Abstract
The source and significance of pulmonary silicate crystals in animals and people are poorly understood. To estimate the prevalence and characterize the pulmonary crystalline material in animals from St. Kitts, tissue samples from dogs, horses, cattle, sheep, goats, pigs, chickens, mongooses, and monkeys were examined by light microscopy, scanning electron microscopy with energy-dispersive x-ray analysis (SEM/EDXA), and x-ray diffraction. Crystalline material was seen in 201 of 259 (77.6%) lung samples as perivascular and interstitial accumulations of heterogeneous crystalline particulate material, free or within macrophages (silicate-laden macrophages [SLMs]), mostly lacking evidence of chronic inflammation or fibrosis. The crystalline material was birefringent, basophilic on acid-fast, and composed of silicas on SEM/EDXA. Mongooses (100%) and monkeys (98%) had the highest prevalence of SLM, followed by cattle and chickens. Lesions were graded on a 3-point scale based on the histologic location and extent of silicates and SLM and were significantly more severe in mongooses (median = 3) than in monkeys (median = 2), dogs (median = 2), and chickens (median = 1). On EDXA, the crystalline material from lungs, air, and topsoil was composed of silicon, oxygen, aluminum, and iron, with a particulate matter size between 2.5 and 10 µm. We hypothesize Saharan dust, volcanic ash, topsoil, and rock quarry dust are potential sources of siliceous dust inhalation and SLM accumulations lacking chronic inflammation (silicosis); dust generation may be potentiated by road vehicle or wind suspension. Future investigations are warranted on the role of silicate inhalation and respiratory comorbidities in people, with monkeys, mongooses, or chickens serving as possible sentinels for exposure.
Keywords
Pneumoconiosis is a lung disease in humans and animals characterized by the inhalation and retention of inorganic dusts. 14 Pneumoconiosis is typed based on the category of inhaled inorganic material. The most clinically relevant materials responsible for pneumoconiosis in humans are those acquired through occupational exposure. Carbon, asbestos, beryllium, talc, and silica are examples of inorganic dusts which can lead to varying degrees of occupational pneumoconiosis in people working in specific industries. 49 One of the most common forms of pneumoconiosis in humans is silicosis, a chronic, progressive lung disease resulting in sustained inflammation and culminating with fibrosis and compromised pulmonary function.11,13,29,41,56
The particulate responsible for the development of silicosis is silica, which is composed of silicon dioxide (SiO2) or silicic anhydride found in either amorphous or crystalline forms and is 1 of the most abundant minerals in the Earth’s crust.19,47 The dosage of inhaled silica dust and duration of exposure results in the development of subsequent pneumoconiosis. 41 Silicon dioxide polymorphs were reported to have clinical significance in animals as recently presented in a study associating pulmonary silicosis with an equine bone fragility syndrome. 3 The horses in this report were environmentally exposed to silicates, a form of silica which is a crustal mineral salt. Silicates are silicon dioxide combined with 1 of a variety of cations (magnesium, aluminum, iron, or calcium) and are a component of much of the Earth’s topsoil.3,19
Pulmonary silicate-laden macrophage (SLM) accumulation unassociated with silicosis has been occasionally reported, including a recent report in European badgers. 51 This nonfibrotic form of pneumoconiosis is usually not associated with a clinical lung disease, and inflammation ranges from none to mild.4,7,18,53
Histologic examination of lung tissue collected from canine autopsy cases submitted during 2017 to the pathology services of Ross University School of Veterinary Medicine (RUSVM) revealed an unusually high prevalence of what appeared to be an incidental finding: peribronchiolar and perivascular brown to black, sometimes birefringent, crystalloid material free or within macrophages, interpreted as SLM. A follow-up retrospective investigation of autopsy cases submitted in the previous 2 years revealed the same finding in the lungs of a wide range of domestic and wildlife animal species. The purpose of this study was to assess the prevalence of SLM in a wide range of animals from St. Kitts, characterize the histologic features in the lung, grade the intensity of the crystalline material accumulations, and identify the source, composition, and structure of the particulate material from lung, air, and topsoil samples.
Materials and Methods
Sampling and Histopathology
The study took place at the Caribbean Island of St. Kitts, part of the federation of St. Kitts and Nevis, (Fig. 1a, b) between 2016 and 2018. Lung samples were collected from a variety of wild and domestic animals (259 animals in total). Demographic data (species, sex, age, and location) were gathered. There were 25 dogs (11 male and 14 female), 3 horses (2 male and 1 female), 18 cattle, 20 small ruminants (7 sheep and 13 goats), 20 pigs, 82 chickens (32 male and 50 female), 40 small Indian mongooses (Urva auropunctata) (21 male and 19 female), and 51 African green monkeys (Chlorocebus aethiops) (29 male and 22 female). The dog and horse lung samples were obtained from the archival tissue of animals submitted for postmortem examination to the pathology services of RUSVM between 2016 and 2017. Lung samples from cattle, small ruminants, and pigs were collected (convenience sampling with no age and sex available) from animals slaughtered at the local abattoir. Free-roaming backyard chicken, mongoose, and monkey samples were kindly donated from other RUSVM research studies and had been reviewed and approved by the Intitutional Animal Care and Use Committee.6,42 Where feasible, air and soil samples were taken on sites overlapping with the location of origin of the sampled animals (N = 49; Fig. 1c).
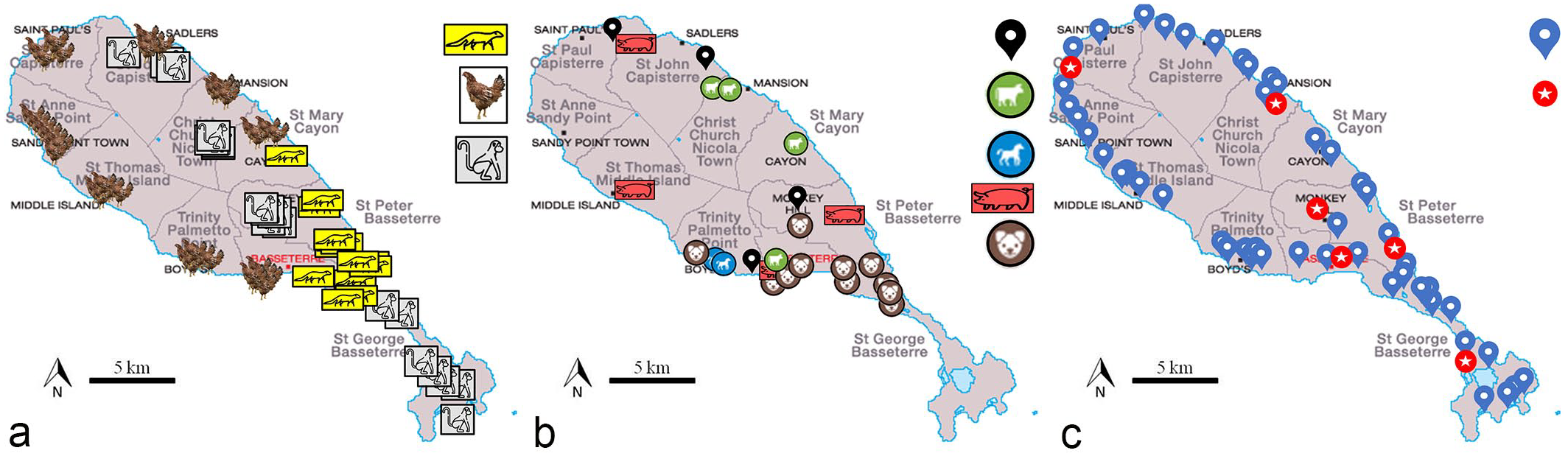
Animal and environmental sampling sites from the island of St. Kitts. Images modified from Google Maps. (a) Sampling sites for the 3 species with the highest prevalence of pulmonary silicate-laden macrophages (SLMs). Symbols represent mongoose, chickens, and monkeys. (b) Sampling sites for the 5 animal species with a lower prevalence of pulmonary SLM. Symbols represent small ruminants (black pin), cattle, horse, pigs, and dogs. (c) Air (blue pin) and topsoil (red pin) sample sites.
A 4 cm × 2 cm × 0.5-cm section of lung was taken from the cranial and caudal lobes of the right lung in larger species, except for those samples obtained from archival tissue and from mongooses and chickens, because of their smaller size. The lung from the mongoose was sectioned across both the right and left middle lobes including the main bronchi and tracheobronchial lymph nodes, while in chickens, a section from the left and right lungs was examined in each case. All samples were fixed in 10% buffered formalin for a minimum of 24 hours, trimmed, routinely processed, embedded in paraffin, cut at 4 µm, mounted on glass slides, and stained with hematoxylin and eosin (HE) and Ziehl-Neelsen acid-fast stain (AF).
Grading System for SLM
All lung samples were histologically evaluated and graded. If a free or phagocytized, partially birefringent crystalline material compatible with silicates was associated with pulmonary fibrosis and macrophage infiltration with intracytoplasmic crystals, the lesion was classified as pulmonary silicosis, as previously described. 3 If fibrosis was absent, this finding was diagnosed as SLMs. 3 An AF was performed to rule out the presence of acid-fast bacteria and to help visualize and grade the crystalline particulate matter. 3 We used a grading system to semiquantitatively categorize the extent (not the severity) and location of peribronchiolar and perivascular crystalline deposits within lung samples, based on a previously published grading methodology to assess suspected cases of pneumoconiosis.7,16 Our grading scheme ranged from 0 to 3, in which grade 0 represented unaffected lung, grade 1 (Fig. 2a) means that up to 33% of peribronchiolar and perivascular spaces were affected (contained silicates), grade 2 (Fig. 2b) 34% to 66% of peribronchiolar and perivascular spaces were affected, and grade 3 (Fig. 2c) more than 66% of the peribronchiolar and perivascular spaces were affected.
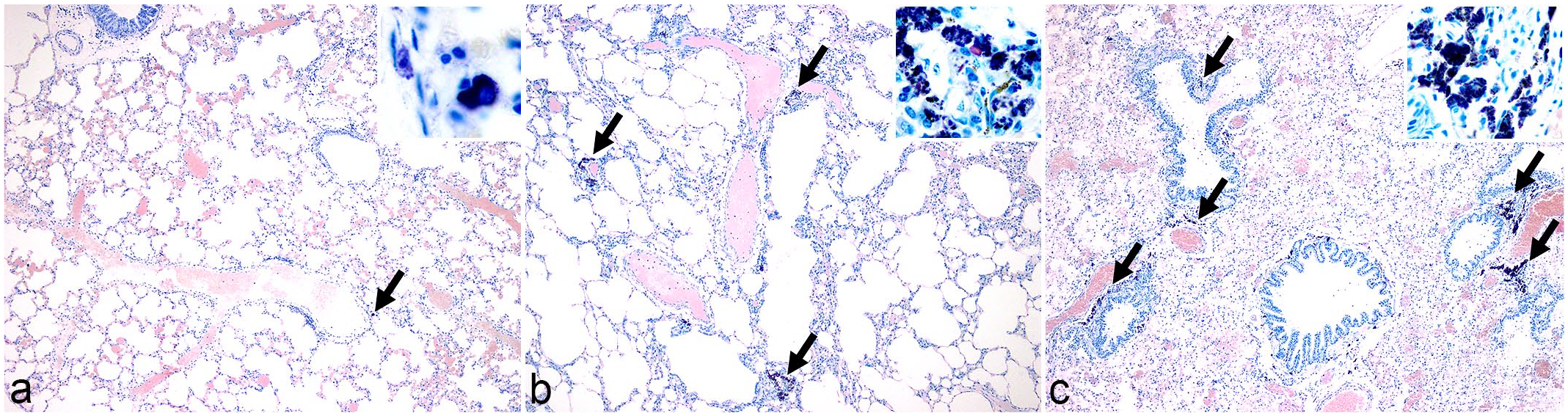
Grading system for pulmonary silicate-laden macrophage (SLM) accumulations (arrows), acid-fast paraffin-embedded sections. Insets: high magnification of silicate crystal depositions. (a) Lung, mongoose, no. 98. Grade 1 SLM. Crystalline material is seen around less than 33% of the larger airways and blood vessels. (b) Lung, monkey 12. Grade 2 SLM. Crystalline material is seen around more than 33% but less than 66% of the larger airways and blood vessels. (c) Lung, mongoose 38. Grade 3 SLM. Crystalline material is seen around more than 66% of the larger airways and blood vessels.
Immunohistochemistry and Histochemistry
To categorize the cellular population present within the areas of crystalline material deposition, 10 formalin-fixed lung samples, 5 from dogs (2 grade 2 and 3 grade 3 SLM accumulations) and 5 from African green monkeys (one grade 1, 1 grade 2, and 3 grade 3), were submitted for immunohistochemistry using IL1R1, a marker for M1 macrophages (inflammatory macrophages). Unstained paraffin-embedded 5-µm sections of lung were placed on Tanner+ slides and submitted to StageBio laboratories. The slides were deparaffinized in xylene and rehydrated through a series of alcohol baths from 100% to 70%. Once rehydrated, they were placed in Biocare’s Decloaking Chamber and heated to 110°C for 15 minutes in Diva Decloaker (Biocare Medical, Pacheco, CA). The slides were then placed onto the intelliPATH FLX autostainer (Biocare Medical, Pacheco, CA) and blocked with Peroxidazed 1 (Biocare Medical, Pacheco, CA) for 5 minutes, Background Punisher (Biocare Medical, Pacheco, CA) for 10 minutes, and 30% horse/30% goat serum for 30 minutes. The primary antibody (interleukin 1 receptor, type I, IL1R1 LifeSpan LS-C471530) 45 was diluted 1:1000 in a Monet blue diluent and incubated on the slides for 60 minutes. The antibody was validated for immunohistochemistry (IHC) use in monkeys and dogs. 45 Once the incubation with the primary antibody was completed, the slides were covered with streptavidin HRP. The streptavidin HRP was diluted 1:1000 (1 ug/ml) in Monet blue diluent and allowed to incubate for 30 minutes. The slides were visualized with Betazoid diaminobenzidine for 5 minutes and counterstained with Mayer’s hematoxylin. TBST (Tris-Buffered Saline Tween-20) was used to rinse the slides in-between each step. The slides were examined using light microscopy.
To assess the presence of fibrosis, paraffin-embedded sections of the same 10 lung samples selected for IHC (5 from canine and 5 from primate species) were stained with Masson’s trichrome and examined using light microscopy. They were selected to include different grades of pulmonary SLM accumulations.
Scanning Electron Microscopy and Energy-Dispersive X-Ray Diffraction Analysis
Three lung samples, 1 each from chicken, African green monkey, and dog, with microscopically visible crystalline material within the pulmonary interstitium (all grade 3), were submitted as formalin-fixed paraffin-embedded (FFPE) blocks to the Joint Pathology Center for scanning electron microscopy (SEM) and energy-dispersive x-ray analysis (EDXA) to elucidate the size and chemical constituents of the crystalline material. Lung sections from FFPE tissues were mounted on a polished carbon planchet for SEM with energy-dispersive x-ray analysis (SEM-EDXA; Hitachi S-3400N scanning electron microscope; Hitachi High Technologies; X-Max 80 energy dispersive x-ray spectrometer; Oxford Instruments). Backscatter images and EDXA spectra and maps were obtained at 20 kV accelerating voltages from several areas across the tissue section. 34
Six additional FFPE lung samples (all SLM grade 3) (2 each from dogs, African green monkeys, and Indian mongooses) were submitted to Metallurgical Engineering Services, Inc. (Richardson, TX, USA, https://www.metengr.com/) to be acid-digested, rinsed, and dried prior to testing the crystals present with a Rigaku Ultima III detector (Rigaku Corporation, Tokyo, Japan). An x-ray diffractometer with a high-precision theta-theta goniometer was used to qualitatively identify the crystalline phases. X-ray intensity counts versus diffraction angle data were collected and processed, and the x-ray diffraction (XRD) patterns were analyzed by automated search methods using the International Center for Diffraction Data databases.
Air and Topsoil Samples Analysis
Air samples were collected from 49 sites (Fig. 1c). The samples were obtained using a Buck Libra Plus LP-5 personal air sampler (A. P. Buck Inc., Orlando, FL, USA) and set to sample 2000 cc/min through 5-µm particulate polyvinyl chloride (PVC) filter cassettes; each air sample was collected over a period of 8 hours. Samples were obtained from varying elevations, and the cassette holder, at each sampling site, was consistently placed at 0.5 m above the ground. The collected filter cassettes from each sampling site were labeled and submitted to SGS Galson Laboratories (https://www.sgsgalson.com/) for XRD analysis to detect cristobalite, tridymite, and quartz within entrapped airborne pollutants.
Topsoil samples were collected from 6 selected sites corresponding to 6 of the air-sampled areas (Fig. 1c). A hand trowel was used to obtain approximately 300 g of soil from each site. Soil samples were placed in airtight bags, labeled, and submitted to Metallurgical Engineering Services. These samples were ground with an aluminum mortar and pestle and deposited onto an aluminum sample holder by XRD and analyzed similarly to the lung samples.
Statistical Analysis
Prevalence was calculated by species and sex. Ninety-five-percent confidence intervals (95% CI) were computed following a binomial distribution. Prevalence between the male and female animals in each species were compared using a Fisher exact test. The lesion grade was considered as an ordinal variable. Lesion grade distributions by species were described using relative frequencies, medians, and interquartile ranges. The grades were compared between species and age groups using the Kruskal-Wallis 1-way analysis of variance followed by a Wilcoxon rank sum test, if the previous test was significant. Wilcoxon rank sum tests were performed to compare the grades between sexes in each species. The threshold of significance was set at 0.05. All calculations and statistical tests were done in Microsoft Excel (Redmond, WA) and R software. 55
Results
Histopathology and Grading of SLM
On gross examination, none of the lungs sampled had visible lesions. Pathology reports from archival samples described no gross abnormalities associated with crystalline deposition. Histological assessment of the 259 lung samples identified 201 samples that contained free or phagocytized (within macrophages) heterogeneously pigmented, brown to black or clear crystalline material (Fig. 3a). The material was highlighted by AF staining (Fig. 3b) and was partially birefringent (Fig. 3c, d). The AF staining appeared to be due to the uptake of the new methylene blue counterstain rather than carbol fuchsin staining of the crystals. The crystals were polymorphic, exhibiting lamellar, acicular, radial, or fibrous morphologies, with postfixation and poststaining dimensions mostly less than 10 µm and ranging in size from less than 1 µm to more than 20 µm in length and up to 7 µm in width (Fig. 3e). These accumulations had a multifocal distribution, predominantly around larger airways (bronchi and bronchioles) or vessels and were partially engulfed by clusters of pulmonary interstitial macrophages. In chickens, the primary location of the SLM was around the secondary bronchi and interstitial tissues, in the interatrial septa (Fig. 4a, b) as well as around parabronchi (Fig. 4c), with formation of granulomas in some cases (large nodular aggregates of macrophages admixed with lymphocytes and plasma cells).
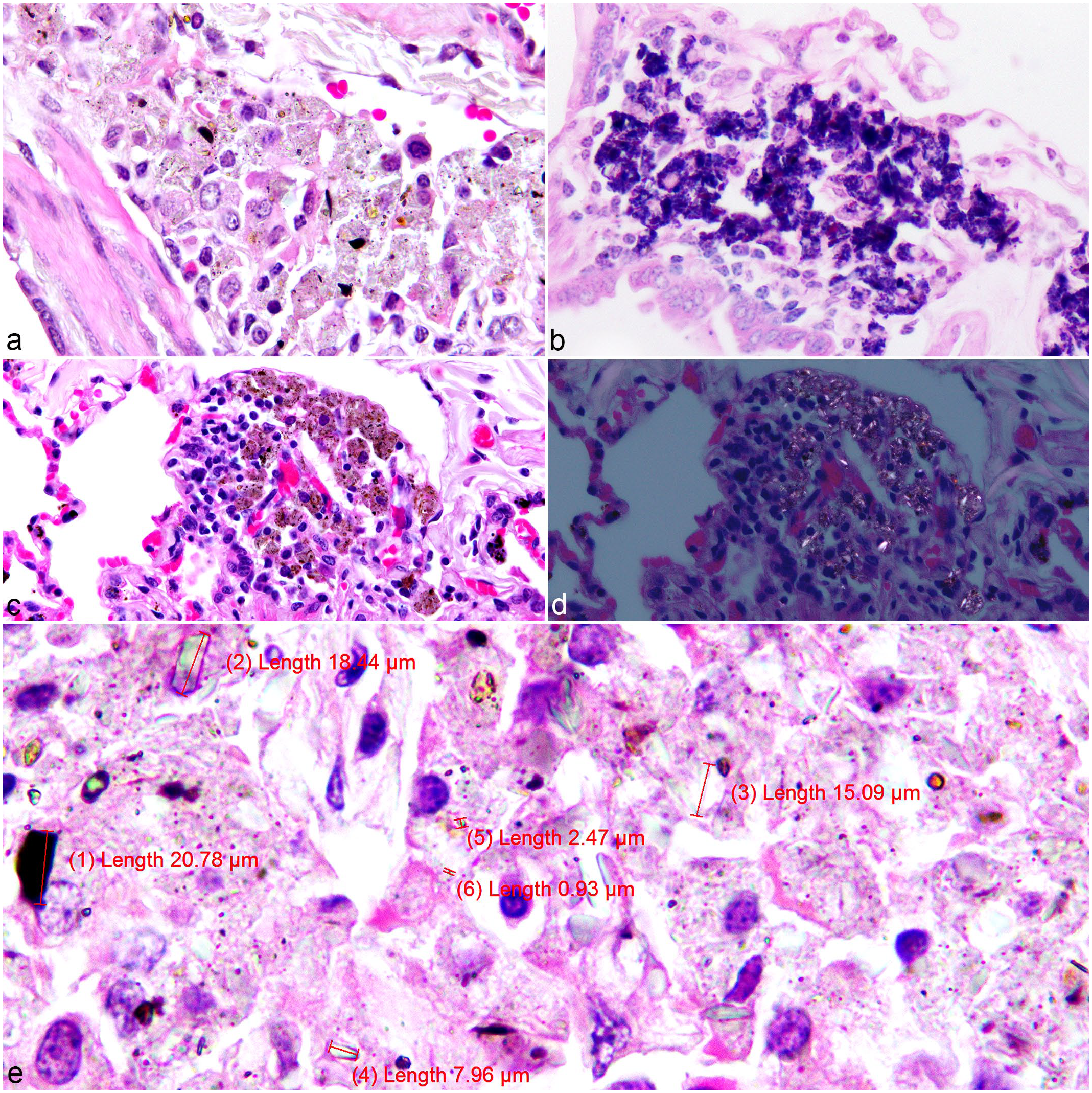
African green monkey, lung, pulmonary silicate accumulations. (a) Case 49. Heterogeneous silicate crystalline material (brown, black, or clear) located around a bronchiole and a blood vessel free or within macrophages. Hematoxylin and eosin (HE). (b) Case 8. Silicate-laden macrophages (SLMs). Acid-fast. (c) Case 43. SLMs, HE. (d) Case 43. SLMs, birefringence under polarized light. HE. (e) Case 49. Measurements of variably shaped silicate crystalline material free or within macrophages ranged in size from less than 1 µm to over 20 µm. HE.
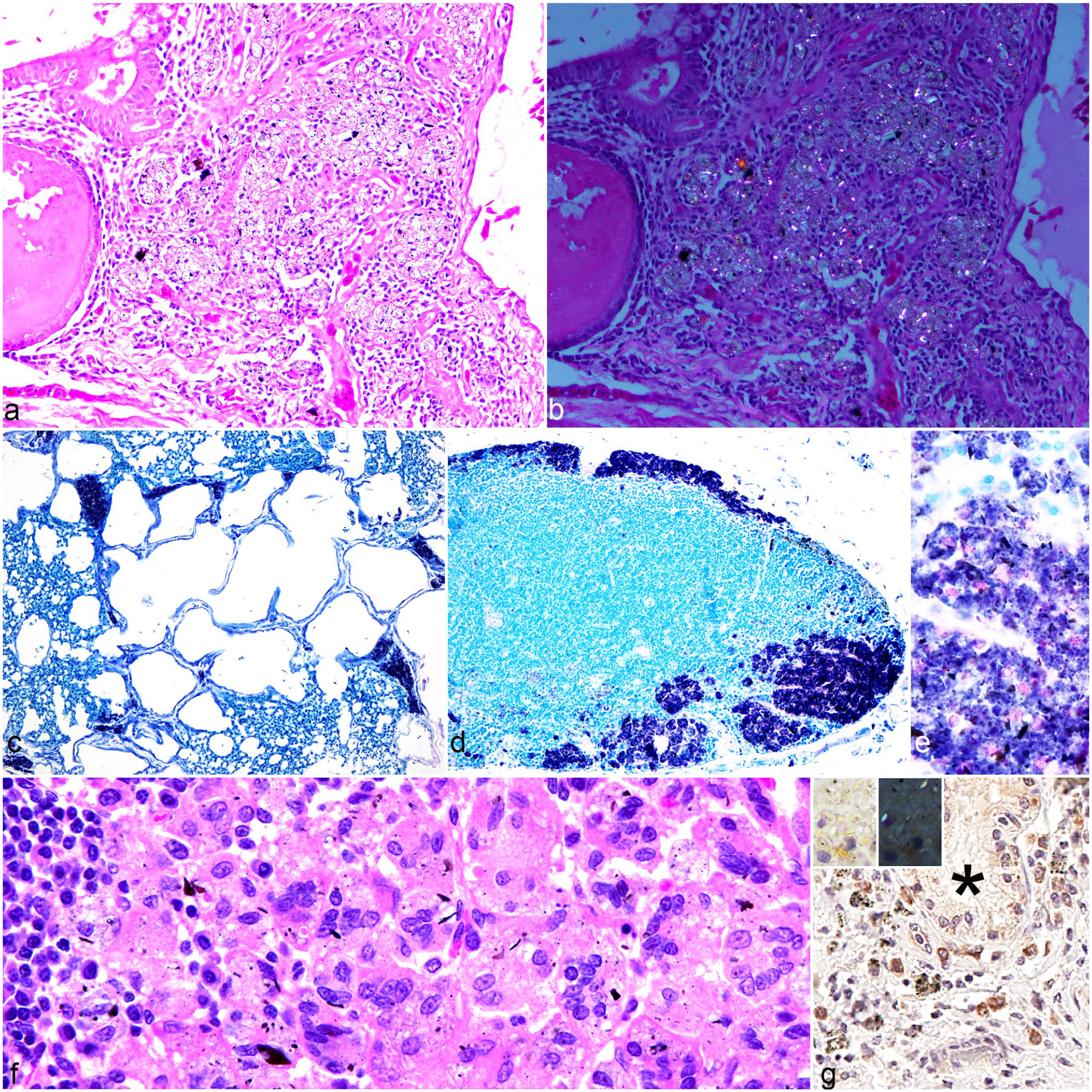
Spectrum of histology changes associated with crystalline silicates in chickens and mongooses. (a) Lung. Chicken 6. Silicate pneumoconiosis: granuloma in the interstitium around a secondary bronchus, composed of silicate-laden macrophages, lymphocytes, and plasma cells. Hematoxylin and eosin (HE). (b) Lung. Chicken 6 (polarized version of Figure 4a). Birefringent or partially birefringent crystalloid material, on polarized light, free or within macrophages, in a pulmonary granuloma around a secondary bronchus HE. (c) Lung. Chicken 18. Basophilic crystalline material accumulating in the area of the parabronchi. Acid-fast stain (AF). (d) Tracheobronchial lymph node. Mongoose 44. Accumulation of basophilic crystalline material in the cortex, free or intracellular. AF. (e) Tracheobronchial lymph node. Mongoose 44, detail from Fig. 4d. Silicate crystals range from pale to deeply basophilic with some associated brown-black crystalline material. AF. (f) Tracheobronchial lymph node. Mongoose 56. Granulomatous lymphadenitis with epithelioid and multinucleated macrophages containing crystalline particulate. HE. (g) Lung. Dog 166. Immunolabeling of macrophages with and without intracytoplasmic crystalline material around a bronchial gland (*). Inset: high magnification of IL1R1-positive macrophages with intracytoplasmic crystals (left), some birefringent on polarized light (right). Immunohistochemistry for IL1R1.
Across all species, the crystalline material was identified in the lungs of 201 of 259 animals (77.6% [95% CI: 72.0–82.5]). Table 1 shows the overall prevalence and grading of the lesion by species. Mongooses and monkeys had the highest prevalence of pulmonary crystalline particulate matter followed by cattle and chickens (more than half of the tested animals had pulmonary crystalline material in these species). Horses and small ruminants had lower but still relatively high prevalence (50.0 [6.8%–93.2%] and 40.0% [19.1%–63.9%], respectively) while the material was only seen in 10% (1.2%–31.7%) of the pig samples. In dogs, the prevalence of crystalline matter was 96.0% (79.6%–99.9%).
Pulmonary SLMs in domestic animals from St. Kitts: Prevalence and grading.
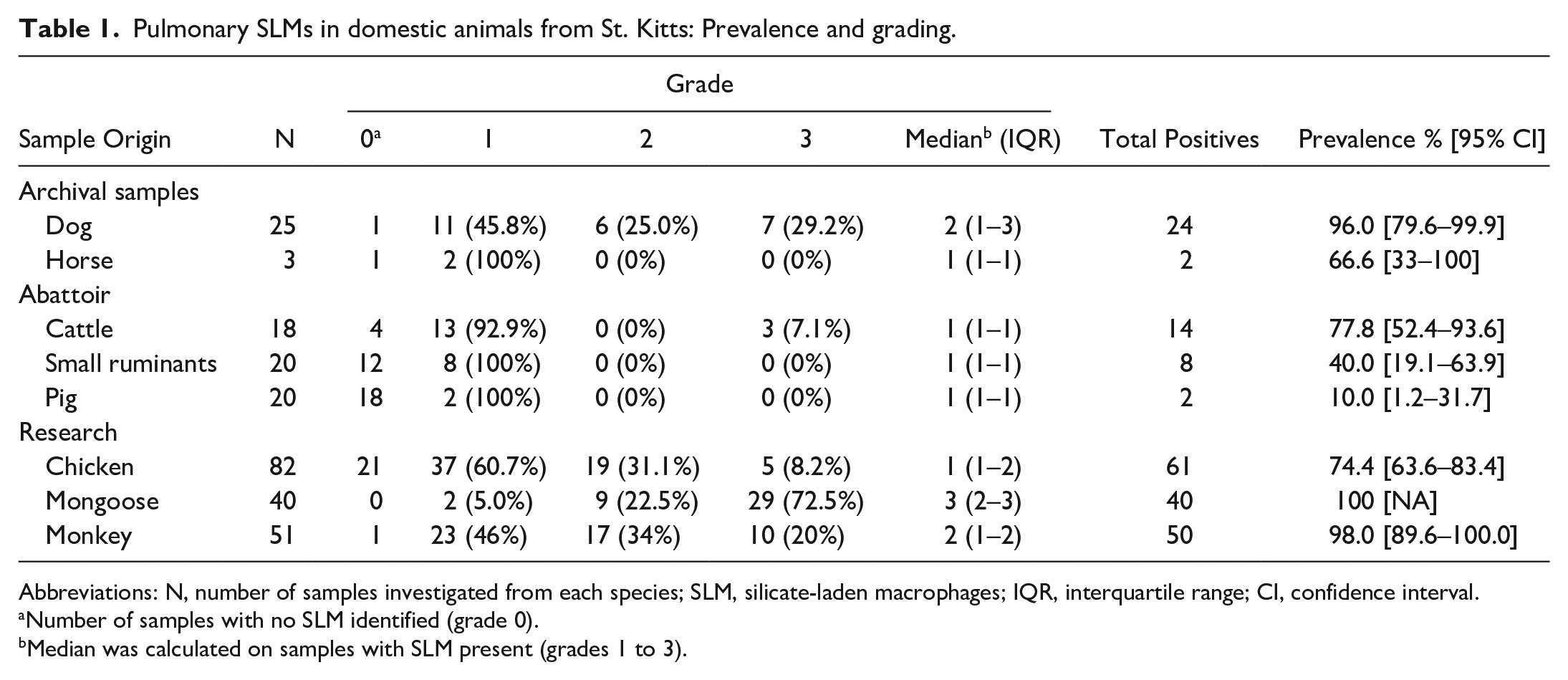
Abbreviations: N, number of samples investigated from each species; SLM, silicate-laden macrophages; IQR, interquartile range; CI, confidence interval.
Number of samples with no SLM identified (grade 0).
Median was calculated on samples with SLM present (grades 1 to 3).
Mongooses had the highest lesion grade average, followed by monkeys, dogs, and chickens. When lesions were present, lesion grades between chickens, mongooses, and monkeys were significantly different (Kruskal-Wallis P value < .001). More specifically, mongooses had the highest lesion grade with a median of 3. The grade was significantly higher than the lesion grade in chickens (median: 2, P value < .001) and in monkeys (median: 1, P value < .001). Mongooses’ ages were estimated based on skull size and tooth wearing 35 by the person performing the postmortem examination. 5 Animals were categorized as either young (comprising juvenile and young adults) or adults (comprising adults and senior adults). A similar trend in the grade distribution was found in both age groups, with grade 3 being more prevalent, but with no statistically significant difference between the 2 age groups (P = .9, Supplemental Table S1). Grades in chickens and monkeys were not significantly different (P = .07).
For chickens, mongooses, and monkeys, for which the sex of animals was reported and the sampling was “convenience” in type, we computed the prevalence by sex (Supplemental Table S2) and found no statistically significant differences between female and male animals in each species (P = .8, 1, and .4, respectively). Female chickens had a significantly higher lesion grade than male roosters (P = .005, Wilcoxon test). There was no association between sex and histologic grade in mongoose and monkey lungs (Table 2).
Pulmonary silicate-laden macrophages in domestic animals—description of lesion grade by sex in the 3 most affected species.
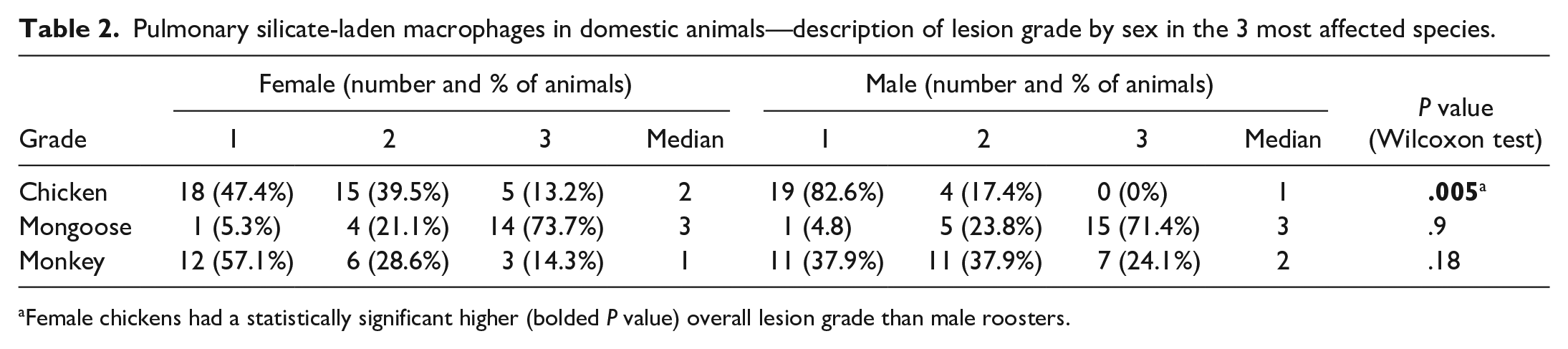
Female chickens had a statistically significant higher (bolded P value) overall lesion grade than male roosters.
Silicosis was only identified in 5 chickens that had granulomas, whereas other animals only had accumulations of SLM. In 4 chickens, the same crystalline material was also identified focally within the serosa of the duodenum and proventriculus, and in 1 of these chickens, it was incorporated within a granuloma located in the pancreatic mesentery. In 6 of the African green monkeys, it was also identified, focally, in the serosa of several abdominal organs as well as within the parenchyma of the spleen and liver of 1 sample. In 4 of the Indian mongooses, the architecture of the tracheobronchial lymph node was effaced to varying degrees (up to approximately 80% of the node) by SLMs (Fig. 4d, e), sometimes inducing multinucleated giant cell infiltration as a part of a granulomatous inflammatory response (Fig. 4f).
Immunohistochemistry and Histochemistry
Immunohistochemistry performed on 10 selected samples across multiple species indicated a strong signal for the presence of type M1 macrophages (IL1R1 cytoplasmic expression) in association with the accumulations of the crystalline material (Fig. 4g). Five of these samples had minimal focal or multifocal mild, early fibrotic changes (not evident on HE, only visible on Masson’s trichrome) in the form of small aggregates of collagen fibers around clusters of free and intracytoplasmic crystalline material. In dogs, fibrosis was present in 1 grade 2 and 1 grade 3 lesion; in monkeys, all 3 lesions with fibrosis were categorized as grade 3.
Electron Microscopy and EDXA
SEM was performed on selected areas of interest. After an area of interest was selected, it was magnified, and the crystalline material found in that area was measured (Fig. 5a–5f). Several measurements were achieved in lung samples from dogs (N = 39 aggregates), chickens (N = 24 aggregates), and monkeys (N = 24). The smaller crystalline aggregates were measured to be less than 10 µm in diameter.
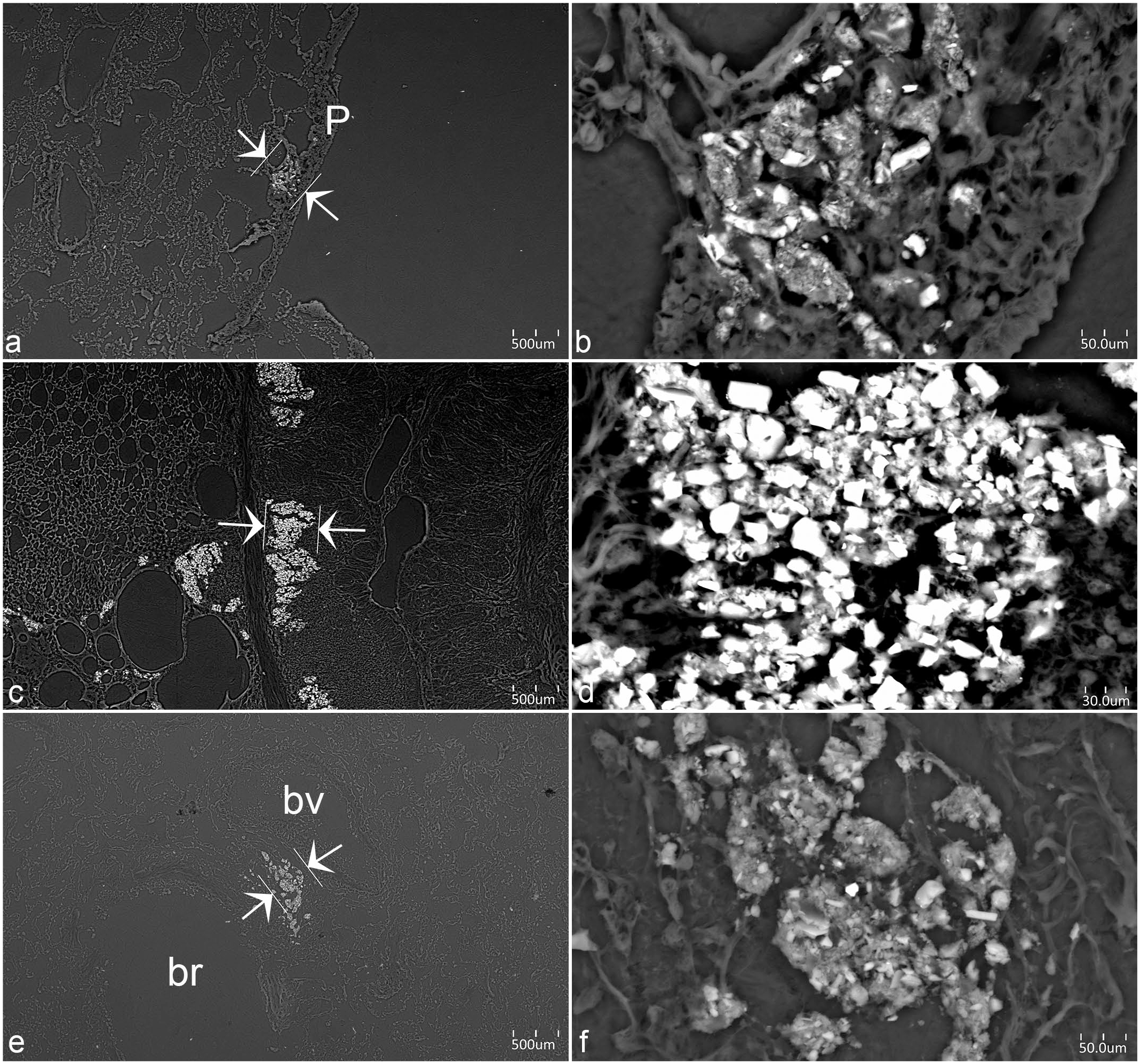
Scanning electron microscopy backscatter images of silicate-laden macrophages (SLMs) in animals from St. Kitts. (a) Lung. Monkey 29. Area of interest for energy dispersive x-ray analysis (EDXA), between white arrows, located subpleurally (P = pleura). (b) Lung. Monkey 29. Detail of area of interest for EDXA, highlighting the crystalline structure of the pulmonary silicates free or within macrophages. (c) Lung. Chicken 37. Multiple area containing crystalline material. Area of interest for EDXA, between white arrows. (d) Lung. Chicken 37. Detail of area of interest for EDXA, highlighting the dense crystalline structure of the pulmonary silicates free or within macrophages. (e) Lung. Dog 53. Area of interest for EDXA, between white arrows, perivascular and peribronchiolar. (bv = blood vessel; br = bronchiole). (f) Lung. Dog 53. Detail of area of interest for EDXA, highlighting the crystalline structure of the pulmonary silicates free or within macrophages.
Seven out of 8 blocks tested with the XRD analysis performed at the Joint Pathology Center contain particles of silicon, oxygen, aluminum, iron, potassium, sodium, and titanium which are consistent with environmental silicates and are typical components of anthracosilicotic dust. Samples taken from the chickens also contained magnesium, the significance of which, if any, is unclear. The main difference between these samples and human samples at the Joint Pathology Center is the relatively low proportion of anthracotic pigment, presumably a reflection of less exposure to combustion products (and cigarette smoke) that is typical for US urban-dwelling adults. The percentage of individual chemical components within the crystalline material and the average percentages by weight of the chemical constituents for each of the examined species are reported in Table 3.
Prevalence of chemical composition for the crystalline material in the lungs of animals determined through dispersive X-ray analysis.
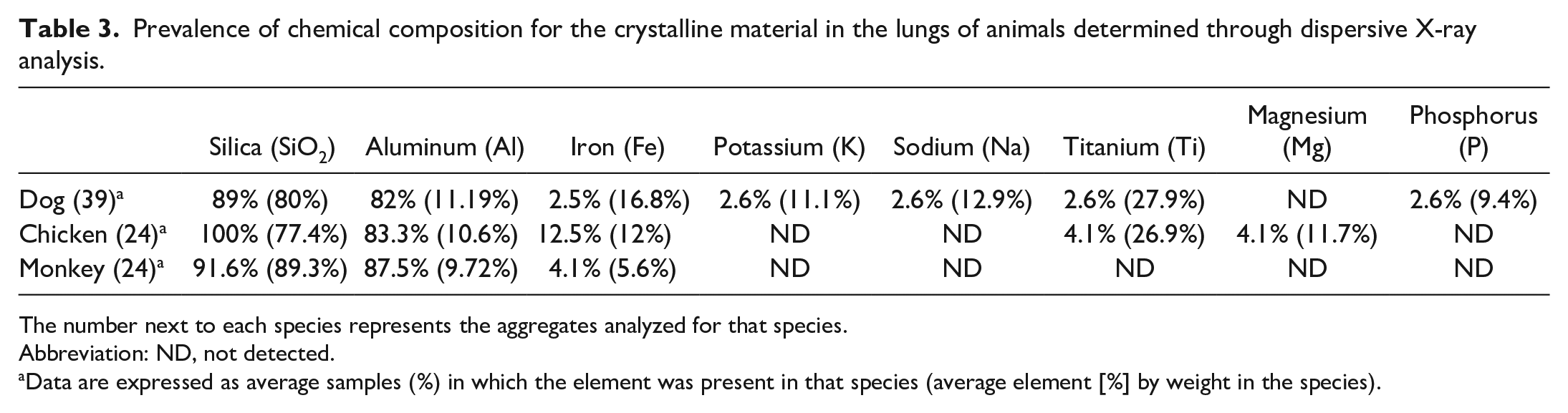
The number next to each species represents the aggregates analyzed for that species.
Abbreviation: ND, not detected.
Data are expressed as average samples (%) in which the element was present in that species (average element [%] by weight in the species).
XRD classified the crystalline structure of the material within the lungs by aggregating the dog (N = 2), African green monkey (N = 2), and mongoose (N = 2) samples. The analysis revealed (Table 4) structural arrangements corresponding to sodium phosphate (6/6), albite (4/6), hematite (4/6), and quartz (4/6).
X-ray diffraction analysis of selected lung samples.

XRD of the particulate material collected from the 5-µm air filters showed the following concentrations: quartz ranged <4.9 to <6.9 µg/m3, cristobalite ranged <4.9 to <6.9 µg/m3, and tridymite ranged <20 to <28 µg/m3. These concentrations are low and are considered below detectable levels by occupational safety and health administration (OSHA) standards. The XRD analysis of the topsoil samples (Table 5) identified structures corresponding to anorthite, albite, labradorite, anorthoclase (all silicate minerals), and rutile, all of which are common rock-forming feldspars. The soil from all collected sites contained these structures in various concentrations, with the exceptions of sample #2, which contained no anorthoclase; sample #3, which contained no anorthite; and sample #5, which contained no albite.
X-ray diffraction analysis of topsoil samples.
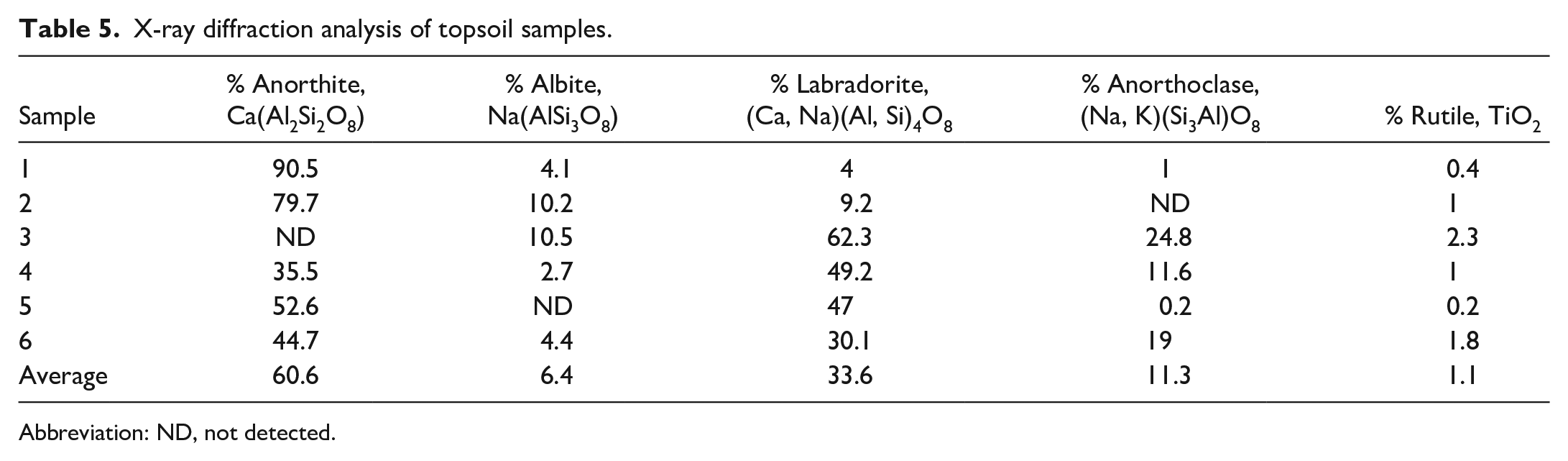
Abbreviation: ND, not detected.
Discussion
In this study, the prevalence of silicate accumulations within the lungs of multiple species on the island of St. Kitts was estimated and characterized histologically and ultrastructurally. Furthermore, the chemical composition and crystalline structure of the material were elucidated to determine its origin. The histologic appearance and location of the largely nonfibrotic pneumoconiosis lesions correspond to what have been reported in similar descriptive studies which describe similar findings in dogs,10,17 kiwis, 53 badgers,15,51 birds,7,21,50 camels,28,32 and a variety of zoo animals 8 with high incidence of pneumoconiosis and associated lesions. 7
The vast majority of the lesions found in our study are consistent with those of nonfibrotic pneumoconiosis resembling the description of macules in humans with pneumoconiosis. 39 A macule (seen mainly in the early stages of coal worker’s pneumoconiosis) is defined as accumulation of anthracosilicotic dust-laden macrophages around blood vessels, respiratory bronchioles, and along interlobular septas.13,18 The lower proportion of anthracotic pigment in our cases may be a reflection of less exposure to combustion products and cigarette smoke than is typical for American adults.
The grading system used in our study allows for the statistical prevalence analysis based on the distribution of crystalline deposits within the lung parenchyma and was adapted from a system for the assessment of silicotic/asbestos nodules within the lungs of occupationally exposed human patients.7,16 Pulmonary SLMs were previously reported in various species including dogs, horses, and otters to be associated with granulomatous inflammation, similar to silicosis in humans.3,17,54 Interestingly, SLMs have also been reported in the lungs of humans, birds, and other species in the absence of associated inflammation or with only mild inflammatory changes.4,7,51 Immunohistochemical investigation on a subset of the cases in this study demonstrated the presence of M1 macrophages associated with SLM accumulations. M1 macrophages are known to inhibit cell proliferation and cause tissue damage, whereas M2 macrophages promote cell proliferation and fibrosis. 46
There are few comprehensive studies documenting the accumulation of silicates in the lungs with no attendant inflammation. A recent study in badgers demonstrated a progressive accumulation of SLMs with no associated inflammation, hypothesizing that badgers, a burrowing species, develop an evolutionary tolerance to environmental dust inhalation. 51 In a study on 110 necropsies from varied mammalian and avian zoo species, 50% of the cases had accumulation of crystal-laden macrophages with no fibrosis or inflammatory cell infiltrates as the main pulmonary finding. 7 Similarly to these studies, the presence of SLMs in the animals of this study did not seem to elicit an inflammatory response in monkeys, mongooses, horses, small ruminants, cattle, pigs, or dogs; however, it did incite early fibrogenesis in a few dogs (1 grade 2 and 1 grade 3 SLM) and 3 African green monkeys (grade 3 SLM).
In the present study, the mongoose was the species with the highest total prevalence and greatest SLM accumulations. These findings could be explained by the behavior of mongooses, their small size, and diverse habitat which includes anthropized environments and dry areas. The small Indian mongoose extensively digs and burrows through the topsoil to create shallow dens, 33 potentially exposing the burrowing mongoose to high concentrations of aerated topsoil, leading to significant pulmonary deposition of inorganic dust. Interestingly, there was no statistical difference in the prevalence of graded lesions between adult and young mongooses, suggesting that the silicate concentration within the pulmonary parenchyma is not influenced by the length of exposure. The tracheobronchial lymph nodes of several mongooses in this study were effaced by crystalline-laden macrophages, presumably originating from lymphatic drainage of the affected lung. This finding is supported by a previous study in badgers with silicates in peripheral lymphatic sites. 51
The species with the next highest prevalence of SLM was the African green monkey (Chlorocebus sabaeus). The monkey lung samples were a combination of monkeys recently caught in the wild and those raised in captivity, which were kept outdoors and used as test subjects from a local research facility. The high prevalence of largely nonfibrotic pneumoconiosis (SLM accumulations) is particularly interesting due to the arboreal nature of this species; however, these monkeys are frequently seen roaming and foraging in close proximity to roadways, and their habitat includes anthropized environments.
Anthropized locations could also explain the high prevalence of crystalline deposits in the lungs of some of the dogs and free-roaming backyard chickens in our study. Roadways in St. Kitts are often unpaved and surrounded by varying amounts of broken and/or disrupted topsoil, which would explain the presence of albite, a topsoil component, in the sampled lung tissue.
Across all species, other than the mongoose, grade 1 SLM accumulation was the most common one. This finding further supports the speculation that individual species' behavior, different environments, and lifestyles contribute greatly to inhalation and, therefore, to the presence of topsoil silicates, within the lung parenchyma. Chickens (Gallus gallus domesticus) have been reported to preferentially “dust bathe” in sand which will aerate particulate material, facilitating inhalation. 52
The presence of nonfibrotic pneumoconiosis in the cattle is presumably due to grazing behavior which would lead to the nares being in close proximity to easily inhaled, loose, topsoil. 52 The lowest prevalence of pulmonary SLM was noted in pigs and small ruminants. This could be explained by the shorter life span of these species and, in the case of pigs, by husbandry practices, such as hard floor pens, which are cleaned frequently, inhibiting the buildup of aerated topsoil and preventing exposure to outdoor soil and rooting behavior. Samples from goats made up most of the small ruminant samples in this study, as this species is present in large numbers on St. Kitts. Goats are foragers and have a preference for ingesting leafy branch material, which makes them less likely to inhale disturbed topsoil when grazing. 21
The crystalline material measured by XRD were less than 10 µm in length, which places them into the class of “respirable” inhaled particulates according to environmental protection agency (EPA) standards, with those of length below 2.5 µm (fine inhalable particles) more easily reaching the lower airways. 1 Particles larger than 10 µm, compatible with silica dusts, were detected in the lungs of the animals in our study. This is explained by the aerodynamic diameter of particles, which can be greater than their particle size (PM). 27 Particles of size between 20 and 100 µm can become aerosolized but settle quickly.22,27,43 The distribution of the crystalline material, especially that of a larger particulate size, may explain the increased prevalence of pneumoconiosis versus SLM observed in the avian species as a result of the peculiarities of the anatomy of the avian respiratory tree. The avian trachea is proportionally 2.7 times longer than a comparably sized mammalian trachea, resulting in a greater deposition of larger particulate material outside the lungs. In addition, avian species lack resident pulmonary macrophages and a mucociliary apparatus, making them highly susceptible to the accumulation of inhaled particulates. 8 Moreover, the presence of air sacs and the specific respiratory cycle in birds could be responsible for crystalline material in the coelomic cavity, which sometimes induces a granulomatous inflammatory response.2,40
In our study, the presence of mild, early-stage silicosis (with fibrosis confirmed on Masson’s trichome stain) in 2 dogs and 3 monkeys was seen in cases that had a more severe accumulation of SLM (1 grade 2 and 4 grade 3 SLMs). These early fibrotic changes are suggestive of the involvement of SLM in the early pathogenesis of pulmonary silicosis. The etiology of pulmonary fibrotic changes in the presence of silica has been described as the direct effect of cytotoxicity leading to the release of lipase and proteases from damaged lung cells, 11 exceeding the antioxidant defense leading to lipid peroxidation and nitrosation and potential pulmonary fibrosis. 13 The development of pulmonary fibrosis secondary to the presence of silica has been speculated to also involve the activation of inflammatory mediators within alveolar macrophages, in concert with type I/II pneumocytes. This “partnered activation” recruits polymorphonuclear cells to the site of injury furthering the production of proinflammatory cytokines and, therefore, reactive oxygen species.11,13,25 Phagocytized silica particles can react with secondary lysosomal membranes, inducing permeability and allowing enzymatic leakage which leads to oxidative damage within neighboring tissue.2,40
Iron contamination is another characteristic that can increase silicate toxicity by heightening the production of hydroxyl radicals.12,48 Iron, as a component of silica, has been reported to contribute to the pathogenicity of silica causing disruption of the pulmonary alveolar macrophage lysosomal membrane leading to macrophage death, oxidative damage, and persistent parenchymal damage. 31 The ferric component of the silica in this study could explain the minimal inflammatory response and collagen deposition observed in trichrome-stained paraffin-embedded sections of the lungs of the most affected species.
Quartz, cristobalite, and tridymite are the crystalline polymorphs most frequently associated with occupational exposure in humans. We found quartz in lung and air samples, as well as cristobalite and tridymite in soil samples. All these polymorphs are known to be fibrogenic and biologically toxic. 13
Overall, the source of silicate crystalline material in tissue and environmental samples in our study was not fully elucidated but would appear to be a combination of Saharan dust (mainly), volcanic ash, topsoil, and local rock quarry, all contributing to siliceous dust inhalation. EDXA performed for this study determined that the chemical composition of the crystalline material was, by percent, largely silicon, oxygen, aluminum, and iron. This identified the crystalline material as silica based on the results of previous studies in which the chemical components of silica have been elucidated. 20 Albite, the material commonly identified within the selected lung and soil samples, is a ubiquitous silica salt and a common component of rock formation. One of the presumed origins of this feldspar in the topsoil, and possibly in the air of St. Kitts, is deposition from the transatlantic Saharan dust plume. The cloud contains predominately quartz (60% by volume) and variable amounts of layered silicates, which would include albite. Iron has also been reported to be a common mineral present in the plume, which has been previously discussed as contributor to the pathogenicity of silica. 24 It was previously shown that PM size in Saharan dust can be up to 20 µm, even though it is mostly referenced as course particles (usually PM2.5–10). 57 This fits the size range of the particles identified in the lungs of the animals in this study (up to around 20 µm).26,57 Pediatric medical records from the island of Trinidad have revealed increased acute asthmatic events and increased prevalence of allergic diseases throughout the Caribbean over a 30-year span, which have been attributed to exposure to the Saharan dust plume.30,44 Particle sizes between PM10 and PM2.5 were statistically associated with emergency visits by asthmatic children during the most active time of the year for the dust plume, compared to days without dust. 9
Volcanic activity was another presumed source of silicate contamination in the topsoil and air. The impact of volcanic eruption and its effects on air quality have been extensively examined where crystalline silica has been identified as particle fractions in dome-collapse ash. The size of these particles has been reported to be less than or equal to 2 µm, classifying them as “fine particulates” with the ability to affect the lower airways.36,37 The impact of volcanic ash on air quality and respiratory health is reflected in the increased occurrence of asthma and exercise-induced bronchoconstriction in children from Montserrat. 23 Size, composition, and concentration of the aerated ash were proven to be the most influential factors in human exposure to respirable particles (under 4 µm) from the Soufrière Hills volcano in Montserrat, which contains cristobalite, a crystalline silica polymorph that can cause silicosis. 38
The 3 species with the higher prevalence of SLM in our study (mongoose, chicken, and monkey) feed closest to the ground and are often near public roads. It was previously demonstrated that ash particles resuspended by road vehicles decrease in concentration exponentially as they move further from the ground, being 3 times lower at a height of 1.8 m (adult) than at 0.9 m (height of a young child). 38
Our sampling sites (for both animal and environmental samples) included a local rock quarry, and there was no significant difference between the concentrations of crystalline material at this site and other locations. In soil samples, we identified 4 silicate minerals in our study: anorthoclase ([Na, K]AlSi3O8), albite (NaAlSi3O8), labradorite ([Na, Ca]1-2Si3-2O8), and anorthite (CaAl2Si2O8), confirming the topsoil as a potential source for silicates.
There are some limitations to this study. Due to the convenient sample strategy, prevalence estimations cannot be extended to the whole island of St. Kitts, and there is a sampling bias in certain species (ie, dogs) as those individuals which had reported evidence of SLMs in previous reports were selected for the study. Additionally, age and sex data were not available for all animals, and the possible involvement of lymph nodes or other tissues was not investigated in all species.
The findings of the present study raise many questions concerning the clinical importance of what has traditionally been viewed as an incidental finding in many animal species. We hypothesize that SLM may represent an early stage in the pathogenesis of silicosis. Further investigations could include the use of 1 of the examined species with higher prevalence of SLM as a sentinel for exposure to topsoil particulate material, such as mongooses, monkeys, or chickens. 8 Confounders such as animal behavior or husbandry practices could alter the severity of presentation and complicate the evaluation of sentinel exposure.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221146095 – Supplemental material for Histology, prevalence, and environmental sources for pulmonary silicates depositions in domestic and wild animals
Supplemental material, sj-pdf-1-vet-10.1177_03009858221146095 for Histology, prevalence, and environmental sources for pulmonary silicates depositions in domestic and wild animals by Randall T. Walker, Oscar Illanes, Anne Conan, Bruce H. Williams, David Hilchie and Pompei Bolfa in Veterinary Pathology
Footnotes
Authors’ Note
Dr Felix Toka (RUSVM), Dr Adrian Nunez, MD, and Dr Ian Jacobs, MD (Pathologist and Pediatrician, JNK General Hospital, Basseterre, SK) for information pertaining to human health impacts; Steve Whittaker, PhD (Yale University) for information pertaining to environmental considerations methods of air sampling; Dr Anna Becker, PhD, DVM (Ross University School of Veterinary Medicine [RUSVM], St. Kitts) for all data pertaining to mongoose ages, sampling locations, and information pertaining to study construction; Randell Thompson for assistance with obtaining retrospective autopsy samples from RUSVM; Dr Paul Orchard, Caribbean imaging master; Dr Curtis Kline (Joint Pathology Center, Silver Springs, MD) for facilitation of performance of scanning electron microscopy images and energy-dispersive x-ray analysis; SGS Galson Labs (Syracuse, NY) for the performance of x-ray diffraction and subsequent results; Metallurgic Engineering Services, Inc. (Richardson, TX) for the performance of x-ray diffraction and energy-dispersive x-ray analysis and subsequent data; Jessica Leuscher (HSRL) for assistance with immunohistochemical analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by intramural grants from the Center for Integrative Mammalian Research, Center 2, Ross University School of Veterinary Medicine (RUSVM).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
