Abstract
We examined the morphologic characteristics of pulmonary macrophages in 42 specimens of Odontoceti (Globicephala macrorhynchus, Grampus griseus, Tursiops truncatus, Stenella attenuata, Stenella coeruleoalba, Berardius bairdii), using light and electron microscopes as well as immunohistochemistry with SRA-E5. SRA-E5-positive alveolar macrophages and pulmonary interstitial macrophages contained graphitic soots, indicating the clearance of airborne, aspirated foreign bodies. Pulmonary intravascular macrophages (PIMs), positive with SRA-E5, were present within pulmonary capillaries, attaching to applied endothelial cells by cell junctions. They showed cytoplasmic tubular structures of micropinocytosis vermiformis and erythrophagocytosis, indicating their contributory role in the clearance of blood-borne particles. The uptake of pathogens by PIMs may be associated with the inducement of acute lung injury, especially bacterial infectious pneumonia. This study revealed for the first time the presence of PIMs in cetaceans.
Pathogens flowing in the blood stream, such as bacteria and endotoxins, aged erythrocytes, and cellular debris, are rapidly removed by macrophages. According to the mononuclear phagocyte system (MPS) theory, macrophages are mononuclear cells derived from the bone marrow, are resident cell types in contact with the blood circulatory system, and play an important role in host defenses. 1,21 Interestingly, it has been reported that MPS-derived cells, called pulmonary intra-vascular macrophages (PIMs), exist within pulmonary capillaries in the order Artiodactyla (cattle, goats, sheep, deer, and swine), in the order Perissodactyla (horses and burros), and in cats. 2,7,21 To our knowledge, however, there have been no reports on PIMs in cetaceans. We examined the detailed morphologic characteristics of resident macrophages in the lungs of cetaceans. This article is the first description of PIMs in cetaceans.
We examined lung samples from 42 animals of Odontoceti caught for the purpose of fisheries by small-type whaling or hand-harpoon fisheries off the coast of Wakayama and Chiba prefectures in 2001–2003, Japan. Specimens comprised 16 Globicephala macrorhynchus, 12 Grampus griseus, six Tursiops truncatus, three Stenella attenuata, two Stenella coeruleoalba, and three Berardius bairdii (Table 1). These samples were provided for our study by the National Research Institute of Far Seas Fisheries, Japan. The age class (adult or immature) of animals was estimated on the basis of standard body length measurements. 8–12,14,15 One or two pieces (4 cm3) of lungs were fixed by immersion in 10% neutral buffered formalin and Zamboni's solution and then embedded in paraffin. For light microscopy, sections were stained with hematoxylin and eosin (HE) and Berlin blue stain. The formalin-fixed lung tissues of G. macrorhynchus and G. griseus were postfixed in 1% osmium tetroxide and embedded in epoxy resin. Ultrathin sections were stained with uranyl acetate and lead citrate and examined with an electron microscope (Hitachi, H7500, Hitachi, Inc., Tokyo) at 75 kV. For immunohistochemistry, a monoclonal antibody (SRA-E5; CD204) against human macrophage scavenger receptor type I protein antigen was used to detect cetacean pulmonary macrophages. 13,17–19 Sections were immunolabeled by the avidin–biotin complex (ABC) method (VECTASTAIN UNIVERSAL Elite ABC KIT, Vector Laboratories, Burlingame, CA). Positive reactions were observed to be brown with 3,3′-diaminobenzidine (DAB). Sections were counterstained lightly with hematoxylin. For double immunohistochemical labeling, lung tissue sections were labeled with a rabbit anti-mouse laminin antibody (LSL Inc., Tokyo) by the ABC method as described above, and the sections were then reacted with SRA-E5 overnight at 4 C. As the secondary antibody, Envision/AP (Dako, Carpinteria, CA) containing goat anti-mouse immunoglobulin G conjugated to an alkaline phosphatase–labeled dextran polymer was used. Positive reactions for anti–laminin antibody and SRA-E5 were observed to be brown with DAB and red with the fuchsin substrate chromogen system (Dako), respectively.
Information about examined animals.
SRA-E5 was a useful marker for detecting pulmonary macrophages in all species examined. Based on the anatomical location, pulmonary macrophages labeled with SRA-E5 were divided into three types: PIMs, alveolar macrophages, and pulmonary interstitial macrophages. PIMs were observed in the pulmonary capillaries of all animals (Fig. 1). Viewed by electron microscope, PIMs were roundish ellipsoids or irregularly shaped mononuclear cells, 10–20 µm in diameter (Fig. 2). In their cytoplasm, tubular invaginations by active pinocytosis, known as tubular micro-pinocytosis vermiformis structures, were seen as the most marked fine structures (Fig. 3). The cellular surface was coated by the electron-dense lamella, known as a fuzzy cell coat or glycocalyx, the structures of which were preserved in the tubular invaginations as central dense structures (Fig. 3). Phagosomes and lysosomes were commonly observed in the cytoplasm; some phagosomes contained degraded erythrocytes (Fig. 3), and lysosomes occasionally contained dense particles with the appearance of siderosomes. In fact, Berlin blue stain demonstrated iron-positive deposits regarded as hemosiderin. These PIMs were apposed closely with the endothelial cells, which were located not inside the air-blood barrier but rather inside the collagenous pulmonary septa. Cell junctions were often observed between PIMs and capillary endothelial cells (Fig. 4). The presence of these fine structures clearly distinguished PIMs from blood monocytes and granulocytes.
Lung; Globicephala macrorhynchus. Double immunohistochemistry with anti–laminin antibody (brown) and SRA-E5 (red). Pulmonary intravascular macrophages (PIMs) immunolabeled with SRA-E5 (arrows) are present inside the capillary basement membrane. ABC method and dextran polymer–alkaline phosphatase. Hematoxylin counterstain. Bar = 16 µm.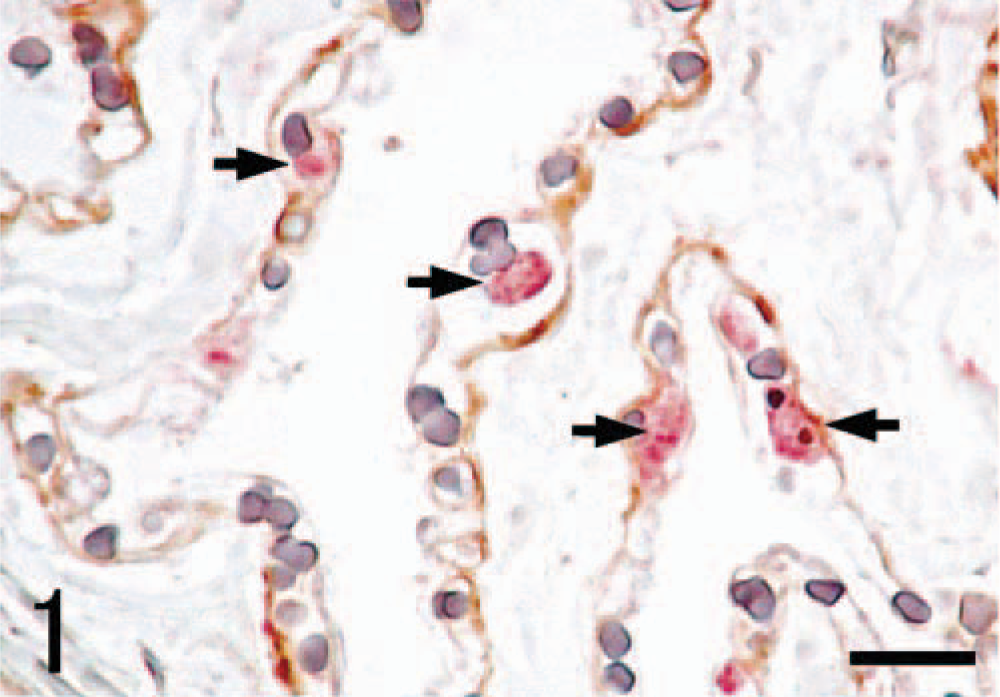
Lung; Grampus griseus. Electron micrograph. PIMs display a roundish ellipsoidal shape and are apposed closely with the endothelial cells. M, PIM; E, endothelial cell; P, type I pneumocyte; Al, alveolar lumen; Ca, pulmonary capillary; Ba, air-blood barrier; S, alveolar septum. Bar = 1 µm.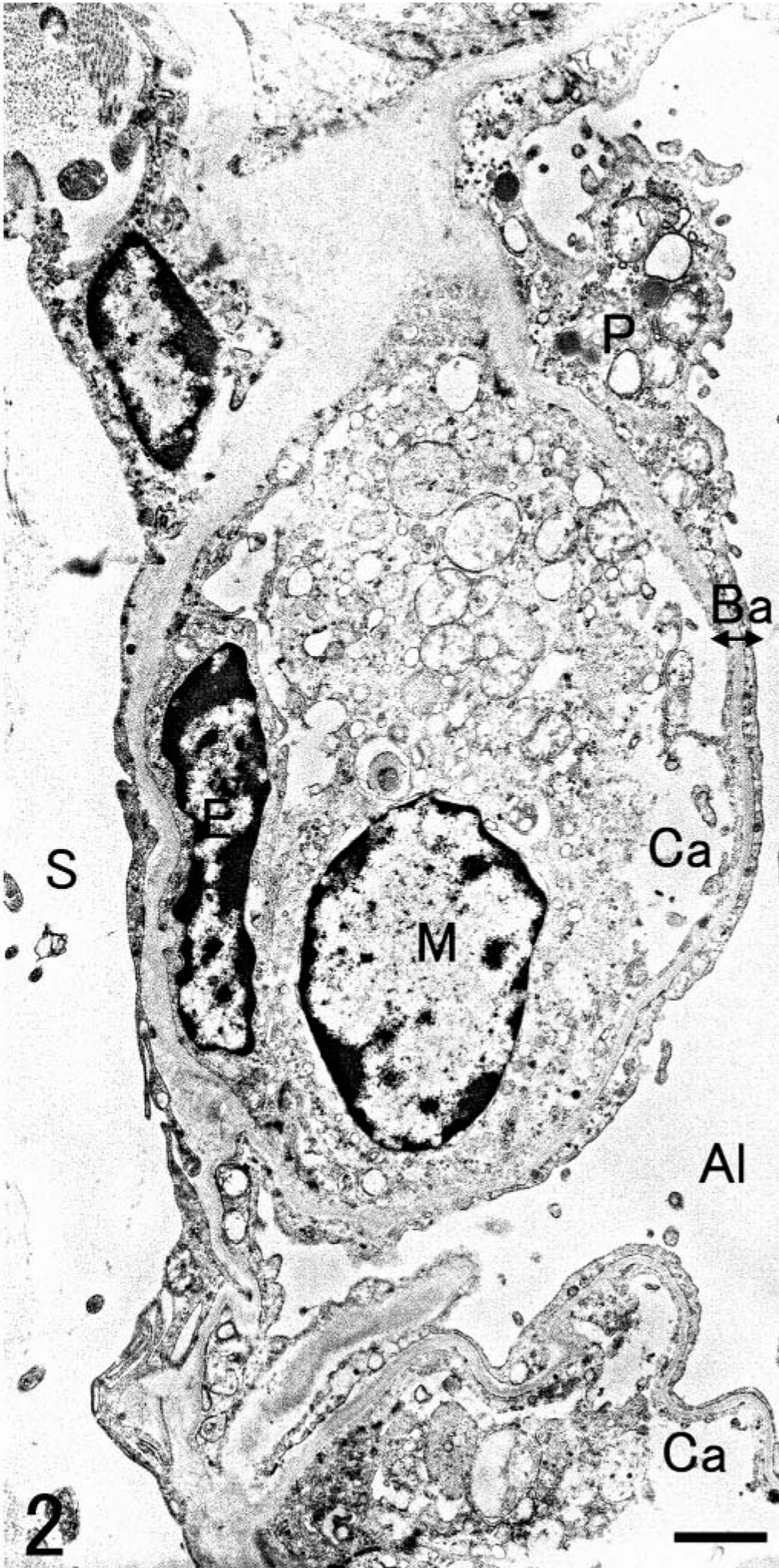
Lung; Globicephala macrorhynchus. Electron micrograph. Tubular structures of micropinocytosis vermiformis (arrows) and degraded erythrocytes (asterisks) are seen in the cytoplasm. The cellular surface is coated by electron-dense lamella known as a glycocalyx (arrowheads). R, erythrocyte. Bar = 0.64 µm.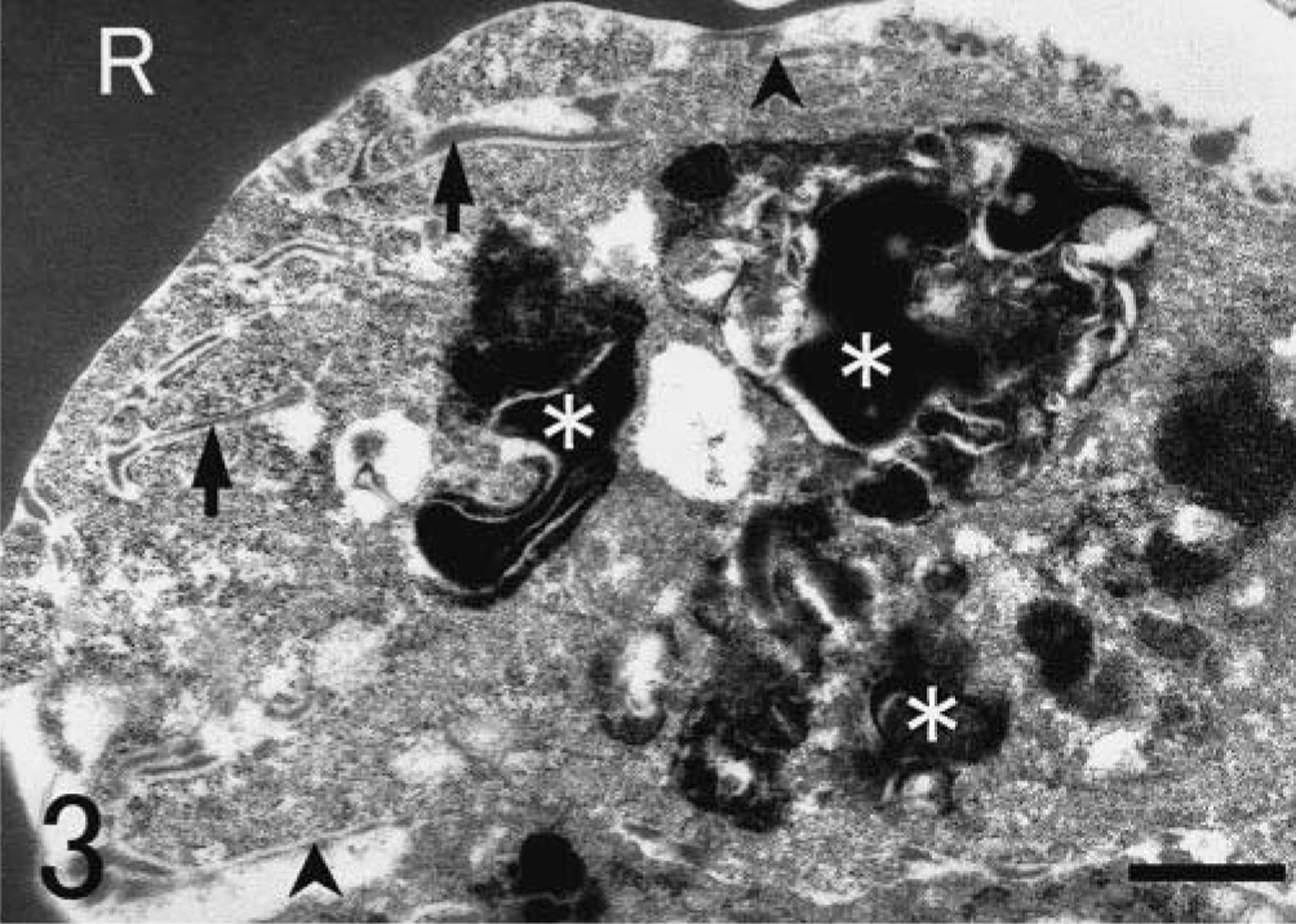
Lung; Grampus griseus. Electron micrograph. A higher magnification of cell junctions (arrows). M, PIM; E, endothelial cell; S, alveolar septum. Bar = 0.25 µm.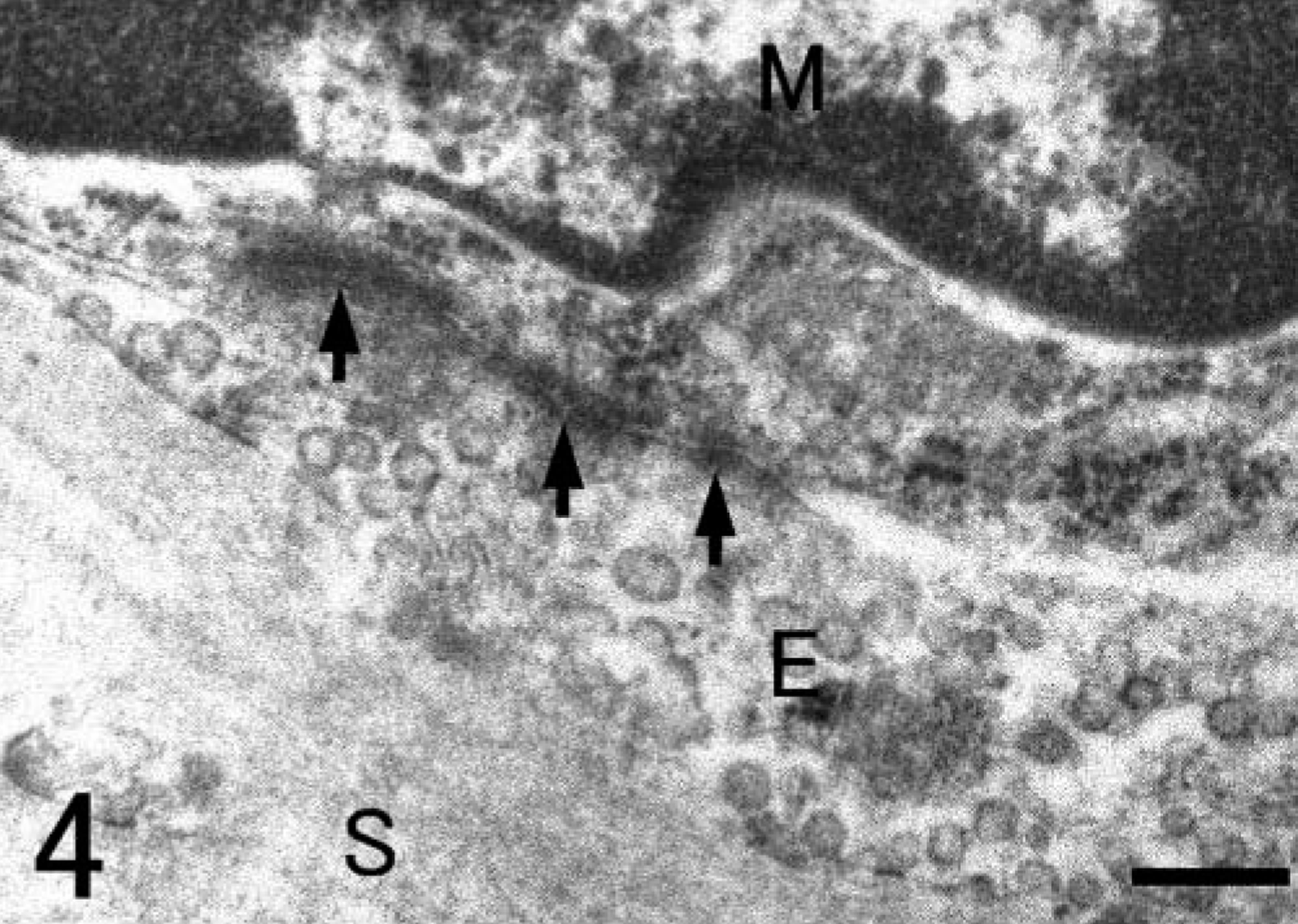
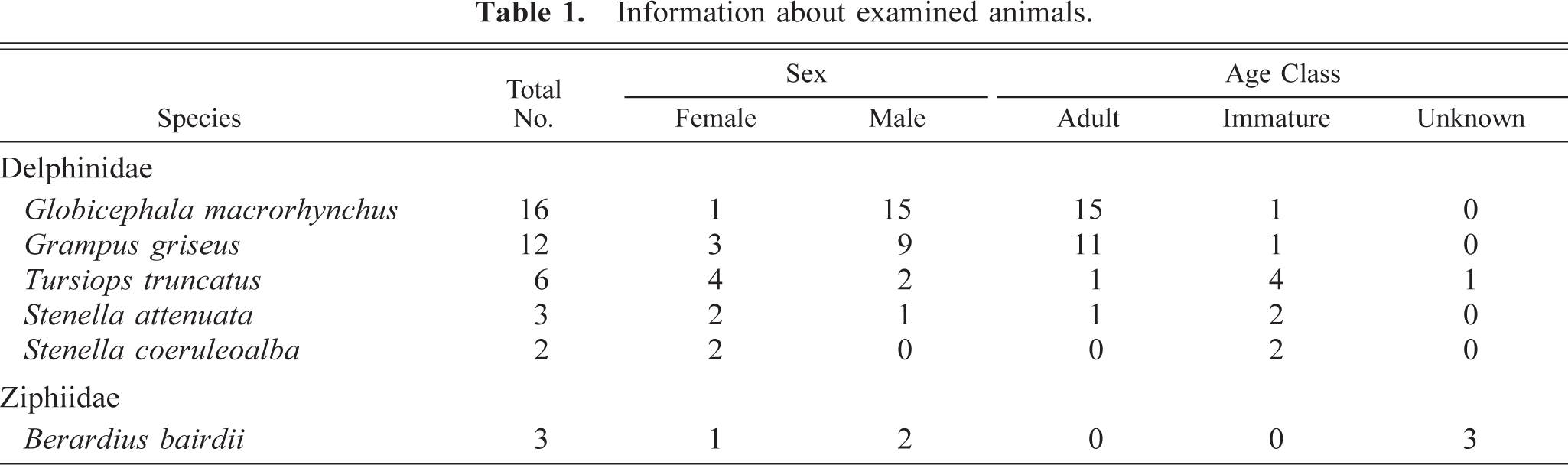
Alveolar macrophages labeled with SRA-E5 were seen in the alveolar lumina. Viewed by the electron microscope, alveolar macrophages possessed some pseudopods protruding from the cell membrane and numerous smaller coated pits and vesicles in the peripheral area of the cytoplasm as well as many larger phagosomes and lysosomes in the central area of the cytoplasm (Fig. 5). In alveolar macrophages, there were no fine structures characteristic of PIMs. In the interstitium under the epithelial layer of bronchioli, spindle-shaped pulmonary interstitial macrophages were diffusely distributed (Fig. 6). Some macrophages in the interstitium had swollen cytoplasm with black particles, apparently graphitic soots or white-yellow pigments (ceroid or lipofuscin) (or both); such cells were seen mainly around the small lymph vessels and sometimes with infiltrated lymphocytes and plasmacytes (Fig. 7).
Lung; Globicephala macrorhynchus. Electron micrograph. An alveolar macrophage in the alveolar lumen possessed some pseudopods and coated pits and vesicles in the peripheral area of the cytoplasm as well as phagosomes and lysosomes in the central area of cytoplasm. Bar = 3.5 µm.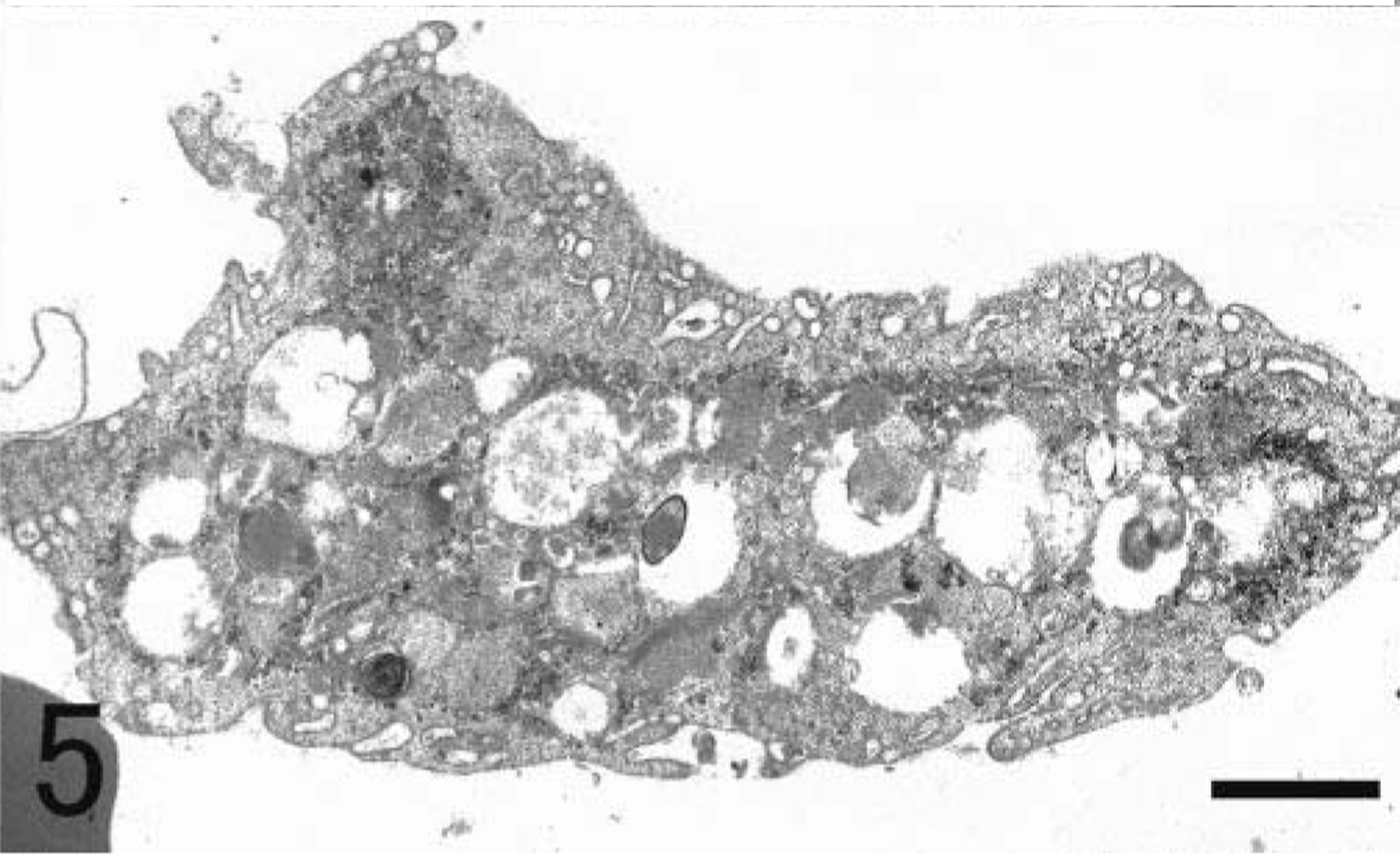
Lung; Stenella attenuata. Immunohistochemistry with SRA-E5 for the bronchiolus. Stellate- or spindle-shaped macrophages reacting to the antibody are diffusely seen in the interstitium (arrows). ABC method. Hematoxylin counterstain. Bar = 25 µm.
Lung; Grampus griseus. Macrophages including black particles and white-yellow pigments in the cytoplasm (arrows) are seen in the interstitium. A lymph vessel (V) is surrounded by these macrophages as well as by lymphocytes and plasmacytes. V, lymph vessel. HE stain. Bar = 16 µm.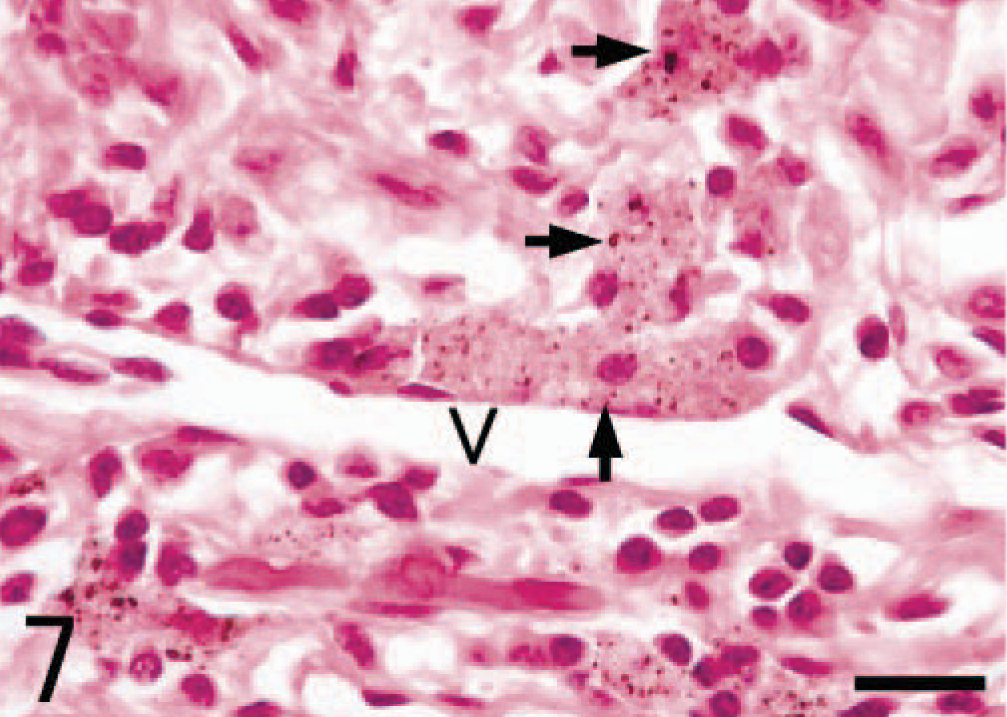
The distribution and morphologic features observed in cetacean PIMs were in agreement with those reported in terrestrial animals routinely possessing PIMs. 20,21 It has been reported that PIM-like macrophages without intercellular cell junctions appeared in rats with experimentally induced biliary cirrhosis and human patients with hepatic dysfunction. 3 PIMs have not routinely been detected in normal primates and rodents. Such cells might have compensated for functions of injured Kupffer cells. In all cetaceans examined here, there were neither significant hepatic diseases nor severe pneumonia. In addition, no pathologic changes were seen in the spleen, kidneys, or lymph nodes (hilar nodes, marginal nodes of the lungs and mesenteric nodes). On the basis of anatomical location and morphologic characteristics, it was thus concluded that cetaceans routinely have PIMs under physiologic situations.
Macrophages in contact with circulating blood play an important role in the clearance of blood-borne particles. Experiments with tracer particles have demonstrated that in domestic animals with PIMs, important components of MPS were located in the lungs as well as in the liver. 1,5,20 More interestingly, tracer particles injected into peripheral veins were rapidly removed by PIMs rather than Kupffer cells. 1,20 Active endocytosis observed in cetacean PIMs indicated that PIMs also play an important role in the direct uptake of aged erythrocytes and circulating particulate materials as a host defense function.
Although some of their effects are beneficial, the presence of PIMs produces contrary effects in hosts. Animals having PIMs show higher sensitivity of the lungs to gram-negative bacterium-derived endotoxin administrated intravenously because activated PIMs through englobement of endotoxin could induce acute lung injury through release of inflammatory mediators. 4,5,7,21 Pulmonary disorders caused by secondary or primary bacterial infection have been reported as a relatively common disease in cetaceans, especially in small-sized cetaceans; causative fatal gram-negative bacteria, which were isolated from septicemia in marine mammals, contained Vibrio spp., Edwardsiella spp., Aeromonas spp., Pseudomonas spp., Pasteurella spp., Klebsiella spp., and Salmonella spp. 6,16 Therefore, PIMs might play a significant role in the inducement and progression of fatal infectious lung lesions in cetaceans.
Alveolar macrophages in cetaceans possessed morphologic features of mature macrophages, exhibiting well-developed endocytosis. Interstitial macrophages, which possessed swollen cytoplasm including graphitic soots, were regarded as derivatives of alveolar macrophages that had been transferred into the interstitium from alveolar lumina. As known to be the case in terrestrial mammals, alveolar macrophages of cetaceans function in the clearance of air-borne, aspirated foreign bodies, which then move into neighboring lymph nodes through lymphoid vessels.
In conclusion, besides alveolar macrophages and pulmonary interstitial macrophages, this study revealed for the first time the presence and morphologic characteristics of PIMs in cetaceans.
Footnotes
Acknowledgements
We are grateful to Japan Small-Type Whaling Association; T. Kishiro in the Cetacean Population Biology Section, National Research Institute of Far Seas Fisheries; T. Hara and H. Sato, cetacean researchers, for providing lung samples; and M. Nakanishi for laboratory equipment and technical advice. This work was supported in part by Grants-in-Aid for Scientific Research B (No. 15380217 to J. Yamate), and for JSPS Research Fellow (No. 16 · 1388 to M. Kawashima), the Ministry of Education, Sciences, Sports and Culture, Japan.
