Abstract
Vulvo-vaginal epithelial tumors are uncommon in mares, and data on the epithelial-to-mesenchymal transition (EMT) and the tumor-immune microenvironment (TIME) are still lacking. This is a study investigating the equus caballus papillomavirus type 2 (EcPV2) infection state as well as the EMT process and the tumor microenvironment in vulvo-vaginal preneoplastic/ benign (8/22) or malignant (14/22) epithelial lesions in mares. To do this, histopathological, immunohistochemical, transcriptomic, in situ hybridization, and correlation analyses were carried out. Immunohistochemistry quantification showed that cytoplasmic E-cadherin and β-catenin expression as well as nuclear β-catenin expression were features of malignant lesions, while benign/preneoplastic lesions were mainly characterized by membranous E-cadherin and β-catenin expression. Despite this, there were no differences between benign and malignant equine vulvo-vaginal lesions in the expression of downstream genes involved in the canonical and noncanonical wnt/β-catenin pathways. In addition, malignant lesions were characterized by a lower number of cells with cytoplasmic cytokeratin expression as well as a slightly higher cytoplasmic vimentin immunolabeling. The TIME of malignant lesions was characterized by more numerous CD204+ M2-polarized macrophages. Altogether, our results support the hypothesis that some actors in TIME such as CD204+ M2-polarized macrophages may favor the EMT process in equine vulvo-vaginal malignant lesions providing new insights for future investigations in the field of equine EcPV2-induced genital neoplastic lesions.
Keywords
Squamous cell carcinoma (SCC) represents the most common malignant cutaneous tumor in horses, accounting for 7%–37% of all skin lesions. It can develop at any site on the skin and mucosa, although nonpigmented skin and mucocutaneous junctions, such as eyelids and external genitalia of both male and female horses, are the preferential sites.40,46,55
Growing evidence has suggested that equus caballus papillomavirus type 2 (EcPV2) is likely the etiological cause of equine SCCs, including penile and preputial SCC,40,46 as well as equine vulvo-vaginal carcinoma.35,38,46,55 In the last decade, numerous studies have investigated the prevalence and the possible role of EcPV2 in inducing penile and preputial epithelial preneoplastic lesions and both benign and malignant tumors. 46 However, the role of EcPV2 in the pathogenesis of genital lesions in mares remains elusive.17,35,38,43,55 Similar to the equine male counterpart, vulvo-vaginal carcinoma represents a malignant tumor that can occur with a de novo onset or arise from precursor lesions, such as plaques and papillomas.52,53
Recently, different equine SCCs have been reported to undergo the epithelial-to-mesenchymal transition (EMT) process. The transition from an epithelial-to-mesenchymal phenotype plays a pivotal role in processes such as physiological embryogenesis and fibrosis.33,50 The reactivation of this process has been proposed as a pivotal driver of tumor progression, invasiveness, and cancer metastasis.10,28 More specifically, EMT consists of a multistep process, characterized by the activation of the main transcription factors (TWIST, ZEB, SNAIL1, and SLUG), which subsequently leads to the loss of epithelial marker expression (E-cadherin, β-catenin, and cytokeratin), promoting the acquisition of a mesenchymal phenotype (N-cadherin and vimentin expression), finally enabling invasiveness during cancer progression. 11
In the last decades, a research field that has provided new insights into tumor progression and potential therapeutic targets is the tumor-immune microenvironment (TIME). It is becoming more evident that tumors develop in a heterogeneous and dynamic ecosystem, wherein the interactions between different players (i.e., stromal cells, endothelial cells, and immune cells) orchestrate tumor reprogramming and drive cancer progression, representing an interesting network from which new potential therapeutic targets can be identified.20,51 Immune cells represent a particularly interesting component of this microenvironment playing a potentially critical role when employed for both prognostic or therapeutic purposes, as demonstrated by the recent successful application of immunotherapeutic protocols. 5 Although data about the immune microenvironment characterization have been reported in equine SCC, 34 there is still a lack of this information for equine vulvo-vaginal epithelial preneoplastic and neoplastic lesions.
EMT and TIME are 2 different and parallel processes. Indeed, in the last few years, a direct and synergistic interaction between the 2 has been highlighted in different types of cancer.14–16,18,47 In particular, EMT seems to enhance immune evasion by increasing the number of intratumoral myeloid-derived suppressor cells and the expression of PD-L1 in several types of cancer. As a result, the signaling pathways stimulated by molecules involved in tumor-immune suppression could lead to the induction of EMT. 47
It is fairly well known that naturally occurring tumors in domestic animals can provide precious opportunities to study cancer in vivo. To date, several studies investigated the EMT process in different equine tumors,2,4,31,45 but there is still a lack of data regarding equine vulvar SCC. As a matter of fact, most information on the biology and features of preneoplastic and neoplastic epithelial lesions in mares are borrowed from the available studies on penile and preputial epithelial lesions in male horses. Therefore, this study aims to provide insights into the biology of vulvo-vaginal epithelial tumors in mares, focusing in particular on the characterization of EMT and TIME, and on their possible interaction in supporting tumor progression.
Materials and Methods
Case Selection
Cases were retrospectively selected from the archives of the veterinary pathology section of the Department of Veterinary Medicine of the University of Perugia and the Institute of Pathology of the University of Veterinary Medicine of Hannover based on the following inclusion criteria: (1) histological diagnosis of epithelial hyperplasia/dysplasia, papilloma, carcinoma in situ, and SCC; (2) confirmed vulvar or vaginal localization of the lesions; and (3) availability of greater than 0.5 cm2 formalin-fixed, paraffin-embedded (FFPE) tumor tissue per section. When available, surgical margins without neoplastic tissue were selected together with neoplastic tissue for histological and immunohistochemical comparisons.
All samples were used for histopathological and immunohistochemical evaluations. For viral DNA testing, in situ hybridization, and gene expression assessment, only 19 samples (8 benign/preneoplastic lesions (BPL) and 11 SCC) were evaluated due to samples’ availability restrictions. Three SCC cases were excluded due to scarce FFPE material availability and poor genomic quality. In addition, 2–3-cm-diameter samples were collected at the slaughterhouse from 6 macroscopically healthy vulvar and vaginal mucosa. These were used as healthy control group (CTRL) for immunohistochemical analysis of EMT, in situ hybridization, and gene expression analysis.
Histological Evaluations
Slides from the 22 cases were blindly evaluated by 2 board-certified veterinary pathologists (IP and mitotic count (MC)). Diagnoses were reassessed and different histological characteristics were described. A differentiation score was also assigned to malignant tumors (0 = undifferentiated, when it was not possible to see signs of squamous differentiation; 1 = poorly differentiated, when rare signs of squamous differentiation were present; 2 = moderately differentiated, when signs of squamous differentiation are present and occasional keratin pearls are seen; 3 = well differentiated, when squamous differentiation is maintained throughout the tumor). MC was evaluated in 2.37 mm2 32 ; and the presence/absence of vascular invasion, ulceration, and necrosis was recorded. The presence and extent of koilocytosis, hypergranulosis, hyperkeratosis (0 = absent; 1 = mild, when focally present; 2 = moderate, when multifocal areas were observed, but less than 50%; 3 = prominent, when >50% of the epithelium was affected), stroma (0 = absent; 1 = fine fibrovascular stroma; 2 = thin to thick bundles of fibrovascular stroma; 3 = thick to desmoplastic stroma), and inflammation (0 = absent; 1 = mild, when focal area or small multifocal infiltrates <10 inflammatory cells where observed; 2 = moderate, when multifocal infiltrates of <50 cells where observed; 3 = severe, when multifocal aggregates >50 inflammatory cells to diffuse infiltration of inflammatory cells were observed) were also assessed. The different inflammatory cell populations were evaluated semiquantitatively both within the neoplastic tissue and, when possible, in the extratumoral tissue (0 = absent; 1 = mild; 2 = moderate; 3 = severe, as assessed for inflammation). The distribution of inflammatory cells and their localization (predominantly intratumoral, predominantly extratumoral, or both) was evaluated, together with the severity in each compartment (0 = absent; 1 = mild; 2 = moderate; 3 = severe, as assessed for inflammation). The tumors were also classified into immune-infiltrated, immune-altered, and immune-deserted. 25 In case of nonconcordance among the 2 pathologists, data were rediscussed and a final agreement was reached.
DNA Extraction and EcPV2 Detection
The presence of the L1, E2, E6, and E7 genes of EcPV2 was tested in 19 samples and healthy mucosa. DNA was extracted from two 5-µm-thick sections as previously described 6 and quantified by QUBIT 3 (ThermoFisher Scientific, Waltham, MA, United States). A volume of 100 ng of DNA was used for viral gene detection and for assessing DNA amplifiability by beta-2-microglobulin (B2M) gene amplification. Real-time polymerase chain reaction (PCR) was performed as previously described, 44 using 200 nM of the probe, 100 nM of each primer, and Taq DNA Polymerase MasterMix (Biorad Laboratories, Berkeley, CA, United States) with the following thermal profile: 95°C for 10 minutes, then 39 cycles of 95°C for 15 seconds, and 60°C for 60 seconds. Oligonucleotide sequences of primers and probes are reported in Supplemental Table S1. The CFX96 Real-Time System (Biorad Laboratories, Berkeley, CA, United States) was used, setting a Cq of 38 as the cut-off for virus positivity.
RNA Extraction and Viral and Host Gene Expression
Total RNA was extracted from 19 samples and healthy mucosa using 5 FFPE sections (5 µm thickness) using RecoverAll Total Nucleic Acid Isolation Kit for FFPE (Invitrogen, ThermoFisher Scientific, Waltham, MA, United States) according to manufacturer’s instructions. SuperScript IV VILO Master Mix (Invitrogen, ThermoFisher Scientific, Waltham, MA, United States) was used to reverse-transcribe 250 ng of total RNA from each sample that was diluted 1:10 for real-time PCR. Gene expression of L1, E2, E6, and E7 viral genes was evaluated using probes and primers reported in Supplemental Table S1 following the protocol used for viral DNA (previous section). The same protocol was applied to directly test the RNA to exclude possible contaminations by EcPV2 genomic DNA. For host relative gene expression, the following genes were tested: C-X-C motif chemokine ligand 8 (CXCL8), interleukins (IL2, IL17A, IL23A, IL12A, IL12B, and IL10), transforming growth factor-beta (TGFB), interferon-gamma (IFNG), AP-1 transcription factor subunit (FOSL1), lymphoid enhancer-binding factor 1 (LEF1), catenin beta 1 (CTNNB1), hypoxia-inducible factor 1 subunit alpha (HIF1A), von Hippel-Lindau tumor suppressor (VHL), and egl-9 family hypoxia-inducible factors (EGLN1, EGLN2, and EGLN3). Primer sequences are reported in Supplemental Table S2. The Power SYBR Green PCR Master Mix (Applied Biosystems, Thermo Fisher Scientific, Waltham, MA, United States) was used, applying the following thermal profile in the CFX96 Real-Time System (Biorad Laboratories, Berkeley, CA, United States): 95°C for 3 minutes, then 50 cycles of 95°C for 15 seconds, and 60°C for 30 seconds and, finally, the melting curve going up in the range 58°C–95°C with an increment of 0.01°C/s. B2M was utilized as a reference. 45 The primer pairs first used in this study are represented in the table by accession numbers and were designed including an intron or spanning an exon-exon junction through Primer3web tool v. 4.1.0 (https://primer3.ut.ee). Technical replicates were included in the reaction, collecting fluorescence data at the end of the extension step of each cycle and constructing the melting curve. Relative expression was calculated through the 2–ΔΔCq method. For samples with no detectable amplification, a Cq of 42 was chosen as the detection threshold.
In Situ Hybridization
The in situ hybridization (ISH) used in this study was the RNAscope ISH Technology (Advanced Cell Diagnostics, Hayward, CA, United States). The presence of EcPV2 E6/E7 oncogenes was investigated directly on neoplastic FFPE samples. The assay was optimized and performed according to the manufacturer’s instructions. The tissue samples were cut at 4-µm thickness, placed on SuperFrost Plus (Fisher Scientific) glass slides, and deparaffinized in a series of xylene and 100% ethanol steps. On each section, the protease was added and pretreated through heating in the dry oven HybEZ for 1 hour at 60°C. The RNA-specific sequence was identified through the binding with the target probe. The following process was the hybridization of the probe with signal-amplifying molecules in 6 successive steps. For each step, the amplifying solution, up to the complete coverage of the slide, was added and subsequently incubated in the dry oven HybEZ for either 30 minutes for AMP-1, AMP-3, and AMP-5 or 15 minutes for AMP-2, AMP-4, and AMP-6. Between each step, the slide was washed with a 1× wash buffer for 2 minutes at room temperature. The detection of the amplified signal was performed using the chromogenic substrate (Fast RED, Abcam, Cambridge, United Kingdom). Slides were then counterstained with Gill’s hematoxylin for 2 minutes at room temperature. In this study, we used RNAscope 2.5 HD Kit (Advanced Cell Diagnostics, Hayward, CA, United States), according to their prescribed procedure. As positive control, a sample of SCC positive for EcPV2 DNA and RNA, confirmed by histology and molecular biology, was used. The slides were evaluated with a Zeiss Axio Scope A1 microscope (Zeiss, Jena, Germany) at increasing magnifications (10×, 20×, and 40×) and were considered positive if red punctuate dots in epithelial neoplastic cells were detected.
Immunohistochemistry
Immunohistochemistry was carried out as previously described.2,34 Briefly, immunolabeling was performed with standard protocols on serial sections, using antibodies antipan-cytokeratin AE1/AE3, E-cadherin, β-catenin, N-cadherin, vimentin, ZEB-1, TWIST-1, and HIF-1α for EMT and anti-CD3, CD20, FoxP3, MUM1, IBA1, CD204, and MPO for characterizing immune infiltrate. Details about protocols and positive controls are summarized in Supplemental Table S3. Avidin-biotin complex (ABC) peroxidase kit (Vector Labs) was used after secondary antibody application. The immunolabeling was revealed with 3,3-diaminobenzidine tetrahydrochloride (DAB) system (Vector Labs) for pan-cytokeratin AE1/AE3, E-cadherin, β-catenin, N-cadherin, vimentin, ZEB-1, TWIST-1, and HIF-1α and with 3-amino-9-ethilcarbazole (AEC Single Solution, Abcam) for CD3, CD20, FoxP3, MUM1, IBA1, CD204, and myeloperoxidase (MPO). Counterstaining was performed with Meyer’s hematoxylin. Negative controls were run by omitting the primary antibody and incubating sections with TBS.
Healthy vulvar tissues obtained from mares at the slaughterhouse (control group) were used to assess baseline expression of EMT-related markers in a normal vulvar epithelium. Positive cells were evaluated manually by counting 10 evenly distributed fields within the tumor periphery/invasive front at 400× magnification (field of view size: 0.0305 mm2) using a Nikon Eclipse E800 microscope (Nikon Corporation, Japan) with a Nikon PLAN APO lens and equipped with a Camera DIGITAL SIGHT DS-Fi1 (Nikon Corporation, Tokyo, Japan). Pictures were acquired with a DS camera control unit DS-L2 (Nikon Corporation, Japan) and stored in a USB device. For control samples, 10 high-power fields (400×) evenly distributed within the mucosal epithelial layer were analyzed.
Statistical Analysis
Descriptive statistics were used to describe basic features of the data, and values are expressed as medians (Mdn) and interquartile range (IQR). Parametric and nonparametric tests were used to test hypotheses. Mann-Whitney U tests were performed to assess differences among groups. Correlation analysis was performed using the Spearman’s test (ρ). Statistical tests were performed with IBM SPSS (version 21). After normality check of gene expression data through the Shapiro-Wilk test, the nonparametric Kruskal-Wallis test was used to assess differences between CTRL, BPL, and malignant epithelial tumors (MET) groups. The posthoc Dunn’s multiple comparison test was applied, and a p-value (p) threshold was set at .05 for the statistical significance. Data analysis was performed with GraphPad Prism 5.04 (GraphPad Software Inc., La Jolla, CA, United States).
Results
Case Selection, Histological Characterization, and Correlations Among Variables
Twenty-two cases of epithelial vulvo-vaginal lesions were retrospectively retrieved. The median age of both BPL and MET bearing mares was 18 years. All the tumors collected were primary lesions at their first presentation, from nonneutered mares. No staging information nor follow-up information was available for the selected cases.
From the 22 retrospectively selected cases, 14/22 (64%) were diagnosed as METs (13 SCC and 1 adenosquamous carcinoma), 1/22 (4%) was an in situ carcinoma, 4/22 (18%) were papillomas, and 3/22 (14%) were classified as epithelial hyperplasia/dysplasia. In situ carcinoma, papillomas, and epithelial hyperplasia/dysplasia were grouped together as BPLs. Representative images of the BPL and MET groups are shown in Fig. 1a–d. Out of 14 cases (57%) of malignant tumors, 8 were graded as well differentiated, 3 (21%) as moderately differentiated, and 4 (29%) as poorly differentiated. No case was classified as undifferentiated. The median MC of all cases was 29.75 mitoses/2.37 mm2 (IQR = 14.12–43.62) and was 17.5 mitoses/2.37 mm2 (IQR = 5.0–30.0) in the BPL group and 37.5 mitoses/2.37 mm2 (IQR = 24.62–44.37) in MET. The MC was not significantly different between the 2 groups but was higher in cases where ulceration was present (p<.05). Vascular invasion was observed in 3/22 cases (14% of cases), which were all SCCs. Data are summarized in Supplemental Table S4.
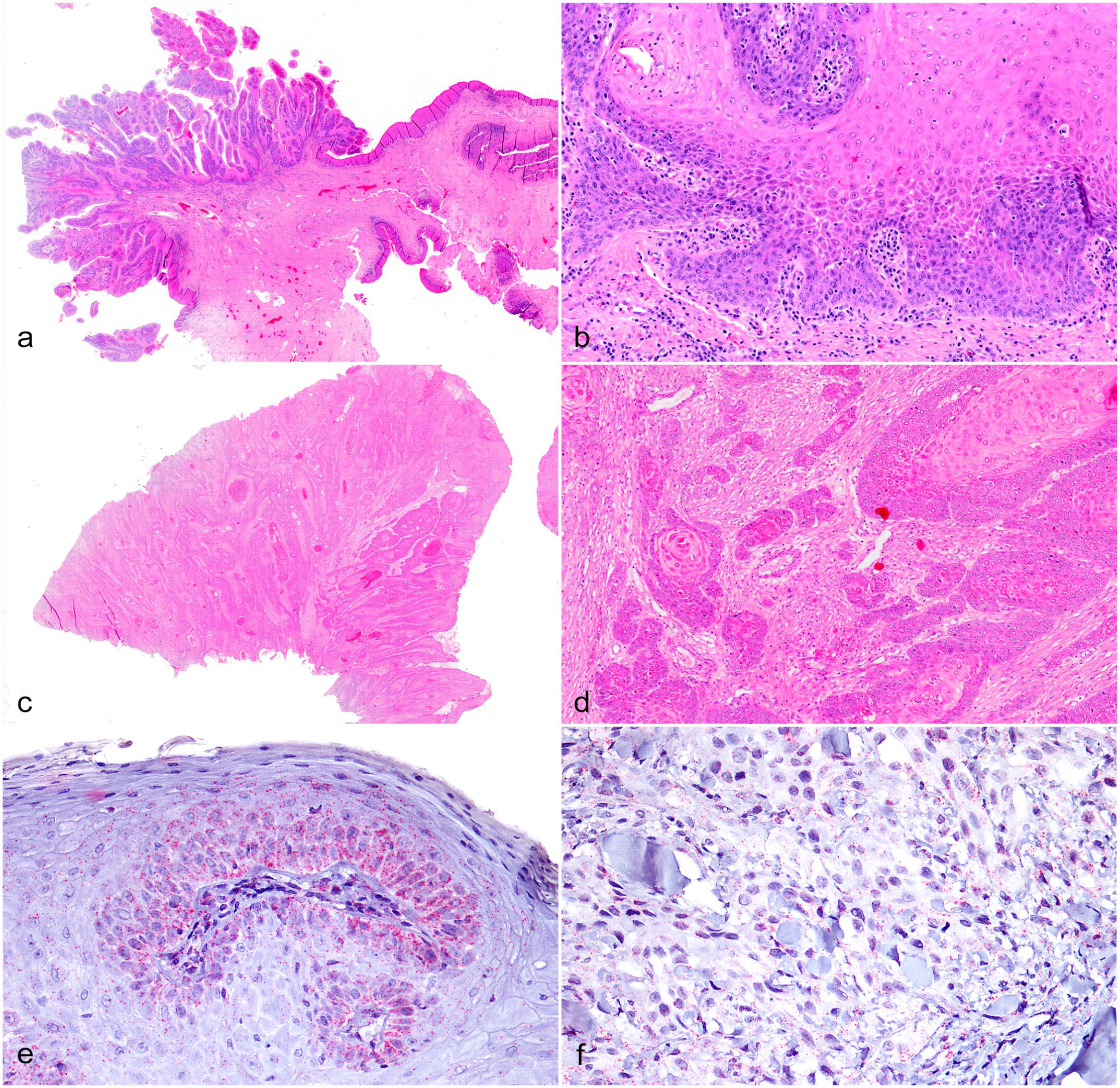
Equine vulvo-vaginal epithelial neoplasia. Vulvo-vaginal mucosa, horse. (a–d) Hematoxylin and eosin. (a) Equine vulvo-vaginal mucosa. Overview of a sample belonging to the benign/preneoplastic lesions (BPL) group. (b) Equine vulvo-vaginal mucosa. Higher magnification of (a). Moderate basal cell proliferation with mild to moderate, multifocal to coalescing, lymphoplasmacytic inflammation. (c) Equine vulvo-vaginal mucosa. Overview of a sample belonging to the malignant epithelial tumors (MET) group. (d) Equine vulvo-vaginal mucosa. Higher magnification of (c). Diffuse desmoplasia. High numbers of mitotic figures. Squamous differentiation and multifocal “keratin pearls.” (e, f) In situ hybridization for E6/E7 oncogenes. (e) Vulvar papilloma. The labeling is represented by magenta dots which appear to be predominantly located in the cytoplasm of basal keratinocytes. (f) Squamous cell carcinoma. Dots are disseminated in the cytoplasm of neoplastic cells.
Further analyzing 22 cases of vulvo-vaginal lesions, the amount of koilocytosis showed a strong inverse correlation with malignancy (p< .001, ρ = –.709), similarly to hypergranulosis, which had a moderately strong inverse correlation (p< .05, ρ = –.524). The presence and extent of hyperkeratosis had a strong correlation with the degree of differentiation (p< .01; ρ = .790). The severity of intratumoral inflammation was also strongly associated with the amount of stroma (p< .001, ρ = .659) and with the overall inflammation of the specimens (p< .001, ρ = .699). On the contrary, the severity of extratumoral inflammation had a strong positive correlation with the degree of differentiation (p < .001, ρ = .895) and with the overall degree of inflammation (p < .001, ρ = .818). The macroscopically normal vulvar mucosae sampled at the slaughterhouse did not show any histopathological alterations.
Vulvo-vaginal Equine MET Invariably Express EcVP-2 Oncogenes, Whereas in BLP Lesions, Viral Replication Was Not Confirmed in All Cases
MET and BPL groups were investigated for the presence of EcPV2 DNA. All samples were positive for EcPV2-L1 viral DNA. Notably, 16 out of 19 (84%) cases were positive for EcPV2-E2 viral DNA. The 3 negative samples included 2 METs and 1 BPL. All cases were positive for EcPV2-E6 and E7 viral DNA (Supplemental Table S5).
Next, MET and BPL groups were investigated in order to verify EcPV2 oncogenes L1, E2, E6, and E7 expression. The L1 gene was expressed (L1+) in 17/19 (89%) samples, with 2 BPLs being negative for L1 expression (L1–). E2 expression was detected in 13/19 (68%) samples; 3 METs and 3 BPLs were negative. On the contrary, 17/19 (89%) samples were positive for E6 as well as for E7, while 2 BPL samples were negative. Out of 19, 16 (84%) samples expressed both E6 and E7 oncogenes. Interestingly, all METs expressed both E6 and E7 mRNAs, whereas expression was detected in only 5/8 (62%) BPLs (Supplemental Table S5). Healthy mucosa samples (CTRL group) were negative for EcPV2-L1 viral DNA detection.
Viral gene expression was investigated in 19 tumors using in situ hybridization. E6/E7 oncogenes expression was detected in 10/19 (53%) samples. Out of 11, 8 (73%) METs were positive, whereas only 2/8 (25%) BPLs were positive. Dots were observed within the cytoplasm of epithelial cells, in both MET and BPL samples (Fig. 1e, f, Supplemental Table S6). The in situ hybridization for E6/E7 oncogenes was negative in the CTRL group.
Intermediate Filaments Rearrangements, “Cadherin Switching” and Activation of the wnt/β-Catenin Pathways Are Features of the EMT Process in Equine Vulvo-vaginal MET
In order to verify if EMT is a feature of equine vulvo-vaginal malignant neoplasia, the expression of the main EMT markers was investigated in tumor cells. Representative pictures of cytokeratin and vimentin immunolabelings in equine vulvo-vaginal MET can be found in Fig. 2a, b. Quantification of cells immunolabeled for intermediate filaments such as cytokeratins (pan-cytokeratin AE3/AE1) revealed a significantly (p ≤ .001) lower number of cells expressing cytoplasmic cytokeratin in the invasive front of METs when compared with BPL and CTRL (Fig. 3a). On the contrary, the number of cells showing cytoplasmic vimentin immunolabeling in the invasive front of METs was slightly higher compared with BPL and CTRL (Fig. 3b).
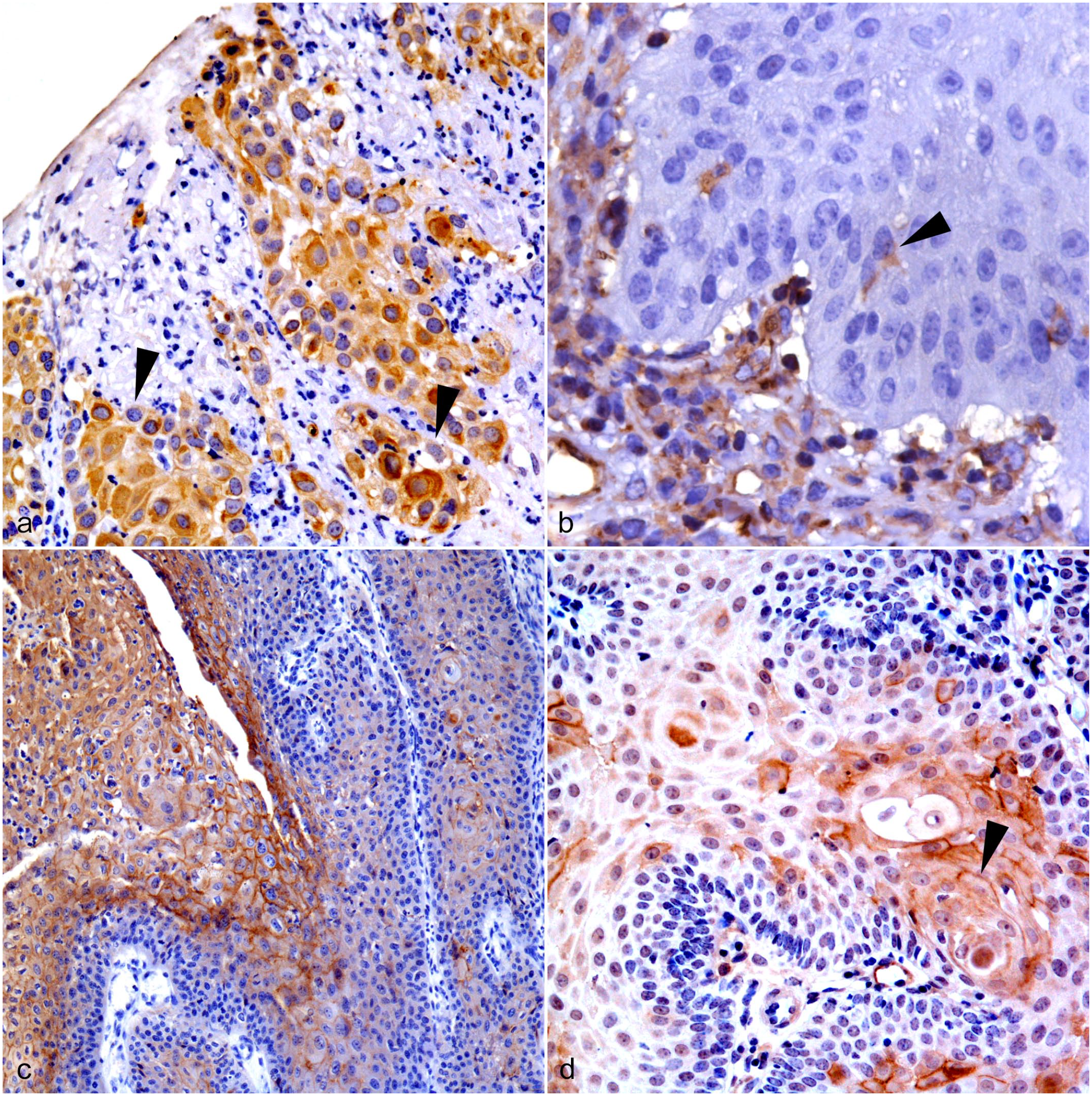
Equine vulvo-vaginal epithelial neoplasia. Vulvo-vaginal mucosa, horse. Immunohistochemistry for (a) cytokeratin AE1/AE3, (b) vimentin, (c) E-cadherin, and (d) N-cadherin in equine vulvo-vaginal malignant epithelial tumors (MET). (a) There are moderate numbers of cytokeratin AE1/AE3 immunolabeled tumor cells. There are a few tumor cells that do not show cytokeratin AE1/AE3 cytoplasmic immunolabeling (arrowheads). (b) There are occasional vimentin immunolabeled tumor cells with weak cytoplasmic labeling (arrowhead). (c) On the left, there are moderate to high numbers of E-cadherin immunolabeled tumor cells with membranous labeling. From the middle to the right, there are high numbers of cells that do not show membranous E-cadherin immunolabeling. (d) There are moderate numbers of membranous N-cadherin immunolabeled tumor cells (arrowhead).
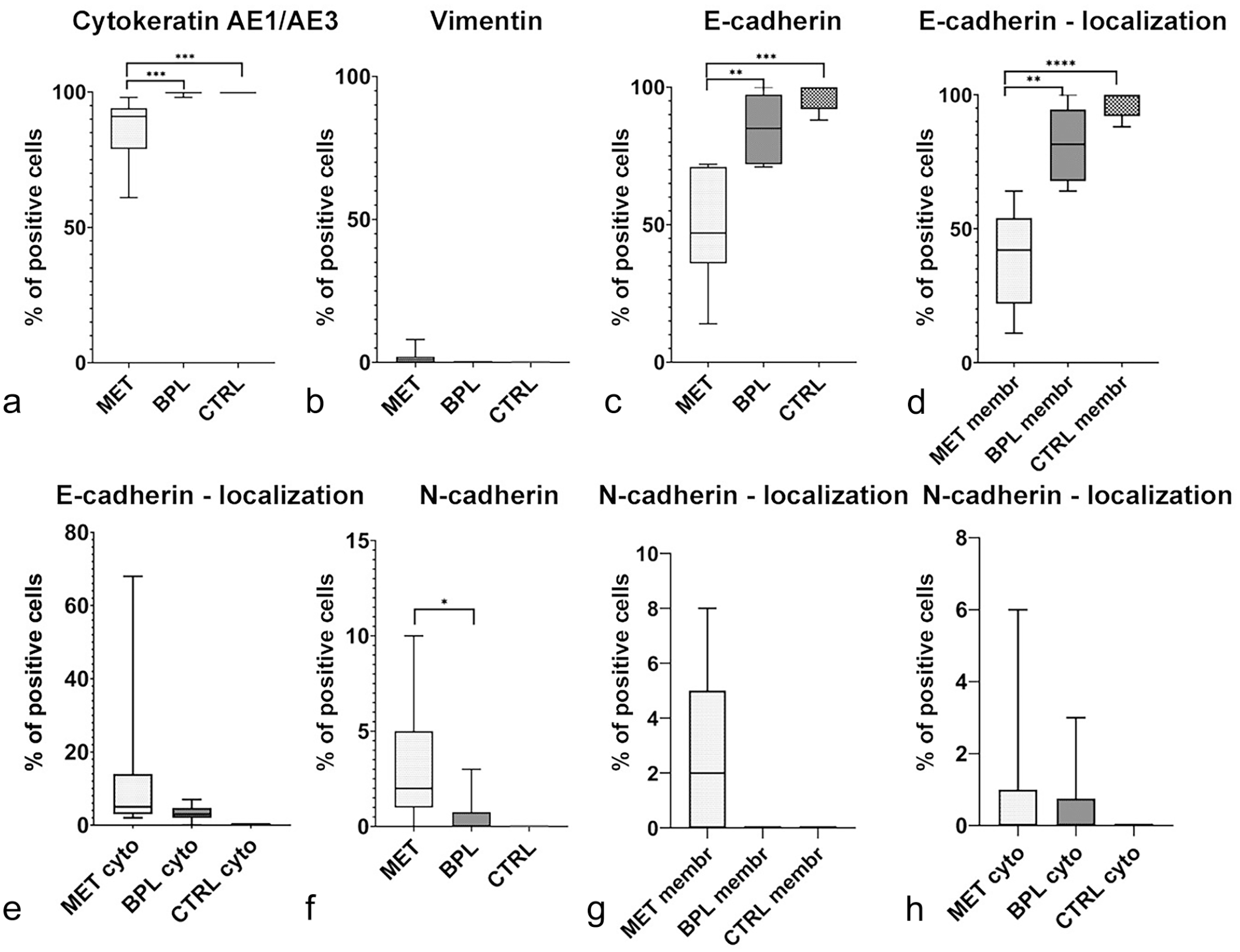
Frequency of cytokeratin AE1/AE3, vimentin, E-cadherin, and N-cadherin expression and subcellular localization in equine vulvo-vaginal benign/preneoplastic lesions (BPL), malignant epithelial tumors (MET), and healthy mucosa (CTRL). (a) Quantitative analysis. Comparison of numbers of cytokeratin AE1/AE3 immunolabeled tumor cells across BPL, MET, and CTRL groups (Mann-Whitney U; ***p ≤ .001). (b) Quantitative analysis. Comparison of numbers of vimentin immunolabeled tumor cells across BPL, MET, and CTRL groups. (c) Quantitative analysis. Comparison of numbers of E-cadherin immunolabeled tumor cells across BPL, MET, and CTRL groups (Mann-Whitney U; **p ≤ .01; ***p ≤ .001). (d) Quantitative analysis. Comparison of numbers of tumor cells with membranous E-cadherin immunolabeling across BPL, MET, and CTRL groups (Mann-Whitney U; **p ≤ .01; ****p ≤ .0001). (e) Quantitative analysis. Comparison of numbers of tumor cells with cytoplasmic E-cadherin immunolabeling across BPL, MET, and CTRL groups (Mann-Whitney U). (f) Quantitative analysis. Comparison of numbers of N-cadherin immunolabeled tumor cells across BPL, MET, and CTRL groups (Mann-Whitney U; *p < .05). (g) Quantitative analysis. Comparison of numbers of tumor cells with membranous N-cadherin immunolabeling across BPL, MET, and CTRL groups. (h) Quantitative analysis. Comparison of numbers of tumor cells with cytoplasmic N-cadherin immunolabeling across BPL, MET, and CTRL groups (Mann-Whitney U). Data are shown as box and whisker plots. The bounds of the box plot indicate the 25th and 75th percentiles, the bar indicates medians, and the whiskers indicate minima and maxima. Membr, membranous; cyto, cytoplasmic.
Focusing on adhesion molecule expression, E-cadherin quantification revealed a significantly lower overall number of cells expressing E-cadherin in the invasive front of METs when compared with BPL (p ≤ .01) and CTRL (p ≤ .001). METs had a significantly lower number of cells with a membranous E-cadherin immunolabeling than BPL (p ≤ .01) and CTRL (p ≤ .0001), whereas the number of cells with aberrant cytoplasmic E-cadherin expression was higher in METs and BPLs compared with the CTRL (Fig. 3c–e). On the contrary, quantification of N-cadherin-expressing cells revealed that the number of cells immunolabeled for N-cadherin was significantly higher in METs compared with BPL (p < .05). In particular, the number of cells with membranous or cytoplasmic N-cadherin immunolabeling was higher in METs compared with BPL and the CTRL (Fig. 3f–h). Representative images of E- and N-cadherin immunolabelings in equine vulvo-vaginal MET can be found in Fig. 2c, d. Despite evidence of intermediate filaments rearrangement and cadherin switching, quantification of the immunolabeling for transcription factors involved in the EMT process showed a lack of expression of ZEB-1 in all samples from all groups. On the contrary, TWIST-1 was detected in only 4 samples from the MET group and was mainly nuclear (Supplemental Figure S1). Additional transcription factors involved in the EMT process, such as β-catenin and HIF-1α, were subsequently investigated.
Representative pictures of β-catenin and HIF-1α immunolabelings in equine vulvo-vaginal MET can be found in Fig. 4a, b. The number of cells expressing β-catenin in the invasive front of METs was significantly lower than in BPL (p ≤ .01) and CTRL (p ≤ .001). The number of cells with a membranous β-catenin expression was significantly lower in METs compared with BPL (p ≤ .01) and CTRL (p ≤ .001), whereas the number of cells expressing nuclear β-catenin was significantly higher in METs than in BPL (p ≤ .01). Interestingly, the number of cells with a cytoplasmic β-catenin expression was higher in both METs and BPLs compared with the normal mucosa (Fig. 5a–d).
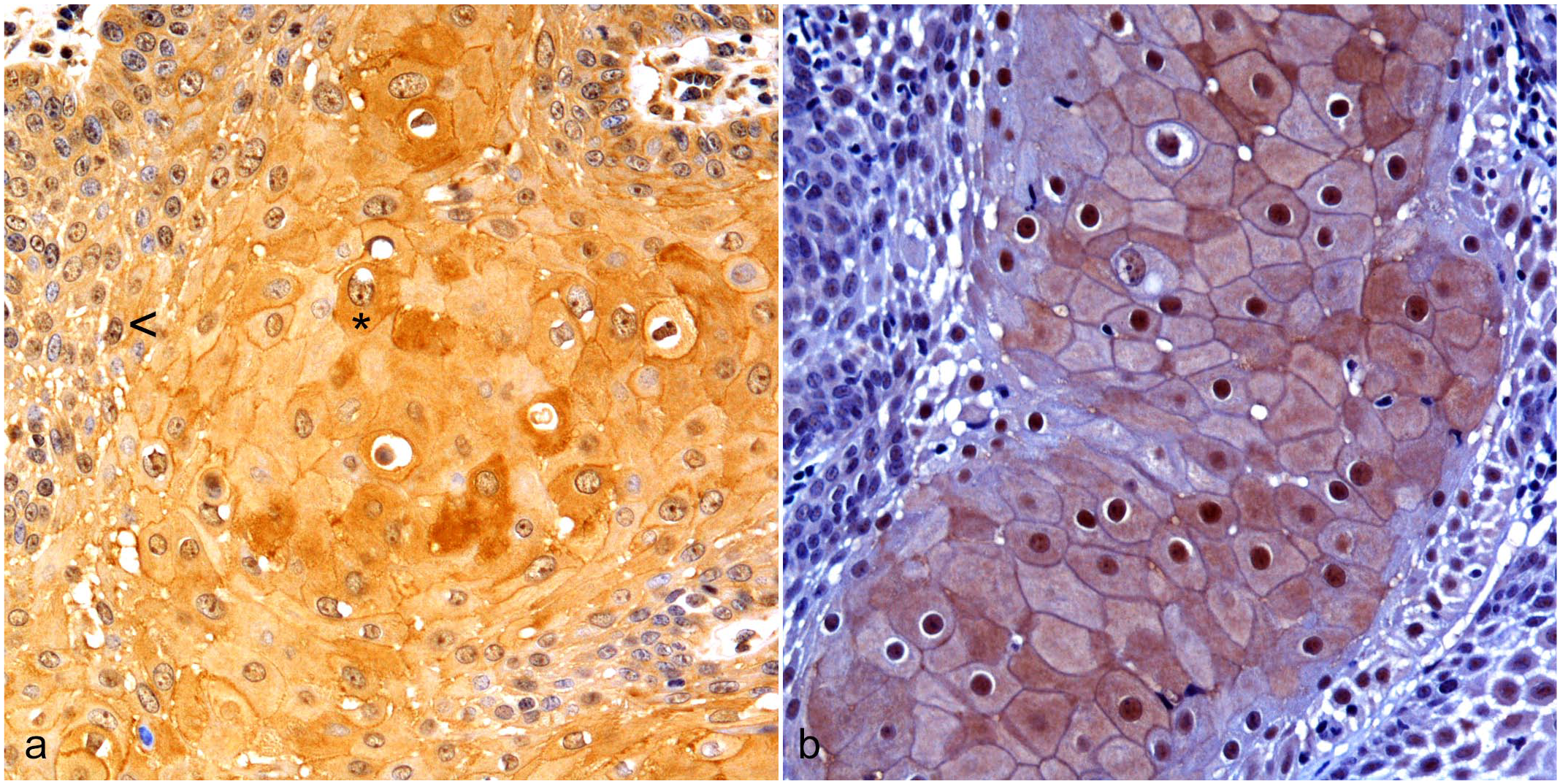
Equine vulvo-vaginal malignant epithelial neoplasia. Vulvo-vaginal mucosa, horse. Immunohistochemistry for (a) β-catenin and (b) hypoxia-inducible factor (HIF)-1α. (a) There are moderate numbers of β-catenin immunolabeled tumor cells. A few tumor cells show weak nuclear immunolabeling (arrowhead), whereas occasional tumor cells have cytoplasmic labeling (asterisk). (b) There are high numbers of HIF-1α immunolabeled tumor cells with frequent nuclear labeling.
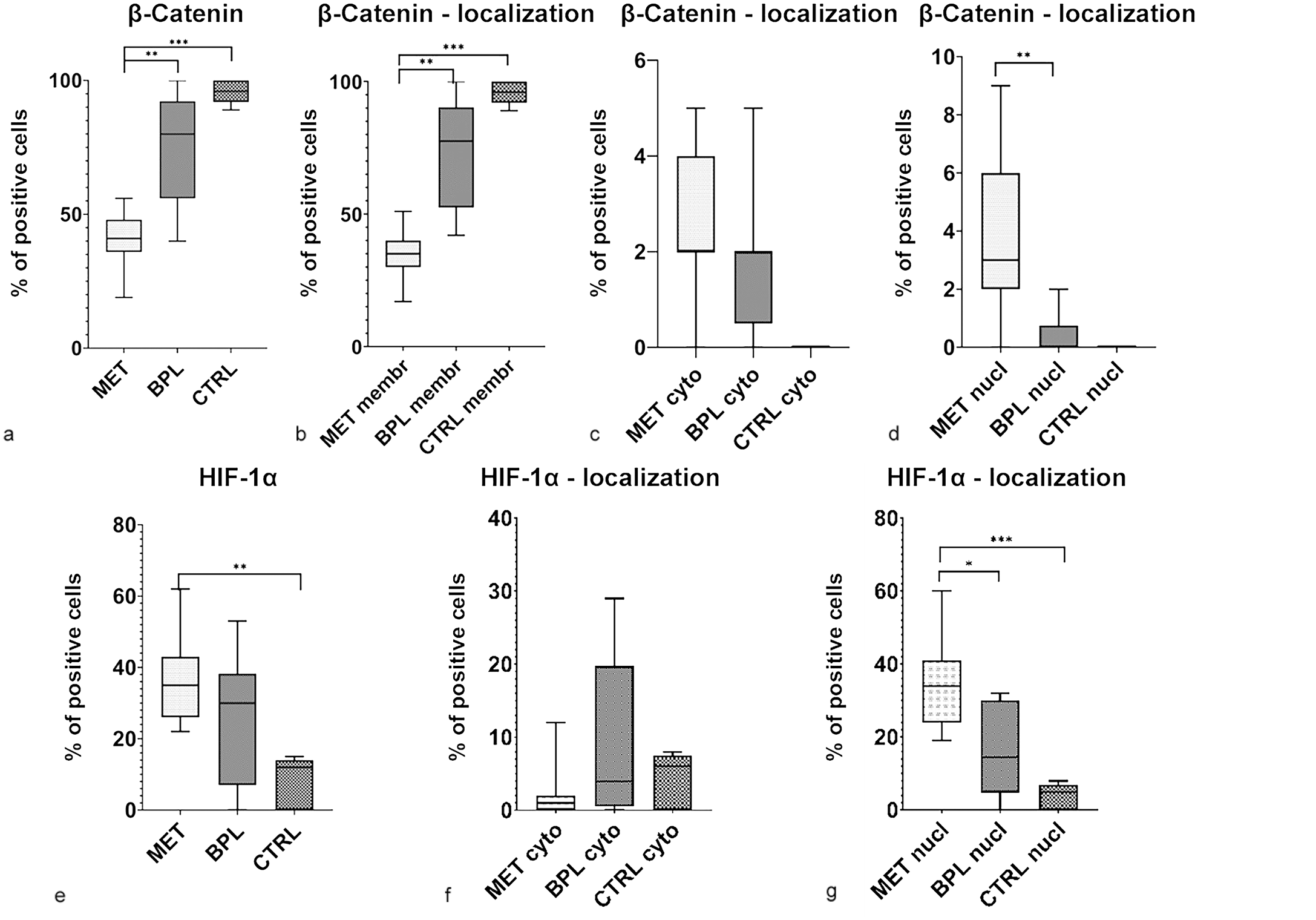
Frequency of β-catenin and hypoxia-inducible factor (HIF)-1α expression and subcellular localization in equine vulvo-vaginal benign/preneoplastic neoplastic lesions (BPL), malignant epithelial tumors (MET), and healthy mucosa (CTRL). (a) Quantitative analysis. Comparison of numbers of β-catenin immunolabeled tumor cells across BPL, MET, and CTRL groups (Mann-Whitney U; **p ≤ .01; ***p ≤ .001). (b) Quantitative analysis. Comparison of numbers of tumor cells with membranous β-catenin immunolabeling across BPL, MET, and CTRL groups (Mann-Whitney U; **p ≤ .01; ***p ≤ .001). (c) Quantitative analysis. Comparison of numbers of tumor cells with cytoplasmic β-catenin immunolabeling across BPL, MET, and CTRL groups (Mann-Whitney U). (d) Quantitative analysis. Comparison of numbers of tumor cells with nuclear β-catenin immunolabeling across BPL, MET, and CTRL groups (Mann-Whitney U; **p ≤ .01). (e) Quantitative analysis. Comparison of numbers of HIF-1α immunolabeled tumor cells across BPL, MET, and CTRL groups (Mann-Whitney U; **p ≤ .01). (f) Quantitative analysis. Comparison of numbers of tumor cells with cytoplasmic HIF-1α immunolabeling across BPL, MET, and CTRL groups (Mann-Whitney U). (g) Quantitative analysis. Comparison of numbers of tumor cells with nuclear HIF-1α immunolabeling across BPL, MET, and CTRL groups (Mann-Whitney U; *p < .05; ***p ≤ .001). Data are shown as box and whisker plots. The bounds of the box plot indicate the 25th and 75th percentiles, the bar indicates medians, and the whiskers indicate minima and maxima. Membr, membranous; cyto, cytoplasmic; nucl, nuclear.
HIF-1α expression was evaluated based on previous findings that reported the involvement of HIF-1α in the EMT process in equine tumors. 2 Vulvo-vaginal METs displayed a significantly (p ≤ .01) higher number of cells expressing HIF-1α compared with normal mucosa. Interestingly, BPLs had the highest number of cells with a cytoplasmic HIF-1α expression. On the contrary, the number of cells expressing nuclear HIF-1α was significantly higher in METs than in BPLs (p < .05) or CTRL (p ≤ .001; Fig. 5e–g). Taken together, these findings were suggestive of an EMT process in vulvo-vaginal METs. Identification of β-catenin nuclear translocation and the HIF-1α nuclear and cytoplasmic expression in the malignant and nonmalignant lesions, respectively, prompted us to further investigate the expression of selected downstream pathway-related genes.
Investigation of downstream genes of noncanonical wnt/Ca2+ and canonical wnt/β-catenin pathways showed that FOSL-1 (p < .05) and LEF-1 (p ≤ .01) were significantly more expressed in METs compared with the CTRL group, while only LEF-1 was also significantly more expressed in BPLs compared with CTRL (p < .05). CTNNB1 gene expression displayed no differences among the groups. Subsequently, investigation of the gene expression of enzymes involved in the cytoplasmic degradation of HIF-1α, namely, EGNL1, EGNL2, EGNL3, and VHL revealed a significantly higher expression of EGNL3 (p < .05) and VHL (p < .05) in the BPL group compared with CTRL lesions. Interestingly, HIF-1A gene expression was significantly higher in both MET (p < .05) and BPL (p ≤ .01) groups compared with CTRL (Fig. 6).
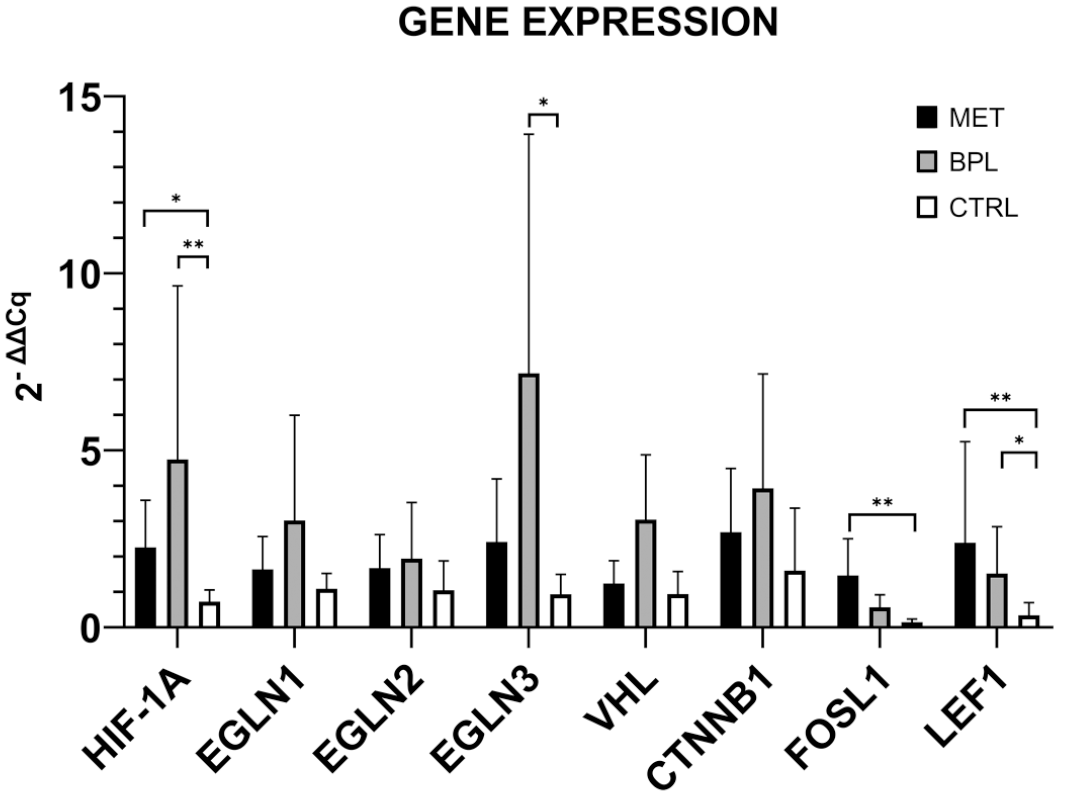
Gene expression levels of hypoxia inducible factor (HIF)-1α and wnt/β-catenin pathways-related genes in equine vulvar lesions samples. Differences, malignant epithelial tumors (MET) versus control (CTRL), benign/preneoplastic neoplastic lesions (BPL) versus CTRL, and MET versus BPL, were evaluated using the Kruskal-Wallis test and applying the posthoc Dunn’s multiple comparisons. The asterisks indicate the statistical significance: *p < .05 and **p < .01.
Spearman’s correlation test analysis of the epithelial marker expression revealed that there were moderately strong positive correlations between the number of cytokeratin-expressing cells and E-cadherin (p ≤ .001, ρ = .793) and β-catenin (p ≤ .01, ρ = .673) immunolabeled cells as well as a there was a moderate positive correlation (p < .05, ρ = .535) between vimentin and N-cadherin expression. Furthermore, moderate negative correlations (p ≤ .01, ρ = –.641) was also detected between the number of β-catenin and vimentin immunolabeled cells as well as the numbers of HIF-1α and E-cadherin immunolabeled cells (p < .05, ρ = –.520). Finally, the number of vimentin immunolabeled cells showed a moderate negative correlation (p ≤ .01, ρ = –.641) with β-catenin immunolabeled cells.
Subcellular protein expression was also investigated for correlations. Membranous β-catenin had a strong positive and moderate negative correlation with membranous (p ≤ .001, ρ = .840) and cytoplasmic (p < .05, ρ = –.479) E-cadherin immunolabeling, respectively. On the contrary, membranous E-cadherin immunodetection showed a moderately strong negative correlation with nuclear β-catenin (p < .05, ρ = –.569). In addition, cytokeratin immunolabeling showed moderately strong positive correlations with membranous E-cadherin (p ≤ .001, ρ = .785) and membranous β-catenin (p ≤ .001, ρ = .717), whereas nuclear β-catenin immunodetection showed a moderate negative correlation (p ≤ .01, ρ = –.647). On the contrary, there was a moderate negative correlation between the number of vimentin immunolabeled cells and membranous E-cadherin (p < .05, ρ = –.515). In addition, there was a moderate negative correlation (p ≤ .01, ρ = .604) between membranous E-cadherin and nuclear HIF-1α immunolabeling. There was also a moderate negative correlation between the number of N-cadherin immunolabeled cells (p < .05, ρ = –.506) and cytoplasmic HIF-1α immunolabeling, whereas there was a moderate positive correlation (p≤ .01, ρ = .601) with nuclear HIF-1α. Furthermore, nuclear HIF-1α also showed a moderate positive correlation (p < .05, ρ = .044) with the number of vimentin immunolabeled cells as well as with nuclear β-catenin immunodetection (p ≤ .01, ρ = .604).
On the transcriptome level, there was a moderately strong direct correlation (p ≤ .001, ρ = .785) between LEF1 and CTNNB1 gene expression.
When analyzing previously investigated features, such as mitoses and differentiation of tumor cells, we found that the overall number of vimentin immunolabeled cells showed a moderately strong negative correlation with tumor differentiation (p < .05, ρ = –.637). There was a strong negative correlation (p ≤ .01, ρ = –.855) between TGFB gene expression and tumor differentiation. On the contrary, there was a moderately strong positive correlation (p ≤ .001, ρ = .708) between FOSL1 and the number of mitoses. Furthermore, the extent of koilocytosis showed a moderately strong positive correlation with overall numbers of cells expressing either cytokeratin (p ≤ .01, ρ = .668) or E-cadherin (p < .05, ρ = .496), while it was negatively correlated with overall numbers of vimentin immunolabeled cells (p < .05, ρ = –.547). In addition, the overall number of HIF-1α-positive cells was associated with tumors where vascular invasion was observed (p < .05).
In conclusion, our last analysis showed a moderately strong negative correlation among the overall number of E-cadherin immunolabeled cells and the extent of tumor inflammation (p < .05, ρ = –.495) and the severity of tumoral inflammation (p < .05, ρ = –.499; p ≤ .01, ρ = –.683), as well as the overall number of β-catenin immunolabeled cells showed a moderately strong negative correlation with the severity of intratumoral inflammation (p ≤ .01, ρ = –.606).
Vulvo-vaginal Equine Papillomavirus-Induced Carcinomas Show a CD204-rich Intratumoral Immune Environment, Associated With Higher MC and the Presence of Ulceration
Once we demonstrated that equine vulvar METs undergo the EMT process, we wanted to characterize the tumor microenvironment by investigating the tumor-associated immune infiltrates. Immune infiltrates in both intratumoral/peritumoral tissue as well as extratumoral tissue were evaluated in all the cases belonging to MET and BPL groups. Extratumoral tissue was available in 18/22 cases. Some markers could not be investigated due to the lack of tissue caused by serial recuts.
All METs were characterized by a mild to moderate infiltration of intratumoral inflammatory cells, whereas, in the BPL group, the intratumoral inflammation varied from absent to moderate. The classification based on T cell infiltration revealed that 5/14 (36%) METs were immune-deserted, 8/14 (57%) showed an immune-altered profile of infiltration, and only one (7%) was infiltrated (Figs. 7a, 8a).
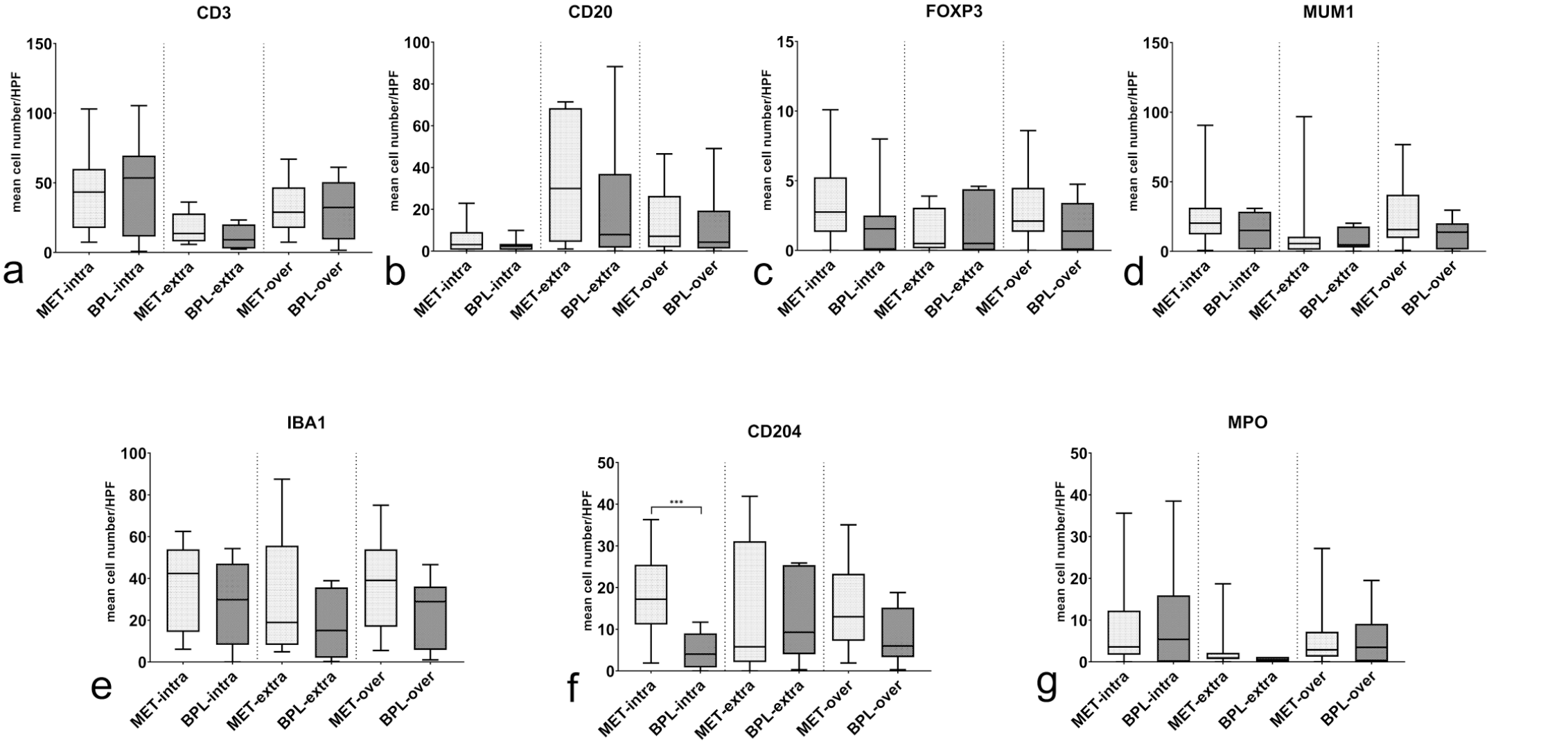
Number of intratumoral, extratumoral, and overall positive immune cells in benign preneoplastic lesions (BPL) and malignant epithelial tumors (MET). (a) Quantitative analysis. Comparison of the number of CD3 immunolabeled immune cells (Mann-Whitney U) (b) Quantitative analysis. Comparison of the number of CD20 immunolabeled immune cells (Mann-Whitney U). (c) Quantitative analysis. Comparison of the number of FOXP3 immunolabeled immune cells (Mann-Whitney U). (d) Quantitative analysis. Comparison of number of MUM1 immunolabeled immune cells (Mann-Whitney U). (e) Quantitative analysis. Comparison of the number of IBA1 immunolabeled immune cells (Mann-Whitney U). (f) Quantitative analysis. Comparison of the number of CD204 immunolabeled immune cells (Mann-Whitney U ***p ≤ .001). (g) Quantitative analysis. Comparison of the number of myeloperoxidase (MPO) immunolabeled immune cells (Mann-Whitney U). Data are shown as box and whisker plots. The bounds of the box plot indicate the 25th and 75th percentiles, the bar indicates medians, and the whiskers indicate minima and maxima. Intra, intratumoral; extra, extratumoral; over, overall.
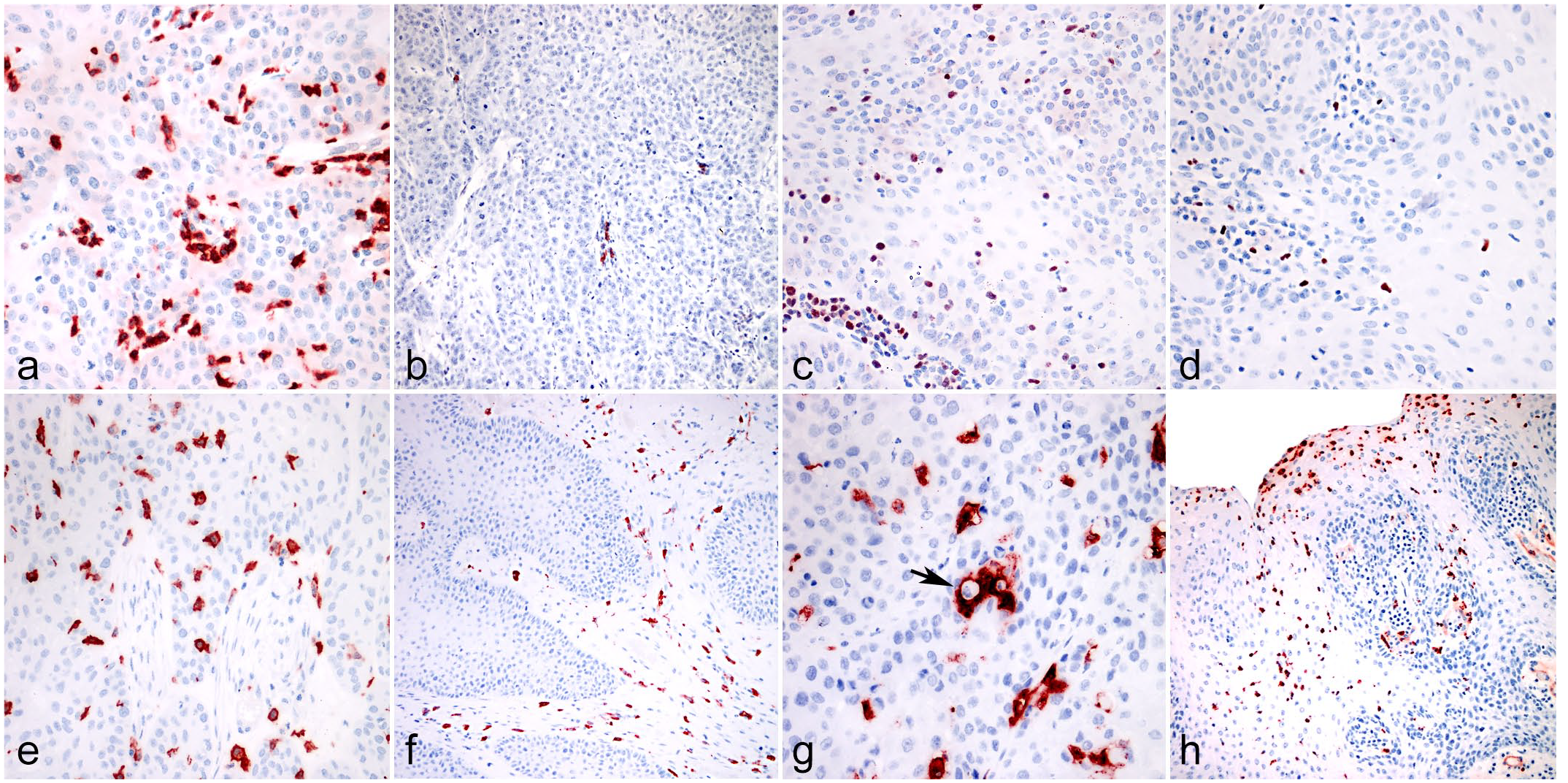
Equine vulvo-vaginal epithelial neoplasia. Vulvo-vaginal mucosa, horse. Immunohistochemistry for (a) CD3, (b) CD20, (c) MUM-1, (d) Foxp3, (e) IBA1, (f, g) CD204, and (h) myeloperoxidase (MPO). (a) Equine vulvo-vaginal squamous cell papilloma. In immune-altered tumors, CD3+ lymphocytes were present in peritumoral stroma and usually did not infiltrate among neoplastic cells. (b) Equine poorly differentiated vulvo-vaginal squamous cell carcinoma. CD20+ B cells were more often localized in the extratumoral tissues, whereas only a few cells were observed in direct contact with tumor cells. (c) Equine well-differentiated vulvo-vaginal squamous cell carcinoma. The number of MUM1+ cells was higher in areas in direct contact with the tumor and occasional positive cells could also be seen among neoplastic cells. (d) Equine well-differentiated vulvo-vaginal squamous cell carcinoma. The number of Foxp3+ cells did not show differences among intratumoral and extratumoral tissues. Foxp3+ cells were often seen infiltrating among tumor cells. (e) Equine poorly differentiated vulvo-vaginal squamous cell carcinoma. IBA1+ cells were often scattered among tumor cells. (f) Equine vulvo-vaginal papilloma. In benign/preneoplastic lesions, CD204+ cells were often limited to extratumoral stroma and did not infiltrate among neoplastic cells. (g) Equine well-differentiated vulvo-vaginal squamous cell carcinoma. Intratumoral CD204+ cells were often intermingled with neoplastic cells. Occasionally, CD204+ cells showed phagocytosis of neoplastic cells (arrow). (h) Equine vulvo-vaginal papilloma. MPO+ cells (mostly neutrophils) were observed predominantly near areas of ulceration.
Comparing the quantity of intratumoral and extratumoral positive cells, results showed that CD3-, MUM1- (Figs. 7d, 8c), and MPO-positive cells (Figs. 7g, 8h) were higher within the intratumoral compartment (p < .001, p= .001, and p < .05, respectively). Instead, CD20 was higher in the extratumoral tissues, whereas few B cells were observed within the neoplastic tissue (Figs. 7b and 8b; p < .001; Supplemental Figure S2). No statistically significant differences were observed for FOXP3 (Figs. 7c, 8d) and IBA1 (Figs. 7e, 8e).
When comparing the expression of the immune markers in the BPL and MET groups, the expression of intratumoral CD204+ cells was significantly higher in the MET group when compared with the BPL group (p = .001; Figs. 7f, 8f–g). No association was observed with the other markers in both intratumoral and extratumoral locations. Moreover, the number of intratumoral CD204+ cells was the only variable that showed a moderate positive correlation with the number of mitoses (p < .05, ρ = .507). Intratumoral CD204+ cells were also associated with the presence of ulceration (p < .05).
The number of intratumoral CD3+ cells showed a moderately strong positive correlation with the number of intratumoral CD20+ cells (p < .05, ρ = .554). It showed a negative association with intratumoral MUM1 (p = .01, ρ = –.538). Intratumoral MUM1 was, in turn, associated with intratumoral CD20 expression (p < .05, ρ = .507). Also, extratumoral CD3 showed a strong positive correlation with extratumoral CD20 expression (p< .01, ρ = .701). Extratumoral CD204+ cells were instead moderately positively associated with extratumoral CD20 and extratumoral CD3 expression (p < .05, ρ = .613 and p < .05, ρ = –.521, respectively). A very strong positive correlation was observed between extratumoral MUM1 and extratumoral FoxP3 (p < .001, ρ = .828), whereas a moderately strong positive correlation was present between extratumoral MUM1 and both extratumoral CD20 (p < .05, ρ = .602), and extratumoral MPO. extratumoral MPO was also positively correlated with extratumoral CD3 (p < .05, ρ = .594). Representative images of different immune markers in METs and BPLs are reported in Supplemental Figure S3.
Gene expression analysis revealed that IL17A expression was significantly higher (p < .001) in METs when compared with both BPL and CTRL. Moreover, CXCL8 (p < .01), IFNG (p < .05), IL12A (p < .05), and IL23 (p < .05) expression was significantly higher in METs when compared with CTRL. Furthermore, a significant upregulation of IFNG (p < .05), IL10 (p < .01), IL12A (p < .05), IL23 (p < .05), and TGFB (p < .05) as well as a downregulation of IL2 (p < .05) was identified in BPL samples compared with CTRL (Fig. 9). The correlation of immune cell markers with RNA expression was observed only for the expression of MPO, which was positively correlated with the expression of IL12B (p < .001, ρ = 0.921) and IL17A (p < .05, ρ = .560). The protein expression of CD204 was instead inversely correlated with the expression of IL10 (p < .05, ρ = –.515). Also, IBA1 expression was positively correlated with the expression of IL12B (p < .05, ρ = .604). Gene expression, comparing 3 groups (MET, BPL, and CTRL), revealed a significantly higher expression of CXCL8 (p < .01), IFNG (p < .05), IL12A (p < .05), and IL23 (p < .05) when comparing METs with CTRL.
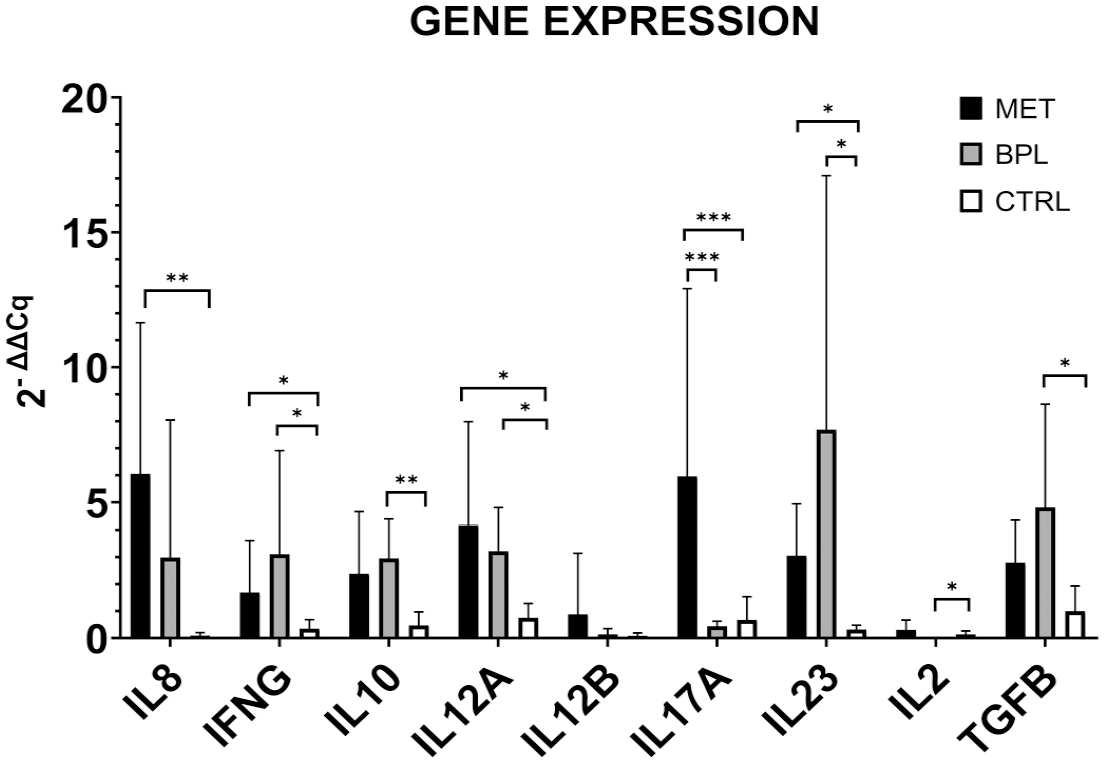
Gene expression levels of immune infiltrate-related targets in equine vulvar lesions samples. Differences, malignant epithelial tumors (MET) versus control (CTRL), benign/preneoplastic neoplastic lesions (BPL) versus CTRL, and MET versus BPL, were evaluated using the Kruskal-Wallis test and applying the posthoc Dunn’s multiple comparisons. The asterisks indicate the statistical significance: *p < .05, **p < .01, and ***p < .001.
Intratumoral CD204+ Cells Are Associated With the Activation of EMT, Particularly With the Process of Cadherin Switching and Nuclear Expression of HIF-1α
The expression of intratumoral CD204 was negatively associated with the expression of different molecules involved in the EMT process, namely, membranous E-cadherin (p < .01, ρ = –.592) membranous β-catenin (p < .01, ρ = –.620), and cytokeratin (p < .01, ρ = –.587). On the contrary, the expression of intratumoral CD204 was positively associated with the membranous expression of N-cadherin (p < .05, ρ = –.457) and with nuclear expression of HIF-1α (p < .05, ρ = –.537).
Discussion
Vulvo-vaginal epithelial tumors are uncommon in mares, and data on these tumors are still lacking. The aim of this study is to characterize vulvo-vaginal epithelial preneoplastic and neoplastic lesions, focusing in particular on the EMT and on the TIME. EMT is a highly coordinated sequential biological process in which epithelial cells lose epithelial biomarker expression and assume a metastable phenotype characterized by mesenchymal biomarker expression.3,11,50 This process has been investigated in different equine tumors,2,4,45 but never in genital epithelial preneoplastic and neoplastic lesions of mares.
This study revealed an overall lower number of cells expressing epithelial markers at the invasive front of METs together with a higher number of cells expressing mesenchymal markers, suggesting an EMT process. Cytoplasmic E-cadherin and β-catenin as well as nuclear β-catenin expression were features of METs, while BPLs were mainly characterized by membranous E-cadherin and β-catenin expression. It is noteworthy to consider that membranous β-catenin localization was positively and negatively correlated with E-cadherin expression on the membrane and in the cytoplasm, respectively, as well as nuclear β-catenin was negatively correlated with membranous E-cadherin expression. Since subcellular translocations of E-cadherin and β-catenin are known to favor the induction of the EMT process,3,41 we postulate from our results that one of the triggers from a benign to a malignant transformation in equine vulvo-vaginal tumors might be represented by the loss of membranous E-cadherin and β-catenin expression as well as by β-catenin nuclear translocation. Nevertheless, our results showed that the expression of downstream genes involved in the canonical and noncanonical wnt/β-catenin pathways was not significantly different between benign and malignant equine vulvo-vaginal tumors. However, future studies are warranted to investigate the gene expression of these 2 pathways more in depth.
Epithelial cells undergoing the EMT process gradually lose cell-cell adhesion molecules, modulate their polarity, and rearrange their cytoskeleton, which becomes dynamic and flexible following the replacement of cytokeratin by vimentin.28,39 Similarly, in our study, cytoplasmic cytokeratin expression was correlated with membranous E-cadherin and β-catenin as well as it was negatively correlated with nuclear β-catenin expression. Furthermore, cytoplasmic vimentin expression was negatively correlated to E-cadherin membranous localization. These findings suggest that E-cadherin and β-catenin translocations influence, or are influenced by, the intermediate filament rearrangements, favoring the EMT process in malignant equine vulvo-vaginal tumors and, thus conferring migratory and invasive features, typical of malignant tumors.3,28,39 In our study, METs also showed another hallmark of EMT known as “cadherin switching” 54 compared with BPL or control tissues.
Another distinctive feature of METs detected in this study was the higher number of cells with nuclear HIF-1α expression compared with BPL or control tissues. The EMT process is reported to be triggered not only by wnt/β-catenin pathways activation or by other transcription factors such as TWIST-1 or ZEB but also by the activation of the HIF-1α pathway, which confers increased tumor cell invasion in different human cancers.9,21,48 Based on the fact that membranous E-cadherin was inversely correlated with nuclear HIF-1α, we postulate that loss of membranous E-cadherin and nuclear HIF-1α expression is part of the malignant transformation of equine vulvo-vaginal neoplasia. In addition, the positive correlation of nuclear HIF-1α with N-cadherin and vimentin expression as well as with nuclear β-catenin leads us to hypothesize that nuclear expression of HIF-1α is associated with the mesenchymal-like state of epithelial tumor cells undergoing EMT in equine vulvo-vaginal malignant neoplasia. Together these findings corroborate the hypothesis that the EMT process is happening in malignant equine vulvo-vaginal tumors, which is further supported by the fact that similar findings have been described in other equine neoplasia undergoing the EMT process.2,4,31
In humans, the occurrence of EMT and its role in tumor progression have been studied in a number of cancers, including several gynecological cancers.8,37,44,56 Human vulvar SCCs are characterized by an aggressive nature and by an infiltrative invasion pattern, which is favored by the EMT process in the neoplastic cells. 19 Human vulvar SCCs undergoing EMT are characterized by a loss of E-cadherin expression, and vimentin and nuclear β-catenin expression. 37 Interestingly, in human vulvar SCCs, the occurrence of EMT is associated with a negative human papillomavirus status 37 and is likely to be associated with p53 mutations.13,26,37 The majority of the malignant equine vulvo-vaginal tumors investigated in this study were found to express E6/E7 oncogenes. This leads us to speculate that, despite a similar expression pattern of EMT markers, the equine counterpart might have a different pathogenesis than the human ones. However, the status of p53 mutations in these tumors should be evaluated in future studies.
Equine vulvo-vaginal METs were characterized by a higher infiltration of inflammatory cells when compared with BPLs. In human medicine, a proinflammatory microenvironment in vulvar high-grade squamous intraepithelial lesions is predictive of vaccine-induced immune infiltration and associated with a better clinical response. On the contrary, cold lesions did not develop such a proinflammatory environment postvaccination. 1 Benign, suspected preneoplastic lesions in mares are usually papillomas, with intraepithelial carcinoma being less common.35,40 For this reason, a comparison with the human counterpart should not be taken as straightforward. Nevertheless, it could be postulated that the mild inflammatory infiltrate observed in equine BPLs could be a predisposing factor for the development of carcinomas and potential histologic criteria to be evaluated to predict responses to a possible future vaccination.
In the MET group, the classification based on tumor T-cell infiltration showed results similar to what was reported in humans. 24 Unlike humans, where HPV-negative lesions represent the majority of cases 23 , all cases of our study group were EcPV2-positive. Unfortunately, follow-up data were not available in our case, but we could hypothesize that different T-cell infiltration patterns could reflect different prognoses or susceptibility to immunotherapy. 25
The presence of a statistically significant increased number of different proinflammatory cellular populations composed of intratumoral infiltration of lymphocytes, neutrophils, and mature plasma cells was observed. Interestingly, the intratumoral infiltration of B cells (CD20+) cells was lower when compared with the extratumoral tissue. These results are similar to what has been reported in equine penile SCCs 34 and may represent the inflammatory response in cases of equine genital EcPV2-induced malignant tumors.
In our study, the number of MPO+ cells was significantly associated with IL17A expression. IL17A expression by MPO+ tumor-associated neutrophils has been postulated to promote EMT processes through JAK2/STAT3 signaling in human gastric cancer. 29 This finding might suggest a possible role of MPO+ tumor-associated neutrophils in promoting the EMT process in equine vulvo-vaginal METs.
Data obtained by the evaluation of CD204 expression showed some of the most interesting results of this study. First, the expression of this marker was higher in METs when compared with BPLs. CD204 is recognized as a possible marker for protumoral M2-polarized macrophages in different cancers, in both humans and animals.6,22,36,42,49 This leads us to postulate that MET infiltration by CD204+ M2-polarized macrophages could be associated with immunoescape processes leading to an immunosuppressed tumor. However, the lack of significantly higher IL10 and TGFB gene expression in equine vulvo-vaginal METs of our study does not seem to support this hypothesis. On the contrary, the protumoral role of CD204+ M2-polarized macrophage population seems to be supported by the association with a higher MC and the presence of ulceration, both variables commonly associated with malignancy and poor prognosis in cancer. Interestingly, intratumoral CD204 expression was also associated with the expression of different key molecules for the EMT process. As a matter of fact, intratumoral CD204 expression was inversely correlated with membranous E-cadherin and β-catenin expression as well as cytokeratin and was instead positively associated with N-cadherin membranous expression and HIF-1α nuclear expression. Taken together, these results seem to indicate an association of intratumoral CD204+ cells with the progression of the EMT process in equine vulvo-vaginal METs. This hypothesis is supported by results in other studies that found a correlation between tumor-associated macrophages and EMT.7,12,27,30 In order to further clarify this aspect, future studies should gather insights on CD204+ M2-polarized macrophages in horses and explore other possible immunosuppressive pathways mediated by this cellular population.
The authors recognize that this study presents some limitations. First, the limited number of cases does not allow us to draw any firm conclusion due to the small sample size. Unfortunately, this is due to the rare occurrence/report of the disease. Second, there was a lack of follow-up and staging data. This did not allow us to associate any of our pathological findings with an actual clinical outcome. In addition, the lack of information about the presence of metastasis, and the lack of eventual samples from metastatic sites, limited the investigations on the EMT process. Third, some data on the transcriptome level did not fully support our findings on IHC quantifications, i.e., gene expression of the wnt/β catenin pathway-related genes or IL10 and TGFB. This might be due to the fact that the RNA was extracted from FFPE materials and this represents a limitation because of an inferior quality of RNA due to the formalin fixation of the samples. However, a future multicentric study should include more institutions and possibly include the presence of clinical data in the inclusion criteria as well as the collection of fresh material for transcriptome analysis.
In conclusion, this is a study investigating the EcPV2 infection state as well as EMT and the tumor microenvironment in vulvo-vaginal BPL or MET lesions in mares. Despite a similar EMT-related protein expression pattern, human and equine vulvo-vaginal cancers share some differences like the negative papillomavirus infection status in the human counterpart that undergoes in EMT. However, our study showed that changes like loss of membranous E-cadherin and β-catenin expression, and increased N-cadherin and vimentin expression as well as nuclear β-catenin and HIF-1α expression are hallmarks of malignant transformation in equine vulvo-vaginal epithelial tumors. In addition, our results seem to support the hypothesis that some actors in TIME, such as M2-polarized macrophages expressing CD204, may favor EMT, providing new insights for future investigations in the field of equine EcPV2-induced genital neoplastic lesions.
Supplemental Material
sj-xlsx-1-vet-10.1177_03009858231207025 – Supplemental material for Vulvo-vaginal epithelial tumors in mares: A preliminary investigation on epithelial-mesenchymal transition and tumor-immune microenvironment
Supplemental material, sj-xlsx-1-vet-10.1177_03009858231207025 for Vulvo-vaginal epithelial tumors in mares: A preliminary investigation on epithelial-mesenchymal transition and tumor-immune microenvironment by Federico Armando, Ilaria Porcellato, Livia de Paolis, Samanta Mecocci, Benedetta Passeri, Małgorzata Ciurkiewicz, Luca Mechelli, Chiara Grazia De Ciucis, Marzia Pezzolato, Floriana Fruscione, Chiara Brachelente, Vittoria Montemurro, Katia Cappelli, Christina Puff, Wolfgang Baumgärtner, Alessandro Ghelardi and Elisabetta Razzuoli in Veterinary Pathology
Supplemental Material
sj-pdf-2-vet-10.1177_03009858231207025 – Supplemental material for Vulvo-vaginal epithelial tumors in mares: A preliminary investigation on epithelial-mesenchymal transition and tumor-immune microenvironment
Supplemental material, sj-pdf-2-vet-10.1177_03009858231207025 for Vulvo-vaginal epithelial tumors in mares: A preliminary investigation on epithelial-mesenchymal transition and tumor-immune microenvironment by Federico Armando, Ilaria Porcellato, Livia de Paolis, Samanta Mecocci, Benedetta Passeri, Małgorzata Ciurkiewicz, Luca Mechelli, Chiara Grazia De Ciucis, Marzia Pezzolato, Floriana Fruscione, Chiara Brachelente, Vittoria Montemurro, Katia Cappelli, Christina Puff, Wolfgang Baumgärtner, Alessandro Ghelardi and Elisabetta Razzuoli in Veterinary Pathology
Footnotes
Acknowledgements
We thank Caroline Schütz, Julia Baskas, Kerstin Rohn, Kerstin Schöne, and Jana Svea Harre for excellent technical support.
Author Contributions
The study was designed by ER, WB, and AG. Pathology evaluation was performed by FA, LdP, IP, BP, LM, MC, CP, VM, and MP. Immunolabeling was conducted and analyzed by FA, LdP, IP, BP, MC, VM, and MP. PCR was performed and analyzed by KC, SM, FF, ER, and CGdC. Data analysis and interpretation were performed by FA, LdP, IP, BP, MC, CB, KC, SM, LM, FF, CGdC, and ER. Figures were prepared by FA, LdP, and IP. The original draft was written by FA, LdP, IP, and ER. The manuscript was reviewed, edited, and approved by all authors. Funding was acquired by ER. The project was supervised by LM, ER, AG, and WB.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Italian Ministry of Health, grant number IZS PLV 15/18 RC and Liguria Region, grant number 22L04.
ORCID iDs
Supplemental Material for this article is available online.
Data Availability
All the paper-related material can be accessible per request to the corresponding author.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
