Abstract
Trigonella foenum-graecum (fenugreek) is a legume widely used as a food supplement in humans and less frequently in ruminants. Toxicity has been described sporadically in ruminants grazing mature fenugreek plants or stubble; however, the pathological features are unclear. This report describes a natural outbreak of intoxication in cattle fed fenugreek straw and the experimental reproduction using 8 sheep and 8 goats. Affected cattle presented clinical signs approximately 1 month after consuming the straw and 100 of 400 cattle (25%) were affected, of which 60 of 100 (60%) died or were euthanized. Clinical signs were characterized by proprioceptive positioning defects with abnormal postures and weakness of hindlimbs. Forelimbs were also affected in severely affected animals, and cattle became recumbent. Locomotion was characterized by trembling, and some cattle showed high-stepping movements of their forelimbs and knuckled over in their fetlocks. Experimental intoxication induced clinical signs only in sheep and were similar to cattle, although with signs starting in the forelegs. Gross and microscopic lesions were similar in spontaneous and experimental intoxications. Macroscopic changes corresponded with muscular hemorrhages and edema, mainly surrounding the peripheral nerves. Microscopic examination only demonstrated lesions in the distal peripheral nerves, which included edema, hemorrhages, and Wallerian degeneration. Neurofilament immunohistochemistry revealed altered axon labeling and S100 showed a decrease in myelin intensity and loss of its typical compact arrangement around axons. Biochemical and hematological abnormalities included elevated levels of muscle and liver enzymes and thrombocytopenia. These findings indicate that fenugreek straw induces peripheral neuropathy in cattle and sheep, but not in goats.
Fenugreek is a legume widely cultivated in the Mediterranean region, North Africa, Yemen, and India, which is used for human food and as a medicinal plant due to its extensive pharmacological properties. 18 It is also used in livestock feed, although its use is more restricted due to the strong odor it imparts to milk and meat. Fenugreek can be used as seeds, leaves, and in animals feed as straw or stubble. Although fenugreek is generally considered safe, some studies have reported toxic effects. 14 However, few publications on toxic plants mention fenugreek. Due to its use as human food supplement, toxicity studies have been carried out in laboratory animals such as rodents and rabbits.18,19 These studies have mainly used various compounds and extracts from seeds and leaves and have shown several adverse effects, mostly related to reproductive disorders and neurotoxicity in the offspring.18,19 In mice, administration of fenugreek sprout seed extracts to pregnant females caused decreased fertility, abortions, fetal mortality, and behavioral and motor skills disorders in the offspring. 18 In this case, an increase of phenolic substances after seed germination was the suspected cause of the adverse effects. Oral administration of fenugreek seed powder to female rats caused a significant decrease in reproductive hormones, decreased ovarian weight, and oophoritis. 12 However, although its chemical composition is well described, the pathogenic mechanisms of its toxicity remain unknown. 19
In ruminants, fenugreek seeds have been used as a food supplement. In sheep, for example, fenugreek has been shown to increase milk production and stimulate appetite.3,22 Higher milk production has been demonstrated in buffaloes. 8 Nevertheless, a few cases of natural adverse effects are reported in association with the consumption of fenugreek straw.2,5,6,17 The first report came from Israel in 1958, where a locomotor disorder was described in cattle after consuming fenugreek straw for 8 weeks. 17 Subsequently, it was experimentally reproduced in cattle and sheep fed fenugreek straw.1,2 Adler et al 1 observed severe skeletal and cardiac muscle degeneration in 2 steers aged 6 and 12 months after eating straw for 3 weeks. The disorder was irreversible, even after straw withdrawal for 4 months, and was considered by the authors to be due to vitamin E deficiency. A locomotor disorder was also demonstrated in an experimental sheep after consuming fenugreek straw for 11 days. 2 These authors did not demonstrate involvement of the nervous system, although they admitted that they could not rule it out. Between 1980 and 2001, 3 outbreaks of a locomotor disorder were described in sheep grazing mature fenugreek plants or stubble in Australia. 5 In only 1 of these outbreaks, nervous lesions were reported microscopically. Spinal cord lesions were identified in acute cases, and Wallerian degeneration in peripheral nerves was identified in chronically affected sheep. In Spain, epidemiological evidence has associated fenugreek straw with a nervous disease in both cattle and sheep in some areas of northern Spain for several years (J. Zabala, unpublished data). 7 In the 1980s, an experimental study was carried out with a native sheep breed from the north of Spain described cardiac and skeletal muscle lesions, as well as spinal cord lesions, but did not report lesions in peripheral nerves. 7 A similar clinical and pathological syndrome with peripheral nerve lesions, named “kangaroo gait,” has been described in lactating sheep in New Zealand, Australia, and the United Kingdom.5,6
Although all these studies have reported similar clinical signs and the disease has been experimentally reproduced, pathological descriptions are incomplete and have shown different results. In cattle, the clinical and pathological findings have only been described in Israel; however, none of these reports studied the nervous system.1,17 In sheep, only 1 natural report has studied the peripheral nervous system. 5 In goats, no natural cases or experimental studies have been reported. The objective of this study is to describe the clinical, biochemical, and pathological results of a spontaneous outbreak of fenugreek straw intoxication in cattle and its experimental reproduction in sheep and goats.
Materials and Methods
Natural Outbreak in Cattle
A farm of extensively raised Limousine beef cattle developed a neurological disease associated with the consumption of fenugreek straw. Affected animals at different stages of the disease were neurologically examined according to a standardized protocol. 10 Necropsies were performed on 3 euthanized cattle in terminal stages of the disease, and samples were taken from all organs, including peripheral nerves, for histological and immunohistochemical studies. Blood was taken from 5 severely affected animals for hematological and biochemical analyses. For histology, samples were routinely processed, paraffin embedded, and sectioned at 4 µm. Staining methods included hematoxylin and eosin (H&E) and Klüver-Barrera. Immunohistochemistry was performed using the Dako Envision system in a semiautomatic stainer (Dako, Cytomation, Denmark), following the manufacturer’s instructions. This method is based on an HRP labeled polymer that is conjugated with mouse or rabbit secondary antibodies. Two primary antibodies were used, a monoclonal mouse anti-human neurofilament protein (IS607, clone 2F11) for axon staining and a polyclonal rabbit anti-S100 (IS504) for Schwann cells. Both antibodies were from Dako and were used undiluted. Antigen unmasking was performed on a Dako PT Link module; it was carried out at 960°C for 20 minutes, in a retrieval solution, at pH 9 (Dako, K-8010). The positive control was a nerve from a normal animal, and the negative control was the sample without the primary antibody, which was replaced by buffer. Hematology was performed in an automatic counter (Sysmex XT-2000i, Roche Diagnostics, Barcelona, Spain) calibrated for bovine values. Enzymes such as creatine kinase (CK), aspartate aminotransferase (AST), alanine aminotransferase (ALT), lactate dehydrogenase (LDH), and alkaline phosphatase (ALP) were analyzed using commercial tests, in an automatic analyzer (BA 400 Biosystem SA, Barcelona, Spain), according to the IFCC (International Federation of Clinical Chemistry and Laboratory Medicine). Total proteins and urea for renal function were also analyzed. The presence of aflatoxins in the fenugreek straw was ruled out. A competitive enzyme-linked immunosorbent assay for total aflatoxins (Code 14104, Biosystem SA, Barcelona, Spain) was used on several samples from the cattle farm and on the fenugreek used in the experimental study.
Experimental Study in Sheep and Goats
Eight adult sheep (4 males from 1 to 9 years old and 4 females from 1.5 to 6 years old) of the Rasa Aragonesa breed and 8 adult goats of mixed breed (2 males and 6 females) were fed fenugreek straw obtained from the cattle outbreak. Fenugreek straw mixed with wheat straw was fed for 1 week and then only fenugreek was given ad libitum. Females were supplemented with 300 g/kg of concentrate and males with 500 g/kg. Two sheep and 1 goat were kept as controls and fed wheat straw. The experiment lasted 3 months. At the end of the experiment, the fenugreek straw was removed. At that time, 2 sheep remained with clinical signs and were then fed only wheat straw.
All animals were bled at the beginning of the experiment, at the onset of the clinical signs, and at the end of the experiment for hematological and biochemical studies. Hematology was performed in some sheep and biochemistry on all animals. Analyses were performed as described above in cattle, on an analyzer calibrated for ovine and caprine values. Clinical signs were assessed daily and body condition weekly. A detailed neurological examination was performed on all animals with clinical signs, similar to that described in cattle. 10 Animals were classified into 3 groups based on the severity of the signs. Mild signs were considered at the onset of the clinical signs and corresponded to animals in which variable signs were restricted to the forelimbs, moderate when signs were observed in both forelimbs and hindlimbs, and severe when sheep were predominantly recumbent. The animals were euthanized depending on the severity of the clinical signs. Two sheep were euthanized with mild signs, one with moderate signs, and three with severe signs. Necropsies were performed and samples were taken for histopathological and immunohistochemical studies, as previously described. Special attention was taken with the peripheral nerves of the limbs, such as the radial and the sciatic.
All the experimental procedures of this study were approved by the Ethic Committee for Animal Experiments of the University of Zaragoza (Project license PI13/18; date of approval: 01 March 2018).
Results
Natural Outbreak in Cattle
Clinical disease was only observed in replacement cattle because fenugreek straw was not fed to feeder cattle due to the strong odor it imparts to the meat. Clinical signs were observed in 100 of 400 cattle (25%) fed fenugreek and appeared about 1 month after consuming the straw. According to the farmer, animals could have consumed about 100 kg/animal. No relationship with age was observed; however, the farmer fed more straw to the youngest and pregnant animals, and therefore these animals were the most commonly and severely affected. Pregnant animals did not present with reproductive problems and, according to the farmer, the fenugreek straw seemed to improve reproductive parameters, as evidenced by a decrease in the general rate of abortion and by a shorter time between calvings. About 60 of 100 affected cattle (60%) died or were euthanized because they were recumbent and unable to feed. Clinical signs were mainly characterized by proprioceptive positioning defects with abnormal postures. Weakness of hindquarters due to hindlimb paresis affecting one or both legs was observed in most animals. In severely affected animals, lameness also involved forelegs and cattle became recumbent. However, they did not lose consciousness and ate when offered feed. When forced to move, locomotion was characterized by trembling and the tail remained raised. Some affected animals showed high-stepping movements in their forelimbs when walking and typically knuckled over in their fetlocks (Fig. 1a, b). Some animals adopted a crouched position due to weakness of legs (Fig. 1a). The least affected animals recovered slowly over months when fenugreek was removed, although all remained permanently and variably disabled. They eventually had to be euthanized for welfare reasons or because they were not admitted to the slaughterhouse. Multiple treatments, including vitamins (B-complex vitamins, such as B1, B2, B3, B6, and B12; Vit D3), minerals (calcium and magnesium salts), digestive stimulants (dexpanthenol), and non-steroidal anti-inflammatory or analgesic drugs (tolfenamic acid, flunixin meglumin), were administered throughout the clinical period without response.
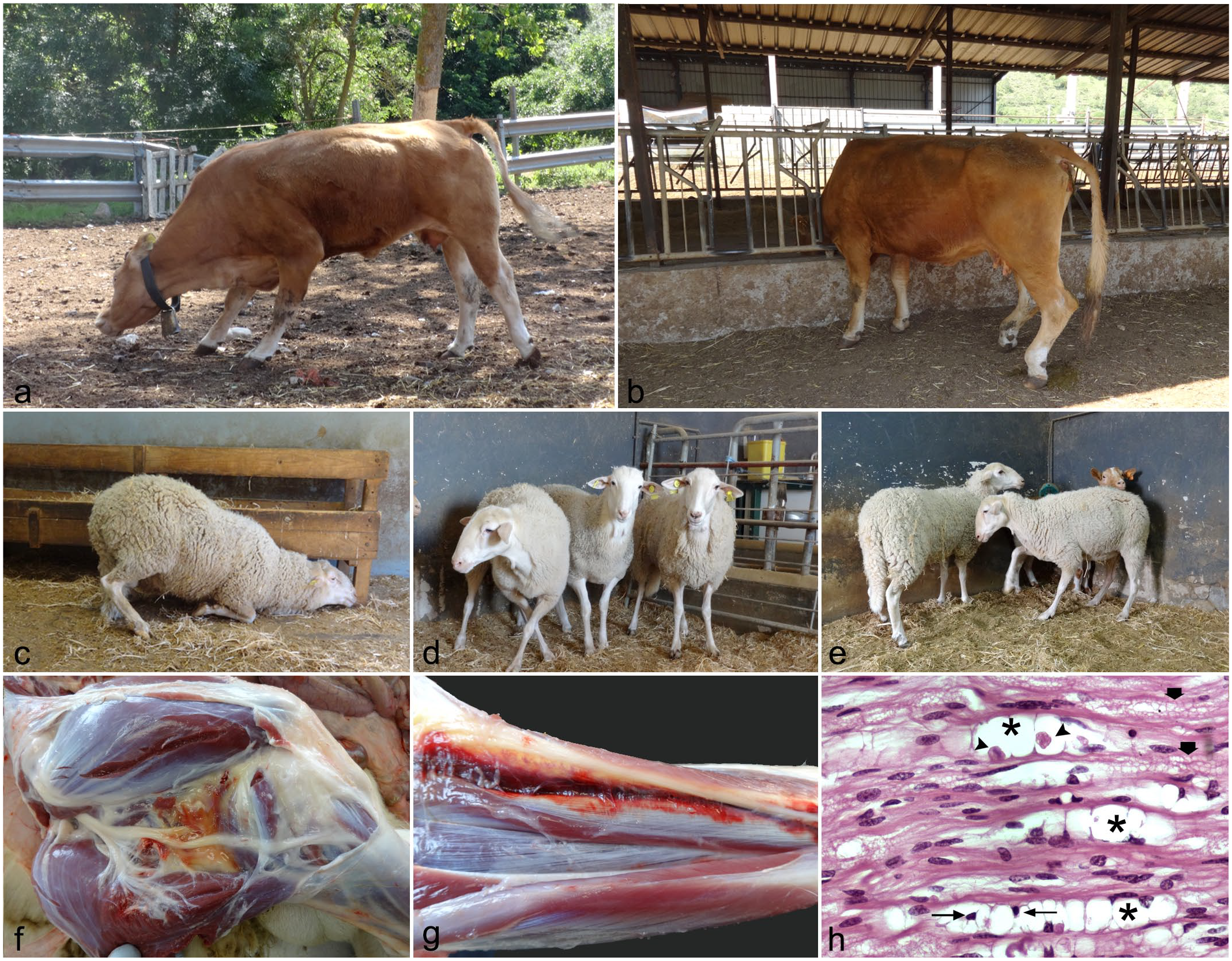
Fenugreek toxicosis in cattle (spontaneous) and sheep (experimental). (a) Intoxicated cattle showing a crouched position and slightly knuckled over in their hindlimb fetlocks. (b) Intoxicated cattle knuckled over in their hindlimb fetlocks and raised tail. (c) Intoxicated sheep in a kneeling position. (d) Intoxicated sheep with crossed forelimbs. (e) Intoxicated sheep with high step movements. (f) Intoxicated sheep with limb edema located in the intermuscular connective tissue and surrounding vascular vessels and nerves (bifurcation of the sciatic nerve into the tibial and peroneal nerves). (g) Severely affected ram with muscle hemorrhages in the forelimb. (h) Sciatic nerve (longitudinal section), affected cattle. Wallerian degeneration with digestion chambers (asterisks) containing granular, fragmented axonal debris (arrow heads) and macrophages (skinny arrows). Small, clear vacuoles within myelin sheaths represent an artifact of formalin fixation (thick arrows). Hematoxylin and eosin (H&E).
Hematological findings in the 5 cattle evaluated did not reveal significant changes. Mild leukocytosis was observed in 2 cattle, and all affected cattle evaluated showed a mild increase of neutrophils and eosinophils. A marked decrease in platelets was observed in 1 animal (Supplemental Table 1). All of these changes were considered unrelated to fenugreek toxicosis. Biochemical abnormalities in all animals evaluated were characterized by moderate to high increases in ALP, AST, and LDH. Creatine kinase was greatly increased in 2 animals (Supplemental Table 2).
Gross lesions in the 3 cattle evaluated included muscular hemorrhages and edema, mainly in the hindlimbs and surrounding peripheral nerves. Muscle atrophy was observed in the most severely affected animals. Microscopic examination in these cattle only demonstrated lesions in the distal peripheral nerves, especially the sciatic nerve, and corresponded with edema between nerve fibers and within individual fibers. In some sections, hemorrhages between nerves, primarily under the epineurium, were observed. The perineurium was separated from the endoneurium and individual nerve fibers by an edematous substance containing cell debris and myelin. Individual nerve fibers showed degeneration of isolated axons with vacuoles containing fragmented debris (Fig. 1h). Neurofilament immunohistochemistry revealed an irregular pattern of axon labeling in transverse and longitudinal sections of nerves, and S100 showed decreased myelin labeling with loss of its typical compact arrangement around axons, and an irregular and granular appearance.
In 1 animal, moderate multifocal granulomatous myositis associated with parasites compatible with Besnoitia besnoiti, suggesting chronic besnoitiosis, was observed in the muscles and nerves of the hindlimbs (Supplemental Figs. S1, S2). Cysts of Sarcocystis spp., without inflammatory reaction, were observed in hindlimb muscles of all cattle. In addition, mild to moderate cholangiohepatitis and small foci of interstitial mononuclear hepatitis were observed in all animals.
Experimental Study in Sheep and Goats
Clinical signs were only observed in sheep. No signs were detected in goats or control animals throughout the experiment. The sheep showed the first clinical signs between the fourth and the sixth week after the start of the experiment (a summary of the onset of clinical signs and their severity can be seen in Supplemental Table 3). Three rams showed signs in the fourth week (24, 26, and 28 days post-exposure [dpe]), 3 ewes in the fifth week (30 and 33 dpe), and the last 2 sheep in the sixth week (36 dpe). Clinical signs started with an altered gait associated with progressive weakness of the forelimbs. Some sheep exhibited bruxism. Some sheep knuckled in their fetlocks, especially in the forelimbs. Some sheep knelt down for a while (Fig. 1c) and others crossed their forelimbs (Fig. 1d). Clinical signs progressed to hindlimb weakness and eventually protracted recumbency with weight loss. When animals were forced to stand up, they showed severe ataxia. In addition, extension and flexion of distal limbs were abnormal, with high-stepping movements (Fig. 1e). The clinical expression was variable between sheep, with some animals showing severe clinical signs at the beginning and others mild. Likewise, the clinical evolution was variable, with some animals showing a rapid evolution and others slower progression. After 3 months of consuming fenugreek, the straw was withdrawn. At this time, there were 2 moderately affected sheep that progressively recovered. A slight recovery was observed in the fifth day after withdrawn, and on the 30th day, the sheep were on their feet, although they still had sporadic falls. At the second month, no nervous clinical signs were evident and the sheep only showed a decrease in weight. Six months after removing the straw, recovery was complete.
Neurological examination showed no mental alteration (prosencephalon) or any other intracranial sign (brain stem or cerebellum). Spinal reflexes were present, and a central nervous system (encephalon and spinal cord) disorder was ruled out. A disorder of the peripheral nervous or muscular system was suspected due to the presence of the following signs: decreased spinal reflexes, abnormal but present postural reactions, weakness-associated recumbency, and generalized hyperesthesia in all 4 limbs that would explain antalgic postures such as crossed forelimbs and occasional dysmetria.
Gross findings were mainly observed in sheep with moderate and severe clinical signs and were characterized by limb edema located in the intermuscular connective tissue and surrounding vasculature and nerves (Fig. 1f). In 2 rams with severe clinical signs, subcutaneous and muscle hemorrhages were observed distally in the forelimbs that were considered, at least in part, to be secondary to recumbency (Fig. 1g). No significant lesions were observed in the other organs.
Microscopically, lesions were similar to those observed in cattle (Fig. 1h), with edema, hemorrhages (Fig. 2a), and Wallerian degeneration and axonopathy in the most distal areas of the peripheral nerves. Digestion chambers and fragmented axons (Fig. 2b) were multifocally distributed and evident in severely affected sheep. In the sheep No. 8, with mild clinical signs, nerve lesions were scarce, patchy, and difficult to find. Lesions were located mainly in the radial nerves in mild and moderate cases and in the sciatic, tibial, and peroneal nerves in severe cases. Neurofilament immunohistochemistry (Fig. 2c, e) showed irregularly labeled axons and S100 (Fig. 2d, f) loss of myelin around the axons. Myelin was fragmented and granular. No significant lesions were observed in the other organs.
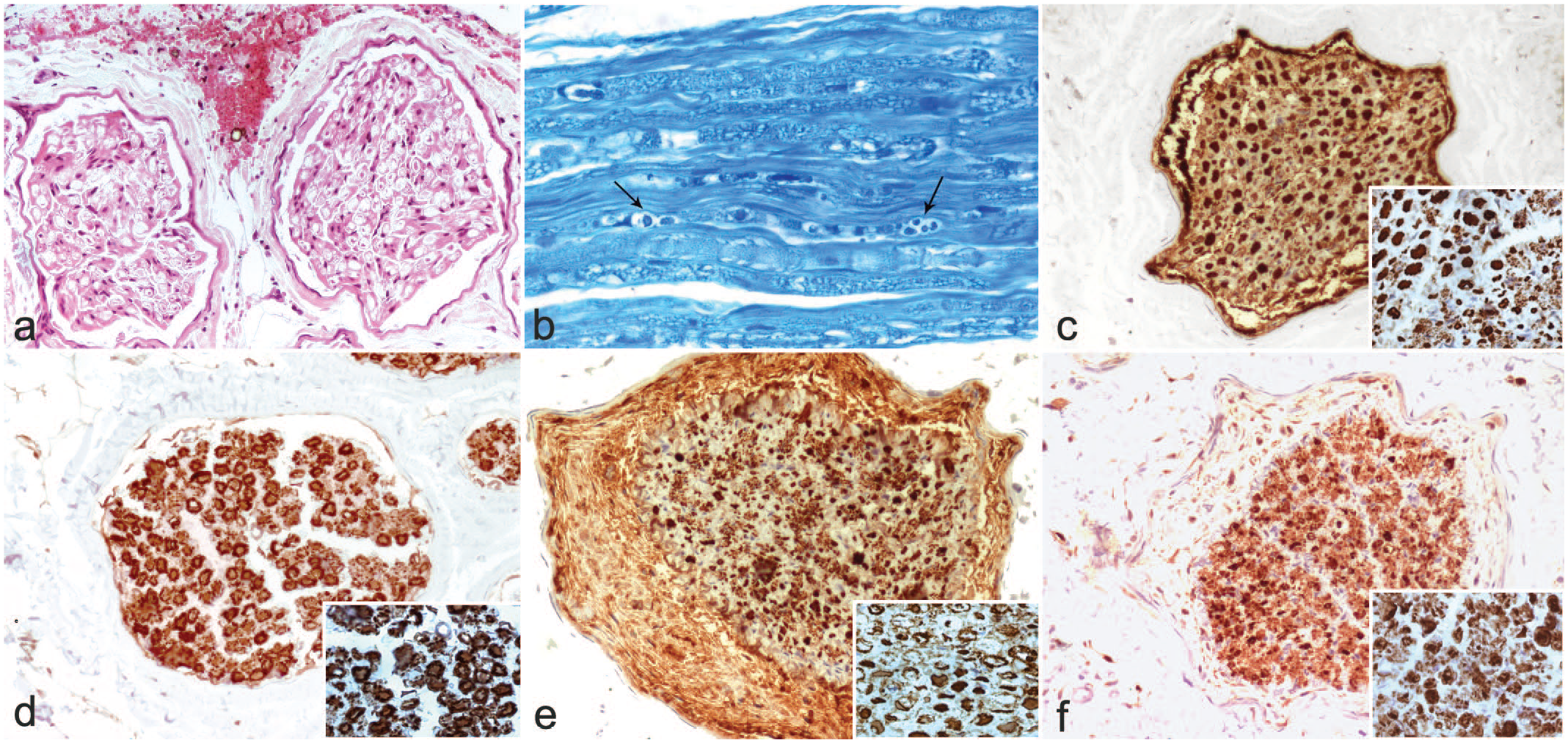
Fenugreek toxicosis in sheep (experimental). Microscopic and immunohistochemical findings. (a) Nerve fascicle with hemorrhages under the epineurium. H&E. (b) Radial nerve (longitudinal) with digestion chambers and fragmented myelin (arrows). Klüver-Barrera. (c) Radial nerve (cross section) from a mildly affected sheep showing a near normal appearance of the axons. Slight edema is observed around the nerve. Inset: well-preserved axons and area with smaller unmyelinated axons in the lower right corner. Immunohistochemistry for neurofilament. (d) Radial nerve (cross section) from a mildly affected sheep showing a near normal appearance of myelin. Inset: myelin showing a normal appearance surrounds axons. Immunohistochemistry for S100. (e) Radial nerve (cross section) from a severely affected sheep showing a highly fragmented appearance of the axons. Note the edematous substance containing cell debris and myelin that separates the perineurium from the endoneurium. Inset: degenerated axons with loss of labeling, more evident in the center. Immunohistochemistry for neurofilament. (f) Radial nerve (cross section) from a severely affected sheep showing a reduced and disorganized myelin. Note the edematous substance that separates the perineurium from the endoneurium. Inset: the myelin is granular and irregular. Immunohistochemistry for S100.
Hematological findings in sheep (Supplemental Table 4 for males and Supplemental Table 5 for females) revealed no significant changes. Biochemical findings included moderately elevated creatine kinase when clinical signs started and less levels when animals were euthanized, except for sheep No. 7 (Supplemental Table 6 for males and Table 7 for females). Sheep No. 6 showed a very surprisingly high level of CK at the onset of clinical signs, but the levels were slightly elevated 1.5 months after fenugreek withdrawal. No significant hematological or biochemical abnormalities were detected in goats (data not shown).
Discussion
The present study has shown that fenugreek straw can cause a nervous disease in cattle and sheep, but not in goats, which is associated with degeneration of the peripheral nerves of the limbs. It has also revealed that there are species differences in the clinical presentation of intoxication in cattle and sheep, and these results are consistent with the few reported natural and experimental studies of fenugreek straw toxicosis in ruminants.1,2,5,6,17 While progressive hindlimb paresis is observed in cattle, forelimbs are affected in sheep. Affected cattle and sheep usually tend to knuckled over their fetlocks with a protracted final recumbency in the later stages of the disease. Involvement of the central nervous system is not typically seen.
However, the appearance and duration of clinical signs seems to depend on the ruminant species and the natural or experimental conditions. Our experimental study in sheep showed the onset of the clinical signs 4 to 6 weeks after consuming the straw, with a progressive course that ended in final recumbency. These findings differ from the experimental study carried out by Adler and Egyed, who observed the first clinical signs 11 days after consuming fenugreek straw; however, they are similar to the natural cases reported by Bourke, in which sheep grazing fenugreek stubble or unharvested crop showed clinical signs 5 to 8 weeks after starting to graze.2,5 These differences could be explained by the amount of fenugreek straw ingested in each case. In the outbreak described by Bourke, only 8% to 12% of sheep grazing the fenugreek developed neurological signs, suggesting variable consumption. 5 In our study, all sheep ate only fenugreek straw and all developed signs. A surprising finding in the natural cases is that some sheep did not develop signs until 2 to 4 weeks after stopping grazing. 5 The present study, in contrast, has shown that withdrawal of fenugreek straw triggered a progressive recovery in moderately affected sheep; recovery was complete after 6 months, suggesting that fenugreek intoxication may be reversible. In natural cases, both acute and chronic presentations have been reported. 5 Acutely affected sheep become recumbent for several days before death, and chronically affected sheep show clinical signs for 6 to 12 weeks. 5 This probably reflects the different severity of the disease, as severely affected sheep become recumbent and eventually die because they are unable to eat. Our experimental study also evidenced a variable severity of the condition among the sheep, both at the onset of signs and throughout the study. Males were more severely affected already at the beginning. No sex differences in susceptibility to fenugreek toxicosis, however, have been reported.
In cattle, on the other hand, comparisons are difficult because only 1 report of natural intoxication has been published so far. 17 Clinical signs started after 8 weeks of consuming fenugreek straw, while in the present case it was after 3 to 4 weeks. An interesting finding from our study is that the appearance and severity of clinical signs in cattle seemed to depend on the amount of straw ingested. Pregnant and young replacement cattle received the greatest amount of straw and were the most severely diseased. This observation agrees with the results observed in sheep.
Differential diagnosis of neuromuscular dysfunction in cattle may include mineral or vitamin deficiencies, such as hypocalcemia, hypokalemia, hypomagnesemia, or vitamin E deficiency; poisoning such as organophosphate toxicity, toxic-related myopathies; and plant poisoning such as plants of the cycad family that induce a distal axonopathy in the spinal cord with hindlimb ataxia.9,11 The initial description considered vitamin E deficiency as a possible cause; this was also suggested by subsequent experimental reproduction in sheep.7,17 However, our results were not consistent with these observations as characteristic muscular lesions were not observed. In addition, in this outbreak, several mineral treatments were utilized without response.
In the cattle outbreak, no reproductive problems were observed. This is not consistent with experimental studies in rodents that have shown that fenugreek seed or leaves can induce reproductive problems, including hormonal dysfunctions, abortions, and delayed development of the nervous system in offspring. 19 These findings suggest that the toxic or beneficial effects may depend on which part of the plant is consumed, the experimental protocols used, or the animal species. 19 In this regard, our study has shown that fenugreek straw could be fed to goats, although more studies are needed to demonstrate a possible improvement in production parameters or its safety after longer periods of ingestion.
In goats, no references of fenugreek intoxication, either natural or experimental, have been published. This is consistent with our experimental study that demonstrated no clinical signs in goats after 3 months of fenugreek straw feeding, suggesting that this species is resistant to poisoning or needs longer periods of straw consumption to develop clinical signs.
Gross lesions of fenugreek intoxication are typically few or absent in both cattle and sheep and are dominated by hemorrhages and edema of the limbs.1,5,7,17 The earliest reports only showed muscular hemorrhages that microscopically corresponded to degeneration. 2 Nervous lesions were not reported and muscle findings could not be confirmed as primary lesions. The gross lesions observed in our study suggested vascular lesions characterized by muscular hemorrhages and edema in the distal legs. These lesions were considered a consequence of recumbency. In our study, the only biochemical changes were in muscular parameters that may also be due to the recumbent position. These results are consistent with biochemical studies in affected sheep in Australia that did not support a primary myopathy. 5
Microscopic studies of the central or peripheral nervous system in cattle intoxicated with fenugreek have not been published.1,17 In sheep, only 3 reports have been published studying the nervous system, 1 experimental and 2 natural cases, showing variable results.5,7 However, only 1 of these studies reported peripheral nerve lesions. 5 Our study has shown that lesions are consistently found in the peripheral nervous system in both cattle and sheep, and are similar in nature, corresponding to Wallerian degeneration. The lesions appear to depend on the clinical stage of the disease. In acutely affected sheep from a natural outbreak, Bourke described lesions only in the spinal cord, with no lesions in the brain. 5 These lesions were consistent with acute edema and were primarily characterized by mild to moderate Wallerian degeneration in the ventral motor tracts and less severe degeneration in the dorsolateral sensory tracts, swollen astrocytes, spongy changes in the neuropil, and prominent blood vessels, occasionally surrounded by lymphocytes. However, the lesions described in the chronic cases were only observed in the peripheral nervous system, without lesions in the brain or spinal cord. The lesions also corresponded to Wallerian degeneration, being severe in the radial nerve and mild in the peroneal nerve. Cuervo and Oregi, in an experimental intoxication, studying only recumbent sheep, observed lesions in the brain and spinal cord. 7 Although the median and sciatic nerves were also sampled, no lesions were reported. These researchers mainly detected groups of basophilic neurons with nuclear pyknosis, gliosis, sometimes around degenerated neurons, and spongiosis with scattered prominent blood vessels in the brain and spinal cord, similar to the spinal cord lesions described by Bourke in acute cases. 5 These authors also observed cerebellar lesions characterized by degeneration of Purkinje cells and loss of neurons of the granular layer. Cerebellar lesions have not been reported by any other author. 5 In the spinal cord and the neuromuscular junction, Cuervo and Oregi observed degeneration of motor neurons and Wallerian degeneration, consistent with the chronic cases reported by Bourke.5,7
Muscular involvement, either cardiac or skeletal, in fenugreek intoxication is intriguing. While cardiac and skeletal muscle lesions were not reported in the natural cases of sheep in Australia, it was a relevant finding in the experimental studies reported in Spain and Israel, and in bovine cases in Israel.1,2,5,7 In these cases, the researchers considered cardiac failure as the cause of the death and severe muscular lesions as the cause of the clinical signs.1,2,7 However, in Australia, biochemical studies of affected sheep did not support a primary myopathy. 5 In our case, no cardiac lesions were found in sheep or cattle, and the skeletal lesions were consistent with secondary recumbency, as suggested by Australian authors. 5 Cuervo and Oregi, 7 also reported marked vascular lesions, mainly characterized by vacuolization of the medial layer of the arterioles. These lesions were considered relevant, explaining the hemorrhages observed macroscopically. However, they have not been found in our study and have not been reported in other studies.1,2,5,17
In our case, the outstanding lesions were observed in the peripheral nervous system in both cattle and sheep, with no involvement of central nervous system and were considered the cause of the clinical signs. Nerve lesions corresponded to axonopathy and Wallerian degeneration and were similar to those reported by Bourke in chronically affected sheep. 5 Although some pyknotic neurons in brain and spinal cord were observed in our study, they were considered a non-significant lesion, without clinical consequences. However, they were a prominent finding in the study of Cuervo and Oregi. 7
Pathology of the peripheral nervous system can be classified into 3 patterns according to the primary target: neuropathy, axonopathy, and Wallerian degeneration.13,16 Distinguishing the primary location may suggest some aspects related to its etiology and pathogenesis; however, discriminating the primary target can be difficult because reciprocal influences between the axon and myelin can lead to degeneration of both structures in chronic cases. 13 Our immunohistochemical studies demonstrated axonopathy and Wallerian degeneration in the most distal areas of the peripheral nerves of the limbs, mainly in the sciatic nerve in cattle and the radial nerve in sheep. Neuropathy was not observed, as the spinal cord did not show lesions. Axonopathy has been described in experimental intoxications, but not in natural cases.5,7 In the latter, only Wallerian degeneration has been reported. 5
The pathogenesis of fenugreek-associated disease is unknown, and direct intoxication or an induced deficiency has been suggested.1,5,7,17 In the first reports in cattle and sheep, vitamin E deficiency was suspected due to the lesions observed in the cardiac and skeletal muscle; however, in our study and in the Australian cases, the findings were not consistent with this deficiency. Moreover, in the natural outbreak in cattle reported in this case, treatment with various minerals and vitamins failed to elicit a response. Alternatively, an interruption in nerve impulse conduction might occur. Disruption may occur due to slight trauma or neuropeptide dysfunctions. 15 In mild cases, as described in neurapraxia cases, motor functions are more affected than sensory functions, leading to muscle weakness without loss of sensitivity. 15 Some plant poisonings can block nervous impulses. Phalaris toxicity causes a locomotor disorder in ruminants similar to that observed in fenugreek toxicosis, which is due to an alkaloid with serotonergic receptor activity. 4 In this poisoning, brown discoloration is observed in some areas of the central nervous system. A delayed neuropathy has been described in organophosphate poisoning with clinical signs and distal peripheral nerve lesions similar to fenugreek toxicosis. 20 Toxic effects are related to acetylcholinesterase interference and are reported in large animals but not in rodents, suggesting that differences in axon length may be the reason. 21 Fenugreek has been shown to have anti-cholinergic activity. 19 Between the 2 anti-cholinergic activities (antimuscarinic and antinicotinic), the clinical signs observed in fenugreek toxicosis suggest an antinicotinic activity with blockade of nicotinic acetylcholine receptors, triggering muscle weakness. In addition, it is consistent with the reversible character, and its effects diminish once the causative agent has been eliminated. Interestingly, in the most severe cases, muscle weakness can be the cause of recumbency and this could lead to secondary damage to the peripheral nerves of the limbs. Secondary limb neuropathy has been demonstrated in downer cow syndrome. 20 Immunohistochemical studies of neuropeptides and their receptors in the spinal cord and neuromuscular junctions may shed some light on fenugreek pathogenesis.
In summary, the present study shows that fenugreek straw induces peripheral neuropathy in beef cattle and sheep, but not in goats. Furthermore, this study shows that clinical signs may be reversible in sheep, with complete, albeit slow, recovery in moderately affected animals, although cattle may remain permanently disabled.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221137020 – Supplemental material for Peripheral neuropathy caused by fenugreek (Trigonella foenum-graecum) straw intoxication in cattle and experimental reproduction in sheep and goats
Supplemental material, sj-pdf-1-vet-10.1177_03009858221137020 for Peripheral neuropathy caused by fenugreek (Trigonella foenum-graecum) straw intoxication in cattle and experimental reproduction in sheep and goats by Bernardino Moreno, Belén Marín, Alicia Otero, Mirta García, Helen Raksa, María I. Guijarro, María Climent, Mariano Morales, Javier Zabala, Juan M. Loste, Cristina Acín and Juan J. Badiola in Veterinary Pathology
Footnotes
Acknowledgements
We thank the farmer and veterinarians of the cattle farm for providing information on the clinical case and for providing the fenugreek straw for the experimental intoxication in sheep and goats.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by the “Centro de Encefalopatías y Enfermedades Transmisibles Emergentes” and the Research Group “Enfermedades priónicas, vectoriales y zoonosis emergentes (Ref. A05_17R).”
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
