Abstract
Despite promising immunotherapy strategies in human melanoma, there are few studies on the immune environment of canine melanocytic tumors. In humans, the activation of immunosuppressive cell subpopulations, such as regulatory T cells (Tregs) that express forkhead box protein P3 (FoxP3), the engagement of immunosuppressive surface receptors like cytotoxic T lymphocyte antigen (CTLA-4), and the secretion of molecules inhibiting lymphocyte activation, such as indoleamine-pyrrole 2,3-dioxygenase (IDO), are recognized as immunoescape mechanisms that allow tumor growth and progression. The aim of our study was to investigate the expression of these immunosuppression markers in canine melanocytic tumors and to postulate their possible role in melanoma biology and progression. Fifty-five formalin-fixed, paraffin-embedded canine melanocytic tumors (25 oral melanomas; 20 cutaneous melanomas; 10 cutaneous melanocytomas) were selected to investigate the expression of FoxP3, CTLA-4, and IDO by immunohistochemistry and RT-qPCR (real-time quantitative polymerase chain reaction). All of the tested markers showed high gene and protein expression in oral melanomas and were differently expressed in cutaneous melanomas when compared to their benign counterpart. IDO expression was associated with an increased hazard of death both in univariable and multivariable analyses (
Human melanoma is recognized as one of the most immunogenic tumors. Melanoma cells bear a high mutational burden compared to other malignancies and can acquire hundreds of mutations per megabase. 1,35,55 Recently, tumor heterogeneity has been demonstrated to further contribute to the host immune response, and it is a better predictor of immunotherapy outcome compared to mutational burden. 22,84 Despite melanoma’s immunogenicity, the host immune response is not effective in controlling tumor progression because the tumor itself is able to mold the immune response to its own benefit through the process of immunoediting. 18 It is currently believed that the interplay between the tumor and the immune system can be divided into 3 phases. The elimination phase is characterized by the development of a tumor-specific immunity, in which tumor-specific CD4+ and CD8+ cells are able to eliminate the tumor. The equilibrium phase is characterized by a dynamic balance between tumor cell variants that survived the elimination phase and the host immune system. Last, during the escape phase, selected tumor cells avoid immune detection and elimination. 19,45 The role of different immune cell populations in the process of immunoediting has been widely investigated in humans. This led to the development of new immunotherapy strategies, including immune checkpoint blockade, that target the host’s immune system to improve or restore protective immune functions, as well as inhibit immunosuppressive pathways activated during the escape phase. This type of treatment has been applied to different types of cancer, especially melanoma, resulting in durable responses, even in patients with metastatic disease. 11,41
The transcription factor forkhead box protein P3 (FoxP3) is involved in ensuring immune homeostasis, but it is also a key molecule suppressing cytotoxic T cell activity in antitumor immune response. 32,38 FoxP3 is expressed in Tregs, which have been associated with a negative prognosis both in human melanoma and other solid cancers. 24,43,63 A recent study also showed that Tregs were less numerous in areas of melanoma regression, confirming their potential role in the establishment of an immunosuppressive environment. 25 Moreover, in a murine melanoma model, it was shown that selective FoxP3 depletion achieved through vaccination led to the depletion of myeloid-derived suppressor cells (MDSCs), the reduction of tumor growth, and the improvement in survival rates, 52 supporting the role of Tregs in tumor progression and growth. The presence of FoxP3+Tregs has been also reported in canine tumors, including melanoma. 12,58,67
Cytotoxic T lymphocyte antigen (CTLA-4), also known as CD152, is a member of the family of immunoglobulin-related receptors expressed on both activated lymphocytes and Tregs and is responsible for T cell regulation and preservation of a normal immune environment. CTLA-4 binds to high-affinity B7 ligands (CD80 and CD86) on antigen-presenting cells (APCs). This leads to the inhibition of T cell responses and proliferation (T cell exhaustion), and antagonizes the binding of the T cell-stimulating receptor, CD28. 16,17,65,72,81 The importance of CTLA-4 in immune responses was shown when fatal autoimmunity was observed in CTLA-4-deficient mice due to the release of self-reactive T cells, suggesting that CTLA-4 is a negative regulator of T cell response. 80 Antitumor immunity is predominantly mediated by T cells and CTLA-4 has been shown to play a pivotal role in cancer-associated immunoediting, particularly in the escape phase. 72,81 Persistent antigen expression by melanoma cells and chronic stimulation of the immune system seems to be critical in the hyperactivation of inhibitory checkpoints on immune cells such as CTLA-4, resulting in the suppression of cytotoxic T cells. 26 CTLA-4 is also the target of Ipilimumab, an immune checkpoint inhibitor that is used to treat melanoma. 10
Another pathway that could contribute to peripheral tolerance and therefore to cancer immunoescape is mediated by indoleamine-pyrrole 2,3-dioxygenase (IDO), an enzyme with immunosuppressive properties postulated to impair the antitumor immune response in melanoma. 61 IDO can be produced by MDSCs, dendritic cells (DCs), macrophages, and tumor cells, and it is believed to inhibit effector T cells by depleting tryptophan in the tumor microenvironment. 29,49,50 Tryptophan catabolites (such as L-kynurenine) can further suppress the proliferation of activated T cells and promote the differentiation and activation of Tregs and CTLA-4 expression. 15,48 Inhibition of IDO, in combination with other immunotherapeutic drugs, can lead to improved response rates during melanoma therapy. 8 IDO blockade can reduce tumor growth, intratumoral immunosuppression, and stimulate robust systemic antitumor effects. 30,33,47 Nevertheless, a recent phase III study did not observe any difference in the group of patients with metastatic melanoma treated with IDO inhibitors when compared to the placebo-treated group. 37 Therefore, further studies are required to better understand the role of this enzyme as a potential therapeutic target.
During the past few years, growing evidence suggests canine melanomas, particularly mucosal melanomas, might be a predictive a preclinical model for human melanoma. 27 Still, further studies are recommended to better characterize the canine disease, including on the immunological front. The aim of this study was to retrospectively investigate the presence of immunoescape mechanisms in canine melanocytic tumors, through the analysis of FoxP3, CTLA-4, and IDO gene and protein expression and to gain more information on the possible similarities between canine and human melanoma immunology.
Materials and Methods
Case Selection
Cases were retrospectively selected on the following inclusion criteria: histological diagnosis of melanoma or melanocytoma, 75 with immunohistochemical positivity for Melan-A and/or PNL2; availability of follow-up information; and minimum follow-up time of 365 days.
Mitotic count was assessed following a proposed standardized method. 44 A telephone survey was conducted with the referring veterinarians, to collect data on the clinical tumor staging, treatment, local recurrence, and the cause of death. Disease-free and overall survival times were calculated from the day of sample registration in our departments.
Immunohistochemical Labeling and Evaluation
Samples were cut into 5-µm sections, mounted on poly-L-lysine coated slides, dewaxed, and rehydrated. Heavily pigmented tumors were bleached overnight at room temperature with 30% H2O2 following a standardized protocol. 58 Immunohistochemistry was performed on serial sections with antibodies against Melan A (pH 9.0 antigen retrieval; 1:150; mouse monoclonal, clones A103-M27C10-M29E3; Abcam), PNL2 (pH 6.0 antigen retrieval; 1:150; mouse monoclonal, clone PNL2; Santa Cruz Biotechnology), FoxP3 (pH 9.0 antigen retrieval; 1:100 dilution; rat monoclonal, clone FJK-16s; Thermo Fisher), CTLA-4 (pH 9.0 antigen retrieval; dilution 1:100; mouse monoclonal, clone F-8; Santa Cruz Biotechnology), and IDO (pH 9.0 antigen retrieval; 1:50 dilution; rabbit polyclonal; Biorbyt) with standardized protocols previously reported. 58 FoxP3 FJK-16s clone is reported to cross-react with canine antigen, 46 whereas anti-IDO and anti-CTLA4 antibodies were stated to cross-react with canine tissue and to be suitable for formalin-fixed, paraffin-embedded (FFPE) material in the datasheet provided by the manufacturer. The labeling pattern observed in the lymph node supported the specificity of both the antibodies when compared to previously reported studies 9,34 (Supplemental Figs. S1–S3). Tris-EDTA (pH 9.0) was used to perform heat-induced epitope retrieval for CTLA-4. Immunolabeling was revealed with 3-amino-9-ethilcarbazole (Dako); Mayer’s hematoxylin was applied as a counterstain. Reactive canine lymph node was used as a positive control for all the antibodies of this study. Negative controls were run by incubating sections with TBS and omitting the primary antibody and by incubating control tissue with isotype-matched antibody (only for monoclonal antibodies) to assess the absence of nonspecific labeling. Positive cells were counted by 2 operators, in 5 high-power field (HPF; FN 20), selecting “hot spots” and avoiding areas of necrosis and/or near ulceration; a mean value was then obtained for each case and expressed as the number of positive cells/HPF. The same method was applied for the evaluation of FoxP3, CTLA-4, and IDO positive cells. The expected labeling was nuclear for FoxP3, both membrane and cytoplasmic for CTLA-4, and granular and cytoplasmic for IDO.
Thirty out of 55 cases were part of the selected cases for our previous study, 58 whereas 25/55 cases were investigated ex novo.
RNA Extraction and Real Time Qualitative Polymerase Chain Reaction (RT-qPCR)
Three to 5 (depending on sample size), 8-µm-thick, sections were cut from paraffin blocks. Normal tissue around the tumor was resected and discarded with the help of a sterile scalpel blade or a sterile needle. RNA extraction was performed with a commercial kit (Invitrogen PureLink, FFPE RNA Isolation Kit) following the manufacturer’s instructions. Residual genomic DNA was removed from the total RNA by DNase I, amplification grade (Thermo Fisher Scientific) following the manufacturer’s specifications. RNA quantity was evaluated by both NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific) and Qubit 2.0 Fluorometer (Life Technologies). Total RNA (500 ng) was reverse transcribed using the SuperScript VILO Master Mix (Thermo Fisher Scientific), according to the manufacturer’s specifications. Successful reverse transcription was confirmed by PCR amplification of the
Primer Combinations and Accession Numbers for Tested Genes.

Statistical Analysis
Diagnostic graphics were used to test assumptions and outliers. We analyzed distributions within the categorical variable “breed” using χ2 goodness-of-fit tests. Differences in number of positive cells and mRNA expression of IDO, FoxP3, and CTLA-4 between diagnoses were analyzed using Kruskal-Wallis and Mann-Whitney tests. Values were expressed as medians with interquartile range (IQR). Correlations were evaluated by using Spearman rank correlation coefficient (ρ). Correlation was defined as high when absolute value of ρ > 0.5, medium when ρ ranged from 0.3 to 0.5, and low when ρ < 0.3.
21
We used the Life Table method to determine survival probabilities. The differences of survival rate according to diagnosis were evaluated by Kaplan-Meier curve and log-rank test. Dogs that died of other causes or were lost to follow-up were considered censored. We used the Cox proportional hazards model to evaluate the influence of parameters on survival. In addition to the univariable analysis, each parameter (IDO [cells/HPF], FoxP3 [cells/HPF], CTLA-4 [cells/HPF],
Results
Sample Population and Mitotic Count
The final caseload was represented by 25 oral melanomas, 20 cutaneous melanomas, and 10 cutaneous melanocytomas. Thirty-five dogs were male (6/35, 17% were neutered) and 19 were female (6, 32% were spayed). For one animal the age was unknown. Most of the dogs were mixed breed (19/55, 35%;
The 6-month and 1-year estimated survival probabilities are shown in Supplemental Table S1. Median survival time for mucosal melanoma was 240 days (IQR = 77–433 days), while it was not reached for cutaneous melanoma or melanocytoma. Log-rank test showed lower survival time for dogs with mucosal melanoma compared to dogs with cutaneous melanoma (
Mitotic counts were higher in oral melanomas (median = 42, IQR = 32–61) than in cutaneous melanomas (median = 14, IQR = 7–50,
Immunohistochemistry
FoxP3, CTLA-4, and IDO positive cells were identified in all oral and cutaneous melanomas, while some cutaneous melanocytoma did not contain any cells expressing these proteins. FoxP3 and CTLA-4 immunolabeling was localized in the nucleus and the cytoplasmic membrane, respectively, in cells with scant cytoplasm, interpreted as lymphocytes. IDO was mostly expressed in the cytoplasm of cells with moderate to abundant cytoplasm (interpreted as histiocytes/macrophages), and rarely in neoplastic cells.
Immunohistochemical labeling revealed that the number of FoxP3+Tregs (Figs. 1–3) was higher in oral melanomas than cutaneous melanomas and cutaneous melanocytomas. Similarly, the number of CTLA-4 positive cells/HPF (Figs. 4–6) was higher in oral melanomas than in cutaneous melanomas and cutaneous melanocytomas. The number of IDO positive cells (Figs. 7–9) was higher in oral melanoma than in cutaneous melanomas and cutaneous melanocytoma. Results are summarized in Figures 10 to 12.
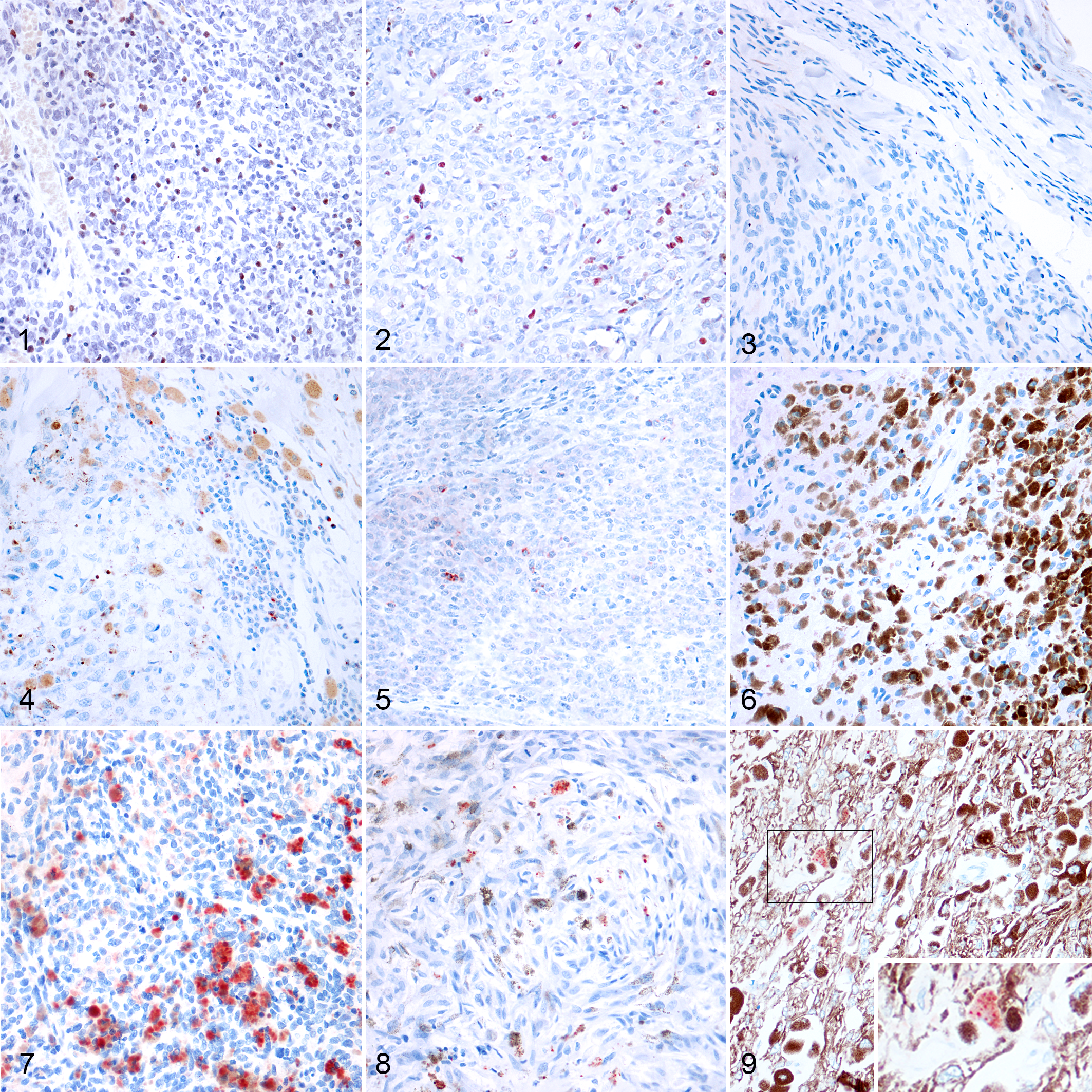
Melanocytic neoplasms, dog. The immunolabeling is nuclear in small cells with scant cytoplasm (lymphocytes). Immunohistochemistry for FoxP3 (AEC chromogen and hematoxylin counterstain).
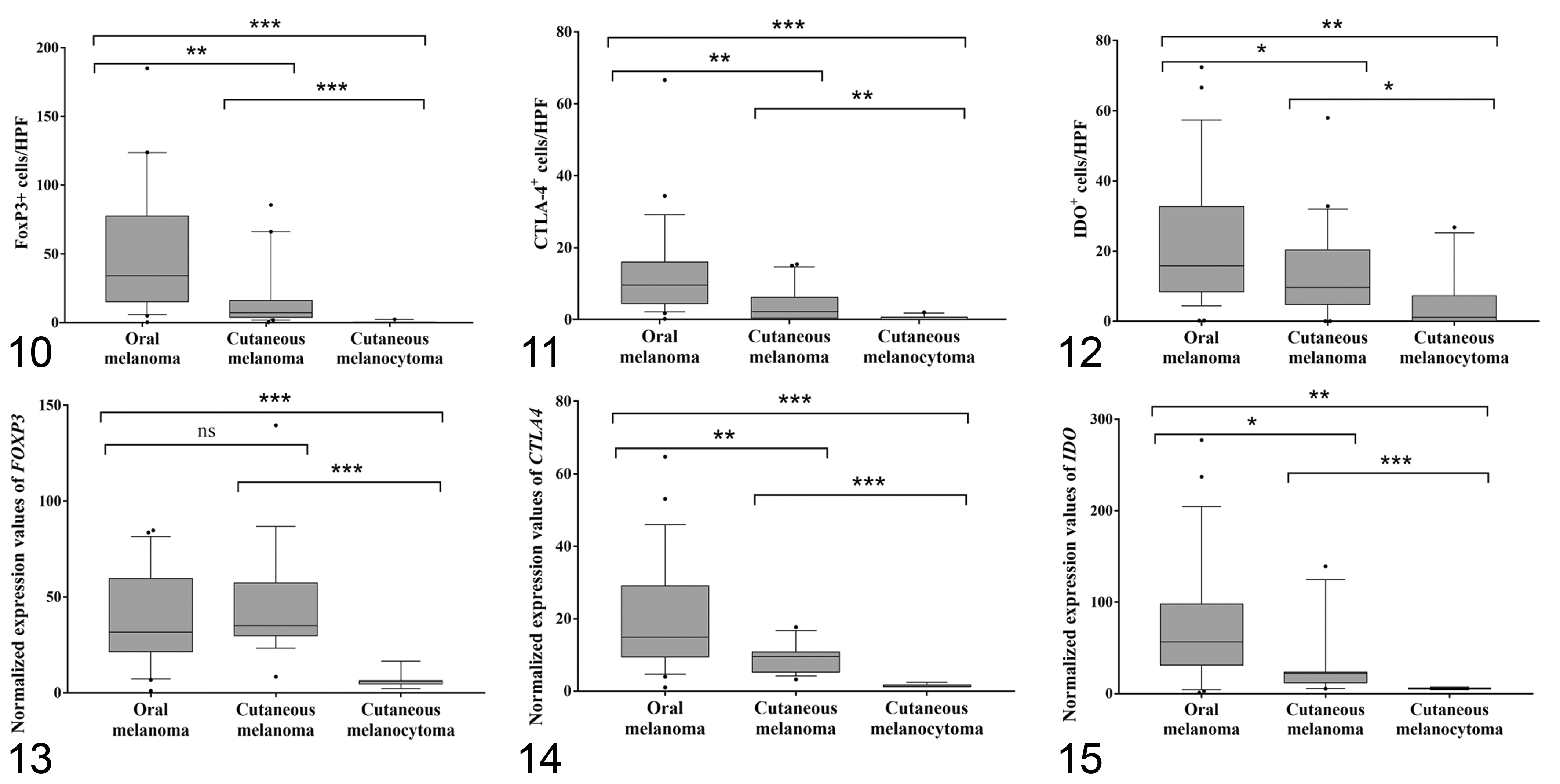
Box plots of the number of FoxP3, CTLA-4, and IDO-positive cells/HPF. The horizontal line in the box is the median, the whiskers are 1.5 times the interquartile range, and the stars are outliers. ns = not significant, *
RT-qPCR
After RNA extraction, samples showed A260/280 ratio ranging from 1.64 to 2.02. Both
Correlations Between FoxP3, CTLA-4, and IDO Protein, Transcripts, and Mitotic Count
All of the examined parameters evaluated with both immunohistochemistry and RT-qPCR showed positive correlations between them and with mitotic counts (Supplemental Table S3). A strong correlation was observed between FoxP3+ cells/HPF and CTLA-4+ cells/HPF (ρ = 0.709,
The correlation between gene expression and immunohistochemical protein expression was moderate for
Prognostic Significance of FoxP3, CTLA-4, and IDO
The univariable Cox analysis (Table 2) showed an increased hazard of death in association with an increased expression of CTLA-4 and IDO, both at the protein and mRNA levels (
Results of Univariable and Multivariable Cox Proportional Hazards Analyses. The Event Was Tumor-Related Death.
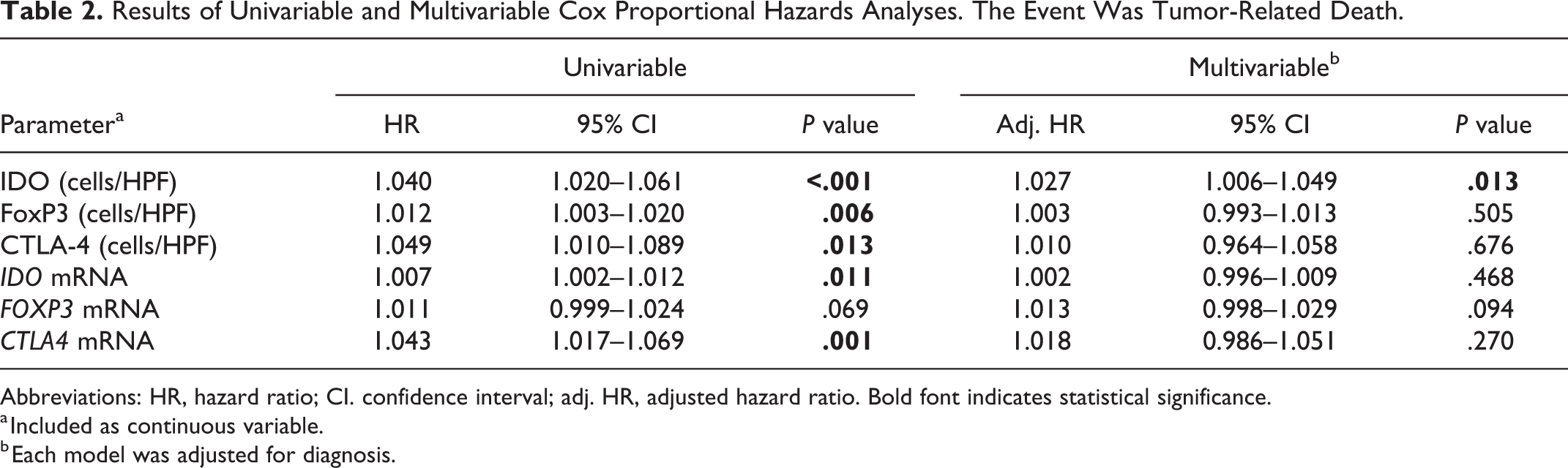
Abbreviations: HR, hazard ratio; CI. confidence interval; adj. HR, adjusted hazard ratio. Bold font indicates statistical significance.
a Included as continuous variable.
b Each model was adjusted for diagnosis.
We investigated the sensitivity and specificity associated with a previously defined IDO cutoff value of 14.7 cells/HPF. 58 This cutoff value showed a 57% sensitivity and 79% specificity. In contrast, a cutoff value of 8.4 cells/HPF was identified in this study with an 82% sensitivity and 68% specificity.
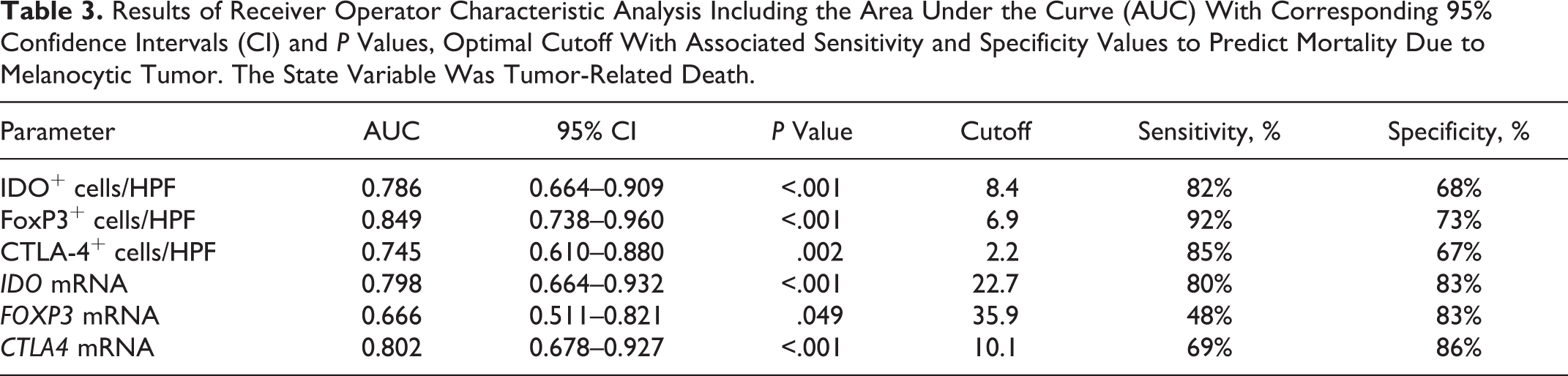
Table 3 shows the other results of ROC analysis and the optimal cutoffs for predicting melanoma-related mortality. The highest area under the curve (AUC) was found for FoxP3+ cells/HPF (AUC = 0.849;
Results of Receiver Operator Characteristic Analysis Including the Area Under the Curve (AUC) With Corresponding 95% Confidence Intervals (CI) and
Dichotomous variables were created for each parameter based on their optimal cutoffs (< or ≥ of the cutoff) and analyzed by Kaplan-Meier survival curves (Figs. 16–21) and Cox models adjusted for diagnosis (Table 4). Categorizing according to their optimal cutoff, FoxP3 (
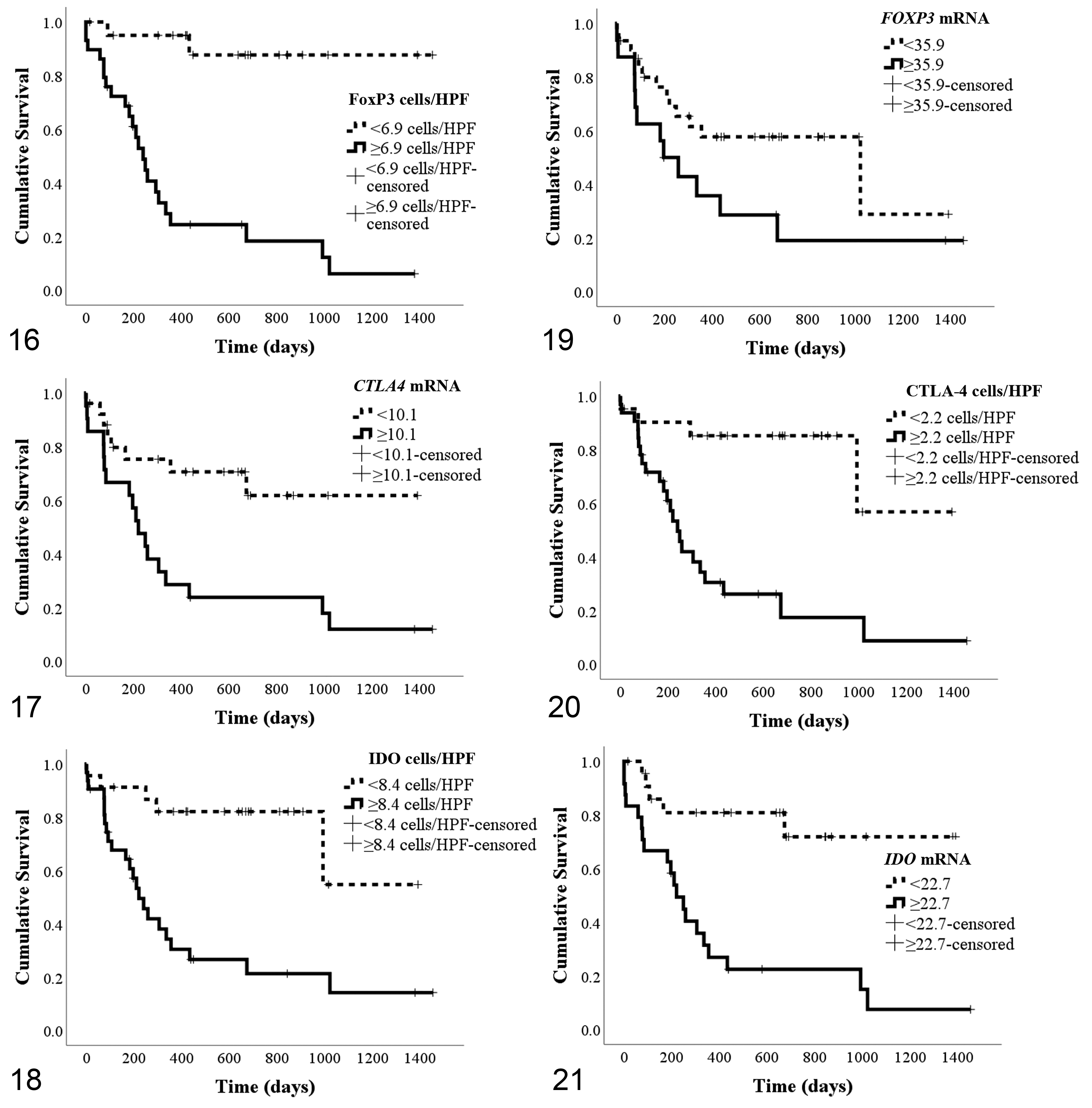
Kaplan-Meier survival curves by optimal cutoff values of FoxP3 (Fig. 16; 6.9 cells/HPF), CTLA-4 (Fig. 17; 2.2 cells/HPF), and IDO (Fig. 18; 8.4 cells/HPF), and
Cox Proportional Hazards Analyses of Parameters According to Their Cutoff (Data of the Entire Cohort). The Event Was Tumor-Related Death.
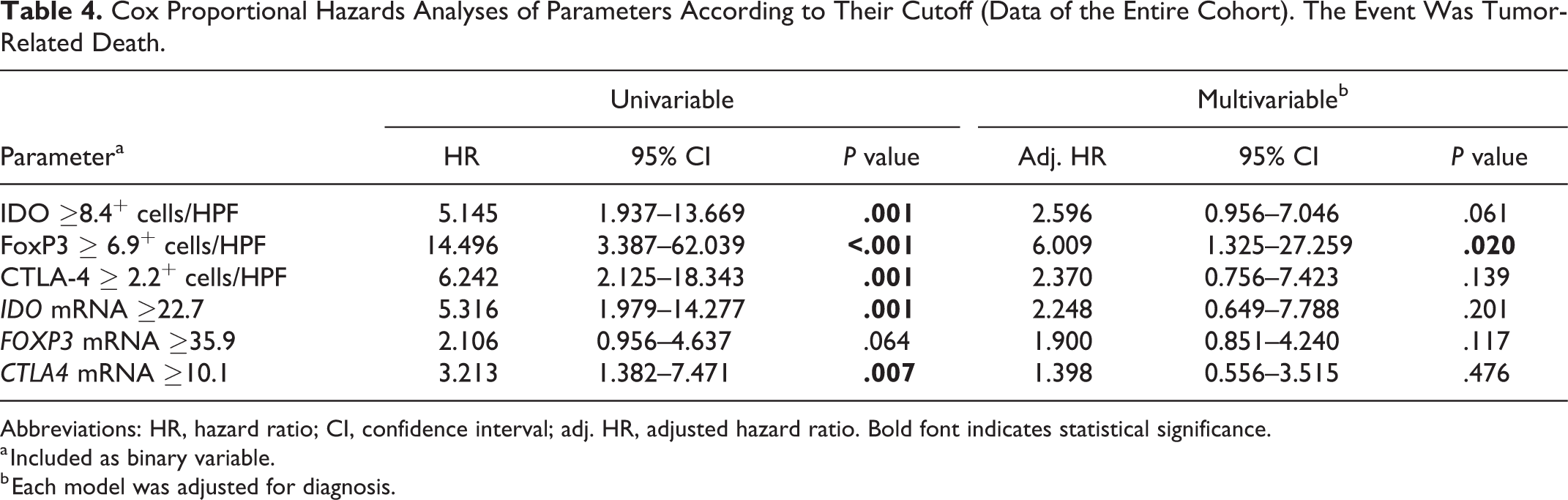
Abbreviations: HR, hazard ratio; CI, confidence interval; adj. HR, adjusted hazard ratio. Bold font indicates statistical significance.
a Included as binary variable.
b Each model was adjusted for diagnosis.
Discussion
In humans, a growing number of studies characterizing the melanoma immune environment has led to the successful use of immunotherapy, particularly by targeting PD-1 and CTLA-4. 54 In veterinary medicine, there are fewer studies on cancer immunity and on the application of immunotherapy. 2,39,58,60
Canine cutaneous melanomas are usually benign and surgical resection is typically curative; still, their behavior can be quite unpredictable. 20,69,76 On the other hand, mucosal melanomas, particularly oral melanomas, show a malignant behavior with a predisposition to the development of lymph node and lung metastasis, similar to human melanomas. 75 Several studies suggest the dog is a valuable spontaneous preclinical model in melanoma research since canine oral melanoma shares numerous similarities with the more rare human disease. 62,74,83
While different aspects of canine melanoma biology have been investigated, 6,7,27,28,31,68,73 the immune environment of canine melanoma is still largely unknown. Our study aims to investigate the immune environment of canine melanocytic tumors to acquire further information on the possible mechanisms of immunosuppression and evasion involved in tumor progression; targets of our investigation are in particular FoxP3, CTLA-4, and IDO.
The mean survival time of dogs with oral melanomas in this study was 240 days. This is higher than is the previously mean survival time of 147 days. 76 This discrepancy could be because of earlier diagnosis, and because of the inclusion of labial melanomas in this study, which are reported to have a longer survival time. This result endorses the necessity for further studies to better characterize oral and mucocutaneous canine melanocyte biology, together with melanoma behavior in association with different sites of origin. A detailed description of the tumor’s anatomical site of origin should be provided by the clinician/surgeon at the time of tissue submission for histopathological analysis; this would enable further evaluations on the prognostic significance of the primary tumor location.
FoxP3 is a transcription factor involved in Tregs development and function, and is currently considered their most specific marker and the main regulator of Treg lineage committment.
3,70
Still, Tregs are a heterogeneous population and better characterization of these cells could include other markers such as CD4, CD25, and CD45RO.
57
In our study, increased FoxP3 immunohistochemical expression and
The correlation between gene and immunohistochemical protein expression was moderate or moderate-to-low for all tested molecules. This could be due to the higher sensitivity of RT-qPCR when compared to the immunohistochemical quantification, but also to poor mRNA quality in FFPE samples, which directly affects the efficiency of some primer combinations (as low as 77.2%). The authors are aware that these are suboptimal values; however, as reported in a previous study, 7 accuracy in gene expression profile should not be compromised.
Markers to identify Tregs are limited and often nonspecific, 64 making the characterization of this T cell subpopulation, within tumor immune environment, difficult and still not completely understood. To overcome this problem, a colocalization of CD4, CD25, and FoxP3 would be useful to correctly identify Tregs subpopulation also in canine melanomas. Moreover, the presence of FoxP3+Tregs could be influenced by tumor site, molecular subtype of the tumor, and tumor stage, adding further bias to their evaluation. 70
During the past few years, immunotherapies with monoclonal antibodies directed against CTLA-4 and PD1 revolutionized the treatment of patients with advanced melanoma in human medicine. 10,36 Still, the role of CTLA-4-expressing cells have been explored in veterinary oncology in a limited manner. In the present study, we described the expression of this molecule in canine melanocytic tumors, suggesting that both CTLA-4 immunohistochemical and gene expression may associated with the histological diagnosis and with an increased hazard of death (univariable analysis), similarly to that reported in human melanomas. 13 However, in the multivariable analysis, CTLA-4 lost its statistical significance, suggesting that CTLA-4 may be not an independent predictor. Still, the association between the protein and gene expression of this marker and the tumor mitotic count, which is considered one of the most affordable prognostic factors in canine melanomas, 4,75 may corroborate the hypothesis of an immunosuppressive role of CTLA-4 in melanoma growth. To the best of our knowledge, this is the first study focusing on CTLA-4 within canine melanoma microenvironment; previous studies aimed at characterizing the expression of this costimulatory molecule in circulating cells during neoplastic disease and in a healthy subject. 77 –79 Our results, although preliminary, highlight the presence of this molecule within canine melanoma and open the path for new investigations on the role of CTLA-4-associated pathways in canine oncology.
IDO can be expressed by different cell types, in particular MDSCs, DCs, macrophages, and tumor cells. It acts both on APCs and T cells, causing immune suppression and facilitating cancer progression. 5 Our results show that IDO immunohistochemical expression was an independent predictor of mortality, even when the model was adjusted for diagnosis (melanocytoma, oral melanoma, cutaneous melanoma). The optimal cutoff value for IDO immunohistochemical expression in this study was set at 8.4 cells/HPF, compared to the 14.7 cells/HPF value that was used in our previous study. In addition to the different characteristics of the sample population, this incongruity can also be explained by the different percentages of sensitivity and specificity associated with this new cutoff. In the present study, the lowest cutoff was associated with a higher sensitivity (82%) in the prediction of death due to melanoma. By setting the IDO cutoff at 14.7 cells/HPF in this case series, the specificity improves, reaching the value indicated in our previous study (79%), but it is not balanced by an adequate sensitivity (57%). The greater accuracy is indicated by the higher AUC in the present study, together with a slightly higher number of cases with a complete follow-up (55 vs 52) in this study, suggests that the lower cutoff is preferred.
One of the possible limitations of our study is related to the fact that we did not stratify our cases by diagnosis. On one hand, the stratification would have led to a drastic reduction in both the sample size and the number of “events” (ie, deaths) to be included for survival analysis. On the other hand, the diagnosis of melanocytoma/melanoma poses often some doubts in the diagnostic routine of the pathologist, particularly in borderline lesions. By avoiding stratification, the cutoffs set for our markers were defined independent of the histologic diagnosis and the pathologists' judgment. Investigations based on a larger study are required to confirm these results and to better assess these markers’ prognostic value.
The role of IDO in canine melanoma may be similar its role in human melanoma, where IDO protein expression has been shown to have a prognostic role in both cutaneous melanoma and nodal metastases. 14,56,66 Gene expression, on the other hand, was significant only in the univariate Cox analysis. The loss of significance could be because of the high mRNA expression variability detected by RT-qPCR due to the use of FFPE material to retrieve mRNA. In fact, even though numerous studies have used this protocol, fresh-frozen tissue are preferred to avoid partial mRNA degradation. 40 Our results suggest that IDO is involved in canine tumor immunoescape and progression, but mechanistic studies are needed to confirm this finding. Also, IDO could be implicated in the activation of Treg cells within the canine melanoma microenvironment, as indicated by the moderate correlation between the variables in this study and in other models. 51,71
Interestingly, all of the markers tested in our study showed a significant difference between the cutaneous melanomas and cutaneous melanocytomas, suggesting increased activation of immunosuppressive pathways in malignant cutaneous tumors compared to their benign counterpart. This finding, if further confirmed by ongoing investigations, could confirm similar mechanisms of immune evasion in the dog as compared to human species. Furthermore, these markers could be useful in prognosticating canine cutaneous melanocytic tumors. If confirmed by prospective studies, the threshold of >6.9 FoxP3+ cells/HPF might be useful in the evaluation of canine melanocytic tumors, as evidenced by the multivariable analysis.
Taken together, the results from our study seem to confirm the presence of immunosuppressive tumor microenvironment mechanisms controlled by FoxP3, CTLA-4, and IDO in canine melanoma, particularly in the most aggressive oral form. After this retrospective investigation, prospective studies on fresh/frozen tissue aiming at the confirmation of these results, including and extending our investigation to other immune populations and to metastatic lesions, have been planned. Further investigations on the immune environment of canine melanocytic tumors should be initiated, aiming to both better characterize canine melanoma biology and immune environment and to possibly employ immunotherapeutic strategies in the canine species.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Porcellato_et_al - FoxP3, CTLA-4, and IDO in Canine Melanocytic Tumors
Supplemental Material, Combined_supplemental_materials-Porcellato_et_al for FoxP3, CTLA-4, and IDO in Canine Melanocytic Tumors by Ilaria Porcellato, Chiara Brachelente, Katia Cappelli, Laura Menchetti, Serenella Silvestri, Monica Sforna, Samanta Mecocci, Selina Iussich, Leonardo Leonardi and Luca Mechelli in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank Gianluca Alunni, Valeria Migni, and Luca Stefanelli for their helpful technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
