Abstract
Protease-activated receptor-2 (PAR2) is a G protein–coupled receptor that is activated by serine proteases. In humans, PAR2 is highly expressed in various cancers, including breast cancer, and is associated with cancer progression and metastasis. However, the expression and roles of PAR2 in canine mammary carcinoma remain unclear. The purpose of this study was to examine the expression of PAR2 in canine mammary carcinoma, the association between PAR2 expression and clinical characteristics, and the role of PAR2 in the metastatic phenotypes of tumor cells. Mammary carcinoma from 31 dogs and 10 normal mammary glands were included in this study, and used for immunohistochemical analysis of PAR2 expression. Normal mammary glands did not express PAR2. In contrast, mammary carcinomas showed PAR2 immunoreactivity in the cytoplasm, and its expression level varied between specimens from negative to strongly positive. The overall survival of dogs with high PAR2 expression was shorter than that of dogs with low PAR2 expression. Moreover, PAR2 expression level was associated with the presence of lymph node involvement, advanced clinical stage, and high histopathological grade. In vitro analyses revealed that a PAR2 agonist accelerated cell migration and invasion in a canine mammary carcinoma cell line. In addition, the PAR2 agonist induced epithelial-mesenchymal transition and actin polymerization. These results suggest that PAR2 expression plays a role in tumor progression and clinical outcomes in canine mammary carcinoma.
Mammary tumors are the most common neoplasms in intact female dogs, accounting for 25% to 50% of all tumors. Approximately 50% of the cases are histologically diagnosed as malignant, and more than 80% of the malignant tumors are carcinomas. 29,32 Canine mammary carcinoma (MC) can be cured by surgery at the time of early detection, but in clinical stages IV and V cases with metastases to lymph nodes or distant sites, the median survival after surgery is about 6 months, and there is no effective treatment. 4 Therefore, the identification of diagnostic, prognostic, and therapeutic molecules is an urgent need.
Epithelial-derived tumor cells have low motility because they are firmly bound to each other. In order to metastasize, tumor cells acquire motility by inducing epithelial-mesenchymal transition (EMT), wherein epithelial cells lose their epithelial traits such as E-cadherin expression and acquire mesenchymal traits such as N-cadherin, snail family transcriptional repressor 1 (SNAI1), vimentin, and α-smooth muscle actin (α-SMA) expression. Tumor cells with reduced cell adhesion caused by EMT acquire motility and metastasize from the primary lesion. 23,33 Transforming growth factor-β (TGF-β) is an important cytokine that induces EMT, 40 and the expression of TGF-β in canine MC cell lines has been reported to be involved in the induction of EMT and metastasis. 14 During invasion and metastasis, tumor cells migrate into the extracellular matrix of the stroma and invade blood vessels and subsequently other organs. Actin polymerization plays an important role in tumor cell migration in the extracellular matrix. G-actin, a monomer, is polymerized to form filamentous F-actin, which forms cellular protrusions on the periphery of tumor cells. 5,17,38
Proteases function not only as enzymes that hydrolyze peptide bonds linking amino acids but also as signaling molecules through the activation of protease-activated receptors (PARs). 16 PARs belong to a subfamily of G protein-coupled receptors that are activated by a unique mechanism of self-activation following cleavage of their extracellular amino termini by proteases. To date, 4 PAR members have been identified: PAR1, PAR3, and PAR4, which are activated by thrombin, and PAR2, which is activated by serine proteases, including trypsin, mast cell tryptase, coagulation factors VIIa and Xa, and tissue factor. 1 PAR2 is known to be activated by serine protease-mediated cleavage of its N terminus. PAR2 is widely distributed throughout the mammalian body. Its biological actions include regulation of immune responses, inflammation, angiogenesis, cell growth, and development. 10,13,21,27,34,36,37 In humans, several studies have reported that PAR2 is expressed in various cancers, such as gastric, pancreatic, and breast cancers, and is associated with their progression and metastasis. 2,20,28 Thus, PAR2 is thought to be involved in cancer pathology. In dogs, data on the physiological and pathological roles of PAR2 are limited. Our previous studies showed that PAR2 activation induced the expression of pro-inflammatory mediators in canine keratinocytes and intestinal epithelial cells. 18,19 However, the expression and role of PAR2 in canine tumors remain unclear. Therefore, the purpose of this study was to examine the association of PAR2 expression in canine MC with clinical characteristics and tumor-associated outcomes. In addition, we investigated the role of PAR2 in tumor cell migration and invasion.
Materials and Methods
Study Population and Samples
Archival formalin-fixed, paraffin-embedded MC tissues were retrospectively obtained from 31 dogs (Supplemental Table S1). All tumors were surgically removed at the Veterinary Medical Center of the University of Tokyo, between December 2009 and December 2015, and diagnosed on the basis of histopathological analysis. In this study, the histopathological diagnosis was based on the 1999 World Health Organization classification. 22 The histological grade was based on a 2013 report by Peña et al. 25 We obtained written informed consent from all dog owners that the removed tumor tissues could be used for research. The samples included 26 simple carcinomas (84%) and 5 complex carcinomas (16%). Eight of 31 cases were grade 1 (26%), 10 cases were grade 2 (32%), and 13 cases were grade 3 (42%). The dogs ranged in age from 5 to 16 years (median, 11 years). All cases were female, and 14 of 31 had been spayed prior to detection of the MC. No dogs were spayed at the time of surgery for the primary MC. The tumor, lymph node, metastasis status, clinical stage, postoperative medication, and survival data (alive, deceased, or lost) were determined by reviewing the medical records and interviews with referring veterinarians. Postoperative medication, including cyclophosphamide, doxorubicin, carboplatin, lomustine, lapatinib, toceranib, piroxicam, firocoxib, and prednisolone, was administered in 18 dogs (Supplemental Table S1). Overall survival (OS) was defined as the time from surgery until the established cause of death (range: December 18, 2009, to April 10, 2017). Disease-free survival (DFS) was defined as the time from surgery until tumor progression and recurrence (range: December 18, 2009, to April 10, 2017). Normal mammary gland tissues obtained from 10 clinically healthy dogs that were necropsied for other experiments were used as controls. All procedures in this study were conducted in accordance with the guidelines of the Animal Care Committee of the University of Tokyo (Approval Number: P17-108).
Immunohistochemistry
Immunohistochemical analysis of PAR2 was conducted on 4-μm-thick paraffin-embedded sections of the mammary gland tissues, as described previously. 18 Briefly, the sections were deparaffinized in xylene and hydrated in graded ethanol. Antigen retrieval was performed by autoclaving at 121 °C for 10 minutes in 10 mM sodium citrate buffer (pH 6.0). Endogenous peroxidase activity was blocked via incubation with REAL Peroxidase-Blocking Solution (Dako) at room temperature for 10 minutes. After 3 washes in Tris-buffered saline containing 0.1% Triton X-100, the sections were treated with 5% skim milk (Wako) to reduce nonspecific reactions. The sections were then incubated with mouse anti-PAR2 monoclonal antibody (diluted to 1:50; clone SAM-11; Santa Cruz Biotechnology) at 4 °C overnight. The cross-reactivity of this antibody with canine PAR2 was confirmed in a previous study. 18 The sections were subsequently incubated with EnVision polymer reagent for mice (Dako) at room temperature for 45 minutes. The reaction products were visualized using 3,3′-diaminobenzidine (Dako). Canine small intestine, which has been shown to express PAR2, 18 was used as a positive control. Negative controls were performed by omitting the primary antibody. Grading of PAR2 expression was based on a method previously reported by Su et al. 27 A total of 1000 tumor cells were randomly observed on each slide and graded based on the percentage of PAR2-positive cells and their labeling intensity: score 0, no reactivity; score 1+, weak immunoreactivity; score 2+, moderate PAR2 expression or less than 50% of cells with high PAR2 expression; and score 3+, high PAR2 expression in 50% and more of the cells (Figs. 1–5). Samples with scores 0 and 1+ were classified into the low PAR2 group, and those with scores 2+ and 3+ were classified into the high PAR2 group.
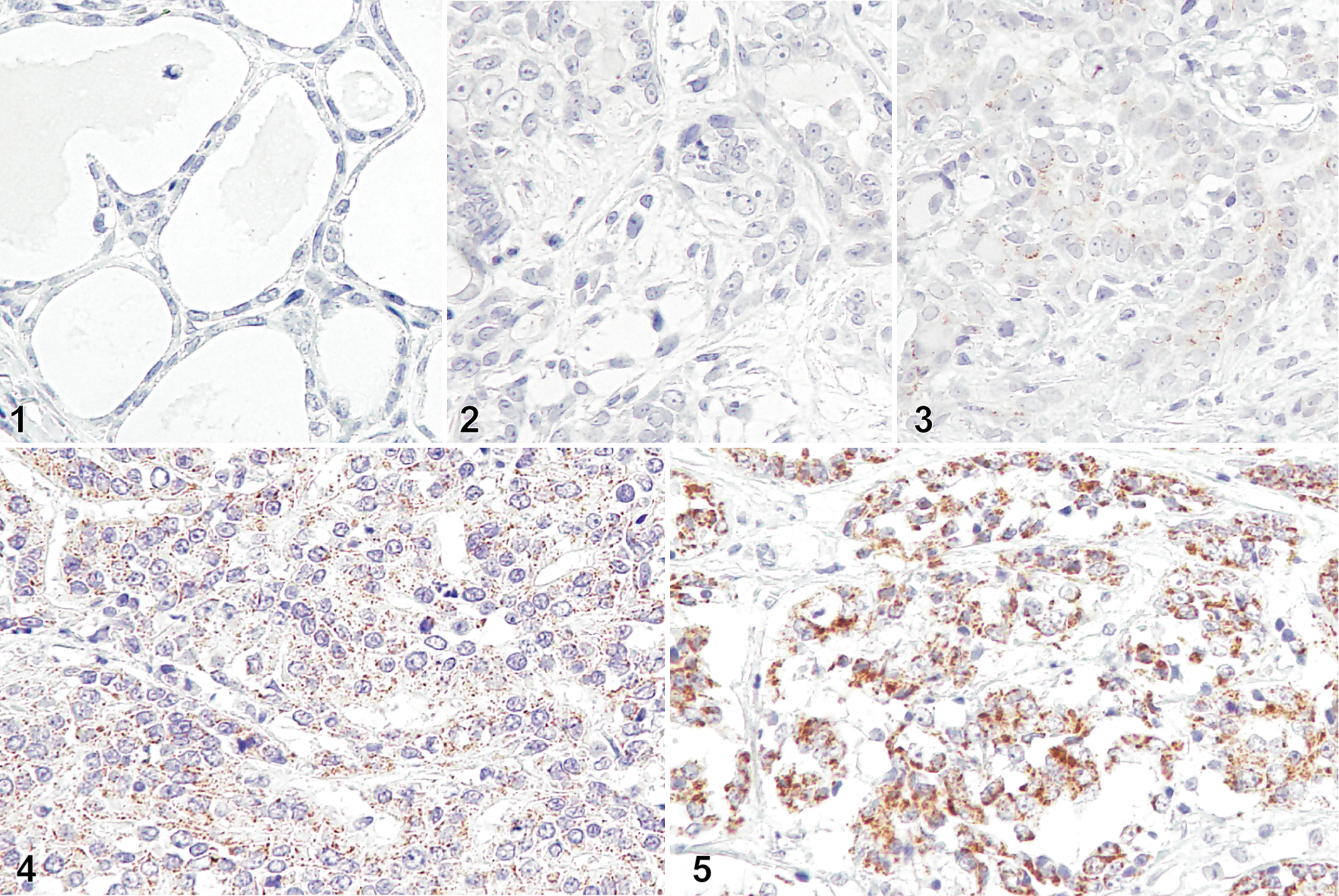
Mammary carcinoma (MC) and normal mammary gland, dogs. Immunohistochemistry for protease-activated receptor-2 (PAR2) counterstained with Mayer’s hematoxylin.
Cell Culture and PAR2 Expression Analysis
We used the CIPp primary canine MC tumor cell line to investigate the effect of PAR2 on primary tumor cells. 35 Cells were cultured in RPMI containing 10% fetal bovine serum (FBS).
Total RNA was extracted from CIPp cells by using TRI Reagent (Molecular Research Center). ReverTra Ace (Toyobo) was used to prepare cDNA from total RNA, according to the manufacturer’s instructions. Amplification of cDNA was performed using TaKaRa Ex-Taq Polymerase (Takara). PAR2 mRNA was detected using the following primer pair: forward 5′-CAGCGTGCAAGGAACCAATAGAA-3′, and reverse 5′-TCCAGTGAGGATGGAGGAAGAGA-3′.
Calcium Imaging
CIPp cells were washed twice with HEPES buffer containing 135 mM NaCl, 5 mM KCl, 10 mM glucose, 2 mM CaCl2, 1.2 mM MgCl2, and 10 mM HEPES (pH 7.4 with NaOH), and loaded with 3 μM Fluo 3-acetoxymethyl ester (Dojindo), and 0.1% Cremophor EL (Nacalai Tesque) in HEPES buffer for 30 minutes at room temperature in the dark. After loading, fluorescence levels (excitation, 480 nm; emission, 515–565 nm) were monitored continuously using a fluorescence imaging system (Hamamatsu Photonics KK). Images were acquired every 1 second at 37 °C. Cells were treated with 100 μM human PAR2 agonist (SLIGKV-NH2; Abcam) or vehicle. The amount of PAR-2 agonist added was based on previous report. 38 The fluorescence intensity (F) was normalized by the fluorescence intensity at the start of the measurement (F0). The experiments were performed in triplicate, and the obtained fluorescence intensity was averaged.
Scratch Wound Healing Assay
CIPp cells were seeded on 24-well plates and grown to 100% confluence in RPMI containing 10% FBS. A wound was made using a pipette tip. Plates were washed with phosphate-buffered saline (PBS) to remove detached cells, and then cultured in RPMI containing 0.5% FBS with 100 μM PAR2 agonist or vehicle for 6 hours. The wound gap was measured at 0 hours and 6 hours after the treatment, and the ratio (%) of wound gap at 6 hours to 0 hours was calculated and presented as the migration ability of cells. The experiments were performed in triplicate.
Transwell Invasion Assay
Twenty hours before the transwell assay, the culture medium of CIPp cells was changed to RPMI containing 0.5% FBS to synchronize the cell cycle. Cells were detached using PBS with 0.5 mM ethylenediaminetetraacetic acid and then washed with FBS-free RPMI. The cells were then suspended in RPMI containing 0.1% bovine serum albumin. The suspension containing 1 × 105 cells was added to the cell culture insert with an 8-µm pore (Corning) coated with diluted Matrigel (Corning). The chamber was placed on 24-well plates filled with RPMI containing 10% FBS. Human PAR2 agonist or vehicle was added to the upper chamber at a concentration of 100 μM. Eight hours after incubation, the membrane was fixed with methanol and stained using Giemsa solution. After removing the cells on the upper side of the membrane with cotton swabs, the invaded cells in 4 randomly selected fields were counted under a microscope (OPTIPHOT-2; Nikon). The experiments were performed in triplicate.
Reverse Transcriptase—Quantitative Polymerase Chain Reaction (PCR)
cDNA was prepared using the method mentioned above. Amplification of cDNA was performed using THUNDERBIRD SYBR qPCR Mix (Toyobo) and an AriaMx Real-Time PCR System (Agilent Technologies). Primers used in the experiment are shown in Supplemental Table S2. The expression levels of each gene were calculated using the ΔΔCt method,
15
and
Western Blotting
CIPp cells were seeded on 24-well plates and grown to 80% confluence in RPMI containing 10% FBS. The cells were treated with 100 μM PAR2 agonist or vehicle for 18 hours. After removing the culture medium, the cells were washed twice with PBS. Protein was extracted from cells using a sample buffer containing 4% sodium dodecyl sulfate, 65 mM Tris-HCl (pH 6.5), 100 mM dithiothreitol, 10% glycerol, and 0.01% bromophenol blue, and the extract was boiled at 100 °C for 5 minutes. Protein extracts were electrophoresed (sodium dodecyl sulfate-polyacrylamide gel electrophoresis) and blotted onto a polyvinylidene difluoride membrane. After blocking with 2% bovine serum albumin, the membrane was incubated with mouse anti-PAR2 monoclonal antibody (diluted to 1:500; clone SAM-11; Santa Cruz Biotechnology), mouse anti-E-cadherin (1:1000; BD Biosciences), rabbit anti-N-cadherin (1:1000; Novus Biologicals), mouse anti-SNAI1 (1:1000; Novus Biologicals), goat anti-vimentin (1:500; Sigma-Aldrich), mouse anti-α-SMA (Dako), and mouse anti-β-actin (1:1000; Sigma-Aldrich) antibodies overnight at 4 °C. After washing with Tris-buffered saline containing 0.1% tween 20, the membrane was incubated with secondary antibodies labeled with IRDye 700 or 800 (LI-COR Biosciences) for 1 hour. After washing again with Tris-buffered saline containing 0.1% tween 20, the bands were detected and quantified using the Odyssey system (LI-COR Biosciences). Bands were normalized to β-actin by dividing by the intensity of the β-actin from the same sample blot. The experiments were performed in triplicate.
F-Actin Staining
CIPp cells were seeded on a coverslip (5 × 104 cells), cultured overnight, and then treated with 100 μM PAR2 agonist or vehicle for 6 hours. After washing twice with PBS, the cells were fixed with 4% paraformaldehyde for 10 minutes. Cell membranes were permeabilized with PBS containing 0.1% Triton X-100, and then incubated with phalloidin labeled with Alexa Fluor 594 (Thermo Fisher Scientific) for 20 minutes at room temperature. After the unreacted dye was removed by washing with PBS, the labeled F-actin was visualized using fluorescence microscopy (ECLIPSE TE-300; Nikon). The experiments were performed in triplicate.
Statistical Analyses
Survival curves according to PAR2 expression in each tumor were generated using the Kaplan-Meier method, and OS was compared between the high and low PAR2 expression groups by using the log-rank test. Censored includes cases that are still alive at the time of analysis (12 of 31 cases; all representing the PAR2 low expression group) and cases that have been unable to be followed up (3 of 31 cases; all representing the PAR2 high expression group). Comparisons of PAR2 expression with tumor size (≤5 vs >5 cm), lymph node involvement (absent vs present), distant metastasis (absent vs present), clinical stage (I-II vs III-V), and histological grade (I-II vs III) were performed using Fisher’s exact test. A univariate analysis based on the Cox proportional hazards model was performed for 7 factors, age (≤11 vs >11 years), tumor size (≤5 vs >5 cm), lymph node and distant metastasis (absent vs present), clinical stage (I-II vs III-V), PAR2 expression intensity (high vs low), spayed (yes vs no), and postoperative medication (yes vs no). Histological grade was excluded because a significant internal correlation was observed with PAR2 expression intensity in Fisher’s exact test (
Univariate and Multivariable Analysis for Overall Survival in Canine Mammary Gland Carcinomaa.
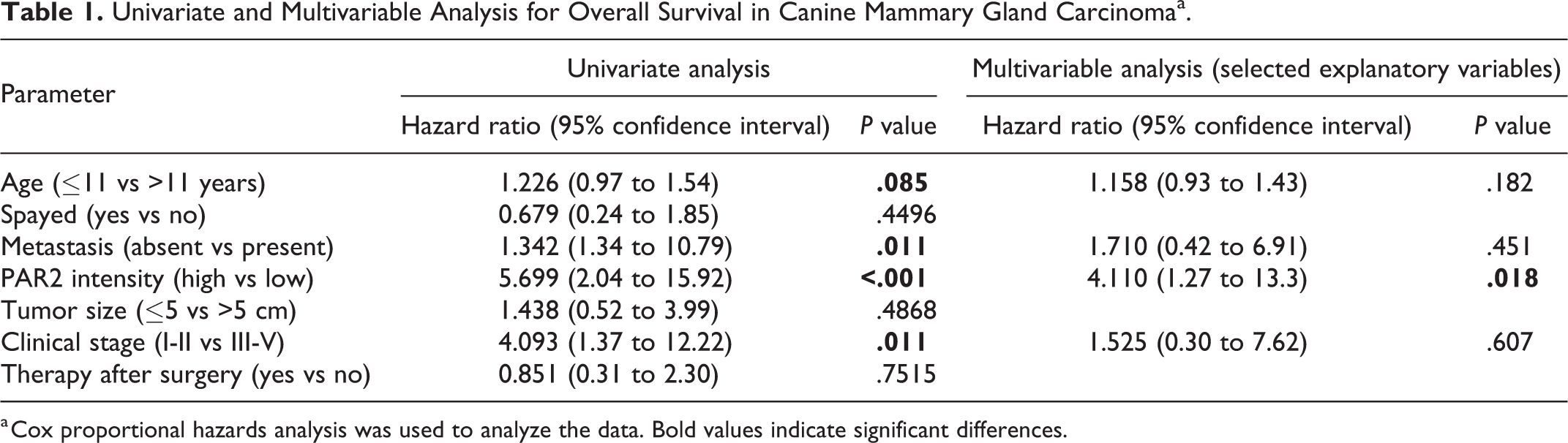
a Cox proportional hazards analysis was used to analyze the data. Bold values indicate significant differences.
Results
PAR2 Expression in Canine MC Was Associated With Shorter Survival and Advanced Clinical Stage
Representative images of PAR2 immunohistochemistry in normal mammary gland and MC are shown in Figures 1 to 5. No PAR2 immunoreactivity was observed in normal mammary glands (Fig. 1). In MC tissues, PAR2 immunoreactivity varied in the cytoplasm of carcinoma cells from absent to strong labeling (Figs. 2–5). PAR2 expression was not observed in neoplastic myoepithelial cells in MC tissues (Supplemental Fig. S1).
Of the 31 dogs with MC, 7 (23%) were PAR2 negative (score 0), 11 (35%) showed weak expression (score 1+), 9 (29%) showed moderate expression (score 2+), and 4 (13%) showed strong expression (score 3+). In both simple and complex carcinomas, only tumor cells were PAR2 positive. No difference was observed in PAR2 expression intensity between simple and complex carcinomas.
During the follow-up period, 16 of the 31 dogs died, and 14 of 16 dogs died due to tumor progression and recurrence. No cause of death, including another concurrent disease process, was identified based on imaging and blood tests in the other 2 cases. No dogs were euthanized. Twelve dogs were alive at the end of the study period. The median OS in dogs with MC was 283 days (range, 11–867 days). To investigate the association of PAR2 expression with the clinical characteristics and outcomes in dogs with MC, each dog was classified into the high or low PAR2 expression group. As shown in Figure 6, the DFS of dogs with high PAR2 expression (median DFS, 132 days; range, 11–701 days) was significantly shorter than that of dogs with low PAR2 expression (median DFS, 482 days; range, 106–867 days;
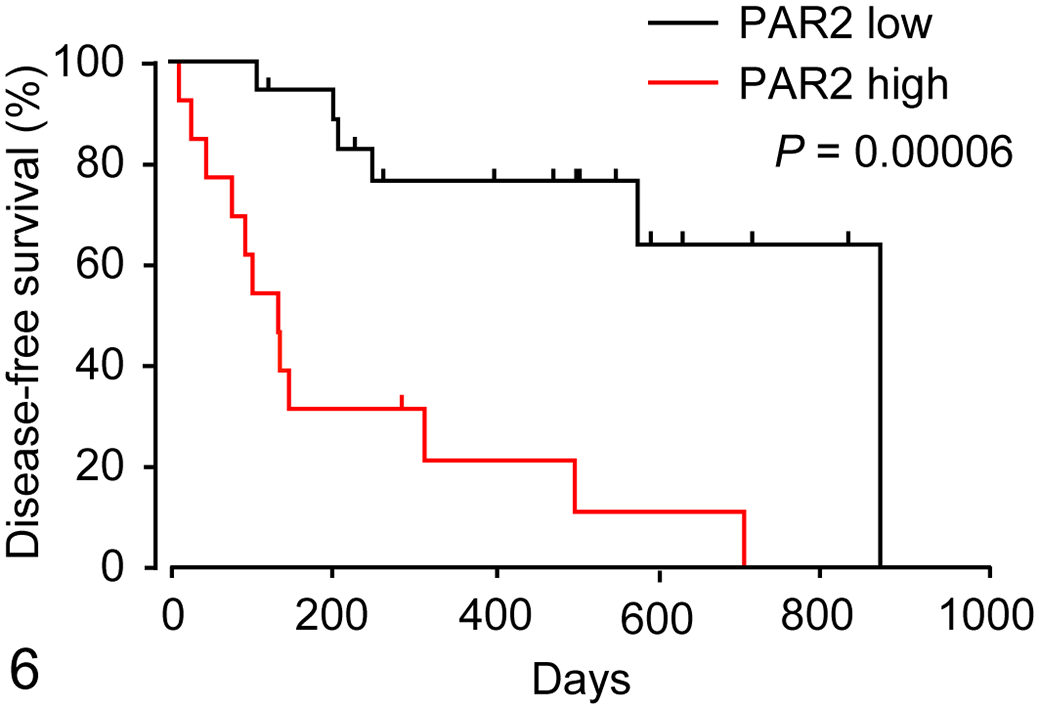
Kaplan-Meier curves of disease-free survival (DFS, Fig. 6) and overall survival (OS, Fig. 7) according to protease-activated receptor-2 (PAR2) expression in 31 dogs with mammary carcinoma (MC). Each dog is classified as high (
On univariate analysis, age (
Association Between PAR2 Expression and Clinicopathological Variables in Canine Mammary Gland Carcinomaa.
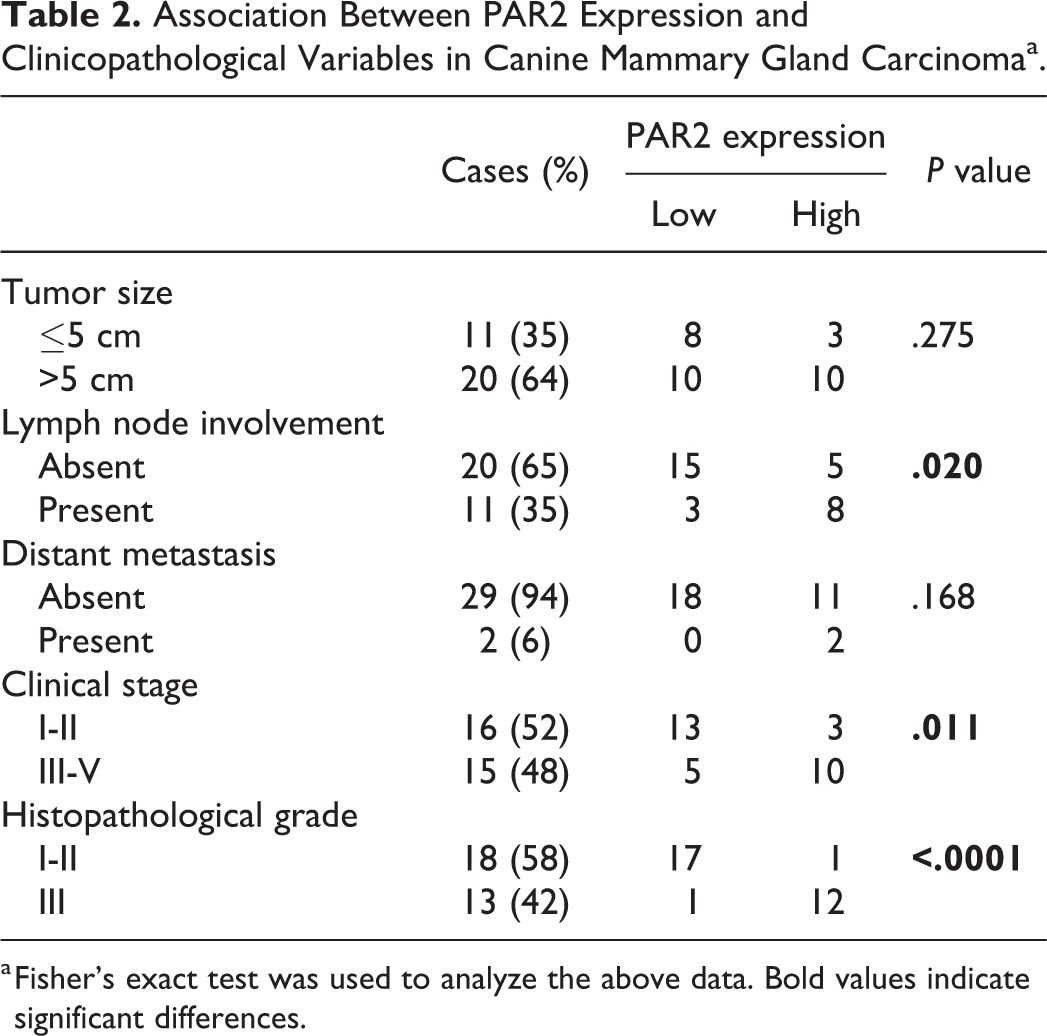
a Fisher’s exact test was used to analyze the above data. Bold values indicate significant differences.
Functional PAR2 Was Expressed in a Canine MC Cell Line
We further investigated the role of PAR2 in tumor progression by using a canine MC cell line, CIPp. The expression of PAR2 mRNA and protein in CIPp cells was confirmed via PCR and western blotting, respectively (Figs. 8, 9). PAR2 couples to Gq/11 protein, and PAR2 activation results in increased intracellular [Ca2+]i. Vehicle treatment did not cause a change in calcium concentration (Fig. 10), whereas treatment with a human PAR2 agonist (SLIGKV-NH2) induced intracellular Ca2+ mobilization (Fig. 11), indicating that PAR2 is functional in CIPp cells and that the human PAR2 agonist can activate canine PAR2.
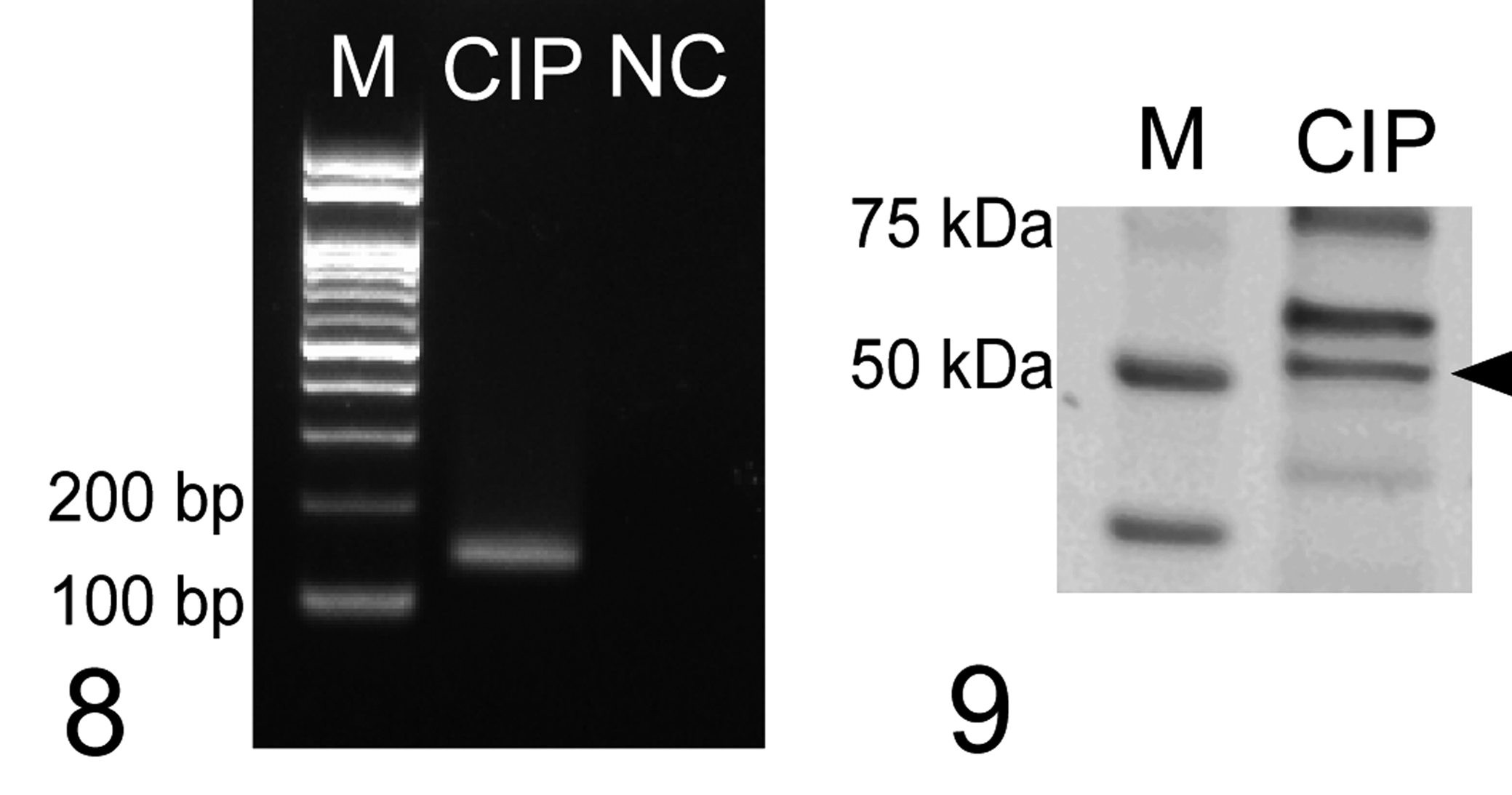
Expression of protease-activated receptor-2 (PAR2) mRNA in a cell line of canine mammary carcinoma, CIPp. Negative control (NC) is prepared via polymerase chain reaction without cDNA. M, marker.
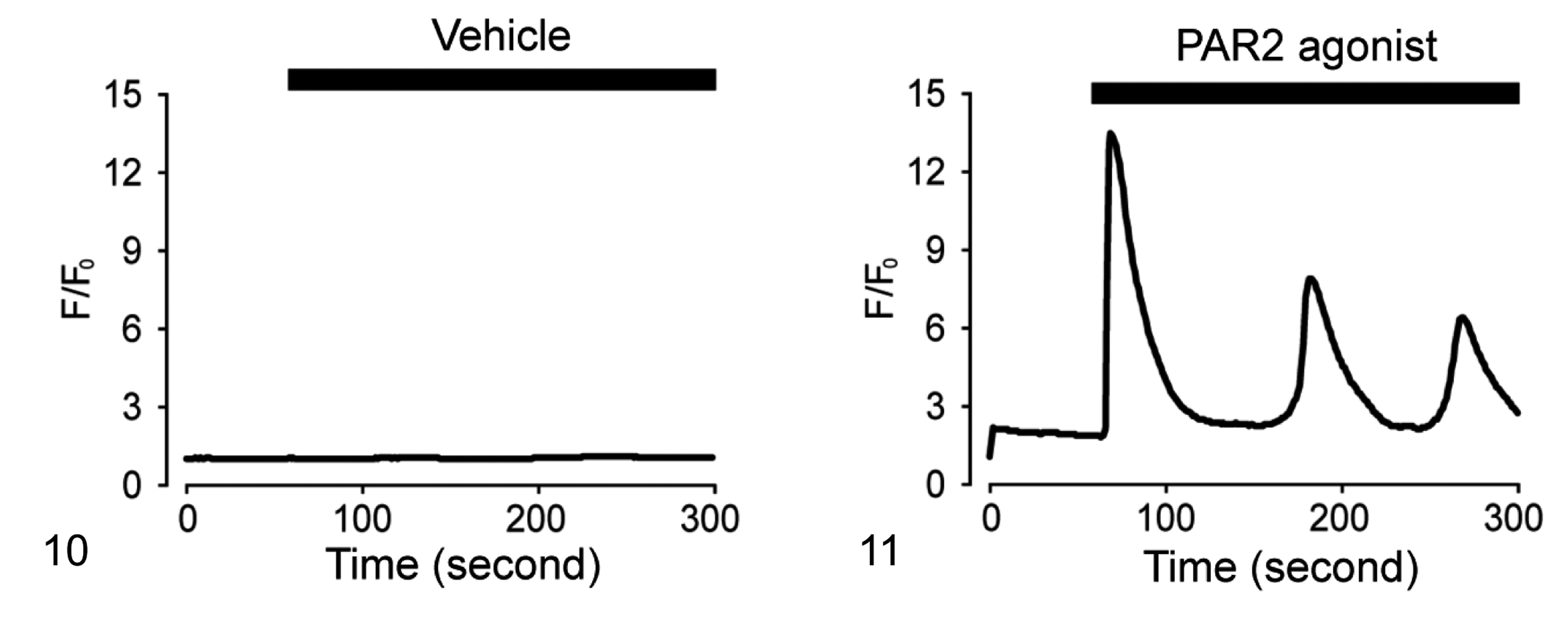
Intracellular Ca2+ response for PAR2 agonist in CIPp. Representative result of Ca2+ imaging. The cells are treated with a PAR2 agonist (SLIGKV-NH2) at the concentration of 100 μM or Vehicle. The values are presented as fold changes in fluorescence intensity (F) against the intensity at 0 seconds (F0).
PAR2 Activation Increased Cell Migration and Invasion
As a high expression level of PAR2 in canine MC tissues was associated with metastasis (Table 2), we investigated the effect of PAR2 activation on cell migration and invasion using the scratch wound healing and transwell invasion assays, respectively. CIPp cells treated with the PAR2 agonist exhibited increased cell migration compared to cells treated with the vehicle (Figs. 12, 13). Furthermore, the transwell invasion assay revealed that the PAR2 agonist induced cell invasion in CIPp cells (Figs. 14, 15). These results suggested that PAR2 activation accelerated cell migration and invasion in canine MC cells.
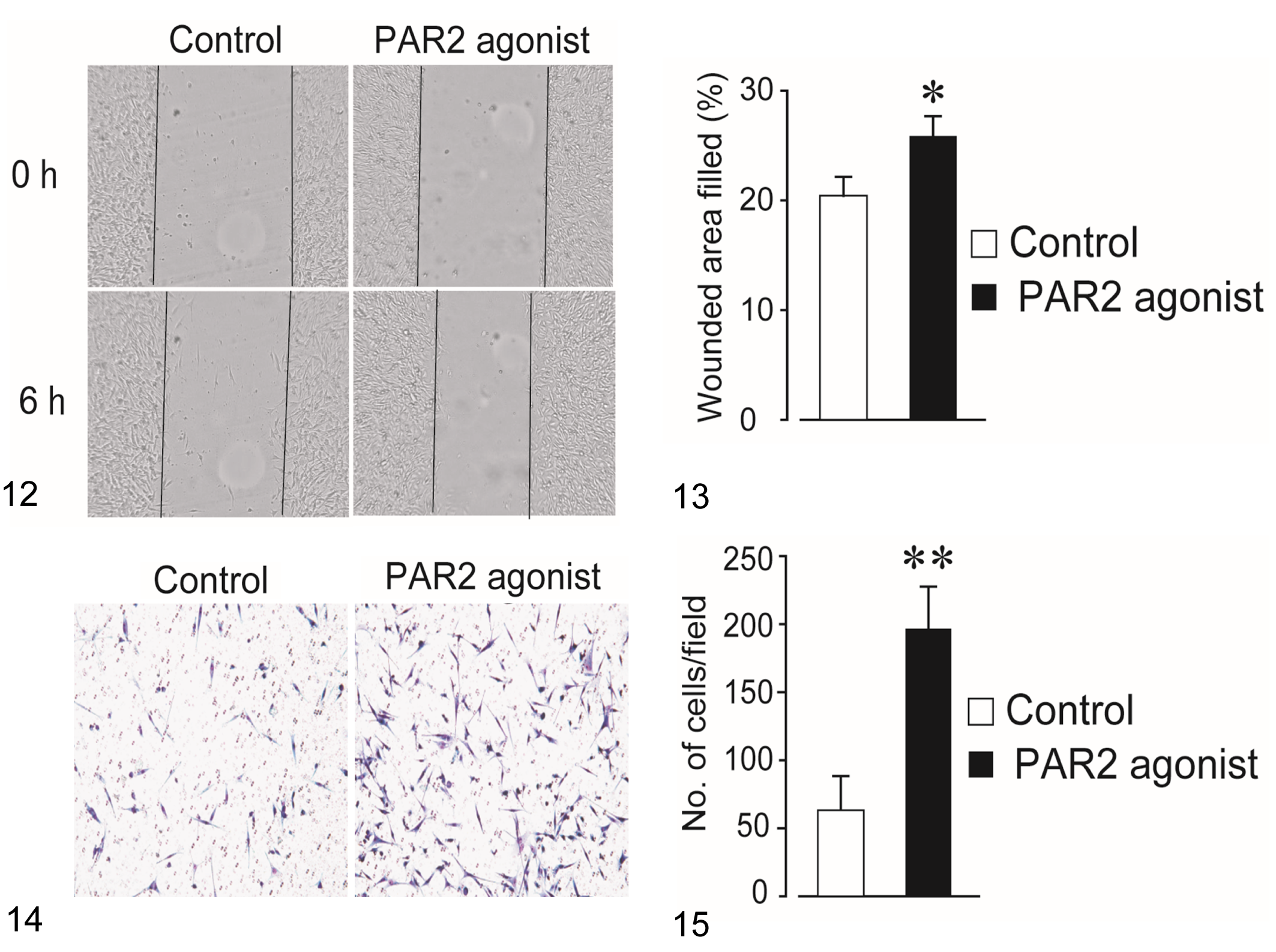
Migration of CIPp cells (a canine mammary carcinoma cell line) treated with a protease-activated receptor-2 (PAR2) agonist (SLIGKV-NH2, 100 μM). Representative images of the scratch wound healing assay at 0 hours and 6 hours after scratching. Black lines show the edge of the cell migration area.
PAR2 Activation Induced EMT and Actin Polymerization
The PAR2 agonist markedly increased the mRNA expression of α-SMA (
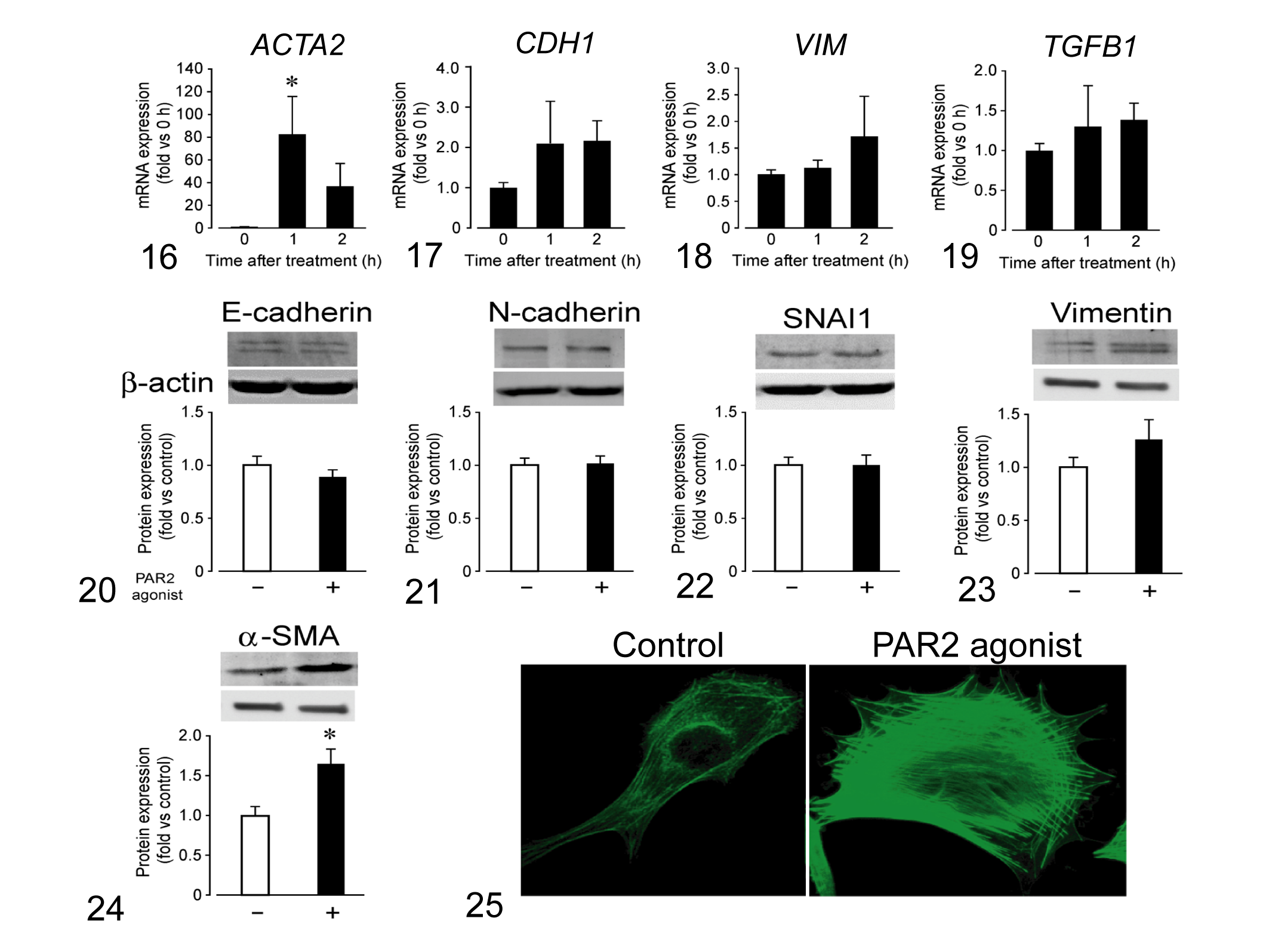
Effect of a protease-activated receptor-2 (PAR2) agonist (SLIGKV-NH2, 100 μM) on epithelial-mesenchymal transition of a cell line of canine mammary carcinoma, CIPp. The mRNA expression levels of E-cadherin (
Discussion
In the present study, we found high PAR2 expression was significantly associated with shorter OS and DFS in dogs with MC, and this finding was consistent with previous studies on human cancers. 3,6,7 In addition, significant associations were observed between PAR2 expression and poor prognostic factors, such as lymph node involvement and advanced clinical stage, in dogs with MC. There was also a significant correlation between PAR2 expression and histopathological grade, an important factor in determining prognosis. These results indicate that PAR2 plays an important role in the clinical outcomes and metastasis in canine MC.
It is known that dogs with a low clinical stage (stages I–III) can have a survival time of 28 months or more. 4 On the other hand, in the advanced clinical stage (stages IV–V), the survival time is about 6 months. 4 The median OS in this study was 283 days, which is relatively short compared to previous reports. The shortened median OS in this study might be due to the fact that 14 out of 31 cases were clinical stages IV to V and histopathological grade III.
EMT is a critical process in tumor metastasis. A previous report showed that PAR2 activation causes EMT through ERK activation in human hepatocellular carcinoma.
31
Another study reported that PAR2 is involved in EMT via TGF-β signaling in colorectal cancer cells.
26
In this study, the PAR2 agonist tended to increase the expression of EMT markers, such as α-SMA and vimentin, but had no effect on
Previous studies have shown that PAR2 activation promotes actin polymerization and contributes to tumor cell migration and invasion in a human breast cancer cell line through the β-arrestin-dependent ERK1/2 or Rab5a signaling pathways. 7,30 In this study, the PAR2 agonist induced the migration and invasion of canine MC cells. Moreover, actin polymerization was increased by the activation of PAR2. These results suggest that PAR2 may be associated with metastasis in canine MC via actin polymerization. Although we did not investigate the involvement of the β-arrestin-dependent ERK1/2 and Rab5a signaling pathways, similar pathways may be involved in canine MC.
The mechanisms underlying PAR2 overexpression in canine MC remain unclear. In humans, inflammatory cytokines, including interleukin-1 and tumor necrosis factor-α, are known to increase PAR2 expression in vascular endothelial cells. 8 However, no reports have documented these inflammatory cytokines in canine MC. Further research is needed to investigate the mechanisms underlying PAR2 overexpression in canine MC.
In human breast cancer, coagulation factor VIIa, tissue factor, and mast cell tryptase are considered the ligands that activate PAR2. 9,12 Although this study did not investigate the ligands of tumoral PAR2 in canine MC, the above serine proteases might be involved. In fact, the number of mast cells has been reported to be significantly increased in canine MC tissues. 39
A novel PAR2 antagonist, l-191, potently inhibited PAR2-mediated functional responses, including the secretion of inflammatory cytokines, cell apoptosis, and migration, in human colon and breast adenocarcinoma cell lines. 11 Furthermore, treatment with the tissue factor–specific therapeutic antibody SC1 ameliorated PAR2-mediated cell migration, metastasis, and tumor growth in human breast and pancreatic adenocarcinoma cells. 41 These findings suggest that blockade of the PAR2 pathway is a potential target for the treatment of canine MC.
This study has some limitations. First, the sample size was relatively small. Second, it is uncertain whether MC is the direct cause of death because autopsies were not been performed on the deceased dogs. Third, the histopathological types of the MC cases included in this study were limited. No difference was observed in the amount of PAR2 expression between simple and complex carcinomas. A previous human study showed that PAR2 expression was observed in patients with medullary breast carcinomas but not in those with infiltrating lobular carcinoma. 31 More extensive study is necessary to examine PAR2 expression in various types of canine MC. In addition, it is not clear whether PAR2 expression changes during lactation, because the ovarian status at the time when normal mammary glands were sampled could not be evaluated in this study. Therefore, it is necessary to compare PAR2 expression in normal tissues at various stages of the mammary gland.
In conclusion, PAR2 is expressed in canine MC cells, but the expression level varied from absent to strong. High PAR2 expression is associated with a poor prognosis and an advanced clinical stage. We also showed that PAR2 activation promotes tumor cell migration and invasion by inducing EMT and actin polymerization. The present results provide insights into cancer progression and suggest a potential novel therapeutic strategy for canine MC.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Kaji_et_al - Protease-Activated Receptor-2 Is Associated With Adverse Outcomes in Canine Mammary Carcinoma
Supplemental Material, Combined_supplemental_materials-Kaji_et_al for Protease-Activated Receptor-2 Is Associated With Adverse Outcomes in Canine Mammary Carcinoma by Kenjiro Kaji, Noriyuki Kaji, Masatoshi Hori, Kosei Sakai, Tomohiro Yonezawa and Shingo Maeda in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Grants-in-Aid for Science Research (KAKENHI Grant Numbers 16H06208 and 19H00968) and Anicom Capital Research Grant (EVOLVE).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
