Abstract

Image 1
C. Lagenidium giganteum.
Lagenidium giganteum is a spore-producing, aquatic oomycete of the southeastern United States. Lagenidium commonly infects the extremities, mammary region, perineum, and trunk. Cutaneous and subcutaneous lesions are progressive, multifocal, firm, and ulcerative. Occult lesions in the thorax or abdomen may involve lymph nodes, great vessels, or lungs. Histologically, pyogranulomatous and eosinophilic inflammation are accompanied by extracellular, negatively staining hyphae with non-parallel walls, spherical to bulbous dilatations, and irregular right-angle branching. Lagenidium hyphae are larger and more visible in hematoxylin and eosin sections when compared with P. insidiosum. Hyphae stain with Grocott’s methenamine silver (Image 1). Ancillary testing is necessary for definitive identification.
Additional reading: Grooters A. Fungal and algal diseases. In: Sykes Jane E, ed. Canine and Feline Infectious Diseases. St. Louis, MO: Elsevier; 2013:672–674.
Contributor: Tessa Brown, University of Georgia
Photo credit: Elizabeth Howerth, University of Georgia
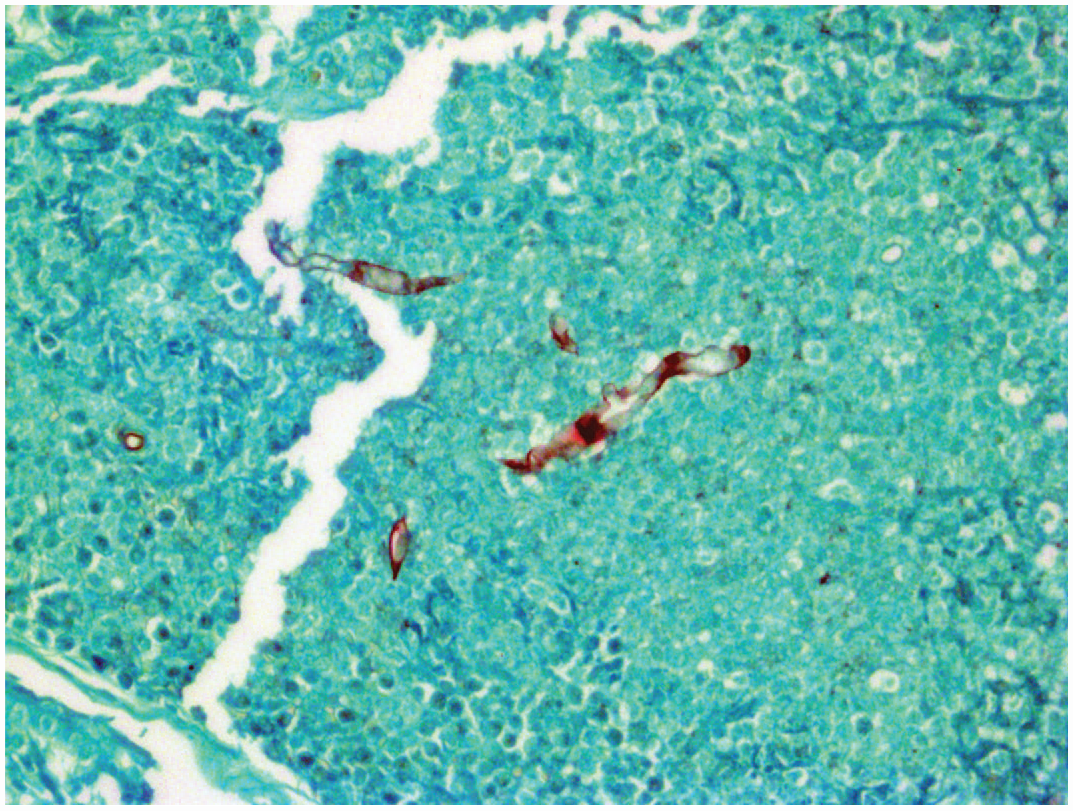
Image 2
A. Perkinsea.
Perkinsea is a direct life cycle protozoa which can cause mass mortalities in tadpoles. Gross lesions include abdominal distention, cutaneous erythema, petechiae, subcutaneous edema, ascites, hepatomegaly, splenomegaly, nephromegaly, pancreatomegaly, and segmental irregular thickening and white discoloration of the intestines. Histologically, alveolate spores are found in the liver, spleen, kidney, and pancreas, where they often outnumber inflammatory and parenchymal cells. Perkinsea spores stain with periodic acid-Schiff, Grocott’s methenamine silver (GMS), and Giemsa. GMS staining reveals abundant spores in areas of necrosis (Image 2). Alloglugea sp. causes digestive xenomas, and Ichthyophonus sp. is a larger organism (up to 250 μm) that causes chronic, systemic granulomatous inflammation. Hoferellus sp. is primarily an intratubular and intraluminal organism within the kidney and urinary bladder.
Additional reading: Isidoro-Ayza M, Grear DA, Chambouvet A. Pathology and case definition of severe Perkinsea infection of frogs. Vet Pathol. 2019;
Contributor: Hannah Coffin, University of Georgia
Photo credit: Elizabeth Howerth, University of Georgia
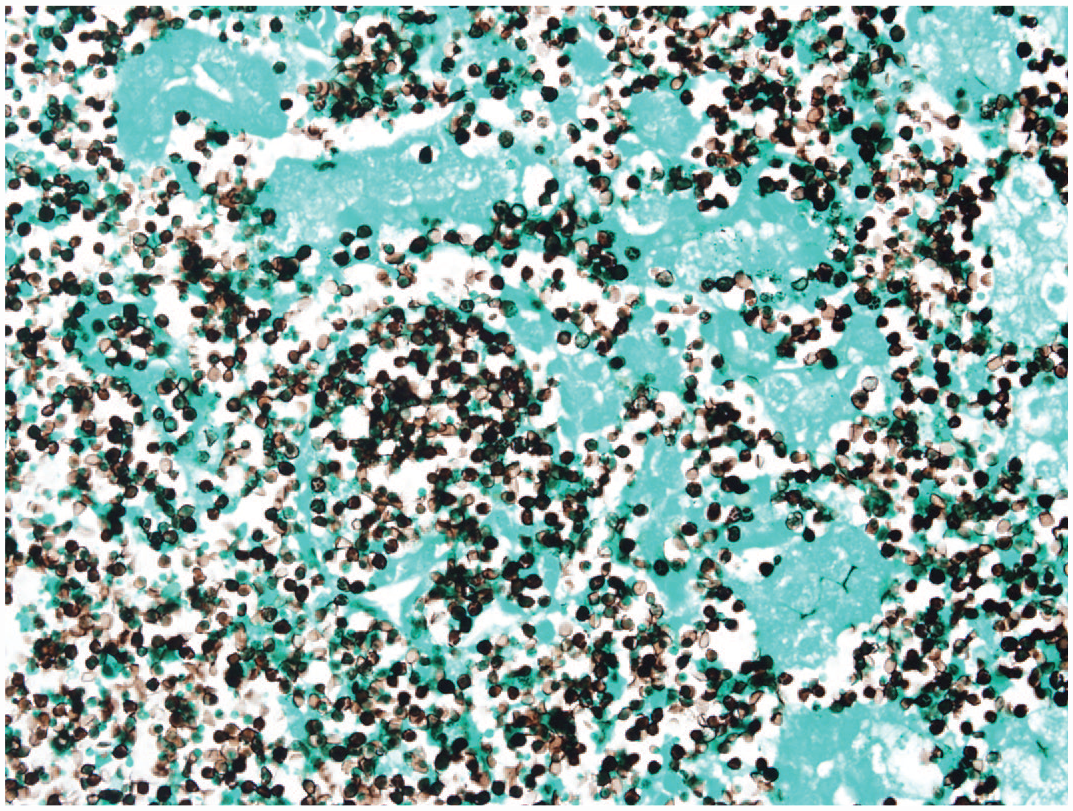
Image 3
B. Saprolegnia sp.
A juvenile yellowfin shiner (Notropis lutipinnis) was euthanized due to erosions and cottony white growths on the caudal fin/peduncle associated with Saprolegnia infection. This ubiquitous oomycete water mold of temperate/tropical fish causes superficial, opportunistic infections associated with immunosuppression, environmental stress (temperatures <59°F), physical trauma, and increased abundance of Saprolegnia spp. at colder temperatures. Microscopic identification of aseptate hyphae, occasionally containing spherical papillate oogonia with centric oospores (arrow, Image 3) that do not extend past superficial muscle layers is diagnostic on wet mounts (Image 3) and hematoxylin and eosin stained tissue sections. Focal infections spread rapidly and mortality occurs in a few days, as disease severity is proportional to surface area affected.
Additional reading: Noga EJ. Fish Disease: Diagnosis and Treatment. 2nd ed. Hoboken, NJ: John Wiley; 2010:156–162.
Contributor: Nicole Glenn, University of Georgia
Photo credit: Alvin Camus, University of Georgia
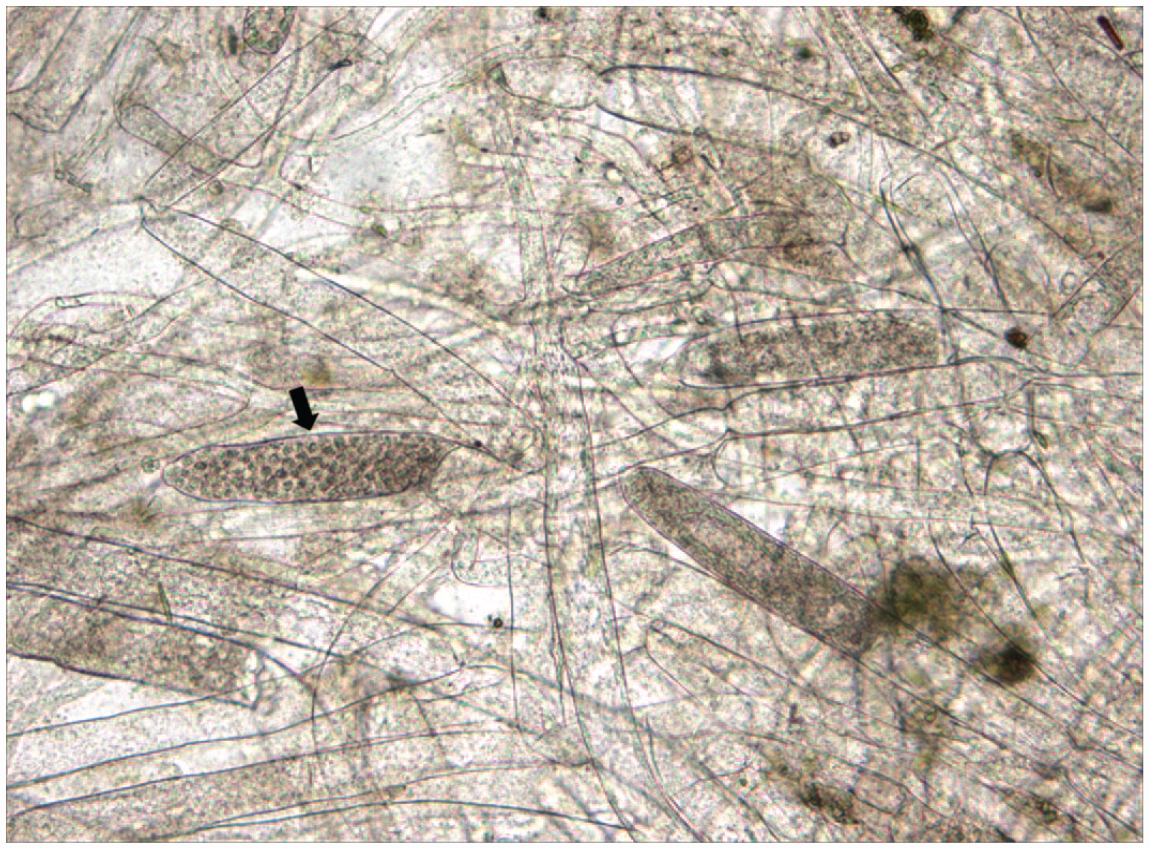
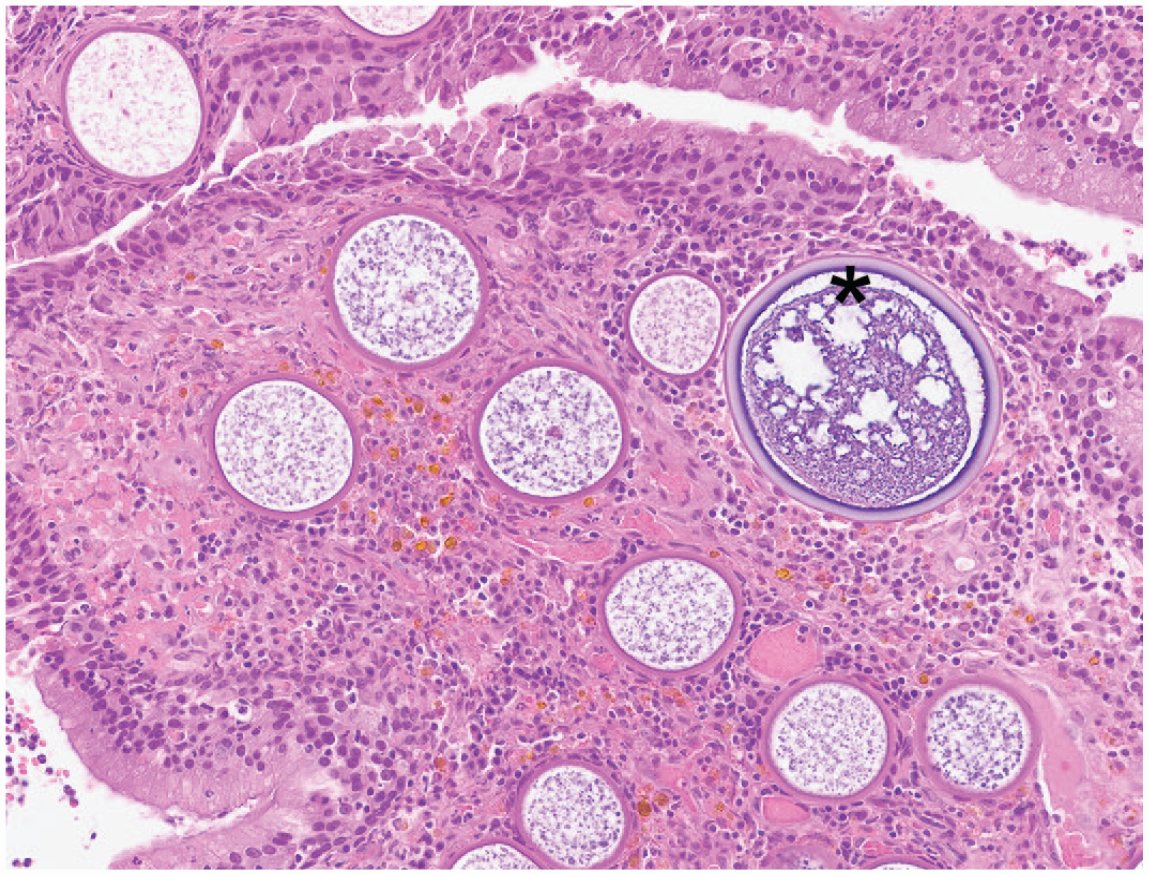
Image 4
C. Rhinosporidium seeberi.
Rhinosporidiosis presents commonly as a unilateral nasal polyp in dogs, with other domestic animals, including horses, less affected. The mature sporangia (asterisk, Image 4) are visible grossly as white pinpoint foci on the polyp surface. Comparatively, C. immitis has smaller endospores, C. neoformans has a thick polysaccharide capsule and is much smaller, and E. crescens often has a thicker inner wall and usually infects the lung. Rhinosporidiosis is most common in tropical climates, and infection occurs via direct exposure in contaminated water. Molecular studies suggest R. seeberi is an endosporulating organism within Mesomycetozoea (“DRIP” clade) and is not a true fungus.
Additional reading: Caswell JL, Williams KJ. Respiratory system. In: Maxie MG, ed. Jubb, Kennedy & Palmer’s Pathology of Domestic Animals. Vol. 2. 6th ed. St. Louis, MO: Elsevier; 2016:579–581.
Contributor: Kevin D. Niedringhaus, University of California, Davis
Veterinary Pathology invites submission of exceptional gross or microscopic images for consideration as an Image Challenge, along with a multiple-choice question and answer. For details, see the Instructions to Authors on the journal website.