Abstract

Clinical History
In December in South Tyrol, Italy, a 5-year-old multiparous Brown Swiss dairy cow was referred for multiple solid dermal and subcutaneous nodules. The animal was housed with 37 cattle of 3 different breeds (Brown Swiss, Holstein Friesian, and Simmental). None of the cattle had prior clinical signs.
Gross Findings
The nodules were alopecic and in some cases ulcerated. They grew rapidly to cover the entire thoracic and abdominal skin (Fig. 1), part of the skin of the udder and hind limbs, as well as the conjunctiva and the vaginal mucosa (Fig. 2). Circular superficial cutaneous plaques were also noted on the body of the cow. No other animals in the herd were affected. On physical examination, the animal showed a good body condition score (score 3/5) and a generally healthy condition. There was no loss of appetite, and the body temperature, heart rate, and respiratory rate were within normal limits.
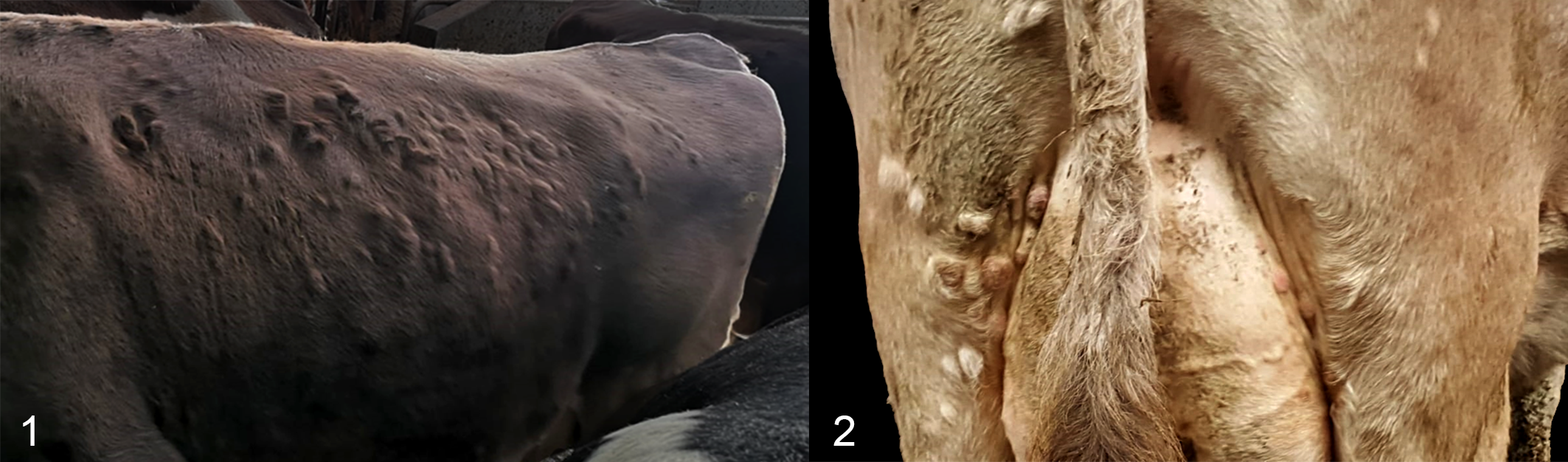
Cutaneous nodules, cow.
Differential Diagnosis
Although the disease was presumed to be noncontagious because only a single cow was affected, and despite Italy having enzootic bovine leukosis (EBL)-free status, bovine leukemia virus (BLV) and bovine herpesvirus-2 infections were considered as differential diagnoses, due to the clinical signs of multiple nodules and the emergence of numerous circular superficial cutaneous plaques. Lumpy skin disease was considered because it causes firm cutaneous nodules up to 5 cm diameter, although Italy is officially free of this disease. Other neoplastic and infectious diseases with cutaneous lesions, such as chronic lymphocytic leukemia, dermatophytosis, and cutaneous actinobacillosis 1 were also taken into consideration.
Laboratory Results
A complete blood count on K3EDTA-anticoagulated whole blood was performed using an impedance method with fluorescence flow cytometry (XN-1000V Hematology Analyzer, Sysmex). Erythrocytes and leukocytes counts were within the reference ranges. 3 The mean corpuscular hemoglobin (MCH) and red blood cell distribution width (RDW) values were slightly above the reference range, and platelet aggregates (pseudothrombocytopenia) were detected and confirmed by microscopic evaluation of a Wright Giemsa–stained blood smear. Serology was negative for serum antibodies to bovine leukemia virus (ID Screen BLV Blocking Gp51, ID.Vet Innovative Diagnostics, Grabels, F) and bovine herpesvirus-2 (ID Screen BHV-2 Indirect, ID.Vet Innovative Diagnostics, Grables, F).
Microscopic Findings
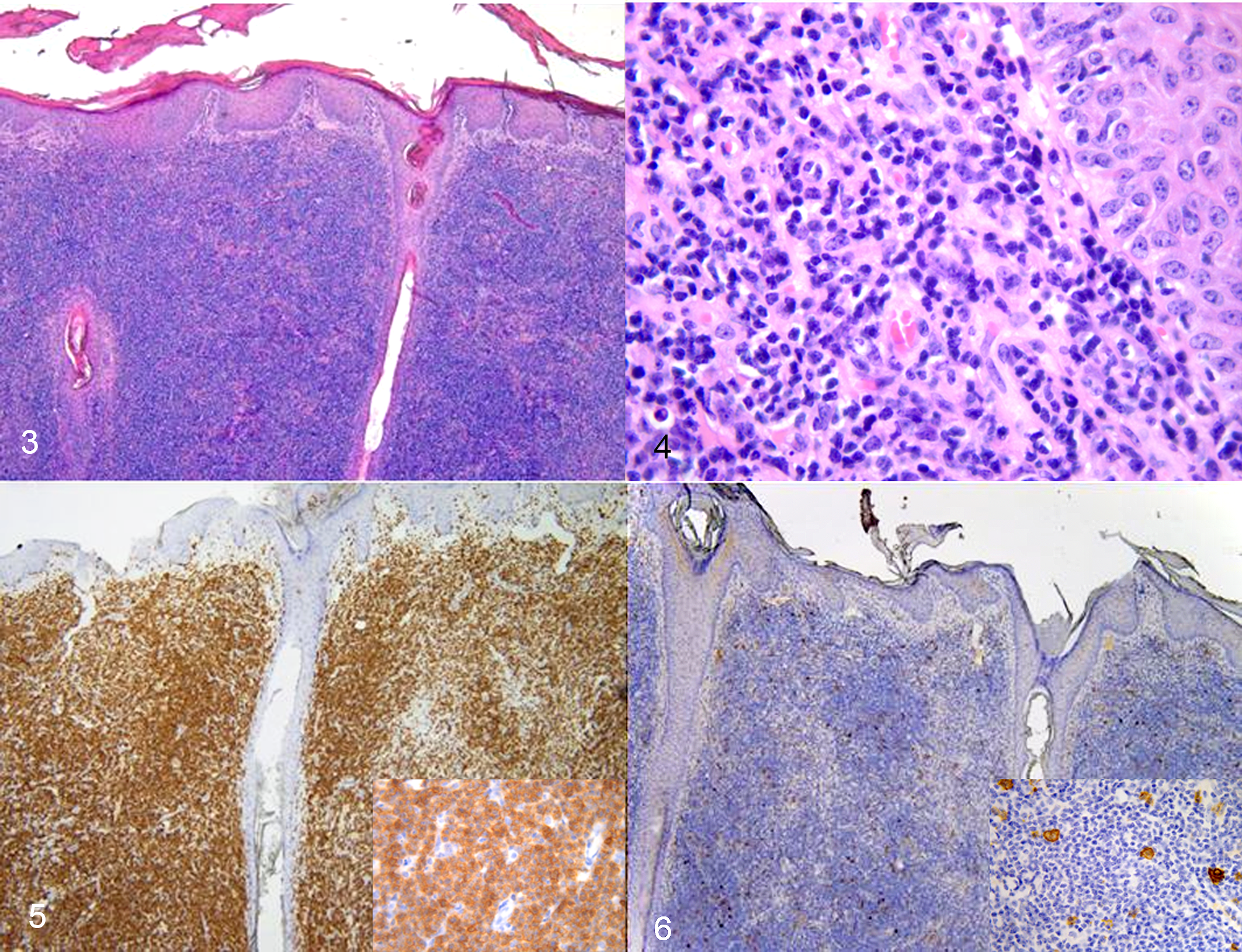
One skin nodule was excised under local anesthesia, fixed in formalin and processed for histological examination. Histologically, the nodule consisted of a dense dermal infiltrate of round cells with multifocal extension into the subcutis. The cells were characterized by small (approximately 5–6 μm), dark and occasionally cleaved nuclei and scant pale cytoplasm. The mitotic activity was about 1 to 2 mitoses in a single high-power field (0.31 mm2). Epitheliotropism was not clearly observed (Figs. 3, 4). Based on the histologic lesions, a diagnosis of a lymphoproliferative process was made.
Cutaneous nodules, cow.
Further Investigations and Diagnosis
Immunohistochemistry (IHC) was performed on 3-μm-thick tissue sections using an automated immunostainer (DiscoveryULTRA; Ventana, Medical Systems), with 2 polyclonal antibodies: CD3 (Dako) applied at 1:100 dilution for 24 minutes at 37 °C, and CD20 (Thermo Scientific) at 1:200 dilution for 60 minutes at 37 °C. An UltraView Universal DAB Detection Kit (Roche) was used as a detection system. Sections were counterstained with Mayer’s hematoxylin. Positive and negative controls were included in the run. There was diffuse membranous immunolabeling of neoplastic cells for CD3 (Fig. 5), while only scattered cells were CD20-positive (Fig. 6).
The histopathologic and immunohistochemistry findings indicated a diagnosis of T-cell lymphoma, highlighting, along with the other clinical findings, a cutaneous form of sporadic bovine lymphoma (SBL). 5 Although epitheliotropism is a reported feature of SBL, it was not clearly observed in this case. On the other hand, BLV is known to cause B-cell lymphoma (enzootic bovine leukosis). 6
A TaqMan-based real time qualitative PCR (rPCR) assay performed on whole blood was negative for lumpy skin disease virus. To evaluate the presence of viral particles, the skin lesion was examined by transmission electron microscopy using a negative staining technique. The sample was homogenized, subjected to 2 freeze-thaw cycles, centrifuged at 4500 × g for 20 minutes, and the supernatant was centrifuged at 10 621 × g for 20 minutes. Supernatant (0.1 mL) was collected and recentrifuged at 90000 × g (Airfuge A100, Beckman) on formvar-coated copper 200 mesh grids (Electron Microscopy Sciences), stained with 2% sodium-phosphotungstate. 9 No viral particles were detected.
According to the laboratory results, microscopic findings, and further investigations, a diagnosis of cutaneous form of SBL was made.
Since the animal was neither contagious nor in manifest pain, and the milk production and the general health status were considered acceptable as well, it was not culled. It was therefore possible to follow the evolution/involution of the nodules. During the 4 following months the nodules gradually disappeared, leaving a necrotic-like area; this was considered a natural regression of the clinical signs. During the same time span, no other cow in the herd showed any similar cutaneous clinical signs, confirming the probable noncontagious nature of the disease and thus its consequent diffusion among the other animals in the stable. Twelve months after the first manifestation of clinical signs, the animal gave birth to a healthy calf. To the best of our knowledge, both the cow and its calf remain healthy and have not developed any manifestation of discomfort or illness.
Discussion
The case was finally diagnosed as a cutaneous form of SBL in a cow. The disease was presumed to be noncontagious, although EBL, BLV, bovine herpesvirus-2 infections, and lumpy skin disease were considered as differential diagnoses based on the clinical signs. Similarly, neoplastic and infectious diseases such as chronic lymphocytic leukemia, dermatophytosis, and cutaneous actinobacillosis were also considered 1 but were ruled out by serology and transmission electron microscopy, and the hematological values did not support bacterial or parasitic infections or leukemia.
Based on histopathology, a diagnosis of a lymphoproliferative process was made but this was not sufficient for ruling out EBL. 2 Along with IHC findings, indicating a diagnosis of T-cell lymphoma, it was possible to state that the clinical findings were indicative of a cutaneous form of SBL. 5
As indicated by this case, SBL not only affects juvenile and young cattle (usually under 1 year of age), but the cutaneous form can affect cattle at all ages with localized skin lesions and occasional generalized lymphoadenopathy. 4 In comparison to the thymic and calf forms, the cutaneous form presents a less aggressive evolution: as for the current case, a natural regression of clinical signs is reported, 7 although this may represent a waning phase, after which the disease may return and involve peripheral lymph nodes and internal viscera. 10
This case highlights the diversity and variety of clinical signs in SBL, and the unique importance of considering diseases other than
Footnotes
Acknowledgements
The authors are grateful to all the staff involved both in field as well as in the laboratory activities. Mrs. Sally Ellis Jones is kindly acknowledged for the English revision of the paper, too.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
