Abstract
During the 12 months of 2006, zygomycotic lymphadenitis was diagnosed in 194 of 198 feedlot steers (0.04% of cattle slaughtered during that period) in a California slaughterhouse as part of bovine tuberculosis surveillance. Mesenteric lymph nodes were involved in 190 cases. Affected lymph nodes were enlarged (2 to 42 cm in greatest dimension), firm, and mottled gray-white to yellow with multiple granular or caseocalcareous foci. Histologically, nodal architecture was effaced by necrosis, granulomatous inflammation, and fibrosis. In approximately 20% of the cases, granulomas were mainly restricted to subcapsular sinuses and afferent lymphatic vessels, causing granulomatous lymphangitis. Nonseptate, irregularly branching hyphae with nonparallel walls and bulbous enlargements were common in necrotic areas and within the cytoplasm of multinucleated giant cells. Fungal cultures were performed on 124 affected lymph nodes using 7 media, but no zygomycetes were cultured. Fungal DNA was amplified from 20 lymph nodes. Amplicons from 16 nodes had nearly 100% homology with sequences for Rhizomucor pusillus; 4 amplicons had (> 98%) homology with Absidia corymbifera sequences. Zygomycosis should be considered in the differential diagnosis for granulomatous lymphadenitis in feedlot steers.
The term zygomycosis denotes infection by one of several genera of fungi of the class Zygomycetes. 2,18,29 The frequency of reports of zygomycosis in humans and animals has increased recently. 22 Several other terms have been applied to infections with these fungi. For example, the term mucormycosis has been used to designate infections caused by various species within the order Mucorales, 33 in the class Zygomycetes. The term phycomycosis had been widely used since 1959 to describe infections caused by fungi previously grouped in the class Phycomycetes. 26 However, in 1976 it was proposed that the archaic term be replaced with zygomycosis. 2,29 The class Zygomycetes is subdivided into the orders of Mucorales and Entomophthorales. Most zygomycotic infections in animals are caused by one of several genera of the family Mucoraceae, within the order Mucorales. 6,32
Zygomycetes are ubiquitous and regularly isolated from soil, foodstuff, and air in many environments where cattle are raised. 4,15 The normal flora of the bovine rumen contains a number of fungal species, among which Aspergillus fumigatus and the zygomycete Absidia corymbifera dominate. 27 Other zygomycetes, including those of the genera Rhizopus and Rhizomucor, are present in the ruminal content of clinically normal cattle. 27 Infection is probably established when the normal balance between the animal and the agents is disturbed. 18 In humans, zygomycosis generally occurs in 2 forms, pulmonary–systemic and rhinocerebral, with the primary target organs being lung and brain, respectively. The latter form is often seen in patients with diabetic ketoacidosis. 21
The main portal of entry for fungi causing systemic bovine zygomycosis is gastrointestinal (especially omasal) mucosa. 5,18,19 The pathogenesis of systemic bovine zygomycosis is not fully understood, but ruminal acidosis and oral antibiotics are thought to be predisposing factors. 5,10 In previous studies, cattle with zygomycotic lymphadenitis did not have clinical disease during antemortem examination, and infections were diagnosed only during carcass inspection at slaughter. 20,35
Culturing zygomycetes from tissue lesions is difficult, 16,19,21 and even when zygomycetes are recovered, identification to species level is time-consuming and may require the expertise of a mycology reference laboratory. Therefore, diagnosis is usually based on fungal morphology in histologic sections or cytologic preparations of impression smears. The presence of broad, nonseptate hyphae with irregular branching is considered characteristic of zygomycosis. However, it is not possible to identify the different orders, families, genera, and species in the class Zygomycetes by morphology alone. 34,41 Fungal DNA extraction, amplification, and sequencing have been described as a valid method for reliable identification of zygomycetes to the species level. 34 In this article, we describe the pathologic and microbiologic findings in lymph nodes infected with zygomycetes, sampled as part of the United States Department of Agriculture (USDA) Tuberculosis Surveillance program.
Material and Methods
Case Selection, Epidemiologic Data, Gross and Microscopic Pathology
From January to December of 2006, 198 lymph nodes with gross lesions from 198 Holstein steers (20 to 24 months old) were submitted to the San Bernardino branch of the California Animal Health and Food Safety Laboratory System (CAHFS), School of Veterinary Medicine, University of California–Davis, from a Southern California slaughterhouse by the USDA as part of bovine tuberculosis surveillance. All lymph nodes received at our laboratory during that period came from the same slaughterhouse, which processed approximately 450,000 steers in 2006. In sum, 198 steers (approximately 0.04% of the animals slaughtered during the study period) had gross lesions in 1 or more mesenteric (n = 191), mediastinal (n = 4), submandibular (n = 2), or cervical lymph nodes (n = 1) (Table 1). The animals originated from 21 different feedlots in Southern California. USDA inspectors routinely examined submandibular, retropharyngeal, bronchial, mediastinal, superficial cervical, prefemoral, supramammary, popliteal, mesenteric, and hepatic lymph nodes for gross lesions. Entire lymph nodes were shipped unfixed on ice packs overnight to our laboratory. The lymph nodes were grossly evaluated, and samples were collected for histology, fungal culture, and molecular-based identification. The samples for histology were fixed by immersion in 10% neutral buffered formalin, embedded in paraffin, sectioned at 4 µm, processed routinely, and stained with hematoxylin and eosin (HE), periodic acid–Schiff (PAS) reaction, and a modified Ziehl Neelsen acid fast–new fuchsin method (modified Ziehl Neelsen staining procedure, No. GPPISOPOO14.01, National Veterinary Services Laboratories [NVSL], Animal and Plant Health Inspection Service, USDA, Ames, IA). Selected sections with extensive areas of necrosis that obscured hyphae were also stained by Gomori’s methenamine-silver (GMS). Lymph nodes in which fungal hyphae were not detected were stained with Gram, as were all lymph nodes with spherical eosinophilic bodies (see below).
Number, Distribution, and Lesions in Lymph Nodes of 198 Cattle With Granulomatous Lymphadenitis
According to USDA and CAHFS protocols, samples are triaged by histology upon receipt at the laboratory. In those cases in which an indication of cause is detected histologically in HE-stained sections (eg, fungal hyphae, bacteria, Splendore–Hoeppli), no further diagnostic workup is performed. All tissues that have inflammation without indication of an etiological agent in HE-stained sections, whether acid-fast bacteria are observed or not, are submitted to NVSL for mycobacterial culture and/or polymerase chain reaction (PCR). In this study, however, no lymph node was cultured for bacteria (including Mycobacterium spp), but most of the lymph nodes were cultured for fungi without waiting for the histopathology results (see below).
The entire gastrointestinal tracts (rumen to rectum with mesenteries) from 10 steers with zygomycotic mesenteric lymphadenitis were opened, rinsed with tap water, and grossly examined. The oral cavity, pharynx, lungs, heart, kidneys, liver, and spleen of these steers were also grossly examined. Histology was performed on any gross lesions.
Fungal Culture
Of the 198 lymph nodes submitted, 124 were randomly selected for fungal culture 12 to 24 hours after slaughter. Samples were macerated using sterile scissors and streaked onto or inoculated into 1 or more of the following media: Sabouraud dextrose agar (SDA) with and without chloramphenicol (Hardy, Santa Maria, CA), inhibitory mold agar, potato flake agar, corn meal agar, Emmons modification of SDA, and Middlebrook H9 broth (County Public Health Laboratory, San Bernardino, CA). Samples of the Middlebrook H9 broth cultures were streaked onto SDA after 48 to 72 hours of incubation at 25°C. The Middlebrook H9 broth commonly used for M tuberculosis culture, was implemented because of its usefulness in the detection of systemic fungi (R. Morales, personal communication). All agar plates were examined for fungal growth weekly for 4 weeks before they were discarded as negative.
DNA Isolation
Total DNA was extracted from 29 randomly selected lymph nodes using a previously described method. 13 Briefly, approximately 2 mm3 of tissue was ground using sterile mortar and pestle and suspended in 1 ml of phosphate buffered saline (PBS). Samples were centrifuged at 20,800 × g for 10 minutes and pellets resuspended in 200 µl of PBS. Furthermore, 300 µl of lysis buffer (500 mM Tris-HCl [pH 9], 20 mM EDTA, 10 mM NaCl, 1% SDS) and 100 µl lysozyme (10 mg/ml) were added to samples followed by 2 hours of incubation at room temperature. Subsequently, 10 µl of proteinase K (20 mg/ml) was added, and samples were incubated in a 37°C water bath overnight. DNA was phenol–chloroform extracted as described previously and resuspended in 50 µl of sterile water. 40
Amplification of Fungal Specific DNA
To obtain fungal DNA amplicons for sequencing, an approximately 700–base pair fragment from the 5′ end of the large subunit of the rRNA gene was amplified using previously described fungal primers TW13 and Ctb6. 36 The amplification reaction (50 µl) contained 100 to 300 ng of total DNA as template, 1.5 mM MgCl2, 150 µM of each deoxynucleotide (Applied Biosystems, Foster City, CA), and 1 U of polymerase (AmpliTaq Gold, Applied Biosystems, Foster City, CA) in PCR buffer (1X PCR Buffer II/Gold Buffer, Applied Biosystems, Foster City, CA). Amplification reactions were conducted under the following parameters: 95°C for 10 minutes; 50 cycles of 94°C for 30 seconds; 50°C for 1 minute; 72°C for 2 minutes; and a final extension at 72°C for 7 minutes followed by a hold at 4°C (GeneAmp PCR System 9700, Applied Biosystems, Foster City, CA). A 5-µl aliquot of the PCR reaction was evaluated on a 3% agarose gel to determine if amplification products of the expected size were obtained. Amplicons were purified (Montage PCR Cleanup Kit, Millipore, Bedford, MA) and sequenced (ABI 3730 DNA sequencer, Davis Sequencing Inc., Davis, CA). Primers used for amplification were also used for sequencing reactions. Both the forward and the reverse fragments were sequenced. Samples that (1) had amplified product but insufficient DNA yield for direct sequencing or (2) were retested for sequence verification were cloned (TOPO TA Cloning Kit, Invitrogen Corporation, Carlsbad, CA). Resulting colonies were randomly selected, evaluated for inserts of the correct size by PCR amplification, and sequenced with primers directed at the T3 and T7 priming sites on the vector.
Analysis of DNA sequences
Consensus sequences were generated from individually sequenced strands from each amplicon and a basic local alignment search tool (BLAST [Basic Local Alignment Search Tool] search performed; VectorNTI Advance 10.3, Invitrogen Corporation, Carlsbad, CA). A multiple-sequence alignment was constructed using representative consensus sequences obtained from lymph node tissues that gave unique sequence results and reference sequences of representative zygomycetes obtained from GenBank. 39 Sequence alignments were manipulated using a multiple-sequence alignment editor (GeneDoc, http://www.nrbsc.org/gfx/genedoc/index.html), and a tree showing sequence relationships was constructed. 37 The Jukes–Cantor model was used for distance estimation; phylogenetic dendograms were built by the neighbor-joining method; and tree topologies were evaluated by performing bootstrap analysis with 1,000 replications.
Results
Pathology of Lymph Nodes
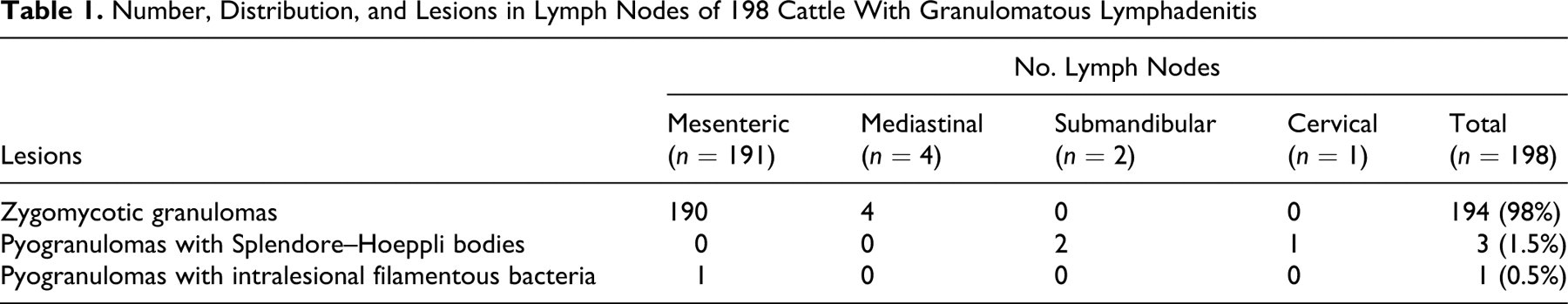
Table 1 presents the anatomic distribution of lymph nodes affected by zygomycotic granuloma. The size of the lymph nodes varied from 2 to 42 cm across their greatest dimension. Lymph nodes were round to oval and firm to hard; in most cases, they had multiple round nodules (0.2 to 1.0 cm in diameter) and were separated by fibrous tissue that elevated and infiltrated the capsule. On cut surfaces, nodal architecture was replaced by multiple to coalescing, sharply delineated, grayish-white to yellow granular areas of caseous necrosis with central calcification (Fig. 1). The entire lymph node was affected in most cases, but in approximately 20% of the cases, only the capsule and cortex were involved.
Histologically, all lymph nodes had granulomatous inflammation. Fungal hyphae were present in 194 of 198 (98%) of the examined lymph nodes. These included 190 mesenteric and 4 mediastinal lymph nodes (Table 1). Lesions were characterized by multifocal to coalescing amorphous necrosis with dystrophic calcification, granulomatous inflammation, chronic active fibrosis, and fungal hyphae. Inflammatory cells consisted mostly of lymphocytes, plasma cells, macrophages, and multinucleated giant cells (Fig. 2). The giant cells were foreign body–type as well as Langhans-type and were both numerous and conspicuous (Fig. 3). Few neutrophils were at the periphery of the granulomas; eosinophils were rare. Variable amount of fibrous tissue was present between the granulomas. When the granulomas occupied only a part of the node (approximately 20% of the cases), they were mainly restricted to subcapsular sinuses and afferent lymphatic vessels, causing granulomatous lymphangitis (Fig. 4).
All fungal hyphae were nonseptate and had nonparallel walls of variable thickness and prominent bulbous enlargements. Branching was nondichotomous, haphazard, or right-angled. Hyphae, 5 to 30 µm in diameter, were seen in areas of necrosis and mineralization, the cytoplasm of giant cells, and connective tissue between granulomas. In HE-stained sections, the hyphae were basophilic in viable tissue and faintly eosinophilic in necrotic areas. The hyphae were PAS-positive (Fig. 3) and argyrophilic with GMS (Fig. 5). Fungal hyphae were not found in vessel walls. In approximately one third of the lymph nodes with zygomycotic infection, extracellular, spheroidal, eosinophilic bodies (20 to 200 µm in diameter) were composed of densely arranged, variably sized radiating material (Fig. 6). The number of these bodies ranged between 1 and 27 per section. They were found in areas of caseous necrosis; a few had fungal hyphae in their centers. The spheroidal eosinophilic bodies were negative for PAS, GMS, and Gram stains.
Four lymph nodes did not contain fungal hyphae: 3 (1 cervical and the 2 submandibular lymph nodes) had pyogranulomas with Splendore–Hoeppli bodies (compatible with actinomycosis or actinobacillosis); 1 mesenteric lymph node had pyogranulomas with Gram-positive intralesional filamentous bacteria (Table 1). No acid-fast bacteria were observed in Ziehl Neelsen–stained sections.
Pathologic Findings in Other Organs
The rumen of 4 of the 10 animals from which the entire gastrointestinal tract was examined had focally extensive areas with black thick papillae. Histologically, this gross lesion consisted of mucosal suppuration and parakeratosis. The reticulum of another animal had transmural perforation by a piece of wire with severe locally extensive suppuration and fibrosis, diagnostic of chronic traumatic reticuloperitonitis. Fungal hyphae were not seen in sections of affected rumens or reticulum. No other gross lesions were observed in the 10 animals examined.
Fungal Cultures and Molecular-Based Studies
Cladosporium sp, Penicillium sp, and A versicolor were cultured from 3 of 124 lymph nodes. No zygomycetes were cultured with any of the 7 mycologic media used.
DNA was extracted from 29 lymph node samples. Products of approximately 600 to 700 base pairs were amplified from 20 of the samples (69%) using broad-range fungal primers. Sequencing of amplified products and subsequent BLAST searches of generated consensus sequences showed 2 main fungal sequence groups among the amplicons generated. Sixteen samples yielded sequences that had mean homology of 99.9% with Rhizomucor pusillus (AF113475). The remaining 4 samples yielded amplicons with 98 to 100% homology with A corymbifera (AF113446). Of these 4 sequences, 1 (S0700565-C) had 98% homology with A corymbifera, whereas the others were 100% homologous. The amplicon from the sample with 98% homology was cloned to ensure the accuracy of the sequence differences found, compared to the 3 other closely related sequences. Four clones were sequenced, and all clones had 100% homology with the directly sequenced amplicon and were 98% homologous to the A corymbifera reference sequence (AF113446). The 4 A corymbifera–related sequences identified from the lymph node granulomas also showed 95 to 98% homology with another culture collection strain of A corymbifera (AF113445). Results of the phylogenetic analysis showed that amplicons generated from lymph node samples clustered tightly with either R pusillus or A corymbifera and were substantially different from other closely related zygomycetes (Fig. 7).
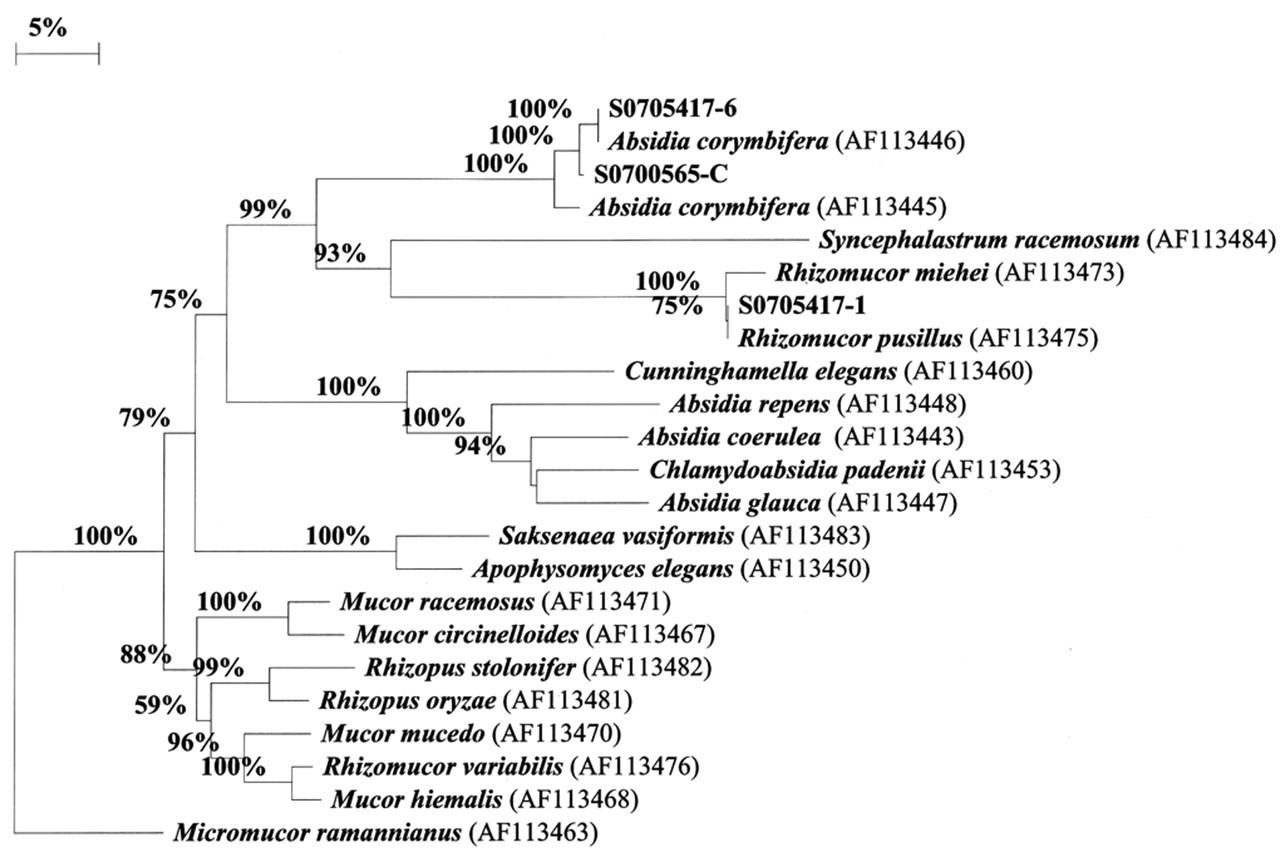
Phylogenetic relationship of fungal sequences obtained from tissue samples from Holstein steers with zygomycotic lymphadenitis to reference sequences of representative zygomycetes. Sequence result for sample No. S0705417-1 is representative of 16 sequences identified with high homology with Rhizomucor pusillus. Sequencing results for sample Nos. S0700565-C and S0705417-6 are representative of sequences most similar to Absidia corymbifera. Numbers at nodes represent bootstrap percentages from 1,000 replications.
Discussion
In this study, the diagnosis of zygomycotic lymphadenitis was based on histologic demonstration of intralesional zygomycetes and a compatible inflammatory reaction. In 20 of the 198 cases, the etiology was confirmed by direct amplification and sequencing of fungal DNA. Our failure to culture zygomycetes from any of the affected nodes may have been related to the 12- to 24-hours postmortem period between slaughter and inoculation of media, because zygomycetes reportedly die soon after the death of the host. 16,30 The 3 nonzygomycete fungi cultured (Cladosporium sp, Penicillium sp, and A versicolor) were interpreted as contaminants because no fungi with structural features other than those of the class Zygomycetes were observed in histologic sections.
The lymphadenitis in this study was uniformly chronic, as demonstrated by the extensive fibrosis and the predominantly mononuclear inflammatory infiltrate. In this respect, our cases differed from reports of bovine lymphadenitis caused by Aspergillus sp because in these reports, there were extensive areas of acute inflammation. 19,20 The lymph nodes in this study were firm, grayish-white or yellow, granular, and caseocalcareous, which is consistent with previous descriptions of zygomycotic lymphadenitis in cattle and other species. 14,16,20 In necrotic areas, fungal hyphae were often eosinophilic, which made them inconspicuous in HE preparations. To avoid overlooking zygomycetes, application of histochemistry for fungi, such as PAS or GMS, should be considered. 9 No acid-fast bacteria were seen in any of the lymph nodes stained with Ziehl Neelsen. Parasitic granulomas, which have been described in lymph nodes from slaughtered cattle, 8 were not found in this study.
Approximately one third of the lymph nodes in this study had spheroidal eosinophilic bodies with radiating internal structure. A few of these bodies had central fungal hyphae. The structure of these bodies is compatible with so-called asteroid bodies, a name widely used in the literature to describe radiating eosinophilic material often associated with fungal elements in cases of zygomycosis. 3,11,12,17,19,20,28,31 However, the expression has been used to describe morphologically distinct structures, and some authors have used this expression interchangeably with the term Splendore–Hoeppli. 16 Although electron microscopic and immunohistochemical studies revealed that asteroid bodies were composed of cytoplasm of surrounding neutrophils and deposits of immunoglobulins, there is still confusion in the literature regarding the origin and composition of these bodies. 17,28,31
In our study, 190 (98%) of the cases involved mesenteric lymph nodes, whereas only 4 (2.1%) involved mediastinal nodes. Similar anatomic distribution was reported in a Danish study: 84 cases (89.4 %) were mesenteric, 7 cases (7.4 %) were mediastinal, and 3 cases (3.2 %), were both mesenteric and mediastinal. 20
We were unable to determine the portal of entry of the fungi. However, in 98% of the cattle, the lesions were distributed along the mesenteric lymph node chain, suggesting that the portal of entry was the intestinal mucosa. In lymph nodes with small lesions, the granulomas were mainly restricted to the cortex and afferent lymphatic vessels in association with granulomatous lymphangitis. This suggests lymphatic spread from the intestine. In these cases, invasion may have occurred (1) by phagocytosis of hyphal fragments or spores that escaped recognition and killing by the mucosal defense system or (2) by direct fungal invasion of mucosal lymphatic vessels, assisted by roughage-induced minor mucosal trauma that healed without a trace. These findings contrast those of previous studies of gastrointestinal zygomycosis, in which necrosis of the mucosa, vasculitis, thrombosis, and hematogenous spread were the most characteristic findings. 5,19 We did not observe either vasculitis or infarction in any lymph nodes. This agrees with reported descriptions of zygomycotic lymphadenitis. 5,19,20 Given that no evidence of distant dissemination was observed in this study, apparently, the lymph nodes contained the infection. We cannot completely rule out that, at least in some cases, the portal of entry was outside the gastrointestinal system. For instance, small lesions in the foot or oropharyngeal and nasal mucosa might have been the route of entry and might have been missed at slaughter.
In our study, DNA sequencing was performed in 20 of the 194 cases histologically diagnosed as zygomycotic lymphadenitis, and A corymbifera or R pusillus was identified. It is possible that other zygomycetes may have been involved in some of the 174 other cases of lymphadenitis in this study.
Both A corymbifera and R pusillus have been found in air, soil, and organic matter worldwide; they have also been detected in a variety of food items, including grains, seeds, nuts, and beans. 32 A corymbifera has been identified as a cause of mesenteric lymphadenitis in cattle, 14,25 but we could not find any published reference to the presence of R pusillus in mesenteric lymph nodes of cattle or any other species. Previous reports, however, have described the recovery of R pusillus from bronchial and mediastinal lymph nodes 7,38 and brain 1,24 of cattle. This agent has also been associated with bovine mycotic abortion. 23 A recent review of zygomycotic infections described 2 modes of transmission for R pusillus—namely, inhalation of spores and percutaneous introduction of spores. 32 However, the presence of R pusillus in mesenteric lymph nodes in our study indicates an oral route of infection.
R pusillus and A corymbifera are considered thermophilic fungi, and their pathogenicity has been linked to their ability to grow in febrile patients. These thermophilic zygomycetes can grow at temperatures up to 48 to 52°C. Nonpathogenic Absidia spp are generally unable to grow at or above 37°C. 32 During 2006, maximum temperatures in the area of origin of the animals (where summer daytime air temperatures frequently approximated 44°C) were similar to the historical maximum for the same area. Rainfall in 2006, however, was well below the historical average for the area, which might have created particularly dusty conditions that may have facilitated ingestion of fungal spores by the cattle. However, because these are chronic lesions, it is difficult to correlate climatic conditions to lesions found at slaughter, because infections may have occurred several months before slaughter.
A form of zygomycosis in humans is frequently seen with ketoacidosis. 22 All the cattle in this study originated from feedlots, production systems in which ketoacidosis is a common problem. However, most cattle slaughtered in the USA originate in feedlots, and zygomycotic lymphadenitis does not seem to be a problem in other areas of the country. The data suggest that factors other than feedlot origin might be implicated in the cases of zygomycosis reported here.
In conclusion, zygomycete fungi were the predominant cause of macroscopic caseous necrosis in mesenteric and mediastinal lymph nodes of healthy-appearing cattle examined at slaughter in California. The gross lesions resembled those of bovine tuberculosis. The unusual number of cases of zygomycotic lymphadenitis in cattle in this study, the high environmental temperatures under which the cattle were housed, and the identification of R pusillus and A corymbifera (2 thermotolerant zygomycetes) as etiologic agents suggest that these infections are associated with environmental factors in the area where these animals originated. The observations of this study suggest that further study of feedlot management and nutrition and environmental conditions may facilitate understanding of the epidemiology and pathogenesis of this condition.
Footnotes
Acknowledgements
We thank Dr Roland Morales (San Bernardino County Laboratory of Public Health) for advice on fungal culture methods and supply of media, Dr C. Gomez for samples supply, Dr Donald Cordes for helpful comments on pathogenesis, Ms S. J. Uzal for reviewing this manuscript, and Ms E. J. Hurley, Ms D Paulson, Mr R. Cazarez, Ms S. Fitisemanu, and Ms S. Kwiek for excellent technical assistance.
This work was performed with financial support from California Animal Health and Food Safety Laboratory. J. Ortega was supported by “Becas para estancias en centros de investigación fuera de la Comunidad Valenciana dirigidas a profesores e investigadores de las universidades y centros de investigación” from Generalitat Valenciana and “Becas José Castillejo” from Ministerio de Educación y Ciencia, Spain.
The authors declared that they received no financial support for their research and/or authorship of this article.
