Abstract
Bacterial infections in chicken eggs often cause mortality of embryos and clinical consequences in chicks but the pathological mechanism is unclear. We investigated the pathological changes and bacterial growth kinetics in dead and live embryos following infection with 2 Escherichia coli strains with a different clinical background and with 1 Salmonella Enteritidis strain. In 2 experiments, 12-day-old embryos were infected via the allantoic sac with 100 µl of 1 to 5 × 102 CFU/ml of one of the bacteria. In experiment 1, only dead embryos were sampled until 4 days postinfection (dpi), and surviving embryos were sampled at 5 dpi. In experiment 2, sampling was performed in dead and killed embryos sequentially at 1, 2, 3, and 4 dpi. The bacteria showed varying pathogenicity in embryos. The yolk sacs of dead embryos showed congestion, inflammation, damaged blood vessels, and abnormal endodermal epithelial cells. Such lesions were absent in the yolk sacs of negative control embryos and in those of embryos that survived infection. The livers and hearts of dead embryos showed congestion and lysed erythrocytes with no morphological changes in hepatocytes or myocardial cells. All bacteria multiplied rapidly in the yolks of infected embryos, although this did not predict survival. However, the livers of dead embryos contained significantly higher bacterial loads than the livers of the embryos that survived infection. The results provide evidence that lesions in the yolk sac, which have been neglected to date, coincide with embryonic mortality, underlining the importance of healthy yolk sacs for embryo survival.
There have been many studies of the physiological and biochemical processes during normal embryogenesis in chicken eggs. 27 As in all oviparous species, the development of chicken embryos depends on an adequate supply of maternally derived nutrients from the yolk, which is the almost exclusive reservoir of nutrients. The yolk sac, which is functionally equivalent to the mammalian placenta, has a vital role as the gateway from the yolk to the developing embryo and mediates the absorption, digestion, and transfer of nutrient components. 23 The yolk sac is composed of 3 germ layers, the endoderm, mesoderm, and ectoderm. 19 The absorption of nutrient metabolites occurs through endodermal endothelial cells by a process called receptor-mediated endocytosis of lipoproteins. The lipoproteins are transported to a vascular system present in the mesodermal layer, which is connected with the embryo. 2,30 Shortly before the embryos hatch, the yolk sac forms a diverticulum attached to the intestine and the remaining yolk is internalized in the abdominal cavity. 4,7,25 The yolk sac is also a major site for hematopoiesis and the synthesis of serum proteins. It also represents a biological barrier and is involved in the defense against pathogens. 13,15,39
Embryonated chicken eggs are extensively used in the laboratory as a versatile test system. Their many applications include the embryo lethality assay, which is based on the percentage of embryo mortality following experimental infection and is widely used to determine the pathogenicity of bacterial isolates. 11,21,26,28 However, detailed pathological studies in embryonic and extraembryonic tissues following infections are often neglected. Although it is well understood that bacterial infection might lead to embryonic mortality, the mechanism of embryonic death is not yet known.
Escherichia coli causes a range of diseases in poultry 24 and is one of the major causes of yolk sac infection, omphalitis, and early mortality of young chicks. 6,16,18,31 Contamination of the egg shell might lead to infection of chicks with E. coli with or without other pathogens such as Bacillus cereus, staphylococci, or Proteus spp. 18,31 Normally, intestinal E. coli isolates from healthy birds are regarded as nonpathogenic, whereas isolates from systemic organs with clinical signs of infection are in the pathotype called avian pathogenic E. coli (APEC). 24
Salmonella have a high zoonotic impact on human health and some serovars cause diseases in poultry. 10 Salmonella invade different types of cells, including epithelial cells, M cells, macrophages and dendritic cells, enabling them to hide from host immune cells and establish an infection. 17 It is generally accepted that vertical transmission of Salmonella might occur through internal or external contamination of eggs. 10 In young chicks, Salmonella infections often have clinical consequences such as unabsorbed yolk, omphalitis, airsacculitis, fibrinous hepatitis, enlarged spleen and kidneys, and dehydration. 1,22
We studied the detailed pathological changes in embryonic and extra-embryonic tissues following infection with 2 different strains of E. coli and an isolate of S. Enteritidis. We measured bacterial growth kinetics in dead and live embryos to link the pathomorphological and histological lesions and to unravel the pathogenesis of embryonic mortality.
Materials and Methods
Bacterial Isolates and Infection
The APEC strain E. coli PA14/17480/5-ovary was collected from the ovary of a layer bird from a commercial farm showing egg peritonitis. The commensal E. coli 2018/SPF-cecum was obtained from the cecum of a clinically healthy specified pathogen free (SPF) layer bird. Phylogenetic groups and virulence-associated genes in E. coli isolates were determined with published multiplex PCR methods 5,8 which revealed that E. coli PA14/17480/5-ovary and E. coli 2018/SPF-cecum belong to phylogenetic groups B2 and B1, respectively. The APEC strain contains 6/8 virulence-associated genes (iss, irp2, iucD, tsh, vat, Cvi/cva), whereas the commensal strain harbors only 1 of 8 genes (tsh) tested (data not shown). The Salmonella included in the study was the reference strain S. Enteritidis ATCC-13076.
All bacteria were grown in Lenox L-broth medium (LB broth, Invitrogen, Thermo Fisher Scientific) at 37 °C for 18 hours, under aerobic conditions. For infection, cultures were washed 3 times and resuspended in sterile phosphate buffered saline (PBS, Gibco) and serial 10-fold dilutions were made from each culture. Bacterial concentrations before and after infection were measured with a spectrophotometer (optical density at 600 nm) and colony-forming unit (CFU) counts of E. coli and S. Enteritidis by directly plating 100 µl of each dilution in duplicates on MacConkey agar (Neogen) and Xylose-lysine Deoxychocolate agar (XLD, Merck).
Infection of Embryos
The method reflects a standard procedure in diagnostics for the isolation and multiplication of avian pathogens and is not covered by the national law for animal experiments in Austria (Tierversuchsgesetz 2012).
Experiment 1
Sixty SPF eggs (VALO BioMedia GmbH, Osterholz-Scharmbeck, Germany) were divided into 4 equal groups and incubated under standard conditions (37 °C, 65% relative humidity). On day 12 of incubation, embryos in groups 1, 2, and 3 were infected with 100 µl of 1.4 to 3.1 × 102 CFU/ml of E. coli PA14/17480/5-ovary, S. Enteritidis ATCC-13076, and E. coli 2018/SPF-cecum, respectively, via the allantoic route as described. 32 Using the same route, embryos in group 4 were inoculated with the same amount of PBS and served as negative control. Following infection, eggs were candled every 24 hours. Only dead embryos were screened and sampled until 4 days post infection (dpi), and the remaining embryos were killed and sampled at 5 dpi. Liver, heart, and yolk sac tissues were collected for histopathological examination and liver and yolk samples from all embryos were processed for CFU count.
Experiment 2
For the second experiment, 48 SPF eggs (VALO BioMedia GmbH, Germany) were allocated into 4 groups with 12 eggs in each. Embryos in groups 1, 2, and 3 were infected with 100 µl of 3.4 to 5.0 × 102 CFU/ml of E. coli PA14/17480/5-ovary, S. Enteritidis, and E. coli 2018/SPF-cecum, respectively, whereas negative controls were only inoculated with PBS. The infection procedure and incubation conditions were the same as for experiment 1. Following infection, dead or live embryos from all 4 groups were sampled sequentially at 1, 2, 3, and 4 dpi. The sampling procedures for histopathology and bacterial quantification were as in experiment 1.
Histopathology
On the day of necropsy, fresh sections of yolk sac, liver, and heart were removed and preserved in 10% neutral-buffered formalin. Tissues were processed routinely and 5-µm sections were stained with hematoxylin and eosin, and analyzed under a light microscope. For yolk sacs, a lesion scoring scheme from 0 to 3 corresponding to normal, mild, moderate, and severe inflammation and pathological changes was established (Table 1, Figs. 1–4).
Description of Lesion Scores in Yolk Sacs of Chicken Embryos.

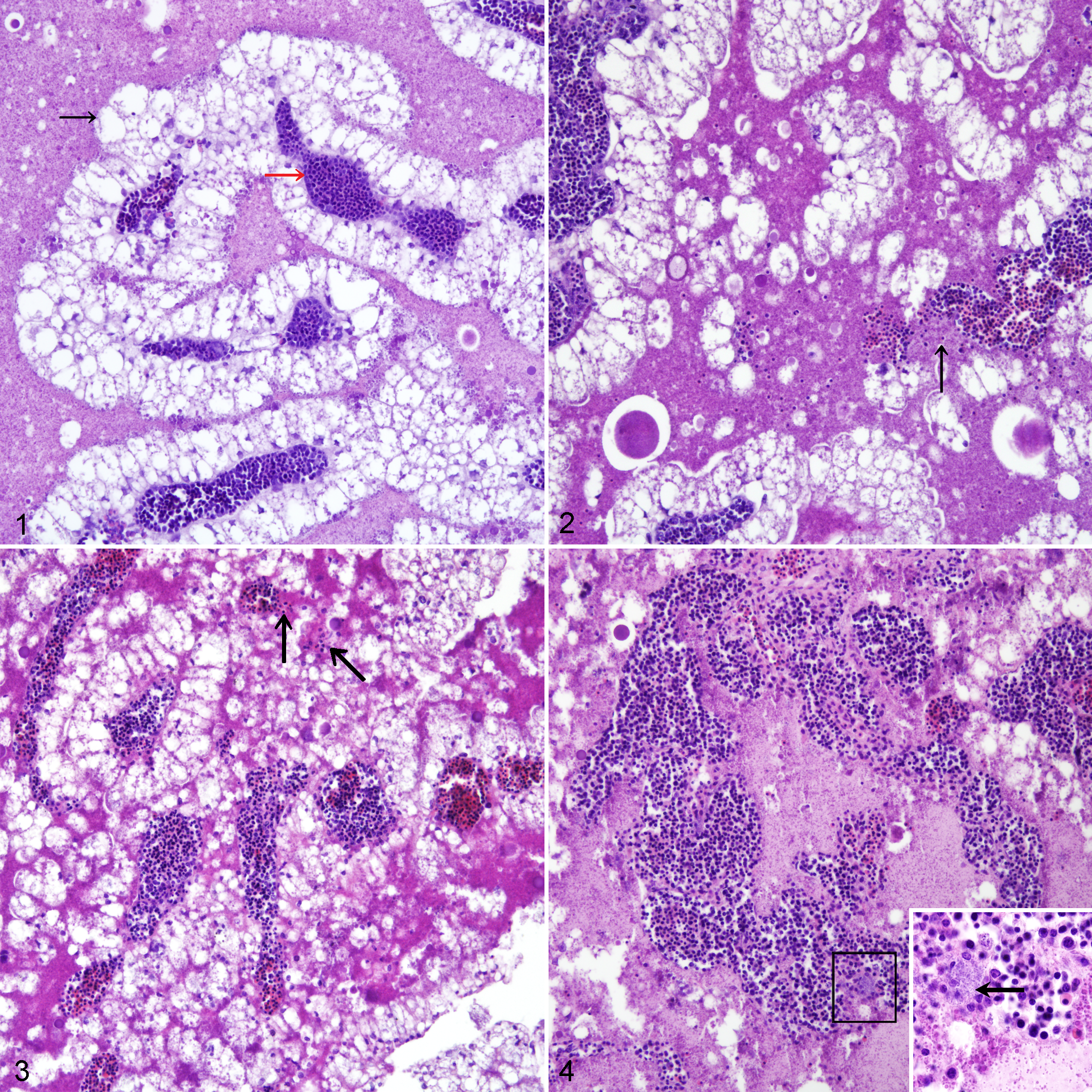
Experimental bacterial infection, yolk sacs, chicken embryos. Embryos were experimentally infected with E. coli PA14/17480/5-ovary, S. Enteritidis ATCC-13076, or E. coli 2018/SPF-cecum prior to histologic lesion scoring. Hematoxylin and eosin (HE).
CFU Count
One milliliter of yolk and 1 g of liver were separately collected during necropsy, homogenized with PBS, and serially diluted in tenfold steps. Suspensions from each dilution were plated in duplicate on MacConkey and XLD agar to quantify E. coli and S. Enteritidis, respectively. The homogenized samples from the negative control group were plated on both agar plates. The plates were incubated aerobically at 37 °C overnight. The total number of E. coli and S. Enteritidis in yolk and liver were expressed as log10 CFU/ml or log10 CFU/g.
Statistical Analysis
In experiment 1, Kaplan-Meier survival curves were plotted based on the cumulative mortality of embryos and evaluated with the log-rank test. Differences in CFU counts in the yolk and liver as well as microscopic lesion scores in yolk sacs from dead and live embryos within a group were analyzed with the t test. Comparisons between live and dead embryos were only possible in groups 1 and 3 as all the embryos in group 2 died due to infection and all the uninfected negative controls in group 4 survived until the end of the experiment. In experiment 2, mean log CFU counts in yolks and livers in different groups at different time points of sampling were analyzed by multiple comparisons with Tukey HSD. The Shapiro-Wilk test was used to test the normality of data. All statistical analysis was performed in Statistical Package for the Social Sciences (IBM SPSS version 24; IBM Corporation). P values below .05 were considered as statistically significant.
Results
Embryo Mortality
In experiment 1, the total mortality rates in the 5-day observation period in groups 1, 2, 3, and 4 were 53%, 100%, 27%, and 0% (Fig. 5). The highest mortality was observed at 2 dpi when 6/15, 10/15, and 3/15 embryos from groups 1, 2, and 3 died. The survival rates of the embryos in all infected groups were significantly lower than that of those in the negative control group, which had no mortality (Fig. 5). In pairwise comparison, survival rates in all groups were significantly different to each other except between groups 1 and 3.
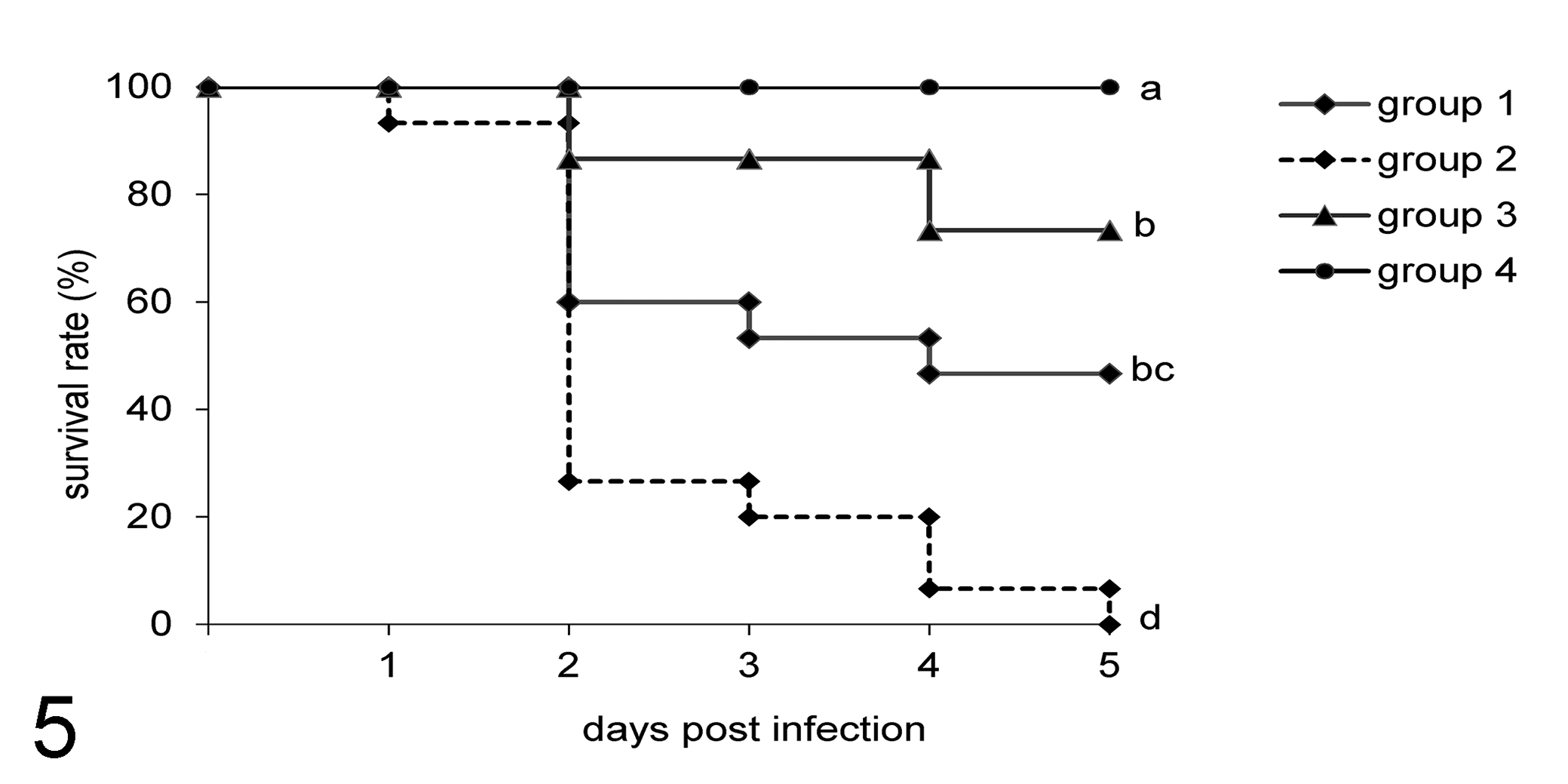
Survival of embryos following experimental infection with different bacteria (experiment 1). Groups containing 15 embryos each are as follows: 1, infected with E. coli PA14/17480/5-ovary; 2, infected with Salmonella Enteritidis ATCC-13076; 3, infected with E. coli 2018/SPF-cecum; 4, uninfected negative control. Differences among groups were analyzed with the log-rank test. Different letters denote statistically significant differences. P < .05.
In experiment 2, live and dead embryos were sampled sequentially at 1, 2, 3, and 4 dpi as shown in Table 2. The mortality rates in infected groups were maximal at 2 dpi. No mortalities were observed in the negative control group (group 4) (Table 2).
Number of Embryos Sampled and Mean Lesion Scores Following Experimental Infection With Different Bacteria in Experiment 2.
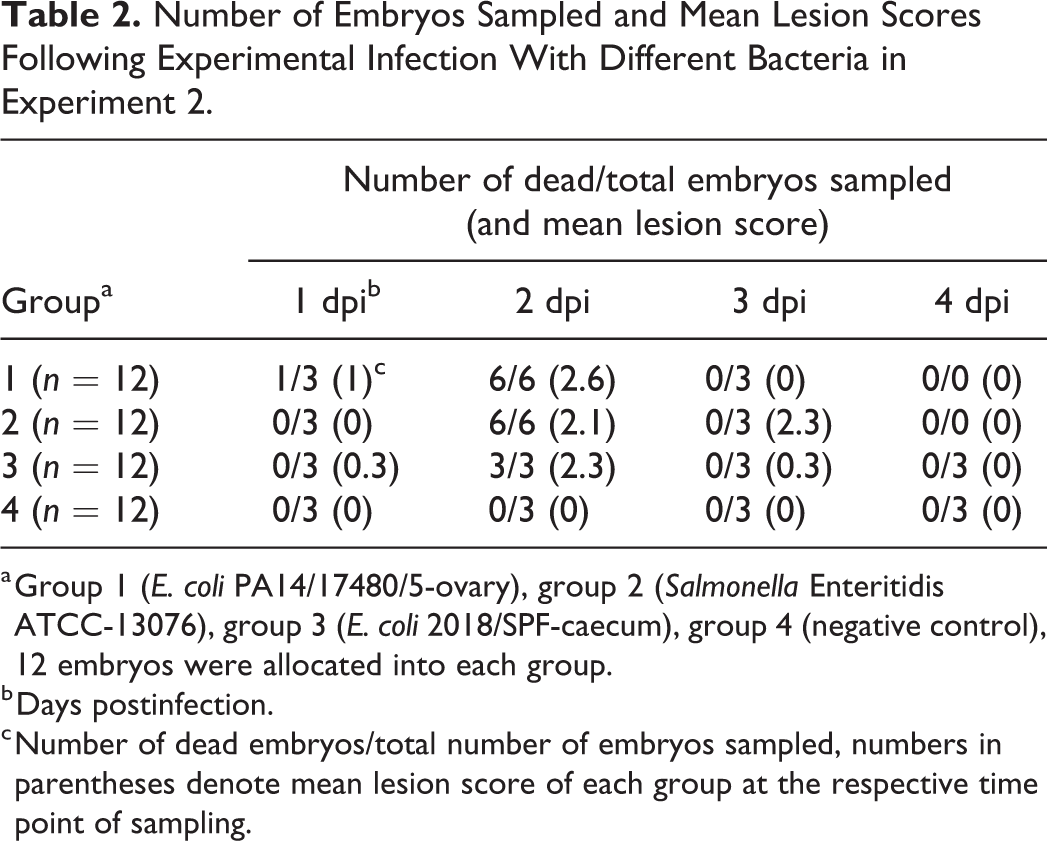
a Group 1 (E. coli PA14/17480/5-ovary), group 2 (Salmonella Enteritidis ATCC-13076), group 3 (E. coli 2018/SPF-caecum), group 4 (negative control), 12 embryos were allocated into each group.
b Days postinfection.
c Number of dead embryos/total number of embryos sampled, numbers in parentheses denote mean lesion score of each group at the respective time point of sampling.
Macroscopic Lesions
The negative control groups had no pathological changes in the yolk sacs and yolk contents from any embryos in either experiment (Figs. 6, 7). In infected live and dead embryos, the yolks were turbid. The contents from dead embryos had an intense smell. The gross lesions did not differ in embryos infected with different bacteria, although we did see differences between dead and live embryos in general. Dead embryos showed signs of congestion and were diffusely reddened all over the skin, head, and neck (Fig. 8). The yolk sacs of these embryos were hyperemic, with engorged blood vessels and black foci indicative of necrosis were present (Fig. 9).
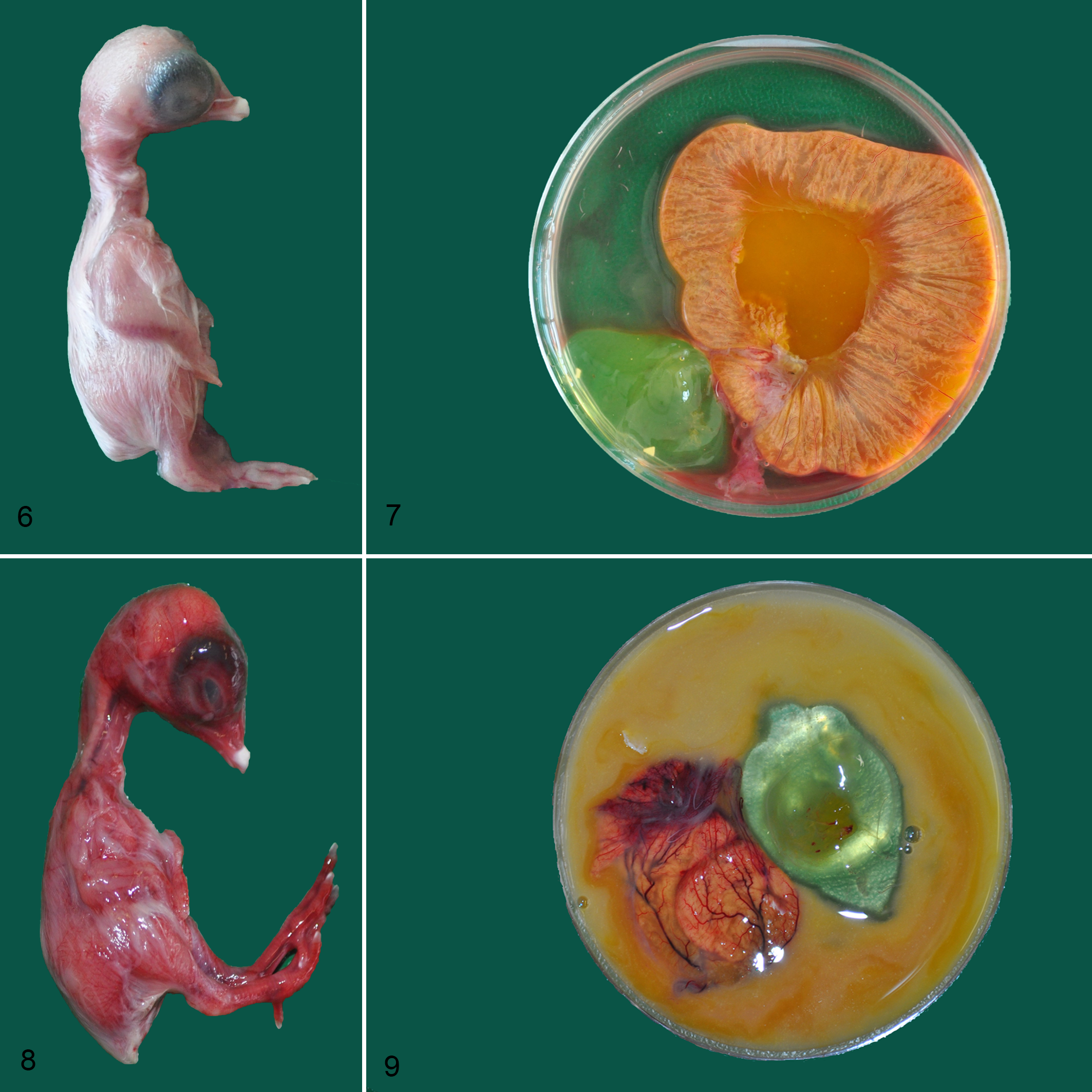
Experimental bacterial infection, yolk sacs, chicken embryos.
Microscopic Lesions
Healthy yolk sacs contained larger, round endodermal epithelial cells with peripherally located nuclei. Blood vessels remained intact and the yolk content was clear without any blood cells or cell debris (Fig. 1).
Infection of embryos with E. coli or S. Enteritidis led to varying degree of microscopic lesions in yolk sacs (Figs. 2–4). These lesions were characterized by congestion, inflammation, damaged blood vessels, and smaller endodermal epithelial cells with loss of normal structure and degeneration, along with the presence of numerous bacteria in the intravascular and luminal regions. Numerous lysed erythrocytes were also apparent.
In experiment 1, the embryos from the negative control group and the surviving embryos from groups 1 to 3 showed no histological lesions in the yolk sac until the end of the observation period. Independent of the bacteria used for infection, we observed lesion scores of 1, 2, and 3 in 3, 14, and 11 dead embryos, respectively (Fig. 10). The differences in lesion scores in yolk sacs between dead and live embryos in groups 1 and 3 were statistically significant.
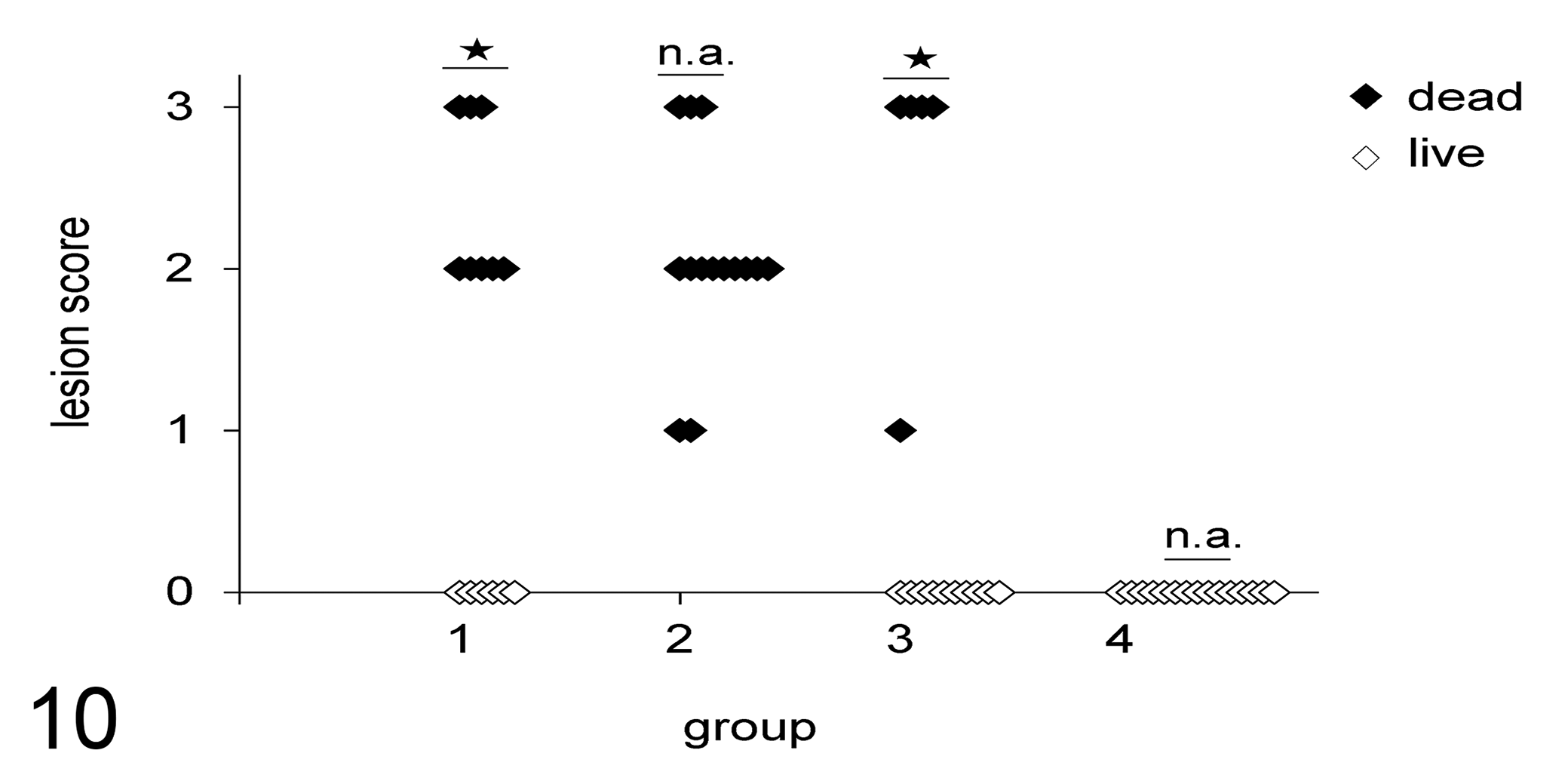
Lesion scores in yolk sacs of live and dead embryos following experimental infection with different bacteria (experiment 1). Groups are as follows: 1, infected with E. coli PA14/17480/5-ovary; 2, infected with Salmonella Enteritidis ATCC-13076; 3, infected with E. coli 2018/SPF-cecum; 4, uninfected negative control. For lesion scoring scheme, refer to Table 1 and Figures 1 to 4. Lesion scores between live and dead embryos within a group were analyzed by t test, and statistically significant differences (P < .05) are denoted by stars. n.a., not applicable.
In experiment 2, no lesions were found in embryos from the negative control group at any time point of the experiment. Independent of the bacteria used in this experiment, we observed scores of 0 (n = 14), 1 (n = 3), or 2 (n = 1) in the yolk sacs of infected embryos killed on the day of sampling, whereas dead embryos had lesion scores of 1 (n = 1), 2 (n = 9), or 3 (n = 8). The highest mean histological lesion scores were observed at 2 dpi, when embryo mortality was at its peak (Table 2).
The microscopic observations in livers of dead embryos from both experiments showed an increased gap between hepatic plates, congestion of blood vessels and sinusoids, a profound increase in the numbers of lysed erythrocytes, and the presence of bacterial colonies (Fig. 11). Numerous bacteria were present in the heart of dead embryos and low numbers of lysed erythrocytes were seen in the myocardium (Fig. 12). No other histopathological changes were observed.
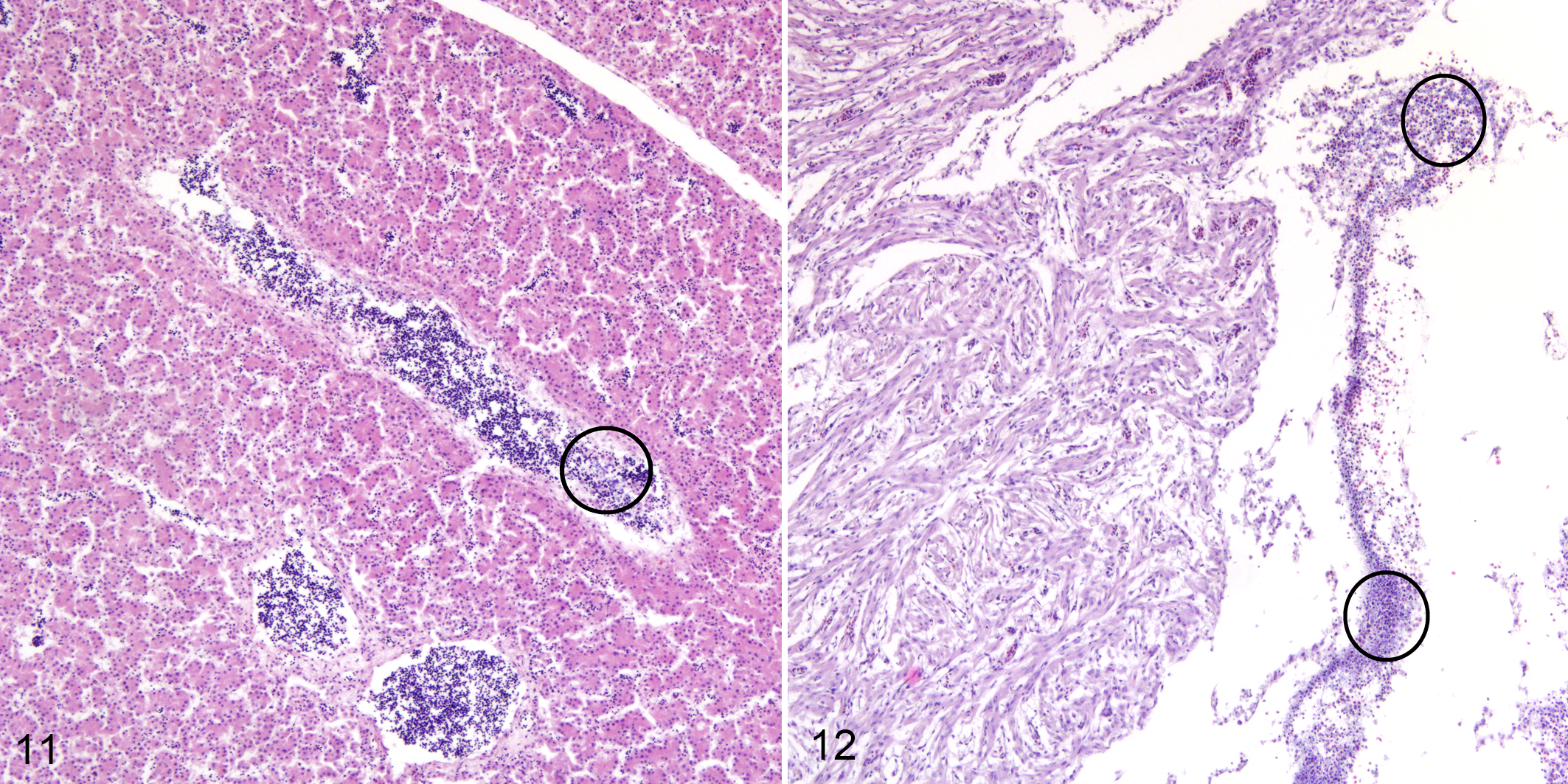
Experimental bacterial infection, chicken embryos.
Bacterial Quantification
The results of CFU counts in yolk and liver samples in experiments 1 and 2 are shown in Figures 13 to 16. In experiment 1, we found no significant differences in bacterial load in the yolks of live and dead embryos in groups 1 and 3, following infection with E. coli. However, the number of bacteria in the livers of dead embryos was significantly higher than in the livers of those that survived infection (Fig. 13, 14). In the second experiment, all 3 bacteria rapidly multiplied within 24 hours post infection in the yolks and livers of all infected embryos. There were no differences in bacterial load in the yolks of the infected groups at 1 and 2 dpi, but at 3 dpi differences were significant with group 2 having the highest mean CFU count. Similarly, the average bacterial count in the livers of group 2 was significantly higher than that in other groups at 3 dpi (Fig. 15, 16).
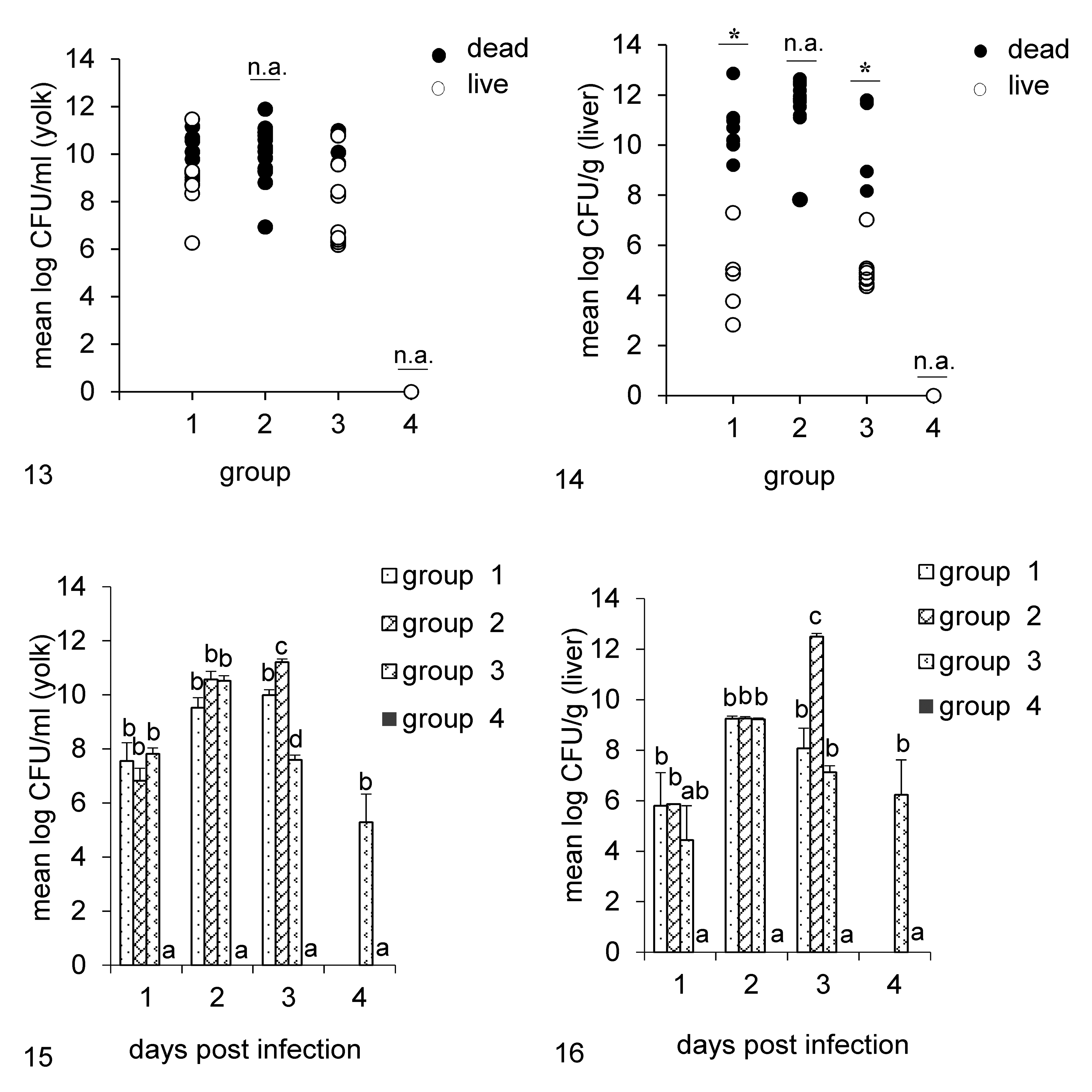
Bacterial quantification following experimental infection of embryos with different bacteria. Groups are as follows: 1, infected with E. coli PA14/17480/5-ovary; 2, infected with Salmonella Enteritidis ATCC-13076; 3, infected with E. coli 2018/SPF-cecum; 4, uninfected negative control.
Discussion
Infection of hatching eggs with bacterial pathogens, through vertical transmission or contaminated incubators, might result in death of embryos, lower hatchability, or poor performance of chicks. These effects are frequently reported in the field and can be reproduced under experimental conditions. 12,20,29 Different bacterial isolates might differ in the ability to kill embryos, based on their virulence potential, which is a fundamental principle of embryo lethality assays. 3,28,37 To understand the pathological mechanism of embryonic death following bacterial infections, we investigated the pathological and bacteriological changes in embryonic and extra-embryonic tissues of dead and live embryos in 2 experimental settings. The first experiment determined the cumulative mortality of embryos due to different bacteria, while the second was intended to observe changes at 24-hour intervals following infection. The selected E. coli strains represented an APEC and a commensal E. coli pathotype on the basis of the clinical condition of the host from which isolates were obtained. 24 The reference strain of S. Enteritidis was also used to test for any species-specific outcomes and to extend the range of bacteria. We observed differences in the pathogenicity of the different bacteria, with the highest mortality rates related to S. Enteritidis, followed by the APEC strain and the commensal E. coli strain.
The CFU counts in the 2 experiments revealed that all the bacterial species were able to rapidly multiply in yolks and livers. However, no significant differences were observed in the bacterial load in yolk. Thus, the growth of bacteria in yolk is not the determining factor for the death of the embryo, which is in agreement with previous findings. 21,28 A higher bacterial load in the livers of dead embryos might have been the consequence of severe lesions in the yolk sacs, which lost their barrier function.
We did not find any differences among bacteria with respect to the resulting macroscopic or microscopic lesions. However, we did find differences in the severity of lesions between dead and live embryos, irrespective of the type of bacteria used for infection. Macroscopically, we observed hemorrhages and congestion in dead embryos and their yolk sacs. Histopathological examination of embryonic and extra-embryonic tissues revealed that the yolk sacs of dead embryos were most affected, with damage of blood vessels and endothelial cells. Interestingly, no embryo that remained alive until the end of experiment 1 showed any lesions. The results of experiment 2 were very similar.
Previous work has shown that yolk sacs are crucial for the normal development of chicken embryos: they support or replace the function of several organs that have not reached full functional capacity in the embryo. Of major importance, endocytotic receptors such as LR8 14 and a trimeric LRP2-cubulin-amnionless complex 2 in yolk sacs are involved in the uptake of VLDL and vitellogenin. In addition, the yolk sacs of chicken embryos also express genes whose products are involved in the production of other nutrient transporters, digestive enzymes, β-defensins, and FcRY receptors for the transfer of maternally derived immunoglobulin Y from egg yolks to the embryonic circulation. 33,34,38 –40 The selective suppression of some of these nutrient receptors in rabbits and mice coincides with compromised embryonic development. 9,35,36 The function of yolk sacs during embryonic development is not limited to nutrient absorption and immune mechanism: yolk sacs also serve as the major site of hematopoiesis. 13,38 In light of these vital functions, the pattern of microscopic lesions we observed in yolk sacs makes it seem likely that essential physiological mechanisms were compromised in dead embryos by the disruption of endothelial cell organization, defects in receptor pathways, or poor expression of essential gene clusters in endodermal sites of yolk sacs, and that these are the most probable causes of embryonic mortality. Although the health of the yolk sac has been largely neglected, it seems to be crucial for the survival of embryos after bacterial infections. Deprivation of nutrients and the poor absorption of yolk might also have clinical consequences in surviving chicks, even after hatching. Previous work showed that chicks that hatched from eggs experimentally infected with E. coli had lower weight gain, prolonged absorption of yolk, and an increased yolk to body weight ratio. 12,20,29
Microscopically, the livers and hearts of dead embryos were congested but we did not see any necrotic changes in the hepatic and myocardial cells. All dead embryos contained a very large number of lysed erythrocytes, although we cannot exclude the possibility of autolysis of dead embryos before fixation in formalin. Based on the large numbers of bacteria in the blood vessels of yolk sacs, we can hypothesize that the bacteria reached the liver and the heart through the circulation.
In conclusion, damage to yolk sac tissues due to bacterial infections correlated with embryonic mortality. The results underline the importance of healthy yolk sacs for the survival of embryos and show the need for further research on this organ, which has previously been neglected despite its physiological importance.
Footnotes
Acknowledgements
The authors thank Dr Alexander Tichy for help with statistical analysis and Dr Graham Tebb for proofreading of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
