Abstract
Vascular mineralization is a hallmark of enzootic calcinosis. Histopathological, ultrastructural, and immunohistochemical investigations were performed on the external carotid arteries of seven sheep naturally poisoned by Nierembergia veitchii. Histologically, moderate to marked hyperplasia of the tunica intima was observed without mineralization. The tunica media exhibited mild to severe mineralization and osteochondroid metaplasia. Sheep with enzootic calcinosis showed arterial overexpression of osteopontin and tissue-nonspecific alkaline phosphatase and immunolabeling for osteonectin and osteocalcin in both intima and media layers of the tested arteries. The main ultrastructural finding in the tunica media was a marked phenotypic change of vascular smooth muscle cells from a contractile phenotype (VSMC-C) into a synthetic phenotype (VSMC-S). In the tunica media, VSMC-S produced matrix and extracellular vesicles, forming mineralizable granules associated with arterial mineralization. VSMC-S were also present in the tunica intima, but matrix and extracellular vesicles and mineralization were not observed. The absence of matrix and extracellular vesicles in the intimal hyperplasia, even in the presence of noncollagenous bone proteins, tissue-nonspecific alkaline phosphatase, and vitamin D receptors, reinforces the hypothesis that the presence of matrix and extracellular vesicles are crucial for the development of vascular mineralization in enzootic calcinosis. It is proposed that the two different VSMC-S phenotypes in calcinosis are due to the expression of at least two genetically different types of these cells induced by the action of 1,25(OH)2D3.
Keywords
Poisoning by calcinogenic plants, known as enzootic calcinosis, is considered the leading cause of arterial mineralization in livestock worldwide. 28 1,25(OH)2D3 (calcitriol) or its glycoside derivatives have been identified as the toxic components of some of these toxic plants. 28 Although enzootic calcinosis has been described for over 100 years, its pathogenic mechanism has not yet been entirely determined. 28
Arterial mineralization of both elastic and muscular arteries is a hallmark of enzootic calcinosis in ruminants. 28 Most reports of enzootic calcinosis in domestic herbivores showed mineralization of tunica intima and media of elastic arteries, which was associated with osteochondroid metaplasia in the tunica media since the early last century3,8,10,11,12,21,22,23,27,34,35,44,45 and in more recent literature.28,36,37 However, reports on enzootic calcinosis in muscular arteries are uncommon. 46 In muscular arteries, such as the external carotid artery, 30 mineralization is not observed in the intima; it is seen only in the media. In sheep poisoned by Nierembergia veitchii, the intima of external carotid arteries showed hyperplasia without mineralization, and the media was mineralized with large areas of osteochondroid metaplasia in the advanced stages of the disease. 46
The process of arterial mineralization in humans involves complex mechanisms not yet fully elucidated, and they have been widely studied for more than 200 years. Arterial mineralization comprises active and dynamic biomineralization events similar to those observed in the bone-forming process.17,48,49 Genetic diseases, hyperlipidemia, chronic kidney disease, diabetes, systemic lupus erythematosus, nicotine, warfarin, and vitamin D poisoning are associated with arterial mineralization in humans.39,48,49,52 Rabbits experimentally poisoned by Solanum glaucophyllum (syn. Solanum malacoxylon) had a marked reduction in contractile apparatus components and a substantial increase in synthetic organelles in aortic vascular smooth muscle cells (VSMC). 5 Later, these cells were designated contractile vascular smooth muscle cells (VSMC-C), which can switch to synthetic vascular smooth muscle cells phenotype (VSMC-S). 13 The role of VSMC in arterial mineralization has been demonstrated in different human vascular diseases.13,15 VSMC have dedifferentiation and phenotypic conversion/transdifferentiation properties, leading to cellular metaplasia and protein expression, and actively participate in the arterial mineralization process.13,29 An overview of the implications of VSMC in enzootic calcinosis in ruminants has recently been published. 28
Since knowledge on pathological mineralization processes in muscular arteries is limited, this study aimed to characterize histopathological and ultrastructural findings and the expression of noncollagenous bone proteins, tissue-nonspecific alkaline phosphatase, alpha-smooth muscle actin, and the vitamin D receptor in the external carotid arteries of sheep naturally poisoned by N. veitchii (enzootic calcinosis). In addition, we discuss the roles of these factors in the arterial mineralization process in poisoning by calcinogenic plants.
Materials and Methods
Animal Samples
The samples used are derived from a previous study. 46 A survey of enzootic calcinosis cases in sheep was conducted in the Veterinary Pathology Laboratory archives of the Federal University of Santa Maria, Rio Grande do Sul State, Brazil. Seven 3-year-old sheep (one male and six females) were selected. Five animals came from Caçapava do Sul (outbreak 1) (latitude: 30° 30′ 49.2″ S; longitude: 53° 28′ 59.5″ W) and two from São Gabriel (outbreak 2) (latitude: 30° 20′ 24.8″ S; longitude: 54° 19′ 19.4″ W) municipalities. Animals were raised in an extensive system in paddocks of approximately 200 hectares of native pasture invaded by N. veitchii. All cases were spontaneous without any control of the amount of plant consumption.
Necropsy of seven sheep was previously conducted during enzootic calcinosis outbreaks. Macroscopic and histologic lesions of the sheep included mineralization of the elastic and muscular arteries, kidneys, lungs, heart and cardiac valves, and muscular layer of the forestomachs, osteopetrosis, hyperplasia of thyroid C cells, and atrophy of the parathyroids. This study considered only samples of the external carotid artery collected near the larynx and fixed in phosphate-buffered 10% formalin. Samples were embedded in paraffin, sectioned at a thickness of 4 μm, and stained with hematoxylin and eosin for light microscopy evaluation.
Morphometric Analysis
The thickness of the carotid artery layers (1 section per animal) in both intoxicated (n = 7) and control (n = 4) sheep was determined using the ImagePro Plus (V3.6; Media Cybernetics, USA). The tunica intima and tunica media were delineated using the area of interest tool (AOI) in 3 different microscopic fields of the same artery for each animal. The Count/Size option determined the average thickness of those areas, resulting in 1 measurement for tunica intima and 1 measurement for tunica media per animal. Measurements of the intima and media layers thickness (µm) were determined, and intima/media ratios (%) were calculated. Mean thickness of each group (4 controls vs 7 intoxicated) was compared and statistically analyzed.
Immunohistochemistry
An immunohistochemical assay was conducted on the external carotid artery samples using the biotin-peroxidase-streptavidin method and revealed with: 3,3′-diaminobenzidine chromogen (ImmunoDetector DAB, HRP, BioSB Inc., Santa Barbara, CA, USA). Antigen retrieval was performed in a pressure cooker with citrate pH 6.0 solution for 3 minutes at 125°C. Primary antibodies were incubated overnight (Table 1). These antibodies have not been validated for use in sheep. Carotid arteries from 4 normal sheep were used as controls to compare the distribution and intensity of immunolabeling with those of the diseased arteries. Tissue sections incubated with nonimmune rabbit serum (1:200) with the omission of primary antibodies were used as negative controls. Immunolabeling of all sections was evaluated according to the following scores: –(absent), + (mild), ++ (moderate), and +++ (strong).
Methods used for immunohistochemistry.
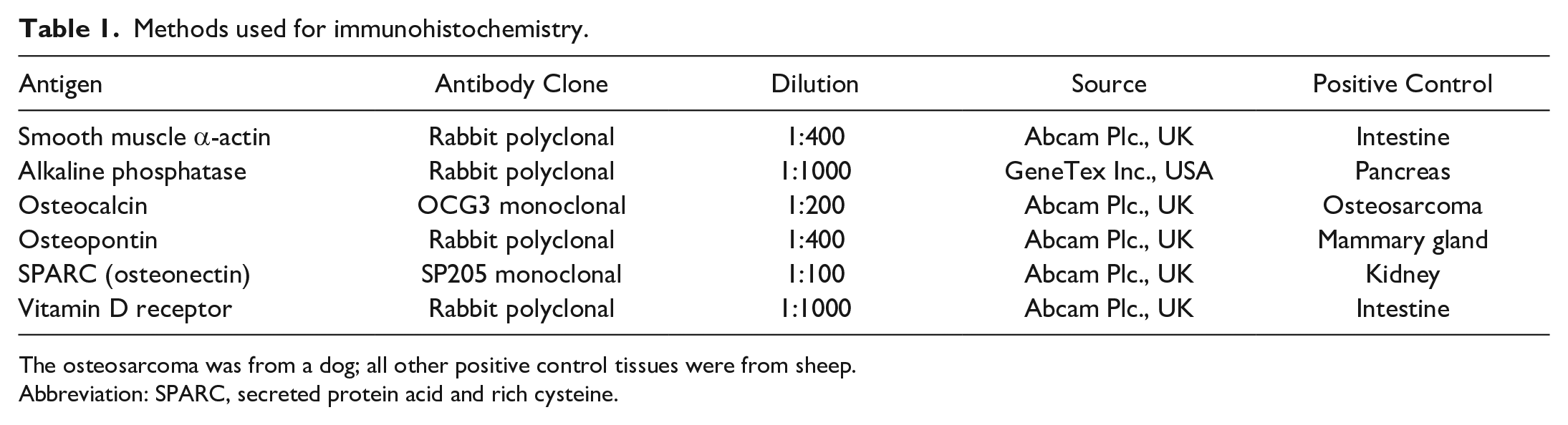
The osteosarcoma was from a dog; all other positive control tissues were from sheep.
Abbreviation: SPARC, secreted protein acid and rich cysteine.
Transmission Electron Microscopy and Ultrastructural Evaluation
Small samples of the carotid artery from 5 poisoned sheep were fixed in 2% glutaraldehyde and 2% paraformaldehyde diluted in sodium cacodylate buffer (0.03 M and pH 7.4), postfixed in 1% osmium tetroxide, dehydrated in ethanol, and included in Epon. Semithin sections were stained with methylene blue, while ultrathin sections were contrasted with uranyl acetate and lead citrate and examined under a transmission electron microscope.
Ultrastructural analysis was conducted to determine changes in vascular cell organelles and extracellular matrix related to N. veitchii poisoning. Arterial ultrastructural changes such as dedifferentiation, cell migration, and mineralization processes, matrix vesicles production and the release of extracellular vesicles, and other morphological changes related to phenotypical changes of VSMC were investigated.13,20,28,46
Statistical Analysis
The morphometric data of arteries and intensity of immunolabeling by each antibody in the arterial layers between groups were compared with the Mann-Whitney U test.
Results
Histopathology
All affected sheep showed moderate to severe non-mineralized intimal hyperplasia in carotid arteries (Figs. 1, 2). The tunica media presented extensive areas with mineralization characterized by variable interstitial deposits of basophilic aggregates (Fig. 1). Focally extensive osteochondroid metaplasia was also observed (Fig. 2).
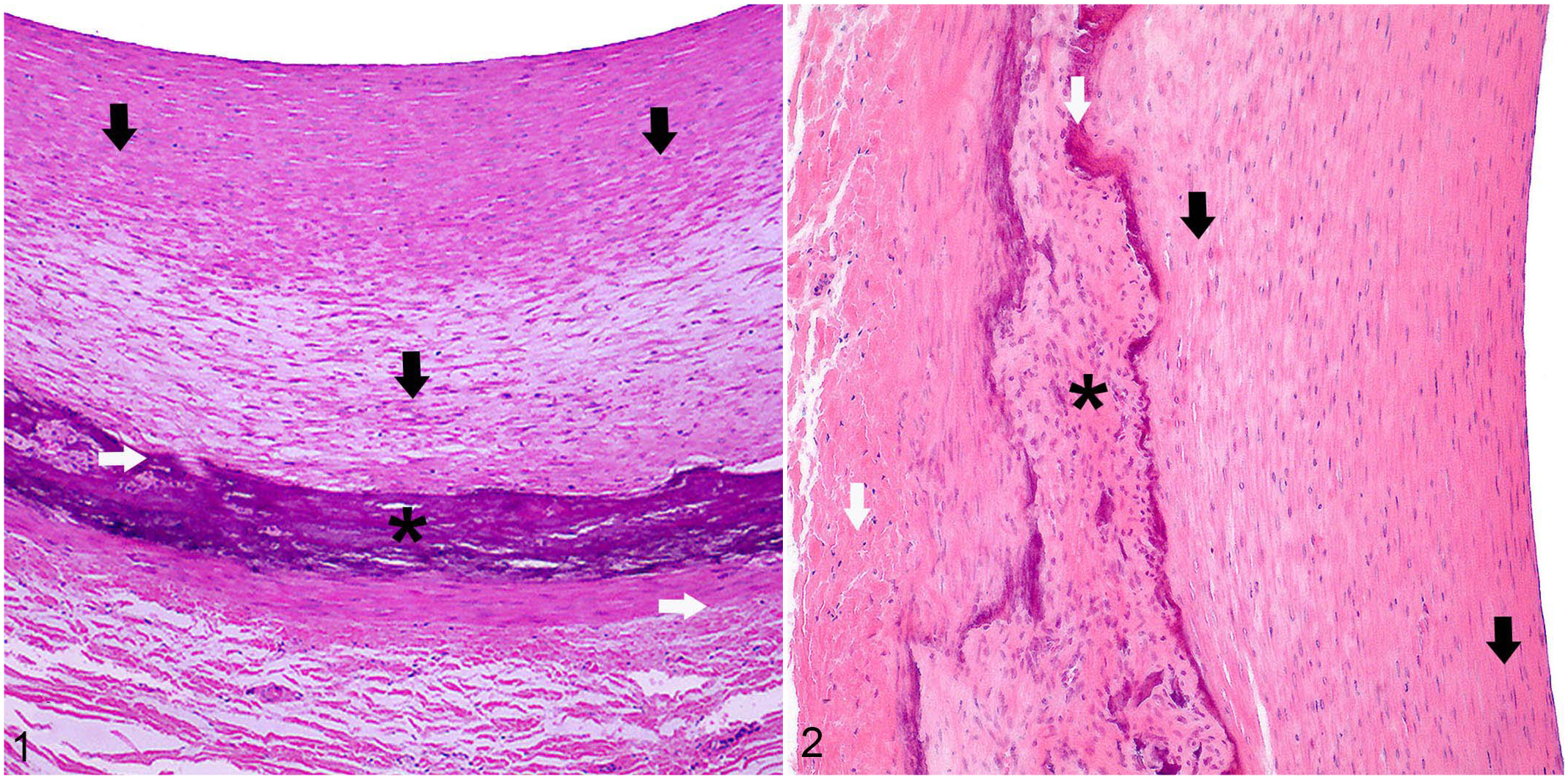
Spontaneous poisoning by Nierembergia veitchii, carotid artery, sheep. Hematoxylin and eosin (HE).
Morphometric Analysis
Carotid arteries from poisoned and control animals were compared by morphometric analysis. The most relevant findings were the increase in the tunica intima thickness of intoxicated animals (mean ± SD, 22.67 ± 3.7 µm in controls [n = 4] vs. 129.67 ± 68 µm in intoxicated animals [n = 7], P = .01) and the intima/media thickness ratio (0.05 ± 0.005 in controls vs. 0.32 ± 0.1 in intoxicated animals, P = .01). No differences were observed between groups in the thickness of the tunica media (498.66 ± 121.99 µm in controls vs. 379.17 ± 100.47 µm, P = .1).
Immunohistochemistry
The immunohistochemical evaluation of carotid arteries is summarized in Table 2. The most relevant differences (P < .01) were the strong immunolabeling of clusters of cells surrounding and within mineralized areas and arterial overexpression of tissue-nonspecific alkaline phosphatase (Fig. 3) and osteopontin (Fig. 4) in the tunica intima and media of poisoned animals compared with control animals. Osteocalcin (Fig. 5) and osteonectin (Fig. 6) were only immunolabeled in arterial tissues of animals with enzootic calcinosis and not in controls. Poisoned sheep also had marked osteocalcin immunolabeling of some clusters of cells in mineralized and injured arterial areas. In addition, sheep with enzootic calcinosis had a stronger (P < .003) α-smooth muscle actin immunolabeling in the intima (Fig. 7) as compared with control animals, but similar immunolabeling as controls in the other arterial layers (P > .05). Vitamin D receptor (Fig. 8) had similar immunolabeling intensities in both affected and control sheep (P > .05).
Immunohistochemistry results of the carotid arteries of sheep poisoned by Nierembergia veitchii (cases 1–7) and control sheep (C1–C4).
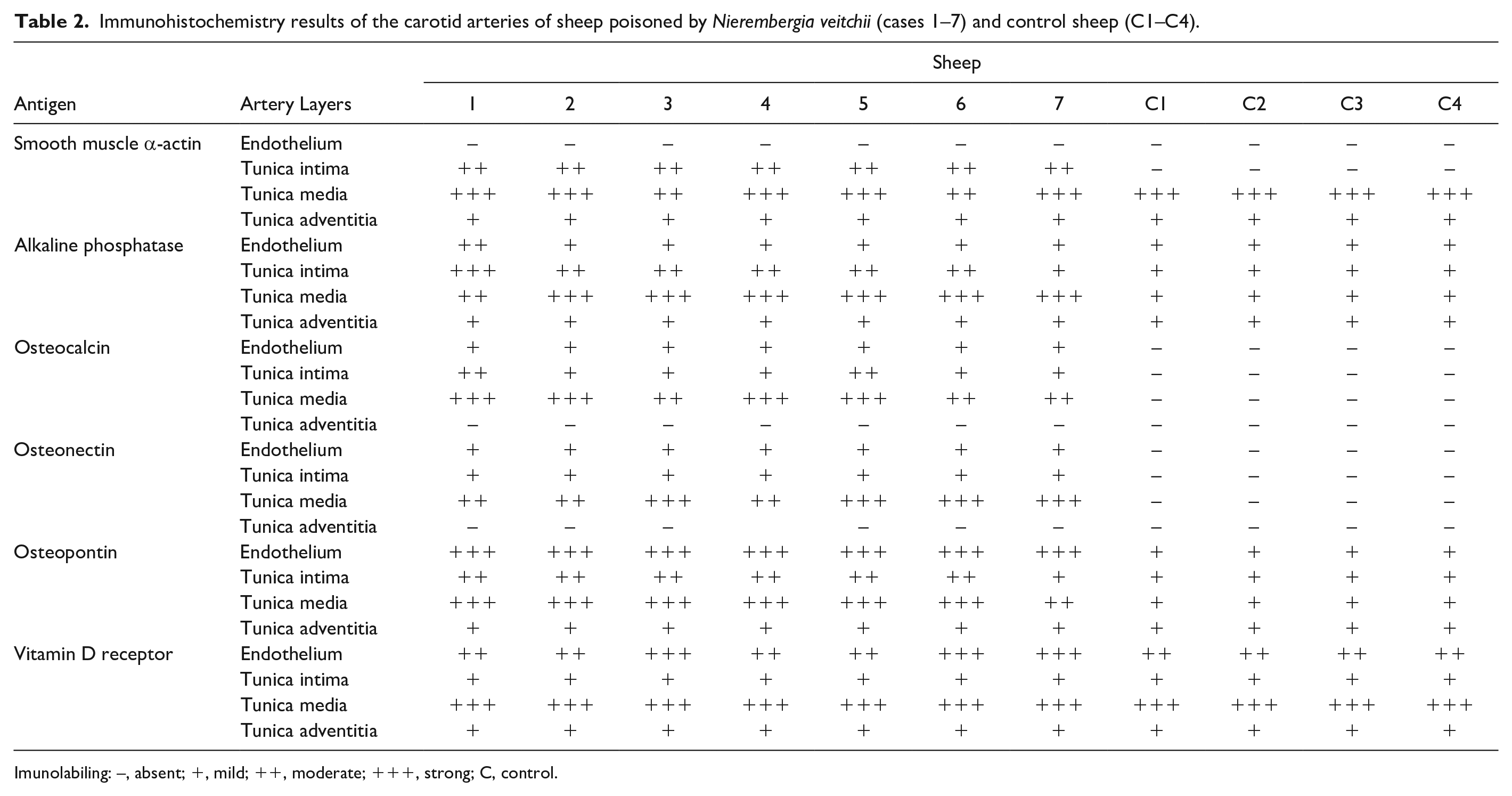
Imunolabiling: –, absent; +, mild; ++, moderate; +++, strong; C, control.
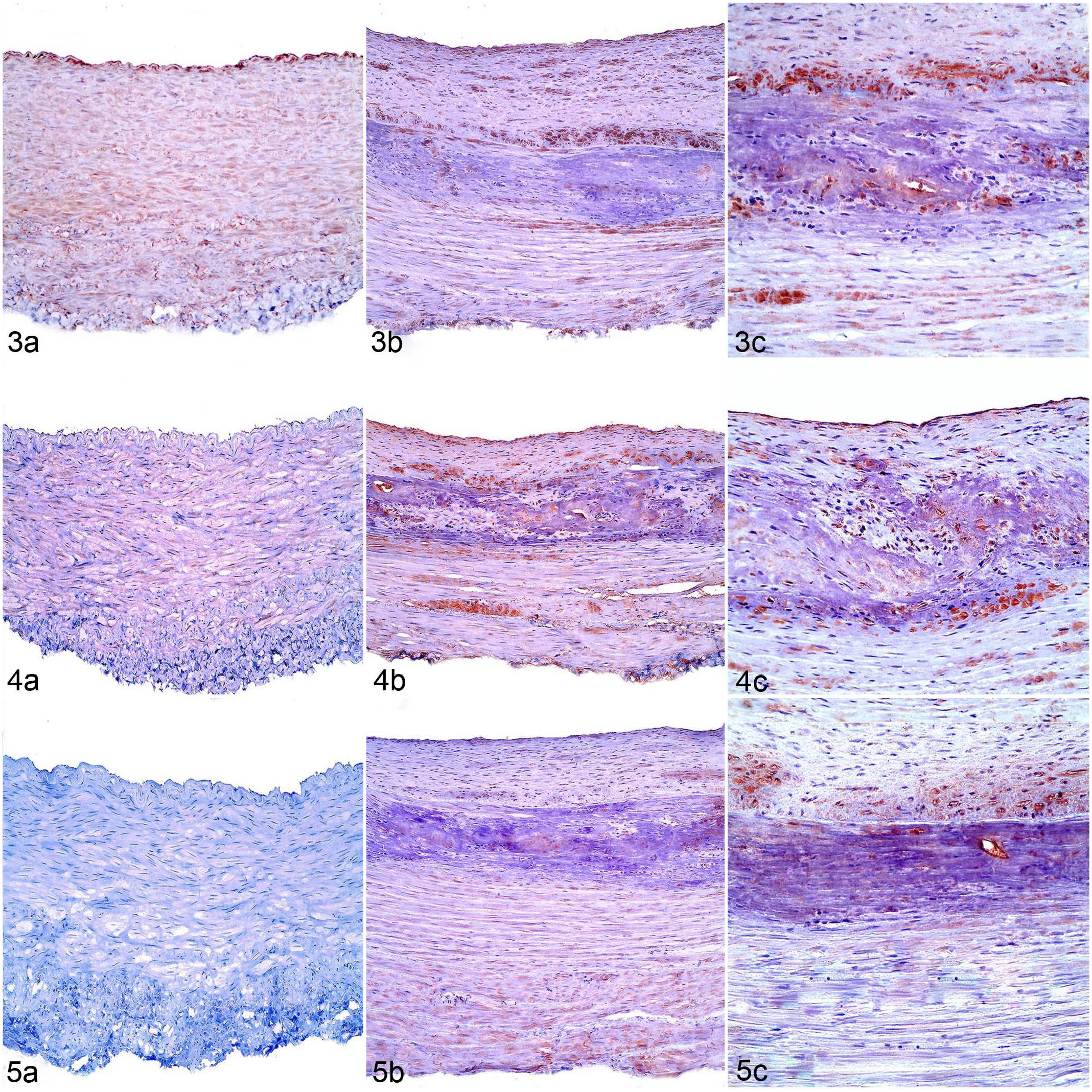
Control animal (a) or spontaneous Nierembergia veitchii toxicity (b, c), carotid arteries, sheep.
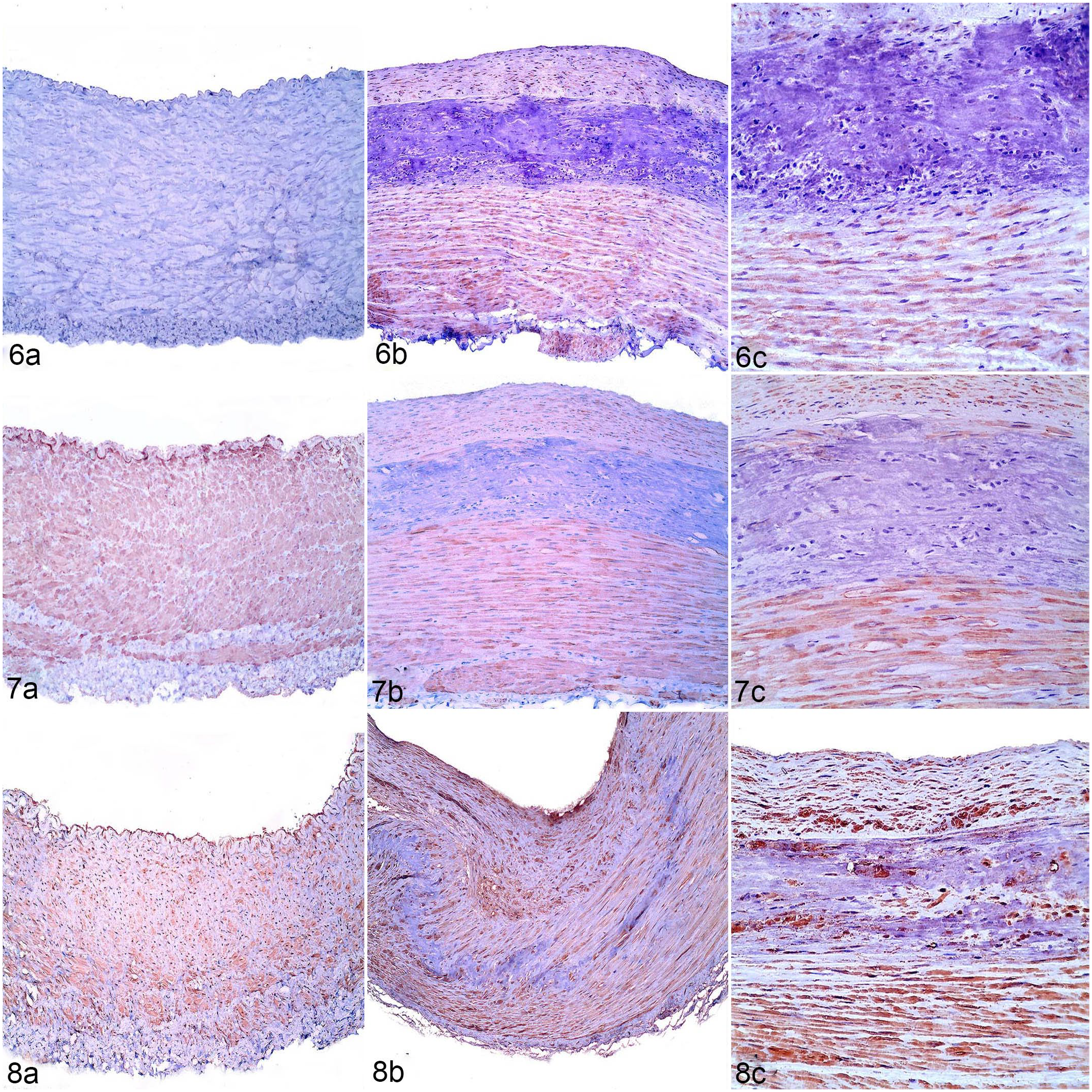
Control animal (a) or spontaneous Nierembergia veitchii toxicity (b, c), carotid arteries, sheep.
Transmission Electron Microscopy
Ultrastructurally, VSMC had evidence of different stages of dedifferentiation processes. Some cells retained their contractile fibril structure and had initial enlargement of the perinuclear space (Fig. 9). Other VSMC (interpreted to have undergone dedifferentiation) still had many contractile elements, while the basal lamina was tortuous or absent in some places. The perinuclear space was enlarged, and the rough endoplasmic reticulum (RER) exhibited slight proliferation. 46 In cells interpreted to be at an advanced stage of dedifferentiation to a synthetic phenotype, there was a pronounced decrease in contractile elements (Fig. 10) and a proliferation of the RER, with cisterns markedly dilated. The fragmentation and loss of portions of the basal lamina were observed, and there was formation of matrix vesicles interpreted as vesicle release from cells (Fig. 10). At this point, cells were classified as VSMC-S. 13 Some VSMC-S were present not only in the media but also the intima, suggesting that they may have migrated to the intima through fenestrae in the internal elastic lamina (IEL), and proliferated to form intimal hyperplasia (Fig. 11). Collagen and elastin were also observed within the interstitium of the hyperplastic intimal layer (Fig. 11). The VSMC-S did not show the release or extrusion of matrix and extracellular vesicles (Fig. 11). The remaining VSMC in the media apparently did not proliferate, but they did release matrix and extracellular vesicles that were mineralized. The interstitium of the media exhibited numerous microscopic mineralization nodules, perhaps formed by the mineralization of extracellular vesicles released by VSMC-S (Fig. 12). A few mineralization nodules originating from the media were observed on the surface of the IEL close to their fenestrae (Fig. 12).
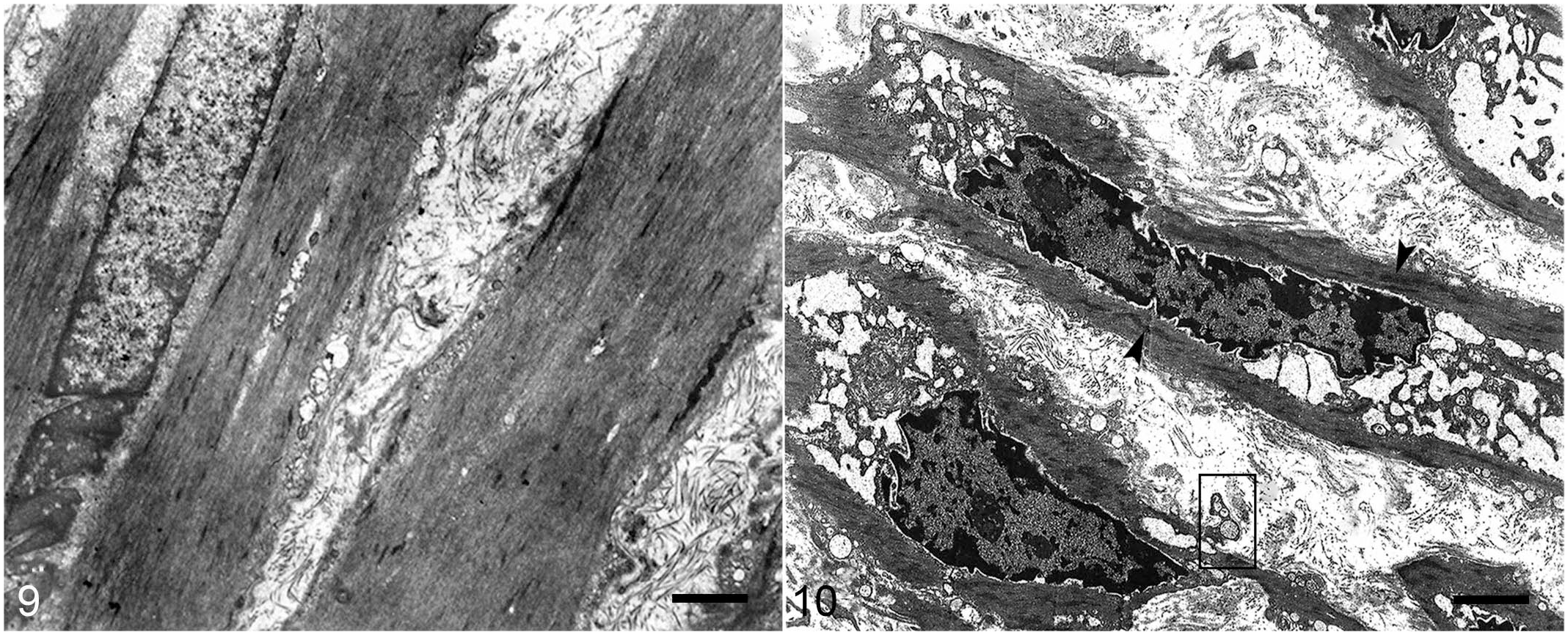
Spontaneous poisoning by Nierembergia veitchii, carotid artery, sheep. Transmission electron microscopy.

Spontaneous poisoning by Nierembergia veitchii, carotid artery, sheep.
Discussion
Vascular and soft tissue mineralization mechanisms involve interactions among VSMC, extracellular matrix components, and chemical signaling. 28 Non-mineralized intimal hyperplasia and medial mineralization associated with bone and cartilaginous metaplasia in the external carotid artery were the main microscopic features observed in this study and have been reported in muscular arteries of animals with enzootic calcinosis.3,12,28,33,35,36,37,38 The morphometric analysis and the positive immunolabeling for alpha-smooth muscle actin in the intima of carotid arteries from sheep with enzootic calcinosis had intimal hyperplasia with numerous VSMC-S, which were not observed in the control animals, suggesting a role of this cell type in the formation of this typical lesion in muscular arteries.
Osteopontin overexpression, observed in all poisoned animals, has been related to arterial mineralization and the stimulation of cell proliferation, migration, and adhesion. 15 Osteopontin is often overexpressed in vascular diseases 26 and it is involved in atherosclerosis and closely related to VSMC proliferation in vascular diseases in humans.9,15 The complex structure of osteopontin enables its interaction with electronegative glutamic and aspartic acid residues, serine threonine kinase substrate sites of proteins, and putative calcium-binding proteins that determine the ability of osteopontin to regulate the formation of apatite crystals. 44 In contrast, osteopontin has inhibitory effects on the arterial mineralization process in vitro.18,43,47
The multiple biological effects of osteopontin are an obstacle to correlating single observations in vivo experimental models. 41 The osteopontin overexpression observed in sheep poisoned by calcinogenic plants with extensive mineralization areas in the media suggests this protein’s association with VSMC proliferation and arterial mineralization and thickening.
In our study, osteocalcin, osteonectin, and osteopontin expression was detected in carotid arteries of animals with enzootic calcinosis and was mainly related to mineralized and injured areas suggesting their participation in the mineralization process. These noncollagenous bone proteins are related to the process of extraosseous mineralization, 38 as observed in arterial osteochondroid metaplasia and areas of mineralization in affected sheep.
These results are similar to those described in carotid arteries with atherosclerosis in humans, 6 the lungs of rabbits experimentally poisoned by Solanum glaucophyllum, 4 and sheep naturally poisoned by N. veitchii. 2 However, the expression of these noncollagenous bone proteins has not been reported in muscular arteries in the veterinary literature.
Our results also showed overexpression of tissue-nonspecific alkaline phosphatase. This finding strengthens the hypothesis that tissue-nonspecific alkaline phosphatase activated by VSMC acts by degrading extracellular pyrophosphate, creating biochemical conditions favorable for hydroxyapatite formation and tissue mineralization.7,31 Mineral deposition and the transdifferentiation of VSMC into osteogenic cells are mechanisms that are dependent on tissue-nonspecific alkaline phosphatase and are related to arterial mineralization. 13 Tissue-nonspecific alkaline phosphatase is ubiquitous in multiple tissues and is also expressed in all carotid layers of control sheep.25,32
The interaction of vitamin D receptors from VSMC with 1,25(OH)2D3 triggers the modulation of gene expression and transcription, as well as cell dedifferentiation, proliferation, migration, and transdifferentiation.14,50 Vitamin D receptor expression was identified in the endothelium and in all the vascular layers of the poisoned sheep studied, as reported previously in sheep with N. veitchii poisoning. 46
In our study, the arterial ultrastructural findings provided evidence of changes of the VSMC phenotype due to the increase in cellular secreting apparatus. In addition, the morphological changes also showed the role of matrix vesicles and the release of extracellular vesicles suggestive of exosomes by VSMC in the initial tunica media mineralization process. The arterial mineralization nidus initiated by matrix and extracellular vesicles, as observed in sheep with enzootic calcinosis, shares similarities with the mineralization process in bone tissues through chondrocytes and osteoblasts.32,40 VSMC can differentiate into a VSMC-S phenotype, osteoblasts, chondrocytes, adipocytes, and macrophages with proliferative, migratory, and extracellular matrix remodeling properties.13,42
Cellular events, such as phenotypic changes, proliferation, migration, by VSMC-S, observed in this study have also been observed in rabbits poisoned by Solanum glaucophyllum and in sheep with enzootic calcinosis caused by N. veitchii poisoning.5,46 A substantial increase of synthetic organelles and a substantial reduction of contractile elements in VSMC-S compared with VSMC-C were reported in sheep poisoned by N. veitchii. 46
Matrix vesicles detected in the tunica media of sheep with enzootic calcinosis are reported in the literature as extracellular bodies surrounded by a polarized membrane.20,51 VSMC-S produce matrix vesicles by selecting membrane proteins and lipids with an affinity for biomineralization. 1 Extracellular biomineralization is initiated in the matrix and extracellular vesicles released by VSMC-S, and occurs in two phases in both physiological and pathological mineralization.1,19,20,24 The first phase comprises early hydroxyapatite crystal development within the matrix and extracellular vesicles and phosphate release through calcium-binding proteins and phospholipids, mainly by alkaline phosphatase activity. High levels of these elements exceed solubility properties, enabling hydroxyapatite deposition on the vesicle’s internal surface. In the second phase, hydroxyapatite nucleation, deposition, and increased biosynthesis of larger crystals occur, which occupy the extracellular space. 1 The homeostatic levels of calcium and phosphate in the extracellular fluid are sufficient to promote hydroxyapatite crystal development. 16 The mineralized nodules observed on the surface of the IEL were previously reported, 46 and possibly are mineralized extracellular vesicles released and mechanically carried by VSMC-S from the media layer during cell migration to the intima. The prior literature suggests that the extracellular vesicles observed in the current study are exosomes, which are known to have a role in biomineralization.
Analyzing the set of histopathological, immunohistochemical, and ultrastructural findings in sheep with enzootic calcinosis, we detected marked changes in the media and intima characterized by the shift of VSMC-C into VSMC-S, overexpression of noncollagenous bone proteins and tissue-nonspecific alkaline phosphatase, and mineralization in the media related to the presence of matrix and extracellular vesicles. From this perspective, we hypothesize that the absence of matrix and extracellular vesicles in the hyperplastic intima explains the lack of local mineralization even in the presence of noncollagenous bone proteins and tissue-nonspecific alkaline phosphatase expression. The ultrastructural findings suggest the role of matrix and extracellular vesicles (suggestive of exosomes) in vascular mineralization in sheep with enzootic calcinosis.
In enzootic calcinosis, differences in the arterial injury between tunica intima and the tunica media of muscular arteries are possibly related to two distinct behavioral phenotypes of the VSMC-S, as follows. One type, after dedifferentiation, appears to migrate from the media to the intima, where it undergoes hyperplasia but does not release matrix and extracellular vesicles, and does not synthesize a mineralizable extracellular matrix or undergo osteochondroid metaplasia. The second type of VSMC-S remains in the tunica media, releasing matrix and extracellular vesicles, producing a mineralizable extracellular matrix, which later undergoes osteochondroid metaplasia. The plasticity of VSMC phenotypes is likely related to genetic factors. 13 Genetic mapping studies are needed to clarify phenotypic drives.
The results of this study, in association with those from previous studies performed by our research group, suggest the following complex mineralization process caused by calcinogenic plants: the toxic principle binds to vitamin D receptors of VSMC and promotes morphological changes via the expression of unknown genes.2,4,5,27,46 The toxic principle triggers cell phenotype conversion events, such as dedifferentiation, transdifferentiation, and osteochondroid metaplasia of VSMC; production of matrix and extracellular vesicles; and expression of noncollagenous bone proteins and tissue-nonspecific alkaline phosphatase, resulting in the medial mineralization of muscular arteries. Further studies are required to elucidate the complex pathogenesis of arterial mineralization completely.
Footnotes
Acknowledgements
Special thanks to Dr. José Augusto Bastos Afonso da Silva (Federal Rural University of Pernambuco—Campus Garanhus) for providing negative control of sheep carotid arteries amid the SARS-Cov-2 pandemic, to Cintia Queiroz (Federal University of Minas Gerais) for her support in the writing of this article, and to Isabel Luana de Macêdo (University of Brasilia) for her technical assistance with immunohistochemistry.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by National Institute of Agricultural Research (INIA) of Uruguay (Project CL 44), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Brazil, (Finance Code 001) and National University of La Plata (Project V270). A postgraduate scholarship was provided to Mizael Machado by INIA, Uruguay.
