Abstract
A cow dairy (n = 2000) in close proximity to a sheep flock had third-trimester abortions and fatalities in cows and calves over a 14-month period. Eighteen of 33 aborted fetuses (55%) had multifocal random suppurative or mononuclear meningoencephalitis with vasculitis. Seventeen of these affected fetuses had intracytoplasmic bacteria in endothelial cells, and 1 fetus with pericarditis had similar bacteria within mesothelial cells or macrophages. Immunohistochemistry for Chlamydia spp. or polymerase chain reaction (PCR) for Chlamydia pecorum or both, performed on brain or pooled tissue, were positive in all 14 tested fetuses that had meningoencephalitis and in 4/4 calves and in 3/4 tested cows that had meningoencephalitis and thrombotic vasculitis. In 1 calf and 11/11 fetuses, C. pecorum PCR amplicon sequences were 100% homologous to published C. pecorum sequences. Enzootic chlamydiosis due to C. pecorum was the identified cause of the late term abortions and the vasculitis and meningoencephalitis in fetuses, calves, and cows. C. pecorum, an uncommon bovine abortogenic agent, is a differential diagnosis in late-term aborted fetuses with meningoencephalitis, vasculitis, and polyserositis.
Bovine abortions are economically devastating with the average reported producer cost in the United States being $555 per abortion. 11 Specifically, for California dairy herds, it has been estimated that abortion losses cost producers approximately $200 million per year. 18 Reports of abortion rates in the United States range between 2% and 5%; however, one report documents an average abortion rate of 10.8% in 10 Holstein herds in northwestern United States. 15,16,19,28 Though often challenging, identifying an underlying cause of abortion is critical given its economic impact. Within several retrospective reports of bovine abortion diagnostics in the United States, a definitive cause was identified in only 23% to 57% of cases. 2 –4,9,20,29 The challenge of diagnosis may be because distinctive gross or microscopic lesions are inconsistent, are obscured by decomposition, or both, and other important diagnostic samples such as placenta and serum from the dam are often not submitted along with the fetus. In those cases where a definitive diagnosis of abortion was made, infectious causes overwhelmingly predominated. Noninfectious causes of abortion (congenital defects, pregnancy toxemia, twinning, dystocia, etc) on average made up less than 3.3% of cases. 3,9,29 Of the infectious abortogenic agents identified, the most commonly reported were bacteria, followed by viruses, and less commonly, protozoa and sporadic fungi. Commonly reported bacteria include Leptospira, Ureaplasma, Listeria, Salmonella, Brucella, Pajaroellobacter abortibovis (epizootic bovine abortion), Campylobacter, and opportunistic bacteria, like Trueperella pyogenes and Streptococcus spp. 2,41 Chlamydiae, which are the focus of this study, are considered an uncommon cause of late-term bovine abortion. In fact, chlamydiae were not mentioned in most large-scale bovine abortion surveys, 2 –4,9,20,29 although only one of these surveys specifically tested for chlamydiae. 29
Chlamydiae are obligate intracellular bacteria with a capacity to infect a wide range of domestic and exotic animals as well as humans. 5 Within the Chlamydiaceae family, the Chlamydia genus includes 14 species that have broad virulence, ranging from asymptomatic to severe and life-threatening. 5,30,43 In ruminants, chlamydial infections are typically associated with either C. abortus, C. pecorum, or C. psittaci. 40 Such infections are often subclinical as evidenced by a reportedly high worldwide seroprevalence of 58% to 100% in healthy dairy cattle, and high seroprevalence of 94% in healthy sheep flocks in Germany. 12,24,31,40 Nonetheless, subclinical infections in cattle still have the capability to cause considerable morbidity and affected herds may suffer significant economic losses due to reduced weight gain, infertility, and decreased milk production. 23,37,39,48
When clinical disease does occur in ruminants, infections with C. pecorum manifest as a wide spectrum of diseases including polyarthritis, conjunctivitis, encephalomyelitis, infertility, enteritis, mastitis, placentitis, abortion, and pneumonia. 14,17,21,33,45,46,49,50 The spectrum of disease may be influenced by the patient’s age at infection; for example, calves infected with C. pecorum shortly after birth commonly develop encephalomyelitis, fibrinous polyserositis with vasculitis, and bronchointerstitial pneumonia. 21,25,38 To the authors’ knowledge, encephalomyelitis associated with C. pecorum infection in adult cattle has not been described. Additionally, in aborted fetal goats infected with C. pecorum, suppurative placentitis and arteritis as well as fibrinosuppurative enteritis were commonly reported. 17 Experimental intravenous challenge of pregnant ewes with C. pecorum led to placental colonization, but failed to result in any significant gross or microscopic lesions. 36 Meanwhile, bovine abortions due to C. pecorum are uncommonly reported in the literature, with limited descriptions of lesions. 49
Chlamydia spp. have a unique biphasic developmental life cycle and demonstrate a strong tropism for mucosal sites (eg, conjunctiva, genital, and intestinal tract) or placenta and fetus. 12,33,34,36 Subsequent bacteremia and dissemination to viscera can occur. 1,14,22,47 Consistent with the tropism of chlamydiae for mucosal sites, shedding has been reported to be nasal, fecal, conjunctival, and vaginal. Accordingly, fecal-oral transmission or inhalation of infected bodily secretions are thought to be important routes of infection. Specifically for C. pecorum, fecal-oral transmission is the most well-established route of infection. 33,51
This diagnostic investigation summarizes the gross, microscopic, immunohistochemical, and molecular diagnostic findings associated with C. pecorum enzootic infection of multiple ages of cattle in a dairy herd in Arizona. In particular, as prior descriptions of fetal lesions in ruminants other than goats have been rare, this study describes lesions from 18 late-term aborted bovine fetuses. Additionally, this study describes the unique finding of C. pecorum–associated meningoencephalitis and vasculitis in 5 adult cows.
Materials and Methods
The dairy farm in Arizona consisted of a milking herd of 2000 Jersey cows, and replacement heifers of about 15% annually. Cows were fed a total mixed ration that met or exceeded National Research Council guidelines according to their stage of lactation. The farm maintained a semi-closed herd with a portion of heifers shipped at 5 months of age to a farm in Texas and returning 4 weeks before calving. Calves were separated from the dams at birth and were administered 4 L of pasteurized colostrum, half given within 4 hours of birth and the remainder 6 hours later. Calves were subsequently fed fresh milk until weaned at 55 days of age. Prior to receiving colostrum, the calves received an oral rotavirus and coronavirus vaccine (Calf guard, Zoetis). During the first week of life, a bovine rhinotracheitis and parainfluenza 3 vaccine was administered intranasally (TSV-2, Zoetis) and a booster at weaning. At 5 months of age, calves received a brucellosis vaccine (RB51, Colorado Serum Company), multivalent clostridia vaccine (Ultrabac 7, Zoetis), and a multivalent respiratory vaccine (Bovishield Gold FP, Zoetis). Three weeks prior to calving, heifers and cows were vaccinated with a multivalent killed respiratory vaccine (Triangle 10, Boehringer Ingelheim), an Escherichia coli vaccine (Enviracor J-5, Zoetis), and Ultrabac 7. Cows received boosters of Bovishield Gold FP and Enviracor J-5, 2 weeks postcalving. The herd’s abortion rate was unknown but estimated at less than 5% per year.
The same farm had a sheep flock of 2000 ewes and 3000 lambs used for milk and meat production, respectively. Sheep that were lambing were kept on pastures and dry lots separated from the cows. In the dry lots, sheep were fed a total mixed ration. Sheep received 2 doses of the Ultrabac 7 four weeks apart and then annually. Orphaned lambs were housed in the calf barn in groups of 5 to 7 and were fed sheep milk. There was no nose-to-nose contact between lambs and calves, but the same personnel handled both lambs and calves. There was no record keeping on sheep health or ewe abortion rates.
Postmortem Examinations
For 14 months (August 15, 2018, to October 14, 2019), Midwestern University’s College of Veterinary Medicine’s Diagnostic Pathology Center received 54 cows, 32 calves, and 33 bovine fetuses for postmortem examination originating from the dairy herd. Submitted animals were predominately (95%) Jersey, and excluding one Angus, all fetuses were Jersey. Cows and calves were submitted following natural death or euthanasia. Aborted fetuses were in the third trimester and rarely submitted with placenta. Complete postmortem examinations were performed on all animals, including histopathology and a variety of diagnostics (bacteriology, virology, polymerase chain reaction [PCR], and immunohistochemistry [IHC]) to establish the cause. During postmortem examinations, representative samples of all organ systems were collected and fixed in 10% neutral-buffered formalin followed by routine processing and paraffin embedding to yield 4-μm-thick sections that were stained with hematoxylin and eosin for microscopic examination. Samples consistently collected included brain, lung, heart, liver, kidney, adrenal glands, fetal abomasum, all stomach compartments of calves and cows, small and large intestine, and variably included spleen, thymus, thyroid gland, pancreas, uterus, gonads, tongue, bone marrow, skin, skeletal muscle, peripheral nerves, eyelid, salivary gland, eyes, lymph nodes, urinary bladder, mammary gland, spinal cord, and umbilicus. Placenta was available only for one case (case 27).
Bacteriology and Immunohistochemistry for Chlamydia spp
Each sample (abomasal fluid, lung, liver, kidney, spleen, brain, thoracic fluid, abdominal fluid, pleural swab, pericardium, placenta) was processed and streaked onto trypticase soy agar containing 5% sheep blood (TSAB), chocolate agar, columbia nalidixic acid (CNA) containing 5% sheep blood, and MacConkey agar followed by incubation at 37 °C for 18 to 24 hours in the presence of CO2 under aerobic conditions. Each sample was also streaked onto TSAB, brucella agar (supplemented with 5% sheep blood, hemin, and vitamin K1), anaerobic laked blood agar (containing kanamycin and vancomycin), and anaerobic blood agar with phenylethyl alcohol (PEA) and incubated anaerobically at 37 °C for up to 5 days. Resulting colonies were subcultured, checked for purity, and identified using the matrix assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF MS) following established protocol.
IHC for Chlamydia spp. was performed by the California Animal Health and Food Safety (CAHFS) Laboratory at UC Davis using a previously described method 17 using 1:100 dilution of the monoclonal primary antibody to Chlamydia lipopolysaccharide (LPS). Liver from birds diagnosed with chlamydiosis served as positive and negative controls. IHC was performed on formalin-fixed, paraffin-embedded (FFPE) 4-μm-thick sections from 9 animals (3 cows, 1 calf, and 5 fetuses). Brain was tested in 8 animals with additional testing on pleura (case 34), and pericardium, epicardium, liver, and intestine (case 6). One cow (case 41) was tested on lung only.
Molecular Diagnostic Testing for Abortogenic Agents
Fresh tissues (brain, pericardium, lung, liver, and kidney) were tested using PCR by the Wisconsin Veterinary Diagnostic Laboratory (WVDL) for detection of bovine abortogenic agents including bovine viral diarrhea virus (BVDV), bovine herpesvirus-1 (BHV-1), Ureaplasma, Leptospira, Neospora, Listeria, and Chlamydia. When multiple aforementioned fresh tissue types were submitted, up to 4 tissues from each animal were pooled for testing. All assays were set up in multiplex with an in-house long oligos internal control. Formalin-fixed, paraffin-embedded brain tissue from 8 animals (3 cows, 4 calves, 1 fetus) with meningoencephalitis was also submitted for Chlamydia spp. PCR. PCR tests for BVDV, BHV-1, Ureaplasma, Leptospira, and Neospora were performed on individual or pooled fresh tissue samples from 10 fetuses. 7,10,32,44 One fetal brain sample (case 5) was also tested for Listeria by PCR assay. 13 PCR for Chlamydia spp. was performed on fresh tissue from 18 animals (2 cows, 1 calf, 15 fetuses) that included brain only (1 cow, 1 calf, 8 fetuses), brain and pericardium (1 fetus), lung (1 cow), and pooled samples of brain, spleen, lung (1 fetus) or brain, lung, liver, and kidney (2 fetuses) or lung, liver, and kidney (3 fetuses). No fresh brain from cows with meningoencephalitis was available for testing.
For 5 of the samples with a Chlamydia spp. positive result, a repeat Chlamydia spp. PCR assay without the internal control was set up to amplify the 222 base pair (bp) partial 16S rRNA region for nucleic acid sequencing. The 25 µl of PCR master mix consisted of TaqMan Universal PCR Master mix (Applied Biosystems), 0.4 μM of forward primer (5′-TCGAGAATCTTTCGCAATGGAC-3′) and reverse primer (5′-CGCCCTTTACGCCCAATAAA-3′), 0.2 μM of Taqman probe (5′-TAM-AAGTCTGACGAAGCGACGCCGC-BQH2-3′), and 5 µl of extracted nucleic acids. PCR was performed in ABI 7500 Real-time PCR System (Applied Biosystems) for 1 cycle of 50 °C for 10 minutes, 1 cycle of 95 °C for 10 minutes, and 40 cycles of 95 °C for 15 seconds, and 60 °C for 1 minute. Amplicons were cleaned up and sequenced using the forward and reverse primers (Functional Biosciences).
To confirm Chlamydia pecorum, a published primer and probe set 35 specific for a 76 bp outer membrane protein A (ompA) gene segment of C. pecorum was adapted for a real-time PCR assay using the same master mix condition (with substitution of primers and probe) and cycling conditions as described for the 16S rRNA PCR assay.
In an attempt to further confirm the C. pecorum detection, a conventional PCR assay for amplification of a 565 to 582 bp Chlamydia sp. outer membrane protein 2 (omp2) gene segment was performed to obtain PCR amplicons for nucleic acid sequencing. The 5 µl master mix consisted of GoTaq Green Master Mix (Promega), 0.5 μM each of published forward primer (PCR-D1) 42 and reverse primer (PCR-D2 42 or an in-house designed PCR-D3 primer [5′-TACCCAWACRGTAATTTTGC-3′]), and 5 µl of extracted nucleic acids for a final reaction volume of 20 μl. 42 The reaction was subjected to 1 cycle of 95 °C for 5 minutes, and 40 cycles of 95 °C for 30 seconds, 48 °C for 30 seconds, and 60 °C for 1 minute, followed by final extension of 72 °C for 5 minutes. PCR products were visualized in 1.5% agarose gel, amplicons were excise from the gel and cleaned-up using QIAquick Gel Extraction Kit (Qiagen). Amplicons were sent to Functional Biosciences Inc for nucleic acid sequencing using the forward and reverse primers.
Sequencing data were analyzed and assembled using Sequencher program (Gene Codes). Sequence identities were compared to published sequences in the public database using Basic Logic Alignment Search Tool (BLAST) available on the National Center for Biotechnology Information website.
Nucleic acid from FFPE brain tissue from 8 animals (3 cows, 4 calves, 1 fetus) with meningoencephalitis was subjected to 16S rRNA and ompA real-time PCR using methods described above.
Results
Thirty-three fetuses were examined, and the results of the postmortem examination and diagnostic tests are summarized in Table 1 (cases 1–18: fetuses with meningoencephalitis), Supplemental Table S1 (cases 1–18: gross and all microscopic lesions), and Supplemental Table S2 (cases 19–33: fetuses without meningoencephalitis). Most fetuses were autolyzed with soft viscera and variable amounts of serosanguinous fluid in body cavities and subcutaneous tissues. Grossly, one 8-month-old fetus (case 6) had fibrinous perihepatitis and pleuritis (Fig. 1) and fibrinous pericarditis and epicarditis (Fig. 2), and 3 others had nontraumatic leptomeningeal hemorrhage (cases 12, 13, and 17). Microscopically, 18 fetuses (cases 1–18) had multifocal random meningoencephalitis with vasculitis in every section of examined brain for each case, which variably included sections from the telencephalon, diencephalon, metencephalon, and myelencephalon. Microscopic lesions were similar among all fetuses, characterized by suppurative (Fig. 3) to mononuclear meningoencephalitis, vasculitis, and gliosis, along with variable hemorrhage, fibrin thrombi, sheets of fibrin in leptomeninges, focal aggregates of neutrophils, spongy change, neuronal necrosis, and aggregating fibrin in areas of inflammation. Seventeen fetuses (excludes case 3) had cytoplasmic bacteria in the endothelium of thin-walled vessels (Fig. 4) and, rarely, within perivascular histiocytes. Bacteria were consistently immunopositive with IHC for Chlamydia spp. (Fig. 5). Case 6 had similar cytoplasmic bacteria in lesions of fibrinous pericarditis and epicarditis and necrotizing enteritis that were also immunopositive for Chlamydia spp. (Fig. 6).
Summary of the Microscopic Lesions and Results of Diagnostic Tests Performed in 18 Fetuses With Meningoencephalitis.
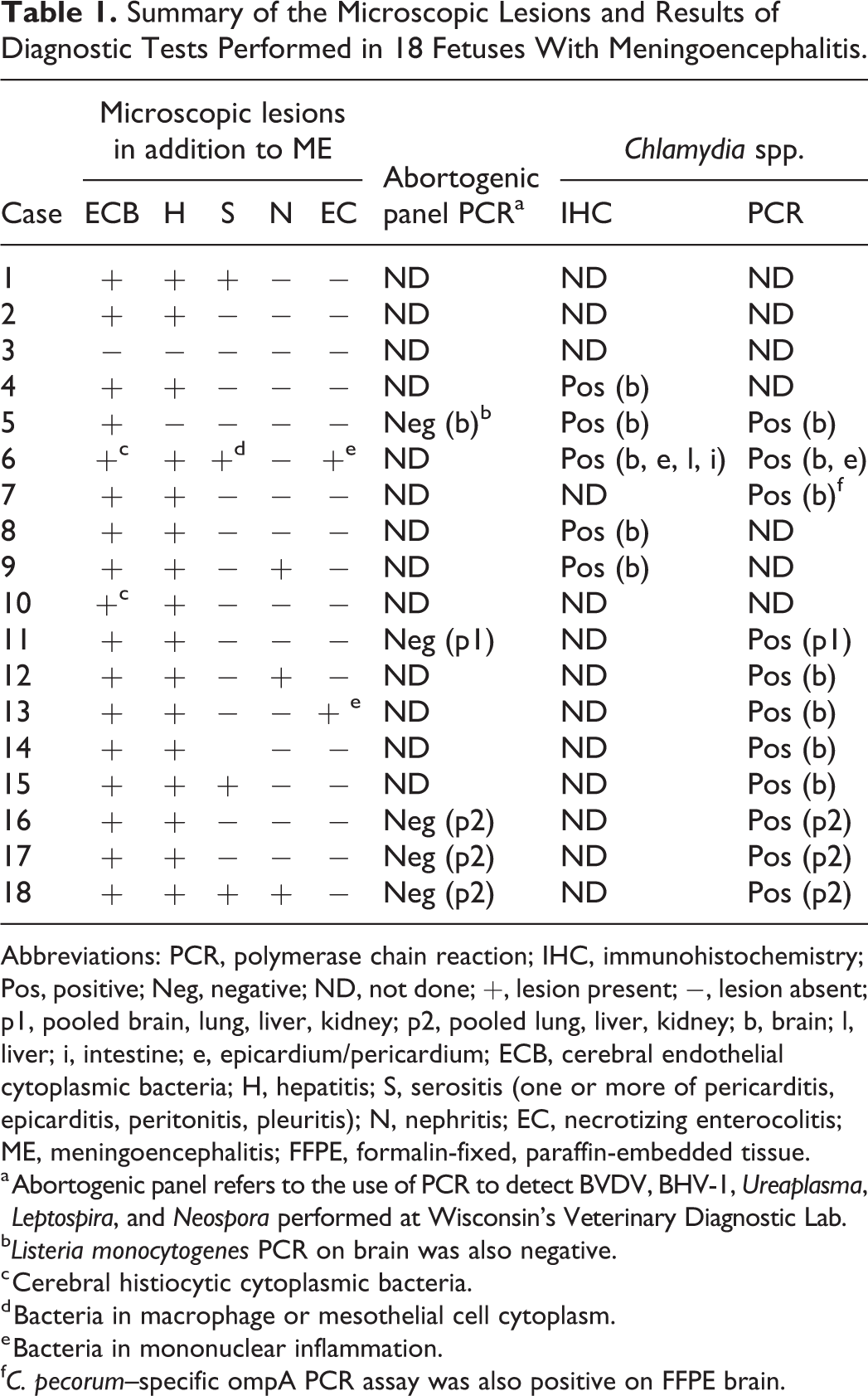
Abbreviations: PCR, polymerase chain reaction; IHC, immunohistochemistry; Pos, positive; Neg, negative; ND, not done; +, lesion present; −, lesion absent; p1, pooled brain, lung, liver, kidney; p2, pooled lung, liver, kidney; b, brain; l, liver; i, intestine; e, epicardium/pericardium; ECB, cerebral endothelial cytoplasmic bacteria; H, hepatitis; S, serositis (one or more of pericarditis, epicarditis, peritonitis, pleuritis); N, nephritis; EC, necrotizing enterocolitis; ME, meningoencephalitis; FFPE, formalin-fixed, paraffin-embedded tissue.
a Abortogenic panel refers to the use of PCR to detect BVDV, BHV-1, Ureaplasma, Leptospira, and Neospora performed at Wisconsin’s Veterinary Diagnostic Lab.
b Listeria monocytogenes PCR on brain was also negative.
c Cerebral histiocytic cytoplasmic bacteria.
d Bacteria in macrophage or mesothelial cell cytoplasm.
e Bacteria in mononuclear inflammation.
f C. pecorum–specific ompA PCR assay was also positive on FFPE brain.
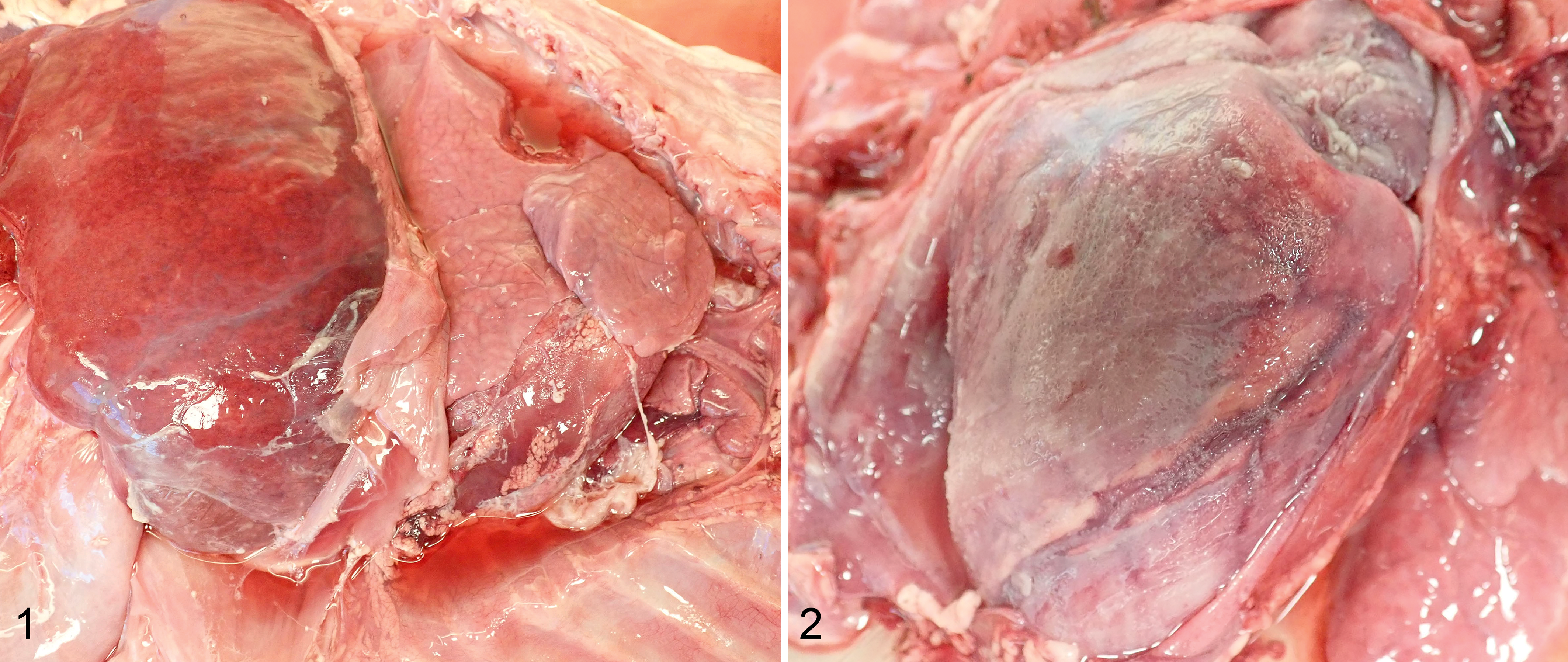
Chlamydia pecorum infection, 8-month-old aborted bovine fetus, case 6.
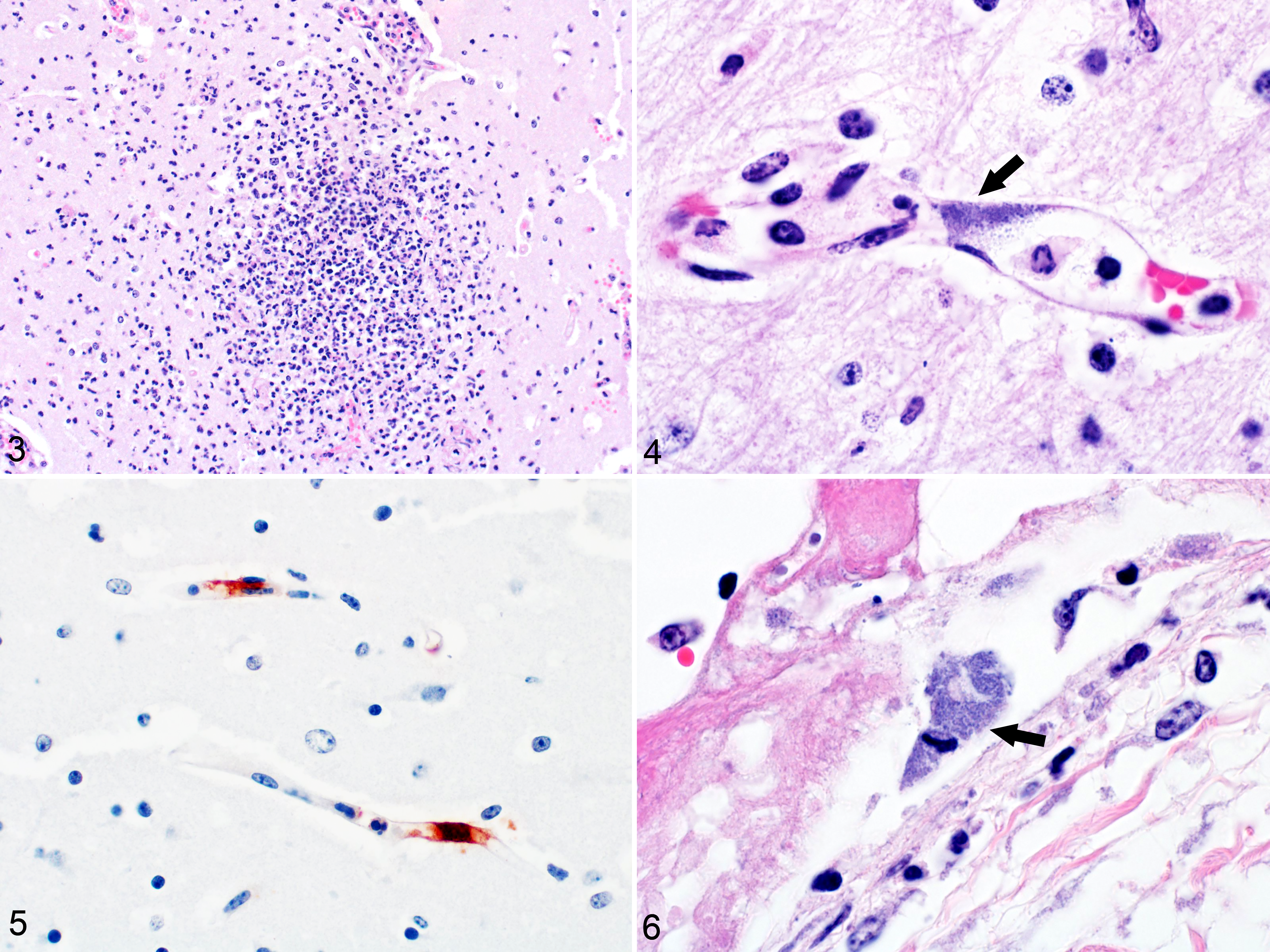
Chlamydia pecorum infection, 7.5- and 8-month-old aborted bovine fetuses, cases 5 and 6.
Eleven of 11 tested fetuses with meningoencephalitis were PCR-positive for Chlamydia spp., 5 of 5 tested were IHC-positive for Chlamydia spp., and none of the 11 tested had significant growth on bacteriology. Overall, 14 of the 18 fetuses with meningoencephalitis were positive for Chlamydia spp. by PCR or IHC or both. Because all 18 fetuses had similar lesions of meningoencephalitis and vasculitis, all were diagnosed with Chlamydia spp. infection.
Four of the 11 16S rRNA gene amplicons from positive fetuses were sequenced, and the resulting 222 bp sequence was 100% homologous to the published C. pecorum 16S rRNA sequence, but with only 1 mismatched base difference from published C. abortus and C. psittaci sequences (Table 2). Because of the relatedness of the 3 Chlamydia spp., a C. pecorum ompA-specific real-time PCR was used to confirm that all 11 tested fetuses were positive for C. pecorum by the specific PCR assay. Subsequently, a conventional PCR targeting the partial omp2 gene segment was used to generate amplicons from all 11 fetuses for nucleic acid sequencing. The 531 bp sequence of omp2 gene fragment from all 11 fetal samples was identical; and was 100% homologous to several published C. pecorum sequences (GenBank ID: CP002608, CP004034, and CP004035).
Results of Chlamydia Spp. 16S RNA, ompA, and omp2 PCR and Amplicon Sequencing on Fresh Tissue in 11 Fetuses (Cases 5–7, 11–18) and 1 calf (Case 43) Affected With Meningoencephalitis and Vasculitis.
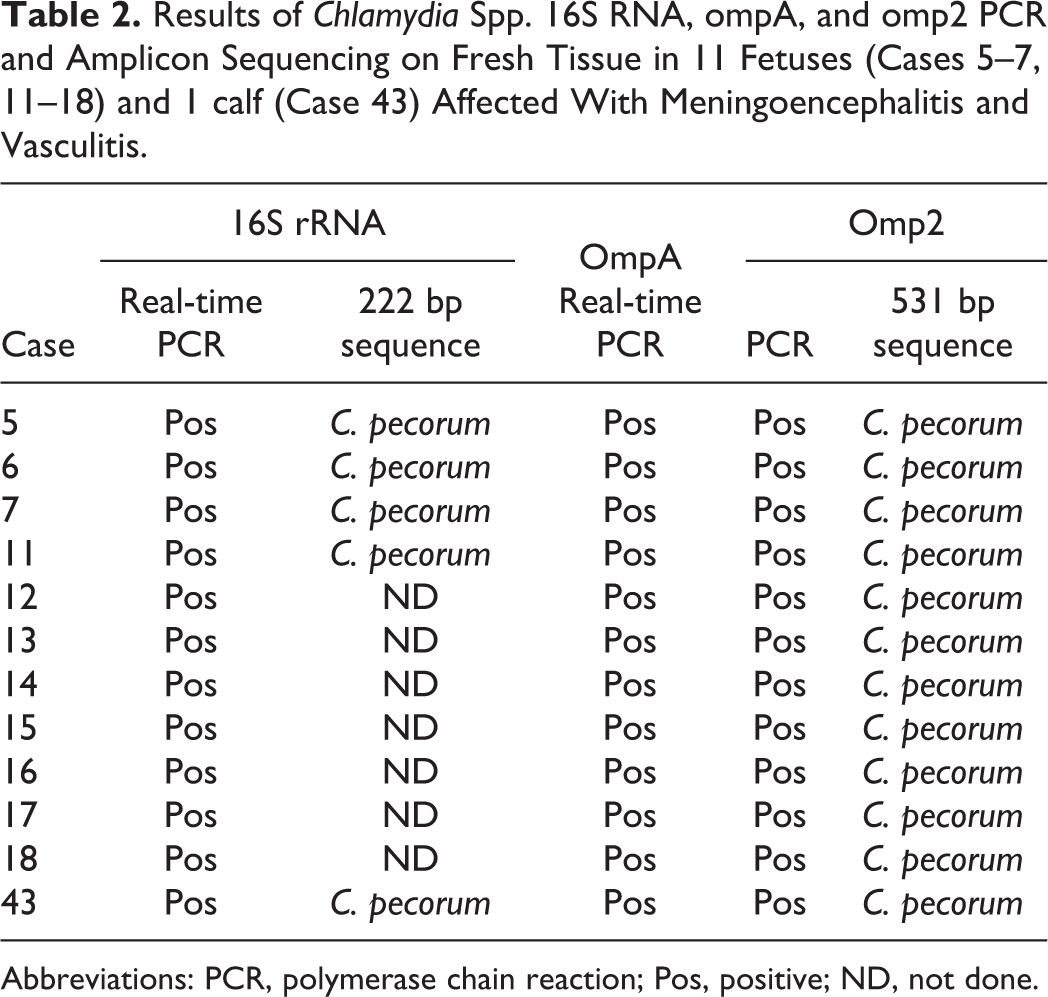
Abbreviations: PCR, polymerase chain reaction; Pos, positive; ND, not done.
Fifty-four examined cows suffered from infectious and noninfectious maladies that caused death or warranted euthanasia. Common diagnoses were mastitis, pneumonia, gastroenteritis, obstructive bloat, and traumatic reticuloperitonitis. The results of diagnostic testing of 8 cows are summarized in Table 3 and gross and microscopic lesions are in Supplemental Table S3. Five cows (cases 34–38) had similar brain lesions to those described for the aborted fetuses, in addition to occurring in sections of mesencephalon (Figs. 7, 8). IHC for Chlamydia spp. in a cow with encephalitis (case 36) and one with encephalitis and pleuritis (case 34) identified intralesional Chlamydia spp. (Fig. 9). FFPE brain tissue from case 38 tested negative by the 16S rRNA assay, but tested positive by a multiplex assay utilizing the C. pecorum-specific ompA PCR assay.
Summary of the Results of Diagnostic Tests Performed in 5 Cows With Meningoencephalitis (Cases 34–38), 3 Cows Without Meningoencephalitis (Cases 39–41), and 4 Calves With Meningoencephalitis (Cases 42–45).
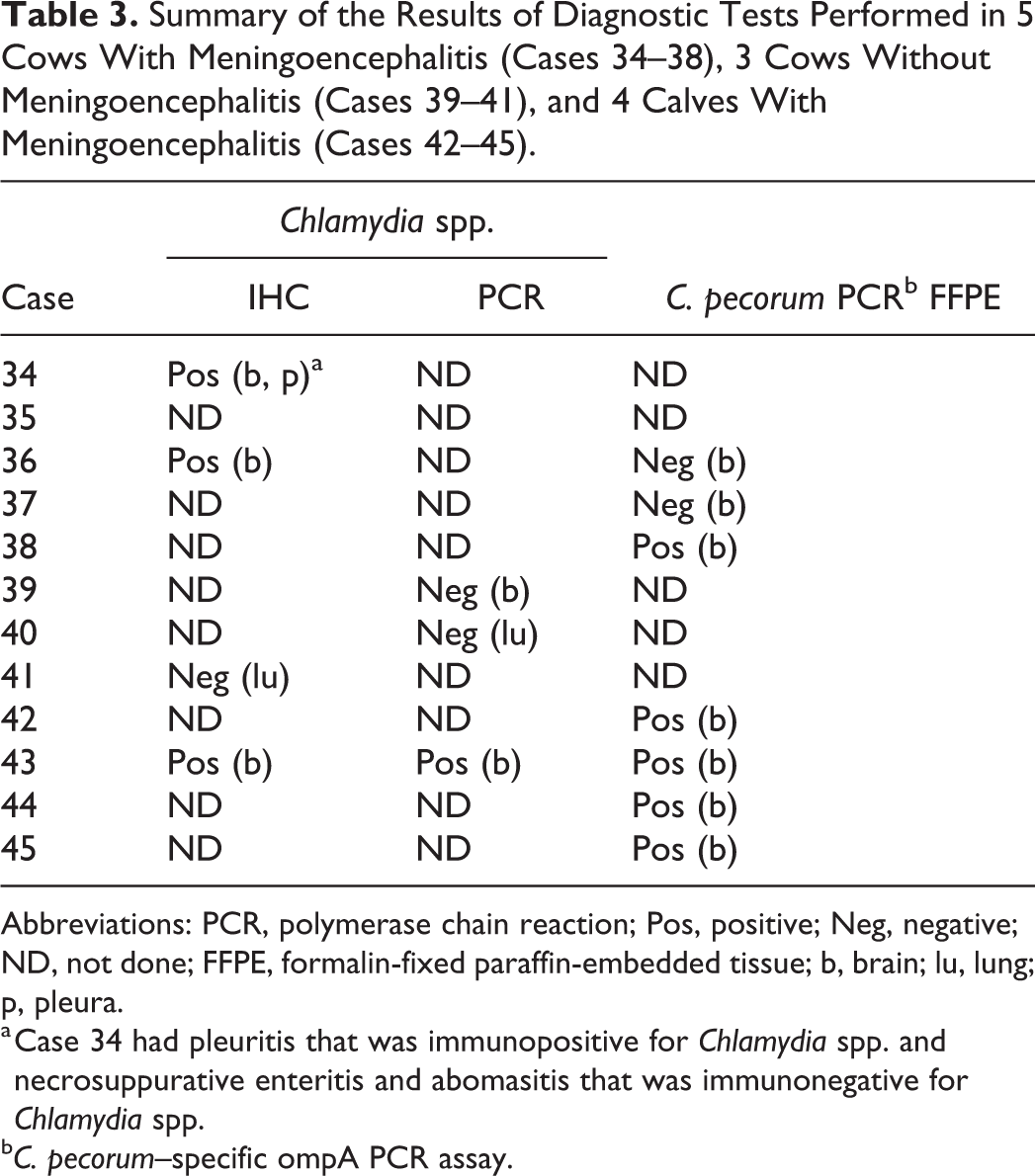
Abbreviations: PCR, polymerase chain reaction; Pos, positive; Neg, negative; ND, not done; FFPE, formalin-fixed paraffin-embedded tissue; b, brain; lu, lung; p, pleura.
a Case 34 had pleuritis that was immunopositive for Chlamydia spp. and necrosuppurative enteritis and abomasitis that was immunonegative for Chlamydia spp.
b C. pecorum–specific ompA PCR assay.
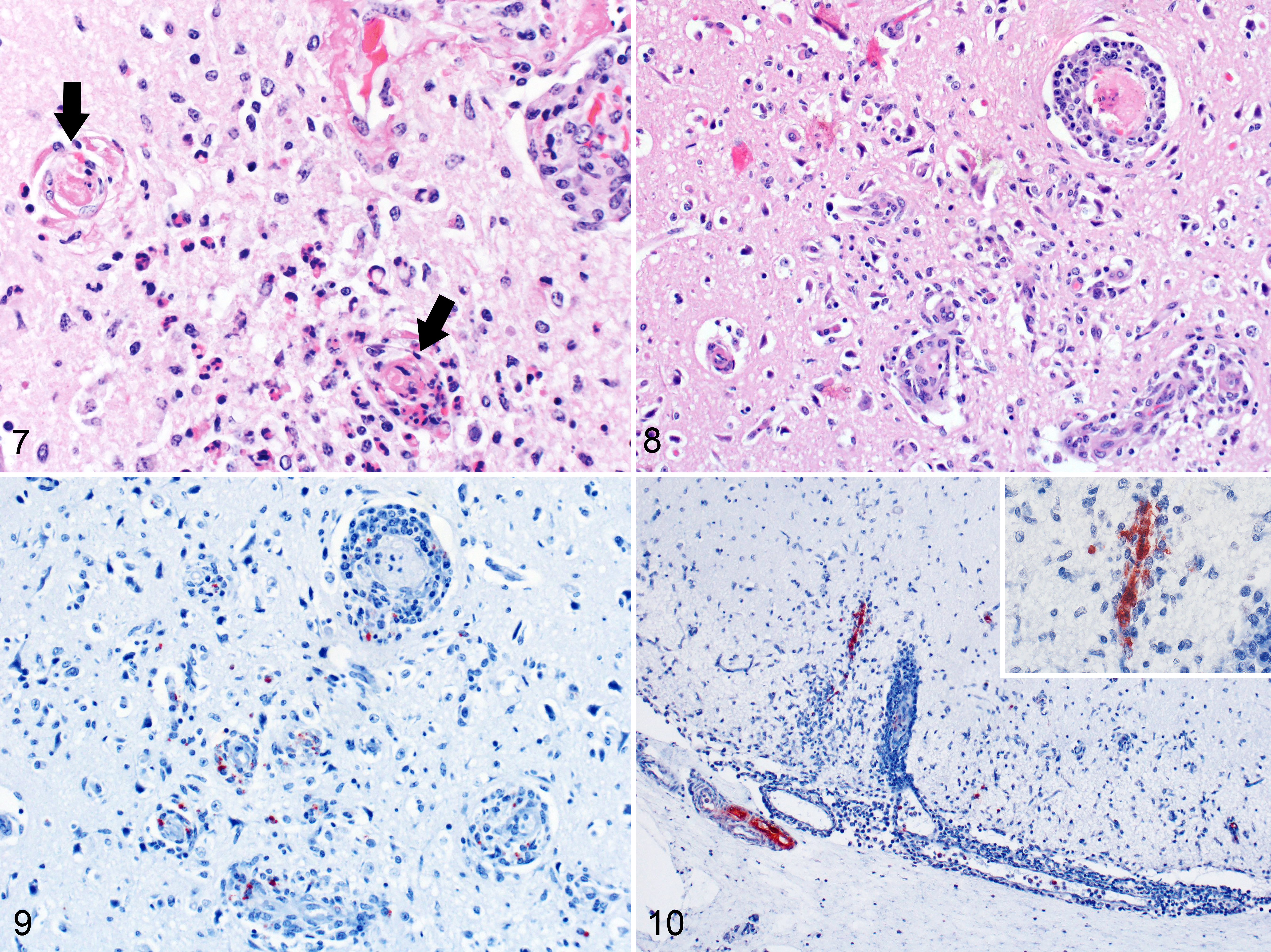
Chlamydia pecorum infection.
Thirty-two examined calves suffered from a variety of infectious maladies that caused death or warranted euthanasia. Common diagnoses were pneumonia, otitis media, and gastroenterocolitis. Mycoplasma bovis was a frequent pathogen. The results of diagnostic tests of 4 calves are summarized in Table 3 and gross and microscopic lesions are in Supplemental Table S3. Four calves (cases 42–45) had similar brain lesions to those described for the aborted fetuses, in addition to occurring in sections of mesencephalon. Virus isolation performed at NVSL in Ames, Iowa, on the brain from 2 calves with encephalitis yielded no growth. Fresh brain from case 43 was PCR-positive for Chlamydia spp., and immunopositive for Chlamydia spp. (Fig. 10).
Fresh brain from case 43 was further evaluated with PCR and sequencing using an identical approach as described for the fetus samples. The calf’s 16S rRNA and the omp2 gene sequences were identical to those of the fetuses. The sample was also positive for C. pecorum–specific ompA real-time PCR assay (Table 2). FFPE brain tissue from all calves tested positive by a multiplex assay utilizing the C. pecorum–specific ompA PCR assay.
Discussion
This report describes enzootic chlamydiosis in a dairy herd. Over a 14-month period, 5 cows, 4 calves, and 18 fetuses were diagnosed with Chlamydia pecorum infection based on similar lesions of meningoencephalitis with vasculitis, and the results of IHC for Chlamydia spp. and C. pecorum PCR and amplicon sequencing. Confirmed infections were those animals with meningoencephalitis that were PCR-positive for C. pecorum on fresh tissue or FFPE brain tissue, which included 11 fetuses, 1 cow, and 4 calves (Table 4). The amplicon sequences in 11 fetuses and in the only tested calf were identical to published C. pecorum sequences for 16S rRNA (4/4 fetuses) and omp2 (11/11 fetuses). A highly likely C. pecorum infection included 3 fetuses and 2 cows with positive IHC for Chlamydia spp. but without PCR or with a negative PCR on FFPE brain (case 36). The remaining animals (4 fetuses and 2 cows) were suspected to be infected by C. pecorum based on analogous lesions of meningoencephalitis with vasculitis—3 of the 4 fetuses did have intralesional bacteria in the cytoplasm of endothelial cells.
Summary of the Number of Fetuses, Calves, and Cows With Suspected, Highly Likely, and Confirmed C. Pecorum Infection According to Histopathology and the Results of Diagnostic Tests (IHC and PCR).
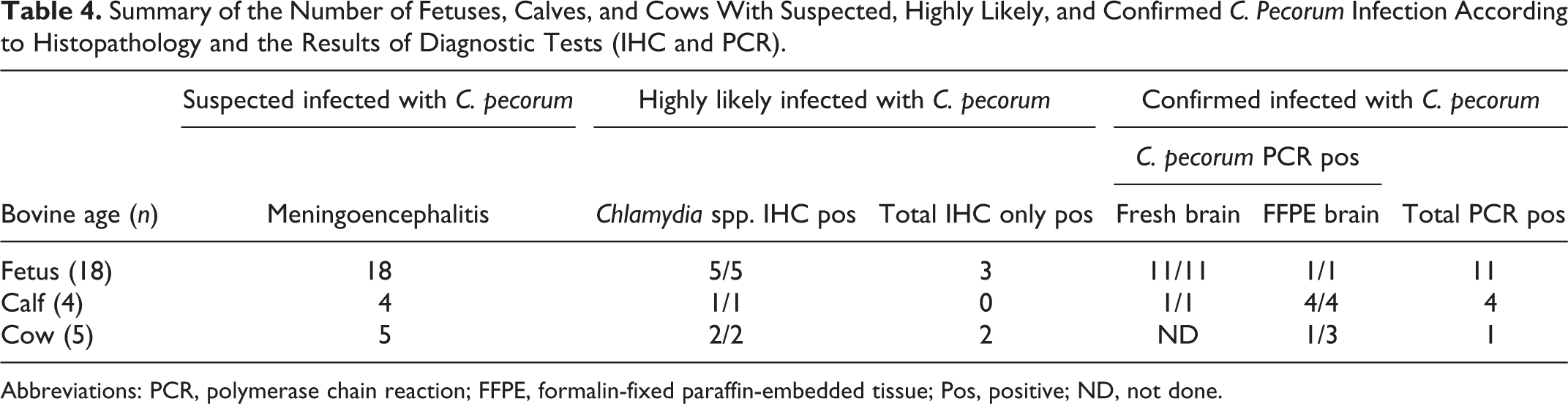
Abbreviations: PCR, polymerase chain reaction; FFPE, formalin-fixed paraffin-embedded tissue; Pos, positive; ND, not done.
Therefore, 18 of 33 fetuses (55%) received for postmortem examination were considered to have aborted due to C. pecorum infection, and bacteriology, PCR, and histopathology excluded other common bovine abortogenic etiologies. Although infected fetuses consistently had meningoencephalitis, the PCR, IHC, and histopathologic findings suggested infections were possibly systemic in some. Case 6 was C. pecorum PCR-positive on brain and epicardium/pericardium, and was positive by IHC for Chlamydia spp. on brain, pericardium, epicardium, intestinal mucosa, and liver. Histologically, it had hepatitis, pericarditis, epicarditis, and enterocolitis. Similarly, case 13 also had enterocolitis and case 14 had necrotizing hepatitis, though IHC for Chlamydia spp. was not performed. The histological lesions and the LPS immunohistochemistry results in the brains of these fetuses resembled what is reported in calves infected by C. pecorum, which described mononuclear perivasculitis, thrombi, neuroparenchymal neutrophil aggregates, malacia, basophilic intracytoplasmic bacteria in endothelium or mononuclear cells, and immunolabeling in endothelium and mononuclear inflammation. 25 Similarly, C. pecorum infections of calves were systemic and caused meningoencephalitis, interstitial pneumonia, fibrinous polyserositis, and epicarditis. 25,49 In ruminant fetuses, the outcome of an infection by C. pecorum may vary among species. In cattle, fetal lesions are not described despite mention of C. pecorum–caused abortion. 49 In small ruminants, fetal lesions associated with C. pecorum abortions are rarely described. 17 Kids aborted from C. pecorum infections developed mononuclear and suppurative hepatitis and fibrinosuppurative necrotizing enteritis. 17 Meanwhile, pregnant ewes experimentally infected with C. pecorum had placental colonization and infrequent abortion without mention of fetal lesions. 36 This study is the first to show that C. pecorum causes meningoencephalitis in aborted bovine fetuses.
Bovine abortions result from infectious and noninfectious causes, and can follow disease of the dam, the placenta, or the fetus. When an abortogenic cause is determined, it is often an infectious etiology. 2,41 Cattle can suffer chlamydial abortions, but this is an infrequent and sporadic cause of late-term abortions, 2,5 so uncommon that Chlamydia spp. are not included in some reviews of major abortogenic agents of cattle, and are not typically part of routine testing at diagnostic laboratories. 2 There are reports of bovine abortion caused by Parachlamydia spp., where Chlamydia-like organisms caused placental lesions. 6 Meanwhile, in small ruminants, C. abortus and to a lesser extent C. pecorum are important abortogenic agents. 5,17 Identification of bovine fetal encephalitis is likely to prompt testing for BHV-1, Neospora, and septicemic bacteria. This report broadens the potential infectious causes of bovine abortions to include C. pecorum, especially when fetuses have meningoencephalitis with vasculitis.
In this report, cases of bovine fetal abortion and cow and calf mortality were directly associated with C. pecorum infection. In cows and calves, meningoencephalitis was diagnosed as the cause of death or the reason for euthanasia. In 2 cows, chlamydial encephalitis was considered unrelated to other comorbidities like pneumonia, mastitis, and gastroenteritis. In 7 cases (3 cows, 4 calves), animals had clinical signs possibly related to chlamydial encephalitis, including recumbency in cases 36 and 37, bloat of unknown cause in case 38, and acute onset of convulsions and opisthotonos in cases 42 to 45. Initially, meningoencephalitis with vasculitis and thrombosis detected in calves and cows was attributed to Histophilus somni thrombotic meningoencephalitis, but bacteriology was consistently negative for H. somni, and lesions lacked coccobacilli. In the calves, the presence of meningoencephalitis, epicarditis, and pneumonia with positive Chlamydia spp. results favored a diagnosis of sporadic bovine encephalomyelitis, a disease of calves less than 6 months of age. 8,25,49 Although well known in calves, C. pecorum meningoencephalitis and vasculitis of adult cattle is not reported. Moreover, in at least one cow (case 34) the lesions of pleuritis were also attributed to C. pecorum infection following positive anti-Chlamydia spp. IHC.
While the study focused on C. pecorum as a cause of meningoencephalitis and abortion, because IHC and PCR for Chlamydia spp. was not performed on all diseased tissues from all cases, it is possible that animals without encephalitis were also infected with C. pecorum. Yet individual or pooled tissues (lung, brain, kidney, spleen, liver) from 2 cows and 4 fetuses without meningoencephalitis were tested for Chlamydia spp. by PCR and were negative.
C. pecorum is an important pathogen of ruminants that causes a range of diseases but can also be subclinical. 49 The bacterium inhabits the gastrointestinal and reproductive tract of ruminants, and organisms may accumulate in the environment through fecal shedding, predisposing to a fecal-oral route of transmission. 49 On-farm epidemiology studies have recognized multiple circulating strains of C. pecorum, which may not have similar tissue tropisms or be equally pathogenic. 25 –27,52
The origin of C. pecorum in this herd is unknown. The introduction may involve shedding from newly acquired cattle or from sheep flocks in close proximity.
The disease investigation of this dairy herd diagnosed meningoencephalitis in fetuses, calves, and cows. Diagnosis of Chlamydia spp. in this study was achieved with PCR and IHC. The PCR amplicon sequencing of partial 16S rRNA and the omp2 gene allowed a diagnosis of C. pecorum infection. Chlamydia pecorum should be considered as an infectious cause of bovine abortions, especially for fetuses aborted in the third trimester and having neutrophilic to mononuclear meningoencephalitis and vasculitis.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_0300985820985288 - Meningoencephalitis, Vasculitis, and Abortions Caused by Chlamydia pecorum in a Herd of Cattle
Supplemental Material, sj-pdf-1-vet-10.1177_0300985820985288 for Meningoencephalitis, Vasculitis, and Abortions Caused by Chlamydia pecorum in a Herd of Cattle by Jason D. Struthers, Ailam Lim, Sylvia Ferguson, Jung Keun Lee, Clemence Chako, Ogi Okwumabua, Matthew Cuneo, Anabell Montiel-Del Valle and Alexandra Brower in Veterinary Pathology
Footnotes
Acknowledgements
We thank the managers, necropsy technicians, and histotechnicans at Midwestern University’s Diagnostic Pathology Center and the pathologists and microbiologists at Wisconsin Veterinary Diagnostic Laboratory for their collaboration and support of this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
