Abstract

Clinical History, Laboratory Results, and Gross Findings
A 6-year-old, castrated, male domestic shorthaired cat presented to the Ohio State University College of Veterinary Medicine Dermatology Service in March 2021 for evaluation of chronic pododermatitis. The owner first noted the lesions in February 2018. Clinical signs included licking of his right forepaw with crusts noted on his paw pads. The licking continued and the lesions progressed and became ulcerated and hemorrhagic. Similar lesions developed on the right hind paw. Various medical therapies were attempted by the referring veterinarian including antibiotics (cevofecin, orbifloxacin, zithromycin), glucocorticoids (prednisolone), and cyclosporine. The cat tested negative for feline immunodeficiency virus (FIV) and feline leukemia virus (FeLV). A biopsy from the right front paw pad in January 2019 revealed chronic pyogranulomatous pododermatitis with ulceration. Special stains (Gram and Fite-Faraco acid-fast) were performed, and no etiologic agents were apparent. The lesions waxed and waned for 3 years, sometimes with several months between recurrences. The lesions recurred in January 2021 with associated lameness, prompting referral to our dermatology service.
Gross Findings
On dermatologic examination, the right forepaw had alopecia and hyperpigmentation on the fifth digit with crusting lateral to the fifth digital pad. There were crusts around the fourth claw with crusting and alopecia cranial to the dewclaw. There was severe crusting and ulceration affecting the metacarpal pad (Fig. 1a). After clipping the fur, draining tracts were apparent in the third through fifth interdigital spaces (Fig. 1b). The right rear paw had draining lesions with alopecia, hyperpigmentation, and proliferative tissue on the lateral metatarsus, lateral to the fifth digit. There was a large crust on the caudal and cranial aspects of the fifth digit. The metatarsal pad had crusting with a large ulcer. There was a hemorrhagic, alopecic, crusting lesion on the medial aspect of digit 4 and draining tracts in the third through fifth interdigital spaces. The skin over the right popliteal lymph node was ulcerated and crusted.
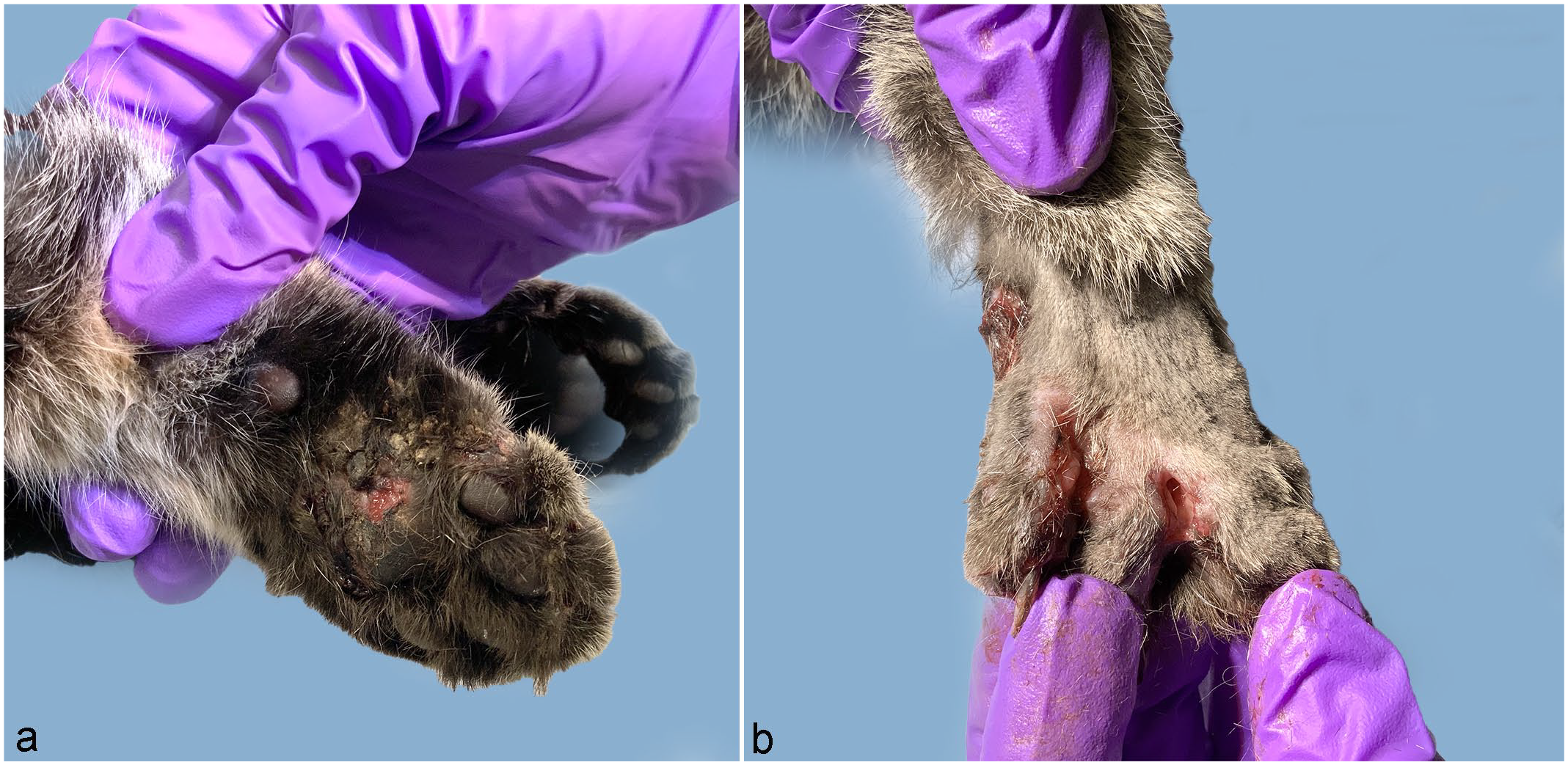
Pododermatitis, skin, cat. (a) Ulceration and crusting of the metacarpal pad. (b) Interdigital draining tracts were apparent following clipping of the fur.
Differential Diagnoses
An infectious pododermatitis and panniculitis was considered as the primary differential diagnosis given the physical examination findings, although no infectious agents were apparent on cytology and no infectious agents were identified on the previous biopsy with Gram and Fite-Faraco acid-fast stains. The etiologic differential diagnoses included fungal (eg, Sporothrix schenckii) and high-order bacterial (eg, Mycobacterium sp., Nocardia sp., and Actinomyces sp.) pododermatitis. Although both Actinomyces sp. and Nocardia sp. may induce pyogranulomatous inflammation, histologically, actinomycosis is often associated with rosette-like bacterial colonies (sulfur granules) surrounded by brightly eosinophilic, club-shaped material (Splendore-Hoeppli phenomenon).4,10 Nocardial infections may also, though less commonly, form sulfur granules and Splendore-Hoeppli material, but are characterized by beaded, branching bacterial filaments arranged individually or in tangled aggregates and shorter coccobacilli. As bacteria may be distributed singly, nocardiosis can be challenging to identify on hematoxylin and eosin (H&E)-stained sections. A modified acid-fast stain (Fite-Faraco) will stain Nocardia spp., but not Actinomyces sp., providing a distinguishing diagnostic feature. 4 In cases of feline atypical mycobacterial panniculitis, organisms are not typically numerous, although occasional nonstaining structures may be observed which are presumed to represent negative-staining mycobacteria.6,8 Acid-fast staining can improve identification of acid-fast bacilli intracellularly within macrophages or extracellularly in free lipid lakes. Finally, a noninfectious dermatitis/panniculitis could not be excluded given the chronic waxing and waning clinical course, persistence of clinical signs despite therapy, and lack of infectious agents apparent on the previous biopsy. 2 Neoplastic diseases such as lymphoma and feline progressive histiocytosis were considered less likely given the previous cytologic and histopathologic findings supportive of an inflammatory process.
Microscopic Findings
Four 6-mm punch biopsies were obtained from the right front and hind paws and submitted to our surgical biopsy service. Histologically, all tissue samples had pyogranulomatous pododermatitis and panniculitis with varying degrees of severity (Fig. 2a–c). All tissues had a dense, nodular to diffuse cellular inflammatory infiltrate that effaced the dermis, adnexa, and subcutis. The infiltrate was composed of numerous epithelioid macrophages, neutrophils, fewer lymphocytes and plasma cells, and rare mast cells and eosinophils. Occasionally, the infiltrate surrounded central regions of necrosis. Regions of inflammation were surrounded by fibroplasia with small amounts of hemorrhage, fibrin, and edema. No infectious etiologic agents were apparent in the H&E sections examined.
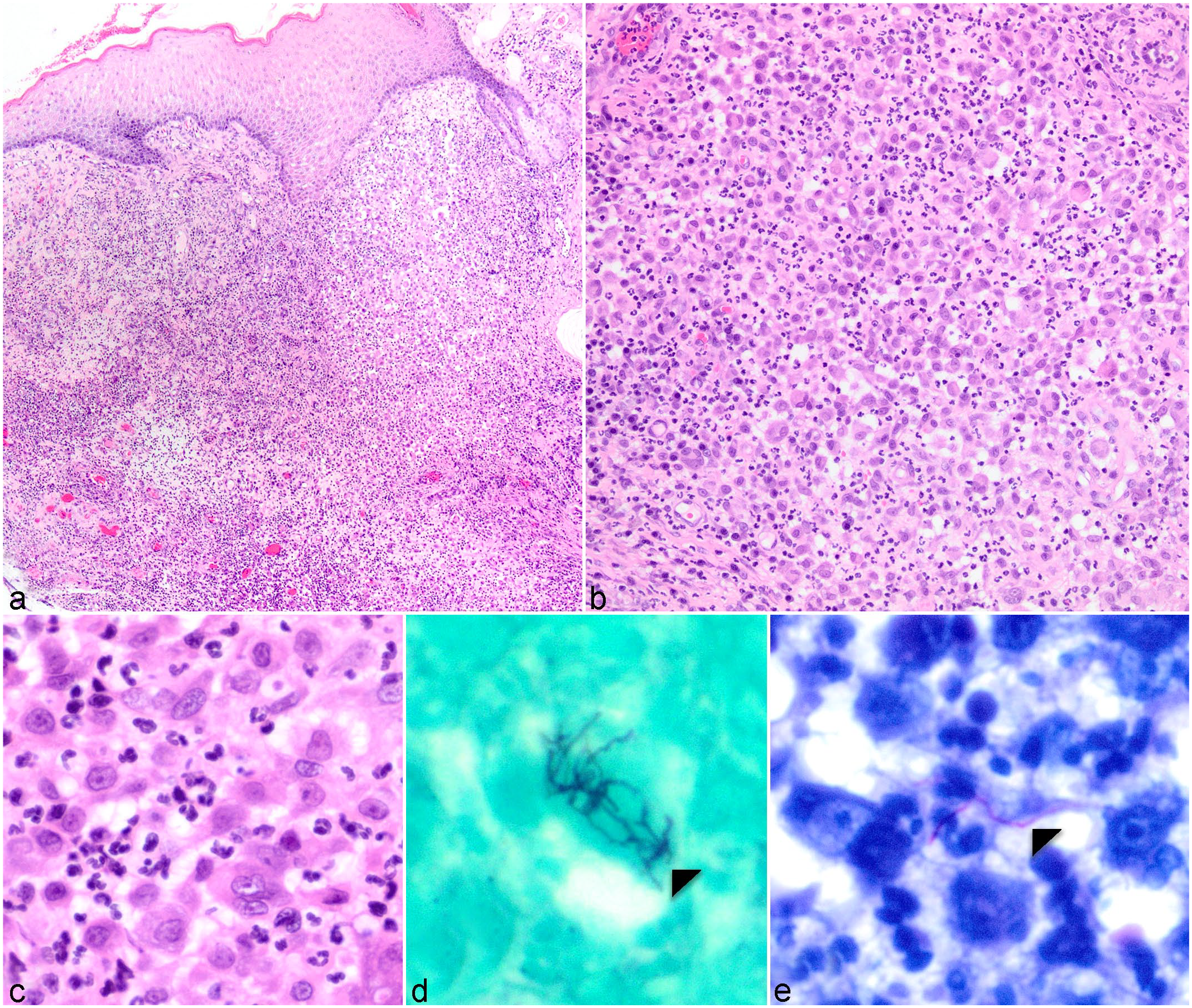
Digital haired skin, cat. (a–c) Nodular and coalescing pyogranulomatous inflammation effaces the dermis, adnexa, and subcutis. (d) Intralesional branching, filamentous bacteria were apparent with Grocott-Gomori’s methenamine stain. (e) Rare acid-fast-positive bacteria were apparent with Fite-Faraco acid-fast stain.
Further Investigations and Diagnosis
An infectious etiology remained the primary differential diagnosis despite no organisms being apparent in the H&E sections. From the dermatologists’ and pathologists’ perspectives alike, important ruleouts remained high-order bacteria such as Mycobacterium sp., Nocardia sp., and Actinomyces sp., as well as fungal infection. Noninfectious differential diagnoses were considered less likely in a cat but may include foreign body panniculitis and sterile inflammatory processes. Plasma cell pododermatitis of cats may contain a significant histiocytic and/or neutrophilic component, although the clinical presentation and histopathology were not supportive of this differential diagnosis.1,3
Bacterial culture (aerobic, anaerobic, and mycobacterial) on clinically obtained samples was submitted, and special stains (Gram stain, Grocott-Gomori’s methenamine stain [GMS], and Fite-Faraco acid-fast stain) were performed. Mycobacterial and anaerobic culture did not result in significant growth. There was growth of <1 mm white, dry colonies on an aerobic Clindamycin Blood Agar plate at 48 hours. Gram stain of these bacteria identified them as gram-positive short rods, and branching, short pink rods were apparent on modified acid-fast stain. Matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF) was performed from the subcultured plate and identified Nocardia nova with a 2.00 score. Similar findings were identified from thioglycolate enrichment broth and confirmed by MALDI-TOF. The GMS staining on histopathologic sections revealed numerous intralesional, predominantly extracellular filamentous bacteria forming branching aggregates (Fig. 2d), consistent with nocardial bacterial morphology. Rare filamentous bacteria stained acid-fast positive by Fite-Faraco acid-fast stain (Fig. 2e). No staining of the bacteria was apparent by Gram stain. The final diagnosis was nocardial pododermatitis.
Discussion
Nocardia spp. are branching, filamentous, aerobic bacteria known to be variably gram-positive, partially acid-fast, and GMS-positive.3,5,9 The genus is named after Edmond Nocard, a veterinarian who first described the causal agent of bovine farcy, a lymphangitis of the distal limbs of cattle, in 1888. Nocardia spp. belong to the order Actinomycetales, an order of organisms that possess a complex cell wall that is believed to facilitate intrahistiocytic survival and chronic infection by preventing phagosome-lysosome fusion and escaping the effects of the oxidative burst.9,10 These organisms are soil saprophytes and opportunistic pathogens. Infections can manifest as either local nonhealing wounds, often with draining tracts and deeper tissue/draining lymph node involvement, or disseminated systemic disease. Human cutaneous nocardiosis has been referred to as a “great imitator,” owing to the variable clinical presentation. 11 In humans, nocardiosis usually develops in association with a preexisting condition, such as impaired immunity or chronic debilitating disease. 12 There is some evidence in the veterinary literature suggesting that immunosuppression may be a predisposing factor. For example, concomitant FIV infection or immunosuppressive therapy has been associated with feline nocardiosis. 7
Nocardial infections can be transmitted to cats via inhalation of soil organisms resulting in pneumonia or via traumatic inoculation of wounds resulting in infection of the subcutis, thoracic cavity, and/or peritoneal cavity. 7 The most common syndromes in companion animals are dermatitis/panniculitis and pneumonia/pyothorax. In a review of 17 cases of feline nocardiosis, disease distribution in cases with cutaneous involvement was variable and included the trunk, limbs, face, eyes, and paws. 7 Several of these animals presented with a known history of a cat fight, and it has been hypothesized that the claws of cats may act as a mechanical vector responsible for subcutaneous inoculation of the bacteria. Disseminated disease (affecting 2 or more body systems) usually develops as initial pulmonary involvement with subsequent hematogenous spread. In general, the prognosis for feline nocardiosis is guarded, although patients with localized infections may respond to appropriate therapy. Delays in diagnosis and treatment tend to compromise response to therapy and disease may become chronic, progressive, and refractory, and insufficient duration of therapy may lead to disease recurrence.
Cats with nodular, fistulous, draining cutaneous and subcutaneous lesions—particularly those that do not respond to standard antimicrobial therapy—can pose a diagnostic challenge. In approaching these cases, differential diagnosis lists should include infectious etiologies such as high-order bacterial and fungal infections. Without additional diagnostic investigation, histopathology may not identify the cause, as these diseases may have highly similar histopathologic appearances and intralesional organisms may not be readily apparent on standard histopathologic stains. In this case, we were unable to visualize intralesional organisms initially or retrospectively in H&E sections despite the apparent abundance of bacteria identified on GMS. Therefore, histologic differentiation of the possible causative etiologic agent and definitive diagnosis require special stains, such as Gram, routine acid-fast (Ziehl-Neelsen), modified acid-fast (eg, Fite-Faraco), and/or silver (GMS) stains. Interestingly, the GMS stain was substantially more sensitive in the detection of intralesional bacteria on histopathology than either Fite-Faraco acid-fast (rare positive organisms) or Gram stain (no apparent staining). This might suggest that GMS is a sensitive screening stain to identify intralesional organisms while using additional diagnostic tests such as culture to further characterize the bacterium. In cases where organisms are few and characterization of the bacterial etiology cannot be made histologically, diagnosis relies on culture or polymerase chain reaction testing.
Pyogranulomatous dermatitis and panniculitis in domestic small animals are associated with a broad array of infectious and noninfectious causes, making diagnosis difficult and necessitating the use of ancillary diagnostic testing. 3 Diagnosis in this case was confounded by the previous biopsy yielding no evidence of infectious etiologies despite special stains. Therefore, the re-biopsy and pursuit of additional diagnostic testing (ie, bacterial culture) were crucial to achieving a definitive diagnosis in this case.
In summary, comprehensive diagnostic testing composed of bacterial culture and supportive histopathologic changes and special stains may be necessary to achieve a final diagnosis in cases of pyogranulomatous dermatitis and panniculitis.
Footnotes
Acknowledgements
The authors would like to thank the staff of the Ohio State University Comparative Pathology and Digital Imaging Shared Resource (particularly, Johanna Rawlings).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
