Abstract
This study evaluated the expression of vimentin and Ki-67 proliferative index (PI) by immunohistochemistry in 30 canine gastric carcinomas (GCs) and a possible association with clinical and pathological features and patient’s survival time. Vimentin immunoreactivity was assessed in neoplastic cells (in primary lesions, emboli, and metastases) and tumor-associated stroma (TAS) of canine GCs. Ki-67 PI was quantified in the neoplastic epithelial component. Vimentin immunolabeling in neoplastic cells was found in 30% of the primary lesions, in 82% of the neoplastic emboli, and in 50% of the metastases; in TAS, it was observed in all cases. A mean of 16% of the TAS was immunolabeled for vimentin. High vimentin immunolabeling in the TAS (>16%) was detected in 40% of cases. The average value of Ki-67 PI was 50%, and 80% of the lesions had Ki-67 PI above 20%. Vimentin immunolabeling in neoplastic cells was more frequent in less-differentiated carcinomas (diffuse [29%] and indeterminate types [75%]) than well-differentiated carcinomas (intestinal type [0%],
Keywords
Gastric cancer accounts for less than 1% of all reported neoplasms in dogs and carcinoma represents 50% to 90% of all canine gastric malignancies. 1 Metastasis has been reported to occur in 70% to 90% of the cases by the time of diagnosis or death, and the most common site of metastasis is regional lymph nodes.1,21 The median age of dogs at diagnosis of gastric carcinoma (GC) ranges from 8 to 10 years, but occasional cases have been reported in dogs younger than 5 years.1,21 Several studies have shown a male predilection, and some authors suggest a breed predisposition in Belgian shepherd, Rough collie, Staffordshire bull terrier, Chow-chow, and standard Poodle. 1
Diagnosis is typically made late in the course of disease. Most dogs with early GC are relatively asymptomatic until the disease progresses to advanced stages. Clinical signs of GC are not specific (i.e., vomiting, anorexia, weight loss, and lethargy) and may be seen with other diseases. 21 Moreover, clients may be resistant to performing consecutive endoscopies and biopsies to monitor the possible multistep progression from gastritis to gastric neoplasia. 16 Therefore, at the time of diagnosis, tumors are often at an advanced stage, resulting in a poor prognosis and limited therapeutic options. 1 Treatment involves surgical resection but is often complicated by diffuse infiltration of the gastric wall, metastases, and frequently a debilitated patient.16,25
In human gastric cancer, molecular markers (such as CEA, CA 19.9, and HER-2) are routinely used for prognosis and predictive purposes.
36
Although some potential prognostic markers have been investigated in canine GC (such as HER-3, HER-2, EGFR, and
The World Health Organization (WHO) classification for domestic animals subdivided GCs based on cellular differentiation into papillary, tubular, mucinous, signet ring cell, and undifferentiated types. 19 However, previous studies demonstrated that some canine gastric neoplastic lesions fit specific histological types only described in the human WHO classification, such as poorly cohesive and mixed carcinomas.1,29 Despite its usefulness in the recognition of morphological patterns, the WHO classification schemes offer little prognostic significance.19,64 The Lauren classification for human tumors may more accurately predict prognosis based on the histopathological features of the lesion 31 and has been successfully adapted to the dog.14,25,44
Vimentin, a 57 kDa protein, is one of the most widely expressed and highly conserved proteins of the type III intermediate filament protein family, known to sustain cellular integrity and provide resistance against stress factors. 49 Typically, vimentin is expressed in various cells of mesenchymal origin, such as fibroblasts, chondrocytes, macrophages, and endothelial cells, but not in epithelial cells. 7 However, vimentin expression has been found to be aberrantly expressed in several human epithelial malignancies (i.e., prostate, breast and lung cancers, as well as in gastrointestinal, and central nervous system tumors). 49 Furthermore, the expression of vimentin in neoplastic epithelial cells may be closely associated with accelerated tumor growth, cell invasion and poor prognosis. 49 To our knowledge, such a relationship has not yet been demonstrated in canine gastric tumors, although vimentin expression in neoplastic epithelial cells has been associated with poor prognosis and shorter survival time in other types of neoplasms in the dog.46,50
For decades, cancer studies were mainly focused on the characteristics of the neoplastic cells themselves. However, it is increasingly acknowledged that TAS contributes to cancer initiation, progression, and metastasis, and thus influences prognosis. 3 For instance, several reports have shown that the increased proportion of TAS, quantified via tumor-to-stroma ratio, was associated with a worse prognosis for humans with cancers of the alimentary tract, including GC.3,23,63 Furthermore, recent studies reported a high proportion of TAS associated with high tumor node metastasis (TNM) stage and diffuse histological type of human GCs.3,27
Tumor-associated stroma consists of a mixture of various stromal cells, such as fibroblasts, immune cells, and endothelial cells, as well as extracellular matrix. Vimentin is expressed in many of the stromal components of the tumor microenvironment, and an increased stromal vimentin expression has previously been associated with a poor outcome in human patients with colorectal, prostatic, ovarian, and GCs.34,41,43,53,66 As far as we know, such a relationship has not been previously investigated in canine gastric or intestinal epithelial tumors, although the expression of vimentin in tumor stroma has been evaluated. 38
Ki-67 is a cellular proliferation marker expressed in cell nuclei during all active phases of the cell cycle (G1, S, G2, and mitosis), but not in the resting phase (G0), 67 so determining the percentage of Ki-67 expressing cells (Ki-67 PI) is widely applied in routine clinical work to assess the proliferative activity of tumor cells.9,12 Ki-67 PI has been extensively evaluated in human and veterinary oncology to help determine the prognosis of different types of neoplasms.20,51,56,65
In this study, we aimed to analyze and quantify vimentin immunoreactivity in GC cells and in the TAS and to assess Ki-67 PI in the neoplastic epithelial cells of canine GCs to determine if there is an association between the expression of these molecules and the clinical and pathologic features of the tumors and the survival time of patients.
Materials and Methods
Sample Collection
Thirty canine GC samples, collected during endoscopic procedures, surgery, or necropsy examination, were selected from the archives of the Laboratory of Veterinary Pathology, ICBAS-UP (Portugal), where they were received between 2004 and 2020.
This study was approved by Animal Welfare Organization (ORBEA) of the ICBAS-UP (Porto, Portugal), authorization Nº 201/2017. All the interventions made on animals were performed in a clinical context based on the best clinical judgment of their attending practitioners. Only well-preserved gastric samples were included in this investigation. Owners gave written informed consent for clinical samples, clinical information, and examination results to be used for research. Clinical information from dogs included in the study were collected from the histopathological forms and, when available, from the medical records. Data collected included epidemiological data (age, sex, breed, body weight), clinical sings, tumor location, presence or development of metastasis, and patient outcome.
Histopathology
Tissues were fixed in 10% buffered formalin and paraffin embedded. Serial consecutive 2-µm sections were stained with hematoxylin and eosin.
Sections were independently examined by 3 veterinary pathologists (MT, FG, and IA), confirmed as GC, and classified according to the diagnostic criteria of the human WHO classification and Lauren classification. The WHO classification was determined by the most prominent histological pattern and the principal cell type of the tumor as tubular, papillary, mucinous, signet ring cell, poorly cohesive, and mixed carcinoma. Tumors were classified as tubular when they contained prominent neoplastic tubules, as papillary when neoplastic cells formed papillary structures, as mucinous when they contained extracellular mucin in >50% of the tumor volume, and as signet ring cell when the great majority of the tumor was composed of malignant cells containing intracytoplasmic mucin with eccentric nuclei. Poorly cohesive carcinomas were composed of poorly cohesive cells that morphologically resembled histiocytes. Mixed carcinoma contained a mixture of well-differentiated and signet ring or poorly cohesive histological components. 4 According to the Lauren classification, tumors were classified into the following categories: intestinal type when they contained rudimentary glands that superficially resembled intestinal glands, diffuse type when they contained cells that failed to form distinct structures, and indeterminate type when they contained equal proportions of intestinal and diffuse characteristics. 31 Thus, intestinal type was considered well-differentiated, and both the diffuse and indeterminate types were considered less differentiated. The presence of neoplastic emboli was considered whenever tumor cells were observed invading through a vessel wall and endothelium or when neoplastic cells were observed within a space lined by lymphatic or blood vascular endothelium. 37
Immunohistochemistry
For the immunohistochemical study, sections were deparaffinized in xylene and rehydrated in graded alcohols. Antigen retrieval was performed in a water bath with Target Retrieval SolutionTM (Dako) for 20 minutes. The NovolinkTM Max-Polymer detection system (Leica, Novocastra) was used for visualization, according to the manufacturer’s instructions. Slides were incubated overnight at 4°C with antivimentin mouse monoclonal antibody (V9; Dako), diluted 1:500 and anti-Ki-67 mouse monoclonal antibody (MIB-1; Dako), diluted 1:50. Sections were rinsed with triphosphate buffered saline (TBS) in each step of the procedure. Color was developed with 3.3-diamino-benzidine (DAB, Sigma), and sections were then counterstained with hematoxylin, dehydrated, and mounted. Negative controls were performed by replacing the primary antibodies by another of the same immunoglobulin isotype at the same concentration. Positive controls tissues were sections of canine mammary tissue known to express vimentin in its stroma and samples of canine lymphoma with high Ki-67 PI.
After vimentin and Ki-67 immunolabeling, sections were independently examined and evaluated by 4 observers (ARF, FS, FG, and IA). When there was a divergence of opinion, a consensual diagnosis was achieved.
Vimentin immunoreactivity was assessed separately within neoplastic cells in the primary lesions, neoplastic emboli, and metastases, and within the TAS. Neoplastic epithelial cells and tumor stroma or inflammatory cells were differentiated based on cell morphology and immunophenotyping when necessary, using immunolabeling for pan-cytokeratin (cocktail AE1/AE3, Menarini, dilution 1:300) in serial sections, following procedures described above. In neoplastic epithelial cells, the immunohistochemical expression for vimentin was considered positive when homogeneous granular cytoplasmic immunolabeling was observed in more than 10% of the neoplastic epithelial cells in the lesion (primary lesions, neoplastic emboli, or metastases). Vimentin immunoreactivity in the TAS was assessed using Free ImageJ Fiji software (https://imagej.nih.gov/ij/download.html), as described below.
The Ki-67 PI was defined as the percentage of positive neoplastic cell nuclei determined by counting at least 1000 nuclei in selected fields, at high magnification (400×).26,39 Areas of highest immunolabeling were searched, and areas of necrosis were avoided. In a previous study conducted by our group, the mean value of Ki-67 expression in normal canine gastric mucosa was 20%. 2 Accordingly, tumors were categorized as having Ki-67 PI ≤20% or >20%.
ImageJ Analysis
For each case, 10 microphotographs were captured with a Nikon Eclipse E 600 microscope, equipped with a digital color camera (Nikon, Tokyo, Japan), at 200× magnification (field of view area = 142.16137 µm2), and saved in JPEG format. 41 Before capturing the images, the color density and white balance were standardized for all images. When vimentin immunolabeling was also found in neoplastic cells, areas of the TAS with no labeled tumor cells were selected to avoid overlapping of vimentin immunoreactivity results in both components. A total of 300 digital images were analyzed.
Briefly, the JPEG image was opened with ImageJ, and the software was set to measure area (µm). Global scale of the image analysis was set as 5.880 pixels = 1 µm, in a pixel ratio of 1. The image was changed to grayscale by setting
Statistical Analysis
Analyses were performed using GraphPad Prim 5 (GraphPad Software Inc., La Jolla, CA). The chi-square test was used to examine the association between vimentin and Ki-67 immunolabeling and several clinical and pathologic characteristics (sex, age, weight, tumor location, histological diagnosis, depth of tumor invasion, neoplastic emboli, and metastatic lesions). Correlation among vimentin immunolabeling in the TAS and Ki-67 PI was determined using Spearman rank correlation analysis. A
Survival time was defined as the period (in days) from diagnosis to animal death/euthanasia. Survival time was censored for dogs that were lost to follow-up. Pearson’s correlation coefficient was used to estimate the correlation between vimentin immunolabeling in the TAS and Ki-67 PI and survival time, and Point-biserial correlation were used to elucidate the correlation between vimentin immunolabeling in the neoplastic epithelial cells and survival time. As survival time was only available for a small number of cases, only the value of the correlation coefficient was reported.
Results
Clinical Data and Gross Findings
The 30 cases included in this study were mostly male (18/30; 60%) and of large breed (26-45 kg) in 17/28 cases (61%); the weight was unavailable in 2 dogs. The mean age of the affected dogs was 10 ± 3 years (range: 5-14 years). The study included 6 crossbreeds, 3 Chow-chows, 2 Poodles, 2 Siberian huskies, 2 Labrador retrievers, 2 Golden retrievers, 2 Collies, 2 West highland white terriers, and 9 other breeds.
In this study, 19 cases died and 11 were lost to follow-up after diagnosis. The median survival time was 15 days (n = 19; 95% confidence interval [CI]: [7-45] days). The diagnoses were based on full-thickness biopsies in 20 cases, biopsies including mucosa, submucosa and tunica muscularis in 7 cases, and biopsies including mucosa and submucosa layers in 3 cases. The tumors were located in the antral region in 13/30 cases (43%), the lesser curvature of the gastric body in 12/30 cases (40%), and both regions in 4/30 (13%); the location was undetermined in 1 case due to the small size of the biopsy sample (Supplemental Table S1).
Histological and Immunohistochemical Findings
The most common WHO histological subtypes were signet ring cell (11/30; 37%) (Fig. 1) and poorly cohesive (8/30; 27%) (Fig. 2), and diffuse type (21/30; 70%) was the most common Lauren classification. Of the 30 tumors, 16 invaded the tunica muscularis, 11 invaded the serosal surface, and 3 were limited to the mucosa. Most cases had neoplastic emboli (16/30; 53%) (Fig. 3), which were usually observed inside lymphatic vessels, both in the tumor stroma (intratumoral area) and in the connective tissue surrounding the neoplastic lesion (peritumoral area). Neoplastic emboli could be evaluated by IHC in 11 cases; an additional 5 cases had neoplastic emboli but had insufficient tissue for IHC. Metastases were microscopically confirmed in 7 dogs (cases 10, 11, 20, 22, 23, 24, and 27) and diagnosed by ultrasound in 1 dog (case 21; Fig. 4; Supplemental Table S1). Metastatic lesions could be evaluated by IHC in 6 cases.
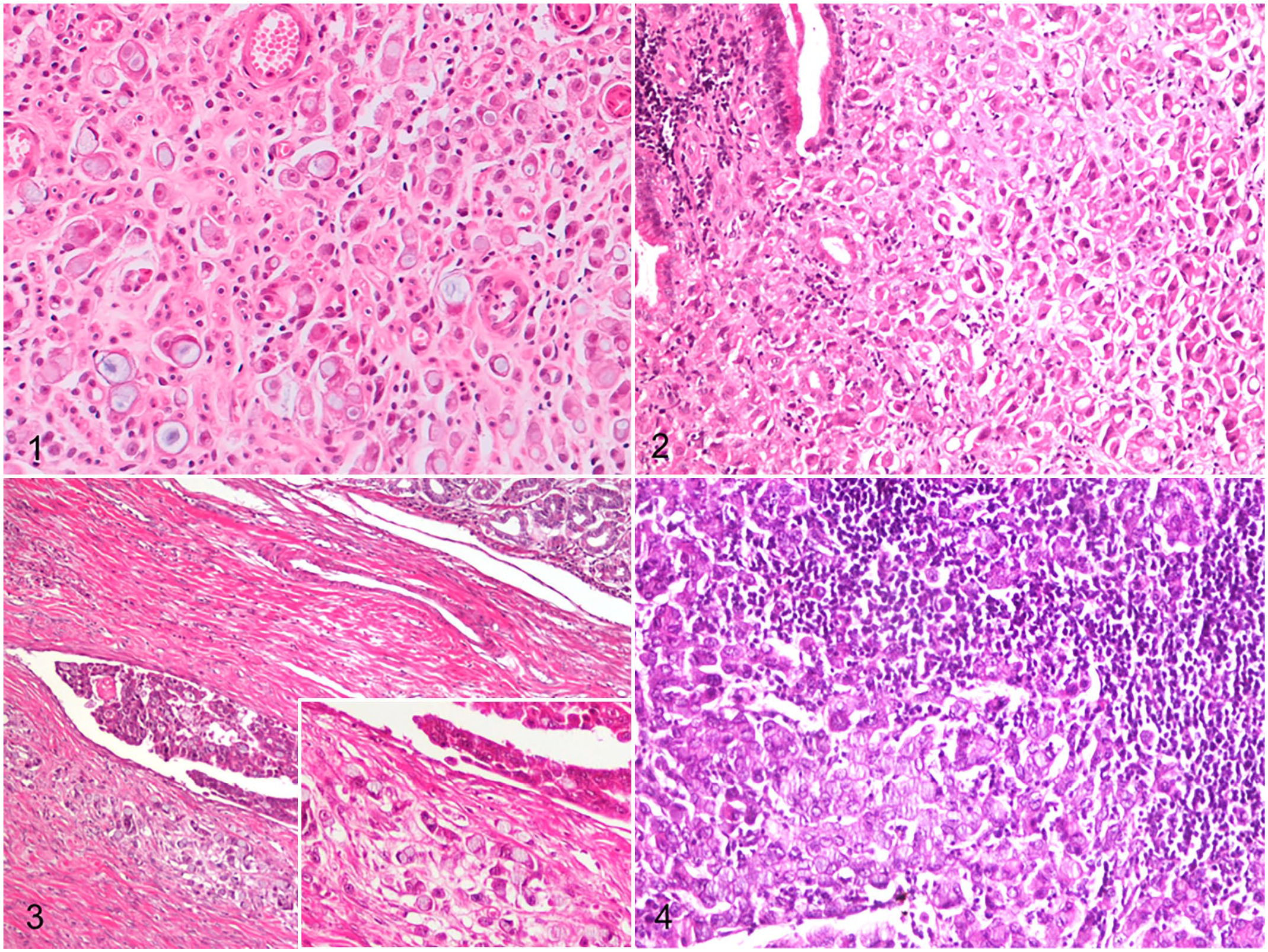
Gastric carcinoma, dogs. Hematoxylin and eosin.
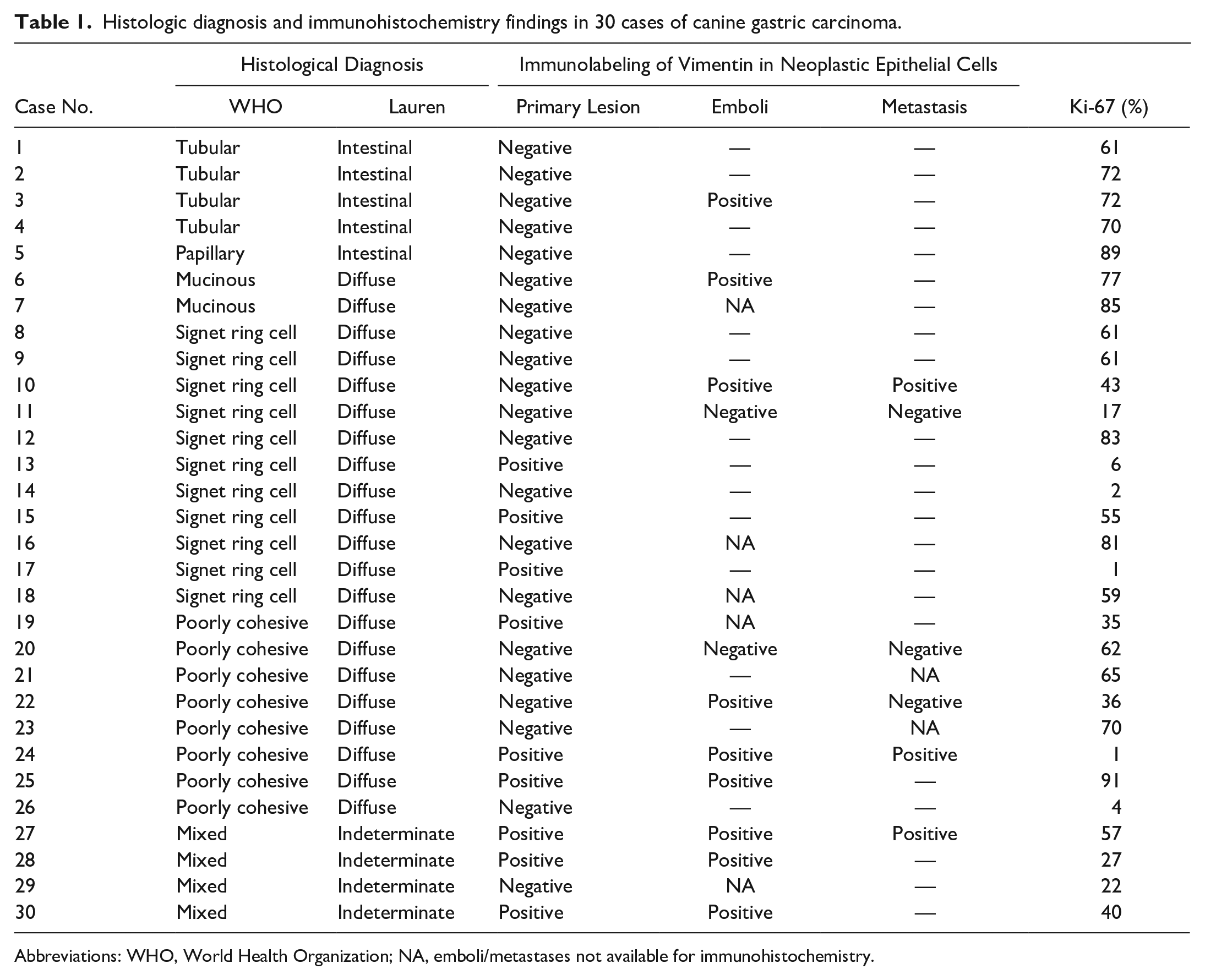
Vimentin was expressed in the cytoplasm of neoplastic epithelial cells in 9/30 cases (30%; Figs. 5–7; Table 1). Of these, 6 were histologically classified as diffuse type (67%) and 3 as indeterminate-type carcinomas (33%). In the intestinal type of GCs, the neoplastic epithelial cells were all immunonegative for vimentin (Fig. 8). Vimentin immunolabeling in neoplastic epithelial cells was more frequent in less-differentiated carcinomas (i.e., diffuse [29%] and indeterminate carcinomas [75%]) than in well-differentiated carcinomas (i.e., intestinal carcinomas [0%],
Histologic diagnosis and immunohistochemistry findings in 30 cases of canine gastric carcinoma.
Abbreviations: WHO, World Health Organization; NA, emboli/metastases not available for immunohistochemistry.
Association of clinical and pathologic features with immunohistochemistry data in 30 cases of canine gastric carcinoma.
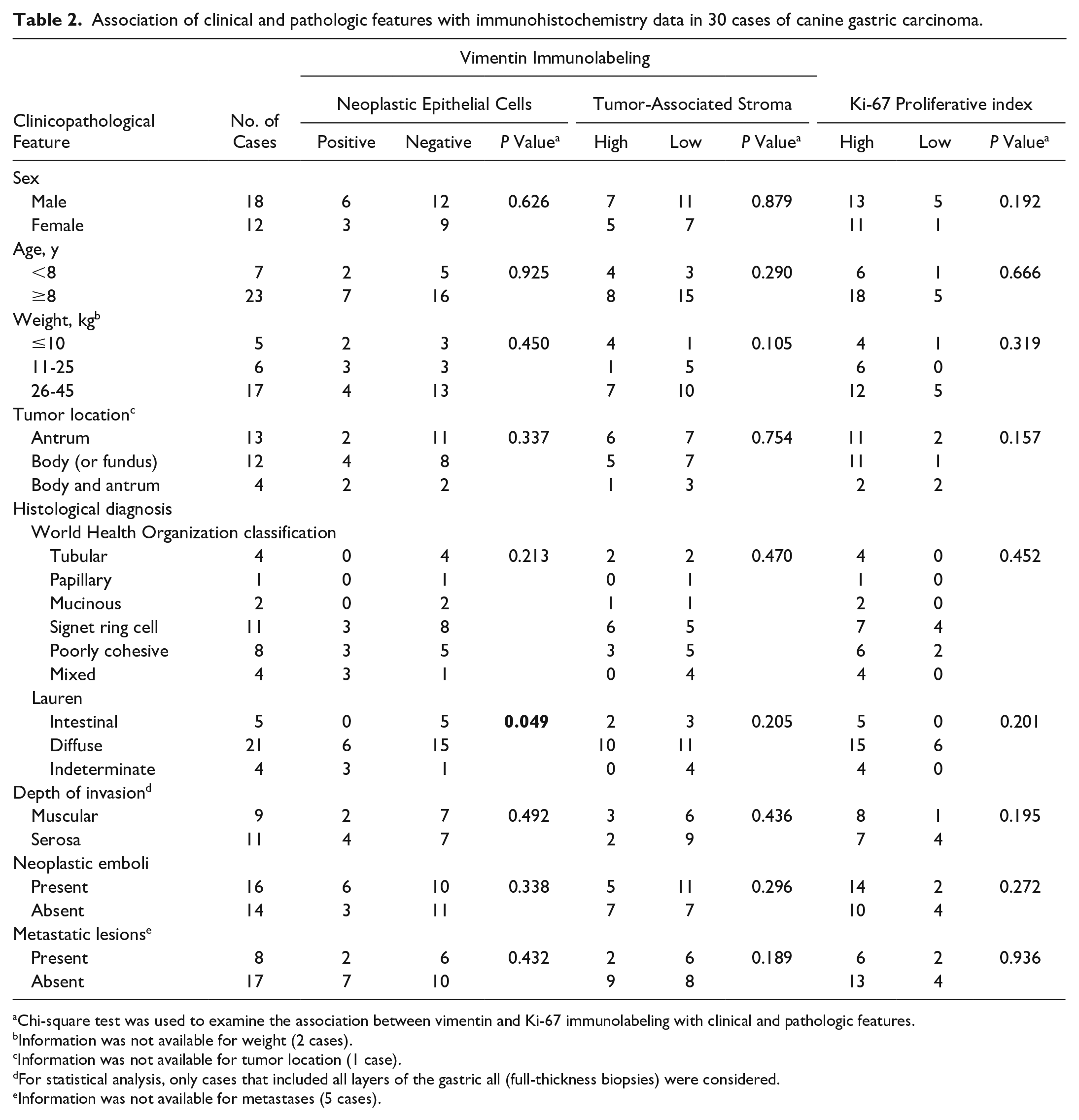
Chi-square test was used to examine the association between vimentin and Ki-67 immunolabeling with clinical and pathologic features.
Information was not available for weight (2 cases).
Information was not available for tumor location (1 case).
For statistical analysis, only cases that included all layers of the gastric all (full-thickness biopsies) were considered.
Information was not available for metastases (5 cases).
Of the 9 carcinomas with vimentin immunolabeling in neoplastic cells in the primary tumor, 6 had neoplastic emboli, and 2 of these had metastatic lesions (one case affecting regional lymph nodes and the other both regional lymph nodes and esophagus). Of the 21 carcinomas with the absence of vimentin immunolabeling in neoplastic cells in the primary tumor, 10 had neoplastic emboli, and 6 had metastatic lesions. However, no significant differences were found between vimentin immunolabeling in neoplastic epithelial cells and the presence of neoplastic emboli or metastases.
Overall, vimentin was expressed in neoplastic emboli in 9/11 cases (82%) evaluated (Fig. 9). In 4 of these 9 cases, vimentin immunolabeling was found in the neoplastic emboli but not in the primary tumor. Of the cases with metastatic lesions analyzed by IHC, vimentin labeling was detected in neoplastic cells within the metastases in 3/6 cases (50%). Of these, one case showed vimentin immunolabeling in metastatic tumor cells but not in the primary tumor (Fig. 10; Table 1).
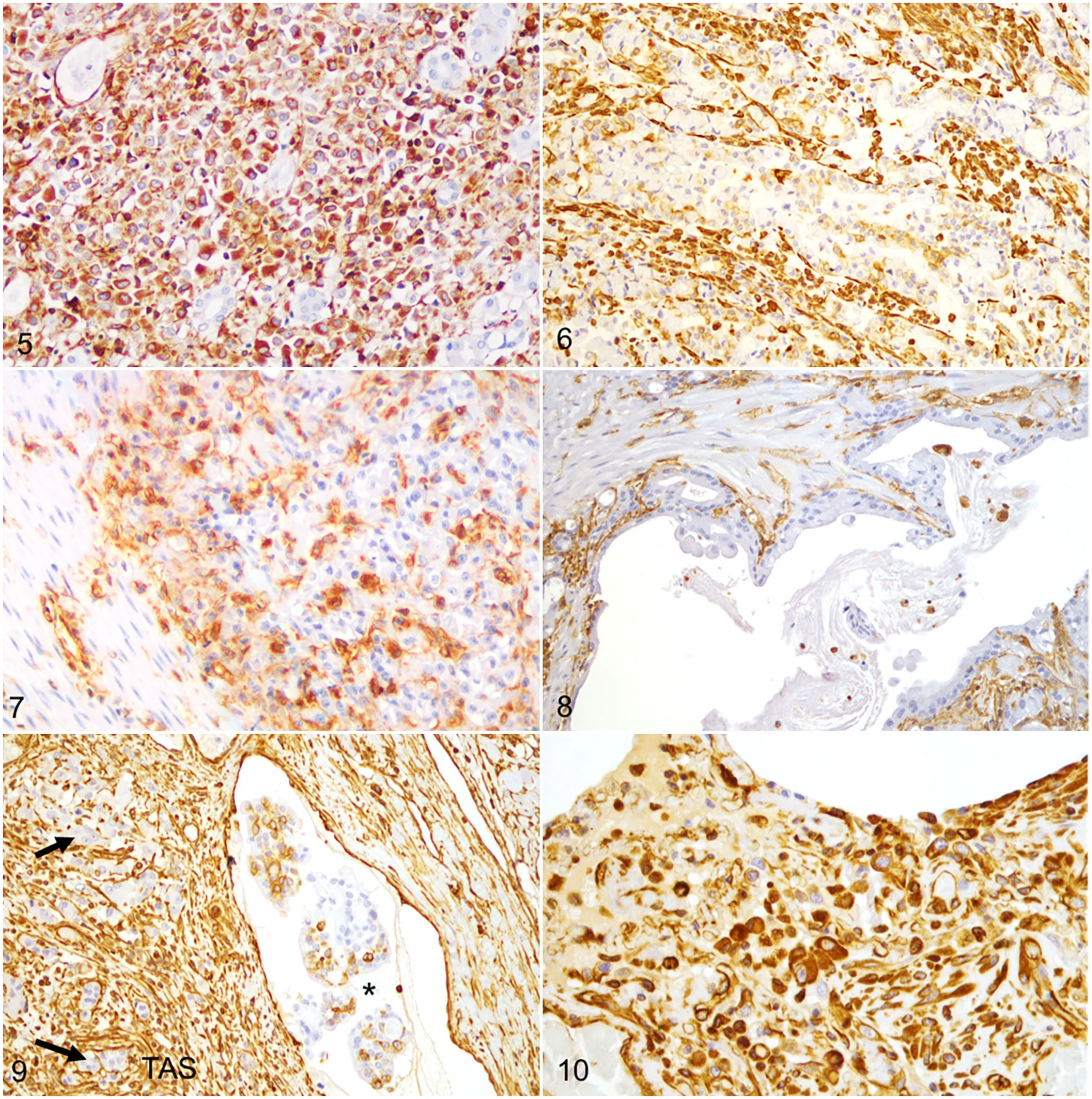
Gastric carcinoma, dogs. Immunohistochemistry for vimentin.
Vimentin immunolabeling was observed in the TAS of all cases. The surface area of the TAS that was immunolabeled for vimentin varied between 3% and 38% (mean 16% ± 8%). Vimentin immunolabeling of the TAS was high (>16%) in 12/30 cases (40%) and low (≤16%) in 18/30 cases (60%; Figs. 11–14). Vimentin immunolabeling was often prominent in the stromal tissue surrounding clusters of infiltrating neoplastic epithelial cells. Vimentin immunolabeling in the TAS was not associated with tumor location, histological diagnosis, depth of tumor invasion, or presence of neoplastic emboli or metastases. Nevertheless, of the cases with high vimentin immunolabeling in the TAS, 10/12 (83%) were of the diffuse type.
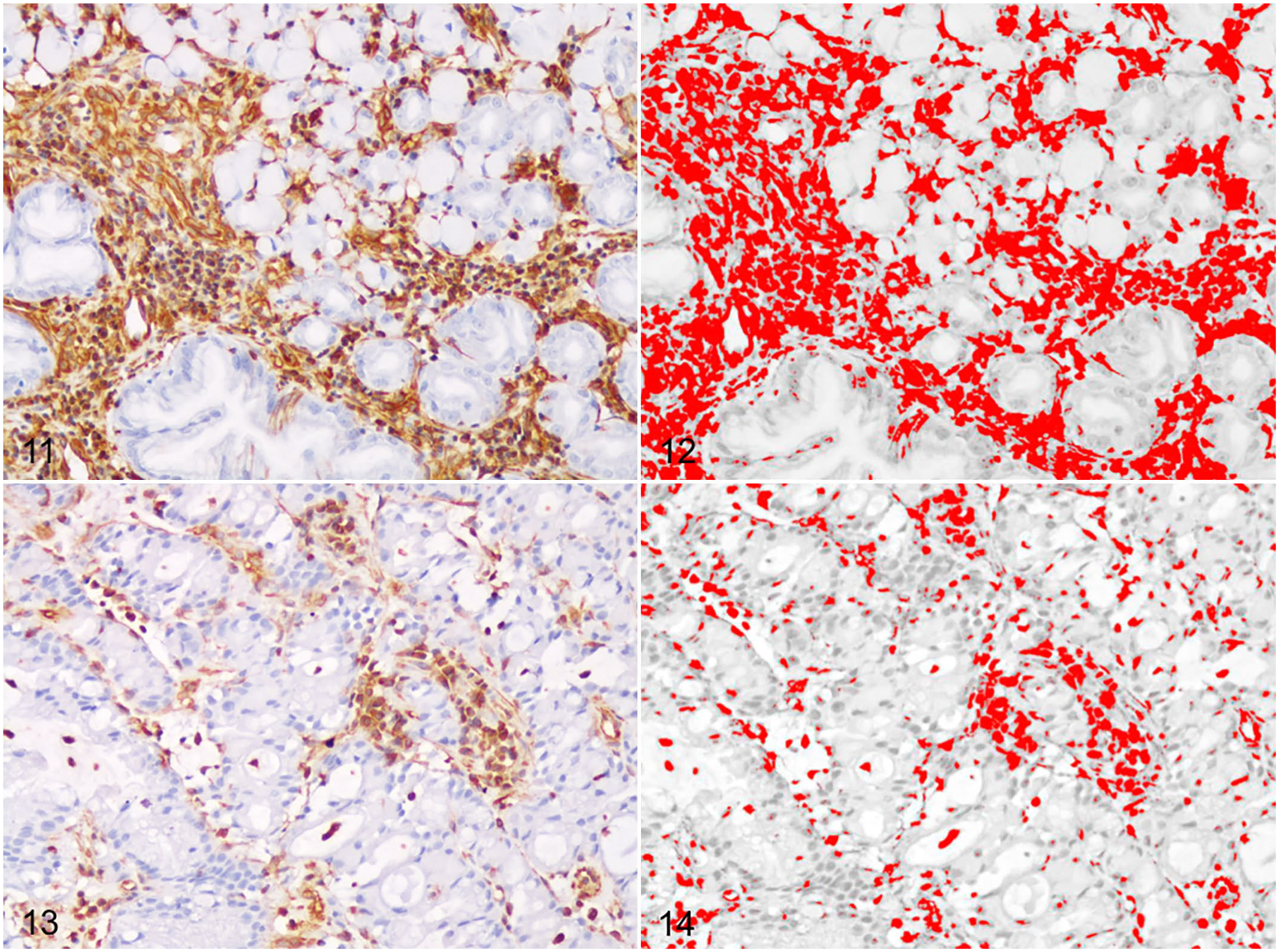
Gastric carcinoma, dogs. Immunohistochemistry for vimentin.
Ki-67 PI of neoplastic cells varied widely from 1% to 91% (mean 50% ± 28%). Of 30 GCs, 24 (80%) had a Ki-67 above 20% (Figs. 15–18). The Ki-67 PI was not associated with any of the clinical or pathologic parameters. A statistically significant positive correlation was found between vimentin immunolabeling in the TAS and Ki-67 PI (
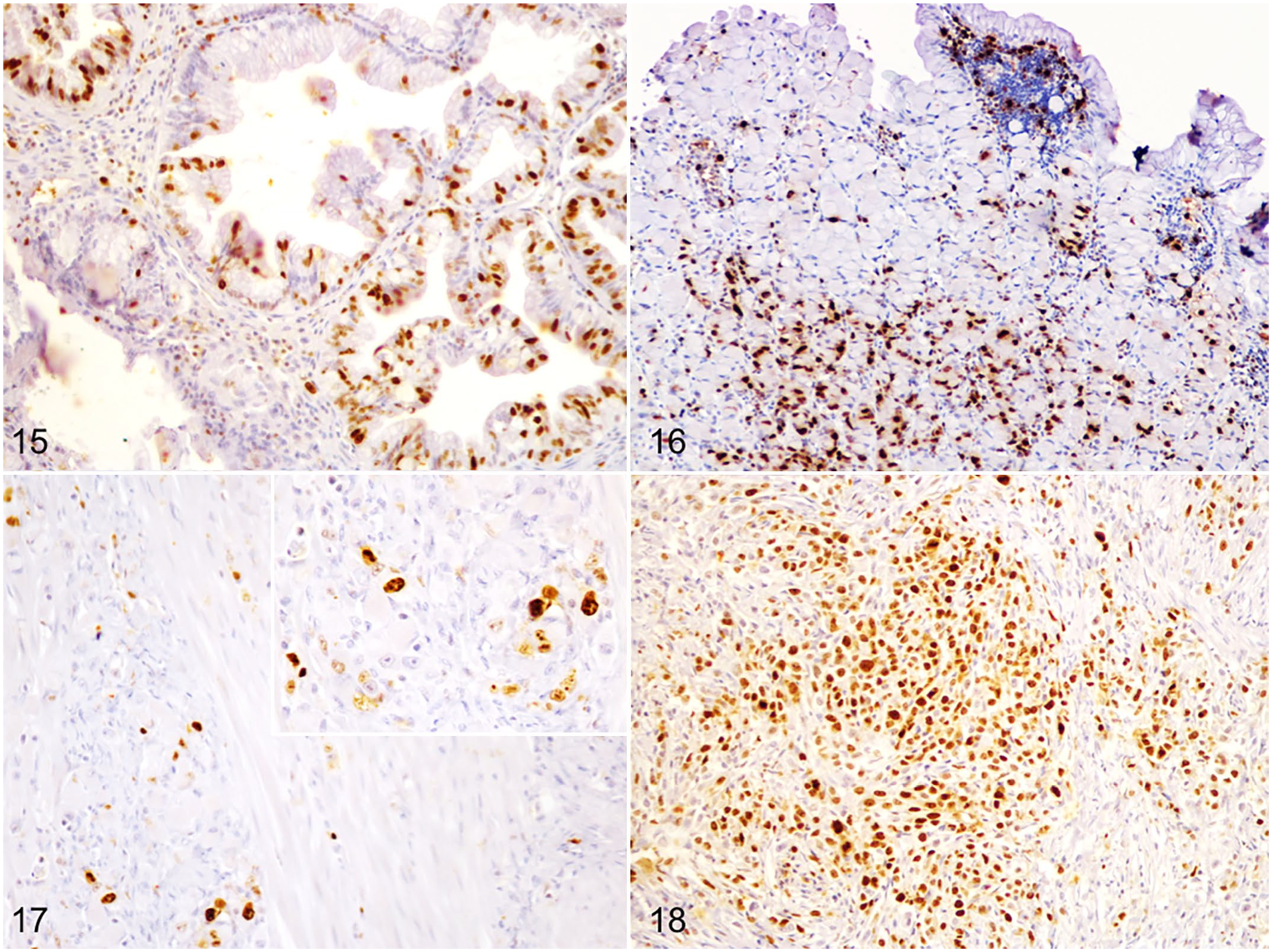
Gastric carcinoma, dogs. Immunohistochemistry for Ki-67.
A weak correlation was found between vimentin immunolabeling in neoplastic epithelial cells and the survival time of the canine patients (
Discussion
In recent years, there have been numerous investigations concerning vimentin’s role in human and canine cancers.46,47,49 In human gastric cancer, vimentin has often been associated with an invasive phenotype and was suggested as a prognostic marker of metastasis. 49 This study evaluated the immunohistochemical expression of vimentin in neoplastic epithelial cells and TAS of 30 canine GCs.
Reinforcing previous data, herein the dogs affected with GCs were mostly males.8,14,55 The mean age of the GCs cases agreed with those reported by others.8,48 Similar to previous data,8,14,55 we found a predominance of signet ring cell and diffuse-type carcinomas, according to the schemes adapted from the human WHO classification and Lauren, respectively. Our data support previous studies in which the lesser curvature and pylorus were the most frequently involved sites of canine GC.14,33
The expression level of vimentin in neoplastic epithelial cells increases during epithelial-to-mesenchymal transition (EMT), which is considered an important change in the adhesion and migration of tumor cells. 22 During EMT, epithelial cells change their phenotype, exhibiting reduction of cell-cell contacts, loss of polarity, increased cell motility and invasiveness, repression of epithelial cell markers [i.e., epithelial cell adhesion molecule, cytokeratin, or E-cadherin], and aberrant up-regulation of certain mesenchymal markers (i.e., vimentin and N-cadherin). 40 Epithelial-to-mesenchymal transition was previously described in canine invasive colorectal tumors, with numerous tumor cells co-expressing E-cadherin and vimentin. 62 Our results revealed that 30% of the cases displayed vimentin immunolabeling in neoplastic epithelial cells of primary lesions. This value is similar to that in previously published data in human gastric cancer series, where vimentin immunolabeling was observed in 6% to 32% of primary GCs.15,28,35,59,60,68 In this study, vimentin immunolabeling was most intense in tumor cells at the deep invasive front, as also reported in human GCs. 54 Indeed, EMT takes place at the periphery of the tumor where cells are more exposed to cytokines and to the extracellular environment that promotes EMT. 5
It is generally agreed that malignancy and prognosis of GC depends on their stage, histological differentiation and infiltrative growth pattern. 54 In this study, vimentin immunolabeling in neoplastic epithelial cells was significantly associated with Lauren’s histological classification, being more frequent in less-differentiated histotypes (diffuse and indeterminate type carcinomas) compared with well-differentiated types (intestinal carcinoma). Similarly, other authors found an association between vimentin expression and diffuse type or poorly differentiated GC in humans.15,28,68 In this study, most canine GC cases with vimentin immunolabeling in neoplastic epithelial cells had neoplastic emboli, but this did not reach statistical significance. Thus, we speculate that vimentin expression may occur in less-differentiated neoplastic epithelial cells, accounting for a more aggressive behavior of vimentin-positive GCs of dogs.
A study of human GCs 24 found no expression of vimentin in primary lesions but strong vimentin expression in some cells invading intratumoral vessels. It was suggested that these cells must undergo mesenchymal transition to survive in the peripheral circulation and be able to implant at metastatic sites. In this study, 4 cases with absence of vimentin in the primary lesion had vimentin immunolabeling in neoplastic emboli.
Our study showed, unsurprisingly, that TAS cells in all carcinomas labeled for vimentin, similar to a previous study in canine gastrointestinal tumors 38 and some studies in human GC.41,43 Furthermore, vimentin immunolabeling was more prominent in the stromal cells surrounding clusters of invasive neoplastic cells.
Vimentin is a canonical marker of EMT, a biological process in which neoplastic cells lose their epithelial morphology and characteristics, invade through the basement membrane, and navigate the surrounding microenvironment.57,61 However, this model has been questioned in part by studies showing collective invasion in which neoplastic epithelial cells invade collectively as a multicellular unit.10,45 In a recent study, Labernadie et al 30 demonstrated that cancer-associated fibroblasts, the major cellular component of the TAS, drive the collective invasion of neoplastic cells through heterotypic interactions between the N-cadherin expressed in cancer-associated fibroblasts and E-cadherin on tumor cells. Along similar lines, vimentin was reported to be necessary for pulmonary adenocarcinoma metastasis by maintaining heterotypic tumor cell-cancer-associated fibroblasts interactions during collective invasion. 45 Taking all the above evidence together, our results suggest that EMT does not occur in the great majority of canine GC cases, and that tumor invasion may be promoted by stromal cells strongly expressing vimentin that surround clusters of infiltrative neoplastic cells. Additional studies are necessary to prove this theory.
Vimentin immunolabeling in the TAS was quantified using computer-assisted imaging. 41 Computer-assisted analysis has been increasingly employed and has proven to be superior to manual visual interpretations, thereby minimizing interobserver variation and providing more accurate and detailed immunolabeling quantification. 17 In our study, vimentin immunolabeling scores in the TAS varied from 3% to 38%, with a mean value of the surface area immunolabeled by vimentin of 16%. Vimentin immunolabeling in the TAS suggests a dynamic change in the microenvironment during tumor progression that can be attributed, for example, to fibroblastic changes, neoangiogenesis, and infiltration by inflammatory cells. Previous data on vimentin expression in the TAS in human GCs are limited, which impairs further comparisons between species. However, the mean of vimentin immunolabeling in the TAS in this study was much higher than the mean recorded in previous studies of human colorectal carcinomas (9% and 6%).34,41 In a study of human gastric cancer, high vimentin mRNA expression was significantly associated with Lauren diffuse-type and recurrence or distant metastasis, and vimentin immunoreactivity was only detected in the tumor stroma. 43 Although a significant association between vimentin immunolabeling in the TAS and histological type has not been demonstrated in this study, most cases with high vimentin immunolabeling in the TAS were of the diffuse type.
Vimentin is one of the markers for identifying fibroblasts, the major cellular component of the TAS. 18 Fibroblasts, being activated by cytokines and at the same time secreting cytokines and/or other soluble factors, were reported to modulate various aspects of tumor progression including tumor cell proliferation.6,52 A positive correlation between vimentin immunolabeling in the TAS and Ki-67 PI in neoplastic epithelial cells was found, that may be attributed to fibroblastic changes induced by tumor cell proliferation, or the other way around.
A study of human GC found a significant association between high vimentin mRNA expression and poor survival. 43 In that study, the immunohistochemical expression of vimentin was only detected in the stromal cells, and the authors hypothesized that the survival of patients with GC was associated with the stromal vimentin expression. Herein, a low negative correlation was found between vimentin immunolabeling in the TAS and survival time. This may be due to the limited number of cases in which follow-up was available.
In humans, high Ki-67 PI has been associated with poor clinical outcome in a variety of malignancies, including gastric cancer.20,42,56 The mean of Ki-67 PI in the GC of this study was 50%, a value higher than the percentage reported in a previous study of canine colorectal carcinomas (17%) 65 but close to the data reported by Lazăr et al 32 (46%) and Joo et al 26 (50%) regarding human GC. Compared to that previously demonstrated for normal canine gastric mucosa, 2 Ki-67 PI was markedly higher in GC tissues; as expected, the expression level increased with malignancy.
In canine gastrointestinal epithelial tumors, some studies have demonstrated an association between Ki-67 expression and tumor malignancy,51,65 whereas others found no correlation between Ki-67 PI and histological parameters of malignancy such as tumor depth and differentiation. 39 The results of this study showed no association between Ki-67 PI and histopathological features of malignancy including tumor differentiation, depth of tumor invasion, and the presence of neoplastic emboli or metastases. These discrepancies between studies are perhaps related to the anatomic location of the tumor in the gastrointestinal tract (stomach vs small intestine vs colon vs rectum), the scoring system adopted for Ki-67 immunoreactivity evaluation, and/or with the sample size.
In this investigation, a moderate negative correlation was observed between Ki-67 PI and the survival time of dogs with GC, which is consistent with previous studies in human GC, where high Ki-67 expression was associated with poor overall survival.32,58 However, further studies including a larger number of canine GC cases, and follow-up studies are warranted to confirm these results.
Limitations of the study include its retrospective nature and the small number of samples, subjected to different sampling methods (partial vs full-thickness biopsies), which were performed by multiple veterinary surgeons using nonstandardized surgical procedures. Owing to the long period of time considered in this retrospective study (2004–2020), parts of the patients’ medical records were lost, and some clinical information data were not recorded on the histopathological forms (i.e., clinical signs, tumor location, metastatic status, and cause of death). Necropsy examination should have been performed on all dogs. Survival time is affected by euthanized, which is based on clinical decisions; moreover, it was only available for 19 out of 30 dogs, which is why the correlation analysis only included the correlation coefficient value. Notwithstanding, our results provide an important contribution to the study and understanding of canine gastric carcinogenesis and encourage further investigations, including a larger number of canine GC cases with clinical follow-up to confirm, with scientific precision, the clinical significance of vimentin and Ki-67 PI and its usefulness as prognostic markers.
In summary, this study is the first, to our knowledge, that investigated immunohistochemical expression of vimentin in both epithelial and stromal compartments in a series of canine GCs. Vimentin immunolabeling in neoplastic epithelial cells was more frequent in less-differentiated carcinomas. A positive correlation was demonstrated between vimentin immunolabeling in the TAS and Ki-67 PI. Furthermore, a moderate negative correlation was found between Ki-67 PI and survival time. In view of available data, vimentin analysis and quantification and Ki-67 PI have potential for providing prognostic information in cases of gastric cancer in dogs.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221117858 – Supplemental material for Vimentin and Ki-67 immunolabeling in canine gastric carcinomas and their prognostic value
Supplemental material, sj-pdf-1-vet-10.1177_03009858221117858 for Vimentin and Ki-67 immunolabeling in canine gastric carcinomas and their prognostic value by Ana R. Flores, Alexandra Rêma, João R. Mesquita, Marian Taulescu, Fernanda Seixas, Fátima Gärtner and Irina Amorim in Veterinary Pathology
Footnotes
Acknowledgements
ARF (SFRH/BD/116373/2016) acknowledges FCT, the Portuguese Foundation for Science and Technology, for financial support. IPATIMUP integrates the i3S Research Unit, which is partially supported by FEDER through the Operational Program for Competitiveness Factors-COMPETE and National Funds through the Portuguese Foundation for Science and Technology (FCT), under the project number PEst-C/SAU/LA0003/2013.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
