Abstract
MicroRNAs (miRNAs) are a class of noncoding RNA molecules playing a crucial role in tumor modulation targeting mRNA. This study aimed to validate the diagnostic potential of a panel of 3 miRNAs previously identified in canine mast cell tumors (MCTs), miR-21, miR-379, and miR-885, as markers of lymph node involvement in terms of histological absence (nonmetastatic: HN0; premetastatic: HN1) and presence (early-metastatic: HN2; overt-metastatic: HN3) of metastasis, in the saliva of mast cell tumor (MCT)-affected dogs by quantitative polymerase chain reaction (PCR). Forty-seven saliva samples were analyzed: 36 from MCT-affected dogs (12 subcutaneous [3 HN0-1 and 9 HN2-3] and 24 cutaneous [9 HN0-1 and 15 HN2-3—MCT]) and 11 from healthy dogs. MCT-group effects were investigated using analysis of variance (ANOVA). The origin of the tumor affected the expression of salivary miR-21 (P = .011) with an increase in cases with subcutaneous MCTs compared with the healthy group (P = .0005) and those with cutaneous MCTs (P = .004). Salivary miR-21 was higher in the HN2-3 class compared with the healthy group (P = .004). Salivary miR-885 was not affected by the presence of MCT, while miR-379 was not detected in saliva. The diagnostic potential of salivary miR-21 in discriminating MCT-affected dogs from the healthy group (AUC = 0.8917), cutaneous from subcutaneous (AUC = 0.8111), and subcutaneous HN0-1 (AUC = 0.7250) and HN2-3 (AUC = 0.9750) classes from healthy samples was demonstrated by receiver operating characteristic curve analysis. Overall, salivary miR-21 was identified as a promising tool, representing a novel approach to detecting MCT-associated epigenetic alterations in a minimally invasive manner.
MicroRNA (miRNAs) are small noncoding RNA that act as molecular orchestrators in almost all cellular pathways, targeting mRNA to block or destroy its translation. 11 MiRNAs are present in all biological fluids, and their regulation depends on the pathophysiological condition. 14 MiRNA expression changes in cancer. 28 The identification of miRNAs involved in tumor progression (oncomiRNAs) and tumor suppression (tumor suppressor miRNAs) has been investigated 3 in both humans and animals. 21 The need for minimally invasive and repeatable-over-time biomarkers for early diagnosis and monitoring of tumor relapse has driven the research toward the use of minimally invasive biosamples, like saliva.15,24,33 Tumor-associated miRNAs, dysregulated in both primary tumors and plasma, are differentially expressed in the saliva of human and canine patients with head and neck cancer 22 and in humans with oral squamous cell carcinoma. 12 The relevance of saliva as an important source for biomarker identification has also been assessed in tumors arising distantly from the oral cavity.19,23 Several studies in human oncology compared the expression profile of miRNAs in different biofluids, 20 while, to the best of the authors’ knowledge, no investigations are reported in canine oncology.
Canine mast cell tumor (MCT) is one of the most frequent neoplasms in dogs with a prevalence from 7% to 21% 31 of all canine skin tumors, and its variable biological behavior requires assessment of the metastatic potential to properly calibrate therapy and estimate prognosis. 1 The standard approach to canine MCT includes tumor staging and histological grading, using both Patnaik 16 and Kiupel 10 grading systems (cutaneous MCTs), or Thompson 29 indices (subcutaneous MCTs). In the last decade, the literature has shown a growing interest in the early detection of lymph node metastasis to accurately stage the disease and suggest appropriate treatment options.7,30,32 Previous studies have detected some MCT-associated miRNAs in plasma and the primary tumor.5,8,34 The miRNA profile of primary MCTs in formalin-fixed paraffin-embedded samples has been previously investigated and suggests that 3 dysregulated miRNAs, miR-21, miR-379, and miR-885, are able to identify lymph node involvement and discriminate between nonmetastatic (HN0-1) and metastatic (HN2-3) tumors. 34 The aim of this study was to demonstrate the potential of saliva as a minimally invasive biological fluid for the detection and quantification of tumor-associated miRNAs suitable for diagnosing and lymph node staging of canine MCT.
Materials and Methods
Saliva Samples Collection
Forty-seven saliva samples were collected using a sterile Dryswab™ (Medical Wire & Equipment, UK) from 36 MCT-affected dogs and 11 healthy dogs cared for at the University Veterinary Teaching Hospital of the University of Milan. Owners signed written consent for the procedure. In the MCT group, dogs with a cytological diagnosis of MCT, that preoperatively staged negative for distant metastases and underwent surgical excision of the MCT and regional 27 or sentinel 6 lymph node(s), were included. For each of these dogs, oncological staging consisted of abdominal ultrasound with the cytological examination of fine-needle aspirates of the spleen and liver, 26 and a peripheral blood smear evaluation was performed. On the day of surgery, after induction of general anesthesia, a sample of saliva was collected by rubbing the sterile swab in the maxillary vestibular area. Dogs were then moved to the operating theater and wide-margin (consisting of 3 cm for the lateral aspect of the neoplasia and 2 deeper noninfiltrated fascial planes) surgical resection of the MCT and regional (n = 4) or sentinel (n = 32) lymph node extirpation were performed. All excised lymph nodes were nonpalpable/normal-sized. The excised MCT and lymph node(s) were submitted for histopathology, fixed in 10% buffered formalin and routinely trimmed and processed. Histopathological evaluation of submitted specimens included: tumor classification (cutaneous or subcutaneous), mitotic count (MC; number of mitosis in 2.37 mm2), tumor grade according to both the Kiupel and the Patnaik grading systems for cutaneous MCT or the Thompson indices for subcutaneous MCT,10,16,29 histologic margins (infiltrated or noninfiltrated), and lymph node classification as reported by Weishaar and colleagues. 30 For the healthy group, we included randomly selected dogs without either MCT or other oncological or systemic diseases that were admitted for routine annual clinical examination and vaccination. The saliva was collected by rubbing the maxillary vestibular area with a sterile swab, with the dog awake. Data regarding the studied population are shown in Table S1. The saliva samples were stored at −80°C until use.
MiRNA Extraction, Retrotranscription, and Quantitative Polymerase Chain Reaction
MicroRNAs were extracted using the miRNeasy Serum/Plasma Kit (Qiagen, Cat. No. 217184). Briefly, the saliva swab was immersed in 2 mL of Qiazol (Qiagen), vortexed, incubated for 5 min at room temperature, and centrifuged at 14,100 × g for 5 min. Then, 3.75 µl (25 fmol final concentration) of the Caenorhabditis elegans miRNA cel-miR-39 (Qiagen, Cat. No. 219610) was added as synthetic spike-in control due to the lack of sequence homology to canine miRNAs. The extraction was then continued following the manufacturer’s recommended procedure, and miRNAs were eluted in 18 µl of RNase-free water. RNA concentrations were quantified with the NanoDrop ND-1000 spectrophotometer (NanoDrop Technologies). Reverse transcription was performed using the TaqMan Advanced miRNA cDNA Synthesis Kit (Applied Biosystems, Cat. No. A28007) following the manufacturer’s instructions.
The quantitative polymerase chain reaction (qPCR) was performed following the MIQE guidelines. 2 Target miRNAs were selected based on the results of a previously reported study. 34 The selected probe assays (Life Technologies) included cel-miR-39-3p (assay ID 478293_mir), miR-21-5p (assay ID rno481342_mir), miR-379-5p (assay ID 478077_mir), and miR-885-5p (assay ID 478207_mir). The qPCR was performed on CFX Connect Real-Time PCR Detection System (Biorad). The reaction included 7.5 µl of 2X TaqMan Fast Advanced Master Mix (Cat. No. 4444557), 0.75 µl of miRNA-specific TaqMan Advance assay (20X), 1 µl of cDNA, and water to reach the final volume (15 µl). The thermal profile was 50°C for 2 minutes, 95°C for 3 minutes, and 40 cycles of 95°C for 15 seconds, and 60°C for 40 seconds. To identify suitable reference miRNAs, a geNorm analysis 9 was performed using Biogazelle’s qbase+ software (www.qbaseplus.com) on the 3 reference miRNAs used in the previous work, 34 namely, miR-122-5p (assay ID rno480899_mir), miR-128-3p (assay ID mmu480912_mir), and miR-101 (custom probe SO_66039417 _6871885). The normalization factor was calculated as the geometric mean of reference miRNAs. The relative expression was calculated using Bio-Rad CFX Maestro™ Software. MiRNAs expression is presented in terms of fold change using the 2-ΔΔCq formula.
Statistical Analysis
The statistical analysis was performed on XLStat software for Windows (Addinsoft, New York, USA) and SPSS 27 (SPSS Inc.). Statistical significance was accepted at a P value ≤ .05. Data were tested for normality and homogeneity of variance using the Kolmogorov–Smirnov and Levene tests, respectively. MiR-885 and miR-21 were not normally distributed and were transformed using square root transformation. Any group effect was investigated using analysis of variance (ANOVA) with miRNA concentrations forming the dependent variables and lymph node and site as the fixed effect. The normality and the homogeneity of variance of residuals were confirmed and post hoc analysis was conducted using Bonferroni post hoc test. A receiver operating characteristic (ROC) was performed to determine the diagnostic performance of miR-21 in discriminating the MCT-affected dogs from the healthy subjects and in predicting the lymph node involvement.
Results
Histology
This study included 36 cases of MCT (Supplementary Table 1), of which 24 were cutaneous and 12 subcutaneous. All cutaneous MCTs were classified as “low grade” according to the Kiupel grading system, and, according to the Patnaik system, 22 of them were classified as grade II and 2 as grade I. Six subcutaneous MCTs had an infiltrative growth pattern, while 6 MCTs, partially demarcated and partially infiltrative, were recorded as “combined” according to Thompson. 29 The MC ranged from 0 to 1, with a higher MC in only 2 subcutaneous MCT cases; one with an MC of 5 and the other of 7. Multinucleated neoplastic cells were present in 6 subcutaneous MCTs and absent in the other 6 cases.
Lymph nodes were classified as HN0-1 in 12 MCT cases (9 cutaneous, of which 7 were HN0 and 2 HN1; 3 subcutaneous, of which 1 was HN0 and 2 HN1) and as HN2-3 in the other 24 cases (15 cutaneous, among which 10 were HN2 and 5 were HN3; 9 subcutaneous, among which 5 were HN2 and 4 were HN3).
Salivary miR-21 was higher in subcutaneous MCTs
Analysis of the salivary miRNAs expression stability by geNorm indicated that the most stable reference miRNAs with the lowest M value were miR-128-3p and miR-101 with average M values of 0.569 and 0.782, respectively. Their mean was used for the normalization of the relative quantification data experiments.
Quantitative PCR was performed on all 47 saliva samples. Two of the 3 selected salivary miRNAs, miR-21 and miR-885, were quantified in all samples, while miR-379 was not detected in saliva. In healthy dogs, the fold change (FC) of circulating miR-21 was 1.02 ± 0.46 standard deviation (SD), while in dogs with MCT, it was 1.74 ± 1.42 SD in the case of cutaneous MCTs or 4.58 ± 3.59 SD in the case of subcutaneous MCTs. The site had a significant effect on salivary miR-21 (ANOVA; P = .011). miR-21 was higher in the saliva of dogs with subcutaneous MCTs compared with the healthy group (ANOVA P = .000) and dogs with cutaneous MCTs (ANOVA; P = .004) (Fig. 1a). No statistically significant difference was detected comparing salivary miRNAs of cutaneous MCT cases and the healthy group.
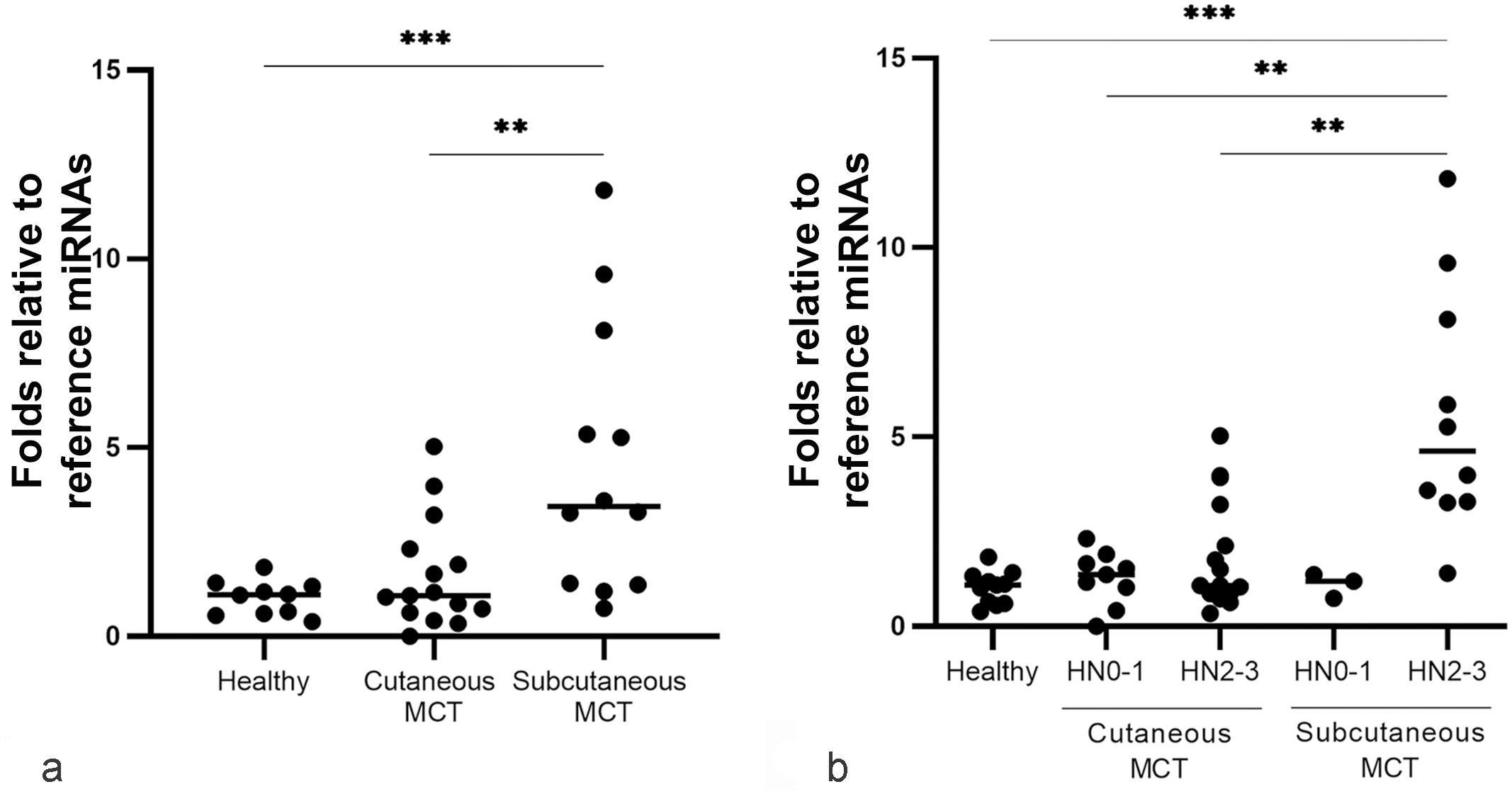
Value plot of miR-21 expression level in the saliva of healthy and mast cell tumor (MCT)-affected dogs. (a) Saliva of healthy dogs compared with the saliva of cutaneous and subcutaneous MCT groups. (b) Healthy saliva samples compared to the saliva of tumor classes, nonmetastatic/premetastatic histological node (HN)0-1 and the early metastatic/metastatic HN2-3. Blackline marks the median. Significance was defined at *P < .05, **P < .01, ***P < .001.
Considering tumor classes based on the lymph nodal involvement, salivary miR-21 was higher in dogs with stage HN2-3 MCT compared with the healthy group (ANOVA; P = .004) (Fig. 1b).
The level of salivary miR-885 was not affected by the presence, absence, or location of MCTs (0.35 FC ± 0.18 SD in healthy dogs, 0.49 FC ± 0.30 SD in dogs with cutaneous MCTs, and 0.63 FC ± 0.41 SD in dogs with subcutaneous MCTs).
Diagnostic Performance of Salivary miR-21
To prove the reliability of salivary miR-21 in discriminating MCT-affected dogs and predicting lymph node involvement, ROC analysis was performed. The area under the curve (AUC) was calculated to estimate the diagnostic potential of the salivary differentially expressed miRNA. Fig. 2 shows the AUC value calculated by comparing the salivary miR-21 levels of subcutaneous MCTs and those of cutaneous MCTs. The AUC was good (0.8111, 95% CI [0.6685, 0.9537]), and the cut-off was set at 3.2734 with a sensitivity of 86.7% and specificity of 66.7% (Fig. 2).
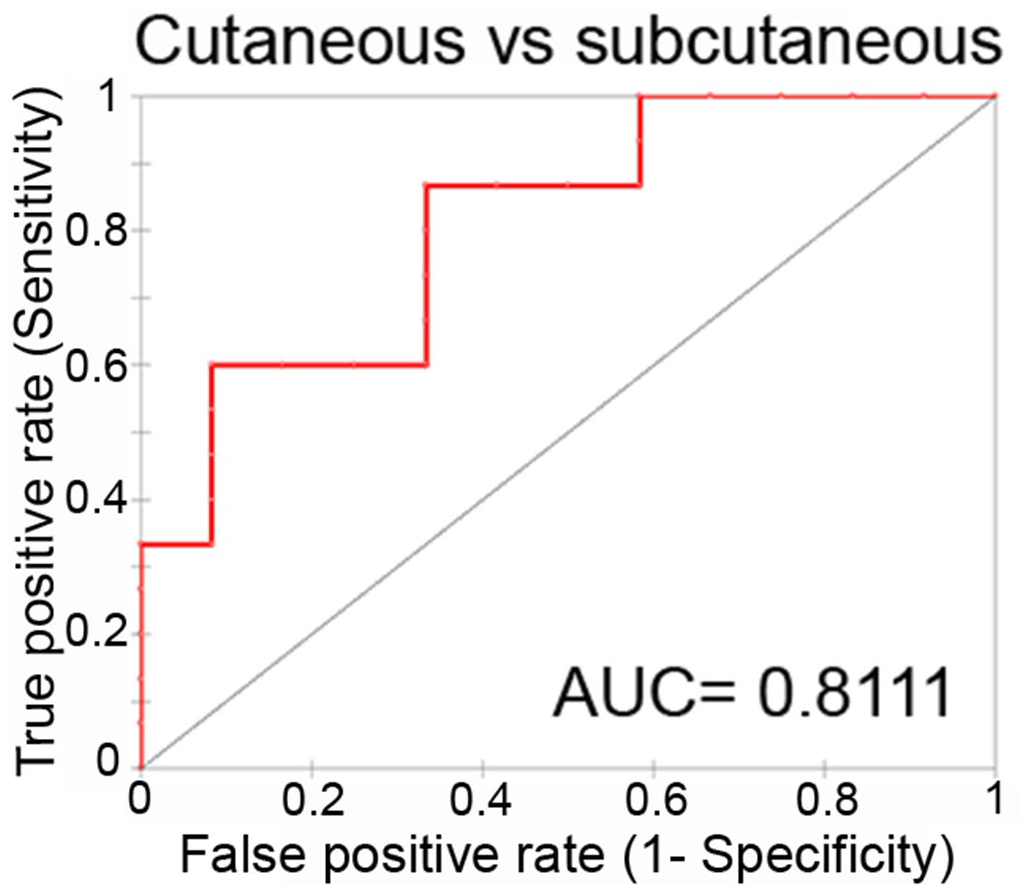
Performance of salivary miR-21 as a biomarker for discriminating cutaneous and subcutaneous affected dogs. Receiver-operator characteristic (ROC) curve analysis. AUC= area under the curve.
The AUCs for salivary miR-21 of subcutaneous and cutaneous MCT versus healthy samples were good (AUC = 0.8917; 95% CI [0.8019, 0.9814]) (Supplemental Figure S1a) and bad (AUC = 0.5667; 95% CI [0.3515, 0.7818]) (Supplemental Figure S1b), respectively. The AUCs of subcutaneous HN0-1 and HN2-3 classes versus healthy samples were sufficient (AUC = 0.7250; 95% CI [0.4778, 0.9722]) (Supplemental Figure S1c) and excellent (AUC = 0.9750, 95% CI [0.9750, 0.9750]) (Supplemental Figure S1d), respectively. Since the expression of salivary miR-21 was the same for cases of cutaneous MCTs with HN0-1 and HN2-3, the ROC analysis was not performed.
Discussion
In this study, the potential of saliva as a reliable minimally invasive biosample for the quantification of MCT-associated miRNAs in dogs was investigated for the first time. Three MCT-associated miRNAs, miR-21, miR-379, and miR-885, previously reported to discriminate between cutaneous MCT-affected and healthy dogs in formalin-fixed paraffin-embedded primary tumors, 34 were quantified in saliva samples of healthy and MCT-affected dogs to investigate their potential as minimally invasive biomarkers. The 3 salivary miRNAs were quantified using a qPCR approach, and the results showed that only salivary miR-21 was modulated in the presence of the subcutaneous MCT. miR-885 was detectable, but its level was not affected by the presence of MCT, and miR-379 was not detected in saliva. Kiupel low-grade and Patnaik grade I or II MCTs can develop occult/early-metastasis/or overt-metastasis to regional/sentinel lymph nodes, which may require dogs to receive adjuvant therapies.7,10,25,30 Therefore, the ability to discriminate the nonmetastatic/premetastatic (HN0-1 class) from the early-metastatic/overt-metastatic (HN2-3 class) lymph nodes may provide a step forward in obtaining more accurate staging. So far, few studies using transcriptomic 17 and miRNomic 34 approaches have investigated epigenetic changes in MCTs. Fenger 5 and colleagues reported overexpressed miR-9 in canine high-grade MCTs. MiRNA-126 is up-regulated in some epithelial and nonepithelial neoplasms, like MCT, 8 and the amounts of miR-21, miR-379, and miR-885 in the tumor are able to predict the spread to lymph nodes in MCT-affected dogs. 34 Increased levels of miR-21 have been observed in the saliva of several cancer-affected human patients, suggesting its role as an oncomiRNA 20 promoting tumor progression. 4 This study confirmed that miR-21 in the saliva of subcutaneous MCT-affected dogs is also higher compared with healthy and cutaneous MCT-affected dogs. The comparison between healthy, HN0-1, and HN2-3 subcutaneous MCT classes showed that the level of salivary miR-21 increased with tumor progression, suggesting that it may serve as a marker for lymph node involvement.
The results of this study suggest that saliva may be a suitable biofluid to detect miRNAs that discriminate lymph node involvement in subcutaneous MCT-affected dogs. MiRNAs in saliva are already regarded as potential candidates for cancer detection in human oncology, 20 demonstrating potential in the identification and monitoring of cancer patients18,23 and in discriminating metastatic from localized tumors. 13 If confirmed in further studies in a broader cohort, the finding of this study might provide a way of excluding early nodal metastases and avoiding unnecessary lymphadenectomy in dogs as well, at least in the HN0-HN1 group. The ROC curve analysis suggested that salivary miR-21 has good diagnostic potential in discriminating subcutaneous MCT cases from the healthy group, while it has excellent diagnostic potential in discriminating HN2-3 subcutaneous MCT cases from the healthy group. Thus, this salivary miRNA might be used as a molecular tool to support the clinical decision-making process.
This study provides new and important insights into the potential of saliva as an alternative, minimally invasive biofluid for the detection of MCT-associated miRNAs. Still, we acknowledge that this study has some limitations. First, the effect of miR-21 upregulation on target genes was not investigated. This information would provide important insights into the molecular pathogenesis of MCT. Second, further experiments involving a higher number of patients and sets of samples are required to validate and strengthen the miR-21 potential use and its reliability as a diagnostic test in veterinary clinical oncology.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221128922 – Supplemental material for Salivary miR-21 is a potential biomarker for canine mast cell tumors
Supplemental material, sj-pdf-1-vet-10.1177_03009858221128922 for Salivary miR-21 is a potential biomarker for canine mast cell tumors by Valentina Zamarian, Damiano Stefanello, Roberta Ferrari, Lavinia E. Chiti, Valeria Grieco, Emanuela DallaCosta, Fabrizio Ceciliani and Cristina Lecchi in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The funding for this research was provided by Linea 2-2018, awarded by Università degli Studi di Milano.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
