Abstract
Massospora cicadina, an obligate fungal pathogen in the subphylum Entomophthoromycotina (Zoopagomycota), infects periodical cicadas (Magicicada spp.) during their adult emergence and modifies their sexual behavior to maximize fungal spore dissemination. In this study, 7 periodical cicadas from the Brood X emergence in 2021 infected by M. cicadina were histologically examined. In 7 of 7 cicadas, fungal masses replaced the posterior portion of the abdominal cavity, effacing portions of the body wall, reproductive organs, alimentary tract, and fat bodies. No appreciable inflammation was noted at the intersections of the fungal masses and host tissues. Fungal organisms were present in multiple morphologies including protoplasts, hyphal bodies, conidiophores, and mature conidia. Conidia were clustered into eosinophilic membrane-bound packets. These findings help uncover the pathogenesis of M. cicadina by suggesting there is evasion of the host immune response and by providing a more in-depth description of its relationship with Magicicada septendecim than previously documented.
Periodical cicadas are a superfamily of insects of the suborder Auchenorrhyncha, which makes up a large component of the order Hemiptera (the true bugs). They have either 13- or 17-year life cycles, the latter of which is the longest of any insect, spending the majority of life underground as nymphs, and then co-emerging in incredible numbers to metamorphose into adults. These adults primarily mate and oviposit in trees over approximately 45 days, during which time males make deafening sounds using their drum-like hollow abdomen with ribbed membranes called tymbals. The conclusion of these 45 days ends their life cycle.3,5,7,14 Periodical cicadas form mixed species cohorts called broods that cluster geographically and temporally. Brood X (also known as the “Great Eastern Brood”) is a brood of 17-year periodical cicadas that emerges throughout the eastern United States and includes three species: Magicicada septendecim, Magicicada cassini, and Magicicada septendecula.3,8,9,14,17 Brood X is the largest brood of 17-year cicadas and emerges at a density up to 1.5 million animals per acre. M. septendecim is a large cicada species with wide, orange stripes on the ventral abdominal surface. 3
Massospora spp. are obligate entomopathogenic fungi in the subphylum Entomophthoromycotina (Zoopagomycota), which includes nearly 250 species that are important pathogens of arthropods. Most fungi in this subphylum, including Massospora spp., are highly host-specific.7,13,16 Massospora spp. have only been reported from annual or periodical cicadas, and cannot be cultured due to their obligate relationships with their hosts. 13 Despite these challenges, morphological and molecular studies have identified both morphological features (eg, spore morphology and size) and genetic loci (eg, nuclear 28S rRNA gene) that can distinguish closely related species, including Massospora cicadina. 13 M. cicadina infects cicadas during their emergence and modifies the sexual behavior of adults to maximize fungal spore dissemination. This altering of the host’s behavior by the pathogen to increase dissemination is termed active host transmission.1,12,13 Massospora fungi are the only pathogen known to be synchronized to the cicada’s long life cycle. 4 The objective of this work is to present pathologic findings of M. cicadina infection in M. septendecim.
During the 2021 Brood X emergence in the eastern United States, 5 male and 2 female, 17-year-old M. septendecim periodical cicadas infected with M. cicadina were collected from the suburban Washington, DC, region. M. septendecim were identified based on key morphological features including abdominal patterning, comparative size, and head markings. These 7 were selected due to the presence of the following characteristic gross lesions of M. cicadina on their posterior abdomen. Grossly, the terminal 3–5 segments of the abdomen of all affected cicadas, including the exoskeleton and portions of reproductive organs contained therein, were absent and replaced by an invasive, white, powdery mass up to 7.1 mm × 7.1 mm × 6.8 mm that extended into the posterior aspect of the coelom (Fig. 1a). No co-infections were noted in these 7 cicadas. Four were deceased, having reached the end of their life cycle, and 3 were humanely euthanized. Multiple animals were collected as control samples, but were found to be infected with various fungal species histologically despite no identifiable gross lesions; therefore, no controls were available.
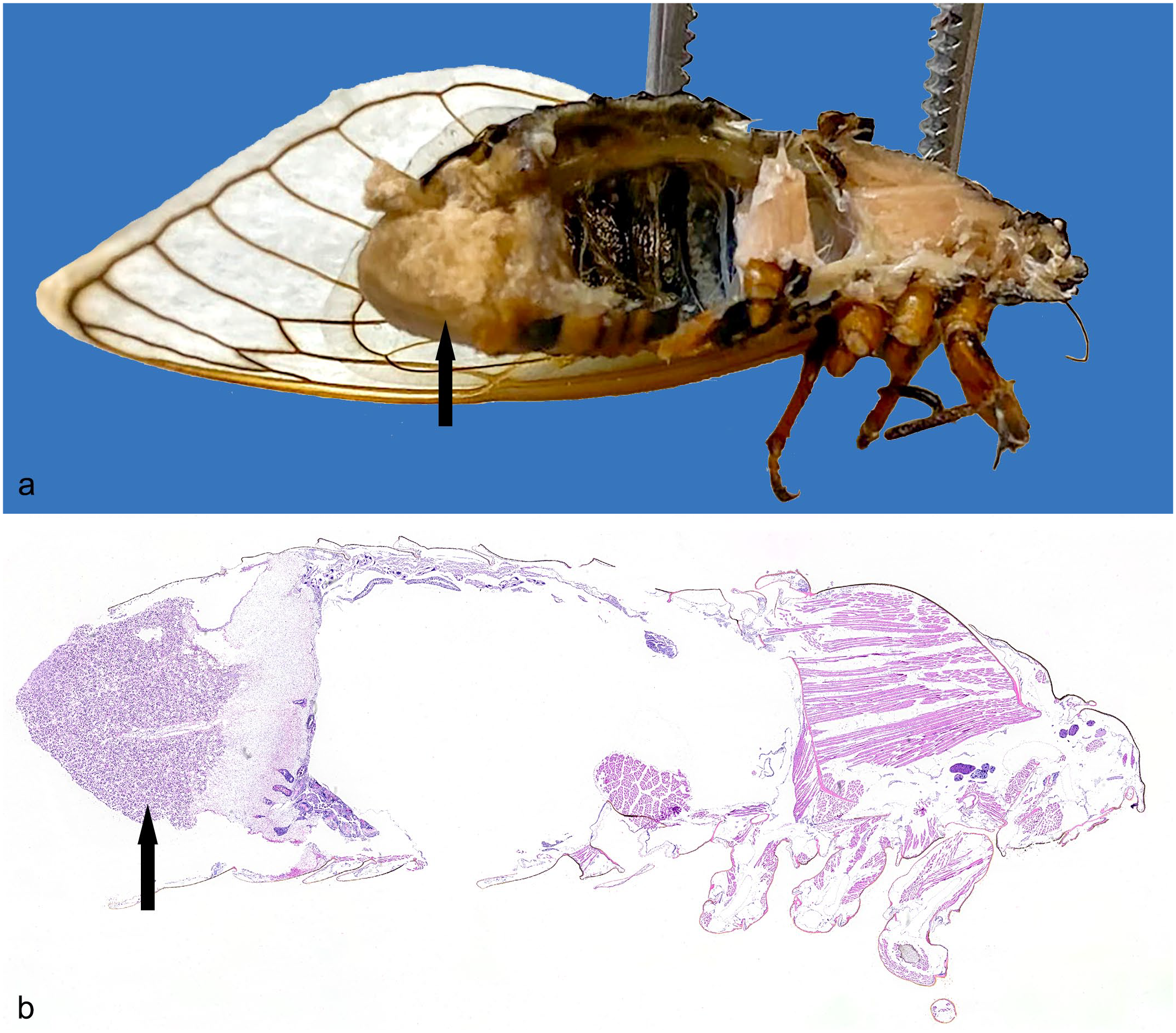
Fungal mass of Massospora cicadina, caudal coelom, bisected specimen, Brood X cicada. (a) A fungal mass (arrow) replaced the terminal segments of the abdomen, including the exoskeleton and reproductive organs, and extended into the posterior aspect of the coelom (fixed specimen). (b) The posterior 30% of the body wall and coelomic cavity were effaced and filled by a fungal mass (arrow). Hematoxylin and eosin (H&E).
The whole body of all cicadas were preserved in 10% neutral-buffered formalin, routinely processed, embedded, and sectioned along a sagittal plane for histology with hematoxylin and eosin (H&E) and Grocott methenamine silver (GMS). The chitinous exoskeleton sectioned satisfactorily without requiring additional processing steps. The following tissues were examined histologically: exoskeleton, skeletal muscle, epidermis, foregut, midgut, hindgut, fat body, hemocytes, trachea, tracheoles, male and female gonads, and the nervous system. 11
All seven cicadas had similar histologic findings. Twenty-five percent to 60% of the posterior body wall and coelomic cavity was effaced and filled by an infiltrative fungal mass that was adhered to the interior surface of the remaining body wall and, in 2 of 7 cicadas, extended anteriorly up to the tymbal. This mass surrounded and replaced up to 90% of the reproductive organs, the alimentary canal, and the visceral fat body (Fig. 1b). At the intersection of the mass and host tissue, there was necrosis with scattered cellular debris and loss of tissue architecture, but no appreciable inflammation. The typical insect immune response to an invading pathogen involves a primary challenge by hemocytes along with melanization of the hemolymph to isolate and eliminate the microorganism. 2 The fungal mass was composed of fungi in multiple stages of development. In 4 of 7 cicadas, approximately 10% of the anterior-most portion of the mass was composed of a mixture of ovoid protoplasts and full-chambered hyphal bodies up to 22 µm in diameter with foamy cytoplasm and 1 to 2 basophilic nuclei. Protoplasts and hyphal bodies are distinguished by the lack or presence of a cell wall, respectively. GMS staining can help to make this distinction as the 2 life stages appear similar with H&E. 10 Protoplasts stain GMS-negative while hyphal bodies are GMS-positive. In those same 4 cicadas, the adjacent 10% of the mass, and in the remaining three cicadas the 10% anterior-most portion, was composed of anuclear empty-chambered hyphal bodies that were up to 40 µm in diameter and lined by a thin eosinophilic membrane. In 4 of 7 cicadas, the intermediate 10% of the mass was composed of 3- to 8-µm diameter conidiophores with nonparallel walls, variable branching, and frequent septation. In 7 of 7 cicadas, the caudal-most portion, comprising the remaining 70% to 80% of the mass and extending outside the animals’ coelom, was composed of numerous, polygonal, fungus-derived packets that were up to 150 µm in diameter and lined by a thin eosinophilic membrane containing dozens of basophilic, irregularly round, verrucose (warty) conidia measuring up to 12 µm × 16 µm and containing one to three, 3- to 5-µm diameter, round nuclei (Fig. 2a). 6
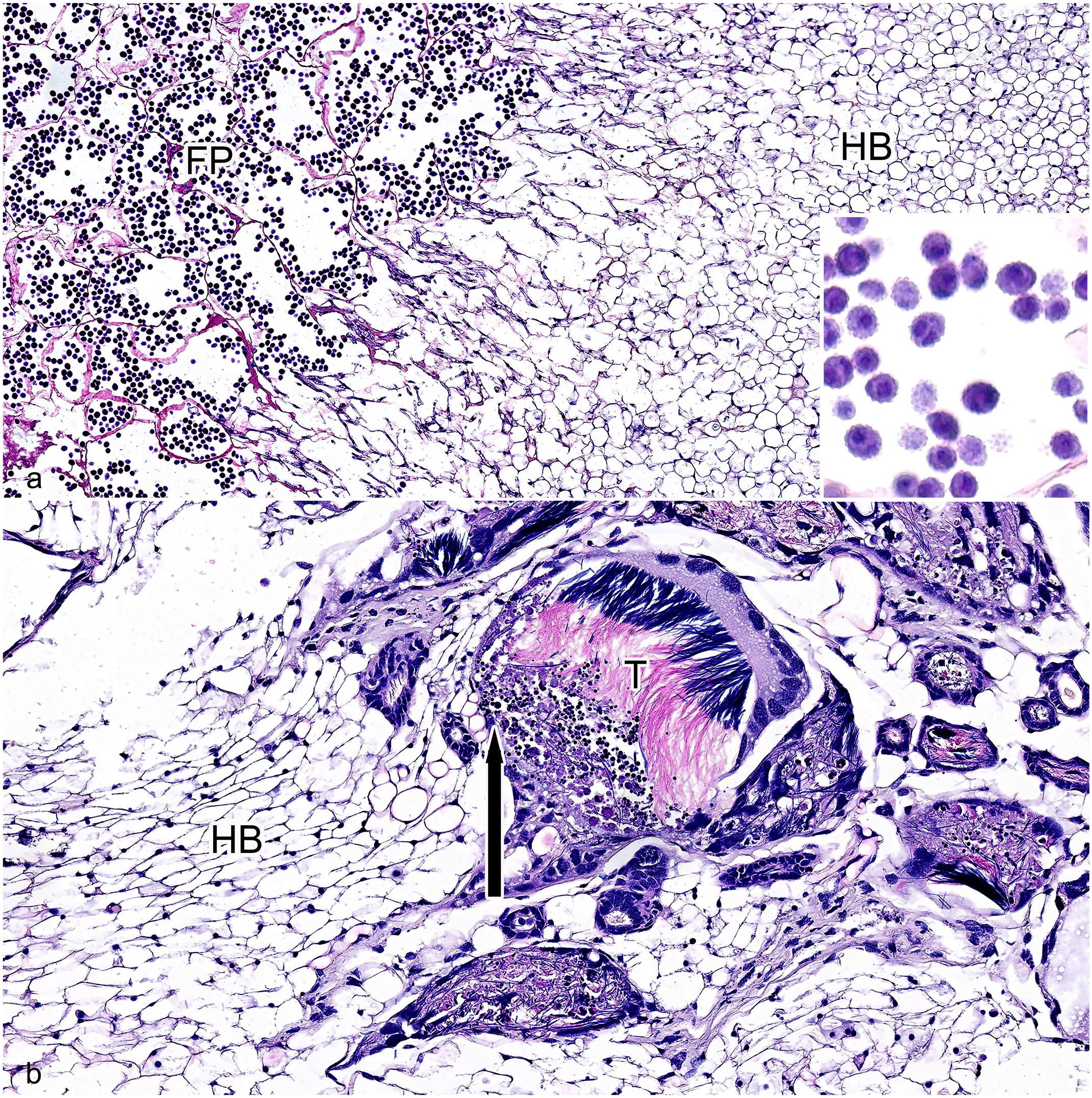
Massospora cicadina infection, caudal coelom, Brood X cicada, H&E. (a) Massospora cicadina fungal morphology. The anterior-most portion of the fungal mass contains anuclear empty-chambered hyphal bodies (HB), up to 40 µm in diameter and lined by a thin eosinophilic membrane. The caudal-most portion of the fungal mass contains numerous polygonal fungal packets (FP), up to 150 µm in diameter and lined by a thin eosinophilic membrane, with dozens of basophilic, irregularly round, verrucose (warty) conidia measuring up to 12 µm × 16 µm with one to three, 3 to 5 µm round nuclei. The intervening portion of the fungal mass was composed of numerous 3 to 8 µm diameter fungal conidiophores with nonparallel walls, variable branching, and frequent septation. Inset: Higher magnification of verrucose conidia within fungal packets. (b) Intersection of fungal mass, as evidenced by hyphal bodies (HB), and host tissue. The fungal mass surrounded and multifocally effaced portions of the testes (T; insect testes have characteristic filamentous sperm). At the intersection of the fungal mass and host tissue, there was necrosis (arrow) and no appreciable inflammation.
Male or female gonads were present in all 7 cicadas. In 7 of 7 cicadas, the gonads were surrounded by and up to 95% of the reproductive organs were replaced by the fungal mass (Fig. 2b). Up to 90% of the visceral fat bodies were also replaced by the fungus in all of the cicadas. Up to 95% of the hindgut was effaced by the mass or heavily infiltrated by fungal growth, and epithelial cells were degenerate with cytoplasmic vacuolation or necrotic with loss of differential staining and scattered cellular and nuclear debris in 6 of 6 insects in which this organ was identified histologically. The hindgut could not definitively be identified in 1 insect, but its normal anatomic location was filled with fungi.
The abdominal body wall was consistently and similarly affected in all 7 cicadas with up to 1/3 of the caudal-most portion transmurally effaced by the mass. The sclerites of the abdominal exoskeleton covering the affected region in 7 of 7 cicadas were necrotic, characterized by fractured and scattered debris, or absent and replaced by the fungal mass. The epidermis lining the abdominal body wall was also affected in all 7 animals. The caudal-most portion was effaced similarly to the exoskeleton while the remaining epidermis, up to the level of the tymbal in 2 cicadas, was variably degenerate or necrotic. The exoskeletal muscle was similarly affected with variable myocyte degeneration, necrosis, or loss. Tracheae and tracheoles adjacent to the affected organs were also obliterated by fungi. The foregut, midgut, hemocytes, and nervous system were unaffected. No fungal organisms were identified anterior to the tymbal.
Massospora cicadina is the only reported Massospora species known to infect periodical cicadas, including M. septendecim. Fungus identification was confirmed morphologically based on their unique verrucose conidia compared with all other Massospora spp. The combined evidence of the host species and distinct formation, shape, and texture of conidia is morphologically diagnostic for Massospora cicadina. 13
It is thought that periodical cicadas are infected by M. cicadina during their emergence at the culmination of their 17-year life cycle. Weeks before the emergence, mature nymphs begin to burrow from their resting places underground toward the soil surface. During this burrowing and emergence is when the cicada likely first encounters infective M. cicadina resting spores in the soil, which have also been dormant for 17 years since the last brood emergence.4,5,7,12,13 Following infection, the fungus remains within the cicada’s abdomen as it ecloses into an adult, eventually causing the caudal-most aspect of its abdomen to slough off, including the genitalia, exposing a white fungal mass.4,5,13 M. cicadina is believed to target the gonads and body wall as part of its life cycle.4,5,13,14 The authors are unaware of any specific research investigating M. cicadina’s impact on the overall Brood X population, despite targeting of the reproductive organs. But we opine that the reproductive success of Brood X is not significantly impacted due to the unfathomably large numbers of cicadas that emerge. The interior of the cicada abdomen contains a hollow chamber that fills almost the entire abdominal volume in males as a key part of their vocalizing. 15 The fungal mass in both males and females was observed to extend anteriorly into the chamber to varying degrees depending on the space available as well as the progression of the infection.
Interestingly, this infection does not cause the death of the cicada. Despite the destruction of the caudal portions of the posterior organs, the remaining viscera in the anterior and mid body were histologically healthy permitting continuation of normal organ functions. While the specifics of how cicadas survive such debilitation is an area of future research, some possibilities include M. cicadina’s role in active host transmission and its production of an amphetamine with neurogenic properties, cathinone, which may enhance the activity of M. septendecim.1,12
Cathinone is suspected to play a role in altering the sexual behavior of diseased males, causing them to mimic the behavior of receptive females in the form of wing-flicks that entice uninfected males into copulating with the diseased males, likely enhancing dissemination of infective and resting fungal spores.1,3-5,7,13 Infective conidia of M. cicadina are thus transmitted through failed mating attempts with both uninfected male and female individuals. These conidial infections develop into M. cicadina resting spore infections, and these resting spores fall to the soil to begin their 17-year hibernation awaiting the next emerging generation.4,5,7,12,13 Resting spores appear histologically similar to conidia except they do not form a plug, but loosely fill the abdomen. 5 Histologically, there was no tissue or inflammatory barrier between the fungal mass and the environment, likely permitting easy fungal dissemination.
The temporally and spatially synchronized life cycles of Magicicada septendecim and Massospora cicadina provide an interesting study on how two organisms can consistently interact despite long periods of dormancy. The 17-year gap between Brood X emergences provides a limited window for pathologists interested in this invertebrate species. These findings help to uncover the pathogenesis of this fungus, as the lesions were minimally inflamed, suggesting evasion of the normal host immune response, and targeting of less-essential tissues (eg, reproductive structures). In addition, a more detailed histopathologic description of the relationship between M. cicadina and M. septendecim than previous studies have documented is provided, which can be used for comparative studies in other fungal infections and diseases of insects.
This article outlines the key gross and histologic features to aid veterinary pathologists in diagnosing M. cicadina mycosis in periodical cicadas. The destruction of the caudal abdomen, including the genitalia and sclerites, by the white, powdery mass is the primary gross finding identified. The histologic correlate was a large fungal mass with various morphologic stages of M. cicadina replacing and invading the body wall and gonads with lesser effects on other tissues.
Footnotes
Acknowledgements
The following technical and administrative staff deserve credit and thanks for their support: from the Naval Medical Research Center and the Walter Reed Army Institute or Research: Stephen Cox, Rex Hartzoge, Theron Jenifer, Brittany Kociuba, Daniel Lozano, Sylriel Peters, Michael Proctor, Jessica Silvers, Jonathan Wilson, and Matthew Wise; from the Joint Pathology Center and the Walter Reed National Military Medical Center: Warren McNeil, Stacey Tamer, Steven McNair, Kenenya Gathers, Ann Brown, Patricia Harris, Andrei Blokhin, Christopher King, and Andrea Cherilus.
Authors’ Note
The views expressed in this article reflect the results of research conducted by the author and do not necessarily reflect the official policy or position of the Department of the Army, Navy, Air Force, Department of Defense, nor the United States Government. Some authors are military service members or federal/contracted employees of the United States government. This work was prepared as part of their official duties. Title 17 U.S.C. 105 provides that “copyright protection under this title is not available for any work of the United States Government.” Title 17 U.S.C. 101 defines a U.S. Government work as work prepared by a military service member or employee of the U.S. Government as part of that person’s official duties. Two authors have begun working at different locations since preparing this manuscript. One (EMC) now works at the Air Force Research Laboratory in San Antonio, Texas and one (JAA) now works at the Uniformed Services University in Bethesda, MD.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
