Abstract
This work aimed to characterize the clinic-pathological presentation of an outbreak of auricular and laryngeal chondritis in pigs. Visits were made to pig farms, where the clinical history was obtained, and clinical and postmortem examinations were performed. In those farms, 3% to 4% of pigs presented otohematomas, which started in the nursery and extended to the finishing phase. Moreover, some finishing pigs presented with respiratory distress, initially characterized as inspiratory dyspnea, associated by an uncommon respiratory stridor and culminating in death. Grossly, nursery piglets had enlarged ears, and on the cut surface, the cartilage was fragmented and associated with blood clots. In the finishing phase, in addition to auricular lesions, the epiglottis and arytenoid cartilages were thickened and distorted, which partially occluded the lumen. Microscopically, the laryngeal and auricular cartilages were fragmented, displayed a loss of matrix basophilia, and were surrounded by lymphohistiocytic inflammatory infiltrate, with occasional multinucleated giant cells and fibrosis. The lesions exclusively affected elastic cartilages. The disease in finishing pigs led to increased mortality and was a differential diagnosis to respiratory challenges. It was not possible to determine the factor that triggered this condition; however, a nutritional association is suspected. To the authors’ knowledge, this is the first report of primary auricular and laryngeal chondritis in pigs.
Keywords
Laryngeal chondritis is reported in cattle, horses, and sheep and can be caused by trauma, followed by secondary bacterial or viral infection, which leads to inflammation that affects the cartilage.4,5,24 Another presentation of chondritis reported in rats, 16 cats, 3 cattle, 1 and dogs 18 is called auricular chondritis, characterized by cartilage fragmentation of the ear and replacement by fibrous tissue. 17
In humans, a systemic inflammatory disease called relapsing polychondritis (RP) is recognized. This condition simultaneously affects the auricular and other cartilages, such as larynx, trachea, and joints. 6 The aforementioned presentations of polychondritis in humans and animals share some clinical and morphological characteristics. However, in pigs, there are no descriptions of such diseases. This work aimed to characterize the clinic-pathological presentation of an outbreak of auricular and laryngeal chondritis in pigs.
Materials and Methods
Technical visits accompanied by a field veterinarian and farm employees were performed at 4 pig farms (1 nursery and 3 growing-finishing farms), with automatized ad libitum feeding, good air condition in the facilities, and pens with flooring consisting of solid and slat concrete. The pigs were housed with a density of 1 animal per 0.27 m2 in the nursery and per 0.9 m2 in the growing/finishing phase. The pigs were vaccinated against influenza A virus, porcine circovirus type 2, and Mycoplasma hyopneumoniae during weaning and 30 days later.
These farms were from a pig production company that comprised 300 pig farms and located in the western region of Santa Catarina State, Brazil (26°51'21'S 52°59'15″W), which reported that approximately 5% of its farms (5 nurseries and 10 finishing) had about 3% to 4% of pigs clinically affected. Data were obtained regarding the occurrence of an atypical disease, including signalment of the affected pigs, clinical signs, and epidemiological data. The affected pigs were clinically evaluated, and some with poor prognosis were selected for postmortem examination. The problem started in August 2020, and morbidity was increased until June 2021.
Two nursery pigs and 5 finishing pigs were submitted for complete postmortem investigation, with particular attention to the cartilages (ear, trachea, bronchi, larynx [epiglottis, arytenoid, thyroid, and cricoid cartilages], joints [costochondral and metacarpophalangeal], and snout) and other sites, such as aorta and eye. All these sites, in addition to the main organs (central nervous system, thoracic, and abdominal viscera), were collected and fixed in 10% buffered formalin.
Afterwards, the laryngeal cartilages were mapped, and fragments of all organs and cartilages collected were routinely processed for histology and stained with hematoxylin and eosin. Histologic sections that contained ear and laryngeal cartilages from affected and control pigs were stained with toluidine blue, Alcian blue (pH 2.5), Masson’s trichrome, and Verhoeff’s elastic stains. The control tissues used in the histochemical techniques were collected from pigs without any relation to those affected by this condition.
Results
The affected pens were composed of pigs from different origins and genetics. All pigs received feed made from the same factory. Approximately, 1 month before the start of the outbreak, there was a change in the brand of the premix given to the pigs (Premix 1). Near the end of the outbreak, a new brand of premix (Premix 2) was introduced in an attempt to elucidate the source of the problem, and the cases started to decline. The premixes were commercial products composed of a homogeneous mixture of minerals, vitamins, and additives that were added to the feed provided to the pigs.
Differences between the compositions of the 2 premixes were observed when comparing the labels. The following elements were present in Premix 1 but not in Premix 2: acidifying additives (butyric, fumaric, and malic acids), flavor additives (milk scent, vanilla scent, thaumatin, and neohesperidine), enzymes (cellulase, glucanase, protease, and xylanaze), saccharides (dextrose and mannanoligosaccharide), antioxidant (ethoxyquin), amino acids (
During the visit to the nursery, the animals were 60-day old, and approximately, 3% of the piglets (60/2100) had clinical signs. The first changes were noted at 50 days of age. In this phase, there was no increase in mortality. The clinical signs were characterized by unilateral or bilateral otohematomas, which initially had a reddish discoloration and mildly increased in volume; afterward, they assumed a slightly domed and smooth surface (Fig. 1a). In other cases, the pigs had ears reduced in size and with a wrinkled surface (Fig. 1b).
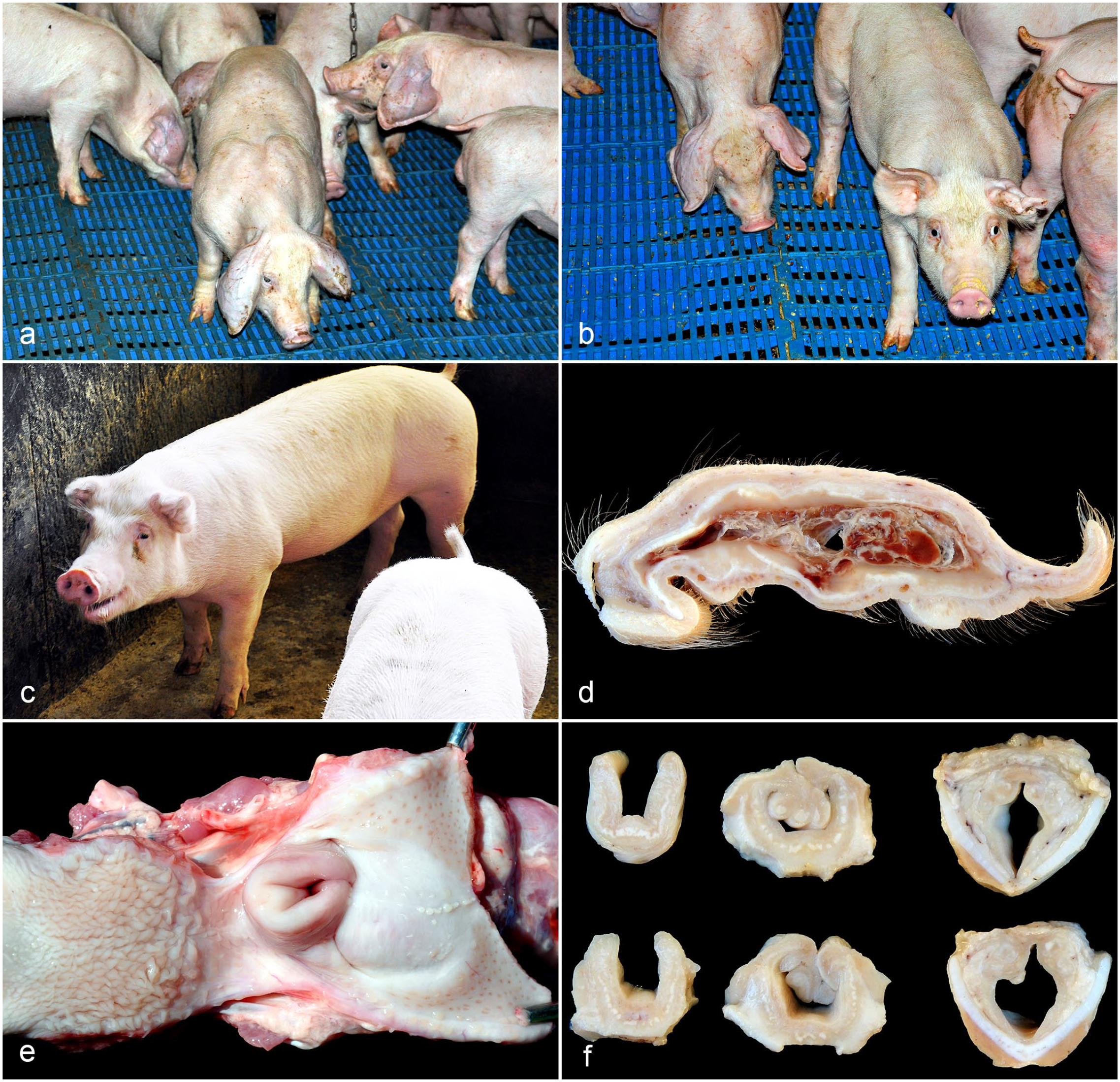
Gross lesions of auricular and laryngeal chondritis in nursery and finishing pigs. (a) Nursery pigs with unilateral or bilateral increases in volume in the ears (otohematoma). (b) Nursery pigs with acute unilateral otohematoma (left) compared with a chronic unilateral otohematoma (right). (c) Finishing pig in orthopneic position and bilaterally shrunken and wrinkled ears (Case 6). (d) Ear on the cut surface. Fragmented cartilage interspersed with whitish material. In the center, there is inflammatory exudate and blood clots (otohematoma) (Case 3). (e) Larynx, thickening of the epiglottis and arytenoid with partial occlusion of the laryngeal lumen (Case 3). (f) Larynx, sequential cut surface of 2 pigs. Distortion and thickening of the epiglottis and arytenoid cartilages is evident, as well as stenosis of the laryngeal lumen. Thyroid cartilage is without alterations (Cases 3 and 4).
At the 3 finishing farms, the pigs were 125- to 140-day old, and approximately, 4% were affected (60/1500 in total). In this phase, the mortality rate was increased (about 2%–3%, compared with the previous production herd with 0.5%) and was mainly attributed to animals with the reported condition. Affected pigs had lower weight gain and displayed ear lesions characterized by a reduction in size and a wrinkled surface. Approximately, 60% of these pigs with ear changes had inspiratory dyspnea, accompanied by an atypical respiratory stridor (Supplemental Video S1). According to reports, those pigs arrived at the farms with ear lesions and later developed respiratory signs. When the affected pigs were moved, they adopted an orthopneic position (Fig. 1c); dyspnea became severe and sometimes progressed to death. In all farms, there were scarce pigs with tail bite lesions, but those affected by cannibalism generally did not have otohematomas and dyspnea. They were therefore not correlated with the condition reported here.
The signalment, clinical signs, and affected cartilage of the pigs that underwent necropsy are described in the Supplemental Table S1. Grossly, nursery pigs had enlarged, hyperemic ears with a domed and smooth surface. On cut section, there was cartilage fragmentation interspersed with whitish material, and inflammatory exudate and blood clots (otohematoma) could be observed in the center of the affected area.
All the finishing pigs presented auricular lesions. In 2/5, the ear lesions were like those described in nursery pigs (Fig. 1d). In 3/5, the ears were reduced in size, firm, wrinkled, and, on cut section, the auricular cartilage was fragmented and interspersed with whitish material (histologically consistent with fibrosis). This presentation was considered a chronic form of ear lesion. In addition, in all cases, there was a marked decrease in the laryngeal lumen due to severe thickening and distortion of the epiglottis and arytenoid cartilages (Fig. 1e). Upon cutting, these cartilages were fragmented and interspersed with fibrosis (Fig. 1f).
Histologically, the auricular cartilages of all pigs (7/7) were fragmented, with the peripheral loss of matrix basophilia, resulting in pink to pale pink matrix. They exhibited perichondrial reaction, which was characterized by multiple layers of a perichondrial fibrous zone composed of fusiform cells. In addition to the perichondrial fibrous zone, there was the formation of multiple nodules of a slightly basophilic, disorganized cartilaginous matrix, with hypertrophic and hyperplastic chondrocytes (nodules of newly formed cartilages). These cartilages were surrounded by an inflammatory infiltrate composed of lymphocytes, plasma cells, and macrophages (Fig. 2a). In some areas, there were also multinucleated foreign body-type giant cells, arranged in a palisade adjacent to the cartilage (Fig. 2b). In addition, there was proliferation of fibrous connective tissue, replacing areas of cartilage loss and being arranged around the remaining cartilaginous tissue.
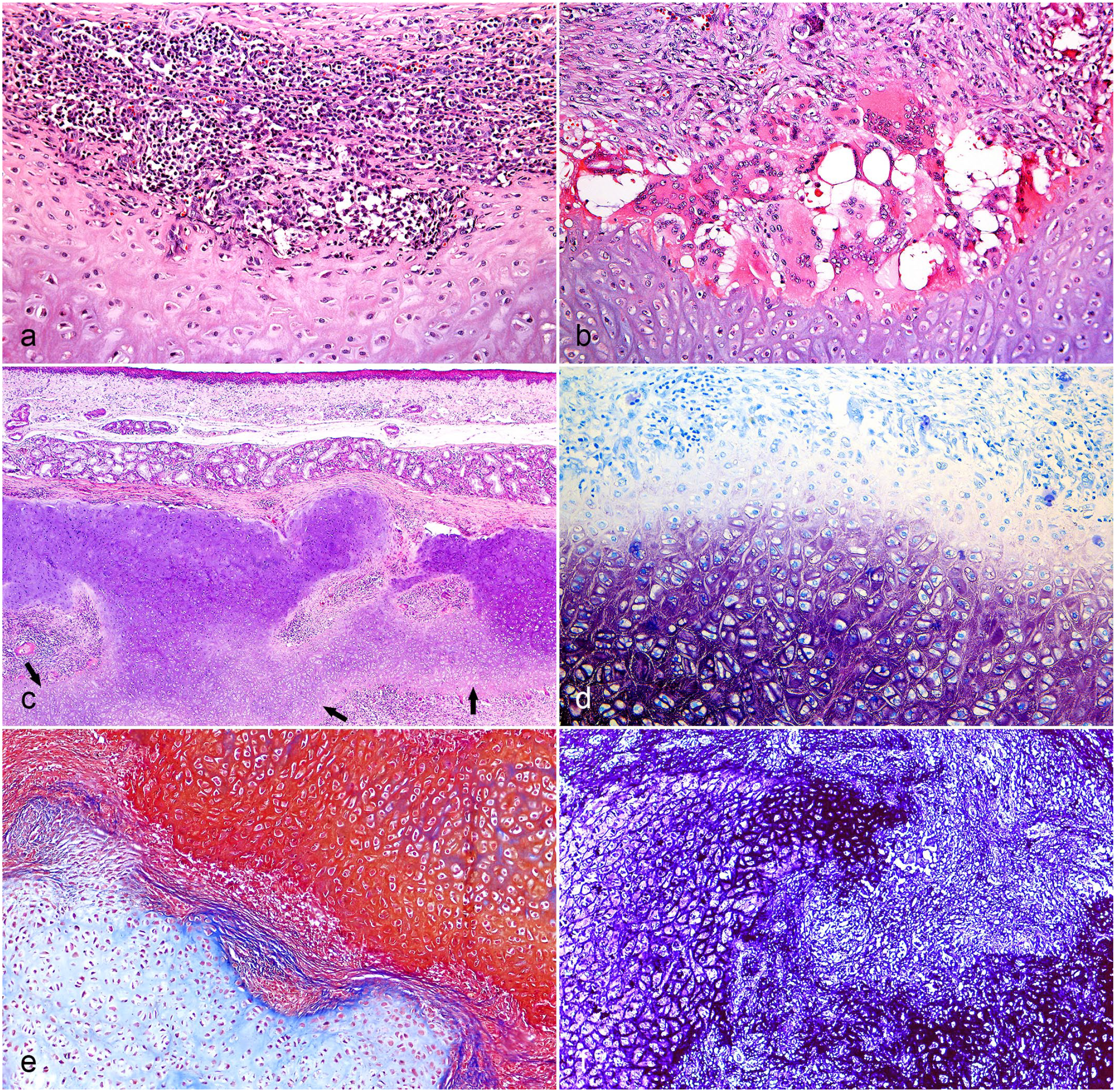
Histologic lesions of auricular and laryngeal chondritis in pigs. (a) Ear. Inflammatory infiltrate composed of lymphocytes and plasma cells forming aggregates adjacent to the affected cartilage that has discoloration (loss of basophilia) of the matrix. Hematoxylin and eosin (HE) (Case 6, finishing pig). (b) Ear. Inflammatory infiltrate composed of lymphocytes, plasma cells, multinucleated giant cells, and macrophages, in addition to the proliferation of well-differentiated fibrous connective tissue. There is also a loss of basophilia at the edge of the cartilaginous island. HE (Case 5, finishing pig). (c) Larynx (arytenoid). Disarrangement and fragmentation of the cartilage. In the center, there is a focally extensive area of newly formed cartilage (arrows) associated with inflammatory infiltrate and proliferation of fibrous connective tissue. HE (Case 3, finishing pig). (d) Larynx (arytenoid). Loss of stain in the cartilage matrix at the periphery of the chondroid islands. Toluidine blue (Case 3, finishing pig). (e) Larynx (arytenoid). There is red stain of healthy and mature cartilage. Adjacent, there is an island of blue tissue compatible with newly formed cartilage. Masson’s trichrome stain (Case 3, finishing pig). (f) Ear. The elastic fibers in the center of this auricular cartilage are evidenced by a strong black stain, unlike the periphery where this stain is not present. Verhoeff’s elastic stain (Case 3, finishing pig).
Cleft formation was frequently observed, which was characterized by the split of the auricular cartilage, allowing the infiltration of inflammatory cells, in addition to fibrin and blood deposition (otohematoma) (Supplemental Figures S1a and S1b [control]). The proliferation of fibrous connective tissue, the presence of cartilage islands, and the occurrence of multinucleated giant cells were more evident in finishing pigs, which suggested chronicity.
The laryngeal cartilages of nursery pigs had no microscopic lesions. However, in all finishing pigs evaluated (5/5), the epiglottic and arytenoid cartilages of the larynx showed lesions identical to those described for the ears (Supplemental Figures S1c and S1d [control]), characterized by a lymphoplasmacytic and histiocytic inflammatory infiltrate, which surrounded the fragmented cartilage with a perichondrial reaction and a newly formed cartilage, interspersed by a proliferation of fibrovascular tissue (Fig. 2c). This injury distorted and increased the volume of the affected laryngeal cartilages, which reduced the laryngeal lumen and possibly hindered the air flow. Multifocal erosions or ulcers of the luminal epithelium (3/5) were also observed.
Other body cartilages, such as trachea, bronchi, larynx (thyroid and cricoid cartilages), snout (nasal septum), joints (costochondral and metacarpophalangeal), and other sites, such as aorta and eye, were also evaluated; however, no lesions were evidenced microscopically.
In the toluidine blue (Supplemental Figures S1e and S1f [control]) and Alcian blue stained sections of the affected auricular and laryngeal cartilages showed loss of staining of the peripheric cartilaginous matrix (Fig. 2d). In the Masson’s trichrome stained sections, there was red staining of the healthy cartilage, and in the nodules of the perichondrium, there was a bluish cartilaginous matrix, morphologically compatible with the newly formed cartilage (Fig. 2e). Verhoeff’s elastic stain highlighted the elastin fibers in the matrices of the affected cartilages (ear, arytenoid, and epiglottis cartilage). However, in the edges of the affected cartilages, there was a decrease and/or loss elastic fibers (Fig. 2f). In the other cartilages of the larynx (cricoid and thyroid), there was no staining of elastin fibers, which confirms them as hyalin cartilages.
Concomitantly with the cartilaginous lesions, the lungs from all growing-finishing pigs (5/5) had a bronchointerstitial pneumonia with necrotizing bronchiolitis, associated with a bronchus-associated lymphoid tissue (BALT) hyperplasia, and fibrinosuppurative bronchopneumonia that affected approximately 10% to 30% of the parenchyma. Besides that, in one of these pigs (1/5), there was abscess formation in the middle of parenchyma. The other organs showed no alterations.
Discussion
The diagnosis of auricular and laryngeal chondritis in the pigs was performed through the association of clinical, macroscopic, and microscopic findings. There are few reports of lesions restricted to the auricular cartilage in other species, such as cats, 3 dogs, 18 cattle, 1 and rats. 16 The morphologic lesions observed by us in pigs are extremely similar to those described for other species and human RP, suggesting that all of these conditions are primary lesions of the cartilage.1–3,6,16,18 This allows us to speculate on a common origin or pathogenesis for these conditions.
The triggering event of RP in humans is not yet understood; however, genetic susceptibility, molecular mimicry with infectious agents, and mechanical or chemical exposure are thought to be involved.2,11 Besides that, autoantibodies against matrilin-1 and collagen types II, IX, and XI are detected in humans affected by RP, which corroborates with the physiopathology, and indicates its autoimmune involvement; however, their detection is not specific nor sensitive to RP. 11 It was not possible to search for autoantibodies described in humans in the affected pigs. In veterinary medicine, a genetic association is described in cases of auricular chondritis of cattle, 4 and in rats, there is an association with ear tags. 13 In our cases, the affected herds were composed of several genetic lines of terminal crossbreed pigs for slaughter that are also used by numerous other producers in the country, which have not experienced the same problem. The affected pigs had neither ear tags nor tattoos.
Trauma to the ears, caused by factors, such as manipulation when handling the animals, was also considered as a triggering factor. However, the field veterinarian had already reviewed these procedures and concluded that this would not be a possible cause since the teams carefully handled the pigs and were well trained.
The feed used on all farms was manufactured by the company itself, and it was a common factor. Although nutritional components have not been described in the literature as possible triggers for polychondritis,2,5 we cannot exclude the possibility of an alimentary association with these cases, as the incidence of new cases continued until they disappeared after the premix change. The chondropathies that are usually associated with nutritional imbalances are primarily chondrodysplasias in veterinary medicine.27,29 However, in human medicine, there are some reports of chondritis, associated with vitamin D deficiency. 19
The difference found among the components cited in the label of the premixes were predominantly represented by acidifying additives, flavor additives, enzymes, saccharides, antioxidants, and other commonly used components of pigs feed, which are already proved to be beneficial to the production and are not expected to cause such problems.7,30 Therefore, at this time, it is not possible to state that any of these components are directly related to the condition reported here.
We hypothesized that in Premix 1, there were some components that were not cited in the label, possibly accidentally added, that, when ingested, led to a change in the morphology of the elastic cartilages, which exposed antigens that were hidden from the immune system and incited the inflammatory response. However, as the etiopathogenesis of diseases, such as human RP is not clearly defined, it is difficult to demonstrate a pathogenic similarity, but the similarity of morphological lesions is a striking factor.2,15
The possibility of a hypersensitivity reaction was considered. In human skin, hypersensitivity can be triggered by metals, and pigs affected by insect bite may trigger an allergic dermatitis that is histologically characterized by perivascular eosinophilic inflammatory infiltrates.10,20,31 However, in our cases, the inflammatory pattern was characterized by a lymphohistiocytic infiltrate surrounding the elastic cartilaginous tissue, which differs from the pattern of a type 1 hypersensitivity.
Cases of primary auricular and laryngeal chondritis in animals and humans are not associated with otohematoma.1,3,6,16,18 In the pigs of this study, otohematomas were frequently observed in the nursery and progressed to a chronic form in the nursery but, most frequently, in the finishing phase. These otohematomas may have developed by self-trauma, due to irritation and pain originated from the inflammatory process.
The differentiation of the reported cases from the otohematomas usually seen in pigs was made by the association of the epidemiological data since the pigs were raised without traumatic handling and housed without stressful agents (e.g. high density and bad air quality), and the histological features contrast with those commonly seen in classical otohematomas, in which an intact cartilage is usually observed, separated from the cutaneous tissue by a collection of blood and fibrinous exudate, and the inflammation present is mild and not directed to the cartilage itself. 9
Humans affected by RP commonly present lesions in the ears, and in half of the cases, the respiratory cartilages are affected, which leads to a worse prognosis.6,28 In the present study, the pigs from the nursery only presented lesions in the auricular cartilages, which were considered as the initial site as finishing pigs had similar but chronic auricular lesions associated with the involvement of the larynx. The disease progression is not yet understood in human cases of RP, 6 and in the reported pig cases, there were no observed factors that could contribute to an understanding of this progression. However, it is assumed that auricular and laryngeal chondritis are due to the same condition as they have microscopic morphologic similarities and consistently affected the same pigs.
In veterinary medicine, the simultaneous involvement of auricular and respiratory cartilages is reported only in an outbreak in cattle. 4 In this work, respiratory distress was present, and the lesions detected in the upper respiratory tract justified the clinical signs. Thus, laryngeal chondritis proved to be a differential diagnosis for respiratory difficulty in finishing pigs.
Toluidine blue and Alcian blue stain proteoglycans and glycosaminoglycans, which are present in the extracellular matrix of the cartilage.25,26 Loss of staining associated with the chronic inflammation of cartilage indicates a loss of matrix components, as described in horses. 5 In Masson’s trichrome stained sections, the healthy and mature cartilage matrix was stained red. The matrix in adjacent islands stained in blue and was considered newly formed cartilage as the elastic cartilage has the ability to regenerate from the perichondrium; the bluish stain indicates newly formed collagen.8,22 The perichondrium is a structure that surrounds the cartilage and is composed of an external, vascularized layer of loose connective tissue, surrounding a fibrous zone and a subchondral zone that fuses with the cartilage matrix. This structure can regenerate and form cartilage tissue; however, it is uncertain which cell is responsible for this differentiation.12,22
In the sampled pigs, the lesions were restricted to the auricular cartilages and 2 of the laryngeal cartilages (epiglottis and arytenoid). In these cases, Verhoeff’s stain demonstrated the elastic nature of these cartilages. 14 The other types of cartilage did not show gross or microscopic lesions. Thus, it is suspected that the immune-mediated reaction is triggered by an antigen of elastic cartilages.
The lung lesions observed were probably caused by respiratory pathogens of the porcine respiratory disease complex, endemic in South Brazil, such as swine influenza A virus, Mycoplasma hyopneumoniae, and secondary bacteria, such as Pasteurella multocida.21,23 These lesions were not constant in all cases and would not justify the clinical signs. Therefore, we cannot associate viral or bacterial infections triggering this immunological reaction against the cartilage.
The pigs in this study were affected by auricular and laryngeal chondritis, which was restricted to the elastic cartilages. This condition led to a high incidence of otohematomas in the herd, in addition to respiratory distress, which resulted in the death of severely affected pigs. Under the circumstances of this study, the identification of the triggering factor was not possible. To the authors’ knowledge, this is the first report of primary auricular and laryngeal chondritis in pigs.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231186101 – Supplemental material for Auricular and laryngeal chondritis in nursery and finishing pigs
Supplemental material, sj-pdf-1-vet-10.1177_03009858231186101 for Auricular and laryngeal chondritis in nursery and finishing pigs by Anderson H. Gris, Manoela M. Piva, Claiton I. Schwertz, Ana P. Mori, Camila Saremba, Daniel M. Simon, Luciana Sonne, Saulo P. Pavarini and David Driemeier in Veterinary Pathology
Footnotes
Author Contributions
AHG, MMP, CIS, APM, CS, and DMS performed the clinical and macroscopic evaluations; AHG, MMP, CIS, LS, SPP, and DD performed histologic evaluations; the manuscript was written by AHG, MMP, and CIS with contribution from the other authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support was supplied by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES)-Finance Code 001, Fundação de Amparo à Pesquisa do Rio Grande do Sul (FAPERGS), and Pró-reitoria de Pesquisa da Universidade Federal do Rio Grande do Sul (Propesq/UFRGS).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
