Abstract
Standardization of tumor assessment lays the foundation for validation of grading systems, permits reproducibility of oncologic studies among investigators, and increases confidence in the significance of study results. Currently, there is minimal methodological standardization for assessing tumors in veterinary medicine, with few attempts to validate published protocols and grading schemes. The current article attempts to address these shortcomings by providing standard guidelines for tumor assessment parameters and protocols for evaluating specific tumor types. More detailed information is available in the Supplemental Files, the intention of which is 2-fold: publication as part of this commentary, but more importantly, these will be available as “living documents” on a website (www.vetcancerprotocols.org), which will be updated as new information is presented in the peer-reviewed literature. Our hope is that veterinary pathologists will agree that this initiative is needed, and will contribute to and utilize this information for routine diagnostic work and oncologic studies. Journal editors and reviewers can utilize checklists to ensure publications include sufficient detail and standardized methods of tumor assessment. To maintain the relevance of the guidelines and protocols, it is critical that the information is periodically updated and revised as new studies are published and validated with the intent of providing a repository of this information. Our hope is that this initiative (a continuation of efforts published in this journal in 2011) will facilitate collaboration and reproducibility between pathologists and institutions, increase case numbers, and strengthen clinical research findings, thus ensuring continued progress in veterinary oncologic pathology and improving patient care.
Reproducibility is the cornerstone of experimental science 126 but irreproducibility of published oncological studies is a crisis in human 14 and veterinary oncology, although we rarely attempt to replicate our research. 80,110 In many instances the lack of reproducibility stems from inadequate description of methods in published studies. 14,88,108,126 Efforts to address this crisis have been implemented in human medicine, including attempted reproduction of published studies and formulation of checklists for use by journal editors and reviewers to ensure inclusion and transparency of detailed methods and materials in publications. 41,88,100,108,126 Less than 10% of observational studies are able to be replicated, and incredibly, less than 20% of preclinical trials can be replicated. 14 If that is the state of oncology studies in human medicine, how do we compare in veterinary oncology? How far have we advanced in the last 40 years?
Attempts to validate existing studies and grading schemes in veterinary oncology are almost nonexistent. Grading schemes and the methods employed require appropriate validation prior to their being adopted and used to provide prognoses or to direct clinical therapy. Yet the philosophy in veterinary pathology seems to be that once a system or method is created, it is put into use and remains in use regardless of whether the system has been validated or not. We do not know how that system or method will perform when different pathologists use it and when it is applied to different populations of patients. Consensus statements that support the use of published grading schemes are not validation. Authors should feel complimented when colleagues attempt to reproduce their methods and study designs. Results will not replicate exactly, but methods must. Validation of grading systems is impossible if the original methods cannot be duplicated by other investigators because methods are flawed or inadequately described. Other limitations stem from inadequate sample sizes from restricted populations without validation in broader populations, leading to overinterpretation of small effects that are statistically significant but perhaps clinically insignificant. Validation of results by multiple institutions in larger, more diverse populations increases the likelihood that findings will have greater prognostic utility and clinical impact. The focus should be on determining parameters that provide proven clinical utility and can stand the test of time.
These issues were brought forward in 2011 through a series of articles in Veterinary Pathology. The present article is a continuum of those efforts but is focused on establishing standardized histopathology methods to evaluate tumors and to develop tumor protocols that will accrue data so that, over time, large data sets with comparable information can be evaluated to enable meaningful conclusions and accurate prognostic information. In this article, we identify key areas for improvement of veterinary oncology studies. We illustrate the problem by starting with an overview of 2 fundamental concepts—method reproducibility and outcome assessment.
Reproducibility of Methods in Veterinary Oncology Studies
One of the major reasons that published studies are not able to be reproduced is that the methods lack sufficient details to allow other users to accurately assess histologic parameters including mitotic figure (MF) recognition, mitotic count (MC), lymphovascular invasion, tumor necrosis, and margin evaluation. 80,100 Currently, the assessment of these parameters requires pathologists to make subjective evaluations without clearly defined methods. Due to the inconsistency of these qualitative evaluations, there can be poor concordance among pathologists, thus preventing meaningful evaluation of grading systems. The Materials and Methods section of published papers should contain descriptions of each method in sufficient detail to allow others to duplicate the methods, reproduce the study, and validate the results. Citing that the methods described in a prior published study were followed is acceptable, providing that any modifications used are described in sufficient detail. However, to promote effective application of methods by other investigators, a summary of the essential components of the methods is needed. Failure of studies to be replicated can be the result of poorly described methods, or not following the methods.
When interobserver variation between pathologists is reported in articles, it is often ascribed to a method that is too subjective (eg, determining degree of atypia or degree of differentiation). However, investigators may not have considered that the original methods were inadequately described, such that pathologists could not consistently follow the method. Stating that MF were counted in 10 consecutive high-power fields (hpf) at 400× is insufficiently detailed for others to reproduce the method, compare results, and validate the data. Since the area within 1 hpf can vary by greater than 200% because of different microscope configurations, then of course there will be interobserver variation in MC if the microscopes used by study pathologists were not configured in the same way. 21,44,81 Not only MC, but all parameters that were enumerated histologically with a microscope or with whole slide images (WSIs) have the potential for error and variability of results if the area enumerated is not defined in mm2. Tumor parameters which have not been described in adequate detail in most articles include neoplastic cell pleomorphism, tumor necrosis, and tumor differentiation. Even with standardized methods, there will be variability that needs to be reported and practical considerations that cannot be standardized.
Standardizing methodology is one step toward consistent results. The methods must be followed carefully, using the same materials (eg, antibodies for immunohistochemistry [IHC]). The methods must be validated by applying them to groups of animals with well-defined outcomes. Accredited clinical pathology labs use standardized methods, they calibrate their instruments, and they use quality control procedures to measure and report the reproducibility, sensitivity, specificity, and reliability, including positive and negative predictive values of test results. Similar principles need to be applied to anatomic pathology, either in the context of pathologists scoring tumor grading parameters using a light microscope, or quantitation of morphologic structures by automated methods. Both of these approaches need to be validated with rigid standards. 24 Once the methods are shown to be reproducible, the next step is to apply them to cases with known outcomes (recurrence, metastasis, survival metrics). Development of interlaboratory proficiency programs to promote accuracy of tumor grading and consistency of ancillary testing is sorely needed. When standardization between laboratories is not possible (eg, when using IHC platforms), then internal validation of such assays becomes important. After the method is validated using cases with known outcomes, only then can the methods be credibly applied to assessment of patient samples. It is important to benchmark a new assay against measures of clinical relevance, and not just equivalent to the current standard diagnostic practices, which may be flawed or can be improved upon.
Technology will continue to spur development of new methods that can be applied to diagnostic cases. It would be helpful if new technologies such as computational pathology (CPATH) to count microscopic structures, or molecular tests, were compared to the existing more subjective parameters (eg, MC, margins, necrosis, nuclear pleomorphism, determined on hematoxylin and eosin [H&E] stained slides). 46 Then, colleagues can compare results, costs of tests, required expertise, and technology to determine the feasibility of adopting the new method. Furthermore, consideration of the cost and infrastructure involved in using newer techniques relative to any improvement in patient prognosis and care are essential to developing a practical approach to diagnostic pathology.
As novel tests are developed, the predictive value of these new tests should be compared to traditional parameters so diagnosticians and oncologists can offer owners the best advice in test selection. Traditional histological or cytological parameters will have subjective limits, but their value is that they can be done by pathologists in almost any location, without specialized equipment or expertise and therefore have the advantage of being widely available in virtually all diagnostic laboratories at a reasonable cost. Furthermore, these traditional parameters and the diagnosis of tumor type help clinicians to decide if and what additional tests are needed (eg, staging, nodal evaluation, molecular or genetic tests). Some animal owners will pay for new techniques at any cost, but others will decline based on other considerations, a few of which are cost of treatments, age of the pet, or concerns of how treatments may affect the quality of life of their pet. None of these are direct factors in how the tumor behaves yet may influence survival statistics used by oncologists and other owners to make important clinical decisions. Molecular diagnostics have had a major impact on tumor evaluation in human medicine and similar tests need to be developed and applied to animal tumors in order to improve the care of our patients and potentially as animal models of human tumors.
Although new methods may initially be restricted to the institutions in which they were developed, standardization and proficiency are critical as these techniques are validated in other laboratories and become routinely used for tumor diagnosis. Centers that develop novel tests (eg, CPATH, artificial intelligence, molecular assays, genetic tests) should have a goal that the methods can be applied uniformly and are described in sufficient detail that other labs can perform and validate the tests. Newly developed, specialized assays should undergo technical comparison to current methods and should be validated for their prediction of patient outcomes to assess their utility. 17,24,92
Assessment of Clinical Outcomes
In addition to a lack of standardized assessment of histologic criteria, reproducibility in veterinary pathology/oncology studies is also limited by a lack of standardized guidelines for outcome assessments of animal patients. 100,125 Factors to be discussed in relation to outcome assessment include analytical methods to define survival data, the accuracy in determining tumor recurrence and metastasis, and approaches to increasing the availability of study populations with well-validated outcomes. A thorough discussion of study design with guidelines on choosing appropriate statistical tests is available through an initiative of the American College of Veterinary Pathologists’ Oncology Committee, and readers are encouraged to familiarize themselves with this information, as well as consulting with a statistician prior to initiating a study. Applying appropriate statistical analyses (multivariable vs univariable) to robust data points is essential to help determine clinical relevance. Prospective studies and studies that standardize treatments are recognized needs. Retrospective studies are plagued with confounders such as different treatments, different doses of chemotherapy or radiation therapy (RT), concurrent medicines, different entry points into the study, incomplete patient records, and changing treatment methods in some patients as a form of rescue therapy for those that are not responding as hoped. When patients are treated, it is essential to group them by the same treatment modalities. Lack of standardized methods in a study compromises reliability of results. Prospective studies are preferred as they can be designed to avoid some of the confounders inherent in retrospective studies, but they are rare in veterinary oncology.
Euthanasia unrelated to tumor progression is a significant confounder. Reported patient survival times are affected by euthanasia, which may be elected due to personal decisions, varying judgements regarding quality of life, owner income, or other factors that do not reflect tumor behavior. In this way, the subjective decision to euthanize introduces variability into survival data, which might reduce the ability to detect an association of grade and survival if one truly existed. How often has an assigned grade or reporting that a tumor in a lymph node resulted in euthanasia and the patient may have lived significantly longer? In this way, different study groups (animals assigned different tumor grades) have intrinsically different likelihoods of being euthanized, and this differential effect is likely to confound the observed survival in animals with low- versus high-grade tumors. Thus, survival time statistics in veterinary oncology are influenced by euthanasia as well as other factors unrelated to tumor and host biology.
Practical decisions such as these support the need for oncology studies to rely on parameters such as disease-free interval, time to tumor progression, recurrence, metastases, and survival rates at defined years posttreatment rather than overall survival times. The start time (T = 0) of survival studies needs to be clearly and consistently defined in the materials and methods section of articles. 86 There are a variety of potential events to establish start time: the day of first treatment is recommended (surgery, chemotherapy, RT) but options include the date of clinical diagnosis, the date of surgical tumor removal, and the date of histopathological tumor diagnosis. 86 This is an example of why the materials and methods section of articles need detailed descriptions of methods, so others can understand what was done and perhaps replicate the study design for validation.
Metastasis needs to be reported as confirmed or suspected. When metastases are identified by diagnostic imaging, they should be reported in journal articles as suspected or metastases as determined by imaging. Light microscopy is required to confirm that metastases are present and are of the same tumor type as the primary tumor. Multiple aggressive tumors can occur in the same patient and are well recognized in breeds such as Golden Retriever, Rottweiler, and Bernese Mountain Dogs. 33 Divergent tumor types may also develop in animals undergoing radiation therapy, underscoring the need to confirm tumor type of suspect metastatic lesions. Local recurrence is defined as the presence of the same tumor within the region of the previous surgical site confirmed via histopathology or cytology. Histopathology is preferred; however, tumor type influences which technique is used and, more important, what is deemed best care for the patient. Future studies should attempt to resolve the variation seen in metastatic and local recurrence rates reported in the current literature, many of which lack histologic lesion confirmation and/or autopsy data. Oncology studies no longer routinely include results of autopsy, 32,109 the perceived value of which seems to have hit a nadir. Client permission to perform autopsies should be pursued with sympathy and empathy but as vigorously as other tests. Autopsy findings greatly increase the objectivity of determining recurrence and metastases, reduce the variability of data, and increase the likelihood of identifying differences between groups, increasing the confidence in study results.
Veterinary oncology studies need institutions and labs to pool their resources so that large numbers of cases with well-validated clinical outcomes can be collected. If these cases have consistent clinical interventions (eg, same therapy) for specific tumor types and accurate patient outcomes, the archived materials are a precious resource. The materials from these studies (histopathology slides, paraffin-embedded tissue blocks, digitized images, statistical data) could be shared with others so that their new methods can be applied to the existing case series with known outcomes. This was done in a study of canine oral and lip melanocytic neoplasms. 16 In addition to a set of oral/lip melanocytic neoplasms from the authors’ diagnostic laboratory, this study also used archived blocks from 2 previous studies. 46,105 The histologic parameters described in those previous studies (nuclear atypia, MC, and pigmentation) were applied to the new case material using the same methods, and these were compared to a new parameter, Ki67 index. This can serve as an example of the value in validating prior studies and introducing a new method which adds confidence to conclusions and makes the well-validated outcomes available for comparisons to a new method. When evaluating existing or new prognostic parameters, it is important to separate the samples on which the original parameters were used (eg, the training set) from an independent set of samples (eg, the test set). Thus, the study’s conclusion is validated on a completely separate patient cohort. Ideally, a separate institution (or multiple institutions) could do these validation studies which would also allow assessment of interlaboratory variability.
Accurate outcome assessment is critical in determining if a therapy is effective or if a test or grading scheme is prognostic. It should be recognized that clinical outcomes may differ between tertiary and primary care patients, and this should be factored into the analysis of the data. Standardization of outcome assessment data is as critical as the standardization of methods involved in tumor assessment. These 2 components of oncology must be linked, because it is useless to create a new tumor grading system without knowledge of patient outcomes.
Supplemental Materials (Guidelines and Protocols)
In 2011, Veterinary Pathology published a series of recommendations and reviews about tumors in animals and how they should be evaluated. The articles in that issue are excellent. They exceed the goals of the present article, but like all journal articles there is no provision to update information, which is now a decade old. Much of the information consisted of literature reviews, and descriptions of the multiple methods to assess a parameter, without prioritizing or choosing one. The present article aspires to be a continuum of the information published in 2011 but with a focus on establishing standardized histopathology methods to evaluate tumors. Here we present Guidelines that represent best practices on how to measure specific parameters and Protocols for how to evaluate specific neoplasms. For selected Guidelines and Protocols, a summary is provided within this article, and additional details are given as Supplemental Materials (available on the journal website). The topics include the following: MC (Guideline 1, Supplemental File 01), Histologic Morphology of Mitotic Figures (Guideline 2, Supplemental File 02), Tumor Margins (Guideline 3, Supplemental File 03), Lymphovascular Invasion (Guideline 4, Supplemental File 04), Tumor Necrosis (Guideline 5, Supplemental File 05), Synoptic Reporting in Veterinary Medicine (Guideline 6, Supplemental File 06), Assessment of Clinical Outcomes (Guideline 7, Supplemental File 07), Lymph Node Analysis (Guideline 8, in progress), Pleomorphism (Guideline 9, in progress), Metastasis (Guideline 10, in progress), Computational Pathology (Guideline 11, Supplemental File 08), Canine Skin and Subcutaneous Soft Tissue Tumors (Protocol 1, Supplemental File 09), Canine Cutaneous Melanoma (Protocol 2, Supplemental File 10), Canine Oral Melanoma (Protocol 3, Supplemental File 11), and Abbreviations and Definitions (Appendix 1, Supplemental File 12).
These Guidelines and Protocols will help accrue standardized data so that studies can be better validated and more effectively compared. The Guidelines and Protocols will be posted on a website (www.vetcancerprotocols.org). The online resource is intended to be a dynamic document that is updated as the described techniques, methodology, and definitions evolve from substantial data-driven results. As guidelines, protocols, and the appendix are updated, each new version will be dated and assigned a version number. The website will list the current version, provide notifications to denote changes, archive old versions, and list changes between versions. Means to cite the website contents—guideline, protocol, and appendix versions—will be posted. Colleagues are invited to contact communication author(s) with suggestions for edits or updates to ensure the guidelines and protocols remain a relevant and useful centralized website. Changes to guidelines and protocols will be made by communication authors and/or an advisory board review. Any updated versions of these documents will be dated, and references cited (see details below). The present system of waiting for publication of a book or a fascicle is outdated. Updates are also needed because errors are possible in the present Guidelines and Supplemental Materials, or it is possible that some important references were missed. The authors hope that readers will bring such errors to our attention by contacting one or more communication authors listed in each of the Supplemental Files; errors or omissions can be quickly and easily updated on the website. Journals will remain vital to our professions as they publish peer-reviewed research and books are valuable because they describe entire topics in detail.
These initiatives attempt to provide focused and current assistance to diagnostic pathologists and clinicians, researchers developing new studies, and journal editors and peer reviewers evaluating manuscripts for publication, as a checklist to ensure that all required data were included, and standard methods were followed. The Guidelines and Protocols describe best practices in assessing the standard parameters used to evaluate tumors and checklists of information that should be gathered about specific tumor types when performing studies. Since the checklists are too extensive for daily diagnostic workflow, a shortened version containing key components and current grading system parameters will be posted on the website in a synoptic format. Some of the Guidelines are for immediate use for diagnosticians to ensure that MF, MC, tumor margins, lymphovascular invasion (LVI), tumor necrosis, and outcome assessments are evaluated in a standardized fashion. The synoptic reporting guidelines (Supplemental File 06) provides a means to communicate, retrieve, and archive essential data. 95 Other Guidelines are newly developed (CPATH) or in progress (lymph node evaluation, pleomorphism, metastasis). At the end of each Supplemental File is a section titled “Future Considerations,” which provides a list of possible ways to improve that method. The Protocols were modeled after those of the College of American Pathologists Protocols and are designed to gather uniform and complete data for the evaluation of neoplasms of domestic animals. Protocols are provided for canine soft tissue tumors/soft tissue sarcomas (STT/STS), canine cutaneous and oral melanomas, and are in process for other tumors. If we do not standardize the methods used to characterize tumors, we will continue to have conflicting data in the literature, hampering progress in understanding tumor behavior and impeding advances in veterinary cancer treatment. Complete data sets and rigorous statistical analysis of the data is needed to identify the criteria which are prognostic. These prognostic criteria need validation. The ultimate goal of standardizing oncology methods (pathology, molecular, clinical) should be to improve patient care.
What are the consequences of not following a Guideline or Protocol? Nothing: no certification or plaques of accomplishment will be awarded or rescinded. They are intended to be “best practices” for our methods and will benefit us and our clients: clinicians, oncologists, surgeons, patients, and their owners. Guidelines and Protocols extend beyond “best practices” as they provide brief literature reviews, areas of weakness, and suggested fields of investigation for future studies to improve a method. The Guidelines and Protocols are based on review of the literature and on the authors’ expertise; they are intended to bring consistency and reproducibility to the evaluation of tumors in animals. They have not been certified by any standards-creating body and they represent the authors’ own interpretation and application of the data reviewed. Whether a governing body will aid in further development in updating these Guidelines and Protocols will depend on the success of the website and how widely it is used. Application of these Guidelines and Protocols may vary for different laboratories and pathologists, depending on the availability of equipment, technical expertise, and on the nature of their caseload.
How widely will the website be used? The authors will use a website analytics platform to determine the number of visitors to the website, time spent on the site, and the number of downloads of the Guidelines and Protocols. This allows determining the approximate geographic location of visitors based on the network they connect from, providing an idea of the level of engagement of stakeholders. We will also create a Facebook group that will link to the site and allow discussion between colleagues as well as providing an opportunity for feedback and sharing of material.
Future Collaboration
A second goal of this initiative is to accrue data on the important parameters that should be evaluated for a specific tumor type so that large data sets with comparable information about these tumor types can eventually be evaluated to provide accurate prognostic information that improves patient care. This will require the collaborative participation of many investigators at various institutions worldwide, specialists from different disciplines, and particularly the involvement of early-career investigators. Edits and updates are encouraged: contact the communication authors of a Guideline or Protocol as listed in the Supplemental Files to make a suggestion or join an author group. Submission of new Guidelines and Protocols are welcomed and can be initiated by contacting the administrators of the website. Guidelines in the future might cover cytologic protocols, cellular and nuclear pleomorphism, proliferative indices, molecular profiling, genetic tests, lymph node evaluation, and checklists for surgical pathology reports. Protocols are needed for many tumors: think of a malignant tumor, or tumors with an existing grading system and write a protocol. Use the current protocols as a template and the existing literature to identify what needs validation and improvement. There is unanimous agreement on the need for Guidelines to standardize parameters and their assessment. However, it is more difficult to achieve agreement on the specific details for each Guideline and Protocol. To compare data between labs, and ultimately to improve patient care, we need to apply the same methods when we evaluate tumors. Using unstandardized methods causes variation in results and is not scientifically or ethically sound.
To facilitate interlaboratory data sharing the authors propose establishment of an international veterinary cancer biobank. Biobanks in human medicine are repositories of human samples that facilitate investigations to advance scientific knowledge and improve patient care 30,89 There are a multitude of these facilities archiving a host of sample types as well as clinical and laboratory data. These biobanks promote international, interinstitutional research by permitting use of the samples and data for research endeavors, allowing comparison between studies, and advancing the pace of medical research. There are veterinary biobanks dedicated to genomic preservation and research of livestock animals and to genomic preservation of wild animals 55 The Cornell University Veterinary Biobank focuses on the investigation of genetic disorders. (https://www.vet.cornell.edu/departments/centers/cornell-veterinary-biobank). The Pfizer-Canine Comparative Oncology and Genomic Consortium biobank is a repository focusing on the molecular level of canine cancer. 75 In contrast to the focus on molecular studies of canine cancer, we propose a biobank to advance veterinary oncology by focusing on diagnostic oncological pathology, housing histological and cytological digital images of tumors and associated clinical records forming a resource for diagnostic investigations. The images and corresponding clinical and laboratory data will be housed at the Veterinary Cancer Center within the Davis/Thompson Foundation and tissue blocks and glass slides will be housed at The Joint Pathology Center, Silver Spring, Maryland. A biobank committee will seek donations of samples and data from published (or soon-to-be published) studies. Samples and data residing in the biobank would then be available for others to use in validation of studies of new prognostic factors, grading schemes, and so on, and can be used to consolidate data from institutions around the globe to increase the total number of patients in a study. Validating studies with robust numbers of cases with different pathologists will increase the likelihood that the results will have longevity and clinical impact.
To encourage multi-institutional studies, we hope that the formation of a veterinary cancer biobank will promote collaboration and inclusion among pathologists and institutions and keep the focus on advancing veterinary oncological pathology. Additionally, deans, department chairs, and division leaders that evaluate employees should consider expanding and redefining how “scholarly excellence,” and advancement of company or college goals are recognized beyond “primary authorships.” All authors in large collaborative studies need to be recognized for their contributions or it may jeopardize participation in multi-institutional studies, which could promote lack of shared resources. Publications that use multi-institutional resources, or consensus reports and meta-analysis, are examples of studies that have many authors, all of whom need recognition. Deans, department chairs, and heads should consider means to encourage and reward investigators who participate in multi-institutional studies and utilize shared resources. Employers should encourage this participation and develop reward systems for scholarship by all authors, not just primary authors. Furthermore, the use of eponyms to identify a method should be replaced by the name of the guideline used to establish the diagnosis or parameter, with the correct guideline version cited in reports and publications.
Journals have requirements for authorship that must be followed. Additionally, journals, editors, and reviewers of manuscripts are gatekeepers for scientific integrity and can establish recommendations or requirements for specific topics. For example, journals should require or strongly recommend that oncology studies that include pathologic assessment of tumors have a pathologist as a coauthor. One or more authoring pathologists should review the case materials to ensure consistency of diagnoses, parameters, and methods. This is critical for studies that combine data from multiple labs with different pathologists, especially when the cases were collected over a period in which the methods or criteria to diagnose tumors may have changed. Communication authors of guidelines or protocols can be contacted to help find a pathologist if needed.
The authors are trying to integrate our concepts, guidelines, and protocols with colleagues at universities, diagnostic laboratories, governmental labs, pharmaceutical companies, residency coordinators, and certifying examination boards nationally and internationally. We are seeking input from as many stakeholders as possible and individuals at any of these institutions are encouraged to reach out to us and we will be contacting them as well. Diagnostic labs across the world are a tremendous resource of case materials and expertise. They are major employers of veterinary pathologists and their collaboration will benefit this project and other initiatives.
Summaries of the Guidelines and Protocols follow this introduction. Readers with an interest in a specific Guideline or Protocol should access the supplemental file or the updated version on the website. At the end of each section in the current manuscript there is a listing of references relative to that section in a textbox; with a complete list of references at the end of the manuscript.
Conclusions
The goal of this project is to help advance veterinary oncology and pathology by promoting standardization of tumor assessment and patient outcomes. This article attempts to establish best-practice reporting standards for diagnosticians and methodological standards for those undertaking prognostic studies using pathology data. Guidelines and Protocols are proposed to increase the uniformity and consistency by which tumors are evaluated, along with suggestions to improve their discrimination and utility in the future.
Key stakeholders each contribute to progress in standardizing tumor assessment and improving the reproducibility of studies.
Editors and reviewers can use checklists provided in the Supplemental Files to ensure that the methods for measuring MC, necrosis, margins, and LVI are sufficiently detailed and objective to be reproducible.
Investigators can design and execute their studies using these guidelines and protocols as a checklist. Data obtained from record review without knowledge of the diagnostic or grading criteria may not be accurate and therefore the study conclusions are of questionable validity. One or more authoring pathologists should review gross descriptions of tumors and glass or digitized histologic sections. The investigators must ensure that best practices are used to assess histologic parameters, and that the methods are described with sufficient detail and objectivity such that the data can be generated in a uniform and valid manner. Recording data objectively should integrate well with synoptic reporting.
Diagnostic pathologists can use the information provided in the Guidelines and Supplemental Materials to critically evaluate the validity of published studies and decide which if any methods should be incorporated into their daily practice. They can help identify the core prognostic parameters from the complete lists in protocols and they can identify features in guidelines that need adjustments. These core criteria can then be added to the workflow of diagnostic pathologists.
Oncologists can utilize this information to correlate with patient response and to develop prognoses and design studies to assess patient outcomes. This article and the associated website is a window for clinicians to see how their perspectives on assessment of tumors might differ from those of the pathologist: fascial planes are not the same to the surgeon as they are to the pathologist, a metric as basic as a high power field is not standardized, and histologic tumor-free distances will not be accurate if tumor margins are not inked by the surgeon.
Patients will ultimately benefit from advancement in the knowledge of tumor behavior and treatment efficacy.
We propose this initiative as a platform to gather new data and integrate it into an evolving system of best practices for tumor evaluation. The ultimate goal is identification and validation of parameters that are maximally prognostic and predictive for aggressive tumors. This will ensure that we are providing sound, factual information to clinical veterinarians to enable them to offer the best treatment options and formulate the most accurate prognoses for their patients and clients. How long will it take to accomplish all of this is unknown, but we need to continue and expand upon what our colleagues started in 2011.
References: Introduction, Standardization
14. Begley CG, Ellis LM. Raise standards for preclinical cancer research. Nature. 2012;
16. Bergin I, Smedley R, Esplin D, et al. Prognostic evaluation of Ki67 threshold value in canine oral melanoma. Vet Pathol. 2011;
17. Bertram CA, Aubreville M, Gurtner C, et al. Computerized calculation of mitotic count distribution in canine cutaneous mast cell tumor sections: mitotic count is area dependent. Vet Pathol. 2020;
21. Bonert M, Tate AJ. Mitotic counts in breast cancer should be standardized with a uniform sample area. Biomed Eng Online. 2017;
24. Boyce RW, Dorph-Petersen KA, Lyck L, et al. Design-based stereology: introduction to basic concepts and practical approaches for estimation of cell number. Toxicol Pathol. 2010;
30. Coppola L, Cianflone A, Grimaldi AM, et al. Biobanking in health care: evolution and future directions. J Transl Med. 2019;
32. Crownshaw AH, McEntee MC, Nolan MW, et al. Evaluation of variables associated with outcomes in 41 dogs with incompletely excised high-grade soft tissue sarcomas treated with definitive-intent radiation therapy with or without chemotherapy. J Am Vet Med Assoc. 2020;
33. Cullen JM, Breen M. An overview of molecular cancer pathogenesis, prognosis, and diagnosis. In: Meuten DJ, ed. Tumors in Domestic Animals. 5th ed. John Wiley; 2017:1–26.
41. Checklists work to improve science. Nature. 2018;
42. Ehrensing G, Craig LE. Intravascular neoplastic cells in canine cutaneous plasmacytomas. J Vet Diagn Invest. 2018;
43. Ellis D, Srigley J. Does standardised structured reporting contribute to quality in diagnostic pathology? The importance of evidence-based datasets. Virchow Arch. 2016;
46. Esplin D. Survival of dogs following surgical excision of histologically well-differentiated melanocytic neoplasms of the mucous membranes of the lips and oral cavity. Vet Pathol. 2008;
55. Groeneveld LF, Gregusson S, Guldbrandtsen B, et al. Domesticated animal biobanking: land of opportunity. PLoS Biol. 2016;
75. Mazcko C, Thomas R. The establishment of the Pfizer-canine comparative oncology and genomics consortium biospecimen repository. Vet Sci. 2015;
80. Meuten D, Munday JS, Hauck M. Time to standardize? Time to validate? Vet Pathol. 2018;
81. Meuten DJ, Moore FM, George JW. Mitotic count and the field of view area: time to standardize. Vet Pathol. 2016;
86. Nguyen SM, Thamm DH, Vail DM, et al. Response evaluation criteria for solid tumours in dogs (v1.0): a veterinary cooperative oncology group (VCOG) consensus document. Vet Comp Oncol. 2015;
88. Oransky I, Marcus A. Project to replicate landmark cancer studies meets mixed results. Accessed April 15, 2021. https://www.statnews.com/2017/2001/2018/replication-cancer-studies/
89. Patil S, Majumdar B, Awan KH, et al. Cancer oriented biobanks: a comprehensive review. Oncol Rev. 2018;
92. Puri M, Hoover SB, Hewitt SM, et al. Automated computational detection, quantitation, and mapping of mitosis in whole-slide images for clinically actionable surgical pathology decision support. J Pathol Inform. 2019;
95. Reagan JK, Selmic LE, Fallon C, et al. Evaluation of information presented within mast cell tumour histopathology reports in the United States: 2012–2015. Vet Med Sci. 2018;
100. Schott CR, Tatiersky LJ, Foster RA, et al. Histologic grade does not predict outcome in dogs with appendicular osteosarcoma receiving the standard of care. Vet Pathol. 2018;
105. Spangler WL, Kass PH. The histologic and epidemiologic bases for prognostic considerations in canine melanocytic neoplasia. Vet Pathol. 2006;
108. Stark PB. Before reproducibility must come preproducibility. Nature. 2018;
109. Stefanello D, Avallone G, Ferrari R, et al. Canine cutaneous perivascular wall tumors at first presentation: clinical behavior and prognostic factors in 55 cases. J Vet Intern Med. 2011;
110. Stefanello D, Buracco P, Sabattini S, et al. Comparison of 2- and 3-category histologic grading systems for predicting the presence of metastasis at the time of initial evaluation in dogs with cutaneous mast cell tumors: 386 cases (2009–2014). J Am Vet Med Assoc. 2015;
125. Webster J, Dennis MM, Dervisis N, et al. Recommended guidelines for the conduct and evaluation of prognostic studies in veterinary oncology. Vet Pathol. 2011;
126. Wen H, Wang HY, He X, et al. On the low reproducibility of cancer studies. Nat Sci Rev. 2018;
Guidelines 1 and 2. Mitotic Count and Morphology of Mitotic Figures
For additional details, see Supplemental File 01–Mitotic Count—and Supplemental File 02—Morphologies of Mitotic Figures.
Mitotic count (MC) will remain an important parameter in the evaluation of tumors as it is easy to accomplish, incurs no additional costs, is predictive of tumor proliferation, and is part of multiple grading schemes that help predict tumor behavior. However, certain components essential for performing reproducible MC must be defined including the region of the tumor where MC should be performed (ie, “hot spot” or areas of highest mitotic density within a tumor 5,12,81,83,119,122 ) and the amount of tumor area in which mitotic figures (MF) will be counted, expressed in standard units of measure (ie, mm2). 81 Standardizing our methods should increase reproducibility, decrease subjectivity, and hopefully improve the predictability of this commonly used and practical prognostic marker leading to improved patient care. Although enumerating MF has long been a mainstay of tumor assessment, until recently there has been no standardization of any element of this parameter in veterinary pathology. Perhaps, the assumptions were that we were all counting the correct structures with the same method, that these methods matched published articles and therefore there was no need to standardize the technique. Unfortunately, some of those assumptions are false. Data from articles that did not define the area in which MF were counted or other details used to determine MC need to be repeated. Performing the MC is considered laborious and subjective with interpathologist variation. 19,82,116,122 Possible causes include counting differently sized areas, poorly defined methods, not following methods, counting too rapidly, counting ambiguous structures, experience level, tumor mitotic heterogeneity, inability to find hotspots, quality of sections (fixation, artifacts), and quality of images (for digital microscopy).
To achieve accurate and consistent counts the MC must be performed carefully following standardized procedures; when this is done, consistent counts can be achieved by pathologists. 119 After we follow standardized methods, these criticisms should be reevaluated for manual and automated MC. MC can be determined by partially automated means, using artificial intelligence (AI; more specifically, deep learning-based algorithms). 7,17,122 MC performed with computer systems can correct for interobserver variations associated with manual counts. They can better identify hot spots, 8 and they can count thousands of fields but may introduce different hurdles. High-quality training datasets that adequately reflect the variability of histopathology sections and scanned images, along with validation of AI methods, are paramount for CPATH to produce accurate and verifiable counts. With high-quality data sets that define MF, atypical mitotic figures (AMF) possibly along with hard negatives such as mitotic like figures (MLF such as inflammatory cells necrotic/apoptotic cells, deformed or compressed nuclei), automated means to perform MC should eventually be able to address potential confounders. Regardless of which mode, manual or automated, we propose that each of these elements needs to be standardized: (1) definition of MF, AMF, and MLF; (2) the size of the area in which MF and AMF are counted; (3) the area of the tumor to be evaluated (such as “hot spots” or areas of high mitotic density); and (4) how to handle confounders. Each of these is described in Supplemental Files 01 and 02, and computational pathology in CPATH is Supplemental Files 08. At the end of each Supplemental File are considerations for future studies which should help improve the method and clarify issues associated with assessing the parameter.
What Morphological Features Define a MF to Be Included in the MC? (See Supplemental File 02)
The morphologic characteristics of MF and AMF and features that distinguish these from MLF are detailed in a recent publication. 39 Mitotic figures and AMF are most easily identified by the short “rods” of chromosomes protruding from the surface of aggregates of nuclear material (Figs. 1 –4). Identification of the different phases of mitosis or the type of AMF are not necessary, but an understanding of the mitotic continuum and that AMF may have prognostic significance should be appreciated. Counting AMF may correlate with poorer prognosis and outcome as seen in some human tumors. 58,74 Definitive MF (Figs. 1 –4) and AMF (Figs. 5 –8) should be included in the MC; however, structures with ambiguous morphology create a dilemma in classification. This is not problematic if the MC is markedly high (eg, >20 MF/2.37 mm2). However, if the MC is close to an established threshold which has clinical significance, then the identity of these candidate structures could be critical (see Supplemental File 01). New thresholds should be established following the guidelines in Supplemental File 01 and those thresholds should be tiered (avoid thresholds based on a <or> single number). Clinicians that request recounts because the MC of a tumor is at a threshold should seek different parameters to help establish the prognosis or direct therapy. We all likely have “nonstandardized methods” that we use while counting MF but would not necessarily like others to know we do them: enumerating doubtful structures under a column labelled “?”; looking at extra fields when no MF were seen; looking at extra fields because there were spaces created by blood vessels, ducts, or cysts; what to do when the tissue sample is <2.37 mm2; and/or looking for MF when the diagnosis of inflammation versus neoplasia is not clear. Practical considerations while performing a MC are listed in Supplemental File 01. Comments from colleagues on these practicalities and other components of the MC are welcomed. Pathologists and laboratories will develop their own procedures to address MC reporting in nonroutine situations. When solutions are found, the protocol will be updated accordingly. Correct identification of histologic structures will improve MC consistency and accuracy obtained from manual (glass or WSI) or CPATH modes.
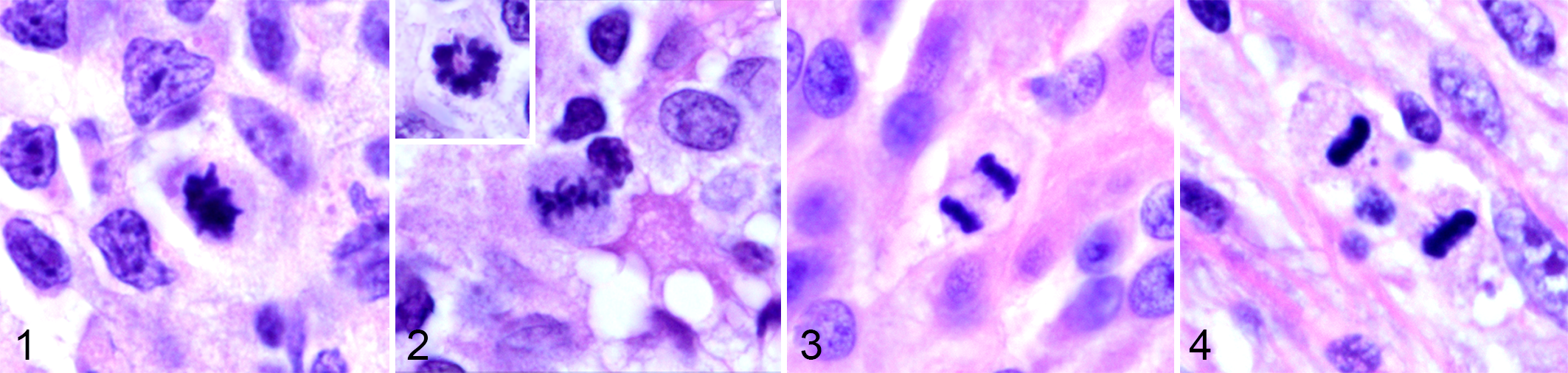
Mitotic figures (MF) are characterized by dark aggregates of nuclear material with short rods and projections.
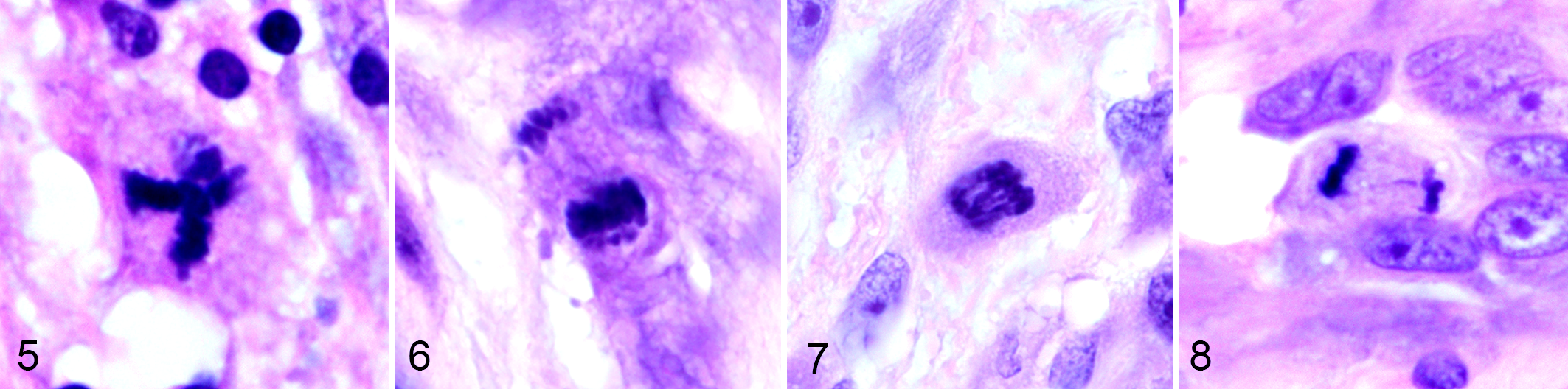
Atypical MF (AMF).
Does the Field Number (FN) of an Ocular Matter?
For light microscopy, absolutely. It is the limiting factor that determines the diameter and therefore the area in the field of view (FOV) when objectives of the same magnification are used. Engraved or printed on some ocular eyepieces is a field number (FN) ranging from 6 to 28 mm. Higher numbers have larger FOV diameters and small increases in the FN will produce large increases in the area of FOV (see Supplemental Files 01 and 12). The diameter of the FOV can be measured with a stage micrometer or it can be calculated by dividing the FN (mm) by the objective magnification. The formula for the area of a circle is used to calculate the area in the FOV. Therefore, a microscope with an ocular FN 18 mm, 40× objective has a diameter of 0.45 mm in the FOV and an area of 0.16 mm2 per “hpf”; FN 26.5 mm, 40× objective has a diameter of 0.66 mm and an area of 0.34 mm2 per “hpf,” which is a 100% larger area, a 2-fold increase (see Fig. 9 and Table S3 in Supplemental File 01). 81
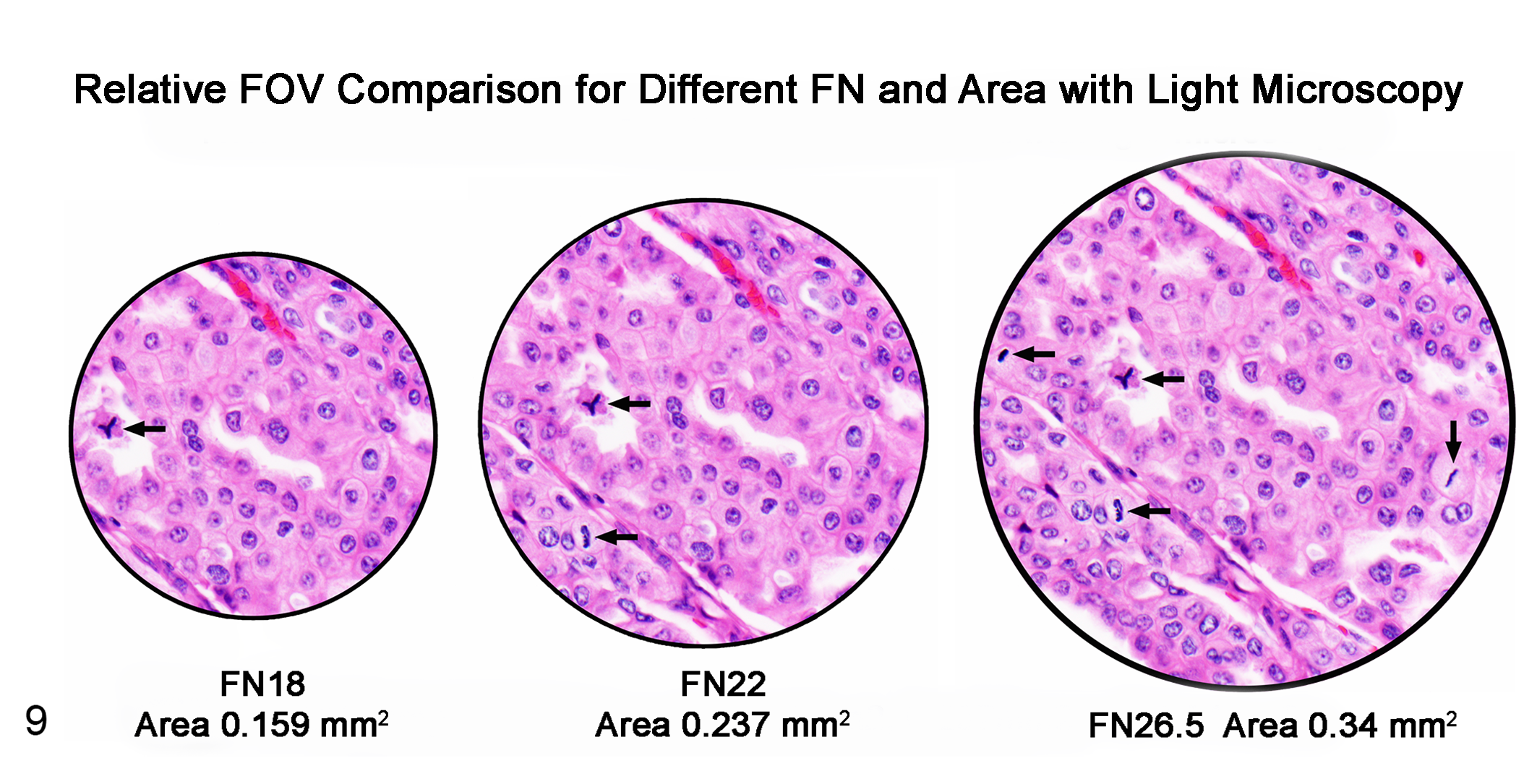
Urothelial carcinoma, dog. Schematic representation comparing field of view (FOV) at 400× magnification. The differences in diameter, area, and the number of cells and MF seen are proportional to the differences seen with oculars: Field number (FN) 18 mm narrow FOV, FN 22 mm and FN 26.5 mm wide FOV. The FOV with ocular FN 26.5 mm has 4 MF, and the area is approximately 100% larger (2×) than the FOV with ocular FN 18 mm which has 1 MF. Older microscopes were commonly equipped with ocular FN 18 (smaller FOV) in the 1980s compared to current microscopes, which commonly have oculars of FN 22 or greater. Mitotic counts (MC) reported in terms of high-power fields (hpf) without specific units of measurement (mm2) cannot be compared to other MC as the area of a hpf can vary 100% to 200% between microscopes. To clarify study results and enable comparison between studies, reports of histologic parameters that are enumerated (MC, nuclear pleomorphism, microvascular density, etc) must be reported in precise units of area (ie, mm2) not in terms of hpf.
Some objectives will have FN and/or NA (numerical aperture) numbers engraved or printed on them. Both are defined in Appendix 1 (Definitions, Supplemental File 12). NA is critical for resolution and depth of field but it is not used to calculate FOV. The higher the NA the greater the resolution, or sharpness of features. All objectives have an FN but it may not be engraved on the objective. The FN of an objective can influence the FOV: however, it is the ocular FN which limits the maximum size of FOV in a standard microscope, not the objective FN.
What Is the Area in 10 hpf?
The area in 10 hpf is not a standard size as it varies up to 200% or more with the objective and the FN of the ocular. 21,44,81 We proposed replacement of the imprecise phrase, 10 hpf with 2.37 mm2 to reflect the area equating to 10 hpf using a 40× objective and a 10×X ocular FN 22 mm, the most common configuration of pathologists’ microscopes today. 81 Furthermore, 10 hpf is nebulous for WSI, which has become routine for many diagnostic laboratories worldwide. A standard size area in mm2 is required so the characteristics of the monitor and the magnification at which the image is reviewed can be configured to a specific area (see Supplemental File 01). Temporarily, retaining the phrase “10 hpf “ together with accurate terminology (2.37 mm2) clarifies communication with clinicians and permits MC to be determined and reported from microscopes or WSI. This is preferred to reporting the FN of the ocular, which may further confuse clinicians and because the FN is meaningless for WSI.
MC reported in terms of hpf without specific units of measurement (mm2) cannot be compared to other MC as the area within one or especially 10 hpf is too variable. 21,44,81 Older microscopes were equipped with ocular FN 18 (smaller FOV) compared to current microscopes, which commonly have oculars of FN 22 or greater (Fig. 9). Most prior animal studies did not define the area (mm2) in which the MC or other histological features were enumerated, or defined the area incorrectly limiting the utility of these data for formulating prognoses for current cases. 22,64,90,98 These studies need to be repeated with standardized methods of determining the basic histological parameters used to evaluate tumors. Concurrently, new methods should be considered and all must eventually be correlated with outcome assessments.
Does the Standard Area Need to Remain 2.37mm 2 ?
No, it can be changed with data-driven results. The total area evaluated can be amended for different tumors or unique situations, for example, total tissue submitted is <2.37 mm2, cystic tumors, and so on. We may discover that different sized areas are needed for tumors with low or high proliferative rates in order to find the area, size, or region of the tumor that is prognostic. Perhaps tumors with low proliferative rates require larger areas to be enumerated (5–10 mm2) or perhaps it is the opposite. What might be more important than a MC in one spot is what proportion of an entire tumor (or section) has low versus high proliferative rates. Greater than 85% of canine cutaneous mast cell tumors (MCT) are indolent; 64,90,98,110 perhaps determining the percent of a MCT that is “cold” (few hot spots) will predict how aggressive the tumor is. For canine oral melanoma, it might be the proportion of the tumor that is “hot” which is predictive. We also do not know how many sections of a tumor should be enumerated for the MC to be most prognostic/predictive? This is true for other histologic parameters as well. These changes require correlating the different methods with known outcomes (recurrence, metastasis, DFI) in many cases to show which method is prognostic. Once a prognostic MC is determined for a specific tumor type, the same size area, same region of the tumor, and means to identify MF and AMF need to be validated if we want to compare results between labs or use published thresholds or tiers of histologic parameters.
When multiple sections or regions are enumerated, should an average MC be reported or the ranges? 83 Various guidelines have been proposed for determining the optimal tumor area for performing the MC in human tumors. Different sized areas are recommended to perform MC for different tumors. Some authors recommend counting a series of 5 or more sets of MC and reporting the average. Others report the highest MC. There are a multitude of scenarios that need investigation to change how we determine MC, and CPATH (Supplemental File 08) will greatly aid these studies because MC can be performed faster, more consistently, and can be performed over differently sized areas in different regions of the tumors. CPATH can report the proportion of a tumor that is hot or cold. Manual counts for these types of studies will be laborious. Studies using CPATH should also include the standard means of determining the MC and compare the various methodologies to known outcomes. Hopefully, these studies will avoid creating MC cutoffs that are based on a single number (above or below) and develop scoring systems, confidence intervals, tiers, and ranges of predictability for MC for different tumors.
Until data-driven results provide new methods, an area equivalent to 2.37 mm2 should be used for MC and should be reported as mm2 rather than stating the FN of the ocular or how the scope is configured.
Where in the Tumor Should the MC Be Performed?
Presently MF and AMF should be counted in regions of hot spots or high mitotic density in viable regions of tumor. The MC should include viable tumor cells adjacent to necrotic areas but must exclude necrotic cells. MC can only be accurately performed in areas of the tumor that are viable. If a necrotic area is encountered while performing a MC, this region is avoided and a viable area of equivalent size to the region of necrosis must be included in the final tally of MF. It is logical to choose regions of high tumor cell proliferation because the cells in these areas may be more aggressive, already may have the potential to metastasize, or have a greater opportunity to form a clone with metastatic potential. Until studies report that a different region is more predictive of outcomes, we should adhere to this method. There are no studies in animals that correlate MC determined in different regions with outcomes. Multiple studies in humans and one in dogs have demonstrated variability in the number of MF in different regions of tumors. 17 We know there is heterogeneity of MF distribution in tumors, but we do not know if it matters, and we will not know until there are outcome assessments correlated to methods. Different regions and differently sized areas of different tumor types are used to perform MC in human tumors, and different cutoffs of MC are used to determine prognoses. Similar studies need to be done with animal tumors, and when these are performed, investigators should include newer technologies as well (molecular, CPATH, etc).
The periphery of some tumors is the preferred site because this is the invasive front, fixation is better, and there is a higher proliferative rate. A study of human breast carcinoma reported that the periphery contained more hot spots (using Ki67) than other regions and percentages of Ki67 positive nuclei obtained at the periphery changed the prognosis. 56 Other studies in humans reported that using Ki67 in hot spots, which were not just at the periphery of breast carcinoma, contributed the most prognostic information as compared to other methods. 107 Additionally, a study of canine cutaneous mast cell tumors found that the regions of highest mitotic activity were not always at the periphery. 17 Selecting the area of a tumor that is predictive of outcome(s) or treatments needs to be found for each tumor type. Until those locations are identified, MC should be performed in regions of hot spots. However, determination of hot spots by routine light microscopy is subjective and a source of interobserver variation. 17,119 A study with canine MCT and one with canine melanoma showed that pathologists were not as capable of finding the hotspots as compared with computer-assisted localization of hot spots. 8,92
Tissue preparation likely has an influence on the MC. Fixation delay is reported to change the MC, however not in a consistent manner 15,31,37,68 One study compared MC in resected breast tumors in women to incisional biopsies of the same tumors. The resected samples had an increase in MC of up to 3-fold, resulting in an increased tumor grade in approximately 25% of carcinomas, which had an effect on prognosis and treatment. 68 Decreased numbers of MFs has been reported when there is reduced tissue quality from delayed fixation, which is likely due to an inability to differentiate between MF and MLF. 31,37 Other studies have shown no measurable effect from fixation delay on the MC. 15 Future studies should evaluate whether or not delays in fixation have an effect on the MC in animal tumors, and recommend best procedures.
Summary
Supplemental Files 01 and 02 detail the standard method of performing a MC and include definitions of MF, AMF, and MLF, contiguous 2.37 mm2 area, hot spots, practical considerations, and future considerations of how the MC can be improved. The present guideline to perform the MC will be modified when data-driven changes necessitate, and the appropriate protocols will subsequently be updated.
A full list of references is available in Supplemental Files 01 and 02.
References: MC MF
5. Al-Janabi S, van Slooten HJ, Visser M, et al. Evaluation of mitotic activity index in breast cancer using whole slide digital images. PLoS One. 2013;
8. Aubreville M, Bertram CA, Marzahl C, et al. Deep learning algorithms out-perform veterinary pathologists in detecting the mitotically most active tumor region. Sci Rep. 2020;
12. Baak JP, Gudlaugsson E, Skaland I, et al. Proliferation is the strongest prognosticator in node-negative breast cancer: significance, error sources, alternatives and comparison with molecular prognostic markers. Breast Cancer Res Treat. 2009;
15. Bergers E, Jannink I, van Diest PI, et al. The influence of fixation delay on mitotic activity and flow cytometric cell cycle variables. Hum Pathol. 1997;
17. Bertram CA, Aubreville M, Gurtner C, et al. Computerized calculation of mitotic count distribution in canine cutaneous mast cell tumor sections: mitotic count is area dependent. Vet Pathol. 2020;
19. Bertram CA, Gurtner C, Dettwiler M, et al. Validation of digital microscopy compared with light microscopy for the diagnosis of canine cutaneous tumors. Vet Pathol. 2018;
21. Bonert M, Tate AJ. Mitotic counts in breast cancer should be standardized with a uniform sample area. Biomed Eng Online. 2017;
22. Bostock DE. The prognosis following surgical removal of mastocytomas in dogs. J Small Anim Pract. 1973;
31. Cross SS, Start R, Smith J. Does delay in fixation affect the number of mitotic figures in processed tissue? J Clin Pathol. 1990;
37. Donhuijsen K, Schmidt U, Hirche H, et al. Changes in mitotic rate and cell cycle fractions caused by delayed fixation. Hum Pathol. 1990;
39. Donovan TA, Moore FM, Bertram CA, et al. Mitotic figures-normal, atypical, and imposters: a guide to identification. Vet Pathol. 2021;
44. Ellis PS, Whitehead R. Mitosis counting—a need for reappraisal. Hum Pathol. 1981;
56. Gudlaugsson E, Skaland I, Janssen EA, et al. Comparison of the effect of different techniques for measurement of Ki67 proliferation on reproducibility and prognosis prediction accuracy in breast cancer. Histopathology. 2012;
58. Jin Y, Stewenius Y, Lindgren D, et al. Distinct mitotic segregation errors mediate chromosomal instability in aggressive urothelial cancers. Clin Cancer Res. 2007;
64. Kiupel M, Webster JD, Bailey KL, et al. Proposal of a 2-tier histologic grading system for canine cutaneous mast cell tumors to more accurately predict biological behavior. Vet Pathol. 2011;
68. Lehr HA, Rochat C, Schaper C, et al. Mitotic figure counts are significantly overestimated in resection specimens of invasive breast carcinomas. Mod Pathol. 2013;
74. Matsuda Y, Yoshimura H, Ishiwata T, et al. Mitotic index and multipolar mitosis in routine histologic sections as prognostic markers of pancreatic cancers: a clinicopathological study. Pancreatology. 2016;
81. Meuten DJ, Moore FM, George JW. Mitotic count and the field of view area: time to standardize. Vet Pathol. 2016;
82. Meyer JS, Alvarez C, Milikowski C, et al. Breast carcinoma malignancy grading by Bloom-Richardson system vs proliferation index: reproducibility of grade and advantages of proliferation index. Mod Pathol. 2005;
83. Meyer JS, Cosatto E, Graf HP. Mitotic index of invasive breast carcinoma. Achieving clinically meaningful precision and evaluating tertial cutoffs. Arch Pathol Lab Med. 2009;
90. Patnaik AK, Ehler WJ, MacEwen EG. Canine cutaneous mast cell tumor: morphologic grading and survival time in 83 dogs. Vet Pathol. 1984;
92. Puri M, Hoover SB, Hewitt SM, et al. Automated computational detection, quantitation, and mapping of mitosis in whole-slide images for clinically actionable surgical pathology decision support. J Pathol Inform. 2019;
98. Romansik EM, Reilly CM, Kass PH, et al. Mitotic index is predictive for survival for canine cutaneous mast cell tumors. Vet Pathol. 2007;
107. Stålhammar G, Robertson S, Wedlund L, et al. Digital image analysis of Ki67 in hot spots is superior to both manual Ki67 and mitotic counts in breast cancer. Histopathology. 2018;
110. Stefanello D, Buracco P, Sabattini S, et al. Comparison of 2- and 3-category histologic grading systems for predicting the presence of metastasis at the time of initial evaluation in dogs with cutaneous mast cell tumors: 386 cases (2009–2014). J Am Vet Med Assoc. 2015;
116. Tsuda H, Akiyama F, Kurosumi M, et al. Evaluation of the interobserver agreement in the number of mitotic figures of breast carcinoma as simulation of quality monitoring in the japan national surgical adjuvant study of breast cancer (NSAS-BC) protocol. Jpn J Cancer Res. 2000;
119. van Diest PJ, Baak JP, Matze-Cok P, et al. Reproducibility of mitosis counting in 2469 breast cancer specimens: results from the multicenter morphometric mammary carcinoma project. Hum Pathol. 1992;
122. Veta M, van Diest PJ, Willems SM, et al. Assessment of algorithms for mitosis detection in breast cancer histopathology images. Med Image Anal. 2015;
Guideline 3. Tumor Margins
For additional details, see Supplemental File 03—Tumor Margins.
Margin assessment is one of the most important histological parameters evaluated in oncology. 59,70,112 Patient management decisions often hinge on the results of margin assessment, and clinicians may value margin assessment as highly or more than a diagnosis. In one survey of 277 veterinary oncologists, the surgical margin description was rated number one in importance, slightly higher than the diagnosis. 76 Supplemental File 03 provides the types of data that are required to standardize the reporting of margins for both clinical management and future studies.
Histologic margin evaluation only needs to be reported on tumors where the aim of surgery is to completely remove the neoplasm (achieve local control). Samples where there was no intent to totally excise the tumor, including intralesional or incisional biopsies are for diagnosis. Certain tumors or the anatomic location of a tumor near vital structures dictate that excision for local control will be attempted but the surgeon realizes that adjacent structures limit how much margin can be safely taken. Therefore, margin assessment by the pathologist may not be critical to the surgeon and communication between surgeon and pathologist will clarify what the surgeon needs to determine adequacy of the margin (eg, thyroid, anal sac tumors, adrenal glands). Consensus was not reached as to whether margins should be reported for benign tumors. Supplemental File 03 provides contrasting philosophies (Why not? vs Why bother?) and the recommendation that considerations such as this should be left at the discretion of the pathologist and or their lab.
For the overall evaluation of surgical margins, the members of the cancer treatment team are the clinician, surgeon, laboratory technologist, and pathologist. The responsibilities of each are detailed in Supplemental File 03. Although terms such as complete, clean, clean but close, narrow, and dirty are ingrained in the clinical and pathology lexicon, practitioners, surgeons, and oncologists should discourage their use and not expect these to be used in pathology reports. Ultimately it is the clinician and/or surgeon that judges if the margin is deemed adequate after consideration of all factors. Pathology reports should state facts and clinicians can use these along with other clinical data to make judgements about adequacy of margins and provide a prognosis. Observations by the pathologist include (1) relationship of neoplastic cells to the surrounding tissue including presence of a capsule, tissue compression, peripheral invasion, and lymphovascular invasion; (2) the distance from neoplastic cells to the narrowest or closest inked lateral and deep margin (histologic tumor-free distance [HTFD]; Fig. 10), the relationship of neoplastic cells to the boundaries of the compartment in which the tumor is located, especially along the deep border. In many cases, measuring the HTFD alone is not enough to determine the adequacy of surgical margins, yet it is the parameter that is often used to determine “completeness”’ of excision by clinicians and pathologists. Metric measurements laterally are considered standard but for the deep margin a metric distance and the type of tissue present at the deep border should be reported.
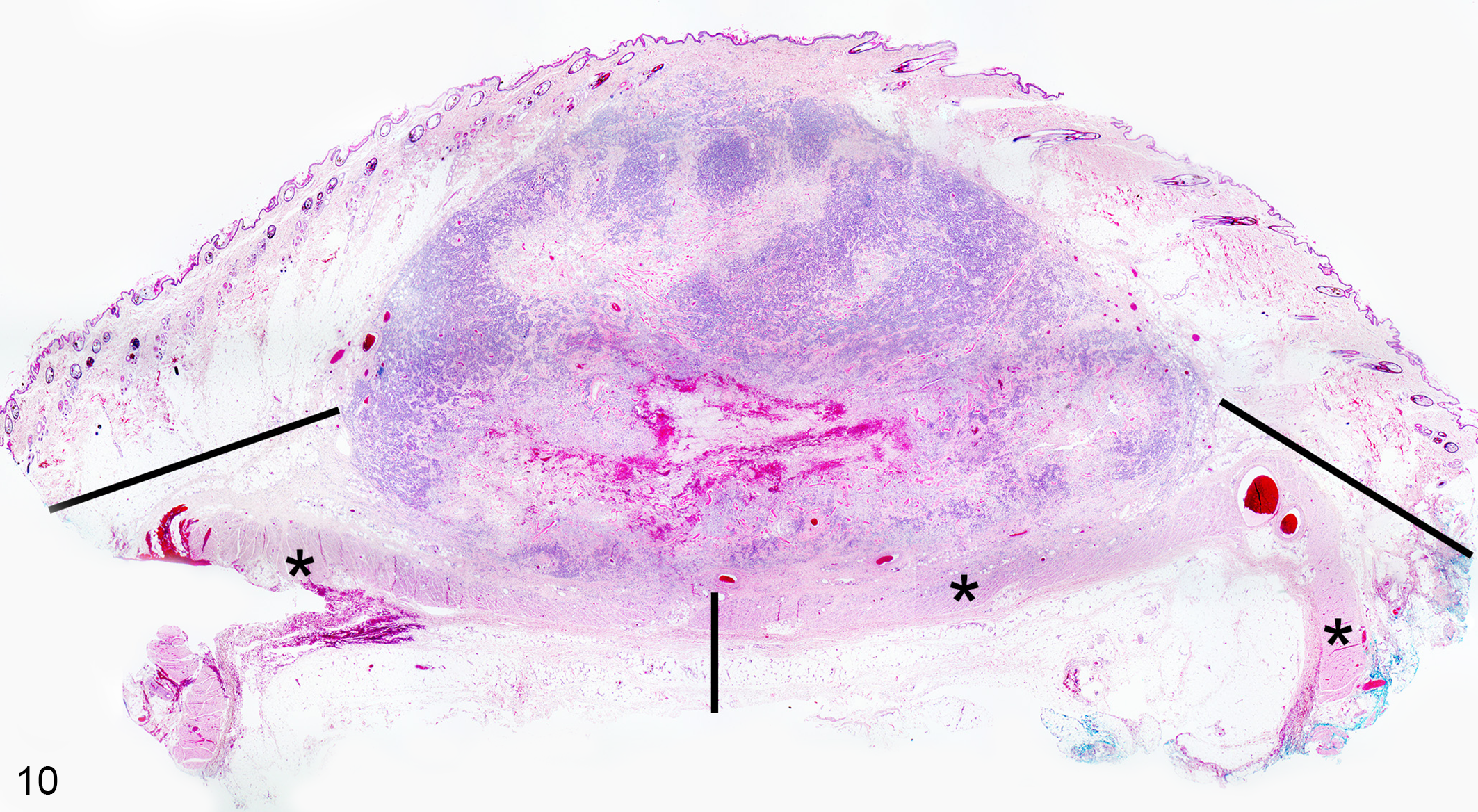
Canine cutaneous mast cell tumor involving the dermis and subcutaneous tissues. The histologic tumor free distance (HTFD) is depicted with horizontal and vertical black lines and can be measured with manual or digital means. Note that ink can be observed at the lateral (or peripheral) margins but is not visible at the deep margin. Therefore, the deep margin measurement represents an approximation given the lack of ink. Additional sections into the formalin-fixed paraffin-embedded block may resolve this issue. A potential tissue barrier within the subcutaneous tissue is the subcutaneous striated muscle (also called panniculus carnosus or cutaneous trunci in the truncal region, denoted by the asterisks). This muscle is not always visible in histologic sections of cutaneous and subcutaneous tumors; it has variable distribution and continuity in different body regions. 4,101
Identification of the gross margin by the clinician/surgeon immediately after tumor excision is necessary in order for HTFD to accurately represent the measurement between the tumor and excision site. However, margins are often not identified at the time of surgery by either surgeons or primary care veterinarians. In one survey of pathology reports, approximately 34% of surgeons or oncologists identified the margins in tissues submitted for pathological examination. 95 Details of how to apply ink have been reported 59 (see Supplemental File 03) and this information is now being included in veterinary school curricula. If ink is not present when the sample arrives at the lab this should be noted. If the gross specimen is not inked by the clinician or the ink cannot be identified in the sections examined (Fig. 10), the significance of any measured margins is questionable. Perhaps communication from diagnostic labs to consumers and letters to editors of clinical journals explaining the importance of ink being applied by clinicians is needed.
Only a small portion of the circumferential surgical margin is evaluated histologically (approximately 0.1% to 0.01% of the total margin). 103 HTFD should be further studied by comparing different methods of margin analysis (radial, tangential, parallel slicing) with outcome assessments for different tumor types. 40,85 Until those studies provide comparative data, radial sections are recommended. Regardless of the method used, any margin measured histologically may not accurately represent the tumor and its relationship to the normal surrounding tissue in the patient. It is important to note that HTFD is made on a histopathology specimen that has undergone shrinkage (ranges reported from 13% to 50%) and can underestimate the surgically obtained margins by up to 40%. 84,117 Most of the shrinkage occurs immediately after removal and prior to fixation. 27,84,117 The important margin is between neoplastic cells and “normal tissues” (non-neoplastic) in the patient and this can only be estimated from histopathology. It is recommended that margins are reported in cm or in mm without decimals since a measurement such as 3.6 mm implies a level of precision and confidence that could be misleading. Furthermore, data are accumulating that the biological behavior of the tumor may be a more important predictor of recurrence than identification of neoplastic cells at a margin. Certainly, this seems to be the case with low-grade canine MCT and STT/STS. Most low-grade MCT do not recur even with tumor cells at the margin and approximately one third of high-grade MCTs will recur when the histologic margins are free of tumor cells. 38 Similarly, for canine STT/STS, greater than 90% do not recur if margins greater than 1 mm are free of neoplastic cells and one study reported that when margins are less than 1 mm, 3 of 41 grade 1 tumors (7%), 14 of 41 grade 2 tumors (34%), and 3 out of 4 grade 3 tumors recurred. 77 These data need validation with many more cases of STT/STS that are grade 3 and have known outcomes; to “rely” on or state there is a 75% recurrence rate based on 4 cases exemplifies a problem in veterinary pathology. The biology of the tumor and the host (immune system, genes) are important factors that influence tumor recurrence and metastases.
There is no standard protocol to follow to determine margins in re-excision specimens. The report cited most frequently is from a study of 41 dogs with STT/STS, in which the final conclusion was that histopathology of re-excision specimens was not predictive of recurrence. 13 Nine of 41 dogs (22%) had tumor in the primary re-excision specimen, whereas 32/41 (78%) had no tumor in the re-excision specimen. Six tumors recurred, 4 of which had no tumor seen in the re-excision specimen and 2 of which had tumor in the re-excision specimen. 13 Attempts to predict tumor behavior based on finding residual neoplastic cells in a re-excision specimen poses a quandary; confounders include the following: how extensively the specimen is sampled and that standardized protocols have not been designed. Determining the significance of the presence or absence of neoplastic cells in a re-excision specimen can be difficult due to several factors including the number of sections examined, the number of neoplastic cells found, and the amount of granulation tissue or fibroplasia which can mimic or obscure tumor cells. In addition to searching for neoplastic cells, we should consider reporting if the re-excisional biopsy extended to normal tissue, and in how many sections this was seen, indicating the “entire” lesion was removed, at least in sections examined histologically.
Of importance to surgeons is the concept of compartmental boundaries, and fascial planes which are used to plan and perform surgical removal of tumors. 45,63 Tissues within anatomic compartments and fascial planes may provide natural barriers to tumor extension. If tissues are functional barriers to tumors, the mechanisms by which this is accomplished needs to be defined. Are they physical barriers that depend on the quantity or quality of the tissue (fascia, muscle, bone) and/or are there inhibitors present that limit tumor growth? Experimental data to support these theories and studies which define the mechanisms in which tumor spread is limited are needed. The peritoneal serosa is a reported barrier to tumor cell migration in vitro. 52 Certainly, the type of tumor, grade of tumor, whether it expands circumferentially, infiltrates, or skips (discontinuous growth pattern) influences how effectively a potential tissue barrier may function. Compartments, fascial planes, and fascia vary depending on anatomical location; therefore, information about location and gross tumor growth patterns (confined, infiltrative) should be provided on the pathology requisition. Surgeons should ink and label the gross specimen and state on the pathology requisition what precise structures are important to them. Pathologists will identify the tumor, structures labelled, and relationship of the tumor to lateral and deep margins (HTFD) and report what tissue is present along the deep margin (adipose, muscle, fascia, etc).
Fascia and fascial planes are not synonymous. Fascia is an anatomical structure described in anatomy and histology texts. There are recent reviews describing fascia for the dog and horse 4 and surgically identifiable fascial planes for the dog. 101 The thickness and tensile strength of fascia is markedly variable in different anatomical locations. Some are thin, barely discernible, torn easily (subcutaneous superficial fascia), while others are thicker, visible grossly and histologically (deep fascia on muscles). 4,101 Fascial planes are described in the surgical literature and can be used to dissect along to isolate and excise tumors from the skin, subcutis, and musculoskeletal system, more so than when removing tumors from internal organs. It has been recommended that excision of potentially aggressive tumors from the subcutaneous tissue include at least one uninvolved fascial plane, deep to the tumor. 50,35,91 The intact fascial plane below the tumor provides a physical barrier of normal tissues such as collagen, muscle, and other tissues that may help prevent extension of the tumor. Pathologists rely on the morphological features of anatomical structures to identify fascia and boundaries of the fascial plane indicated by the surgeon.
If one or more fascial planes of tissues are removed with a tumor, the surgeon should state on the pathology requisition what tissues are to be examined in relationship to the tumor. If the surgeon wants to know how close a tumor is to the deep margin and what tissues are present along the deep margin, then those regions need to be inked grossly and labelled. The pathologist reports if tumor extends to the deep margin (which the surgeon inked) and the composition and integrity of the deep margin. A report that states the HTFD is 4 mm to the deep margin but there is no fascia present is interpreted quite differently by the surgeon and medical oncologist from a report that states the HTFD is 4 mm and sections from the deep margin include dense collagenous fascia which is free of neoplastic cells. This information helps the surgeon and oncologist decide if the margin is adequate and consider treatment options. Pathologists can attempt to identify fascia with H&E, consider histochemical stains for collagen, or may visualize the fascia in the context of its adjacent skeletal muscle.
When a delineated anatomic compartment is not obvious, the HTFD is of critical importance. HTFD for lateral and deep margins in samples from skin and subcutis tumors should be reported separately. In a review of surgical biopsy reports of canine cutaneous mast cell tumors, details about the margins and consistency of how histologic margins were reported were generally lacking. 95 For example, while some margins were reported in 92% of cases, lateral and deep margins were described separately in 77% of cases, margin direction was only given in 16% of cases, and descriptions of the deep margin were only available in 11% of cases. 95 The deep margin may be difficult for surgeons to visualize intraoperatively. Synoptic reports organized by required data elements should help ensure complete data are reported and prevent omission errors (Supplemental File 06). Tumor and host biology (eg, microenvironment and immunologic factors) may be more important predictors of outcome than the presence or absence of cells at the margin. 51 Multiple tumor parameters with scoring systems assessed simultaneously may aid in indicating intrinsic tumor biology and behavior. At the end of Supplemental File 03 are considerations for future studies (M1-M4 or R0-RX). 70,112,128
References: Margins
4. Ahmed W, Kulikowska M, Ahlmann T, et al. A comparative multi-site and whole-body assessment of fascia in the horse and dog: a detailed histological investigation. J Anat. 2019;
13. Bacon NJ, Dernell WS, Ehrhart N, et al. Evaluation of primary re-excision after recent inadequate resection of soft tissue sarcomas in dogs: 41 cases (1999–2004). J Am Vet Med Assoc. 2007;
27. Clarke BS, Banks TA, Findji L. Quantification of tissue shrinkage in canine small intestinal specimens after resection and fixation. Can J Vet Res. 2014;
38. Donnelly L, Mullin C, Balko J, et al. Evaluation of histological grade and histologically tumour-free margins as predictors of local recurrence in completely excised canine mast cell tumours. Vet Comp Oncol. 2015;
40. Dores C, Milovancev M, Russell D. Comparison of histologic margin status in low-grade cutaneous and subcutaneous canine mast cell tumours examined by radial and tangential sections. Vet Comp Oncol. 2018;
45. Enneking WF, Spanier SS, Goodman MA. A system for the surgical staging of musculoskeletal sarcoma. Clin Orthop Relat Res. 1980;
50. Fulcher RP, Ludwig LL, Bergman PJ, et al. Evaluation of a two-centimeter lateral surgical margin for excision of grade I and grade II cutaneous mast cell tumors in dogs. J Am Vet Med Assoc. 2006;
51. Galon J, Mlecnik B, Bindea G, et al. Towards the introduction of the “Immunoscore” in the classification of malignant tumours. J Pathol. 2014;
52. Gao Z, Ye Y, Zhang W, et al. An anatomical, histopathological, and molecular biological function study of the fascias posterior to the interperitoneal colon and its associated mesocolon: their relevance to colonic surgery. J Anatomy. 2013;
59. Kamstock D, Ehrhart E, Getzy D, et al. Recommended guidelines for submission, trimming, margin evaluation, and reporting of tumor biopsy specimens in veterinary surgical pathology. Vet Pathol. 2011;
63. Kawaguchi N, Matumoto S, Manabe J. New method of evaluating the surgical margin and safety margin for musculoskeletal sarcoma, analysed on the basis of 457 surgical cases. J Cancer Res Clin Oncol. 1995;
70. Liptak JM. Histologic margins and the residual tumour classification scheme: is it time to use a validated scheme in human oncology to standardise margin assessment in veterinary oncology? Vet Comp Oncol. 2020;
76. McGill L, Blue J, Powers B. Report of the ad hoc committee on oncology to the ACVP membership and interested pathology community. Vet Pathol. 2002;
77. McSporran KD. Histologic grade predicts recurrence for marginally excised canine subcutaneous soft tissue sarcomas. Vet Pathol. 2009;
84. Miller JL, Dark MJ. Evaluation of the effect of formalin fixation on skin specimens in dogs and cats. Peer J. 2014;
85. Milovancev M, Townsend KL, Gorman E, et al. Shaved margin histopathology and imprint cytology for assessment of excision in canine mast cell tumors and soft tissue sarcomas. Vet Surg. 2017;
95. Reagan JK, Selmic LE, Fallon C, et al. Evaluation of information presented within mast cell tumour histopathology reports in the United States: 2012–2015. Vet Med Sci. 2018;
101. Schroeder MM, Skinner OT. Fascial plane mapping for superficial tumor resection in dogs. Part I: neck and trunk. Vet Surg. Published online January 21, 2021. doi:10.1111/vsu.13569
103. Selmic LE, Ruple A. A systematic review of surgical margins utilized for removal of cutaneous mast cell tumors in dogs. BMC Vet Res. 2020;
112. Stromberg PC, Meuten DJ. Trimming tumors for diagnosis and prognosis. In: Meuten DJ, ed. Tumors in Domestic Animals. 5th ed. John Wiley; 2017:35-51.
117. Upchurch DA, Klocke EE, Henningson JN. Amount of skin shrinkage affecting tumor versus grossly normal marginal skin of dogs for cutaneous mast cell tumors excised with curative intent. Am J Vet Res. 2018;
128. Wittekind C, Compton C, Quirke P, et al. A uniform residual tumor (R) classification: integration of the R classification and the circumferential margin status. Cancer. 2009;
Guideline 4. Lymphovascular Invasion
For additional details, see Supplemental File 04—Lymphovascular Invasion.
Neoplastic cell invasion of blood vessels or lymphatics is widely recognized as evidence of tumor aggressiveness and potential malignancy in both humans 47,79 and animals, 54,94 but despite the importance of this parameter, criteria to definitively identify lymphovascular invasion (LVI) and distinguish from pseudo-vascular invasion or retraction artifact are lacking in the veterinary literature. This lack of stringent, standardized criteria may have led to misdiagnosis of LVI in veterinary oncology studies. Assessing LVI with criteria of varying stringency has revealed key insights into the biological behavior of human cancers as has the distinction between blood vascular and lymphatic invasion. 69,79,118 In veterinary medicine, LVI is recognized as a marker of potential tumor malignancy but this parameter has only been extensively evaluated in canine and feline mammary tumors 54,94 without establishment of strict criteria for LVI diagnosis or comparison of blood vascular and lymphatic invasion.
Mimickers of LVI, such as pseudo-vascular invasion and retraction artifacts, are not adequately addressed in the veterinary literature; images of each can be found in Supplemental File 04 and on the website. Pseudo-vascular invasion is the presence of neoplastic cells within vascular spaces, but the cells are not present because of tumor invasion of vessels. Displacement of neoplastic cells into vessels secondary to manipulation of the neoplasm at the time of biopsy, surgical excision, grossing procedure, or tissue sectioning (ie, “floaters”) can result in pseudo-vascular invasion 79,118 This is also reported for non-neoplastic lesions in the thyroid. 79 Neoplastic cells may protrude or impinge into adjacent vascular lumens without true invasion in which case endothelial cells cover the surface of the impinging tumor. However, endothelium may also line the surface of neoplastic cells which have invaded through the vascular endothelium but have undergone re-endothelialization, necessitating searching for other criteria of LVI to confirm which is the correct interpretation.
Distinguishing between these various manifestations of pseudo-vascular invasion and true LVI relies on identification of more robust LVI criteria. The 2 most definitive criteria used to define LVI in human tumors are thrombus adherent to intravascular tumor and tumor cells invading through the vessel wall and endothelium. Additional criteria are listed in Supplemental File 04 along with a complete reference list. These criteria should be used to assess LVI in tumors from animals. 79
Retraction artifact, another mimicker of LVI, forms an artifactual space surrounding tumor foci and can be distinguished from intravascular neoplasia by the absence of an endothelial cell lining. Retraction artifact is seen in epithelial tumors in which tumor cells retract from surrounding stroma (Figure S15 in Supplemental File 04). This event is fairly common with circumanal gland tumors.
Studies of human breast, thyroid, and prostate cancer show that widespread metastases are more commonly associated with blood vascular invasion in contrast to lymphatic invasion. 79 Animal tumors may show similar distinctions between blood and lymphatic vascular invasion, warranting detailed descriptions of the type of vessels invaded (ie, if a muscular wall is discerned in the involved vessels) or use of immunohistochemical markers to distinguish blood from lymphatic vessels. A variety of immunohistochemical markers have been used to identify endothelial cells in blood and lymphatic vascular channels in humans and animals. 49,104,123,127 Some markers, such as CD31 and Factor VIII related antigen, do not discriminate between lymphatic and blood vascular endothelium, whereas others, such as lymphatic vessel endothelial receptor 1 (LYVE-1), D2-40, and prospero-related homeobox gene-1 (PROX-1), are specific for lymphatic endothelium. 49,57,93,104,123,127 Use of IHC endothelial markers has been shown to facilitate identification of LVI in tumors in humans 87 and in mammary and plasma cell tumors in dogs. 42,104 Validation of IHC markers and antibodies used to differentiate lymphatic versus blood vessels for the different animal species is a necessity. Although IHC confirms the identity of the vascular structure it does not confirm true LVI and, in fact, is not one of the more stringent criteria of LVI.
Studies of tumor lymphovascular density (LVD) in humans have been correlated with LVI in a number of human tumors (complete list of references in Supplemental File 04). LVD is an enumeration of lymphatics within a defined area of a tumor and is used as an indicator of lymphangiogenesis and therefore probable lymph node metastasis. Both LVD and LVI are used as predictors of lymph node metastases in human breast cancer, and peritumoral lymphatic vessels may be the main route for dissemination of the tumor. Intratumoral microvascular density (IMD), the quantitation of blood vessels (number/mm2) in tumors, is used as an indicator of angiogenesis or vasculogenesis and by extension LVI and the ability of a tumor to metastasize. New blood vessels in a tumor are required for tumors to grow beyond several millimeters 72 as they are believed to facilitate metastasis and are associated with more aggressive neoplasms in humans and animals. Although IMD has been assessed in a number of animal tumors and has been associated with higher grade or more malignant histological features (ie, canine: soft tissue sarcomas, mammary gland tumors, seminomas, cutaneous squamous cell carcinoma and cutaneous mast cell tumors; full reference listing in Supplemental File 04) there have been no comprehensive studies of intratumoral versus peritumoral vascular density nor associations between IMD and blood vascular or lymphatic vascular invasion in domestic animals. Future veterinary studies comparing intratumoral versus peritumoral microvascular density and correlation with nodal and systemic metastases are warranted.
A thorough reassessment of LVI is needed in veterinary oncology with attention to the specific details described in Supplemental File 04 under future considerations. These studies should use the criteria outlined to determine if LVI is present, especially focusing on the more definitive features: invasion through vessel wall and endothelium and thrombus adherent to the tumor. Studies should include detailed descriptions of criteria used to establish presence of LVI and clarify the importance of lymphatic versus blood vascular invasion. Quantitation of blood and lymphatic vessels (IMD, LVD) may benefit from the use of CPATH, and both subjective and quantitative analyses should be correlated with nodal and systemic metastases and, most importantly, known patient outcomes.
References: LVI
42. Ehrensing G, Craig LE. Intravascular neoplastic cells in canine cutaneous plasmacytomas. J Vet Diagn Invest. 2018;
47. Falvo L, Catania A, D’Andrea V, et al. Prognostic importance of histologic vascular invasion in papillary thyroid carcinoma. Ann Surg. 2005;
49. Fitzgibbons PL, Connolly J, Bose S. Protocol for the examination of resection specimens from patients with invasive carcinoma of the breast. Coll Am Pathol. 2020;
54. Goldschmidt M, Pena L, Rasotto R, et al. Classification and grading of canine mammary tumors. Vet Pathol. 2011;
57. Halsey CH, Worley DR, Curran K, et al. The use of novel lymphatic endothelial cell-specific immunohistochemical markers to differentiate cutaneous angiosarcomas in dogs. Vet Comp Oncol. 2016;
69. Lin X, Zhu B, Liu Y, et al. Follicular thyroid carcinoma invades venous rather than lymphatic vessels. Diagn Pathol. 2010;
72. Luong R, Baer K, Craft D, et al. Prognostic significance of intratumoral microvessel density in canine soft-tissue sarcomas. Vet Pathol. 2006;
79. Mete O, Asa SL. Pathological definition and clinical significance of vascular invasion in thyroid carcinomas of follicular epithelial derivation. Mod Pathol. 2011;24
87. O’Donnell RK, Feldman M, Mick R, et al. Immunohistochemical method identifies lymphovascular invasion in a majority of oral squamous cell carcinomas and discriminates between blood and lymphatic vessel invasion. J Histochem Cytochem. 2008;
93. Pusztaszeri MP, Seelentag W, Bosman FT. Immunohistochemical expression of endothelial markers CD31, CD34, von Willebrand factor, and Fli-1 in normal human tissues. J Histochem Cytochem. 2006;
94. Rasotto R, Berlato D, Goldschmidt MH, et al. Prognostic significance of canine mammary tumor histologic subtypes: an observational cohort Study of 229 cases. Vet Pathol. 2017;
104. Sleeckx N, Van Brantegem L, Fransen E, et al. Evaluation of immunohistochemical markers of lymphatic and blood vessels in canine mammary tumours. J Comp Pathol. 2013;
118. Van den Eynden G, Van der Auwera I, Van Laere S, et al. Distinguishing blood and lymph vessel invasion in breast cancer: a prospective immunohistochemical study. Br J Cancer. 2006;
123. Von Beust B, Suter MM, Summers B. Factor VIII-related antigen in canine endothelial neoplasms: an immunohistochemical study. Vet Pathol. 1988;
127. Wennogle SA, Priestnall SL, Suarez-Bonnet A, et al. Lymphatic endothelial cell immunohistochemical markers for evaluation of the intestinal lymphatic vasculature in dogs with chronic inflammatory enteropathy. J Vet Intern Med. 2019;
Guideline 5. Tumor Necrosis
For additional details, see Supplemental File 05—Tumor Necrosis.
The extent of tumor necrosis has been correlated with tumor biological behavior and is a parameter used in grading schemes for human tumors. Tumor necrosis has also been included as a grading scheme parameter in animals, primarily in dogs with STT/STS but is also used in other grading schemes (canine primary pulmonary carcinoma). Criteria for determining the percent of tumor necrosis in all species have not been adequately described. 28,65 Necrosis within a tumor is often subjectively and vaguely used to suggest a tumor is aggressive. In humans, the percent of tumor necrosis has been determined by estimating the amount seen grossly and histologically, whereas animal studies have not indicated if gross observations were used in combination with histological assessment, or if only histological assessments were evaluated. 28,36,65,67,115,120 Measuring or estimating percentage of tumor necrosis is especially challenging in larger tumors.
In grading human soft tissue sarcomas, necrosis was found to be 1 of 3 parameters correlating with patient survival and tumor metastasis, along with tumor differentiation and MC. 115 The thresholds for scoring necrosis histologically were no necrosis (score 0), less than 50% of tumor necrosis (score 1), and greater than 50% tumor necrosis (score 2), but how a pathologist was to estimate those percentages was not detailed. A grade of 2 could also be assessed for any neoplasm whose gross appearance was described as “mainly necrotic” by a surgeon or pathologist even if no necrosis was seen on the submitted sections. 115 We do not recommend this last criterion be adopted for animal tumors, and later authors and grading systems, as well as current College of American Pathologist protocol for assessment of soft tissue tumors in humans, require microscopic confirmation/validation of macroscopic evidence suggesting necrosis. 28,67 This brings us to the problems associated with gross interpretation of necrosis (and to a lesser extent, its histologic interpretation). Even for an experienced pathologist, the gross diagnosis of necrosis may be problematic, and most pathologists in veterinary pathology will not see the gross specimen. Areas of edema or exudate may be interpreted as areas of necrosis grossly, and areas of hemorrhage, which are often associated with necrosis, may far exceed the boundaries of actual necrotic tissue. These problems are further compounded by certain histologic lesions such as myxomatous change, cystic space formation, edema, hemorrhage, and exudate, which can resemble or obscure necrotic areas. Gross/macroscopic assessment of necrosis requires histologic confirmation. For large tumors, processing an adequate number of sections may not be practical in a diagnostic setting due to cost, but should be done in research studies. The number of sections examined at trimming and/or submitted for histopathology for routine diagnostic cases is likely far fewer in veterinary than human pathology. If gross assessment is to be used as a parameter, numerous confounders must be clarified in future studies. This requires documentation of systematic sampling of both necrotic and viable tissue during the gross examination and confirmation of necrosis by histological evaluation. Alternatively, we can abandon the use of gross assessment and only use light microscopy. This would be straightforward, but if gross assessment of tumor necrosis improves the prognostic utility of grading systems then it would be lost as a parameter.
Although it seems obvious that the means to assess various histologic parameters need to be defined prior to implementation, this has not always happened, for example, the area in which MF were counted was never standardized and the same seems true for percent necrosis. The percent of tumor necrosis in STT/STS is included in grading schemes, yet the means to assess necrosis has not been clearly defined or standardized. Was the percent necrosis determined by examination of the tumor during gross sectioning, and were areas appearing necrotic confirmed microscopically? Was the percent necrosis used in the grading system based on visual estimate of necrosis in random histologic tumor sections? Was a consistent portion of the tumor submitted for microscopic examination? A recent publication suggested preparation of 1 tissue block for each 2 cm diameter of soft tissue tumors. 97 Since no formulae for number of blocks/slides per tumor have been described in published grading systems for dogs this seems like a good starting point, but no studies using this guidance were referenced.
Supplemental File 05 provides guidelines for recording and scoring extent of tumor necrosis on gross and histologic tumor evaluation, which should enable evaluation of the utility of this parameter to assess tumor prognosis and patient outcomes. The scoring system proposed is based on prior reports as indicated above but includes an unusual percentage of <10% for future studies and explains the logic for this. Additionally, for necrosis to be objectively assessed as a parameter for future grading schemes, new studies must determine if gross assessment of necrosis can be documented in a standardized fashion and if this parameter correlates with outcome assessment independently or as part of a grading system. For this to be accomplished, grossing personnel must include sections of tumor sites which appear necrotic, hemorrhagic, or edematous, regions typically avoided in most grossing procedures. Most veterinary pathologists will only have microscopic sections to estimate necrosis and these sections are likely to be a small percentage of the entire tumor. Furthermore, in many cases, the gross description will be inadequate unless grossing personnel are instructed to search and report the percent of the entire tumor that appears necrotic. The usual practice of only sampling viable tissue for histological examination might bias the utility of tumor necrosis as an independent or a component parameter in grading systems. Importantly, the size of the tumor, method of sectioning, number of cut surfaces examined grossly and histologically must be documented and, at some point, standardized. Based on size of tumor, a recommendation is needed for how many sections should be examined grossly and microscopically. It seems obvious that if pathologist A examines 5 histologic sections and pathologist B only 1 section of a tumor with 5 cm3 dimensions that the data gathered will not be comparable.
This brings us to the dilemma of how to currently approach reporting tumor necrosis. Given the lack of established guidance, the pathologist can estimate necrosis either visually with glass slides, WSI, or measure necrosis with annotation software in WSI. If WSI has drawing software, simply outline the entire tumor circumference (X) as well as the areas of necrosis (Y), followed by calculation of X/Y = % necrosis in one section (Figures S17A, B) in Supplemental File 05. In the absence of software or if using a microscope then visually estimate with varying magnifications (to confirm areas are indeed necrotic) if the percent necrosis is <50%>. The range of <50%> seems like a wide target and perhaps that is sufficient for estimates. We “assume” prior studies that estimated necrosis in canine tumors only used histology, but how representative the slide(s) are of overall tumor necrosis is unknown and inconsistent sampling of the tumor, purposely avoiding areas of necrosis in tissue selection can skew any determination of percent necrosis in histologic sections. Given the wide target of greater than or less than 50% necrosis, it may be possible to assess this level of necrosis histologically, even with inconsistent sampling. However, determining a 10% threshold of necrosis, as reported in one study indicating that dogs with tumors with >10% necrosis were 2.7 times more likely to die of tumor related causes may prove problematic. 65
Future studies can clarify how to determine the percent of tumor necrosis, particularly in larger tumors, and establish a standardized means of gross tissue selection for histologic examination. Various means of assessing necrosis in histologic sections can be compared and statistically evaluated. Results of standardized assessments for tumor necrosis can be compared to outcomes in univariable and multivariable analysis in concert with other histologic parameters and prognostic utility determined.
References: Necrosis
28. Coindre JM. Grading of soft tissue sarcomas: review and update. Arch Pathol Lab Med. 2006;
36. Dobromylskyj MJ, Richards V, Smith KC. Prognostic factors and proposed grading system for cutaneous and subcutaneous soft tissue sarcomas in cats, based on a retrospective study. J Feline Med Surg. 2021;
65. Kuntz CA, Dernell WS, Powers BE, et al. Prognostic factors for surgical treatment of soft-tissue sarcomas in dogs: 75 cases (1986–1996). J Am Vet Med Assoc. 1997;
67. Laurini JA, Blanke CD, Cooper K, et al. Protocol for the examination of specimens from patients with gastrointestinal stromal tumor (GIST). Coll Am Pathol. Accessed April 15, 2021. https://documents.cap.org/protocols/cp-gist-2017-v4001.pdf
97. Roccabianca P, Schulman FY, et al. In: Kiupel M, ed. Tumors of Soft Tissue. Vol 3. Davis Thompson DVM Foundation; 2020.
115. Trojani M, Contesso G, Coindre JM, et al. Soft-tissue sarcomas of adults; study of pathological prognostic variables and definition of a histopathological grading system. Int J Cancer. 1984;
120. Vayrynen SA, Vayrynen JP, Klintrup K, et al. Clinical impact and network of determinants of tumour necrosis in colorectal cancer. Br J Cancer. 2016;
Guideline 6. Synoptic Reporting in Veterinary Medicine
For additional details, see Supplemental File 06—Synoptic Reporting in Veterinary Medicine.
Synoptic reporting (as opposed to the traditional narrative reporting) is a method for reporting specific pieces of prognostically relevant data in a discrete format in pathology reports. 96 In human medicine, these have progressed from individual efforts 73 to being mandated by the College of American Pathologists (CAP) for accreditation. 29 In general, a synoptic pathology report consists of pairs of data elements (the item being measured) and responses (the measurements being reported). These pairs may be either required or optional. Table 1 lists data elements on the left and responses to the right; it also compares a synoptic report with a narrative report. The narrative report also includes descriptors that are not required; in a synoptic report, these could be included in a separate descriptive section.
Comparison of Synoptic and Narrative Reportsa.

a The same information in each report is in the same color font. The synoptic format allows easier retrieval of important tumor parameters and the checklist format helps ensure that all parameters are reported.
Synoptic reporting has been shown to make pathology reports more readable to clinicians and patients, 96 as well as making reports more likely to include all data elements needed; however, synoptic protocols include data elements that have been established as diagnostic, prognostic, or predictive. 60,61,78,106 To develop an effective synoptic report typically requires the efforts of pathologists and clinicians, who develop the checklist of required and recommended items after reviewing the relevant literature. 26 Currently, there are 2 main groups producing templates in human medicine, CAP and the International Collaboration on Cancer Reporting (ICCR). Both require a committee of pathologists, oncologists, and other interested representatives (eg, World Health Organization working groups, etc) to develop a new protocol.
A number of studies have found that synoptic reporting produces reports that are more likely to contain all significant pieces of information than narrative reports. For pancreatic tumors, 100% of synoptic reports had information about small vessel and perineural invasion, compared to 66% and 84% of narrative reports, respectively. 53 In addition, the stage could be determined in 100% of synoptic reports compared to 56% of narrative reports. In a comparison of melanoma reports, MC, histologic subtype, predominant cell type, vascular and lymphatic invasion, neurotropism, desmoplasia, and distance to the nearest margin were all reported significantly more frequently in synoptic reports than narrative reports, both at the teaching institution responsible for the study and the outside reports sent into the teaching institution for a second opinion. 61 Omission of important data elements has also been reported in a survey of surgical pathology reports (n = 368) of canine cutaneous mast cell tumors. Margins were reported in 92% of cases; however, lateral and deep margins were described separately in 77% of cases, margin direction was only given in 16% of cases, and descriptions of the deep margin component were only available in 11% of cases. 95 A review of tumor margins for canine MCT and STT reported that only 7.5% of published articles reported quantitative methods to evaluate margins and that the majority reported histologic margins as “complete” or incomplete” (dichotomously). 2
While full implementation of standardized reporting would allow for easy automated data collection, 43 even simple implementations of synoptic reporting can allow for significant automated information extraction. For example, if all deep margins are listed as “DEEP MARGIN: <xx>mm” on a line by itself, it is comparatively easy to extract all margins from reports using standard text search and manipulation tools (eg, grep, cut). Parameters which have been determined to be predictive on the basis of standardized studies can be incorporated into synoptic reports, ensuring inclusion of information critical for patient care.
From the beginning of synoptic reporting, clinicians have reported increased satisfaction with synoptic versus narrative reports. 73 A study of treating physicians and pathologists in Canada found that both groups thought information was easier to find in synoptic reports, facilitated a consistent approach to interpretation of diagnostic and prognostic factors, and provided higher overall satisfaction. 66 While pathologists felt that reports took approximately 25% to 50% longer to complete, treating physicians did not notice a difference in the length of time it took pathology reports to be completed.
The major problem in veterinary medicine is a lack of knowledge about factors involved in prognosis. As discussed in the other Supplemental Files in this document, there is little standardization of methods used in determining prognostic factors. There are also no standards for terminology, such as immunohistochemical findings (eg, “positive” vs “immunoreactive” vs “present”), which hinders design of standardized reports. Another issue for many pathologists, particularly in academia, is the effect switching to synoptic reports would have on resident training. Given the necessity of writing descriptions for boards and the lack of universal adoption of synoptic reporting, residents still require experience in writing narrative reports. This can be mitigated by requiring narrative reports in other resident educational settings (such as rounds) to provide practice in writing narrative reports for neoplasms. The checklist approach provides trainees with the important criteria to be searched for and therefore is educational. Possible adoption of the synoptic reporting concept is worthy of discussion by a broad group of stake holders representing various pathology practice settings (state diagnostic labs, commercial labs, teaching hospital labs, governmental reporting, industry, toxicologic pathology, and international organizations). If there is traction toward adoption of synoptic reporting, then further engagement and interaction between coordinators of training programs and certifying examinations (internationally) could mitigate potential issues.
Many pathologists are concerned about increased time to finish reports with synoptic reporting, including physicians; however, when synoptic reports have been implemented 66 many of these concerns have been deemed technology related rather than issues with the reporting format. As with many new processes, we assume that once the pathologists become familiarized with the new format, there will be a decrease in time needed to write these types of reports. A standardized formatted template will be created and added to the website we propose. In veterinary medicine, no current laboratory information management system (LIMS) can use synoptic reporting, which may seem like an obstacle to implementation of synoptic reporting. However, any word processor can be used to implement synoptic reporting without specialized software; 43 all that is required is to type the data element, a separator (such as TAB), and the response. Templates can be saved containing required and optional data elements, making it easier for pathologists to fill out reports quickly. These can then be copied and pasted into any LIMS or word processor for subsequent reporting.
Finally, another major obstacle to implementation of synoptic reporting is a lack of awareness of synoptic reporting and its benefits in veterinary medicine. Establishing working groups with pathologists and oncologists to develop guidelines for specific neoplasms would help promote awareness and develop reporting checklists that would benefit both pathologists and treating clinicians. Knowing that templates for synoptic tumor reports were created by oncologists and pathologists working together should add confidence to the format.
The next step beyond synoptic reporting is standardized reporting, that is, having a standardized, specific set of responses for each required question. Ultimately, 106 this can lead to automated staging and grading, as well as improving data harvesting for future research and clinical applications. The addition of free text fields associated with standardized options would allow for customization of reports while retaining standardization for further applications. As standards are developed, templates for synoptic reporting will need to be revised. To allow for standardization, once created, each report template will be assigned a version number. The website will list the current version, provide notifications to denote changes to templates, archive old versions, and list changes between versions. Finally, the template version could also be added as a required data element to reports, making it clear which version was used for any particular report. A similar approach is planned for updated versions of guidelines, protocols, and appendices, analogous to what is presently done by CAP.
References: Synoptic Reporting
2. Abrams BE, Putterman AB, Ruple A, et al. Variability in tumor margin reporting for soft tissue sarcoma and cutaneous mast cell tumors in dogs: a systematic review. Vet Surg. 2021;
26. Chamberlain DW, Wenckebach GF, Alexander F, et al. Pathological examination and the reporting of lung cancer specimens. Clin Lung Cancer. 2000;
29. College of American Pathologists. Cancer Protocol Templates 2020. Accessed April 15, 2021. https://www.cap.org/protocols-and-guidelines/cancer-reporting-tools/cancer-protocol-templates
43. Ellis D, Srigley J. Does standardised structured reporting contribute to quality in diagnostic pathology? The importance of evidence-based datasets. Virchow Arch. 2016;
53. Gill AJ, Johns AL, Eckstein R, et al. Synoptic reporting improves histopathological assessment of pancreatic resection specimens. Pathology. 2009;
60. Kang HP, Devine LJ, Piccoli AL, et al. Usefulness of a synoptic data tool for reporting of head and neck neoplasms based on the college of American pathologists cancer checklists. Am J Clin Pathol. 2009;
61. Karim R, Van Den Berg K, Colman M, et al. The advantage of using a synoptic pathology report format for cutaneous melanoma. Histopathology. 2008;
66. Lankshear S, Srigley J, McGowan T, et al. Standardized synoptic cancer pathology reports—so what and who cares? A population-based satisfaction survey of 970 pathologists, surgeons, and oncologists. Arch Pathol Lab Med. 2013;
73. Markel SF, Hirsch SD. Synoptic surgical pathology reporting. Hum Pathol. 1991;
78. Messenger DE, McLeod RS, Kirsch R. What impact has the introduction of a synoptic report for rectal cancer had on reporting outcomes for specialist gastrointestinal and nongastrointestinal pathologists? Arch Pathol Lab Med. 2011;
95. Reagan JK, Selmic LE, Fallon C, et al. Evaluation of information presented within mast cell tumour histopathology reports in the United States: 2012–2015. Vet Med Sci. 2018;
96. Renshaw AA, Mena-Allauca M, Gould EW, et al. Synoptic reporting: evidence-based review and future directions. JCO Clin Cancer Inform. 2018;
106. Srigley JR, McGowan T, MacLean A, et al. Standardized synoptic cancer pathology reporting: a population-based approach. J Surg Oncol. 2009;
Guideline 7. Assessment of Clinical Outcomes
For additional details and definitions see Appendix 1 and cRECIST v 1.0, 86 see Supplemental File 07—Assessment of Clinical Outcomes.
Standardized methods of histologic and outcome assessment parameters for animal tumors are essential if we wish to compare studies and apply the data to clinical cases. The Guidelines herein are an attempt to address this goal and complement existing literature. Outcome assessment of clinical patients is required for determining the predictability of histologically determined prognostic measures (eg, tumor classification, grade, etc). Outcome assessment data need to be collected as carefully and accurately as the techniques used to assess tumors. 125 Some criteria are subjective, clinical, and out of the realm of pathology. Clinicians must carefully select and standardize clinical outcome measures to avoid potential confounders. For example, reporting either disease- or progression-free interval is preferable to median survival time, in order to avoid the confounding effect of timing of euthanasia, which reflects individual biases present within owners and clinicians. Pathologists play a critical role in accurately determining both progression- and disease-free intervals by providing definitive determination of whether the same tumor recurred and/or metastasized given the appropriate tissue. While obtaining samples for histopathology presents more of a challenge than noninvasive imaging modalities and many advances have been made in this realm, microscopic examination of tissues remains the gold standard.
Histopathologic assessment also has advantages over cytologic evaluation as tissue architecture provides more information. Histologic examination is needed in order to differentiate fibroplasia from recurrent perivascular wall tumor at the original excision site and in re-excision specimens. However, even with histopathology it is difficult to differentiate these 2 processes or to find tumor cells in re-excision specimens. There is no standard means to evaluate these cases (clinically and histologically), and in at least one study of STT/STSs, presence or absence of tumor in re-excision specimens did not accurately predict recurrence. 13 Future studies could include imaging modalities, and correlate outcome with the presence of normal tissue at the margins of resected samples (eg, no spindle cells of any type). The surgical procedure as well as the tumor type and quantity of tumor within the specimen influences the feasibility of identifying tumor cells at the surgical margin, which impacts the concern for local recurrence.
Genetic studies have shown human and animal breed susceptibilities to develop specific tumor types and multiple tumors in the same host. Multiple aggressive tumors can be present in the same dog (Golden Retriever, Rottweiler, Bernese Mountain dogs, and others). 33 Given this tendency, it is essential to make a definitive diagnosis of tumors in metastatic sites. Combining methodologies is ideal but practical considerations of costs and emotional factors affect study results. When results from the clinical impression (which often includes diagnostic imaging) and histopathology differ, the relevant clinicians and diagnosticians should collaborate as a multidisciplinary team to reconcile the findings and optimize delivery of patient care. Imaging cannot determine whether the tumor suspected of being in the lungs is the same tumor as was excised previously. These methods to identify suspected neoplasia are the methods of choice for clinical settings but not research models. Light microscopy remains the gold standard to develop ground truths for metastasis and recurrence. We can substitute other methods for histopathology, but the data should be labelled suspected neoplasia/metastases (eg, as determined by imaging or physical exam) but not confirmed unless histopathology is used. In the future, molecular testing of suspected tumor tissue may be superior to histopathology.
Other Supplemental Files have detailed how to assess parameters used to evaluate a tumor, recurrence, margins, and metastases. In order to use morphologic diagnoses, margins, LVI, MC, lymph node status, or CPATH to predict tumor behavior and/or to select treatment options, oncologists must acquire sufficient outcome assessment information to allow interpretation of tumor parameters. Knowing actual survival times of geriatric pets or including pets in which no treatments were performed provides control groups to which treatments and outcomes can be compared. Determining the least invasive means to characterize tumor behavior is ideal but cannot be accomplished without adequate outcome assessment studies. Supplemental File 07 utilizes and expands upon published guidelines for conduct and evaluation of prognostic studies 125 and for response assessment in canine solid tumors, 86 citing specific information gained from studies of canine soft tissue sarcoma and canine mast cell tumor.
Definitions and terminology used for clinical outcome measures should be consistent across studies to facilitate comparisons in the literature. Appendix 1 summarizes some of the most important terminology for readers, especially pathologists who may be less familiar with these clinical definitions, for ease of reference while using the online resource. However, complete definitions are found in widely adopted references, 86,125 and readers are encouraged to consult these publications for details. The materials and methods section of articles must contain complete and clear definitions of the outcomes measured and the modalities used, such that others can replicate the study. Recurrence or metastasis are straightforward to a pathologist; however, survival can be defined in multiple ways. Determining survival probabilities at defined time points (1, 2, 5 years) is critical to compare different prognostic studies; moreover, reporting hazard ratios between groups is considered the best indicator of the prognostic significance for a given parameter (eg, MC) or treatment effect. Clinicians are positioned to determine outcomes, through careful use of correct terminology and diagnostic methodology, and report the findings with appropriate statistical methods. They also are the individuals that must balance patient care and individual owner scenarios while striving for accurate outcome data, which is a difficult task, emphasizing the need for transparency and consistency in outcome reporting.
Standardized criteria, such as RECIST and RECIST 1.1, 102,114 should be used to document the patient’s response to treatment and progressive disease. The RECIST 1.1 criteria have been robustly evaluated for use in human clinical trials and can be easily adapted to the evaluation of veterinary patients. Pathologists, oncologists, surgeons, clinicians, and students should be familiar with the terms explained in these articles, which indicate response to treatment and include Complete response (CR), Partial response (PR), Progressive disease (PD), Stable disease (SD), and Not evaluable (NE). 86 Documented progression is needed in the cases of questionable lesions, or a minimum size is required to determine whether neoplastic disease is present within a lymph node. Additionally, there may be specific anatomical locations evaluated depending on the tumor type. For example, prostate cancer may favor bone metastases, pulmonary carcinoma in cats requires assessment of all digits, and hemangiosarcoma is the most common metastatic tumor to the brain of dogs. Ideally, imaging will be used in concert with biopsy or autopsy in order to confirm recurrence and metastasis with the utmost accuracy.
Metastasis should be subdivided into confirmed and suspected. Metastases determined by imaging only should be labelled suspected. Light microscopy is required to confirm metastases are present and are of the same tumor type. The preferred methodology of evaluation in humans, the CT scan, should be used if possible as it avoids some of the technical problems associated with the use of radiographs, whereas ultrasound is not an acceptable method of assessing disease state. 86 The use of functional imaging (PET/SC scans) is increasingly common to better determine sites of disease; however, it cannot be used for measuring purposes. Following these standardized criteria will ensure that studies can be reproduced and compared between institutions, resulting in more useful correlates of clinical data to prognostic information, and ensuring progress in veterinary oncologic pathology.
Euthanasia is a reality of veterinary medicine, and oncology studies that use pets must carefully evaluate how decisions to euthanize influenced survival times. Reported patient survival times are affected by euthanasia which may be elected due to perceived pet value, owner income, primary versus referral centers, or other factors which do not reflect tumor behavior. When patients are euthanized, clinicians should determine and/or record the cause of death with as much accuracy as possible. If euthanasia is due to an unrelated disease process, this must be noted. If euthanasia is caused by the neoplasm being studied, and cachexia is present, then histologic confirmation of the extent of the neoplastic disease helps verify clinical observations and reliability of study conclusions. Oncology studies no longer include results of autopsy, 32,109 the perceived value of which seems to have hit a nadir. Permission to perform autopsies should be pursued as autopsy greatly increases the confidence in results from the case. Studies should set a goal of autopsies on at least 20% of the cases.
References: Outcome Assessments
13. Bacon NJ, Dernell WS, Ehrhart N, et al. Evaluation of primary re-excision after recent inadequate resection of soft tissue sarcomas in dogs: 41 cases (1999–2004). J Am Vet Med Assoc. 2007;
32. Crownshaw AH, McEntee MC, Nolan MW, et al. Evaluation of variables associated with outcomes in 41 dogs with incompletely excised high-grade soft tissue sarcomas treated with definitive-intent radiation therapy with or without chemotherapy. J Am Vet Med Assoc. 2020;
33. Cullen JM, Breen M. An overview of molecular cancer pathogenesis, prognosis, and diagnosis. In: Meuten DJ, ed. Tumors in Domestic Animals. 5th ed. John Wiley; 2017:1–26.
86. Nguyen SM, Thamm DH, Vail DM, et al. Response evaluation criteria for solid tumours in dogs (v1.0): a veterinary cooperative oncology group (VCOG) consensus document. Vet Comp Oncol. 2015;
102. Schwartz LH, Seymour L, Litiere S, et al. RECIST 1.1—standardisation and disease-specific adaptations: perspectives from the RECIST Working Group. Eur J Cancer. 2016;
109. Stefanello D, Avallone G, Ferrari R, et al. Canine cutaneous perivascular wall tumors at first presentation: clinical behavior and prognostic factors in 55 cases. J Vet Intern Med. 2011;
114. Therasse P, Arbuck SG, Eisenhauer EA, et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst. 2000;
125. Webster J, Dennis MM, Dervisis N, et al. Recommended guidelines for the conduct and evaluation of prognostic studies in veterinary oncology. Vet Pathol. 2011;
Guideline 11. Computational Pathology
For additional details, see Supplemental File 08—Computational Pathology for Tumor Histopathology.
Computational pathology (CPATH) is an umbrella term used to broadly encompass computerized/automated gathering of information on disease in patients. 1 Although CPATH may use a large variety of information sources (raw medical data: histology images, radiology images, gene sequences, clinical records), Supplemental File 08 focuses only on automated image analysis (AIA) of microscopic tumor images, particularly whole slide images (WSI). When used appropriately, CPATH is a promising tool which uses microscopic images (input data) and automatically produces output information (counts or scores of patterns, classification of images, etc). It allows the evaluation of large amounts of data (such as histological tumor sections) on an unprecedented scale, which is likely to reveal novel trends of prognostic importance. As AIA is a relatively new modality of analysis in veterinary pathology with a vast number of relevant methods, this field can be overwhelming with respect to terminology, technical aspects, requirements for developing algorithms, performance validation, and implementation strategies. Therefore, Supplemental File 08 aims to give an overview of relevant terms, general considerations of CPATH methods, and specific recommendations for individual prognostic parameters. Generally, 2 broad categories of AIA approaches are applicable for microscopic tumor prognostication: (1) algorithms with manual adjustment (most commonly using thresholding) and (2) advanced data-driven approaches. Algorithms with manual adjustment use a set of simple, often programmer-designed image processing steps based on the color information of individual pixels, which are especially useful for scoring immunohistochemical labeling intensity. For instance, brown or red pixels of immunohistochemical images (depending on the chromogen used) that belong to an individual cellular compartment (membrane, cytoplasm, nucleus) can be classified based on their color intensity and cells are graded (usually in 0, 1+, 2+, 3+) based on preset intensity thresholds. Complex patterns such as MF can only be poorly predicted by thresholding-based algorithms.
Data-driven approaches learn to retrieve meaningful patterns from images in order to derive the desired information using AI. AI can be used with traditional machine learning methods (such as Bayesian or random forest networks; for more details, see Awaysheh et al 11 ) that require “hand-crafted” (by developer) information about relevant features of the pattern, or more sophisticated deep learning methods (using artificial neuronal networks with multiple hidden layers) that autonomously extract relevant features (decision criteria are unknown to developers, “black box”). Deep learning is generally more powerful than traditional machine learning methods but necessitates larger amounts of data. For histological images, supervised learning (as opposed to unsupervised learning) is a very useful method that learns by “feedback” from ground truth labels assigned to the input images. Creating those labels is a very time-consuming task and is prone to several biases (see Supplemental File 08).
Possible applications of AIA for tumor prognostication are seemingly limitless and various benefits of these approaches have been determined in previous studies. 7,107,111 Compared to manual assessment by pathologists, AIA has higher reproducibility, may have higher accuracy, may increase efficiency of repetitive tasks (such as counting of MF), and can carefully assess vast amounts of data per case (every image section of multiple WSIs at high magnification) without fatigue. AIA of immunohistochemical labeling intensity was reported to have higher reproducibility and improved prognostic value compared to the manual approach by pathologists for Ki67 index in human breast cancer, 107 and membrane-binding biomarkers in human esophageal adenocarcinomas. 48 Deep learning approaches for MF identification in H&E stained tumor sections have been developed for human 7,121 and canine breast cancer 7 as well as canine mast cell tumors. 18 Deep learning-based algorithms are comparable with pathologists for counting MF (in the same tumor regions) 121 and outperform pathologists in identifying the “hot spot” regions in WSI. 8 However, correlation of algorithmic MC to patient outcome has not yet been investigated in human and animal tumors. For automated metastasis identification in H&E sections, deep learning–based algorithms can be used for prescreening of images, and a computer-assisted approach has been shown to have higher sensitivity and diagnostic speed compared to the unassisted pathologist. 111 Recent studies on tumors from humans reported that the systems used could even predict if a tumor was benign, carcinoma in situ, or invasive carcinoma 6 as well as predict genetic alterations and gene expression from H&E tumor sections. 62
Algorithms are not flawless, have multiple sources of error (depending on the algorithmic approach and available dataset), and therefore require very careful validation (see Supplemental File 08). While algorithms with manual adjustment have a high ability to explain algorithmic predictions, data-driven approaches are often considered a “black box” as decision criteria of the algorithms are typically unavailable. Although algorithms are 100% reproducible (same result for the same image using the same model), they may not necessarily cope with variability introduced via biological and pre-analytic factors (tumor type, tissue types present, section preparation, and image acquisition). For example, a deep learning–based algorithm for MF may perform poorly on images obtained from a WSI scanner that was not used for the training images. 7 If not part of the training data, algorithms can be compromised by images with very poor tissue or image quality (artifacts, poor fixation, etc). In contrast to algorithms with manual adjustment, data-driven algorithms are, however, capable of learning a certain degree of image variability and training datasets should include realistic variability that reflects the intended use. Performance evaluation should be done with great care, and data-driven approaches can be assessed by mathematical evaluation (see Supplemental File 08), 1 whereas algorithms with manual adjustment are often only assessed visually by a pathologist. 3
While diagnosis of tissue structures by pathologists is greatly influenced by surrounding context (spatial awareness), there may be limitations to spatial awareness of algorithms, depending on training data and approach. For example, a deep learning–based MF algorithm may detect MF outside the tumor tissue; pathologists would exclude this region for their assessment. However, there are possible methods to improve algorithms, such as by using a preceding algorithm for segmentation of the tumor area and exclusion of necrotic and non-neoplastic tissue. Besides the numerous hurdles in development of AIA algorithms, there are practical issues to consider for bringing AIA into diagnostic workflows. Basic requirements include consistent tissue preparation steps, a digital image acquisition workflow, appropriate IT infrastructure, and sufficient computational power. Increasing implementation of digital microscopy in veterinary laboratories 20 will augment access to WSI and facilitate AIA. Nevertheless, acceptance of AIA may be hampered by unfamiliarity, limited research results and a poor ability to explain machine learning-based algorithms (“black box”). However, there are approaches that can convert the “black box” into a more transparent “glass box” that are likely to have higher acceptance. For example, some algorithms can be implemented as computer-assisted prognosis systems (as opposed to fully computerized decisions) that always require review by a pathologist. These approaches will improve the reliability of the computer assisted prognosis system and allow the reviewing pathologist to retain responsibility in making final decisions with regard to these prognostic parameters. AIA could greatly improve tumor prognostication by providing vast amounts of reproducible and possibly accurate information on the tumor section, but interpretation of the result remains the responsibility of the pathologist.
References: CPATH
1. Abels E, Pantanowitz L, Aeffner F, et al. Computational pathology definitions, best practices, and recommendations for regulatory guidance: a white paper from the digital pathology association. J Pathol. 2019;
3. Aeffner F, Wilson K, Bolon B, et al. Commentary: roles for pathologists in a high-throughput image analysis team. Toxicol Pathol. 2016;
6. Aresta G, Araujo T, Kwok S, et al. BACH: grand challenge on breast cancer histology images. Med Image Anal. 2019;
7. Aubreville M, Bertram C, Donovan T, et al. A completely annotated whole slide image dataset of canine breast cancer to aid human breast cancer research. Sci Data. 2020:
8. Aubreville M, Bertram CA, Marzahl C, et al. Deep learning algorithms out-perform veterinary pathologists in detecting the mitotically most active tumor region. Sci Rep. 2020;
11. Awaysheh A, Wilcke J, Elvinger F, et al. Review of medical decision support and machine-learning methods. Vet Pathol. 2019;
18. Bertram CA, Aubreville M, Marzahl C, et al. A large-scale dataset for mitotic figure assessment on whole slide images of canine cutaneous mast cell tumor. Sci Data. 2019;
20. Bertram CA, Klopfleisch R. The Pathologist 2.0: an update on digital pathology in veterinary medicine. Vet Pathol. 2017;
48. Feuchtinger A, Stiehler T, Jutting U, et al. Image analysis of immunohistochemistry is superior to visual scoring as shown for patient outcome of esophageal adenocarcinoma. Histochem Cell Biol. 2015;
62. Kather JN, Heij LR, Grabsch HI, et al. Pan-cancer image-based detection of clinically actionable genetic alterations. Nat Cancer. 2020;
107. Stålhammar G, Robertson S, Wedlund L, et al. Digital image analysis of Ki67 in hot spots is superior to both manual Ki67 and mitotic counts in breast cancer. Histopathology. 2018;
111. Steiner DF, MacDonald R, Liu Y, et al. Impact of deep learning assistance on the histopathologic review of lymph nodes for metastatic breast cancer. Am J Surg Pathol. 2018;
121. Veta M, van Diest PJ, Jiwa M, et al. Mitosis counting in breast cancer: object-level interobserver agreement and comparison to an automatic method. PLoS One. 2016;
Protocol 1. Canine Skin and Subcutaneous Soft Tissue Tumors
For additional details, see Supplemental File 09—Canine Skin and Subcutaneous Soft Tissue Tumors.
This protocol is intended for use with soft tissue tumors arising in the skin and subcutaneous tissues of dogs which are predominantly of mesenchymal tissue origin and which are commonly referred to as soft tissue sarcomas (STS). 23,65,77,97 Modifying a name generally meets with resistance and lack of unanimity. The term sarcoma suggests the group of neoplasms are malignant; however, present outcome assessment data indicate that the majority do not recur and metastases are infrequent. 23,65,77,97 Thus, it is proposed to remove sarcoma from the acronym. These neoplasms are predominantly mesenchymal; however, a subset (namely nerve sheath tumors) are not solely derived from the mesoderm, therefore, soft tissue mesenchymal tumor is not entirely accurate. These neoplasms can be accurately encompassed by the term soft tissue tumors (STT) (which is admittedly vague); however, ensures that more users of this term will be satisfied. The purpose of this protocol is to provide standards for accruing data so that, over time, large data sets with comparable information can be evaluated to enable meaningful conclusions and accurate prognostic information.
The term STT/STS encompasses a wide range of tumor types in humans. 25 The different types are much more limited in animals, and although the veterinary terminology and various grading schemes have, in many instances, been borrowed from the human literature, the types of neoplasms which commonly comprise soft tissue tumors in humans are very different from the tumor types typically encountered in animals. This is exemplified by liposarcomas, which are common in humans and rare in dogs, and perivascular wall tumors (PWT), very common in dogs, are rare in humans. Furthermore, STS in humans have extensive molecular profiles to help subtype them, which are not established for canine tumors. The common denominators between species appears to be an origin in nonepithelial, extraskeletal soft tissues exclusive of the hematopoietic system. 23,28,34,65,77,97,115 This protocol is intended for use with the following types of tumors: perivascular wall tumors (PWT), nerve sheath tumors (NST), fibrosarcoma, myxosarcoma, leiomyosarcoma, liposarcoma, rhabdomyosarcoma, or unclassified spindle cell tumor/sarcoma arising in the dermis or subcutis. PWT and NST are the most common types of STT/STS and their biological behavior is primarily indolent. 97 The effect of grouping of disparate tumors within the same grading scheme needs to be compared to grading tumors segmented into specific histological diagnoses so that important predictive parameters may be determined.
The 2 current schemes used for grading dog STT/STS 55,77 are both patterned after Trojani’s (French) grading of human STS but with slight modifications. 115 Unlike the original human grading scheme, however, the studies of dog STT/STS only evaluated 3 histological features. Four additional histologic features evaluated by Trojani but not found useful for human tumors were not assessed in the dog STT/STS grading studies. Our existing scheme needs to be broadened to determine if parameters originally rejected for human STS may, in fact, be prognostic in dogs. Some criteria, such as determination of the percentage of necrosis via gross and/or histological evaluation, are poorly defined in the human literature and were not clarified in the veterinary manuscripts. 23,65,77 Percent necrosis for human tumors was determined by estimating the amount seen grossly and histologically (see Supplemental File 05). 28,86,99,115 In addition, there are a number of distinctions between the grading systems used for human tumors and how they are applied to dogs, which have not been addressed in the canine papers; 23,28,65,77,115 in particular, the need to determine histologic tumor type and confirmation of the diagnosis of sarcoma prior to applying the human grading systems. The methods described to assign scores for necrosis, MC, and differentiation for canine tumors are not detailed enough such that others can replicate them, and the number of dogs reported with high-grade STT/STS that have outcome assessments is small. These studies need to be repeated with additional parameters evaluated, more detailed description of methods, and greater case numbers paired with standardized outcome assessments. The protocol in Supplemental File 09 provides details of the histological findings that should be noted in STT/STS which will enable more thorough assessment of these tumors and should provide a database for performance of studies and validation of grading schemes.
For any proposed veterinary tumor grading system, the tumor type should be designated as precisely as possible and the criteria used to arrive at that diagnosis be provided (H&E, IHC, etc). Each graded element must be clearly defined. For instance, the means to grossly or histologically assess percent necrosis (Supplemental File 05) must be clarified if this is an element of a grading system and others are expected to duplicate the method. 65 Histologic classification of some types of STT/STS is difficult. A particular conundrum is differentiating PWT from NST. Histologic features characteristic of PWT and NST have been described, but there is overlap of histologic patterns found in these 2 tumor types, 9,10,71,97,113,124 which can complicate definitive diagnosis in routinely stained sections. How specific can, or should our diagnoses be from HE slides and how does this influence differentiation scores used to grade these tumors? Examples: Should PWT be subtyped, and similarly as NST is not just one tumor, should neurofibroma, Schwannoma, and malignant NST be identified? Classification of some tumors may require IHC or other ancillary tests. In veterinary medicine, the costs for these tests are incurred by owners, and if the tests are declined, it is unreasonable to expect a precise classification of some of these tumors with H&E alone. These practical factors influence our diagnoses and grading systems.
Present canine studies have not determined if identifying tumor type is predictive of tumor behavior. Until we use a grading system for specific tumor types as well as for the entire group of STT/STS, we will not know which approach is more predictive. A grading scheme that can be applied to any tumor within the STT/STS group is easier to apply than requiring identification of the specific tumor type before grading, particularly in instances in which a definitive diagnosis cannot be made with evaluation of routinely stained tissue sections. However, future studies should validate if this is “best practice.” On the one hand, identifying the precise tumor type may have prognostic information unrelated to a specific grade, and on the other hand, perhaps all tumors within either the entire STT/STS group or within specified subsets of the group (eg, PWT/NST) may behave according to assigned grades. For instance, PWT and NST can be grouped together based on H&E histologic morphology and outcomes can be determined to see if there are any differences in outcome assessment if these 2 tumor types are evaluated separately. Definitive identification of these 2 tumor types may involve IHC or electron microscopy. If the biological behavior of these 2 tumors was such that distinguishing them at the H&E level was not needed, that information would have practical use for a diagnostic pathologist and oncologist. The only means to determine the prognostic utility of grouping or separating tumor types within the STT/STS category is to perform studies which evaluate outcomes related to the STT/STS group as a whole and also evaluate outcomes in relation to specific histologic type of tumors. Studies must have sufficient numbers of animals within each tumor grade to generate statistically significant findings. This latter issue will be a problem for uncommon tumors, such as liposarcoma, for which it may be problematic to find enough high-grade tumors with accurate outcome assessments, but using criteria in which 2 tumor types (ie, PWT and NST) comprise more than 80% of the cases to predict how uncommon tumors behave needs to be validated.
Future considerations should address existing and new grading systems for STT/STS (see Protocol 1). The present grading system should be followed with methods described in sufficient detail to permit other investigators to duplicate the methods and the scoring systems. Consideration should be given to assessment of weighted scores for parameters, such as differentiation or MC, in determining grade and correlation with outcome assessment. Additional histological features should be evaluated for their prognostic utility, for instance, tumor cellularity, presence of atypical nuclei or multinucleated giant cells, and presence of lymphovascular invasion (see Supplemental File 04). The benefit of applying a new, better-detailed scoring system for histological differentiation should be assessed as this is the most subjective parameter in human tumors and likely canine tumors. The use of a defined area in mm2 should be applied to all parameters enumerated in a grading system. New grading systems should be compared to older systems, and there must be sufficient numbers of animals in each tumor grade to enable interpretation of results. Studies should be initiated to assess the criteria for diagnosis of NST and PWT and the reproducibility of the criteria. Standards for assessment of re-excision biopsy specimens should be established and results correlated with outcome assessment. Finally, the use of computational pathology and molecular profiling should be explored in determining grades and outcomes of STT/STS. A complete list of references is available in Supplemental File 09.
References: STT
9. Avallone G, Helmbold P, Caniatti M, et al. The spectrum of canine cutaneous perivascular wall tumors: morphologic, phenotypic and clinical characterization. Vet Pathol. 2007;
10. Avallone G, Stefanello D, Ferrari R, et al. The controversial histologic classification of canine subcutaneous whorling tumours: the path to perivascular wall tumours. Vet Comp Oncol. 2020;
23. Bostock DE, Dye MT. Prognosis after surgical excision of canine fibrous connective tissue sarcomas. Vet Pathol. 1980;
25. Byerly S, Chopra S, Nassif NA, et al. The role of margins in extremity soft tissue sarcoma. J Surg Oncol. 2016;
28. Coindre JM. Grading of soft tissue sarcomas: review and update. Arch Pathol Lab Med. 2006;
34. Dennis M, McSporran K, Bacon N, et al. Prognostic factors for cutaneous and subcutaneous soft tissue sarcomas in dogs. Vet Pathol. 2011;
65. Kuntz CA, Dernell WS, Powers BE, et al. Prognostic factors for surgical treatment of soft-tissue sarcomas in dogs: 75 cases (1986–1996). J Am Vet Med Assoc. 1997;
71. Loures F, Conceição L, Lauffer-Amorim R, et al. Histopathology and immunohistochemistry of peripheral neural sheath tumor and perivascular wall tumor in dog. Arq Bras de Med Vet Zootec. 2019;
77. McSporran KD. Histologic grade predicts recurrence for marginally excised canine subcutaneous soft tissue sarcomas. Vet Pathol. 2009;
86. Nguyen SM, Thamm DH, Vail DM, et al. Response evaluation criteria for solid tumours in dogs (v1.0): a veterinary cooperative oncology group (VCOG) consensus document. Vet Comp Oncol. 2015;
97. Roccabianca P, Schulman FY, et al. In: Kiupel M, ed. Tumors of Soft Tissue. Vol 3. Davis Thompson DVM Foundation; 2020.
99. Rubin BP, Cooper K, Fletcher CD, et al. Protocol for the examination of specimens from patients with tumors of soft tissue. Arch Pathol Lab Med. 2010;
113. Suzuki S, Uchida K, Nakayama H. The effects of tumor location on diagnostic criteria for canine malignant peripheral nerve sheath tumors (MPNSTs) and the markers for distinction between canine MPNSTs and canine perivascular wall tumors. Vet Pathol. 2014;
115. Trojani M, Contesso G, Coindre JM, et al. Soft-tissue sarcomas of adults; study of pathological prognostic variables and definition of a histopathological grading system. Int J Cancer. 1984;
124. Vučićević I, Marinković D, Kukolj V, et al. Immunohistochemical distinguishing between canine peripheral nerve sheath tumors and perivascular wall tumors. Acta Veterinaria. 2019;
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211013712 - International Guidelines for Veterinary Tumor Pathology: A Call to Action
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211013712 for International Guidelines for Veterinary Tumor Pathology: A Call to Action by Donald J. Meuten, Frances M. Moore, Taryn A. Donovan, Christof A. Bertram, Robert Klopfleisch, Robert A. Foster, Rebecca C. Smedley, Michael J. Dark, Milan Milovancev, Paul Stromberg, Bruce H. Williams, Marc Aubreville, Giancarlo Avallone, Pompei Bolfa, John Cullen, Michelle M. Dennis, Michael Goldschmidt, Richard Luong, Andrew D. Miller, Margaret A. Miller, John S. Munday, Paola Roccabianca, Elisa N. Salas, F. Yvonne Schulman, Renee Laufer-Amorim, Midori G. Asakawa, Linden Craig, Nick Dervisis, D. Glen Esplin, Jeanne W. George, Marlene Hauck, Yumiko Kagawa, Matti Kiupel, Keith Linder, Kristina Meichner, Laura Marconato, Michelle L. Oblak, Renato L. Santos, R. Mark Simpson, Harold Tvedten and Derick Whitley in Veterinary Pathology
Supplemental Material
Supplemental Material, sj-pdf-10-vet-10.1177_03009858211013712 - International Guidelines for Veterinary Tumor Pathology: A Call to Action
Supplemental Material, sj-pdf-10-vet-10.1177_03009858211013712 for International Guidelines for Veterinary Tumor Pathology: A Call to Action by Donald J. Meuten, Frances M. Moore, Taryn A. Donovan, Christof A. Bertram, Robert Klopfleisch, Robert A. Foster, Rebecca C. Smedley, Michael J. Dark, Milan Milovancev, Paul Stromberg, Bruce H. Williams, Marc Aubreville, Giancarlo Avallone, Pompei Bolfa, John Cullen, Michelle M. Dennis, Michael Goldschmidt, Richard Luong, Andrew D. Miller, Margaret A. Miller, John S. Munday, Paola Roccabianca, Elisa N. Salas, F. Yvonne Schulman, Renee Laufer-Amorim, Midori G. Asakawa, Linden Craig, Nick Dervisis, D. Glen Esplin, Jeanne W. George, Marlene Hauck, Yumiko Kagawa, Matti Kiupel, Keith Linder, Kristina Meichner, Laura Marconato, Michelle L. Oblak, Renato L. Santos, R. Mark Simpson, Harold Tvedten and Derick Whitley in Veterinary Pathology
Supplemental Material
Supplemental Material, sj-pdf-11-vet-10.1177_03009858211013712 - International Guidelines for Veterinary Tumor Pathology: A Call to Action
Supplemental Material, sj-pdf-11-vet-10.1177_03009858211013712 for International Guidelines for Veterinary Tumor Pathology: A Call to Action by Donald J. Meuten, Frances M. Moore, Taryn A. Donovan, Christof A. Bertram, Robert Klopfleisch, Robert A. Foster, Rebecca C. Smedley, Michael J. Dark, Milan Milovancev, Paul Stromberg, Bruce H. Williams, Marc Aubreville, Giancarlo Avallone, Pompei Bolfa, John Cullen, Michelle M. Dennis, Michael Goldschmidt, Richard Luong, Andrew D. Miller, Margaret A. Miller, John S. Munday, Paola Roccabianca, Elisa N. Salas, F. Yvonne Schulman, Renee Laufer-Amorim, Midori G. Asakawa, Linden Craig, Nick Dervisis, D. Glen Esplin, Jeanne W. George, Marlene Hauck, Yumiko Kagawa, Matti Kiupel, Keith Linder, Kristina Meichner, Laura Marconato, Michelle L. Oblak, Renato L. Santos, R. Mark Simpson, Harold Tvedten and Derick Whitley in Veterinary Pathology
Supplemental Material
Supplemental Material, sj-pdf-12-vet-10.1177_03009858211013712 - International Guidelines for Veterinary Tumor Pathology: A Call to Action
Supplemental Material, sj-pdf-12-vet-10.1177_03009858211013712 for International Guidelines for Veterinary Tumor Pathology: A Call to Action by Donald J. Meuten, Frances M. Moore, Taryn A. Donovan, Christof A. Bertram, Robert Klopfleisch, Robert A. Foster, Rebecca C. Smedley, Michael J. Dark, Milan Milovancev, Paul Stromberg, Bruce H. Williams, Marc Aubreville, Giancarlo Avallone, Pompei Bolfa, John Cullen, Michelle M. Dennis, Michael Goldschmidt, Richard Luong, Andrew D. Miller, Margaret A. Miller, John S. Munday, Paola Roccabianca, Elisa N. Salas, F. Yvonne Schulman, Renee Laufer-Amorim, Midori G. Asakawa, Linden Craig, Nick Dervisis, D. Glen Esplin, Jeanne W. George, Marlene Hauck, Yumiko Kagawa, Matti Kiupel, Keith Linder, Kristina Meichner, Laura Marconato, Michelle L. Oblak, Renato L. Santos, R. Mark Simpson, Harold Tvedten and Derick Whitley in Veterinary Pathology
Supplemental Material
Supplemental Material, sj-pdf-2-vet-10.1177_03009858211013712 - International Guidelines for Veterinary Tumor Pathology: A Call to Action
Supplemental Material, sj-pdf-2-vet-10.1177_03009858211013712 for International Guidelines for Veterinary Tumor Pathology: A Call to Action by Donald J. Meuten, Frances M. Moore, Taryn A. Donovan, Christof A. Bertram, Robert Klopfleisch, Robert A. Foster, Rebecca C. Smedley, Michael J. Dark, Milan Milovancev, Paul Stromberg, Bruce H. Williams, Marc Aubreville, Giancarlo Avallone, Pompei Bolfa, John Cullen, Michelle M. Dennis, Michael Goldschmidt, Richard Luong, Andrew D. Miller, Margaret A. Miller, John S. Munday, Paola Roccabianca, Elisa N. Salas, F. Yvonne Schulman, Renee Laufer-Amorim, Midori G. Asakawa, Linden Craig, Nick Dervisis, D. Glen Esplin, Jeanne W. George, Marlene Hauck, Yumiko Kagawa, Matti Kiupel, Keith Linder, Kristina Meichner, Laura Marconato, Michelle L. Oblak, Renato L. Santos, R. Mark Simpson, Harold Tvedten and Derick Whitley in Veterinary Pathology
Supplemental Material
Supplemental Material, sj-pdf-3-vet-10.1177_03009858211013712 - International Guidelines for Veterinary Tumor Pathology: A Call to Action
Supplemental Material, sj-pdf-3-vet-10.1177_03009858211013712 for International Guidelines for Veterinary Tumor Pathology: A Call to Action by Donald J. Meuten, Frances M. Moore, Taryn A. Donovan, Christof A. Bertram, Robert Klopfleisch, Robert A. Foster, Rebecca C. Smedley, Michael J. Dark, Milan Milovancev, Paul Stromberg, Bruce H. Williams, Marc Aubreville, Giancarlo Avallone, Pompei Bolfa, John Cullen, Michelle M. Dennis, Michael Goldschmidt, Richard Luong, Andrew D. Miller, Margaret A. Miller, John S. Munday, Paola Roccabianca, Elisa N. Salas, F. Yvonne Schulman, Renee Laufer-Amorim, Midori G. Asakawa, Linden Craig, Nick Dervisis, D. Glen Esplin, Jeanne W. George, Marlene Hauck, Yumiko Kagawa, Matti Kiupel, Keith Linder, Kristina Meichner, Laura Marconato, Michelle L. Oblak, Renato L. Santos, R. Mark Simpson, Harold Tvedten and Derick Whitley in Veterinary Pathology
Supplemental Material
Supplemental Material, sj-pdf-4-vet-10.1177_03009858211013712 - International Guidelines for Veterinary Tumor Pathology: A Call to Action
Supplemental Material, sj-pdf-4-vet-10.1177_03009858211013712 for International Guidelines for Veterinary Tumor Pathology: A Call to Action by Donald J. Meuten, Frances M. Moore, Taryn A. Donovan, Christof A. Bertram, Robert Klopfleisch, Robert A. Foster, Rebecca C. Smedley, Michael J. Dark, Milan Milovancev, Paul Stromberg, Bruce H. Williams, Marc Aubreville, Giancarlo Avallone, Pompei Bolfa, John Cullen, Michelle M. Dennis, Michael Goldschmidt, Richard Luong, Andrew D. Miller, Margaret A. Miller, John S. Munday, Paola Roccabianca, Elisa N. Salas, F. Yvonne Schulman, Renee Laufer-Amorim, Midori G. Asakawa, Linden Craig, Nick Dervisis, D. Glen Esplin, Jeanne W. George, Marlene Hauck, Yumiko Kagawa, Matti Kiupel, Keith Linder, Kristina Meichner, Laura Marconato, Michelle L. Oblak, Renato L. Santos, R. Mark Simpson, Harold Tvedten and Derick Whitley in Veterinary Pathology
Supplemental Material
Supplemental Material, sj-pdf-5-vet-10.1177_03009858211013712 - International Guidelines for Veterinary Tumor Pathology: A Call to Action
Supplemental Material, sj-pdf-5-vet-10.1177_03009858211013712 for International Guidelines for Veterinary Tumor Pathology: A Call to Action by Donald J. Meuten, Frances M. Moore, Taryn A. Donovan, Christof A. Bertram, Robert Klopfleisch, Robert A. Foster, Rebecca C. Smedley, Michael J. Dark, Milan Milovancev, Paul Stromberg, Bruce H. Williams, Marc Aubreville, Giancarlo Avallone, Pompei Bolfa, John Cullen, Michelle M. Dennis, Michael Goldschmidt, Richard Luong, Andrew D. Miller, Margaret A. Miller, John S. Munday, Paola Roccabianca, Elisa N. Salas, F. Yvonne Schulman, Renee Laufer-Amorim, Midori G. Asakawa, Linden Craig, Nick Dervisis, D. Glen Esplin, Jeanne W. George, Marlene Hauck, Yumiko Kagawa, Matti Kiupel, Keith Linder, Kristina Meichner, Laura Marconato, Michelle L. Oblak, Renato L. Santos, R. Mark Simpson, Harold Tvedten and Derick Whitley in Veterinary Pathology
Supplemental Material
Supplemental Material, sj-pdf-6-vet-10.1177_03009858211013712 - International Guidelines for Veterinary Tumor Pathology: A Call to Action
Supplemental Material, sj-pdf-6-vet-10.1177_03009858211013712 for International Guidelines for Veterinary Tumor Pathology: A Call to Action by Donald J. Meuten, Frances M. Moore, Taryn A. Donovan, Christof A. Bertram, Robert Klopfleisch, Robert A. Foster, Rebecca C. Smedley, Michael J. Dark, Milan Milovancev, Paul Stromberg, Bruce H. Williams, Marc Aubreville, Giancarlo Avallone, Pompei Bolfa, John Cullen, Michelle M. Dennis, Michael Goldschmidt, Richard Luong, Andrew D. Miller, Margaret A. Miller, John S. Munday, Paola Roccabianca, Elisa N. Salas, F. Yvonne Schulman, Renee Laufer-Amorim, Midori G. Asakawa, Linden Craig, Nick Dervisis, D. Glen Esplin, Jeanne W. George, Marlene Hauck, Yumiko Kagawa, Matti Kiupel, Keith Linder, Kristina Meichner, Laura Marconato, Michelle L. Oblak, Renato L. Santos, R. Mark Simpson, Harold Tvedten and Derick Whitley in Veterinary Pathology
Supplemental Material
Supplemental Material, sj-pdf-7-vet-10.1177_03009858211013712 - International Guidelines for Veterinary Tumor Pathology: A Call to Action
Supplemental Material, sj-pdf-7-vet-10.1177_03009858211013712 for International Guidelines for Veterinary Tumor Pathology: A Call to Action by Donald J. Meuten, Frances M. Moore, Taryn A. Donovan, Christof A. Bertram, Robert Klopfleisch, Robert A. Foster, Rebecca C. Smedley, Michael J. Dark, Milan Milovancev, Paul Stromberg, Bruce H. Williams, Marc Aubreville, Giancarlo Avallone, Pompei Bolfa, John Cullen, Michelle M. Dennis, Michael Goldschmidt, Richard Luong, Andrew D. Miller, Margaret A. Miller, John S. Munday, Paola Roccabianca, Elisa N. Salas, F. Yvonne Schulman, Renee Laufer-Amorim, Midori G. Asakawa, Linden Craig, Nick Dervisis, D. Glen Esplin, Jeanne W. George, Marlene Hauck, Yumiko Kagawa, Matti Kiupel, Keith Linder, Kristina Meichner, Laura Marconato, Michelle L. Oblak, Renato L. Santos, R. Mark Simpson, Harold Tvedten and Derick Whitley in Veterinary Pathology
Supplemental Material
Supplemental Material, sj-pdf-8-vet-10.1177_03009858211013712 - International Guidelines for Veterinary Tumor Pathology: A Call to Action
Supplemental Material, sj-pdf-8-vet-10.1177_03009858211013712 for International Guidelines for Veterinary Tumor Pathology: A Call to Action by Donald J. Meuten, Frances M. Moore, Taryn A. Donovan, Christof A. Bertram, Robert Klopfleisch, Robert A. Foster, Rebecca C. Smedley, Michael J. Dark, Milan Milovancev, Paul Stromberg, Bruce H. Williams, Marc Aubreville, Giancarlo Avallone, Pompei Bolfa, John Cullen, Michelle M. Dennis, Michael Goldschmidt, Richard Luong, Andrew D. Miller, Margaret A. Miller, John S. Munday, Paola Roccabianca, Elisa N. Salas, F. Yvonne Schulman, Renee Laufer-Amorim, Midori G. Asakawa, Linden Craig, Nick Dervisis, D. Glen Esplin, Jeanne W. George, Marlene Hauck, Yumiko Kagawa, Matti Kiupel, Keith Linder, Kristina Meichner, Laura Marconato, Michelle L. Oblak, Renato L. Santos, R. Mark Simpson, Harold Tvedten and Derick Whitley in Veterinary Pathology
Supplemental Material
Supplemental Material, sj-pdf-9-vet-10.1177_03009858211013712 - International Guidelines for Veterinary Tumor Pathology: A Call to Action
Supplemental Material, sj-pdf-9-vet-10.1177_03009858211013712 for International Guidelines for Veterinary Tumor Pathology: A Call to Action by Donald J. Meuten, Frances M. Moore, Taryn A. Donovan, Christof A. Bertram, Robert Klopfleisch, Robert A. Foster, Rebecca C. Smedley, Michael J. Dark, Milan Milovancev, Paul Stromberg, Bruce H. Williams, Marc Aubreville, Giancarlo Avallone, Pompei Bolfa, John Cullen, Michelle M. Dennis, Michael Goldschmidt, Richard Luong, Andrew D. Miller, Margaret A. Miller, John S. Munday, Paola Roccabianca, Elisa N. Salas, F. Yvonne Schulman, Renee Laufer-Amorim, Midori G. Asakawa, Linden Craig, Nick Dervisis, D. Glen Esplin, Jeanne W. George, Marlene Hauck, Yumiko Kagawa, Matti Kiupel, Keith Linder, Kristina Meichner, Laura Marconato, Michelle L. Oblak, Renato L. Santos, R. Mark Simpson, Harold Tvedten and Derick Whitley in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Christof A. Bertram gratefully acknowledges financial support received from the Dres. Jutta und Georg Bruns-Stiftung für innovative Veterinärmedizin.
ORCID iD
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
