Abstract
Clostridium perfringens type D epsilon toxin (ETX) is responsible for a severe and frequently fatal neurologic disorder in ruminant livestock. Light microscopic, immunohistochemical, and ultrastructural studies have suggested that ETX injury to the cerebral microvasculature, with subsequent severe, generalized vasogenic edema and increased intracranial pressure, is critically important in producing neurologic dysfunction. However, the effect of ETX on brain capillary endothelial cells in vitro has not been examined previously, to our knowledge. We exposed a well-characterized human blood–brain barrier cell line to increasing concentrations of ETX, and demonstrated a direct and dose-dependent endotheliotoxic effect. Our findings are concordant with the primacy of vasculocentric brain lesions in the diagnosis of acute epsilon toxin enterotoxemia in ruminant livestock.
Introduction
Clostridium perfringens type D epsilon toxin (ETX) causes an important, and often fatal, neurologic disorder of worldwide distribution in lambs and goats and, less commonly, cattle.2,13 When the brain is exposed to large doses of circulating ETX absorbed from the intestine, the clinical course is acute or peracute.2,13
ETX is a member of the aerolysin family of pore-forming toxins. One domain of ETX binds to endothelial receptors, where it utilizes lipid rafts and caveolin to oligomerize into a pre-pore on the surface of the cell membrane. Another ETX domain then inserts into the membrane lipid bilayer and forms an active pore. ETX can be transiently prevented from exerting its deleterious effect by prior administration of the prototoxin, presumably by competitive inhibition of endothelial receptors. The ETX-induced endothelial cytotoxicity appears to be caused, in part, by a rapid decline, via pores, in cytoplasmic potassium levels, with a concomitant influx of sodium and chloride ions. This process, which seems to involve adenosine triphosphate (ATP) depletion, leads to cell necrosis. 11
Histologic and ultrastructural studies in sheep and laboratory rodents have suggested that the principal action of this neurotoxin is a damaging effect on the cerebral microvasculature, probably after binding to a specific endothelial receptor.2,11,13 Capillary endothelial injury leads to loss of blood–brain barrier (BBB) integrity and a marked increase in vascular permeability, resulting in severe, generalized, and diagnostic vasogenic edema, which leads to greatly elevated intracranial pressure, convulsions, coma and, often, death.2,13 Moreover, ETX produces similar microvascular damage to the blood–retinal barrier (BRB), 3 which resembles the BBB in most important respects. Loss of endothelial barrier antigen immunoreactivity, a marker of an intact BBB and BRB in rats, is correlated with brain and retinal microvascular injury.4,8
Although the cerebral capillary endothelium seems to be the major target of ETX, a direct, deleterious effect of this toxin on cultured endothelial cells in vitro has not been demonstrated. The only study to examine the effect of ETX on cultured endothelial cells was unable to show any resulting endothelial injury, but the target endothelium was derived from ovine, caprine, and bovine aorta, 12 rather than brain microvascular endothelium, which differs from endothelia elsewhere. 7 The BBB possesses tight interendothelial junctions, a paucity of micropinocytotic vesicles, and surrounding astrocytic end-feet, resulting in highly localized control of the composition of the perineuronal fluid. 7 We designed a study to determine whether ETX has a direct and damaging, dose-dependent effect on cerebral endothelial cells in vitro, and investigated this ETX-microvascular interaction on target endothelial cells of brain origin.
Materials and methods
The BBB immortalized human cerebral microvascular endothelial cell (hCMEC/D3) line (catalog SCC066; Millipore, Temecula, CA) was used.15,16 This cell line was derived from human temporal lobe microvessels isolated from brain tissue collected at surgery. In the first passage, cells were sequentially immortalized by lentiviral vector transduction with the catalytic subunit of human telomerase (hTERT) and SV40 large T antigen, following which endothelial cells were selectively isolated by limited dilution cloning, and the clones were extensively characterized for brain endothelial phenotype. This stable cell line shows robust proliferation, while retaining the morphologic and known biochemical phenotype of differentiated BBB endothelium over many passages. 10
The ETX (Commonwealth Serum Laboratories [CSL], Melbourne, Australia) used in our study was produced by growth of a highly toxigenic strain of C. perfringens type D. The prototoxin was activated at pH 7.5 by trypsin (0.08% weight/volume) for 30–45 min at 37°C to form the active epsilon toxin. The batch of toxin used contained 386 L+/mL (where 1 L+ = the amount of toxin neutralized by 1 international unit of antitoxin) and 69,000 mouse minimal lethal doses (MLD)/mL. 4 The toxin was then frozen at −80°C, at which temperature it is very stable (CSL, Dr. Anthony Cameron, pers. comm., 2017) until used. The trypsin-activated toxin was used at a dose rate of 1:100, 1:200, and 1:800, diluted in complete cell culture medium (catalog SCME004, EndoGRO-MV; Merck Millipore, Sacramento, CA). This was the same batch of toxin that was used to produce ETX-specific microvascular damage in the brain and retina in previous studies.3,4,8
For light microscopy, 1 × 105 endothelial cells/mL were grown for 24 h to confluence at 37°C in humidified air with 5% CO2 atmosphere in EndoGRO-MV culture medium containing 5% serum. Adherent cells were then removed with trypsin–EDTA, resuspended to 1 × 105 cells/mL, and added to each pre-coated collagen well (catalog 08-115; Millipore) in a Nunc Lab-Tek II chamber slide system (cat 154453; Thermo Fisher Scientific, Scoresby, Victoria, Australia) and incubated for 24 h. This system consists of a removable, polystyrene medium chamber attached to a specifically treated, glass microscope slide (25 × 75 mm) to ensure a consistent growth surface. The culture medium was then removed from the cells, and 1 mL of ETX dilutions were added to the wells. Each dilution of toxin had a corresponding control well to which an equal volume of the complete culture medium was added, and 3 replicates were studied for each treatment. Endothelial cells were incubated in the presence of ETX for 5 min, and the Lab-Tek slides were then fixed in −20°C acetone for 10 min and stained with hematoxylin and eosin (H&E). The identity of the cell line as endothelial was confirmed using CD31 immunohistochemistry (mouse monoclonal antibody, JC70A MO823; Dako, Carpinteria, CA) as an endothelial marker.
For transmission electron microscopy (TEM), 1 × 105 endothelial cells/mL were grown for 48 h to confluence at 37°C in humidified air with 5% CO2 atmosphere in EndoGRO-MV culture medium containing 5% serum. ETX was diluted to 1:100, 1:200, and 1:800 in complete medium and added to each pre-coated collagen flask; a control flask contained tissue culture medium alone. After incubation, adherent cells were removed with trypsin–EDTA and centrifuged at 300 × g for 5 min to obtain a pellet of cells. The pellet was then washed in phosphate-buffered saline (PBS) and fixed in 1.25% glutaraldehyde in PBS with 4% sucrose overnight in a microfuge tube. The pellet was then postfixed in 2% osmium tetroxide, dehydrated in a graded series of ethanols, and embedded in epoxy. Ultrathin sections were cut at 80 nm thickness and stained with uranyl acetate and lead citrate.
For scanning electron microscopy (SEM), endothelial cells were maintained on a filter membrane for fixation (4% paraformaldehyde/1.25% glutaraldehyde in PBS with 4% sucrose, pH 7.2). Filters were washed in buffer (PBS with 4% sucrose) for 5 min, followed by post-fixing in 2% osmium tetroxide for 1 h. Filters were then dehydrated through a graded series of ethanol before application of 1:1 hexamethyldisilazane:100% ethanol for 15 min, followed by 100% HDMS for 20 min. Samples were air dried before mounting on stubs and coating. Samples were imaged (Qanta FEC 450; FEI, Netherlands) and visualized using xT microscope control software.
In order to quantify the effect of increasing concentrations of ETX on cultured human brain endothelial cells, 2 methods were used. First, a 3-(4,5-dimethylthiazole-2-yl)-2,5-diphenyltetrazolium bromide (MTT; catalog 11465007001; Roche, Basel, Switzerland) colorimetric cell viability assay 9 was used to calculate the number of viable endothelial cells after exposure to ETX. Briefly, MTT, a yellow tetrazole that readily penetrates viable eukaryotic cells, is converted by viable cells with active metabolism to a purple formazan product with an absorbance maximum near 560 nm. The quantity of formazan, which is related to the number of viable cells present, is measured by recording changes in absorbance at 540 nm using a plate-reading spectrophotometer. The measured absorbance is directly correlated to the number of viable cells standardized to medium-only controls (n = 5).
Second, cell counts were performed in order to obtain the mean number of cells/well. A specimen area of 1.5 × 104 µm2 at 400× magnification was selected, and the number of cells present in 5 such squares (5 squares/replicate, 3 replicates/treatment) counted for each concentration of ETX. For each method of analysis, 2 controls were used, namely complete culture medium only, and trypsin in complete culture medium. Data were compared using ANOVA, and all values were expressed as mean ± SD.
To confirm the specificity of ETX-induced cytotoxicity in vitro, the endothelial cell culture was pre-incubated with an ETX monoclonal antibody (C. perfringens type D epsilon monoclonal antibody–IRP 640; CPDN 5B7 and IRP 579 [CPDN 4D7]; U.S. Department of Agriculture, Animal and Plant Health Inspection Service, Centre for Veterinary Biologics, Ames, IA). The cells were then incubated with ETX as described above.
Results
In control cultures of brain endothelium examined by light microscopy, nuclei were round-to-ovoid, and occasionally slightly elongate, with generally evenly distributed coarse-to-fine chromatin clumps. The cytoplasm was markedly elongated and palely eosinophilic in H&E-stained microscope slides, and cytoplasmic boundaries were sometimes elongated (Fig. 1). A few binucleate cells were present and occasionally mitotic figures were seen. Endothelial cells exposed to ETX (Figs. 2, 3) were more sparsely distributed than in control cultures, and became conspicuously more depleted with increasing concentrations of ETX. Injured endothelial cells at higher concentrations frequently contained a pyknotic nucleus and shrunken, hypereosinophilic cytoplasm (Fig. 3); at lower concentrations of ETX, nuclei were degenerate with chromatin condensation, and the cytoplasm less intensely eosinophilic (Fig. 2).
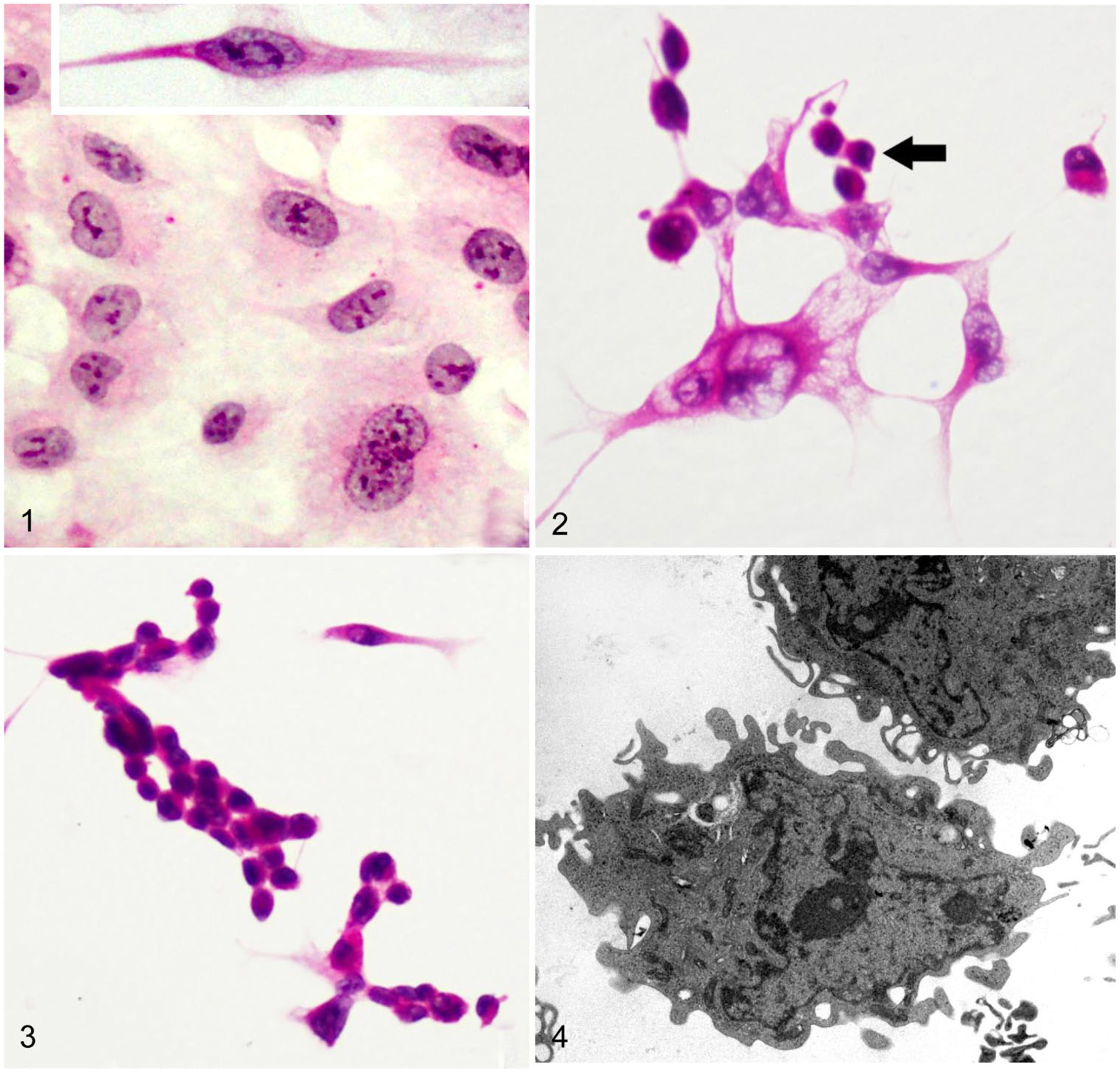
Untreated human cerebral capillary endothelial cells in culture with round-to-ovoid nuclei and a variable amount of palely eosinophilic cytoplasm, which was sometimes (inset) markedly elongated. H&E.
Control endothelial cells (Fig. 4) examined by TEM were usually polygonal and sometimes in close apposition to neighboring cells, with adherens-type junctions occasionally seen at sites of cell contact. The oval nucleus was more-or-less centrally disposed with one or more nucleoli; the fine chromatic granules were uniformly distributed throughout the nucleus, and the nuclear envelope was smooth and well-defined. The cytoplasm contained large quantities of rough and smooth endoplasmic reticulum, prominent Golgi complexes, many vesicles, mitochondria, numerous bundles of fine filaments, and occasional microtubules; microvilli projected from the cell surface. Endothelial cells exposed to ETX (Fig. 5) had condensation of nuclear chromatin (hyperchromasia) or nuclear pyknosis, resembling death by necrosis. The cytoplasm was either shrunken and more electron-dense than control endothelium or showed organelle swelling and degeneration. In a few endothelial cells, vacuoles contained condensed nuclear remnants and degenerate cytoplasm, suggesting that they had phagocytosed effete endothelial cells.
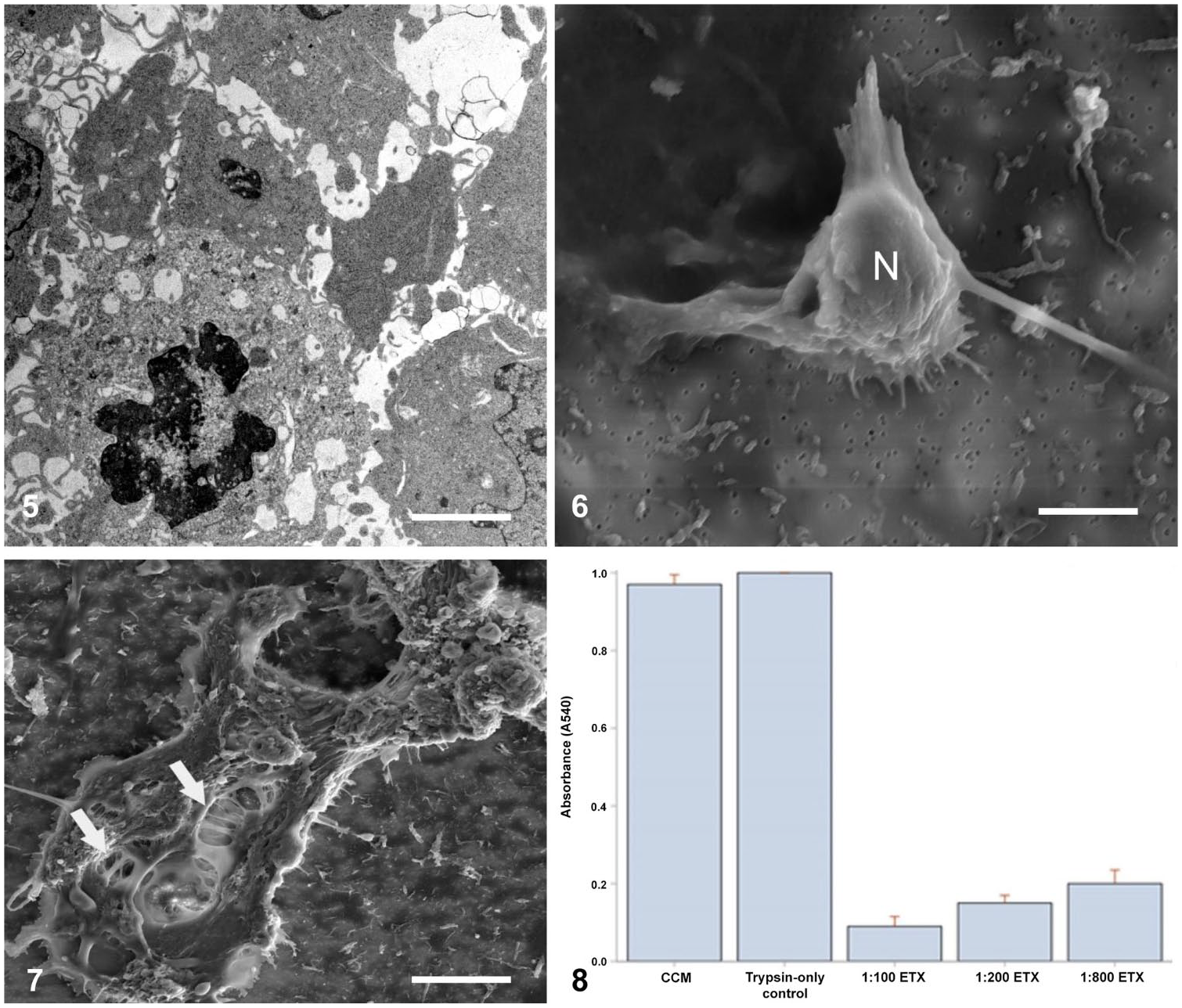
TEM image of cerebral capillary endothelial cells in culture exposed to ETX (1:200 dilution). Endothelial cells show marked condensation of nuclear chromatin and either shrunken, more electron-dense cytoplasm, or swelling of cytoplasmic organelles.
Control cerebral microvascular endothelial cells (Fig. 6) examined by SEM revealed a nucleus, which was rounded in outline with a prominent, raised profile, and various degrees of cytoplasmic spreading. In ETX-treated cultures (Fig. 7), the endothelial cytoplasm was often attenuated, shrunken, and more irregular in outline, with numerous large, variably sized, hollow cytoplasmic defects in the form of pits or craters, which exposed subendothelial structures; the nucleus was less conspicuous.
The effect of ETX on cell viability, as assayed by the MTT method, confirmed dose-dependent endothelial cell damage and depletion, which increased significantly with increasing toxin concentration (Fig. 8). Furthermore, trypsin-only controls demonstrated no significant difference when compared with complete media controls, thereby excluding trypsin as a possible adjunct cytotoxic agent. This result was confirmed by cell counts: control (198 ± 15), 1:800 dilution (122 ± 21), 1:200 dilution (90 ± 12), and 1:100 dilution (51 ± 15; data presented as means ± SD, with p < 0.0001 at each toxin concentration, compared with the control, ANOVA).
Pre-incubation of the endothelial cultures with a specific anti-ETX monoclonal antibody, prior to addition of the ETX-containing culture supernatant, abolished the cytotoxic effect of this toxin on endothelial cells, confirming the endotheliotoxic action of ETX.
Discussion
We have shown that ETX causes a direct and dose-dependent, cytotoxic effect on cultured human cerebral endothelial cells. Moreover, the light microscopic and TEM appearance of these injured endothelial cells suggested that necrosis rather than apoptosis was the likely mode of cell death, the nucleus often being pyknotic. Control endothelium examined by SEM resembled that reported previously. 6 The crater-like cytoplasmic defects produced by ETX have also been described in these cells in vitro under conditions of hypertension, ischemia, and hypoxia, 5 and after exposure to the neurotoxin, tunicamycin. 1
Although the endothelial cell line that we used was of human origin, we nevertheless demonstrated that ETX is capable of injuring brain endothelium directly in vitro and it is likely that it would similarly damage cultured cerebral endothelial cells from species known to be susceptible to EXT neurotoxicity, such as ruminant livestock, particularly sheep, and laboratory rodents. The susceptibility of human-derived brain microvascular endothelial cells to ETX is also important given that this potent neurotoxin has the potential to be used as a bioterrorism agent against human populations, 17 and it has been suggested that ETX might be associated with multiple sclerosis in humans. 14
The death of cultured brain microvascular endothelium exposed to ETX was also supported by the fact that: 1) this cytopathic effect was neutralized by a specific ETX monoclonal antibody; 2) the effect of trypsin, used to activate ETX, on these endothelial cells was no different from a control consisting of culture medium alone; and 3) the same toxin preparation caused identical experimental microvascular damage in the brain and retina in vivo3,4,8 as produced by naturally occurring ETX exposure.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
