Abstract
A high prevalence of AA-amyloidosis was identified in a breeding colony of northern tree shrews (Tupaia belangeri) in a retrospective analysis, with amyloid deposits in different organs being found in 26/36 individuals (72%). Amyloid deposits, confirmed by Congo red staining, were detected in kidneys, intestines, skin, and lymph nodes, characteristic of systemic amyloidosis. Immunohistochemically, the deposited amyloid was intensely positive with anti-AA-antibody (clone mc4), suggesting AA-amyloidosis. The kidneys were predominantly affected (80%), where amyloid deposits ranged from mild to severe and was predominantly located in the renal medulla. In addition, many kidneys contained numerous cysts with atrophy of the renal parenchyma. There was no significant association between concurrent neoplastic or inflammatory processes and amyloidosis. The lack of distinctive predisposing factors suggests a general susceptibility of captive T. belangeri to develop amyloidosis. Clinical and laboratory findings of a female individual with pronounced kidney alterations were indicative of renal failure. The observed tissue tropism with pronounced kidney alterations, corresponding renal dysfunction, and an overall high prevalence suggests amyloidosis as an important disease in captive tree shrews.
The term amyloidosis refers to a group of protein-misfolding disorders, characterized by extracellular deposition of fibrillary proteins with a specific β-pleated sheet structure in various organs and tissues. The large group of different amyloidoses is identified and distinguished by their amyloid protein of origin. 2 Amyloid light-chain (AL) amyloidosis is a form of systemic amyloidosis largely associated with plasma cell neoplasms in domestic animals. 36 Reactive systemic amyloidosis is the result of a pathologic deposition of AA amyloid following elevated serum amyloid A levels during chronic infectious, inflammatory, or neoplastic diseases.
An increased occurrence of systemic AA-amyloid deposits in tissues of related individuals or within a genetic lineage is referred to as familial amyloidosis. For instance, predispositions for developing systemic AA-amyloidosis can be found in different cat (eg, Abyssinian and Somali) and dog breeds (eg, Shar Pei). 8,23 Several other animal species, such as captive cheetahs, caracals, black-footed cats, and brown layer chickens, appear to be genetically predisposed to amyloidosis as well. 14,24,25,33
The northern tree shrew, Tupaia belangeri, is a squirrel-sized mammal native to the tropical regions of Southeast Asia that gained attention in biomedical research as an alternative model for human diseases and therapeutic responses. 4,17 In 1997, a breeding colony of T. belangeri was established at the Institute of Zoology of the University of Veterinary Medicine Hannover with founding animals from the German Primate Centre in Göttingen, Germany. Life expectancy of T. belangeri in captivity is reported to range between 9 and 12 years. 11 However, individuals of this breeding colony showed a decline in general health condition at a notably younger age following the introduction of individuals from the University of Bayreuth to the colony. Since a retrospective review of necropsy reports revealed a high incidence of pathological kidney alterations, further detailed investigations were conducted on an animal cohort of 36 individuals.
The aim of the present study was to characterize amyloidosis in T. belangeri with regard to a potential association with lineage and SAA1 gene mutations, sex, age, the specific pathological pattern of amyloid deposition in the kidney, and presumptive pathogenesis.
Material and Methods
Animals
Tree shrews of the breeding colony of the Institute of Zoology at the University of Veterinary Medicine Hannover were housed solitarily or in harmonic pairs in wire mesh cages (size 150 cm × 150 cm × 80 cm per individual; EBECO), equipped with one nest box per cage and a variety of branches and wooden resting shelves at 25 ± 2 °C and 50% relative humidity. 29 The diet consisted of Tupaia pellets (Altromin), fresh fruits, and living insects. Water was provided ad libitum. Thirty-six individuals were included in the present study. Twenty-six individuals (cases 1–26) that died naturally or had to be euthanized due to medical reasons between 2013 and 2020 were subjected to a gross postmortem examination by the attending veterinarians. Organs were removed and stored at −80 °C until further processing. The investigations include an additional nine animals (cases 27–35) from different lineages and places of birth that had died between 2004 and 2013 and were stored at −80 °C without preceding autopsy. These animals had lived at the Institute of Zoology for 5.5 to 9.5 years prior to their death. Selected organs or whole carcasses were examined at the Department of Pathology.
One individual (case 36) was subjected to clinical examination, including abdominal ultrasonography at the Clinic for Small Mammals, Reptiles and Birds at the University of Veterinary Medicine Hannover, and fresh blood and urine samples were taken antemortem. Blood was collected in ethylenediaminetetraacetic acid and serum tubes and a hematological and biochemical profile was generated using automated analysis systems (Siemens Advia120 and RAPIDLab1260). Collected urine was applied to a commercial urine reagent strip (Reactif 11M) according to the manufacturer’s instructions. The animal was euthanized for medical reasons, and autopsy was performed.
Histopathology and Immunohistochemistry
For histological investigation, tissues were fixed in 10% buffered formalin for 24 hours and subsequently embedded in paraffin wax. Tissue sections of 2 to 3 µm thickness were stained with hematoxylin and eosin (HE). Amyloid deposits were detected by Congo red (CR) staining. Severity of amyloidosis was graded based on the amount of deposits found in the kidneys, as this was the most frequently affected organ. The semiquantitative grading scale divided amyloid deposits in the kidneys into absent, mild (scattered deposits in the renal papilla; <10% of the renal medulla affected), moderate (>10% to 50% of renal medulla alterations), and severe (>50% of renal medulla alterations) forms. Renal cysts were classified as “−” (absent), “+” (sporadic, few cysts), “++” (multiple cysts), and “+++” (numerous cysts with prominent atrophy of renal parenchyma).
Amyloid was further characterized by immunohistochemistry (IHC) using an automated platform (Ventana ULTRA; Ventana Medical Systems) with mouse monoclonal anti-AA antibodies (clones mcC, mc4, mc29 18 ). Formalin-fixed, paraffin-embedded sections were incubated with mouse anti-AA antibodies mc4 (1:100; amYmed) or mc29 (1:1000; amYmed) overnight at 4 °C after peroxidase (3% H2O2, 10 minutes) and protein blocking (Zytomed Systems), followed by incubation with anti-mouse immunoglobulins conjugated with horseradish peroxidase (Jackson ImmunoResearch Europe). Chromogen detection was performed by 3,3-diaminobenzidine (Zytomed Systems) and nuclear counterstain by hemalum. Negative controls omitting primary antibodies were included in all staining procedures. Human samples with known AA amyloid deposits served as positive controls.
Molecular Genetic Analysis
We extracted genomic DNA from native muscle tissue samples stored at −70 °C (not formalin-fixed) through a standard ethanol fractionation with concentrated sodium chloride (6 M NaCl) and sodium dodecyl sulfate (10% SDS). The concentration of genomic DNA was adjusted to 50 ng/µL per sample. Genomic DNA of 6 amyloidosis-affected and unaffected T. belangeri individuals each was used for sequencing all 4 exons and their exon-intron boundaries of the serum amyloid A1 gene (SAA1, gene ID TSDBG00029867 and transcript ID TSDBT00029867.1). Reference sequence information was taken from the Tree Shrew Database (http://www.treeshrewdb.org/cgi/idsearch3.pl?id=TSDBG00029867). Forward and reverse primer pairs as well as an internal primer for each exon were designed with Primer3 (http://frodo.wi.mit.edu/primer3/) using the reference genome sequence of T. belangeri (http://www.treeshrewdb.org/cgi/idsearch3.pl?id=TSDBG00029867). Product sizes were between 249 and 693 bp (Supplemental Table S1). Polymerase chain reaction (PCR) was performed in 32 µL reaction volumes containing 2 µL genomic DNA, 40 μM deoxyribonucleoside triphosphates, 10 pmol of each primer, and 1 U of Taq polymerase in the reaction buffer supplied by the manufacturer (Qiagen). After a 4-minute initial denaturation at 95 °C, 35 cycles of 30 seconds at 95 °C, 45 seconds at 59 °C, and 50 seconds at 72 °C were performed in a T Professional Thermocycler (Whatman Biometra). Samples were sequenced with forward and reverse sequencing primers on a Genetic Analyzer 3500 (Applied Biosystems by Life Technologies), and resulting sequences were analyzed using Sequencher 4.8 (Genes Codes). Sequences were screened for mutations using the T. belangeri reference sequence. 39 We employed SIFT 30 and PolyPhen-2 1 to predict variant effects detected within SAA1. A case-control analysis for variants detected was performed using SAS, v 9.4 (Statistical Analysis System).
Statistical Analysis
The prevalence of amyloidosis was calculated and a potential tissue tropism was evaluated by comparing the number of individuals with amyloid deposits in each studied organ. Potential risk factors including sex, origin (Bayreuth lineage or non-Bayreuth), and concurrent diseases (neoplastic or inflammatory processes) were evaluated for an association with amyloid deposits using separate Fisher’s exact tests, with P ≤ .05 considered significant. Potential differences in age and body mass at time of death between groups were analyzed using 2-sample t tests for factors with 2 categories (amyloidosis, renal cysts, sex, origin, concurrent disease) or one-way analyses of variance (ANOVA) for factors with 3 or more categories (severity of amyloid deposits in the kidneys, renal cyst classification). The level of significance was P ≤ .05. Body mass was subdivided by sex to account for the known sexual dimorphism in T. belangeri. 37 A possible relationship between the severity of amyloid deposits and presence of renal cysts, as well as extent of cystic alterations was analyzed using Fisher’s exact tests.
Results
Repeated occurrence of pathological kidney alterations during routine gross postmortem examinations led to a retrospective investigation of tissue samples and necropsy reports of 36 T. belangeri (22 females, 14 males) aged 2.8 to 10 years (Supplemental Tables S2 and S3) originating from 5 different facilities. Amyloidosis was identified in 26 cases (72%; 15 females and 11 males) by hematoxylin eosin and Congo red staining. Of these, amyloid deposits were restricted to the kidneys in 8 animals (31%) and to the intestines in 5 animals (19%). Multiple organs were affected in 13 cases (50%), of which 10 had involvement of both the kidneys and intestine, and the other 3 had amyloid within kidneys and skin; kidneys, intestine and skin; or kidneys, intestine, skin, spleen, mucous membranes, and tongue. Of the 26 animals with amyloidosis, 21 (80%) had amyloid deposits in the kidneys. Renal amyloid deposits were located predominately in the interstitium of the medulla and papilla, and were associated with atrophy of collecting tubules (Figs. 1, 2). In addition, amyloid deposits were observed in the walls of renal arteries. Fourteen (54%) of the 26 cases of amyloidosis showed cystic dilatations of renal tubules, associated with parenchymal atrophy within the cortex and medulla (Fig. 1). Intestinal amyloid deposits were largely restricted to the lamina propria (Figs. 3, 4). Immunohistochemistry revealed the presence of AA amyloid (Figs. 4 –8), with a strong positive signal with mc4, weak reactivity with mcC, and no signal with mc29 antibodies.
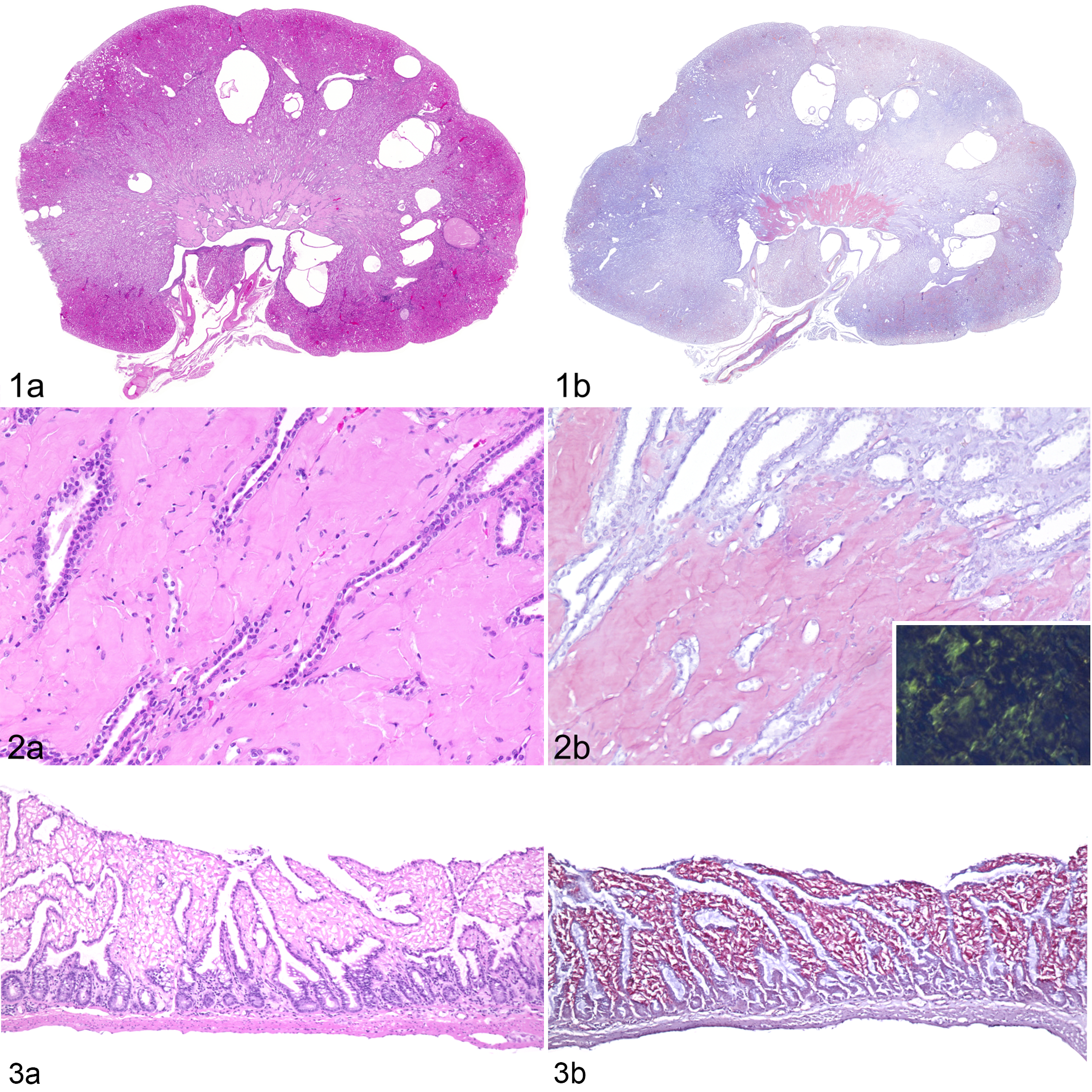
AA-amyloidosis, northern tree shrew, case 3.
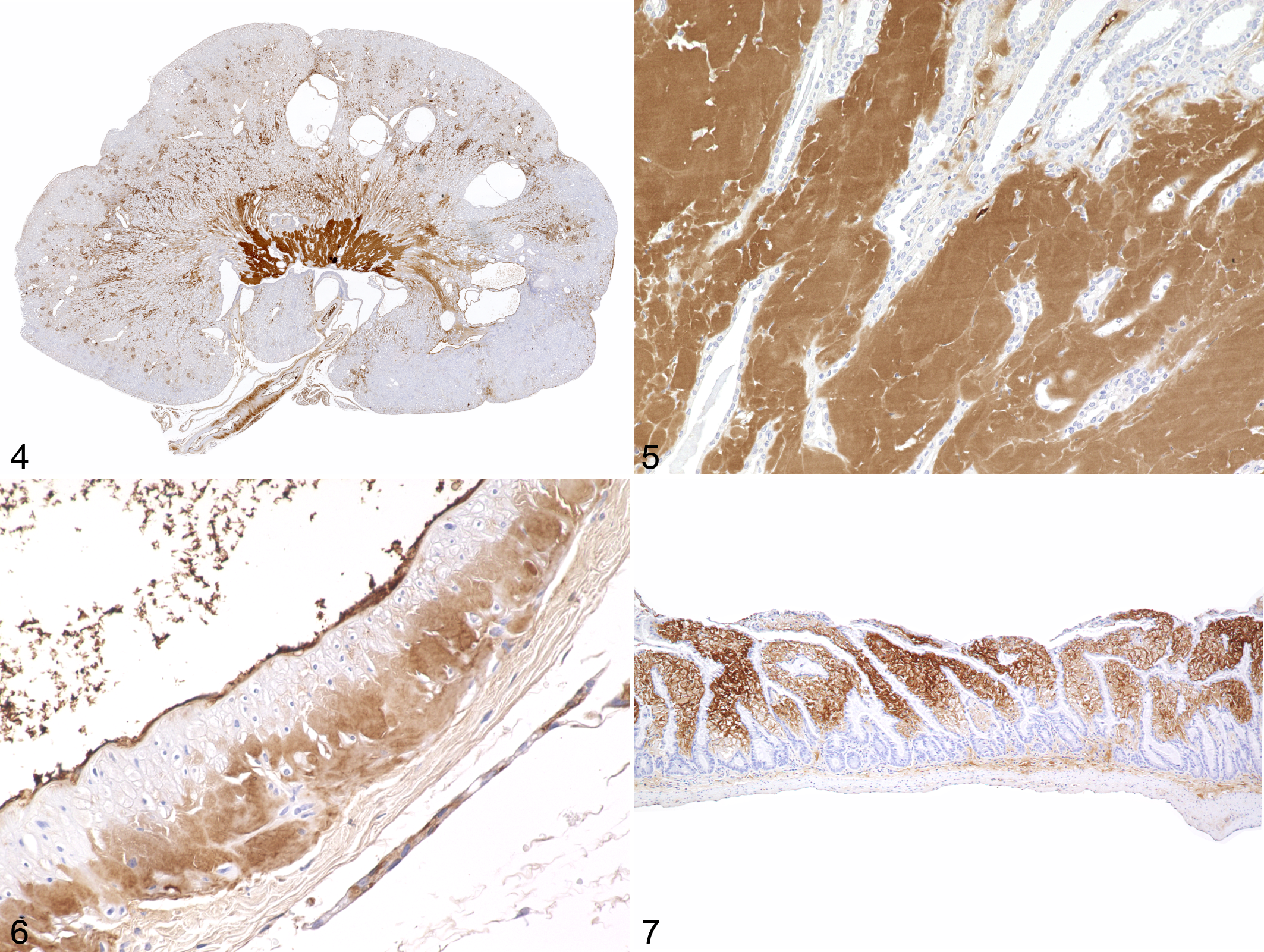
AA-amyloidosis, northern tree shrew, case 3. Immunohistochemistry with antibody mc4.
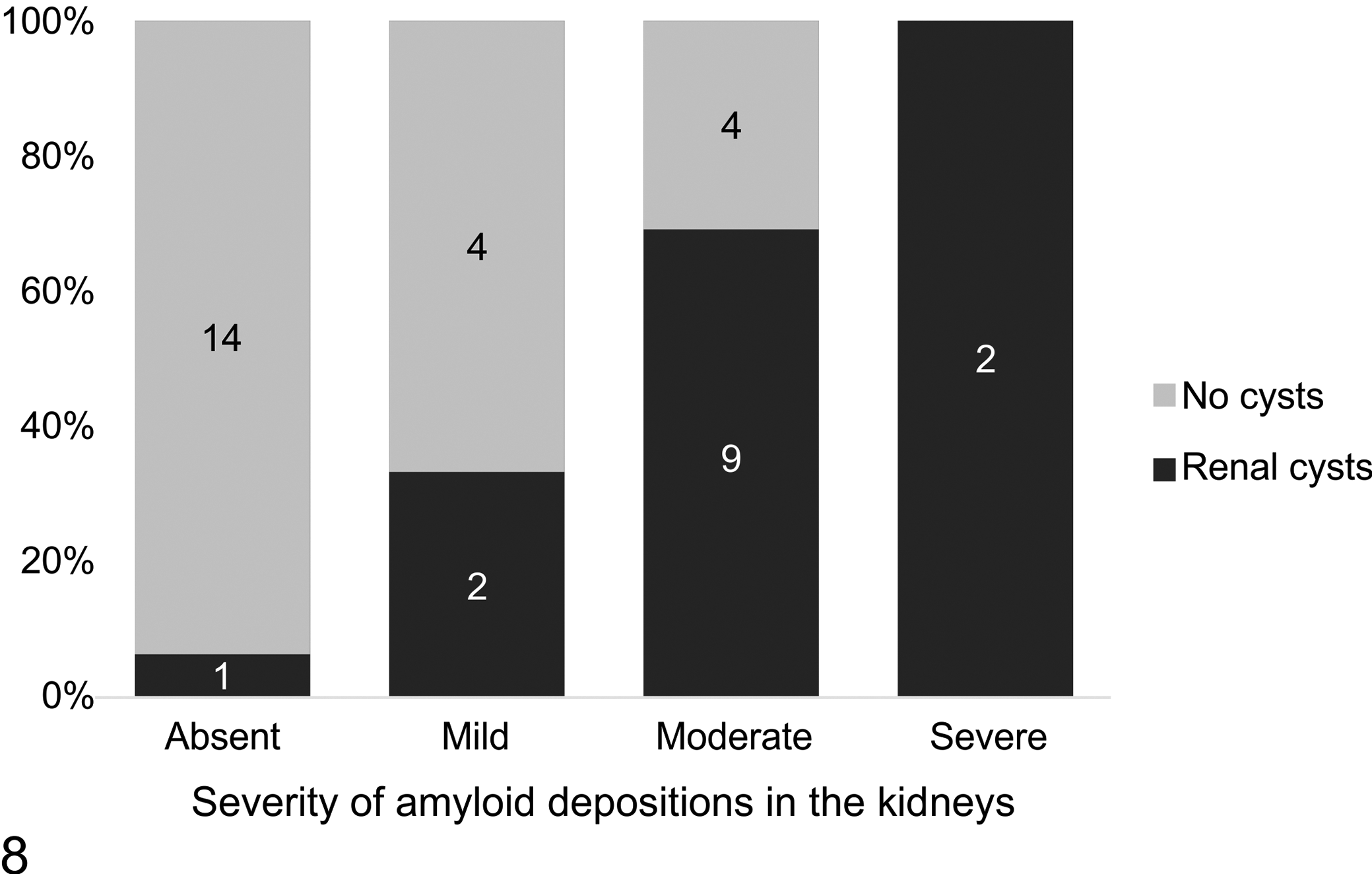
Prevalence of renal cysts with different grades of amyloid deposits in the kidneys. The number of cases is shown.
There was no significant co-occurrence of amyloidosis with neoplastic (9/13 vs 17/23, P = 1, Fisher’s exact test) or with inflammatory processes (4/7 vs 22/29, P = .37, Fisher’s exact test). The probability of amyloidosis was furthermore not influenced by animal origin (P = .413, Fisher’s exact test): 20/26 (77%) individuals with Bayreuth ancestry and 6/10 (60%) cases with other origin had amyloid deposits. Sex ratio in the data set was skewed toward females, but frequency of amyloidosis did not differ significantly between sexes (P = .706, Fisher’s exact test). Furthermore, the mean age at time of death did not differ significantly between animals with amyloidosis and unaffected individuals (2-sample t test, T = 0.538, n 1 = 26, n 2 = 10, P = .599). Body mass of individuals in the given data set ranged between 123 g and 242 g. An outlier of 343 g was excluded from statistical analysis, as this was due to a case of pathological gestation with massive fluid accumulation in the uterus after maceration of fetuses. Mean body mass differed significantly between females with amyloidosis (162.43 ± 32.98 g) and females without amyloidosis (200.57 ± 37.42 g; 2-sample t test, T = −2.289, n 1 = 15, n 2 = 7, P = .043), whereas body mass variation did not differ within the males (2-sample t test, T = 0.134, n 1 = 11, n 2 = 3, P = .899).
Within the 26 cases of amyloidosis, the severity of amyloid deposits in the kidneys was classified as absent in 5 (19%), mild in 6 (23%), moderate in 13 (50%), and severe in 2 (8%) cases. There was no statistically significant association between sex (P = 1, Fisher’s exact test) or origin (P = .476, Fischer’s exact test) and the extent of amyloid deposits. Neither age (F[3, 22] = 1.336, P = .288) nor body mass (F[3, 21] = 1.337, P = .289) were statistically associated with different grades of renal amyloidosis. The youngest individual with amyloidosis was 3 years and 4 months of age (case 1). The prevalence of renal cysts increased with increasing severity of amyloid deposits in the kidneys, with 33% of mild, 69% of moderate, and 100% of severe forms showing cystic dilatation of renal tubules. The influence of the grade of renal amyloidosis on the occurrence of renal cysts was significant (P < .001, Fisher’s exact test; Fig. 8) and the extent of these cystic alterations (from scattered, few cysts to numerous cysts with prominent atrophy of renal parenchyma) was associated with severity of amyloid deposits (P = .003, Fisher’s exact test). Only one individual (case 15) had numerous cysts with prominent atrophy of renal parenchyma without evidence of amyloid deposits. Renal cysts (n = 14) were only observed in cases older than 5 years of age.
Additional clinical and laboratory data were provided by case 36, a 6-year-old female that showed a progressive reduction in general condition and activity, while food intake remained stable. Abdominal sonography under sedation with midazolam (0.5 mg/kg, midazolam, 5 mg/mL, Rotexmedica) revealed multiple smaller renal cysts in highly irregular right and left kidneys without definable corticomedullary junctions (Fig. 9). A single large (1.23 × 1.22 cm) anechogenic septated cyst was found on the cranial pole of the right kidney (Fig. 10). The qualitative and semiquantitative urinalysis revealed leucocytes and elevated levels of urobilinogen, ketones, bilirubin, protein, and glucose. Compared with available reference values for Tupaia spp., 10,11,15,22,34,38,41,42 the hematological and biochemical profiles had low concentrations of alkaline phosphatase, aspartate aminotransferase, and creatine kinase; elevated creatinine, chloride, and glucose; and reduced albumin and total protein levels. The calcium-to-phosphorus ratio was 2.1:2.7 (Supplemental Table S4). Postmortem examination revealed multifocal bilateral cysts up to 0.7 cm diameter in the cortex and medulla of the kidneys. Amyloid deposits were observed in histological sections of kidneys and the mucosa of the small intestine.
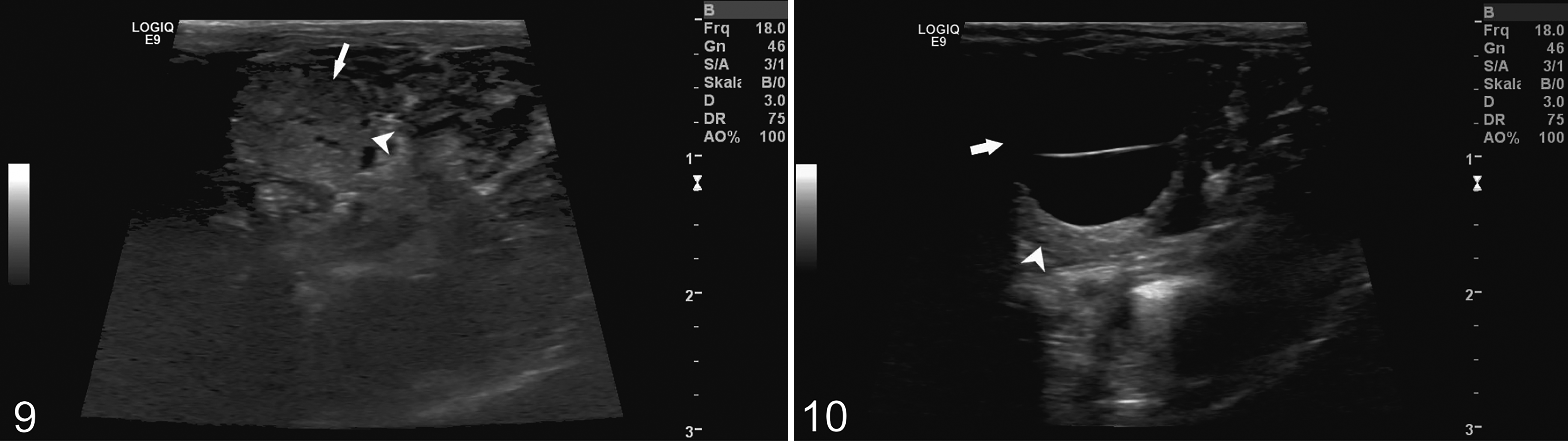
AA-amyloidosis, northern tree shrew, case 36. Ultrasonography.
Sequence analysis of the SAA1 gene revealed a missense mutation within exon 3 (SAA1: c.147G>A) causing an exchange of valine with isoleucine (p.V49I; Suppl. Fig. S1) compared with the T. belangeri reference sequence. This variant was found in amyloidosis-affected and unaffected animals. Of 6 amyloidosis cases tested, 3 were homozygous A/A and 1 was heterozygous G/A. Of the 6 unaffected animals tested, 2 were homozygous A/A and 1 was heterozygous G/A. SIFT 30 prediction gave a score of 0.28, indicative for a tolerated variant. Prediction with PolyPhen-21 failed to return a score because the mutation was classified as unknown. There was no association of the SAA1: c.147G>A variant with the amyloidosis status (P = .82), severity of amyloid deposits in the kidneys (P = .97), lineage (P = .33), sire (P = .24), dam (P = .36), or sex (P = .79).
Discussion
A high prevalence of systemic amyloidosis was identified in captive T. belangeri in the present study, with 72% of examined individuals showing deposits of AA-amyloid. Deposits were most frequent in kidney and small intestine.
AA-amyloidosis is the form of amyloidosis most commonly reported in mammals and birds. A relation between significant chronic inflammatory disease and systemic AA-amyloidosis has been discussed for example in captive cheetahs (Acinonyx jubatus), Dorcas gazelles (Gazella dorcas), and California sea lions (Zalophus californianus). 5,25,28 Within the tree shrews of the investigated breeding colony, however, there was no significant difference in the prevalence of amyloidosis between groups with and without concurrent inflammatory or neoplastic diseases. Similar findings have been reported on other nondomestic species, such as island foxes (Urocyon littoralis), 12 black-footed cats (Felis nigripes), 33 or common marmosets (Callithrix jacchus). 21
AA-amyloidosis has also been associated with chronic stress in several species. Studies on Syrian hamsters, 13 laboratory mice, 19 and white Pekin ducks 6 reported a link between amyloidosis and social stress related to crowding conditions, and an increase in SAA was found in cows and pigs subjected to isolation and transport stress. 20,32 Interestingly, this increase was not observable in a longitudinal study of chronic stress in pigs induced by unpredictable temporal food deprivation. 26 The impact of persistent stressors on AA-amyloidosis therefore remains debatable.
Tree shrews are solitary or pair-living, strongly territorial mammals that are relatively susceptible to stress. The pronounced territoriality of males in particular has been used to establish a chronic psychosocial stress model for experimental studies on depressive disorders. 17 Tree shrews of the investigated breeding colony were housed either solitarily or in harmonic pairs, but auditory, olfactory, and visual stimuli of conspecifics were present at all times. Thus, an impact of stress on the development of AA-amyloidosis cannot be excluded in the present study.
The absence of a clearly identifiable underlying condition and the high prevalence of amyloidosis in the colony suggest a species-specific predisposition for amyloid A deposition, comparable to the familial amyloidosis of Siamese and Abyssinian cats. 23 SAA is of polymorphic nature, and a number of isotypes have been identified that are preferentially deposited as amyloid. 3,35 For example, a unique AA peptide is suspected to play a crucial role in the development of AA-amyloidosis in pigs via cross-seeding polymerization. 16 A species-related factor that alters either SAA synthesis or enzymatic degradation may explain the observed accumulation of systemic AA-amyloidosis in the studied T. belangeri. Analyses revealed no clear association of the detected SAA1 missense mutation with the occurrence of amyloidosis in investigated animals. However, development of amyloidosis requires an inflammatory stimulus in addition to genetic preconditions. In the present study, premature death of genetically predisposed individuals prior to amyloid deposition is also possible. The role of the detected allelic variation in the complex of amyloid formation in T. belangeri therefore remains to be explored. Moreover, the suspected impact of the Bayreuth lineage on the predisposition for amyloidosis could not be verified based on the given data set. The lack of a sufficient number of dyads hampered a complete pedigree analysis. A comprehensive genomic investigation of deposited amyloid A and exploration of the allelic composition of genes encoding the amyloid fibril protein in T. belangeri will be needed to confirm a suspected familial or species-specific predisposition. Nevertheless, amyloidosis did occur in cases originating from 5 different facilities and therefore seems to be a common finding in captive tree shrews.
The potential transmissibility of AA amyloid may contribute to the observed high prevalence in a captive environment. For example, AA amyloid fibrils extracted from the feces of amyloidosis-positive cheetahs were capable of inducing systemic amyloidosis in experimentally infected mice. 40 Contact to contaminated fecal matter in a captive setting may therefore increase the incidence of amyloidosis. A comparative study on wild T. belangeri could elaborate on this factor and help disentangle extrinsic (eg, husbandry, stress) and intrinsic (familial or genetic predisposition) influences.
Amyloid deposits in the kidneys were almost exclusively observed in the renal medulla and papilla. Extracellular amyloid deposits in the medullary interstitium may lead to physical impairment and obstruction of collecting tubules and blood vessels, causing focal ischemia and tubular dilatation and cyst formation, as described in mice. 9 While the destructive effects on renal parenchyma are evident in microscopic sections, an additional toxicity of amyloidogenic precursor proteins has been discussed in amyloidosis-associated kidney disease 7 and may have added to the observed kidney disease.
Given the extent of amyloidosis and the associated pathologic changes, especially in the kidneys, renal dysfunction of affected individuals can be expected. A decline in general condition and weight loss were noted in the clinical record of the majority of cases diagnosed with systemic amyloidosis with renal manifestation at postmortem examination. Moreover, a significantly lower body mass was observed in female tree shrews with systemic amyloidosis, compared with unaffected conspecifics. Blood analysis of case 36 showed increased creatinine, consistent with renal failure. Moreover, the decrease in blood albumin and total protein in connection with proteinuria indicates protein-losing nephropathy probably due to amyloidosis. The observed hyperglycemia could be an acute stress response due to transport and handling during abdominal sonography. However, increased urinary glucose and ketones in case 36 are indicative of a diabetic condition. Spontaneous diabetes mellitus has been previously reported in tree shrews. 27 Even though no reference values are available for phosphate in Tupaia spp., the calcium-to-phosphate ratio of 2.1:2.7 determined in the blood sample of case 36 appears critical. In small mammals, physiological serum concentrations of calcium and phosphate are expected within a ratio of 1.5–2:1. A dysregulated calcium and phosphate homeostasis is common in chronic kidney disease due to secondary renal hyperparathyroidism. 31
In conclusion, this study identified AA-amyloidosis as a common disease of captive T. belangeri. The pronounced amyloid deposits in the kidneys and associated renal dysfunction suggest amyloidosis as a major factor for morbidity and mortality in this population of tree shrews. The genetic background of the suspected familial form, pathogenesis, clinical aspects, and diagnostic findings of the characterized AA-amyloidosis require further investigation.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211066847 - AA-amyloidosis in captive northern tree shrews (Tupaia belangeri)
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211066847 for AA-amyloidosis in captive northern tree shrews (Tupaia belangeri) by Annette Klein, Ute Radespiel, Felix Felmy, Tina Brezina, Malgorzata Ciurkiewicz, Jessica Schmitz, Jan Hinrich Bräsen, Reinhold Paul Linke, Sina Reinartz, Ottmar Distl and Andreas Beineke in Veterinary Pathology
Footnotes
Acknowledgements
The authors are indebted to the late Elke Zimmermann for the establishment of the captive colony at the Institute of Zoology and her continuous interest and work on Tupaia belangeri. We also appreciate the work of the veterinarians Annette Klaus, Marko Dubičanac, and Annika Kollikowski as well as excellent technical assistance by Edda Christians, Mahtab Taleb-Naghsh, Angelika Meinel, and Nicole Wagner. We also thank two anonymous reviewers for helpful comments on the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
