Abstract
Odontogenic neoplasms demonstrate unique histopathological features and are thought to arise from the germinal tissues of the developing tooth germ, effectively restricting their anatomic origin to the tooth-bearing regions of the jaw and directly associated soft tissues of the oral cavity. Ectopic odontogenic-like neoplasms located in the skin of cats, rabbits, and human beings challenge these assumptions. Here we describe the clinical, pathological, and immunohistochemical features of 6 spontaneously occurring odontogenic-like neoplasms arising in the cutaneous tissue of the cheek in client-owned pet rabbits, including ameloblastoma-like (n = 3), ameloblastic fibroma-like (n = 2), and ameloblastic carcinoma-like neoplasms (n = 1). Microscopically, all the cheek tumors featured neoplastic epithelium exhibiting odontogenic architectural structures (plexiform ribbons, anastomosing trabeculae, follicles, cysts, and irregular structures with rounded botryoid protuberances) and 1 or more cardinal odontogenic epithelial features (basal palisading, antibasilar nuclei, and central stellate reticulum–like cells). The pancytokeratin, cytokeratin 5/6, cytokeratin 14, and vimentin immunohistochemical patterns of these odontogenic-like lesions were most similar to those of jaw-associated ameloblastoma and differed from those of cutaneous trichoblastoma. All neoplasms were narrowly excised, and for lesions with clinical follow-up information, none had evidence of recurrence 1–7 months after surgical removal. Although evidence suggests that these odontogenic-like tumors of the rabbit cheek may be derived from ectopic rests of transformed tooth germ, the histogenesis of these lesions remains unresolved.
Odontogenic tumors occur at any stage of life in a variety of toothed vertebrates and are thought to result from neoplastic transformation of the germinal tissues of the tooth forming apparatus—odontogenic epithelium, pulp ectomesenchyme, or various combinations of these germinal tissues. Ameloblastoma is an odontogenic neoplasm thought to arise from transformed odontogenic epithelium and, as a result, is generally presumed to be anatomically confined to the dentate jaws. The pathogenesis of ameloblastic fibroma is more complex. It is thought to be the result of an inductive signaling process between the neoplastic odontogenic epithelium and subjacent pulp ectomesenchyme. Although local tissue destruction and local invasion of the jaw are often present, the vast majority of odontogenic tumors are considered benign as they do not metastasize to distant anatomic sites. Ameloblastic carcinoma, however, demonstrates malignant cytological features (pleomorphism, increased nuclear/cytoplasmic ratio, nuclear hyperchromatism, increased mitotic activity, abnormal mitoses), vascular/perineural invasion, regions of necrosis, and partial or complete spindle cell morphology in some lesions. 5 Ameloblastic carcinoma is exceedingly rare in veterinary species, and, as a result, its clinical behavior is poorly characterized.
Clinical signs of oral tumors in rabbits include ptyalism, dysphagia, dropping food, anorexia, and oral hemorrhage. 11 From the few cases described in the peer-reviewed literature, leporine odontogenic tumors tend to occur in adult animals and have a predisposition for the maxilla. Odontogenic neoplasms that have been described arising from the mandible or gingival mucosa of rabbits include ameloblastoma,2,10,11,16 complex odontoma, 11 and ameloblastic fibroma. 17 Ameloblastomas arising ectopically from the cheek tissue of 2 rabbits have also been described in the peer-reviewed literature. 10 In human patients, approximately 80% of ameloblastomas are found in the mandible, followed by the maxilla; ameloblastomas arising from the sinonasal tract are rare. 5 Different investigators have come to markedly different conclusions regarding the prevalence of oral odontogenic tumors in rabbits. In a retrospective study of 18 spontaneous oral tumors in rabbits published by a Japanese group of investigators, odontogenic tumors accounted for 28% of all the identified oral tumors. 11 However, in a more extensive review of 1238 tumor-like system-wide lesions in client-owned rabbits examined at a German diagnostic laboratory, only 3 ameloblastomas were identified, resulting in the markedly different occurrence rate of 0.24%. 2 In a separate German study involving 2970 client-owned rabbit pathology submissions (both necropsy and surgical biopsies), no odontogenic tumors were identified, and tumors of the oral cavity represented only 0.2% of the necropsy cases. 3 A portion of this discrepancy in lesion incidence may relate to which oral lesions are actually classified as odontogenic (eg, cementoma and giant cell epulis in the Japanese study). 11 The incidence of leporine oral lesions and odontogenic neoplasms apparently varies by the cited study and perhaps by geographic region. For human odontogenic tumors, rates of reporting are known to vary markedly by geographic region. 1
Adamantinoid trichoblastoma (the term adamantinoma is an antiquated term for ameloblastoma) is a well-documented entity in human dermatopathology that morphologically mimics an ameloblastoma and has also been variably referred to as adamantanoid basal cell epithelioma, cutaneous lymphadenoma, or lymphoepithelial tumor of the skin.4,9 This rare cutaneous lesion in human patients has a predilection for the head and neck and is thought to arise as a variant of trichoblastoma. Adamantinoid trichoblastoma shares histopathologic similarities with ameloblastoma (palisading basal cells with reverse nuclear polarity and central stellate reticulum–like cells), thereby justifying the entity name.15,18
Basal cell adenoma is a salivary gland–derived tumor of human patients that arises in the major salivary glands and very rarely in the minor salivary glands. 5 This neoplasm can demonstrate solid, trabecular, tubular, or membranous architectures and may have peripheral palisading, features reminiscent of an ameloblastoma.
Extragingival peripheral ameloblastoma is an exceedingly rare tumor of human patients arising from the buccal mucosa.8,13 This tumor demonstrates odontogenic features like peripheral palisading of basal cells with antibasilar nuclei and central stellate reticulum–like cells.
Trichoblastoma is a benign tumor of haired skin arising from the germinal epithelial tissue of the hair bulb. Neoplastic cutaneous lesions occur relatively commonly in rabbits, and trichoblastoma is the most common tumor reported in the skin of rabbits. 2 Whether cutaneous adamantinoid trichoblastoma, basal cell adenoma, or extragingival peripheral ameloblastoma exist in rabbits has not been determined.
Here we describe the clinical and pathologic features of 6 ectopic odontogenic-like neoplasms arising as primary lesions within the rabbit cheek and lacking an oral component, including ameloblastoma-like (3), ameloblastic fibroma–like (2), and ameloblastic carcinoma–like neoplasms (1). We describe the clinical, microscopic, and immunohistochemical features of these ectopic odontogenic-like lesions and compare them to the normal structures in the rabbit skin, oral mucosa, cutaneous trichoblastoma, and jaw-associated ameloblastoma.
Materials and Methods
Rabbit Tissues
Spontaneously arising odontogenic-like lesions from the cheeks of 6 client-owned pet rabbits were obtained (Table 1). All the specimens were initially submitted as standard surgical biopsies and subsequently submitted for second opinions to 2 of the authors (B.G.M or E.E.B.L). Radiographs were available for 4 cases. For each case, an excisional biopsy was performed under general anesthesia following standard protocols. The biopsied tissues were fixed in buffered formalin, trimmed into cassettes, embedded in paraffin wax, and routinely processed for histological examination. Four-micrometer-thick sections were stained with hematoxylin-eosin, examined microscopically, and digitally photographed. For 5 of the cases, sections were also stained with Congo red following routine methods. Congo red staining was not performed for the sixth case as the paraffin block was no longer available.
Patient signalment and clinical features of odontogenic-like neoplasms in the cheek of pet rabbits.

Abbreviations: FS, female spayed; L, left; MN, male neutered; ND, not done; Neg, negative; PE, physical examination; R, right; yo, years old.
Control rabbit tissues and neoplastic lesions (normal skin, normal oral mucosa, a jaw-associated ameloblastoma, and a cutaneous trichoblastoma from the cheek) were obtained retrospectively from surgical biopsies of client-owned pet rabbits in the University of California, Davis Veterinary Medical Teaching Hospital’s pathology archives and were processed in a similar manner to the odontogenic-like lesions of the cheeks.
Immunohistochemistry
Immunohistochemistry (IHC) procedures were performed at the UC Davis Veterinary Medical Teaching Hospital Histopathology Laboratory. Formalin-fixed paraffin-embedded tissues were sectioned at 4 µm and adhered to positively charged glass slides. Antigen retrieval after deparaffinization was achieved by Proteinase K (Dako, Ref. S3020) exposure for 7 minutes at room temperature for pancytokeratin (panCK) or heat-induced epitope retrieval at 95°C to 100°C for 30 minutes for CK 5/6 (EDTA Buffer pH8; Thermo Fisher Scientific, Ref. AP-9004-500) and for CK 14 and vimentin (10-mM citrate buffer, pH6).
The primary murine monoclonal antibodies utilized in this study were panCK (Lu-5; Biocare Medical, Ref. CM043C), CK 5/6 (Biocare Medical, Ref. CM105B), CK 14 (keratin 14 Ab-1; Thermo Fisher Scientific, Ref. MS-115-P), and vimentin (Dako, Ref. M7020). The detection method was the same for all the IHC assays (Mouse-on-Canine HRP-Polymer; Biocare Medical, Ref. MC541H). The chromogen used was Vector NovaRED Substrate Kit (Vector Laboratories, Ref. SK-4800). The slides were counterstained with hematoxylin followed by Bluing (Thermo Fisher Scientific). Each IHC test was performed in parallel with known positive/negative control tissue (normal rabbit skin). An additional negative control lacking the detection antibody was performed for each IHC assay.
The IHC staining was graded based on modified criteria set forth by Kok et al: −, 0%; +/−, 1%–10%; +, 11%–50%; ++, 51%–75%; and +++, 76%–100% positive tumor cells. 7 At least 3 different stained slides/tissues were examined to assess the labeling features of normal skin and oral mucosa tissues.
Results
Rabbits
The age of the rabbits in this study ranged from 6 to 8 years and included 2 neutered males, 3 spayed females, and 1 rabbit of unknown sex (Table 1). Four cases had a history of a slow growing mass prior to excision: 2 months duration in 3 rabbits and 12 months duration in 1 rabbit. One case was identified at the time of surgery. In 3 cases, the masses were identified incidentally during routine physical examination. In 1 case, the mass was identified by the owner prior to physical examination. It was not stated how the masses were identified in the remaining 2 cases. Clinical examination revealed that the masses were freely movable in the cheek tissue and not adhered to the oral mucosa, bone, or teeth. Radiographic and/or computed tomography studies were performed in 4 cases (cases 1–4), which confirmed there was no bone or tooth involvement. None of the animals had clinical signs referable to the tumors.
All 6 masses were removed with excisional biopsy, and surgical margins were narrow (narrowest histologic tumor-free margin of 50 µm–1.1 mm). Case 4 had radiation therapy following surgical removal. Follow-up information was available for 4 cases (cases 2–5), all of which lacked regrowth at recheck examination 1–7 months after the surgery.
Microscopic Features
Ameloblastoma-like lesions
Histologically, the ameloblastoma-like lesions (cases 1–3) were circumscribed, unencapsulated to pseudoencapsulated masses with compressive (noninvasive) margins. Cases 2 and 3 were submucosal (located medial to the buccal musculature) with no direct interaction between the cutaneous or mucosal epithelium and the lesion. Case 1 was subcuticular (external to the buccal musculature), and the lesional epithelium was multifocally contiguous with the overlying epidermis. No minor salivary gland tissue was evident in any of the examined sections. The lesions were composed of abundant neoplastic odontogenic-like epithelium (OLE) featuring peripheral palisading of cuboidal to columnar epithelial cells, variable antibasilar nuclei, and variably prominent centrally located stellate epithelial cells (stellate reticulum–like cells, Fig. 1a). The OLE was arranged in various architectural structures including plexiform ribbons, anastomosing trabeculae, solid follicles, cysts, and irregular structures with botryoid protuberances (“ink drop” architecture). The epithelium was frequently associated with minimal to moderate amounts of subepithelial basophilic ectomesenchyme. In some areas, the OLE had features of differentiated, palisading ameloblasts with antibasilar nuclei and thin subjacent ribbons of brightly eosinophilic collagenous stroma (hyalinized collagen, Vickers-Gorlin effect; Fig. 1b). No cytological atypia or mitotic figures were identified. Islands of neoplastic OLE had variable central keratinization (central squamous metaplasia) and scattered intraepithelial mineralization. In case 3, the associated stroma had abundant metaplastic bone, and the neoplastic tissue had central ischemic necrosis. In case 1, the neoplastic OLE had a nodular arrangement separated by eosinophilic fibrous stroma. In case 2, multiple regions of the OLE had black melanin-like pigment granules. Surgical excision was judged to be complete in all cases, and the narrowest histologic tumor-free margins were 1.1 mm, 70 µm, and 100 µm in cases 1, 2, and 3, respectively.
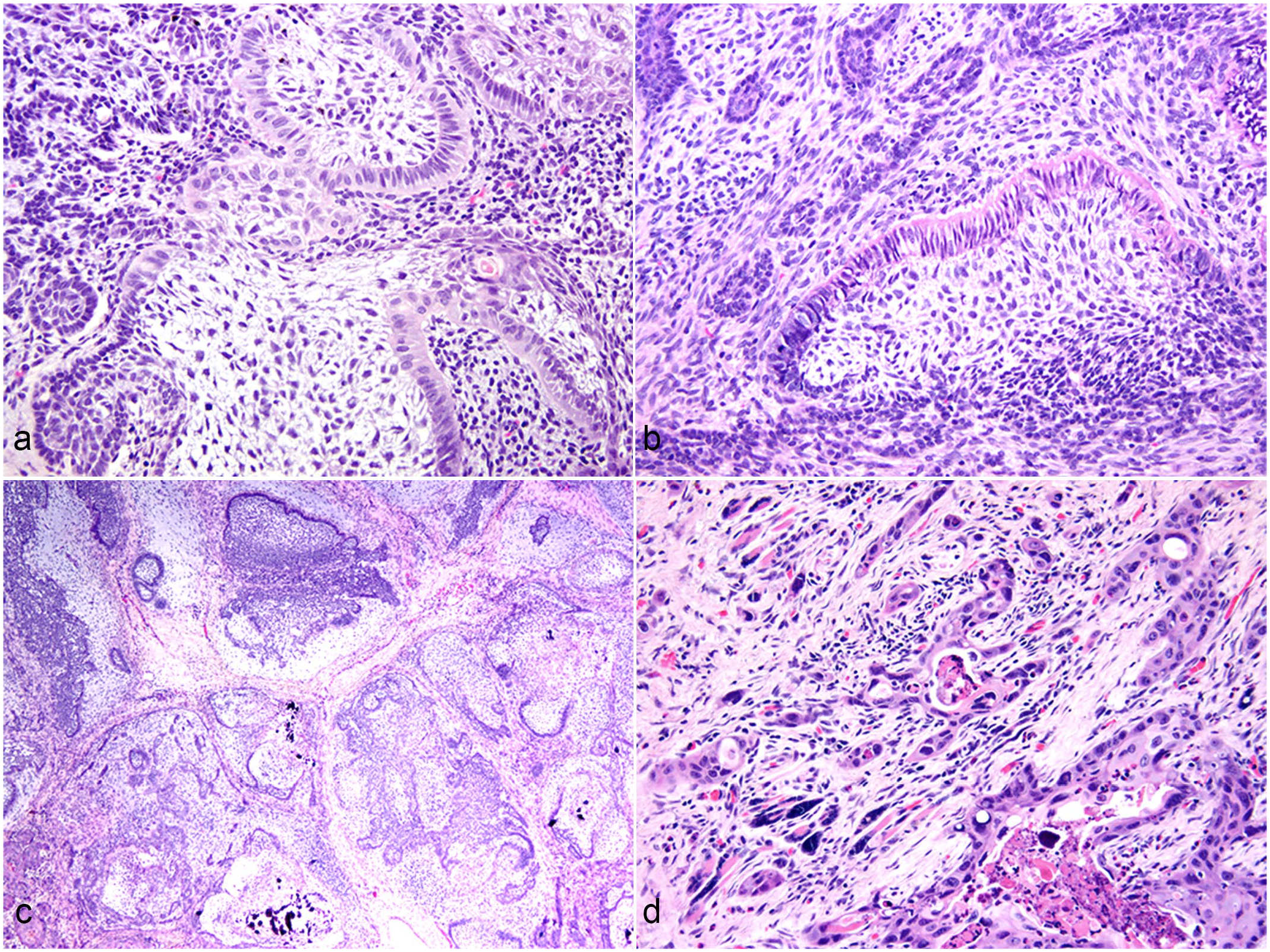
Ectopic odontogenic-like lesions, cheek, rabbit. Hematoxylin & eosin. (a) Neoplastic epithelium forms peripheral palisades with antibasilar nuclei and central stellate reticulum–like cells (case 2). (b) A thin band of hyalinized collagen is present along the basal aspect of the palisading epithelium (case 1). (c) Neoplastic epithelium is associated with abundant basophilic pulp-like ectomesenchyme and is organized into nodules (case 4). (d) Neoplastic epithelium invades the subjacent skeletal muscle tissue and is associated with myocyte atrophy/regeneration, a collagen-rich stroma, and mild inflammation (case 6).
Ameloblastic fibroma-like lesions
Cases 4 and 5 were submucosal with no direct interaction between the neoplastic epithelium and either the skin or oral mucosa. No minor salivary gland tissue was identified. These cases comprised neoplastic OLE that demonstrated the histological features described above for the ameloblastoma-like lesions, and the neoplastic OLE was embedded within abundant pulp-like ectomesenchyme (ameloblastic fibroma-like, Fig. 1c). The pulp-like tissue comprised variably dense (typically low density) spindloid mesenchymal cells with bland cytological features within an abundant basophilic, proteoglycan-rich matrix. In the majority of the examined sections, the neoplastic OLE and surrounding ectomesenchyme were arranged in a nodular pattern separated by fibrous connective tissue stroma. In case 5, the neoplastic OLE rarely formed cap-like structures surrounding the induced pulp-like ectomesenchyme and hyalinized subepithelial collagen (Vickers-Gorlin effect). Neither the neoplastic OLE nor the induced pulp-like ectomesenchyme demonstrated cytological atypia. Scattered foci of extracellular mineralization were present in both the OLE and ectomesenchyme. Surgical excision was judged to be complete, and the narrowest histologic tumor-free margins were 50 µm in both cases.
Ameloblastic carcinoma-like lesion
Case 6 was subcuticular and multifocally contiguous with the epithelium of the skin. No minor salivary gland tissue was identified. The lesion demonstrated regional microscopic features consistent with a diagnosis of ectopic ameloblastoma, as described above. Adjacent to the region of the tumor demonstrating odontogenic-like features, the neoplastic epithelium multifocally invaded into the endomysium of adjacent skeletal muscle tissue, entrapped myocytes, and exhibited pronounced stromal desmoplasia with intraepithelial and stromal inflammation (Fig. 1d). Regions of the lesion demonstrated features consistent with carcinoma including cutaneous ulceration, central comedonecrosis, loss of basal palisading, loss of organized cellular differentiation, pronounced epithelial anisokaryosis, epithelial multinucleation, and 5 mitotic figures in 2.37 mm2. Surgical excision was judged to be complete, and the narrowest histologic tumor-free margin was 120 µm.
Congo red and Immunohistochemistry
In Congo red–stained sections (cases 1, 2, 4, 5, and 6), the extracellular material was determined to be Congo red negative. Some of this extracellular material was presumed to be dystrophically mineralized necrotic debris and tissue. The immunohistochemical results of the normal rabbit skin and oral mucosa are listed in Supplemental Table S1 for panCK, CK 5/6, CK 14, and vimentin. As expected, no epithelial structures in the skin or mucosa were positive for vimentin. The majority of the cutaneous and mucosal epithelial structures were diffusely positive for panCK. However, for CK 5/6, only the outer layers of the skin and oral mucosa labeled positively (stratum spinosum and stratum corneum/superficial layer). For CK 14, the cutaneous basal layer, stratum spinosum, follicle, and dermal adnexa labeled positively, while in the oral mucosa, the basal layer and, to a lesser degree, stratum spinosum labeled positively.
The results of the IHC panel were similar for all 6 odontogenic-like neoplasms, regardless of the final diagnosis (Table 2). Strong, diffuse panCK and CK 14 labeling was present in basal neoplastic epithelial cells (ameloblast-like cells) and central stellate reticulum. Diffuse CK 14 labeling of the neoplastic epithelium was similar for the jaw-associated ameloblastoma (Fig. 2a) and cheek-associated ameloblastoma-like tumor (Fig. 2b) and was different from the cutaneous trichoblastoma, in which the neoplastic epithelium was negative (Fig. 2a inset). PanCK labeled the central squamous metaplasia, while CK 14 did not.
Immunohistochemistry results for rabbit neoplasms, including six cases of odontogenic-like neoplasms (Cases 1-6).
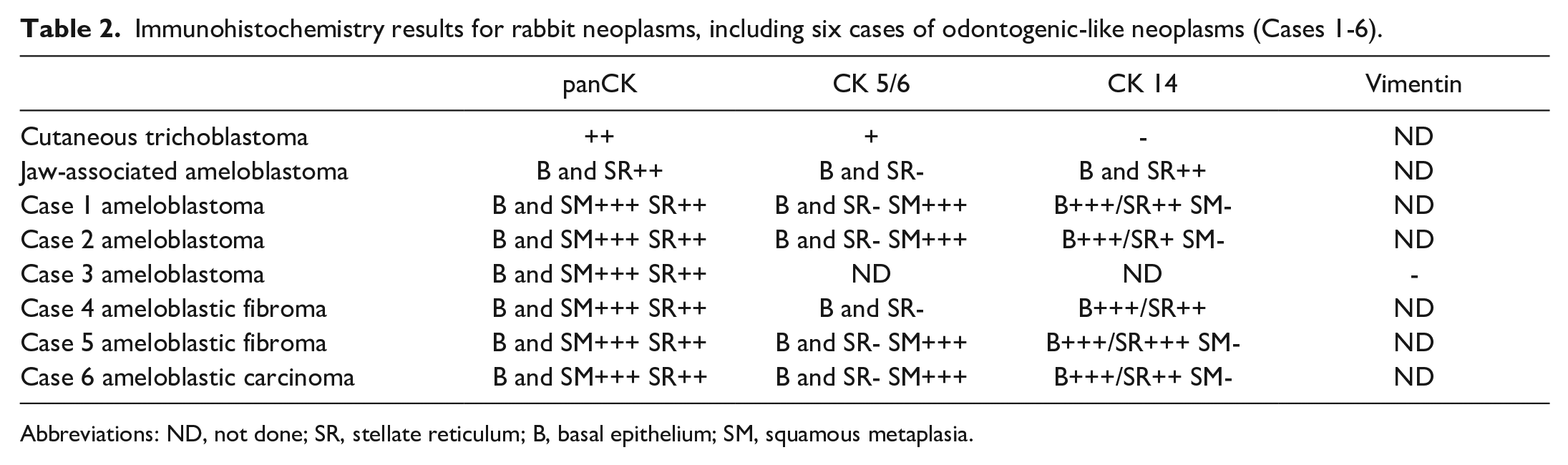
Abbreviations: ND, not done; SR, stellate reticulum; B, basal epithelium; SM, squamous metaplasia.
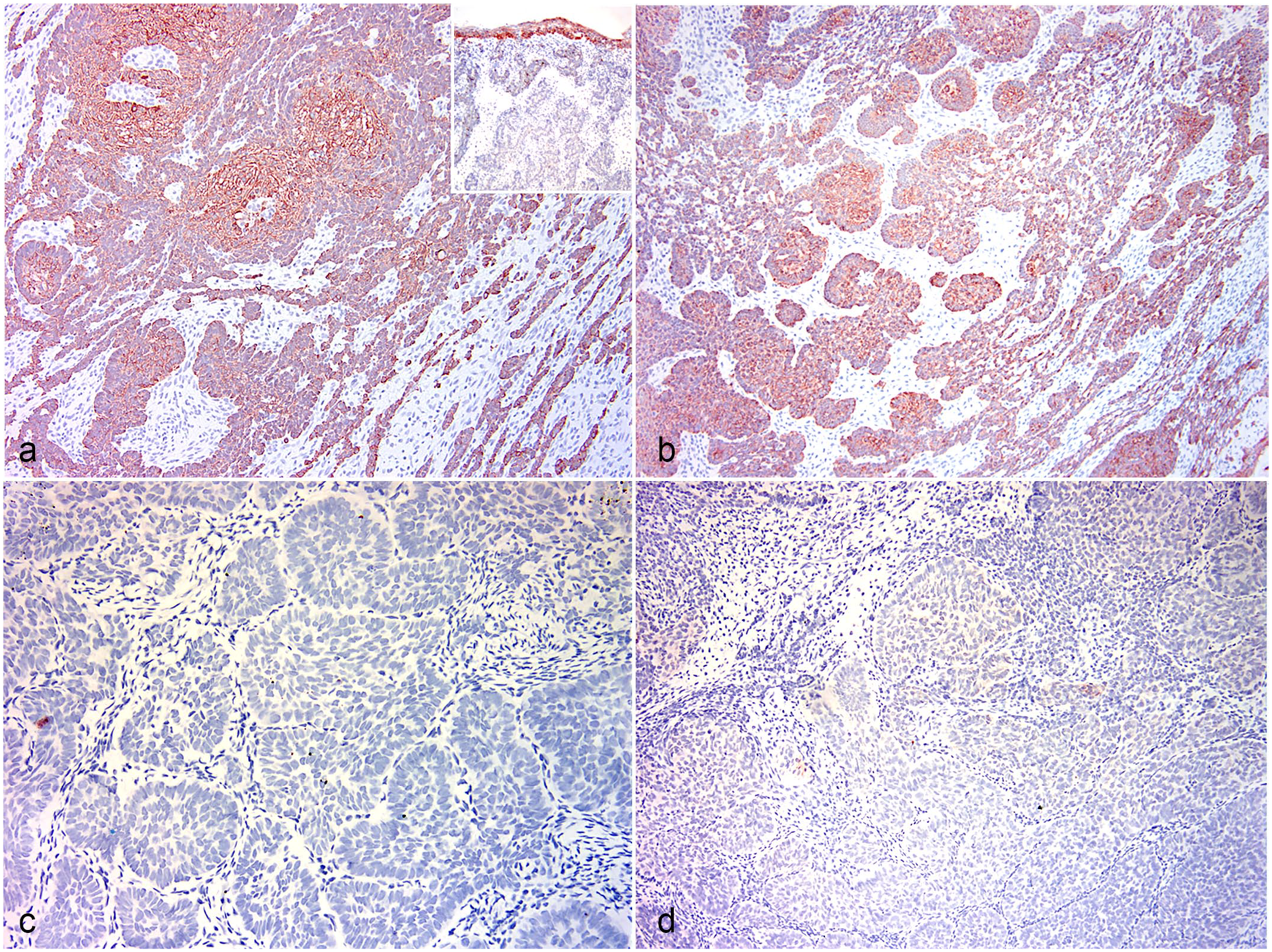
Rabbit. Immunohistochemistry (IHC). (a) Jaw-associated ameloblastoma lesion. Diffuse immunolabeling of the basal and central portions of the neoplastic epithelium. IHC for cytokeratin (CK) 14. Inset: skin. Cutaneous trichoblastoma. Neoplastic epithelium does not stain while the overlying epidermis stains positively. Case 2. IHC for CK 14. (b) Ectopic ameloblastoma-like lesion, cheek. The basal and central portions of the neoplastic epithelium stain diffusely. Case 1. IHC CK 14. (c) Jaw-associated ameloblastoma lesion. The neoplastic epithelium does not stain. IHC for CK 5/6. (d) Ectopic ameloblastic fibroma-like lesion, cheek. The neoplastic epithelium does not stain. Case 4. IHC for CK 5/6.
CK 5/6 labeling was largely negative in the neoplastic epithelium of the jaw-associated ameloblastoma (Fig. 2c) and cheek-associated odontogenic-like tumors (Fig. 2d). As expected, vimentin-positive staining was not identified in any of the epithelial structures. Collectively, these IHC results for the cheek-associated odontogenic-like tumors are most similar to the results for the jaw-associated ameloblastoma and differ from the labeling pattern for cutaneous trichoblastoma (Table 2).
Discussion
Here we describe 6 neoplasms with odontogenic features arising as primary lesions in the cheek tissue of rabbits. Three of these lesions had microscopic features of ameloblastoma, 2 were most consistent with ameloblastic fibroma, and 1 lesion had features consistent with ameloblastic carcinoma. In all cases, the lesional neoplastic epithelium was architecturally arranged as plexiform ribbons, anastomosing trabeculae, follicles, cysts, and “ink drop” structures and was organized into peripheral palisades of cuboidal to columnar cells with antibasilar nuclei (reverse polarity) and central epithelial cells with features of stellate reticulum. Collectively, these microscopic findings are generally considered to indicate an odontogenic histogenesis (ie, derived from the epithelium of the enamel organ). However, because the ameloblastic carcinoma lesion demonstrated regional features consistent with both ameloblastoma and squamous cell carcinoma, 2 possible pathogenic mechanisms should be considered: divergent differentiation of an odontogenic-like tumor or a collision lesion of ameloblastoma and squamous cell carcinoma.
The immunohistochemical findings for normal skin and oral mucosa were largely in agreement with previously published results. 7 A slightly reduced proportion of cells stained positively in the current study relative to this previous immunohistochemical study of trichoblastoma in the rabbit. 7 However, the small number of control lesions examined (a single trichoblastoma and one jaw-associated ameloblastoma lesion) is a recognized weakness of this study, and some variation in IHC staining intensity in rabbit trichoblastoma lesions has been previously documented, particularly for CK 14. 7 The immunohistochemical labeling pattern of the cheek-associated lesions was most similar to that of the jaw-associated ameloblastoma (the basal and stellate reticulum–like cells did not label with CK 5/6 but labeled positively with CK 14) and differed from the labeling pattern of the trichoblastoma lesion arising in the skin of the cheek (neoplastic epithelium in the trichoblastoma lesion labeled positively with CK 5/6 and did not label with CK 14).
The proposed ectopic occurrence of primary odontogenic tumors in the cheek tissue of rabbits seemingly violates the dogma that these tumors arise exclusively in the maxillofacial skeleton (central) or in the oral mucosa overlying the dentate jaws (peripheral) where the germinal tissues of the tooth are located. 1 Multiple lesions with odontogenic features apparently occur outside of the dentate jaws of human patients, including adamantinoid trichoblastoma, salivary gland–derived basal cell adenoma, sinonasal ameloblastoma, and extragingival peripheral ameloblastoma. There are at least 2 hypotheses that could explain the ontology of these ectopic odontogenic-like lesions: (1) The lesions arise from ectopic germinal rests of odontogenic epithelium, or (2) the lesions represent a form of phenotypic mimicry and arise in situ from the cutaneous adnexa or minor salivary glands (variant of trichoblastoma or basal cell adenoma).
Amyloid-producing odontogenic tumor (APOT, also known as amyloid-producing ameloblastoma) has been described previously in the facial skin of 3 cats. 6 According to the authors of that report, these ectopic odontogenic lesions, arising from the supraorbital skin or labial skin, demonstrate the same histopathological features as APOT arising from the dentate jaws. Based on the IHC results for ameloblastin, amelogenin, and sheathlin, the authors hypothesized that residual germinal rests of odontogenic epithelium may exist in the facial skin outside the oral cavity. 6 Although this panel of antibodies could also be used in the dissection of the ectopic odontogenic-like lesions discussed here, the antibodies would need to be validated for use in rabbit tissues.
In human patients, the cutaneous adamantinoid trichoblastoma lesion exhibits odontogenic-like features and is thought to arise from the hair follicle. The histogenesis of this rare cutaneous lesion of human patients has been debated since at least 1959. 14 Although adamantinoid trichoblastoma shares odontogenic features with the rabbit lesions and both the basal cells and central cells immunohistochemically label with panCK, it also features prominent lymphocytic infiltration, 19 which was not identified in any of the rabbit lesions. In addition, other immunohistochemical studies indicate that adamantinoid trichoblastoma derives from stem cells of the hair follicle bulge, expressing CK15 and PHLDA1. 12
The authors of the study described here do not believe it is possible to unequivocally confirm the histogenesis of these rabbit lesions given the current data. However, the balance of the evidence supports a histogenesis featuring ectopic germinal rests: (1) IHC results are more consistent with jaw-associated ameloblastoma than cutaneous trichoblastoma; (2) morphologically, the lesions described here encompass a variety of odontogenic-like lesions, not simply ameloblastoma; and (3) to date, the lesions have only been identified in the rabbit cheek tissue and not elsewhere in the rabbit skin. Unequivocal proof of lesional histogenesis will likely require some combination of molecular testing and/or additional IHC assays.
While maxillary ameloblastomas in rabbits requiring maxillectomy are associated with a poor prognosis, adequate lesional resection appears to be possible for these cheek-based tumors, as all cases for which postexcisional information was available lacked recurrence at follow-up. However, the sample size of this study was small, and follow-up data were only available for 7 months. Therefore, definitive conclusions on the prognosis of these cheek lesions must be interpreted with caution.
Footnotes
Acknowledgements
We are grateful for the histopathology expertise of the histotechnologists and wish to express our gratitude to Alyssa Osier of Zoetis for assisting with tissue processing and slide scanning. We also thank the veterinary clinicians who submitted case material, especially Hannah Attarian of Arizona Exotic Animal Hospital, Julianne Richard from Cummings School of Veterinary Medicine at Tufts University, Ross Weinstein of Peninsula Dog and Cat Clinic, and Sandra McRoberts of PetCare Veterinary Clinic.
Disclaimer
The views expressed in this article are those of the authors and do not necessarily reflect the official policy of the Department or Defense or the U.S. Government.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by the UC Davis Dermatopathology & Dermatology Consulting Service.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
