Abstract
Ischemic myelomalacia secondary to fibrocartilaginous emboli (FCE) is an idiopathic disease in humans and animals. On the other hand, congenital spinal cord malformations result from neural tube defects in fetal development (ie, spinal dysraphism), with structural anomalies referred to collectively as myelodysplasia. Spinal dysraphisms are frequently accompanied by skin and vertebral abnormalities because of the embryogenic relationship. In this observational case study, we report the pathologic findings of 13, 18- to 24-weeks-old pigs from a large conventional operation that presented with acute paraparesis. Ischemic myelomalacia secondary to FCE was observed in 5 of 13 examined pigs. Congenital spinal cord malformations located between the caudal thoracic and sacral spinal cord were identified in 7 pigs, with structural abnormalities that ranged from diplomyelia/split cord malformation to segmental spinal dysgenesis (myelodysplasia) to caudal agenesis. Concurrent myelomalacia and congenital spinal cord malformations in the same or different sites were noted in 2 pigs. No spinal lesion was observed in 3 pigs. Although gross vertebral abnormalities were not observed herein, intervertebral instability due to minor defects in the articular facets, as well as other unidentified factors, is suspected to contribute high incidence of FCE. It is likely that these congenital malformations were previously underdiagnosed or are possibly new conditions associated with continuous inbreeding and genetic improvement in the modern swine industry.
Accompanying the continuous acceleration of growth performance in the modern swine industry, hindlimb lameness has become one of the main clinical problems seen in finishing pigs. In addition to disorders of the musculoskeletal system, diseases that cause insults to the spinal cord, such as ischemic spinal cord injury (myelomalacia) secondary to fibrocartilaginous embolism (FCE), are a possible cause of hindlimb weakness. 19 Although ischemic myelomalacia is infrequently diagnosed in pigs and other species, a high incidence of this condition has been reported in a large group of conventional finishing pigs with rear limb paresis or paralysis. 3 Although the pathogenesis of ischemic myelomalacia involves some idiopathic components, the formation of FCE in that report was presumptively attributed to a combination of genetic, performance, behavioral, and nutritional issues acting in conjunction with chronic diskospondylitis as the likely predisposing factor.3,12
Spinal dysraphism is an umbrella term that describes several congenital malformations that affect the spine, spinal cord, or nerve roots. It is a potential cause of hindlimb weakness, although reports in veterinary medicine are rare, and not associated with spinal cord ischemic changes. 4 In human medicine, most cases of spinal dysraphism are diagnosed at birth or in early infancy, 18 while others are diagnosed in older children or even adults.16,17 Although these conditions are well-described, there is considerable confusion as to the classification and terminology applied to these abnormalities.16,29 An updated classification of spinal dysraphism has been proposed based on clinical and neuroradiological features.2,7,23,29 The open type of spinal dysraphism includes conditions such as craniorachischisis, myelomeningocele, and myelocele, in which neuroglial tissue and/or meninges are exposed to the environment through a congenital bony defect. Closed spinal dysraphism differs in that these lesions are not exposed to the environment due to normal development of overlying bone and/or skin and can be further subclassified based on the presence of a subcutaneous mass. Within this group is a rare form of spinal dysraphism, resulting in the caudal divergence of a single spinal cord into 2 or more hemicords, which includes both diplomyelia (spinal cord duplication) and diastematomyelia (spinal cord splitting). These conditions have been more recently designated as split cord malformations (SCMs), which are further classified as types I and II based on imaging hallmarks.16,17 Type I malformations are characterized by the presence of a rigid bony or cartilaginous septum that gives rise to 2 dural tubes containing completely separated hemicords, whereas type II malformations lack a rigid septum and the 2 spinal cords are contained within a single dural tube.25,29 Along with SCMs, segmental spinal dysgenesis (SSD) and caudal agenesis (CA)/caudal regression syndrome (CRS) are included in the category of closed spinal dysraphism without a subcutaneous mass. 29 SSD and CA/CRS differ embryologically by their segmental location along the longitudinal axis of the embryo. In SSD, the intermediate segment of the notochord is primarily involved as opposed to the caudal segment in CA/CRS.23,29
Within domestic animals, spinal dysraphism is most frequently described in calves.4,22 In 1 case study, myelodysplasia was described as involving up to 30% of calves in a herd over a period of at least 15 years. 24 Otherwise, it is generally presented as individual cases. Beginning in early 2021, a series of 18- to 24-week-old conventional pigs with acute onset of hindlimb lameness was submitted to our laboratory. In addition to ischemic myelomalacia secondary to FCE, anomalies with different morphologies, resembling subtypes of closed spinal dysraphism in human medicine, were observed through the continuous case collection. In this study, we present a detailed description of the spinal cord lesions, including ischemic myelomalacia and different spinal dysraphic conditions, that were found in these pigs, and discuss their diagnosis, clinical relevance, potential embryogenesis, etiology, and possible relationship to ischemic myelopathy.
Materials and Methods
Cases and Samples
Starting in February 2021, a conventional swine production unit housing 2400 grow-finish pigs reported acute onset of hindlimb lameness in approximately 1.5% to 2.0% of the pigs, which were typically 22- to 24-weeks-old. The pigs were derived from a heavily muscled, high-lean commercial genetic line crossbred with another commercial genetic line with enhanced reproductive performance. Considering the poor prognosis of pigs with paraparesis, affected animals were euthanized by stunning on the farm. Thirteen pigs were selected by the farmers between February and August 2021 and submitted to South Dakota State University Animal Disease Research & Diagnostic Laboratory for complete necropsy. External examination of the pigs showed no defects on the skin, tail, and limbs. Except for pulmonary congestion and distended urinary bladders, no remarkable gross lesions were observed in brains, visceral organs, and hindlimb joints. The spine between the middle thorax and sacrum was sectioned longitudinally in a parasagittal plane with a bandsaw. The spinal cord, brain, intervertebral disk, and other visceral organs were sampled and submitted for aerobic bacteria culture and virus isolation with American Association of Veterinary Laboratory Diagnosticians (AAVLD) validated procedures in our laboratory. Another set of the collected tissues was fixed in 10% neutral buffered formalin. A detailed gross examination of the spinal cord was performed after fixation.
Histopathology
Formalin-fixed tissues were embedded in paraffin. Four-micrometer-thick sections were stained with hematoxylin and eosin (HE) for histologic examination. Sections were stained by periodic acid–Schiff (PAS)-light green for cases with evidence of ischemic myelomalacia to identify potential cartilaginous emboli.
Results
Case 1 to 3: Ischemic Myelomalacia Secondary to Fibrocartilaginous Embolism, No Spinal Dysraphism
In cases 1 to 3, 1- to 3-cm-long segments of spinal cord parenchyma, randomly distributed between the mid thoracic and sacral vertebrae were soften and shrunken, and were sometimes associated with well-demarcated subdural hemorrhages (Fig. 1a). After formalin fixation, associated spinal cord parenchyma was dark red on cross-sections. The affected regions were well-demarcated, but there was loss of distinction between the gray and white matter.
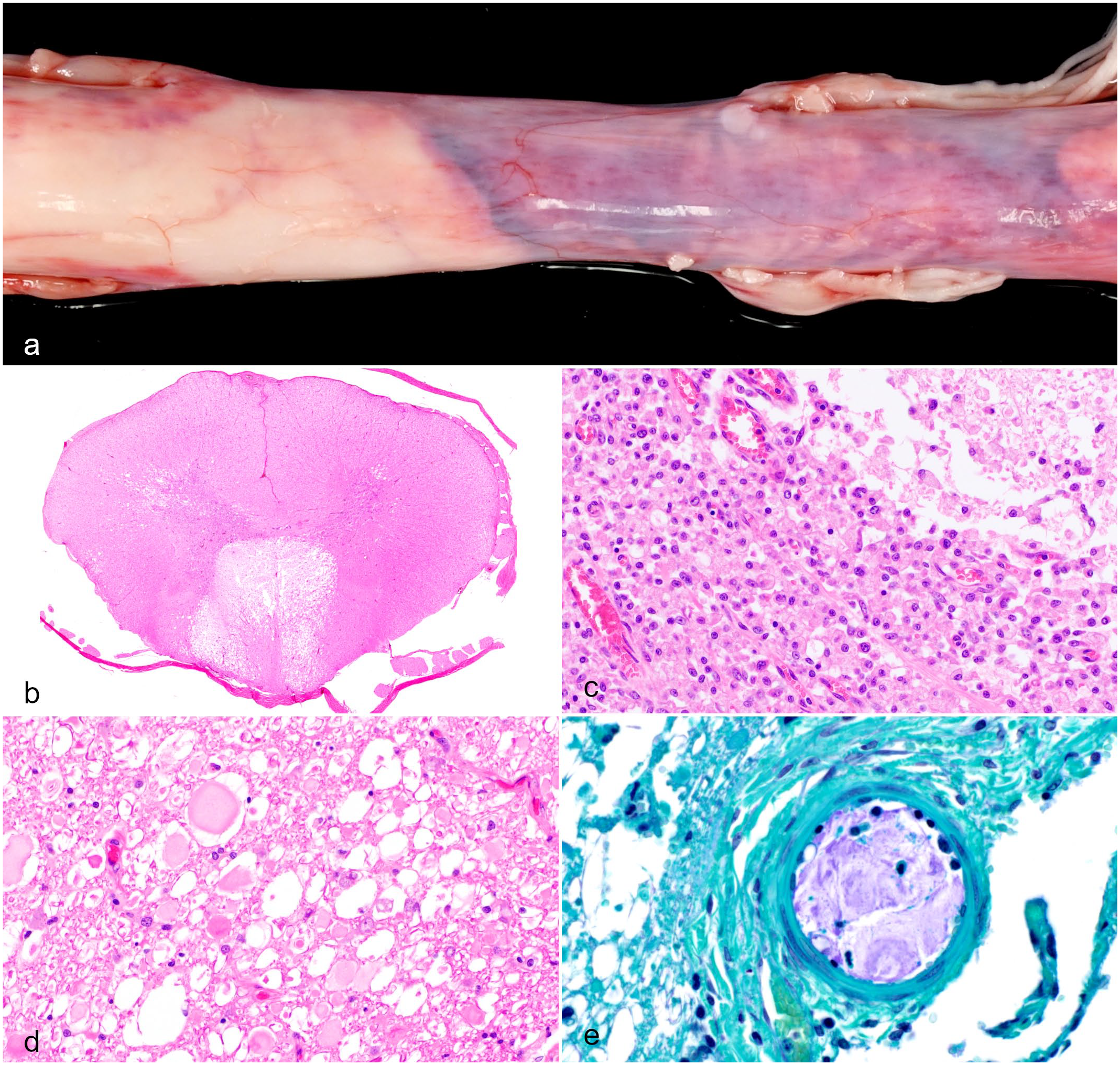
Ischemic myelomalacia, spinal cord, pig. (a) Sharp demarcation of dark red discoloration (at right) in the thoracolumbar segment of the spinal cord. Case 1. (b) Myelomalacia effaced the majority of white matter in the ventral funiculus and extended into the adjacent ventral horns of the gray matter. Case 2. Lumbar spinal cord. Hematoxylin and eosin (HE). (c) The affected gray matter was infiltrated by sheets of gitter cells and exhibited neuropil rarefaction (upper right). Case 2. HE. (d). White matter showed Wallerian degeneration, characterized by distended myelin sheaths with swollen axons, fragments of necrotic debris, axon dropout, and axonophagia. Case 2. HE. (e). A leptomeningeal artery was lined by hypertrophic endothelial cells with luminal occlusion by granular to amorphous cartilaginous material (blue staining with periodic acid Schiff-light green stain). Case 1.
Histologic examination revealed both uni- and bilateral, asymmetric, but well-demarcated areas of myelomalacia (Fig. 1b). The white matter and adjacent gray matter were effaced by sheets of macrophages with a few lymphocytes (Fig. 1c). In the adjacent gray matter, there was degeneration, necrosis, and loss of neurons and glial cells with rarefaction of the neuropil. In adjacent white matter, there was extensive Wallerian degeneration, characterized by distended myelin sheaths with swollen axons, necrotic debris, axon dropout, and infiltration of macrophages (Fig. 1d). The central canal was slightly distended by blood or segmentally ruptured. In some sections, the small arteries and arterioles in the spinal cord and leptomeninges were lined by hypertrophic endothelial cells and were cuffed by 1 to 3 layers of lymphocytes. Variable numbers of spinal and leptomeningeal arterioles were partly to completely occluded by fibrocartilaginous emboli (Fig. 1e). Collectively, spinal cord changes were interpreted as an ischemic sequel to vascular FCE.
The sagittal section of vertebrae exhibited open physes with additional physeal plates. In the intervertebral disk, the nucleus pulposus was composed of numerous notochordal cells with foamy to vacuolated cytoplasm or clear, nonstaining cytoplasm. The matrix was basophilic and granular. No obvious inflammation was observed in examined sections. No significant gross or histological lesions were observed in the associated vertebrae or intervertebral disks, and spinal cord architecture was normal with the exception of the ischemic changes described above.
Case 4: Diplomyelia (Type II SCM) With Myelodysplasia (Segmental Spinal Dysgenesis)
In case 4, the spinal cord had a 1.5-cm-long, intradural-extramedullary crescentic mass at the level of the caudal thoracic vertebrae that arose from the spinal cord with no obvious compression (Fig. 2a). Histologically, the mass was composed of disorganized white and gray matter with a central canal, which was interpreted as a duplicated spinal cord with disoriented tissue architecture (Fig. 2b). Both the main and duplicated spinal cords were located within the same dural tube (Fig. 2b). Right caudal to the mass, the main spinal cord was segmentally distended. Histologically, the area of distension was characterized by disorientated white and gray matter (Fig. 2c, d).
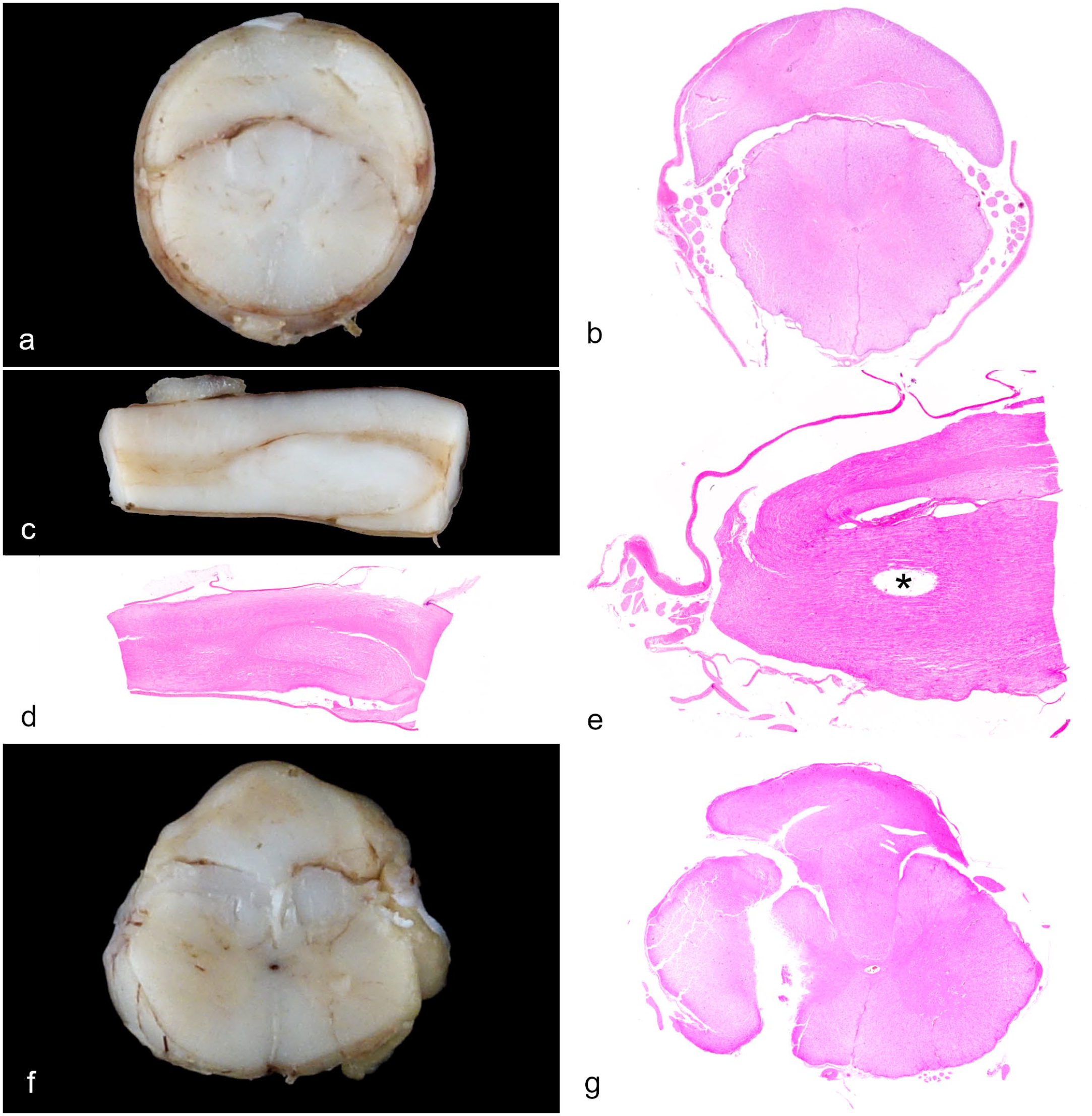
Closed spinal dysraphisms. (a) Diplomyelia/type II split cord malformation (type II SCM), caudal thoracic spinal cord. A crescent mass is attached to the dorsal aspect of the spinal cord. Case 4. (b) Histology of Fig. 2a. A duplicated spinal cord with disorganized architecture (upper) was attached to the dorsal aspect of the spinal cord (lower). Both were enclosed by the same dura mater. Case 4. Hematoxylin and eosin (HE). (c) Myelodysplasia, thoracolumbar spinal cord. Caudal to the duplication, the white matter was segmentally distended with infolded axons that distorted the gray matter. Case 4. (d) Histology of Fig. 2c. Case 4. HE. (e) Caudal agenesis, lubmar spinal cord. The spinal cord was abruptly terminated at the level of the midlumbar segment . In longitudinal sections, it was evident that the duplicated spinal cord on the dorsal aspect was folded back and fused with the main spinal cord at its termination. The empty space (asterisk) was a tissue processing artifact. Case 5. HE. (f) Diplomyelia/type II SCM with myelodysplasia, lumboscral spinal cord. A nerve mass was attached and partially incorporated into the dorsal aspect of the spinal cord. Case 6. (g) Histology of Fig. 2f. The nerve mass was composed of disorganized gray and white matter and was connected with the spinal cord where a vertical defect formed on the right side. Case 6. HE.
Case 5: Diplomyelia (Type II SCM) With Early Termination (Caudal Agenesis) and Ischemic Myelomalacia
In case 5, there was a crescentic mass on the dorsal aspect of the spinal cord at the level of the cranial lumbar segment, consisting of a duplicated spinal cord in histology as described above. Caudal to the mass, the spinal cord abruptly terminated (Fig. 2e). A longitudinal section showed that the dorsal aspect of the duplicated spinal cord was enfolded and fused with the main spinal cord at its termination (Fig. 2e). In addition, foci of ischemic myelomalacia were noted in the caudal thoracic spinal cord (not illustrated).
Case 6: Diplomyelia (or Hamartomatous Myelodysplasia) With Partial Agenesis and Focal Wallerian Degeneration
In case 6, there was a fluctuant intradural mass attached to the dorsal aspect of the spinal cord at the level of the cranial lumbar vertebrae. On cross-section, the spinal cord was partially encompassed by the mass on its dorsal aspect (Fig. 2f). Serial cross-sections showed that the mass was composed of an irregular area of gray and white matter where the neurons, glial cells, and axons were disoriented. The neural mass was incorporated into the dorsal median sulcus and gradually merged with the spinal cord (Fig. 2g), in which the right side was separated by a large vertical fissure (presumptive, processing artifact). In the sections from the caudal lumbar spinal cord, approximately 25% of the parenchyma on the right side was absent. In the tissues on the right side of the spinal cord that were present, the dorsal gray matter column was malformed, medially displaced, and dorsally connected to the neural mass, which contained a central canal–like structure (not illustrated). Conversely, the left portion of the spinal cord had no obvious congenital abnormality. Occasional small foci of Wallerian degeneration were presented in some sections; however, myelomalacia and vascular FCE were not observed. In the sacral spinal cord, the mass merged with the intermediate column of gray matter (Fig. 3a). There were duplicated central canals (Fig. 3a, inset) and the spinal cord lost its normal symmetry and polarity. On the right side, the dorsal and ventral columns of gray matter were poorly developed, approximately 50% of the white matter and associated spinal nerves were absent. The remaining axons were disorientated. A small, isolated island of gray matter was present at the ventrolateral margin (Fig. 3a).
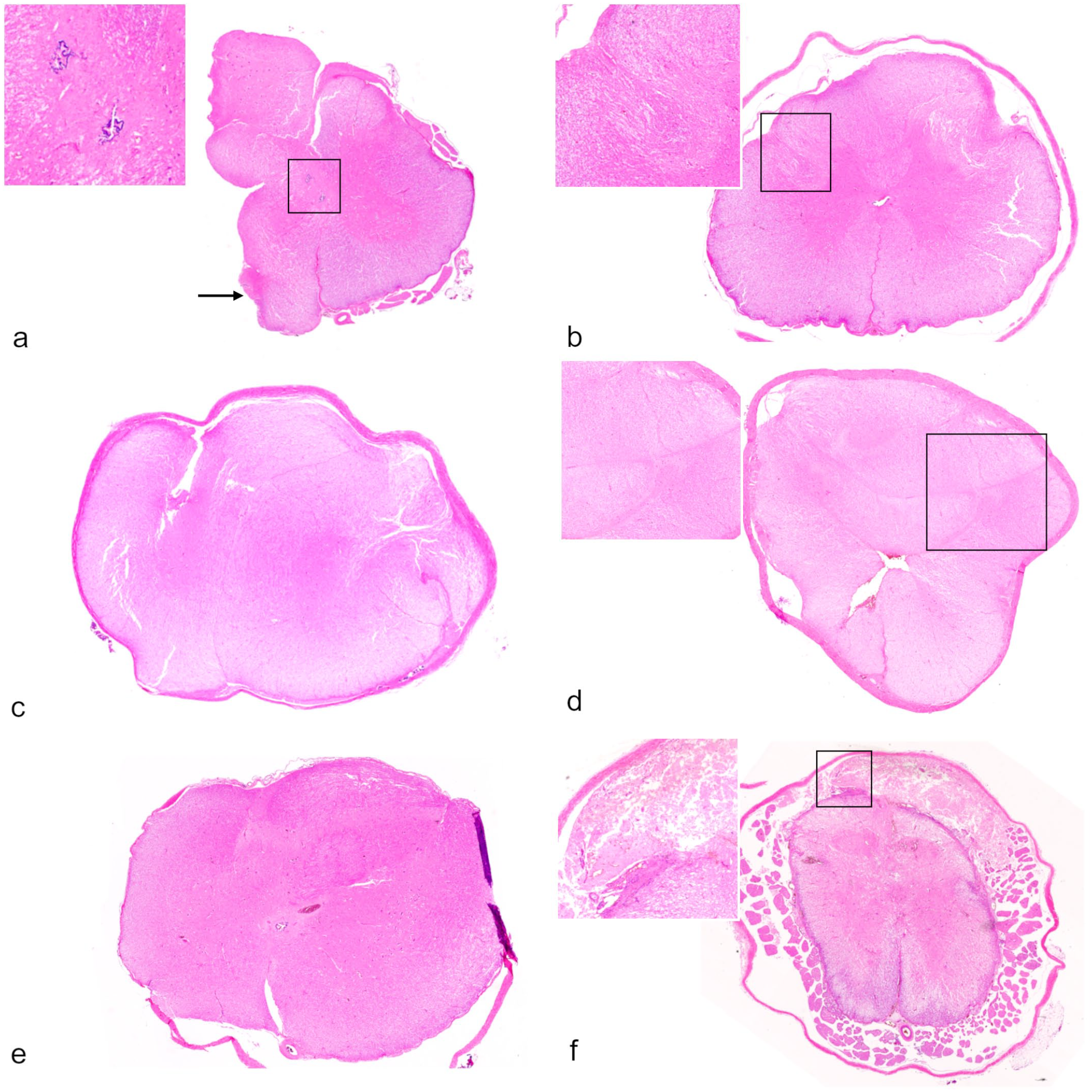
Closed spinal dysraphisms. (a) Myelodysplasia, there were extensive defects on the right side, duplication of central canal (inset), a small aberrant island of gray matter (arrow) at the ventrolateral margin, and a dorsal nerve mass. Case 6. Hematoxylin and eosin (HE). (b) Myelodysplasia (segmental spinal dysgenesis/caudal agenesis), spinal cord. Swirling axons surrounded the dorsolateral sulci (inset). Case 7. HE. (c) Myelodysplasia (segmental spinal dysgenesis), thoracolumbar spinal cord. The spinal cord was divided by a thin fibrous stroma, resulting in 2 separated portions. Each had gray and white matter. Case 7. HE. (d) Myelodysplasia (segmental spinal dysgenesis), thoracolumbar spinal cord. The spinal cord was divided by a thin line of fibrous tissue (inset). There were 5 small islands of gray matter interspersed with disorientated white matter. Case 7. HE. (e) Myelodysplasia (segmental spinal dysgenesis), lumbar spinal cord. There was indistinct gray and white matter differentiation, and the tissue architecture was markedly disarranged without identifiable horns, funiculi, sulci, and the absence of dorsal and ventral median fissures. Case 9. HE. (f) Myelodysplasia (caudal agenesis) and ischemic myelomalacia, lumbosacral spinal cord. Nearly all the tissue architecture of the sacral spinal cord was effaced by ischemic myelomalacia. Note, there was an intradural and extramedullary nerve mass (inset) on the dorsal aspect of the spinal cord, which was affected by myelomalacia as well. The spinal rootlets were intact, reflecting the different routes of blood supplies. Case 10. HE.
Case 7: Myelodysplasia (Segmental Spinal Dysgenesis)
At the level of thoracolumbar vertebrae, the spinal cord of case 7 had a grossly contour without distinct demarcation of the gray and white matter. Histologically, various anomalies were observed in a series of sections. The first anomaly was characterized by axons around the dorsolateral sulci that were bilaterally whorled and associated with distorted adjacent dorsal gray matter columns (Fig. 3b). The second anomaly was characterized by asymmetrical division of the spinal cord by a thin fibrous tissue in the middle, resulting in 2 independent islands of gray matter encompassed by disoriented axons (Fig. 3c). The third anomaly was composed of 5 small, intermittently connected islands of gray matter surrounded by irregular white matter in which the axonal bundles were disoriented (Fig. 3d). The central canal was absent in the affect spinal cord segment.
Cases 8 and 9: Myelodysplasia (Caudal Agenesis)
At the level of the lumbar (case 8) and lumbosacral (case 9) segments, the spinal cords had deformed contours with indistinct gray and white matter differentiation. Histologically, myelodysplastic lesions were present, which were characterized by loss of symmetry, abnormal arrangement of gray matter without identifiable horns, disorganized white matter with loss of identifiable funiculi and sulci, absence of dorsal and ventral median fissures (Fig. 3e), and a segmental absence of the central canal (case 8).
Case 10: Myelodysplasia (Caudal Agenesis) and Ischemic Myelomalacia
At the level of lumbosacral vertebrae, the spinal cord of case 10 was softened with extensive myelomalacia noted on the cross-sections. Microscopic examination demonstrated ischemic myelomalacia as a consequence of fibrocartilaginous emboli occluding leptomeningeal arterioles, with nearly all gray and adjacent white matter destroyed in the most severely affected area. The remaining white matter showed extensive Wallerian degeneration. In addition, there was an intradural extramedullary neural mass on the dorsal aspect of the sacral spinal cord (Fig. 3f). The morphological details of this mass were completely obliterated by necrosis, with few remaining degenerated and necrotic neurons present. The spinal rootlets surrounding the spinal cord were intact.
Cases 11 Through 13: Unremarkable
No remarkable lesions were noted in cases 11 through 13.
Discussion
This study documents the occurrence of ischemic myelomalacia due to FCE (cases 1–3, 5, 10; Fig. 1) and several types of spinal cord malformations (cases 4–10, Fig. 2) in 18- to 24-week-old pigs, which are similar to spinal cord malformations that have been described in human and other species, usually as individual case reports or review articles. In 2 cases (cases 4 and 5; Fig. 2a, b), a dorsoventral duplication of the spinal cord was asymmetrically separated by a thin fibrous septum and enclosed within a common dural sheath. This anomaly is morphologically compatible with diplomyelia and type II SCM reported in humans,16,17 calves, 10 dogs,20,26 and foals. 9 It is noteworthy that these cases had more than 1 anomaly, including dorsoventral duplication of the spinal cord accompanied by myelodysplasia (Fig. 2c, d) or early termination (Fig. 2e). Concurrent diplomyelia and myelodysplasia with early termination of the spinal cord at the lumbar segment have also been reported in a 2-year-old German Shepherd Dog. 20 In 1 case (case 6, Fig. 2f), a disorganized neuroglial tissue mass was partially incorporated into the dorsal aspect of the spinal cord, resembling an incomplete separation of spinal cord duplication (diplomyelia). A similar lesion referred to as “hamartomatous myelodysplasia” was recently reported in a horse. 27 In this study, both terms were listed for case 6. Myelodysplasia is broadly defined as a spinal cord malformation that results from segmental dysgenesis of the neural tube, whereby part of the spinal cord fails to develop properly. This term was used to describe cases 7 to 9, even though the morphological appearances of this lesion differed slightly among these cases (Fig. 3b–e). In human medicine, the anomalies located in the thoracolumbar and lumbosacral spinal cord, respectively, were classified into SSD and CA/CRS. 29 Similar cases of myelodysplasia without skeletal deformities have been reported in cattle, but were restricted to the lumbar cord. 13 In case 10, an intradural-extramedullary neuroglial mass, most likely a form of myelodysplasia, was observed in the sacral region; however, its morphology was obscured by ischemic myelomalacia (Fig. 3f). Likewise, SSD with CA was reported in a newborn Holstein calf, in which a neuroglial mass developed in the sacral segment. 28 The co-occurrence of multiple spinal anomalies with variable but overlapping morphologic features within the same animal may result from 1 or more defects in the neural tube development during early pregnancy. Considering that all the cases were collected from the same pig farm within a certain period, it is plausible that the spinal anomalies had a common pathogenesis but exhibited a wide spectrum of morphological expressions.
While the acute onset of hindlimb lameness could be explained by ischemic myelomalacia secondary to the FCE in some pigs, 3 the clinical impact of the congenital spinal malformations is supported by the lack of myelomalacia in 4 pigs presenting with paraplegia. Considering the likely embryologic origin of the spinal anomalies in these pigs, the later onset of clinical signs that might be attributed to these lesions is difficult to explain. From a perspective of comparative pathology, spinal dysraphism typically presents as a pediatric disorder, frequently identified during early childhood with the onset of neurologic deficits. 18 However, a poorly characterized subset of patients with SCM may become symptomatic later in adulthood.16,17 Similarly, SCM was identified in the cervical spinal cord in a 9-year-old Thoroughbred gelding with sudden onset lameness of the right forelimb. 9 Known as tethered cord syndrome in human medicine, spinal anomalies could cause adhesions with adjacent tissues, which in the case of sudden or repetitive flexion or extension of the spine could result in stretching of the spinal cord.5,17 Likewise, a recent case report described a tailless Holstein calf with caudal spinal dysraphism in which a tethered spinal cord in the lumbosacral region caused ataxia and paresis with analgesia of the hindlimbs. 14 In our cases, the presence of adhesion could have been overlooked due to the presence of ischemic myelomalacia or obscured by bandsaw artifacts. Finally, in human medicine, closed spinal dysraphism can be an incidental finding in some patients that have no or very subtle neurological clinical signs and never seek medical care. 16 For pigs with lesions resembling SSD and CA, the severity of the clinical deficit would depend on the residual spinal cord function. Thus, it is possible that subtle clinical signs were not observed by owners before the onset of lameness. The cause of the acute lameness in the 3 pigs (cases 11–13) that had no remarkable pathologic findings is uncertain. Although a reasonably careful gross and histologic examination was performed in all animals, it is possible that tissue sampling biases could have missed more subtle spinal anomalies and/or myelomalacic lesions in these pigs.
An unusually high prevalence of fibrocartilaginous embolism in finishing pigs has been documented in earlier case reports, in which many predisposing factors were documented.3,12 During the period of rapid growth, high lean body type and marginal levels of nutrients are likely associated with higher pressures within the intervertebral disks and spinal instability, making them more susceptible to traumatic injuries. In our case series, pigs were derived from a heavily muscled, high-lean commercial genetic line used for rapid growth. Chronic bacterial diskospondylitis that has been described previously 12 was not a feature in our case series. However, the spinal dysraphic lesions present in our pigs have not been described in previous case reports of FCE.3,12 Given the close relationship between the embryologic development of the spinal cord and vertebrae,2,21,31 it is possible that minor defects in the vertebrae or intervertebral disks could have contributed to spinal instability. An ancillary spine imaging examination25,30 would be required to identify small defects in the vertebrae or disks, although it is not always available in diagnostic laboratory or regional animal hospital settings.
The spinal malformations described in this study are consistent with defects in early embryonic notochord formation. 15 The formation of SCM is associated with a fusion of unzipped notochord to the endoderm with the establishment of a transient communication, named an accessory neurenteric canal between the yolk sac and amniotic space during gastrulation. Abnormalities in the closure of this connection disrupt the midline integration of 2 paramedian notochordal anlagen along the rostral lip of the primitive node, resulting in duplication. The persistence of an accessory neurenteric canal might interfere with the subsequent neurulation process, resulting in myelodysplastic lesions. According to this theory, a duplication of the spinal cord could be formed laterally. In the present study, the dorsoventral duplication (cases 4 and 5) or neuroglial mass (case 6) with an irregular central canal in the distal thoracolumbar segment might be explained by an asymmetric or incomplete splitting of the notochord and the overlying neural plate. Alternatively, from an embryogenetic point of view, the malformations in the thoracolumbar region could be more compatible with SSD, in which defects in the spatial arrangements of somites or the notochord result in asymmetries in the neural tube. 29 However, the malformations in the lumbosacral region fit better with CA/CSR, as a result of developmental error of the tail bud, which is responsible for the development of the caudal notochord during the secondary neurulation.28,29 Nonetheless, embryogenesis is a continuous process and defects in this process can be single or multiple, thus, the exact mechanism underlying the formation of spinal dysraphism remains unclear. However, in the pigs in this study, it is reasonable to conclude that the neurulation cascades were most likely disrupted within a short window between 14 and 18 days of gestation. 31
As suggested above, the type of malformation may depend more on the critical timing of an insult relative to ontogenesis than the nature of the insult itself, if we presume the cause to be an exogenous agent. Some chemical hazards or teratogenic viruses 32 had been considered as possible causes. However, in this series, the farm was participating the “no antibiotics ever” program with minimal medication and using commercial feed with regular ration formulation. The farm record showed no changes in the abortion rate or reproductive performance over the past year. It is generally agreed that most neural tube defects are the result of multi-gene predispositions together with an additive contribution from nutritional, physical, and environmental factors.2,7,8 As compared with humans, individual and nutritional variations were greatly reduced, if not eliminated, in the modern pork industry. Although there was strong evidence demonstrating a genetic component in the etiology, it was still challenged by numerous candidate genes, each individually insufficient to disrupt the embryogenesis of the neural tube.6,11 Conversely, the accumulation of recessive mutations in some genes, such as those related to folate metabolism, 1 could be enhanced through inbreeding and predisposed to congenital malformations as observed in our case series.
To our knowledge, the congenital spinal malformations described in the pigs in this report have not been reported previously. Since the diagnosis of spinal cord disease in large animals requires facilities and personnel in a laboratory to perform complete and careful dissections of the spine, it is possible that spinal cord disorders in these animals are underdiagnosed. Alternatively, they could be novel due to an adverse effect of continuous inbreeding and genetic improvement in the modern swine industry. We believe more swine cases will be identified in the future. With higher economic impacts and scientistic relevance on the comparative pathology, further investigations on genetic regulation and embryogenesis of these spinal dysraphisms are merited.
Footnotes
Acknowledgements
The authors thank Amanda Brock, Scott Goodpaster, Kelsey Garber, and Dr. Benjamin Hause at South Dakota State University for their assistance with histopathology and metagenomic assay.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was, in part, funded by SDSU Agricultural Experiment Station Hatch funds and the SDSU Animal Disease Research & Diagnostic Laboratory.
