air capillary necrosis characterized by expansion of the interstitium with karyorrhectic debris (arrowhead), lymphocytic infiltration, and hemorrhage. (f) Gizzard. Mild, multifocal, gizzard necrosis with disruption of the mucosa-submucosa, accumulation of karyorrhectic debris and fibrin within the submucosa (arrowhead), and loss of koilin. Insets (a–f): Colocalization of viral antigens with areas of necrosis. Influenza A nucleoprotein immunohistochemistry.
Four black-headed gulls were examined microscopically, but the histological interpretation was hindered by advance autolysis. Within the limits of tissue preservation, moderate air capillary necrosis was observed, and organs, such as the brain, kidney, and liver exhibited only minimal mononuclear inflammation. On the other hand, viral antigens were abundant in the brain, heart, and lung (Table 1). Ovary was sampled from one black-headed gull, and immunohistochemical labeling revealed rare viral antigens within the blood vessels of the ovarian stroma, but no overt histological changes were detected.
Discussion
During the 2020–2021 and 2021–2022 outbreaks of HPAIV in the United Kingdom, there has been an increased detection of HPAIV H5N1 in seabirds of the Stercorariidae and Laridae families.5,7,16 This investigation of naturally acquired HPAIV infection revealed that gross lesions were limited to pancreatic necrosis in the herring gull and gizzard ulceration in the great skua. The pancreatic changes were less conspicuous compared with those in Galliformes and Anseriformes and required immunohistochemical confirmation. Microscopic evaluation confirmed a multisystemic HPAIV infection, including neuro-, pneumo-, lymphoid-, and cardiomyotropism, which was likely contributory to the mortalities seen. In addition, acute reproductive damage in female great skua was noted. Overall, skua and gull birds are highly susceptible to developing lesions in multiple organ systems following HPAIV infection.
The most common and severe lesion in all birds examined was pancreatic necrosis associated with viral infection (except in the black-headed gulls where the pancreas was unavailable), followed by splenic necrosis and pneumonia (except in the long-tailed skua). Such lesions are similar to those reported from experimentally challenged common gulls (Larus canus), black-headed gulls, and herring gulls with preclade 2.3.4.4.b and 2.3.4.4b isolates of HPAIV H5N1,10,24,49,53 as well as report of naturally infected sandwich terns (Thalasseus sandvicensis) with the contemporaneous H5N1.49,50 The multisystemic infection and lesions observed in skua and gull birds, including vascular tropism, are similar to those typically seen in HPAIV-infected Galliformes poultry,20,40 emphasizing the susceptibility of skua and gull birds to the contemporaneously circulating HPAIV 2.3.4.4b H5N1.
Avian influenza viruses are known for the preferential binding to the α-2,3 sialic acid residues. 14 Based on lectin histochemistry on other gull species, including American herring gulls (Larus smithsonianus), laughing gulls (Leucophaeus atricilla), and ring-billed gulls (Larus delawarensis), the respiratory epithelium commonly express both α-2,3 and α-2,6 sialic acids, whereas the intestinal tracts express predominantly α-2,3 and rarely α-2,6 sialic acids. 19 Historically, gull species from the Order Laridae have been associated with infection with LPAIV, including H11, H13, and H16 subtypes.25,35,57 These infections have been primarily associated with replication in epithelial cells of intestine and thus fecal-oral transmission of LPAIV in the black-headed gulls has been proposed. 26 In the case of HPAIV, natural infection has been reported in great skuas, European herring gulls, black-headed gulls, and great black-backed gulls (Larus marinus).1,2,7,13,15,37,41,43 Intestinal infections have rarely been observed with experimental studies using H5N1 clade 2.2 viruses in common gulls 24 and laughing gulls infected with an “Eurasian-lineage” of H5N1. 9 However, the cellular tropism of HPAIV has not been reviewed in detail in the previous studies, hence limiting the understanding of the pathobiology.
The viral tropism of the enteric or respiratory system has significant implications for the dynamics of disease transmission. In this report, viral immunolabeling in the trachea, proventriculus, gizzard, and duodenum of the great skua were more frequently detected than those great skua from the previous epizootic event in Great Britain in 2021. 7 The immunolabeling of the glandular epithelium of the gizzard in particular further confirms virus-specific immunolabeling of epithelial cells within the digestive tract. In addition, similar respiratory and enterotropism of HPAIV was observed in the long-tailed skua, herring gull, and black-headed gull in this study. Importantly, viral RNA was more abundant in the oropharyngeal compared with the cloacal swabs, with similar differences observed between respiratory and enteric tissues, which could be related to the expression pattern of α-2,3 and α-2,6 sialic acids in the respiratory and intestinal epithelium. 19 Contrary to the proposed enteric tropism and adaptation of Gs/Gd HPAIV in wild waterbirds,11,12 the dual tropism for the respiratory and enteric systems suggests that the contemporary HPAI 2.3.4.4b viruses are highly permissive within the skua and gull birds.
The pathway of incursion in free-ranging seabirds is not understood but has been proposed to be either independent incursion or onward introductions from species movements between colonies and the movement of seabirds between mainland and islands particularly during the breeding season.16,50 Herring gulls and great skua can opportunistically predate or scavenge on other birds,16,28,34,58 and this was observed in the outbreak in gannet colonies. Furthermore, contact transmission between common gulls and European herring gulls has been documented previously in experimental infections with HPAIV H5N1 clade 2.2 and H5N8 clade 2.3.4.4b.24,53 More recent HPAIV H5N1 outbreaks (June and August 2022) in wild bird rescue centers/hospitals in England (East Sussex and Cornwall) have been confirmed in herring gulls. After epidemiological assessment, the most likely source of infection appeared to be the introduction of infected/diseased herring gulls within the premises, which had then transmitted the disease to the resident gulls of the same species within and among enclosures (Animal and Plant Health Agency, unpublished data). During the breeding season, skuas and gulls often gather in large numbers to nest, feed, and bath, which increases the likelihood of close contact and potential transmission of HPAIV, particularly if virus replication occurs in the respiratory and intestinal tracts. 16 Infections through such contact can lead to birds from other colonies becoming exposed and infected, who can then disseminate the virus to new localities and susceptible avian species. The HPAI outbreak in the great skua colony in the United Kingdom has resulted in approximately 10% mortality during the summer of 2022, and outbreaks in captive herring gulls reported in this study have had mortality rates ranging from 20% to 50%. Although not examined in this study, subsets of skua or gull birds that develop mild or subclinical disease could potentially facilitate transmission over larger areas.
The distribution and ecology of seabird populations also challenge the current understanding of HPAIV transmission at a global level. Both long-tailed skua and great skua are transitory migrant birds. Long-tailed skuas are a passage migrant in the United Kingdom and breed in Arctic region, 23 whereas great skuas migrate to the northernmost isles of the United Kingdom in summer for breeding and return to the coasts of Spain and Africa, and as far as Brazilian and Argentinian coasts for wintering.27,44 This is in contrast to the black-headed gulls that are found across the United Kingdom, 31 and herring gulls are found throughout the year around the UK coastline and inland around rubbish tips, fields, large reservoirs, and lakes, especially during the winter months. 33 Recent ring-recovery data revealed that great skua, European herring gulls, and black-headed gulls migrate between Europe to Iceland and other North Atlantic islands, and to North America. 13 The pelagic and migratory nature of gulls have led to the suggestion of intercontinental dissemination and shaping of influenza A virus evolution.22,25,48,55,59 Furthermore, these seabirds are often found in areas with high seal populations plus other scavenging mammals that can predate on sick or dead birds, and result in exposures of other host populations to infectious materials either directly or indirectly through the environment. 17
Apart from the increased mortality in seabirds during 2022, which has resulted in an immediate impact upon populations, there is a significant deficit in knowledge on the impact of infectious diseases on population structures across these species. However, a trend toward a reduction in breeding abundance in the United Kingdom for herring gulls, black-headed gulls, and great skuas has been noted.31–33 The pathogenic mechanism of HPAIV on reproductive organs of wild bird is poorly documented. Previous reports have demonstrated epithelial labeling of viral antigen in the oviduct of common buzzards and peregrine falcons infected with HPAIV. 56 In domestic poultry, both HPAIV and LPAIV infections can lead to short to long-term reductions in egg production or embryonic death because of viral-induced lesions in the ovaries, oviduct, or conceptus.8,36,52,54 There has been an increased detection of reproductive diseases in laying poultry, both Galliformes and Anseriformes, during the 2022 epizootic season in the United Kingdom, which can be attributed to virus infections (Lean F, unpublished). However, the impact on the layer poultry sector, where an abundance of eggs is produced daily, cannot be compared with seasonal reproductive cycle in seabirds and as such, the longer-term impact on population densities for these species will require monitoring to assess population recovery.
In this study, great skuas and black-headed gulls were frozen and thawed for necropsy, which can introduce artifacts that could affect histopathological interpretation and the antigenicity of virus proteins for IHC. In addition, the small sample size for long-tailed skua and black-headed gull, and the gender skew (predominantly female great skuas and male gull birds) may limit meaningful comparison. It is important to note that the availability of carcasses received from wild bird surveillance is dependent on natural events. The age of birds could also potentially have an impact on disease outcome. It was documented from the 2 outbreaks from captive settings that only the younger herring gulls, but not the older birds, were clinically affected. It would have been useful to sample birds of different age groups and disease states, but such work has not been possible due to resource constraints during multiple disease outbreaks. Finally, this study only evaluated a subset of species within Stercorariidae and Laridae. Other seabird families, such as Alcidae, Pelecanidae, Phalacrocoracidae, and Procellariidae, have tested positive for HPAIV 2.3.4.4b during the epizootic; 4 therefore, further study is warranted to understand the susceptibility of other seabird species and the associated lesions with HPAIV infection.
In conclusion, this study demonstrates the susceptibility and pathology of a subset of Laridae and Suliformes following naturally acquired infection with HPAIV H5N1 clade 2.3.4.4b. The rapid mortality is associated with multisystemic dissemination of virus and resultant tissue damage. Epitheliotropism in the respiratory and digestive systems enables shedding of virus and facilitates transmission. Reproductive pathology is also noted amongst the female great skua, but the longer-term impact on population fecundity warrants further investigation.
Supplemental Material
sj-xlsx-1-vet-10.1177_03009858231217224 – Supplemental material for Highly pathogenic avian influenza virus H5N1 infection in skua and gulls in the United Kingdom, 2022
Supplemental material, sj-xlsx-1-vet-10.1177_03009858231217224 for Highly pathogenic avian influenza virus H5N1 infection in skua and gulls in the United Kingdom, 2022 by Fabian Z. X. Lean, Marco Falchieri, Natalia Furman, Glen Tyler, Caroline Robinson, Paul Holmes, Scott M. Reid, Ashley C. Banyard, Ian H. Brown, Catherine Man and Alejandro Núñez in Veterinary Pathology
Supplemental Material
sj-xlsx-2-vet-10.1177_03009858231217224 – Supplemental material for Highly pathogenic avian influenza virus H5N1 infection in skua and gulls in the United Kingdom, 2022
Supplemental material, sj-xlsx-2-vet-10.1177_03009858231217224 for Highly pathogenic avian influenza virus H5N1 infection in skua and gulls in the United Kingdom, 2022 by Fabian Z. X. Lean, Marco Falchieri, Natalia Furman, Glen Tyler, Caroline Robinson, Paul Holmes, Scott M. Reid, Ashley C. Banyard, Ian H. Brown, Catherine Man and Alejandro Núñez in Veterinary Pathology
Supplemental Material
sj-xlsx-3-vet-10.1177_03009858231217224 – Supplemental material for Highly pathogenic avian influenza virus H5N1 infection in skua and gulls in the United Kingdom, 2022
Supplemental material, sj-xlsx-3-vet-10.1177_03009858231217224 for Highly pathogenic avian influenza virus H5N1 infection in skua and gulls in the United Kingdom, 2022 by Fabian Z. X. Lean, Marco Falchieri, Natalia Furman, Glen Tyler, Caroline Robinson, Paul Holmes, Scott M. Reid, Ashley C. Banyard, Ian H. Brown, Catherine Man and Alejandro Núñez in Veterinary Pathology
Supplemental Material
sj-xlsx-4-vet-10.1177_03009858231217224 – Supplemental material for Highly pathogenic avian influenza virus H5N1 infection in skua and gulls in the United Kingdom, 2022
Supplemental material, sj-xlsx-4-vet-10.1177_03009858231217224 for Highly pathogenic avian influenza virus H5N1 infection in skua and gulls in the United Kingdom, 2022 by Fabian Z. X. Lean, Marco Falchieri, Natalia Furman, Glen Tyler, Caroline Robinson, Paul Holmes, Scott M. Reid, Ashley C. Banyard, Ian H. Brown, Catherine Man and Alejandro Núñez in Veterinary Pathology
Supplemental Material
sj-xlsx-5-vet-10.1177_03009858231217224 – Supplemental material for Highly pathogenic avian influenza virus H5N1 infection in skua and gulls in the United Kingdom, 2022
Supplemental material, sj-xlsx-5-vet-10.1177_03009858231217224 for Highly pathogenic avian influenza virus H5N1 infection in skua and gulls in the United Kingdom, 2022 by Fabian Z. X. Lean, Marco Falchieri, Natalia Furman, Glen Tyler, Caroline Robinson, Paul Holmes, Scott M. Reid, Ashley C. Banyard, Ian H. Brown, Catherine Man and Alejandro Núñez in Veterinary Pathology
Supplemental Material
sj-xlsx-6-vet-10.1177_03009858231217224 – Supplemental material for Highly pathogenic avian influenza virus H5N1 infection in skua and gulls in the United Kingdom, 2022
Supplemental material, sj-xlsx-6-vet-10.1177_03009858231217224 for Highly pathogenic avian influenza virus H5N1 infection in skua and gulls in the United Kingdom, 2022 by Fabian Z. X. Lean, Marco Falchieri, Natalia Furman, Glen Tyler, Caroline Robinson, Paul Holmes, Scott M. Reid, Ashley C. Banyard, Ian H. Brown, Catherine Man and Alejandro Núñez in Veterinary Pathology
Footnotes
Acknowledgements
We thank the scientists at APHA for their laboratory work and colleagues at Scotland’s Rural College and NatureScot for the support.
Author Contributions
F.Z.X.L., M.F., N.F., G.T., C.R., P.H., and C.M. involved in conceptualization of the investigations. F.Z.X.L., N.F. and A.N. performed the necropsies. F.Z.X.L. conducted formal analysis. A.N., A.C.B., S.M.R., I.H.B., and C.M. provided project leadership, financial, and laboratory resources. F.Z.X.L. wrote the original draft. All authors reviewed and edited the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the UK Department for Environment, Food, and Rural Affairs (Defra); the devolved administrations of the Scottish and the Welsh Governments (grant numbers: SV3006, SV3032, SV3400, SE2213).
Ethical Statement
No ethical approval was required as carcass and tissue were derived from diagnostic investigations.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
