Abstract
Since 2005, clade 2.2 H5N1 highly pathogenic avian influenza (HPAI) viruses have caused infections and morbidity among numerous species of wild waterfowl in Eurasia and Africa. However, outbreaks associated with clade 2.3.2 viruses have increased since 2009, and viruses within this clade have become the dominant strain of the H5N1 HPAI virus detected in wild birds, reaching endemic status in domestic birds in select regions of Asia. To address questions regarding the emergence and expansion of clade 2.3.2 viruses, 2 waterfowl species repeatedly involved in outbreaks of H5N1 HPAI viruses, bar-headed geese (Anser indicus) and ruddy shelducks (Tadorna ferruginea), were inoculated with a representative virus. All of 3 infected ruddy shelducks exhibited neurologic signs and died within 4 to 5 days. Two of 3 infected bar-headed geese had transient weakness but all survived. Viral shedding was predominately via the oropharynx and was detected from 1 to 7 days after inoculation. The severity and distribution of microscopic lesions corresponded with clinical disease and influenza-specific immunohistochemical staining of neurons. The predominant lesions were in the brain and were more severe in ruddy shelducks. Increased caspase-3 reactivity in the brains of all infected birds suggests a role for apoptosis in H5N1 HPAI virus pathogenesis in these species. These results demonstrate that similar to clade 2.2 viruses, a clade 2.3.2 H5N1 HPAI virus is neurotropic in some waterfowl species and can lead to neurologic disease with varying clinical outcomes. This has implications for the role that wild waterfowl may play in transmission of this virus in endemic regions.
Keywords
Infection of wild birds with H5N1 highly pathogenic avian influenza (HPAI) viruses was first documented in late 2002, when numerous species of ducks, geese, and swans died at a nature park in Hong Kong. 9 Subsequently, in 2005 and 2006, H5N1 HPAI virus epidemics at Lake Qinghai, China, affected thousands of migratory waterfowl, the majority of which were bar-headed geese (Anser indicus) with smaller numbers of ruddy shelducks (Tadorna ferruginea).5,24,50 Four genetically distinct H5N1 HPAI viruses were responsible for the largest of these outbreaks and were classified as clade 2.2 based on phylogenetic characterization and sequence homology of the H5 hemagglutinin gene. 5 From 2006 to 2009, sporadic outbreaks of clade 2.2 H5N1 HPAI viruses occurred among wild birds in Central Asia, Europe, Africa, and the Middle East.14,22,39 Ruddy shelducks and bar-headed geese were again among the bird species affected, and laboratory-based trials confirmed fatal outcomes in individuals of both species following inoculation with clade 2.2 viruses.3,23
Starting in 2007, clade 2.3.2 H5N1 HPAI viruses were detected in wild birds in Hong Kong and appeared to be the predominant H5N1 HPAI virus among wild birds in Mongolia in 2009 to 2010.40,44 These viruses have caused numerous outbreaks in the vicinity of Lake Qinghai, China, as well as Russia, Japan, Mongolia, and South Korea.11,15,18,20,40,42,48 In addition to wild birds, clade 2.3.2 H5N1 HPAI viruses have been detected and associated with disease outbreaks in poultry in Romania, Bulgaria, Vietnam, and India.26,29,37
Bar-headed geese and ruddy shelducks migrate along the central Asian flyway and intermingle with other waterfowl at breeding and wintering sites. 5 This intermingling potentially allows for exchange of influenza viruses between these host populations that could ultimately lead to altered transmission patterns and host pathogenicity. Clade 2.3.2 viruses were isolated from carcasses of ruddy shelducks and bar-headed geese in 2009 and 2010 on Lake Qinghai, China, as well as on numerous lakes in Mongolia, suggesting that as with clade 2.2 viruses, clade 2.3.2 viruses are associated with a fatal outcome in these birds.11,15,18,40 To date, no experimental infection experiments with clade 2.3.2 viruses have been performed in waterfowl.
The present experimental infection of bar-headed geese and ruddy shelducks with a clade 2.3.2 H5N1 HPAI virus was performed to better understand the pathogenesis, virulence, and viral shedding patterns in 2 waterfowl species commonly infected with these viruses in the wild. Such information, in conjunction with bar-headed goose and ruddy shelduck satellite telemetry data,11,12,27 will provide insight into the potential of these species to contribute to short- or long-term maintenance and dissemination of these new H5N1 HPAI viruses.
Methods
Animals
Captive-reared birds were purchased from Chenoa Farms (Martin, TN) at 16 to 20 weeks of age. Upon arrival, all birds were banded and bled to confirm seronegative status for avian influenza virus. Birds were housed individually in isolator units at a US Department of Agriculture (USDA)–certified biosafety level 3–enhanced facility at the Southeast Poultry Research Laboratory (SEPRL), Agricultural Research Service, USDA in Athens, Georgia. 1 Birds were allotted a 4-day acclimation period in the isolator units prior to inoculation. Commercial waterfowl maintenance feed (Mazuri, Richmond, IN) and water were provided ad libitum.
Birds were cared for in accordance with the guidelines of the Institutional Animal Care and Use Committee as outlined in the Guide for the Care and Use of Agricultural Animals in Agricultural Research and Teaching 6 and with the approval of Institutional Animal Care and Use Committees at both SEPRL and the University of Georgia.
Viruses, Inoculation, and Sampling Scheme
Four ruddy shelducks and 3 bar-headed geese were intranasally inoculated via the choanal cleft with 0.1 ml of brain-heart infusion (BHI) medium containing 104 median embryo infectious dose (EID50) of A/bar-headed goose/Mongolia/X53/2009 (H5N1). This low dose, relative to previous H5N1 HPAI virus challenge studies, was chosen to more closely mimic natural exposures and therefore assess the potential for subclinical infections. 19 One individual of each species was inoculated with a sham solution consisting of an equal volume of allantoic fluid diluted 1:30 in BHI medium.
Bird activity and behavior were evaluated at least once daily throughout the duration of the trial, and birds were weighed when handled for swab collection. The oropharyngeal and cloacal cavities of each bird, including sham-inoculated controls, were swabbed prior to inoculation (ie, 0 days postinoculation [DPI]), as well as 1 to 7, 10, and 14 DPI. Oropharyngeal and cloacal swabs were immediately placed in serum-free Eagle’s minimum essential medium (MEM) supplemented with antibiotics (100 μg penicillin G with 100 μg streptomycin sulfate/ml) and stored at –70°C until testing. Any bird exhibiting severe clinical disease, including neurologic signs (eg, depression, seizures, ataxia), was euthanized by intravenous sodium pentobarbital injection (100 mg/kg body weight). All remaining virus-challenged and sham-inoculated birds were euthanized on 14 DPI. Birds were considered infected if they shed virus via the oropharyngeal or cloacal cavities and either died during the period of shedding or subsequently seroconverted.
Virus Isolation
Virus isolation from swabs was performed in 9- to 11-day-old, specific pathogen-free, embryonating chicken eggs following standard procedures. 47 Viral titers were determined by microtiter end-point titration in Madin-Darby canine kidney (MDCK) cells for all positive samples. 4 Briefly, serial 10-fold dilutions were made of clarified media for all virus isolation positive swab samples, which were then inoculated onto confluent monolayers of MDCK cells. Resulting viral titers were calculated using the Reed and Muench method 36 and reported as median tissue culture infectious dose (TCID50)/ml of swab media. The minimum detectable titer was 101.8 TCID50/ml. Samples that were positive by virus isolation with a titer below the detectable limit were reported as 101.0 TCID50/ml.
Serologic Testing
Testing on pre- and postinoculation sera from all birds was performed using a commercial blocking enzyme-linked immunosorbent assay (FlockCheck AI MultiS-Screen Antibody Test Kit; IDEXX Laboratories, Westbrook, ME), which detects antibodies directed against the nucleoprotein of influenza A viruses. Postinoculation serology was performed on sera collected just prior to euthanasia on 14 DPI but not on sera from birds that died or were euthanized prior to 14 DPI.
Histopathology and Immunohistochemistry for Influenza Viral Antigen
All birds were necropsied immediately after death, with routine tissue collection from all major organ systems for histopathologic and immunohistochemical evaluation. Tissues were fixed in 10% neutral-buffered formalin, routinely processed, embedded in paraffin, sectioned at approximately 5 μm, mounted on glass slides, and stained with hematoxylin and eosin. Duplicate slides were deparaffinized and rehydrated and immunohistochemically stained as previously described with a mouse-derived monoclonal antibody (P13C11, developed at SEPRL) specific for type A influenza virus nucleoprotein antigen as the primary antibody. 33 The primary antibody was detected through application of a biotinylated goat anti–mouse IgG secondary antibody using a biotin-streptavidin detection system (Supersensitive Multilink Immunodetection System; Biogenex, San Ramon, CA). Fast red TR (Biogenex) was used as a substrate chromagen and hematoxylin as a counterstain. The presence of positive-staining antigen was subjectively scored for each tissue and ranged from negative or “–” for no staining; “+” for 1 to 2 small foci or <10 scattered, individually stained cells; “++” for 1 larger focus or numerous smaller foci of stained cells; and “+++” for abundant and widespread, immunohistochemically positive cells.
Detection of Apoptosis by Caspase-3 Immunohistochemistry and TUNEL Assay
For caspase-3 (cleaved) immunohistochemical staining of deparaffinized brain sections of H5N1 HPAI virus–infected and uninfected bar-headed geese and ruddy shelducks (excluding the individual that was inoculated but not infected, No. 7), the primary antibody, polyclonal rabbit anti–caspase-3 (cleaved) antibody (CP229; Biocare Medical, LLC, Concord, CA), was diluted 1:200 using Dako Antibody Diluent (S0809; Dako, Carpinteria, CA) with a staining time of 60 minutes. Antigen retrieval was heat-induced epitope retrieval (HIER) using target retrieval solution (10×), pH 9.0 (S2367; Dako). Endogenous peroxidase was quenched using 3% hydrogen peroxide (H312-500; Fisher Scientific, Fair Lawn, NJ). All other blocking was completed with Power Block (HK085-5K; Biogenex). As a negative reagent control, the primary antibody was replaced with purified rabbit immunoglobulins (NC495H; Biocare Medical, LLC). SuperPicTure polymer kit (Invitrogen, Camarillo, CA) was used as an amplification system, and the substrate-chromogen system was diaminobenzidine (DAB) (K3466; Dako). Tissue sections were counterstained with Gills II hematoxylin and bluing; dehydrated in alcohol levels of 70%, 95%, and 100%; cleared in xylene; and mounted with xylene-based mounting medium.
Labeling of apoptotic cells of deparaffinized brain sections of H5N1 HPAI virus–infected and uninfected bar-headed geese and ruddy shelducks (excluding the inoculated ruddy shelduck that did not become infected, No. 7) was performed with a terminal deoxynucleotidyl TUNEL (transferase-mediated dUTP nick end labeling) assay (TUNEL apoptosis detection kit for paraffin-embedded tissue sections, biotin-labeled POD; GenScript, Piscataway, NJ) per the manufacturer’s instructions with the following exceptions. The DNase I incubation step was omitted, and the blocking step consisted of a 10-minute incubation with 3% hydrogen peroxide. HIER was performed using citrate buffer (HK086-9K; Biogenex) at pH 6.0 for 30 minutes at 95°C to 100°C prior to incubation with proteinase-K (50×; 1 mg/ml; S3020, Dako).
For both caspase-3 and TUNEL assays, positive tissue controls consisted of formalin-fixed, paraffin-embedded rat lymphoid tissue, as well as same-bird lymphoid tissues (ie, cloacal bursa and cecal tonsil). Brains and lymphoid tissues of noninfected birds served as negative controls. For assessment of reactivity with caspase-3, the average number of positive-staining cell nuclei in ten 400× fields within the cerebrum of infected birds was compared with similar areas within the cerebrum of conspecific, sham-inoculated control birds. In all infected birds, cell numbers were assessed in fields with and adjacent to microscopic lesions that corresponded to positive immunohistochemical staining for influenza virus antigen in ruddy shelducks. Because no TUNEL reactivity was evident in the brains of sham-inoculated control birds, infected birds were assessed for TUNEL staining similarly to those for caspase-3, but staining was classified as weak (average of 0–5 cells/400× field), moderate (average of 5–10 cells/400× field), or strong (average of >10 cells/400× field) reactivity.
Results
Morbidity and Mortality
Mild lethargy was observed during daily examination of 2 of the 3 infected bar-headed geese (Nos. 1 and 2) on 6 and 7 DPI. In addition, bar-headed goose No. 2 had unilateral opacity in the right eye from 6 to 7 DPI, which subsequently resolved. All bar-headed geese survived to the end of the trial on 14 DPI, and body weights of infected geese increased between 5.6% and 14.4% of preinoculation weights. The 3 infected ruddy shelducks that excreted detectable levels of virus (Nos. 4–6) died or were euthanized due to severe neurologic disease by 5 DPI. Clinical signs were severe and rapid in onset in these birds. Clinical signs in the euthanized ruddy shelducks (Nos. 4 and 5) included severe ataxia and seizures that lasted 24 hours or less. Death in the noneuthanized ruddy shelduck (No. 6) occurred on 4 DPI with no premonitory clinical signs; however, the postmortem neck and head orientation of the carcass was indicative of severe torticollis at the time of death. At the time of death, body weights of infected ruddy shelducks had decreased between 9.2% and 23.5% versus preinoculation weights.
No clinical signs of disease were observed in one of the virus-inoculated but noninfected ruddy shelducks (No. 7) or in the sham-inoculated control birds.
Viral Shedding and Seroconversion
Virus was detected in both oropharyngeal and cloacal swabs from infected bar-headed geese and ruddy shelducks. However, oropharyngeal viral shedding predominated in both species and was detected in all infected bar-headed geese beginning on 1 DPI, followed by continual shedding through 6 or 7 DPI. Peak viral shedding titers in all 3 bar-headed geese occurred on 4 DPI and ranged from 104.6 to 105.3 TCID50/ml. Oropharyngeal shedding in infected ruddy shelducks was first detected on 1 to 3 DPI, followed by continual shedding until death; titers were highest on the day of death in the 2 euthanized ducks (Nos. 4 and 5; 5 DPI), as well as in the ruddy shelduck that was found dead (No. 6; 3 DPI). In the latter bird, detection of oropharyngeal shedding was limited to 3 DPI. Peak oropharyngeal titers were lower in ruddy shelducks versus bar-headed geese. In addition, the duration of shedding was shorter in ruddy shelducks than in bar-headed geese, which was likely a result of the acute mortality observed in the former. Cloacal shedding was detected in all infected ruddy shelducks and in 2 of the 3 infected bar-headed geese; however, viral shedding by this route was reduced relative to oropharyngeal shedding in both species (Table 1). The inoculated ruddy shelduck that failed to become infected (No. 7) and the sham-inoculated birds had no detectable shedding via the oropharyngeal or cloacal cavities throughout the sampling period.
Mortality and Viral Shedding Data From Bar-Headed Geese and Ruddy Shelducks Intranasally Inoculated With H5N1 HPAI Virus, Clade 2.3.2.
BHGE, bar-headed goose; DPI, days postinoculation; RUSD, ruddy shelduck. The inoculated but uninfected ruddy-shelduck (No. 7) is not included in these data.
aViral shedding is expressed in median tissue culture infectious dose (TCID50/ml); duration is the total range of days postinoculation during that viral shedding was detected.
All bar-headed geese seroconverted by 14 DPI. Ruddy shelduck No. 7 was seronegative on 14 DPI, as were the sham-inoculated birds. Seroconversion was not assessed in the ruddy shelducks that died or were euthanized prior to 14 DPI.
Pathology and Immunohistochemical Staining for Influenza Virus
The ruddy shelduck found dead on 4 DPI (No. 6) was in good nutritional condition and had multiple, well-demarcated foci of hemorrhage and necrosis scattered throughout the pancreas. The ruddy shelducks euthanized on 5 DPI (Nos. 4 and 5) were in good nutritional condition and had diffuse congestion of the meninges, as well as multiple well-demarcated areas of hemorrhage and necrosis in the pancreas, and No. 5 also had diffuse pulmonary congestion. All sham-inoculated birds, infected bar-headed geese, and the virus-inoculated but noninfected ruddy shelduck (No. 7) were in fair to good nutritional condition and had no apparent gross lesions.
Microscopic lesions were more frequent and severe in infected ruddy shelducks versus bar-headed geese and were most pronounced in the cerebrum of both species (Table 2). Cerebral lesions included mild perivascular aggregates of lymphocytes and plasma cells throughout the gray and white matter in all infected bar-headed geese (Fig. 1), with necrosis of individual neurons adjacent to the affected vessels in 2 (Nos. 2 and 3). One bar-headed goose (No. 2) had multiple, small hemorrhages in the cerebellar white matter and Purkinje cell layer, whereas bar-headed goose No. 3 had similar hemorrhages in the cerebral gray matter. The cerebrums of infected ruddy shelducks also had perivascular, lymphoplasmacytic inflammation with occasional rarefaction of the adjacent neuroparenchyma; in 2 of these ducks (Nos. 4 and 5), inflammation rarely infiltrated vessel walls (vasculitis) and occasionally extended to the meninges. Two of these ducks also had neuronal necrosis adjacent to areas of inflammation (Nos. 4 and 5; Fig. 2). Ruddy shelducks Nos. 4 and 5 had focal to multifocal small hemorrhages along the cerebellar surface and within the molecular layer. Marked congestion of blood vessels was evident throughout the brains of virus-inoculated, infected, and sham-inoculated birds.
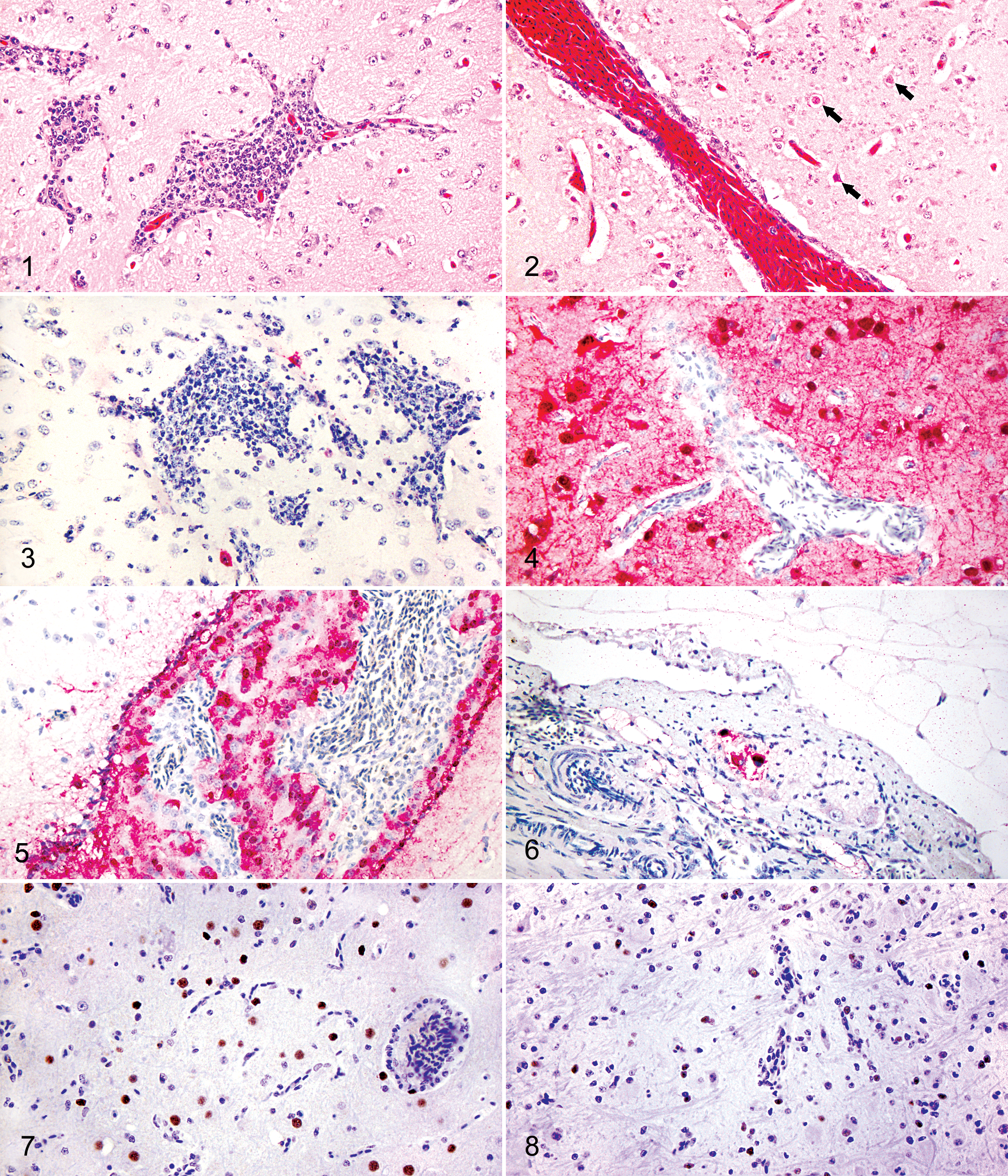
Histopathology and Immunohistochemical Staining in Bar-Headed Geese and Ruddy Shelducks Intranasally Inoculated With H5N1 HPAI Virus, Clade 2.3.2.
Immunohistochemical staining for influenza antigen was subjectively quantified as –, negative; +, minimal (eg, 1–2 foci); ++, moderate (ie, multiple foci); and +++, abundant and widespread. Histologic lesions are listed under each tissue; X, lesions present; 0, no lesions present.
aLymphoid tissues included cecal tonsil and/or cloacal bursa.
bA small number of scattered mucosal epithelial cells and nerve ganglia within the corresponding mesenteric plexus were immunohistochemically positive for influenza virus antigen.
Positive immunohistochemical staining for influenza virus antigen in the brains of infected bar-headed geese was limited to the cytoplasm of a few individual or small groups of scattered neurons in the cerebrum (Fig. 3). In contrast, the distribution of virus antigen in the cerebrums of ruddy shelducks corresponded with microscopic lesions, with extensive areas of positive immunohistochemical staining of the cytoplasm and nuclei of many neurons as well as neuronal cell processes (Fig. 4). Positive immunohistochemical staining also occurred in ependymal cells and associated choroid epithelial cells (Fig. 5), the meninges, and the neuroparenchyma corresponding to areas of perivascular cuffing, necrosis, and rarefaction. Smaller and fewer foci of immunohistochemically positive neurons were in the Purkinje cell layer and, less commonly, microglial cells, granule cells, and neurons in the molecular and granular layers, and white matter tracts of the cerebellum were immunohistochemically positive in ruddy shelduck Nos. 4 to 6 (Table 2).
Several tissues other than brain had microscopic lesions, which were most common in the pancreas and lymphoid tissues (ie, cloacal bursa and cecal tonsil). The pancreas of all infected bar-headed geese (Nos. 1–3) had variable zymogen depletion and congestion; Nos. 1 and 3 had multifocal, acinar degeneration and No. 3 also had a focus of periacinar, lymphoplasmacytic inflammation. Ruddy shelducks also had variable amounts of zymogen depletion in the pancreas, and all birds had scattered, single-cell (Nos. 5 and 6) to diffuse disorganization of acini with shrunken cytoplasm, pale nuclei, and vacuolation (No. 4) with a small number of corresponding, immunohistochemically positive acinar cells. No significant renal lesions were observed in bar-headed geese; 2 ruddy shelducks (Nos. 4 and 5) had mineral within multiple tubular lumens (also present in the sham-inoculated ruddy shelduck), and No. 4 had mild, tubular urate deposition and associated heterophilic inflammation.
Bar-headed geese had mild, perivascular, lymphoplasmacytic hepatitis similar to that observed in the sham-inoculated control bird. The livers of ruddy shelduck Nos. 4 to 6 had variable intracytoplasmic lipid; moderate lymphoplasmacytic (and heterophilic in Nos. 5 and 6), perivascular inflammation; and multifocal hemosiderin deposition within hepatocytes and Kupffer cells. Ruddy shelduck No. 6 had diffuse disruption of normal hepatic architecture with multiple, necrotic foci as well as single-cell necrosis. No lesions were evident in hearts of bar-headed geese, whereas ruddy shelduck Nos. 4 and 5 each had a mild focus of necrosis that affected a small number of myocytes with corresponding, positive immunohistochemical staining in No. 5. All infected bar-headed geese had mild to moderate lymphoid depletion in the splenic white pulp with moderate congestion, whereas ruddy shelducks had more severe lymphoid depletion, and No. 6 had multiple foci of lytic necrosis. Most inoculated birds, as well as sham-inoculated birds, had multifocal hemorrhages within the splenic red pulp. The lymphoid germinal centers in the spleen, cloacal bursa, and cecal tonsils had mild to moderate depletion of lymphocytes in all infected bar-headed geese, with more severe depletion and multifocal necrosis in the cloacal bursa of all infected ruddy shelducks. Lymphoid tissues in birds of both species had variable amounts of individual cell apoptosis, and a few scattered lymphoid cells in the cloacal bursa were immunohistochemically positive in ruddy shelduck Nos. 4 and 5. Ruddy shelduck No. 4 also had multiple foci consisting of 1 to 3 immunohistochemically positive lymphocytes. The adrenal glands of all infected ruddy shelducks had mild, multifocal degeneration of cortical cells with rare, individual cell necrosis (the adrenal gland was analyzed for 1 of the 3 inoculated bar-headed geese). Ruddy shelduck Nos. 4 and 5 had a small number of immunohistochemically positive neurons within ganglia of the myenteric plexus (Fig. 6), and No. 5 also had a focus of immunohistochemically positive cells in the neurohypophysis of the pituitary gland. Vascular congestion was present throughout numerous tissues, including lung, kidney, liver, and small intestine, of some inoculated and sham-inoculated individuals of both species.
No significant lesions were observed in the following tissues: skin, trachea, lung, skeletal muscle, gonads, esophagus, proventriculus, ventriculus, small intestine, and large intestine. Incidental lesions included a focal, caseous, heterophilic granuloma with bacteria in the intestinal lumen of ruddy shelduck No. 4 and numerous bacterial emboli in the spleen, lung, kidney, ovary, and liver of ruddy shelduck No. 6. Microscopic lesions in the ruddy shelduck that failed to become infected (No. 7) were limited to lymphoplasmacytic, periportal, and centrilobular hepatitis and diffuse hepatic lipidosis, as well as a small number of aggregates of lymphocytes and plasma cells in the renal interstitium. No avian influenza virus–specific immunohistochemical staining was observed in the following tissues from any individual: spleen, liver, proventriculus, adrenal gland, kidney, large intestine, lung, esophagus, trachea, skin, skeletal muscle, and gonads. In addition, no positive immunostaining was evident in tissues of the inoculated but uninfected ruddy shelduck (No. 7) or sham-inoculated controls (Table 2).
Staining for Apoptosis
The majority of caspase-3 expression was in the nuclei of glial cells, specifically oligodendrocytes and microglia, with less common nuclear staining in neurons and astrocytes. Cells expressing caspase-3 were distributed throughout the brain but were generally more numerous adjacent to and within areas with inflammation and necrosis. The average numbers of cells that expressed caspase-3 in the cerebrum were approximately 40% to 60% greater in infected bar-headed geese and 50% to 160% greater in infected ruddy shelducks versus the sham-inoculated birds. In general, staining intensity of cells in ruddy shelducks was greater than in bar-headed geese (Figs. 7 and 8), and was highest in ruddy shelduck Nos. 5 and 6. Cells expressing capase-3 were also increased within the cerebellar white matter and Purkinje cell and granular layers, as well as occasionally within the neuroparenchyma subjacent to the meninges of infected ruddy shelducks, although cell numbers were not quantified in these sites. Cells expressing caspase-3 were not observed within perivascular inflammatory infiltrates.
TUNEL staining for DNA fragmentation was generally less intense and less frequent in cells in the brains of infected birds for both species as compared with caspase; however, the identities of positively stained cells were consistent with those expressing caspase-3. Bar-headed geese Nos. 1 and 3 demonstrated weak TUNEL reactivity, whereas bar-headed goose No. 2 had relatively stronger reactivity. All infected ruddy shelducks had moderate reactivity. TUNEL reactivity was not evident in the brains of sham-inoculated birds.
Discussion
The unprecedented wild bird mortality caused by clade 2.2 H5N1 HPAI viruses prompted research focused on understanding the epidemiology, pathogenesis, and transmission dynamics of these viruses, including field investigations5,11,22,24,27 and challenge studies in multiple avian species.3,13,25,32 More recently, clade 2.2 H5N1 HPAI viruses in wild birds have been replaced with clade 2.3.2 viruses in select geographic regions (eg, Vietnam, Thailand, China, Japan, Mongolia, and Russia), creating the need for additional information on the impacts of these emerging viruses on the health of wild bird populations and the potential role that migratory waterfowl may play in the epidemiology of these viruses.11,12,15,16,27,40,42,44 Bar-headed geese and ruddy shelducks were selected for experimental infection with a clade 2.3.2 H5N1HPAI virus in the present study because they have been involved in H5N1 HPAI virus outbreaks concerning both clade 2.2 and 2.3.2 viruses and have been implicated in the spread of these viruses based on their migratory status, abundance, degree of interspecies mixing, gregariousness, and virus prevalence.5,7,10,12,15,18,22,24,35,40,50
Viral shedding profiles of waterfowl in the present study were similar to those of domestic and wild waterfowl experimentally inoculated with clades 0, 1, and 2.2 H5N1 HPAI viruses, in which shedding from the oropharynx was often of higher magnitude and longer duration than via the cloaca.2,3,19,23,25,34,45 Bar-headed geese shed higher titers of virus of longer duration from the oropharyngeal cavity than ruddy shelducks, likely in part due to varied postinoculation survival periods. The higher survival rate and increased titers and duration of viral shedding in bar-headed geese in the present study suggest that this species may represent a greater risk than ruddy shelducks for the dissemination of H5N1 HPAI virus in a natural setting. Conversely, due to the more severe effects of disease in ruddy shelducks, this species may be a better sentinel for passive H5N1 HPAI virus wildlife surveillance. Previous evaluation of ruddy shelducks inoculated with a clade 2.2 virus resulted in no detectable cloacal shedding by polymerase chain reaction, whereas oropharyngeal shedding was detected in 2 of 2 inoculated ducks. 23 Bar-headed geese inoculated with a clade 2.2 virus shed via the oropharynx and cloaca for 7 to 8 days, and titers were higher in oropharyngeal (105.10 EID50/ml) than in cloacal secretions (102.55 EID50/ml). 3 This pattern may reflect the continuing evolution of H5N1 HPAI viruses and an associated shift in transmission dynamics from the digestive tract as the predominant site of shedding (ie, fecal-oral route) to respiratory or oropharyngeal transmission, evident since late 2002. 45
Clinical signs associated with clade 2.3.2 H5N1 HPAI virus infection were short-lived and minimally evident in bar-headed geese in the present study, whereas ruddy shelducks had fatal neurologic disease similar to that observed in wild and domestic waterfowl infected with clade 0, 1, and 2.2 H5N1 HPAI viruses.3,5,9,19,21,23–25,34 Ruddy shelducks inoculated with a clade 2.2 H5N1 Korean virus were ataxic and lethargic, which culminated in 100% mortality from 5 to 9 DPI. 23 All of 5 bar-headed geese infected with a clade 2.2 H5N1 Mongolian virus were depressed and had transient ocular opacity, with gradual resolution of clinical signs in 3 individuals and neurologic disease and death from 6 to 7 DPI in the remainder. Clinical disease lasted an average of 5.3 days in survivors and 2.5 days in those that succumbed. 3 Results from the present study suggest that bar-headed geese may be less susceptible to severe neurologic disease associated with clade 2.3.2 vs clade 2.2 viruses; however, this is difficult to assess due to limited sample sizes. Experimental inoculation was unsuccessful in 1 ruddy shelduck in the present study that had no detectable shedding or clinical disease, no histopathology in the brain, and no immunohistochemical evidence of antigen in any tissues and failed to seroconvert.
The clade 2.3.2 H5N1 HPAI virus used in the present study appears to have retained the neurotropism in bar-headed geese and ruddy shelducks that has been observed in numerous waterfowl species infected with preceding clades. The cerebrum appeared to be the major site of H5N1 HPAI clade 2.3.2 virus replication and was the site of the predominant lesions in both bar-headed geese and ruddy shelducks. These lesions included perivascular, lymphoplasmacytic cuffing and neuronal necrosis in both species, as well as multifocal vacuolation of neuropil and hemorrhage, lymphoplasmacytic meningitis, and rare vasculitis in ruddy shelducks. Influenza virus antigen was not detected within endothelial cells of ruddy shelducks in the present study. This finding is consistent with previous experimental inoculations of ruddy shelducks, bar-headed geese, and other waterfowl species; however, it contrasts results in several experimentally inoculated swan species, in which immunohistochemically positive endothelial cells corresponded to severe vascular lesions.2,3,17,19,23 As pointed out in these previous studies, the pathogenetic mechanisms and associated morbidity and mortality with H5N1 HPAI virus infection may differ among waterfowl species. Ensuing disease in birds that survive the peracute phase of infection may be associated more with virus replication and damage within the parenchyma of numerous tissues (eg, brain, autonomic nervous system, pancreas, endocrine organs, and heart) than those with early and extensive vascular damage, including apoptotic endothelial cells and microthrombosis. 46 If virus replication occurred in the endothelium of ruddy shelducks in the present study, replication levels may have been short-lived (ie, cleared soon after infection) or minimal, leading to lack of virus antigen detection. Viral antigen within the parasympathetic ganglia of the intestinal submucosa and myenteric plexus of ruddy shelducks in the present study has also been documented in Canada geese (Branta canadensis) and wood ducks (Aix sponsa) experimentally inoculated with a Vietnamese clade 2.3.2 virus and a Mongolian clade 2.2 virus, respectively (World Health Organization, www.who.int/influenza/gisrs_laboratory/201101_h5fulltree.pdf; accessed March 5, 2013).2,32 Also similar to ruddy shelducks in the present study, severe neurologic disease in tufted ducks (Aythya fuligula) and pochards (Aythya ferina) infected with a clade 2.2 H5N1 Turkish virus was associated with multifocal viral encephalitis and corresponding positive immunohistochemical staining. 19
Histopathology outside of the brain in bar-headed geese in the present study was rare, whereas most infected ruddy shelducks had mild pancreatic, cardiac, adrenal, and pituitary necrosis, similar to ruddy shelducks previously inoculated with a 100× higher dose of a clade 2.2 virus. 23 Previously circulating H5N1 viruses caused necrosis of the adrenal gland, pancreas, heart, skeletal muscle, and spleen of domestic and wild waterfowl.3,5,19,24 However, unlike in ruddy shelducks infected with a clade 2.2 virus, necrosis of the nasal epithelium was not observed in birds in the present study. 23 Furthermore, bronchointerstitial pneumonia with colocalized immunohistochemical staining in tufted ducks and pochards inoculated with a clade 2.2 virus 19 was not observed in either ruddy shelducks or bar-headed geese in the present study. Despite the lack of lesions in the upper and lower respiratory tracts in birds in the present study, the choanal inoculation route with immunohistochemically positive ependymal and choroid plexus epithelial cells in the brain support possible virus access to the brain via neurons of olfactory receptors and nerves leading to the olfactory bulb, as has been shown in rodent species infected with H5N1 HPAI virus and other neurotropic viruses (eg, eastern equine encephalitis and Venezuelan equine encephalitis viruses).31,38,49 The inciting natural transmission route among waterfowl may occur via inhalation of virus-laden droplets from nearby infected birds or waterborne virus into the nasal cavity while drinking, eating, bathing, and preening. Infected birds in the present study had variable lymphoid depletion of follicles in the cloacal bursa, cecal tonsils, and spleen, consistent with virus-induced immunosuppression, also supportive of a slightly more chronic disease course. The lack of histopathology and viral antigen in the gastrointestinal tract of infected birds in the present study is consistent with the minimal cloacal shedding.
Caspase-3 expression and, to a lesser extent, TUNEL reactivity in the brains of bar-headed geese and ruddy shelducks infected with H5N1 HPAI clade 2.3.2 virus suggest that apoptosis plays a role in viral pathogenesis. The numbers of cells expressing caspase-3 were increased in the brains of both species but were higher in ruddy shelducks, which also had more severe microscopic brain lesions with increased intensity and frequency of influenza-specific immunohistochemical staining. The TUNEL assay detects DNA fragmentation (ie, degradation) and reveals cells undergoing apoptosis; caspases are proteases activated during apoptosis and are necessary for protein cleavage, DNA fragmentation, and nuclear collapse. 43 Some viral gene products block apoptosis, a strategy to prevent premature cell death and therefore maximize viral replicative potential in the cell. Some viruses promote apoptosis, thereby contributing to cytotoxicity and potentially to production of virus progeny. 30 In mice as well as humans, infection of astrocytes, microglial cells, and/or neurons with neurotropic H5N1 HPAI virus led to release of proinflammatory cytokines and apoptosis (ie, cytopathic effect) in these cells.28,51 Apoptosis may be a more predominant feature of infection with more virulent subtypes of HPAI viruses, as TUNEL reactivity and expression of caspases were elevated in porcine alveolar epithelial cells infected in vitro with H5N1 HPAI viruses but not with other H5 influenza viruses. 8 In vitro and in vivo mouse studies suggested that neuraminidase of influenza A and B viruses activates sufficient levels of transforming growth factor–β to induce apoptosis in influenza virus–infected cells. 41 More detailed investigations into apoptotic signals and virus-infected cells are needed to evaluate the role of apoptosis in the pathogenesis of H5N1 HPAI influenza viruses in wild bird species.
In summary, experimental infection of 2 waterfowl species common to the Central Asian and East Asian–Australasian flyways, where H5N1 HPAI clade 2.3.2 virus is present, revealed predominantly oropharyngeal shedding and viral neurotropism, with interspecies variability in clinical outcomes and pathology. Oropharyngeal viral shedding was of longer duration and higher peak titers in bar-headed geese, which were relatively resistant to clinical disease but had mild to moderate lesions in the brain. In contrast, infection was fatal in ruddy shelducks, which had more severe microscopic lesions, especially in the brain, as well as abundant viral antigen and increased numbers of apoptotic cells in the brain. These results are consistent with the recent findings of Gilbert and coauthors, 11 in which sick and dying wild bar-headed geese were less commonly observed than ruddy shelducks and other waterfowl species during H5N1 HPAI clade 2.3.2 virus outbreaks in Mongolia. Samples collected from these birds proved more useful in detecting HPAI virus outbreaks than surveillance of healthy wild birds and environmental (fecal) samples.
Results from the present study support the notion that infected bar-headed geese can likely carry a clade 2.3.2 H5N1 HPAI virus over moderate distances and therefore potentially serve as a source of these viruses to comingling birds.10–12,15,18 Recent satellite telemetry data suggest that bar-headed geese and ruddy shelduck flight patterns (eg, travel rates and long, uninterrupted flight periods) may lessen the likelihood that these migratory waterfowl species transport H5N1 HPAI viruses over long distances, although the potential for contacting other wild birds and poultry during migration exists, especially for bar-headed geese. The authors suggest that relay transmission involving multiple infected individuals during migration may be a more likely role for bar-headed geese in viral dispersal. 27 Additional outbreak investigations in wild birds in Mongolia from 2005 to 2011, including bar-headed geese and ruddy shelducks, suggest that birds migrating to and from Mongolia are unlikely to be competent continuous reservoirs of H5N1 HPAI viruses. 11 The subclinical effects of H5N1 HPAI viruses in migratory waterfowl under natural conditions—namely, their ability to migrate—are unknown. Thus far, clade 2.3.2 virus outbreaks have been localized, and whether these local events expand to larger, more widespread areas has yet to be determined. As such, the emergence of clade 2.3.2 viruses should be continually monitored to assess potential spread and adaptation to wild birds.18,27,44
Footnotes
Acknowledgements
We thank Joan Beck, James Doster, and Kira Moresco (SEPRL) and Becky Poulson (Southeastern Cooperative Wildlife Disease Study [SCWDS]) for technical support. Deb Carter (SCWDS) performed TUNEL assays, and Abigail Butler and Patricia Rowe (Athens Diagnostic Laboratory) performed caspase immunohistochemistry.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Institute of Allergy and Infectious Diseases, National Institutes of Health (NIH), Department of Health and Human Services under contract No. HHSN266200700007C, and Agricultural Research Service, Department of Agriculture under project numbers 6612-32000-048-00D and 6612-32000-063-00D, and the Food and Agriculture Organization of the United Nations. The opinions expressed herein are those of the author(s) and do not necessarily reflect the views of any of the funding agencies or organizations.
