Abstract
Oral melanoma (OM) is a highly aggressive tumor of the oral cavity in humans and dogs. Here we review the phenotypic similarities between the disease in these 2 species as the basis for the view that canine OM is a good model for the corresponding human disease. Utility of the “canine model” has likely been hindered by a paucity of information about the extent of the molecular genetic similarities between human and canine OMs. Current knowledge of the somatic alterations that underpin human tumorigenesis and metastatic progression is relatively limited, primarily due to the rarity of the disease in humans and consequent lack of opportunity for large-scale molecular analysis. The molecular genetic comparisons between human and canine OMs that have been completed indicate some overlap between the somatic mutation profiles of canine OMs and a subset of human OMs. However, further comparative studies featuring, in particular, larger numbers of human OMs are required to provide substantive evidence that canine OMs share mechanisms of tumorigenesis with at least a subset of human OMs. Future molecular genetic investigations of both human and canine OMs should investigate how primary tumors develop a metastatic gene expression signature and the genetic and epigenetic alterations specific to metastatic sites. Such studies may identify genetic alterations and pathways specific to the metastatic disease which could be targetable by new drugs.
Human OMs are frequently considered part of the larger group of “mucosal melanomas” together with melanomas arising from the anogenital regions, oropharynx, paranasal sinuses, conjunctiva, esophagus, and middle ear. 76 Mucosal melanomas are a rare clinical entity, with a reported incidence of 800 cases per year in the United States (in contrast to cutaneous melanomas which are 100 times more common). 102 Head and neck sites represent 55% of cases, with a quarter arising within the oral cavity. 102 Human OMs are clinically characterized by a high fatality rate due to early metastatic spread to regional and distant sites. Canine OM shares the same deadly clinical course as well as histopathologic features and treatment, but in contrast with the human counterpart, it represents the most common oral malignancy and form of canine melanoma. 28 Therefore, the dog has been considered for a long time to be a good preclinical model for human OMs in several aspects of the disease, with relatively large cohorts of tissue samples available for molecular genetic investigations. This article reviews the current knowledge on driver genetic alterations shared between canine and human OMs based on genomic and transcriptomic analyses, and critically points out the concomitant lack of comparative studies focusing on genetic and epigenetic alterations responsible for tumor progression toward the metastatic spread in the 2 species, which represents the most common cause of OM-related deaths.
Why Is Canine OM a Better Preclinical Model for Human OM Than Other Animal Models?
As is the case for most cancers, mouse models are regarded as the “gold standard” for studies investigating melanoma development, progression, and therapeutic response, given the genetic homology between humans and mice, and the ease of mouse breeding. 99 Syngeneic mouse models (typically in C57BL/6J mice 73 ), spontaneous, carcinogen-induced or transgenic cell lines, human patient-derived tumor xenograft models, 21 and knock-in (eg, BRAF V600E 42 ) or knock-out (eg, PTEN 16 ) genetically engineered models have provided powerful tools for melanoma research and contribute immeasurably to the understanding of melanoma biology and accelerated therapeutics entering clinical trials. However, there are concerns as to whether mouse xenograft models of human melanoma in immunodeficient mice accurately recreate the human tumor microenvironment, 36,83 as well as the limitations that genetically engineered murine models lack the genetic heterogeneity of the spontaneous human disease, and have lower levels of genetic instability. 77,92 Such deficits, coupled with the absence of the natural steps of cancer development and progression in mouse models, may explain why only 5% of anticancer drugs are translated from murine preclinical models to treatments with clinical utility in humans. 43 Therefore, there is a clear and unmet need for models that can recreate the mechanisms of human tumor formation and progression, including the role of the tumor microenvironment.
To address this need, there has been steady growth of comparative oncology initiatives over the last 30 years, including the study of spontaneous cancers in dogs. Dogs with naturally occurring cancers are considered to be a better model for equivalent human cancers than artificially created rodent models. The value of the “canine model” to increasing understanding of human tumor development and progression is exemplified by comparative human–canine tumor genome analyses that have expedited the identification of clinically relevant human tumor molecular subtypes and genes associated with metastatic progression. 68,84 Dogs display a greater degree of genetic homology with humans than do mice, 52 they are immune competent hosts with immunoedited tumor microenvironments, and significantly they live in a shared environment with humans and so are exposed to many of the same environmental factors. Tumor initiation and progression in dogs is spontaneous and mimics that in humans. 54 As in humans, dogs are affected by the spontaneously occurring and highly aggressive OM, which metastasizes commonly to regional lymph nodes and distant organs, mirroring the clinical course of human disease. 90 OM has comparable anatomical and histological presentation in both species and the identification of similar somatic mutations and dysregulated cell signaling pathways suggests that canine OM may be able to reproduce the biological and clinical complexity of human OM. 87,100 Nevertheless, there is currently no effective treatment for OM metastatic disease in either humans or dogs, and OM has a very poor prognosis in both species.
Clinical Presentation and Histopathologic Features of Canine and Human OM
Oral melanoma arises from malignant transformation of neural crest-derived melanocytes that reside within the oral mucosal epithelium. 62 In contrast to human cutaneous melanomas, for which ultraviolet (UV) light exposure is a significant etiological factor, 44 OM is a highly aggressive non-UV-driven disease in both humans and dogs, exhibiting rapid growth and invasion. 28 There is an indication of inherited susceptibility in both species. There is a higher incidence of OM in dog breeds such as Cocker Spaniel, Miniature Poodle, Anatolian Sheepdog, Gordon Setter, Chow Chow, and Golden Retriever, 18 and a higher incidence of human OM within the Japanese population (accounting for 7.5% of all melanomas), and a significantly lower incidence in Caucasians. 3 In both species, OM is invariably fatal due to a high metastatic propensity and the recalcitrance of tumor metastases to conventional treatments (see below). A third of human cases present with lymph node metastasis at diagnosis and the lungs are one of the most reported sites of early distant metastasis. 93 Similarly, the tumor metastasizes to regional lymph nodes and the lungs in 75% of canine cases. 103 This results in a postresection 5-year survival of only 25% to 33% in humans, and less than 20% of dogs survive 1 year after surgery. 70,102,103 In both species, OM commonly presents in the gingiva and hard palate, as brown-black macules or nodules sometimes with ulceration. 79,102,105 However, a proportion of tumors (30% of all canine melanoma cases 67 and 50% of head and neck mucosal melanomas in humans 102 ) are amelanotic, which may contribute to their late diagnosis. Immunohistochemistry can be used to detect specific melanocytic markers to help in the diagnosis of amelanotic tumors. In particular, S100 protein, melan A, tyrosinase, and HMB45 are the most frequently used markers for the diagnosis of human OMs as they react in the majority of melanocytic tumors. 102 In canine OMs, a “cocktail” containing melan A, PNL-2, and tyrosinase-related proteins 1 and 2 provides the highest diagnostic sensitivity, whereas HMB45 and tyrosinase have very low sensitivity. 63 The majority of human OMs arise de novo from normal mucosa, although around a third of cases are thought to develop from preexisting melanocytic lesions like atypical melanocytic hyperplasia, which is characterized by equivocal histopathologic features like hyperchromatic and angulated nuclei with rare mitosis. 40 Such lesions may be dormant for several years before vertical invasion, and similar histological growth patterns have been noted in dogs. 39,87 Interestingly, there is a considerable difference between the relative incidence of OM in dogs and humans. In dogs, OM is both the most common oral malignancy and the most common form of melanoma, 28 and accounts for around 7% of all canine cancers. 63,103 In contrast, human OM accounts for only 0.5% to 0.7% of oral malignancies, and less than 7% of all melanomas. 70,102
Histologically, the disease presents comparably in dogs and humans, with tumors growing in sheets, nests, or bundles, frequently with an associated in situ/intraepithelial component. Both species have spindle, epithelioid, plasmacytoid, or mixed cell cytomorphologic subtypes, with rarer whorled/dendritic, balloon cell, signet-ring cell, clear cell, and adenoid/papillary cell subtypes in dogs. 4,63 Some putative prognostic histological features are invasion of surgical margins by tumor cells, the presence of vascular invasion, high mitotic count (>10 mitoses in 10 high-power fields), and ulceration, which are associated with a worse prognosis in human disease. Negative prognostic histopathologic features reported for canine OM are nuclear atypia (≥30% of atypical nuclei in 200 cells counted), mitotic count (≥4/10 high-power fields), the presence of lymphatic invasion, and a Ki67 index ≥19.5 (where Ki67 index is defined as the average number of positive nuclei in 5 areas of a 1 mm2 optical grid at 400×). 7,59,69,71,86,91 However, given the rarity of human OM and a general lack of appropriate follow-up data for canine OM cases, there is no fully validated histological prognostic biomarker that is able to evaluate and inform on treatment options for this disease.
Human melanoma of the head and neck can be staged according to the American Joint Committee on Cancer Staging system (AJCC) 8th edition, where tumor staging begins at T3 (tumor limited to mucosa and submucosa) with worse prognosis with increasing stage. 20 T1 (in situ) and T2 (between 1 and 2 mm thick) are omitted because of the innate aggressiveness of the disease. Other prognostically relevant staging systems for mucosal melanomas of the head and neck have often focused on the depth of infiltration. Which staging system is superior remains still controversial. 10,74,86 Historically, canine OM is staged according to the World Health Organization (WHO) tumor, node, metastasis (TNM) system, but this system is largely based on primary tumor size, which has not been proven to be prognostically significant, and therefore its use remains uncertain. 32 Given the inadequate staging systems, there is a need for improved prognostic markers for OM.
Treatment of Canine and Human OM
Surgical excision of oral melanomas remains the cornerstone of treatment for human disease and the achievement of clear margins following radical surgery provides the best outcome. 47,71 Although melanomas display a low radiosensitivity, radiotherapy is increasingly used to accomplish local control, but no benefit with respect to overall survival has been demonstrated. 81 Interferon-α2b adjuvant therapy is commonly used despite clinical trials including only a handful of OM cases, and there is evidence that it may be less effective than traditional chemotherapy (combining temozolomide and cisplatin). 46,50 A plethora of new targeted therapies, using inhibitors to mutated BRAF, MEK, and KIT, and immunotherapy targeting cytotoxic T lymphocyte associated antigen-4 (CTLA-4), programmed death-1 (PD-1) and ligand (PD-L1), have been trialed for metastatic cutaneous melanoma. However, mucosal melanoma is commonly excluded from trials due to its extremely low incidence and survival rates. 60
As for human disease, the most common treatment for canine OM includes surgery with wide excision, although the recurrence rate even when clean excision margins are achieved is still 3.2% to 10%. 8,22,94 Radiotherapy is often used alone for unresectable tumors or as an adjunct for control of locoregional disease. 75 Although not common practice, lymphadenectomy of both regional mandibular and medial retropharyngeal lymph nodes may provide better staging information given the complexity of lymphatic drainage and the presence of multiple lymphocenters in the canine head, and the propensity of canine OM to metastasize contralaterally to the primary tumor. 38,89 Interestingly, 40% of dogs with normal-sized lymph nodes have cytologic or histologic evidence of metastatic disease, 101 but the overall sensitivity of fine needle aspiration cytology for detecting nodal metastatic disease in canine OM is low, ranging from 63% to 78%. 8,24,101 In human melanomas, combined ultrasound and fine needle aspiration cytology (after identification of sentinel nodes by lymphoscintigraphy) are good indicators of sentinel lymph nodes metastasis, with sensitivity >80%, 97 and this approach potentially avoids the surgical procedure of sentinel lymph node biopsy. Chemotherapy alone, or as adjuvant treatment with platinum agents (mainly carboplatin), has failed to prove any significant survival advantage compared to local treatment alone, although a few studies have demonstrated an increased time to tumor progression when using chemotherapy in combination with radiotherapy. 8,12,26 The treatment of canine OM cases with targeted therapy and immunotherapy, as used to treat human disease, is in its infancy despite evidence of shared pathway activation and tumor antigens (eg, disruption of MAPK and PI3K/AKT pathways). 25,100 Immunotherapy in dogs is hindered by incomplete knowledge of the canine immune system and the ability of dogs to mount an immune response against humanized monoclonal antibodies. However, targeting and modulating the canine immune system by checkpoint blockade, adoptive cell transfer, or cancer vaccines is an attractive prospect. 37 Presently, there is limited characterization of the PD-1 checkpoint in canine melanoma in therapeutic targeting. However, dogs with advanced cancer often exhibit defective antitumor immunity and T cell exhaustion, 37 and PD-1 and PD-L1 genes are highly conserved among canine breeds. 55 There is limited evidence that PD-L1 might be expressed by the majority of canine oral melanomas and PD-1 is highly expressed on tumor-infiltrating lymphocytes, suggesting a rationale for PD-1/PD-L1 inhibitors as novel therapeutic agents for canine oral melanoma. 56 Adoptive cell therapy involving the targeting of the melanoma differentiation antigen, human glycoprotein 100 (hgp100), has been assessed in dogs using allogeneic dendritic cell and whole tumor cell vaccines expressing hgp100. 1,31 Furthermore, immunization of patients with abnormally expressed immunogenic tumor antigens is an immunotherapy yielding high tumor versus normal cell specificity and clinical efficacy. 41 Although originally aimed at informing human trials, a DNA vaccine containing xenogeneic plasmid DNA encoding the human tyrosinase gene (ie, integral for melanin synthesis), was demonstrated to be safe in a phase I trial involving dogs with stage II/III oral melanoma, and able to induce tyrosinase-specific antibody responses, and improve survival. 6,51 In 2007, this led to approval by the US Food and Drug Administration and US Department of Agriculture of the Oncept DNA vaccine expressing xenogeneic human tyrosinase for veterinary use in dogs with stage II/III oral melanoma after surgical control of disease. Oncept is still the only approved but still debated therapeutic for canine OM and the only therapeutic vaccine approved in both species.
Molecular Mechanisms of Human and Canine OM Tumorigenesis
Despite a long-standing debate regarding whether the origin of any tumor is a single normal cell or multiple initiating cells, the existence of a shared set of truncal mutations in neoplastic cells is compelling evidence for a common evolutionary origin from a single cell. 17 Truncal mutations are in the trunk of the phylogenetic tree that are shared by all neoplastic clones. 107 Genetic alterations responsible for cancer tumorigenesis are defined as driver mutations. Driver mutations confer a fitness advantage on the neoplastic cells, compared to passenger mutations which do not have any effect on fitness. 17 Driver mutations are usually truncal events, accumulating over time in tumor cells according to the branching evolution model (where clones are believed to diverge from a common ancestor and evolve in parallel in the tumor mass). 17 Two types of “limb” pathways can arise from the main trunk: events that induce or accelerate metastasis and events that induce resistance to therapy. 11
The application of next-generation sequencing methods has revealed that genetic alterations in canine OM resemble those of human mucosal melanoma, with both diseases having a high copy number and low single nucleotide mutations reflective of non-UV-driven initiating molecular events. 27,35,104
Genomics of Primary OM
Common initiating events in human mucosal melanoma include loss of heterozygosity in TP53, a tumor suppressor gene encoding for the p53 protein which regulates cell cycle progression, DNA repair, cellular senescence, and apoptosis. 30 When functional, the p53 protein accumulates in cells with DNA damage, arrests the cell cycle, and allows time for repair, inducing apoptosis when the damage is too great. 96 Mutations of TP53 are the most frequently reported in human cancers, 96 and loss-of-function mutations have been reported in human mucosal melanomas. 34,104 In a recent comparative exome sequencing study, 104 15% of human mucosal melanoma cases (and 2 of 4 OM cases) were characterized by a total of 8 different TP53 mutations. TP53 loss-of-function mutations result in the accumulation of DNA damage, with consequent genetic instability that promotes the accumulation of further genetic and epigenetic alterations. 96 Similarly, TP53 gene mutations have been reported in 8 - 13% of canine OMs. 104
MDM2 is a proto-oncogene, able to inactivate p53 binding to its N-terminal transactivation domain or inducing proteasome-mediated degradation. 98 Amplification of MDM2, with significant copy number gain, has been demonstrated in both human and canine mucosal tumors. 34,35,65 Consequently, it is possible in both canine and human OMs that silencing of the protective action of p53 pathway against tumor development is obtained by MDM2 gene amplification rather than through TP53 mutation or loss-of-function.
NRAS is a proto-oncogene belonging to the family of RAS genes, commonly activated by point mutations that results in constitutive activation of the NRAS protein with intrinsic GTPase activity and subsequent hyperactivation of downstream mitogen-activated protein kinase growth-related pathways. 58 Somatic mutations of NRAS have been reported in canine OMs, at position Q61, matching the most frequent somatic alteration found in NRAS-mutated melanomas in humans. 28
PTEN is a tumor suppressor gene encoding a protein (PTEN) that acts as a brake on the PI3K/AKT pathway with the consequent regulation of cell growth, survival, and migration. 96 Human and canine mucosal melanomas both have disruption of PTEN by nonsense or frameshift mutations, causing a loss of gene function followed by uncontrolled neoplastic cell proliferation. 100 However, diminished PTEN expression has not been correlated with reduced survival. 104
The gene c-Kit encodes the type III transmembrane tyrosine kinase KIT protein (CD117), and oncogenic mutations in the autoinhibitory intracellular juxtamembrane domain activate and recruit many downstream effectors, including MAPK and PI3K/AKT, known to promote cancer development. 48 Mutations in c-Kit frequently occur in human oral melanoma and may occur in 16% of metastatic oral mucosal melanomas. Overall survival of cases with c-Kit mutation appears to be shorter compared to wild-type cases. 5,15,53 However, there has been discord between the presence of KIT and activating c-Kit mutations, with only c-Kit activating mutations being predictive of response to KIT inhibitors. 95,106 Likewise, although variable c-KIT receptor expression has been documented in canine melanomas, its presence and implications on survival is not fully established as well as for activating mutations in c-KIT. 14,64,66
BRAF V600E mutations are the most common mutation, found in up to 60% of human cutaneous melanomas or melanocytic nevi, but they are only rarely present (<5%) in their mucosal counterpart, 19 and in canine OMs (<4%). 61 The rarity of BRAF mutations, together with a lack of UV mutational signatures in NRAS or PTEN, in addition to the anatomical location of human and canine OMs, all suggest non-UV-driven pathways for their development and progression. 28
Although established driver genes for human OM have been shown in a recent comparative exome sequencing study 104 to be mutated in canine OMs, the same study found that the 65 primary canine OMs analyzed lacked mutations in a small number of genes (SF3B1, GNAQ, POLE, PRTD, and DMX1.2) recognized as drivers for a subset of human mucosal melanomas. Conflicting data are also reported for other genes potentially implicated in the development of canine and human OMs. For example, PTPRJ (a putative tumor suppressor gene) has been reported to carry a truncating mutation in only 3% of canine OMs 100 but inactivation of this gene was described as the most commonly (26%) mutated gene in canine OMs in another study. 36 Similarly, pathogenic germline mutations of BRCA2 (another tumor suppressor gene) have been reported in human OMs in one study 100 but no pathogenic germline variants have been described for the same gene in another study. 63 Table 1 lists genes believed to drive development of both canine and human OMs.
Abbreviations: CNV, copy number variant; SNV, single-nucleotide variant.
a% of mucosal melanomas affected; % of oral melanomas affected not stated, from Newell et al. 65
Transcriptomics of Primary OMs
In 2015, the expression profiles of 12 canine OMs were compared with those of 4 normal oral mucosal tissue biopsies. 25 The investigation focused on differentially expressed genes involved in the MAPK/ERK and/or PI3K/AKT signaling pathways and identified overlap between genes involved in the same pathways that exhibited differential expression between 45 human cutaneous melanomas and 7 normal human skin samples. It is unclear if a non-hypothesis-driven analysis of the biological process and pathways implicated in canine OM development was undertaken. A recent study compared gene expression profiles of 8 canine OMs with those of 3 “healthy” oral tissue biopsies by analysis of RNA-seq data. Pathways including “cytokine receptor interaction”, “ECM receptor interaction”, and “focal adhesion” were overrepresented among those annotated to genes that exhibited differential expression. 78
Molecular Mechanisms of Human and Canine OM Metastatic Progression
After gaining several truncal driver mutation-associated hallmarks of malignancy, such as self-sufficiency in growth signals, insensitivity to growth-inhibitory signals, altered cellular metabolism, evasion of apoptosis, and limitless replicative potential, 96 neoplastic cells may acquire a “metastatic gene expression signature” and potentially give rise to distant metastases. Metastatic dissemination is a complex and inefficient process, in which not all the neoplastic cells in a primary tumor have the same potential to disseminate, 23 and fewer than 0.01% of cancer cells ultimately pass through the extracellular matrix, enter the circulation, travel through the blood or lymphatic systems, extravasate, and grow at the distant site to develop into metastases. 13 Metastatic dissemination occurs from different subpopulations of the primary tumor, often in a parallel rather than a serial fashion. This means that distant metastases are thought to be seeded early in the development of cancer, at the same time of regional metastases. The idea that melanoma dissemination follows a linear progression from the primary site to regional to distant metastases is not considered correct, despite the clinical evidence of draining lymph node metastasis developing earlier than distant metastases. Indeed, the faster growth of regional metastasis may be correlated to their physical proximity to the primary tumor, increasing the probability of repeated seeding events. 82 This might reflect that the lymphatic system is built for transport of fluid and cells away from tissues and therefore potentially able to increase the chances of reseeding. A recent study showed that in a melanoma mouse model, all animals had lung metastases that originated from both lymph node lesions and directly from the primary tumor, supporting the hypothesis of parallel dissemination. 72 Moreover, circulating melanoma cells have been detected at all stages of human cutaneous melanoma (including early stages), supporting the idea of parallel dissemination to regional lymph nodes (via the lymphatic system) and distant sites (via the blood) early in tumor progression. 80
The particular way that melanoma cells disseminate to distant sites (early in tumor development, arising from both the primary tumor and the metastatic regional lymph node) should be considered when trying to uncover the genetic and epigenetic alterations underpinning the metastatic process in human and canine OMs. Genomic and transcriptomic analyses of the metastatic disease should not be confined to the regional metastasis and those considered to be representative of all other metastatic sites, as some subclones could have arisen directly from the primary site, with branch (private) mutations different from the lymph nodal metastasis. On this basis, where possible, all metastatic tumors in each case (dogs and human) should be sampled to facilitate screening for recurrent metastasis-specific genetic and epigenetic alterations, which could potentially be targeted in both canine and human OMs.
Comparative Genomics of Primary and Metastatic OMs
Most genomic profiling studies of human metastatic melanoma (almost exclusively with cutaneous localization) have explored genome evolution in response to targeted therapy. 2,85 A recent study compared the somatic mutation profiles of primary and metastatic cutaneous melanomas (both lymph node and visceral metastases) by next-generation sequencing of 25 genes relevant to cutaneous melanoma development. 57 A high level of concordance between the mutational patterns of paired primary and metastatic cutaneous melanomas was demonstrated, with particularly high levels of consistency observed for BRAF (the mutation pattern at primary and metastatic sites was consistent for 95% of tumors). However, given that human and canine OMs only rarely present with mutations in BRAF, it cannot be assumed that the same level of concordance in mutation pattern is true for primary and metastatic human and canine OMs. The same study further demonstrated that there is a discrepant mutation pattern (primary vs metastatic) for 76% of cases when considering all mutations (including those of uncertain or unknown significance). In another recent study, whole-exome sequencing of single or multiple metastases in human cases with advanced melanoma arising from different sites was performed to assess genomic events leading to malignant transformation and progression from primary to regional and distant metastasis. 7 This study reported that most mutations are truncal events and metastatic divergence is a late event. However, despite including several melanoma subtypes in this latter study, only 2 mucosal melanomas were analyzed and neither were from the oral cavity. 9 A comparative exome sequencing study 104 featuring human mucosal and canine oral melanoma included a limited comparison of primary tumors and locoregional or distant metastases. Median overlaps in somatic mutations of 42% (human, 8 comparisons) and 44% (canine, 6 comparisons) were observed. However, the study featured a single human primary OM, and the exomes of insufficient canine OM metastases were analyzed to be able to screen for potential recurrent metastasis-specific mutations that may have contributed to metastatic progression. Figure 1 depicts the hypothetical model of canine OM evolution from initiating cell to metastatic disease in OM. To the best of our knowledge, comparative genomic analyses of canine primary and metastatic OMs have yet to be undertaken.
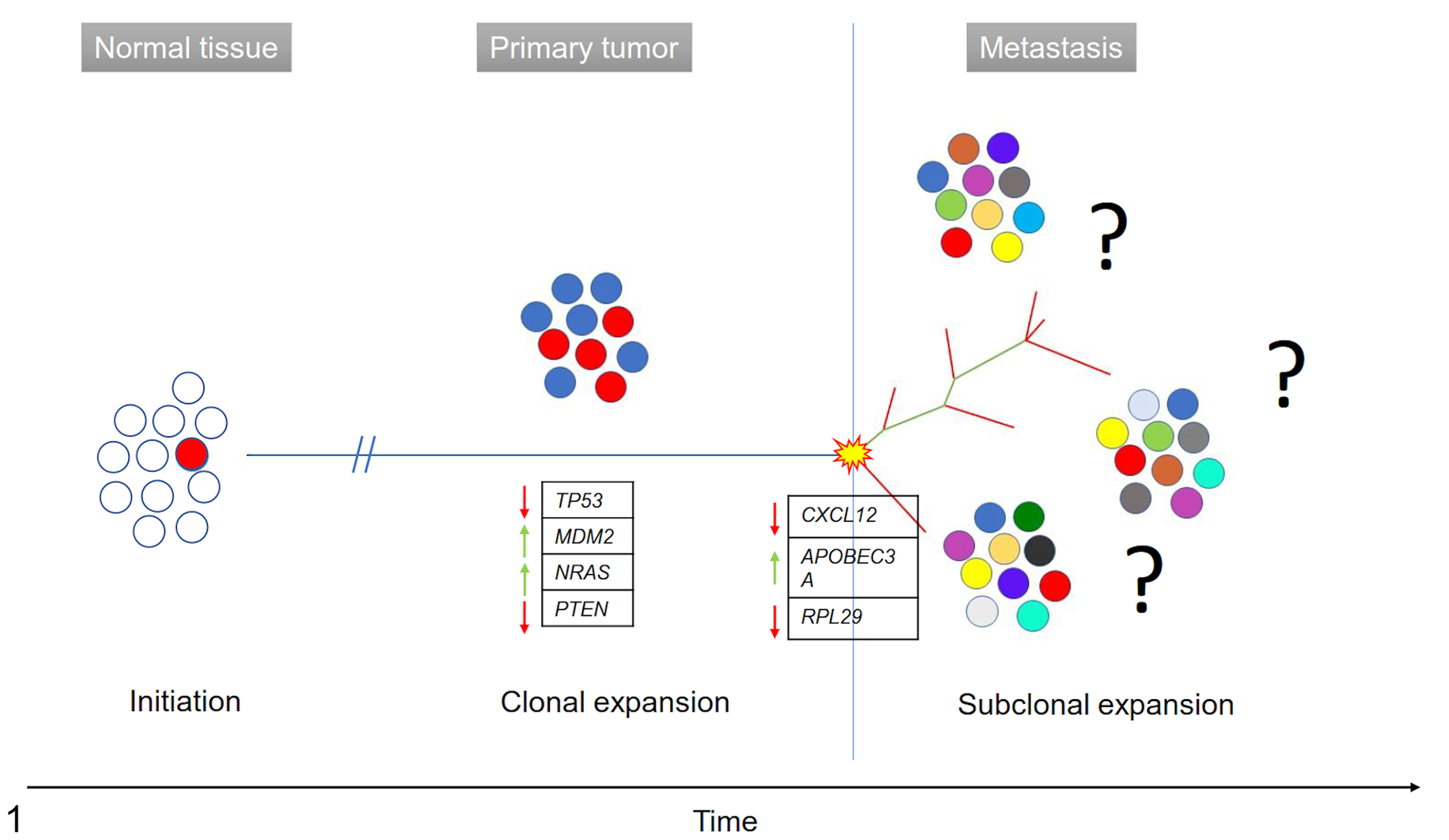
Hypothetical model of canine oral melanoma evolution from initiating cell to metastatic disease. TP53, MDM2, NRAS, and PTEN are driver mutations acquired by the neoplastic melanocytes during clonal expansion. Appearance of a metastatic phenotype is most likely related to branch mutations (eg, alterations in expression of CXCL12, APOBEC3A, RPL29) acquired just before subclonal expansion. Horizontal blue line: ubiquitous/truncal mutations; green line: shared/branch mutations; red line: private mutations. Yellow star: mutational burst. Red arrow: downregulated genes; green arrow: upregulated genes.
Comparative Transcriptomics of Primary and Metastatic OMs
For human cutaneous melanomas, gene expression profiling of nevi, primary melanomas, and melanoma metastases identified a distinct metastatic gene expression signature, with 2602 transcripts significantly correlating with sample type and stage of progression. Interestingly, gene expression signatures characteristic of the tumor metastases were observed at in situ or minimally invasive stages of the disease, suggesting an early development of genetic and/or epigenetic alterations favoring the metastatic dissemination in melanoma progression. 33 Only one study of canine OM metastasis-associated gene expression has been performed, in which the global gene expression profiles of 18 metastasizing and nonmetastasizing primary OMs were compared. 9 Among the genes exhibiting statistically differential expression, the expression levels of 3 genes (CXCL12, APOBEC3A, and RPL29) could be used in a linear discriminant analysis classifier to categorize metastasizing and non-metastasizing primary OMMs with accuracies of 94% and 86%, respectively. 9
Long non-coding RNAs (lncRNAs) are reported to be recurrently altered in many types of human cancers, including human melanomas. 49 Recently, lncRNAs have also been implicated in metastatic dissemination to the regional lymph nodes of human OMs, 45 with several upregulated and downregulated lncRNAs in the metastatic tissue. Moreover, in cis-regulation analysis (which can precisely identify the cis-acting genomic sequences regulating gene transcription 88 ) the metastasis-associated gene PLEKHA5 and microphthalmia-associated transcription factor (MITF) were predicted to be targets of dysregulated lncRNAs. However, review of the literature suggests that comparative gene expression profiling of canine primary OMs and metastases have yet to be performed.
Conclusions
Genomic analysis of primary tumors has demonstrated that the somatic mutation profiles of canine OMs resemble those of a large subset of human OMs in regard to mutations in shared driver genes, with an overall low point mutation frequency, but a high structural variation burden. 104 However, OMs have typically been poorly represented in studies of human mucosal melanomas. Consequently, for unequivocal demonstration of the extent of the genetic similarity between canine OMs and at least a subset of human OMs there is a need for large-scale analysis of the somatic mutation profiles of human and canine melanomas arising in the oral cavity. Although research specifically on OM development has been relatively limited, there has been even less investigation of the genetic and epigenetic events that may contribute to OM metastatic progression in either humans or dogs. The potential value of using dogs affected by OM as a spontaneous model for human OMs is not only to improve understanding of the development of the tumor, but also to define the molecular events that promote OM metastatic progression. Studies should focus on understanding the acquisition of a metastatic gene expression signature by primary OMs and on unravelling the molecular mechanisms contributing to metastatic dissemination, given that human and canine OM-affected mortality is almost entirely attributable to metastatic progression. The higher incidence of canine OM means that, in principle, study of OM metastatic progression in dogs is more readily achievable. Moreover, the approach of “first-in-dog” could also be applied to the development of drugs targeting metastatic disease for OM. This concept is not new and a Comparative Oncology Trials Consortium has been launched by the National Cancer Institute in the United States, with the aim of designing and executing clinical trials in dogs with cancer to assess novel drugs, in order to ease the path for these therapeutic agents to be used in human cancer patients. 29 It is important to acknowledge that there are barriers to comparative OM metastasis research. Human OM is rare and therefore there are potentially research funding constraints. Distant canine OM metastases are not biopsied in the course of routine diagnostic and treatment protocols, and institutional and/or grant funding would be required to fund the dog owner-consented necropsies via which canine OM metastases could be collected. Nevertheless, it is difficult to deny that the potential gains from this line of enquiry are more than sufficient justification to seek to overcome the practical challenges.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

