Abstract
The peripheral nervous system (PNS) relays messages between the central nervous system (brain and spinal cord) and the body. Despite this critical role and widespread distribution, the PNS is often overlooked when investigating disease in diagnostic and experimental pathology. This review highlights key features of neuroanatomy and physiology of the somatic and autonomic PNS, and appropriate PNS sampling and processing techniques. The review considers major classes of PNS lesions including neuronopathy, axonopathy, and myelinopathy, and major categories of PNS disease including toxic, metabolic, and paraneoplastic neuropathies; infectious and inflammatory diseases; and neoplasms. This review describes a broad range of common PNS lesions and their diagnostic criteria and provides many useful references for pathologists who perform PNS evaluations as a regular or occasional task in their comparative pathology practice.
Keywords
Neuropathology of the peripheral nervous system (PNS) is an underappreciated area in comparative pathology. Despite its widespread distribution within the body, in the absence of clinical signs associated with PNS disease ganglia and nerves are seldom included in the list of organs and tissues collected and processed for histopathologic evaluation in either diagnostic or experimental pathology settings. Accordingly, many comparative pathologists have limited familiarity with the normal structures, specialized processing needs, and range of common lesions that might be encountered when analyzing the PNS.
This lack of familiarity led to the decision to hold a half-day symposium on the “Peripheral Nervous System” as one of the educational sessions at the 2019 annual meeting of the American College of Veterinary Pathologists (ACVP). The session was co-chaired by 2 neuropathologists, while speakers were chosen for their experience with neuropathology in the diagnostic pathology, comparative pathology, and toxicologic pathology arenas. Attendance at each talk ranged from 150 to 200 attendees, indicative of a robust interest in the comparative pathology of PNS diseases. For this reason, the symposium team has produced this concise but data- and reference-dense review of important principles of anatomy, tissue sampling and processing, and lesion identification and interpretation regarding the PNS.
Biology of the PNS
The PNS represents the outlying offshoots of the central nervous system (CNS) that allow centers in the brain and spinal cord (CNS) to exert control over organs and processes throughout the body. The distinction between the CNS and PNS is pragmatic rather than absolute since the integration of the 2 systems transcends neuroanatomical boundaries. 113 There is truly only one interdependent nervous system. Nonetheless, our appreciation of the nervous system is facilitated by seeing the structures and functions of the PNS and CNS as anatomically distinct entities.
Unlike CNS diseases, where neuropathological sequelae are impacted to some degree by neuroanatomical, neurobehavioral, and neurochemical divergence among species and strains, across sexes, and at different ages, 14,217 the PNS exhibits a relatively consistent anatomical organization and functional capacity in all vertebrates. The PNS is composed of ganglia (collections of neurons) and nerves that present information from peripheral receptors to the CNS. Signals may be differentiated based on the type of input they convey. Somatic information consists of consciously perceived sensory and motor (sensorimotor) signals typically originating from widely distributed sensory receptors or passing to large muscle masses. Autonomic information occurs as unconsciously received input from 3 divisions. 61,130 The autonomic parasympathetic division is a slower-acting network that tends to dampen basal activities. The autonomic sympathetic division is a quick-reacting network that mobilizes organs for action (often termed a “fight or flight” response) in the face of acute or prolonged stress. The enteric nervous system is a quasi-independent division of the autonomic nervous system that coordinates parasympathetic-like functions in digestive organs.
Basic PNS Anatomy
The neural net comprising the vertebrate PNS consists of many far-flung branches, including bilaterally paired cranial nerves that connect to the brain (cranial nerves [CN] I-X, XII) and cranial spinal cord (CN XI) as well as paired spinal nerves that project from specific spinal cord levels. 46,58,177,238,250 The first cranial nerve (CN I, the olfactory nerve) originates as widely dispersed axons arising from bipolar sensory neurons residing in the olfactory neuroepithelium, which lines the ethmoid turbinates in the dorsocaudal nasal cavity. The olfactory neurons are sustained by sustentacular cells in the olfactory mucosa, while their unmyelinated axons are encompassed by olfactory ensheathing cells (a site-specific glial cell similar to Schwann cells in other nerves). The second cranial nerve (CN II, the optic nerve) arises as axons projecting from the ganglion cell layer near the inner surface of the retina. This nerve is unique in that developmentally it represents a part of the CNS, and not the PNS; the glial cells in the optic nerve are astrocytes and myelin-producing oligodendrocytes, and the nerve is enveloped in meninges that are continuous with those covering the brain. The other 10 cranial nerves and the spinal nerves are all PNS components since their neurons, located within specific nuclei within the CNS, are neural crest derivatives and the myelin sheaths of PNS nerve fibers are produced by Schwann cells.
Ganglia in the PNS (Fig. 1) are composed of neurons of diverse sizes and with different functions, satellite glial cells, and variable numbers of fibroblasts (interspersed in collagen), macrophages, mast cells, and melanocytes. 39 The satellite glial cells are thought to fulfill astrocyte-like functions for the neurons they encircle, such as giving metabolic support, environmental control, and neurotransmitter clearance. The ganglia are surrounded by a capsule of connective tissue. Nerves are composed of bundles of axons (neuronal processes) that are insulated by thick (for “myelinated”) or thin (for “unmyelinated”) layers of myelin; thicker myelin sheaths permit more rapid transmission of signals (Fig. 2). Nerve fibers are encompassed by 3 layers of fibrous connective tissue: endoneurium, a fine mesh that separates individual nerve fibers; perineurium, which bundles multiple nerve fibers into one of several fascicles within the nerve; and epineurium, which is a dense sheath that surrounds the entire nerve. 243 Nerves may contain many other cell types including capillary endothelial cells, fibroblasts, and occasional lymphocytes (especially in nonhuman primates), macrophages, and mast cells (particularly in rodents). The composition of major spinal nerves varies among species and across strains. 19
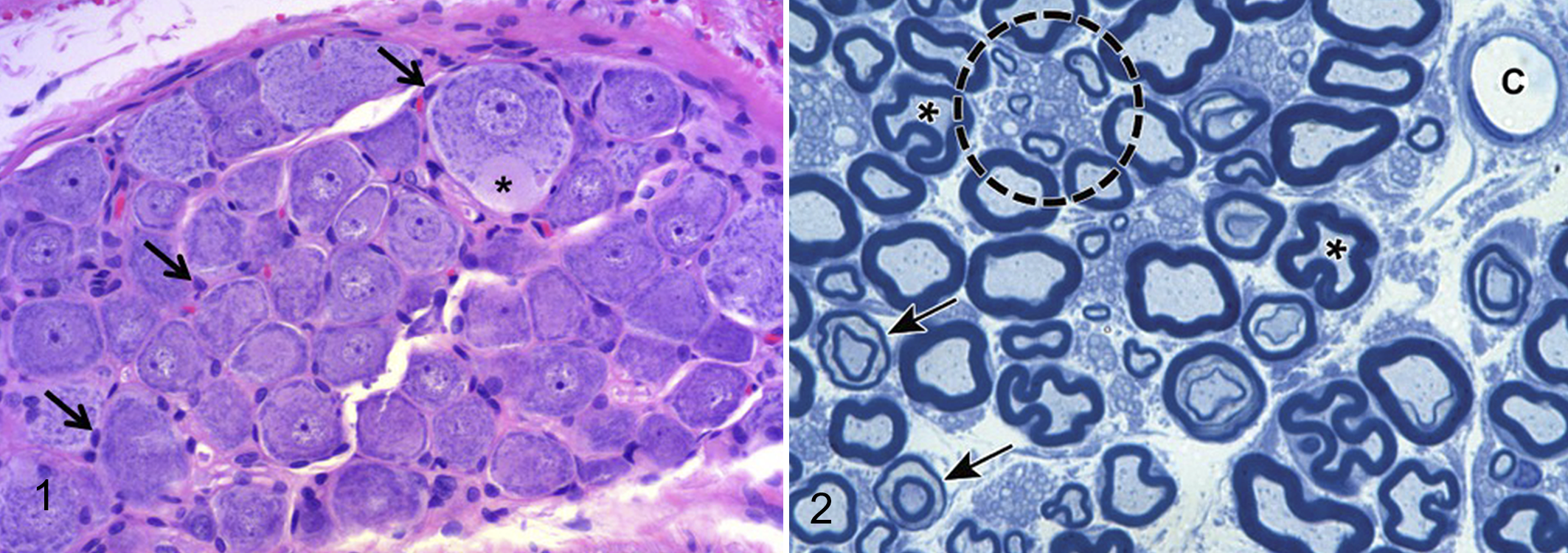
Normal thoracic dorsal root ganglion (DRG), adult Sprague-Dawley rat. The ganglion contains large and small sensory neurons as well as supporting satellite glial cells (arrows). One large neuron exhibits an axon hillock (asterisk), which may be identified by the absence of Nissl substance (ie, absence of dark basophilic cytoplasmic granules) at the location where the axon projects from the cell. Hematoxylin and eosin (HE).
The cells that give origin to somatic sensory nerve fibers in the PNS are located outside the CNS, especially in the sensory ganglia of cranial nerves and the dorsal root ganglia (DRG) for spinal nerves (Fig. 3). 113 The key sensory ganglia for cranial nerves are the trigeminal (or Gasserian * ) ganglion, for the trigeminal nerve (CN V); the geniculate ganglion for the facial nerve (CN VII); the spiral and vestibular ganglia (embedded in the labyrinth of the inner ear) for the vestibulocochlear nerve (CN VIII); and the dorsal/superior (or jugular) and caudal/inferior (or nodose) ganglia of the vagus nerve (CN X). 46,177,238,250 The sensory neurons in DRG have long processes, one extending distally into a nerve and the other passing centrally through the dorsal spinal nerve root to enter the spinal cord. These DRG sensory fibers may ascend within the white matter as primary sensory spinal tracts located in the dorsal funiculus (or “dorsal columns”); 121 the 2 primary collections of ascending sensory nerve fibers within this funiculus comprise the fasciculus gracilis, located near the midline and carrying fibers from spinal nerves serving the hind limb, and the fasciculus cuneatus, located laterally (just medial to the dorsal horn of the spinal cord gray matter) and carrying input from the forelimb-associated spinal nerves. 14,46,113 Some sensory fibers from the DRG instead provide input to interneurons in the spinal cord gray matter, which respond by sending new signals to peripheral effector organs (to complete reflex arcs) or higher brain centers (eg, within the spinocerebellar tracts of the spinal cord white matter). The only pure motor nerves are the ventral spinal nerve roots. These structures carry somatic motor fibers from the neurons in the ventral horns of the spinal cord gray matter for each body segment. The ventral spinal nerve roots merge with sensory nerve fibers just ventral and distal to ganglia to form larger nerve trunks, which pass to the periphery as mixed nerves containing both sensory and motor fibers. Some distal nerves are “sensory predominant” in that they contain mainly sensory elements. The locations and nomenclature of sensory-predominant nerves vary with the species. 19
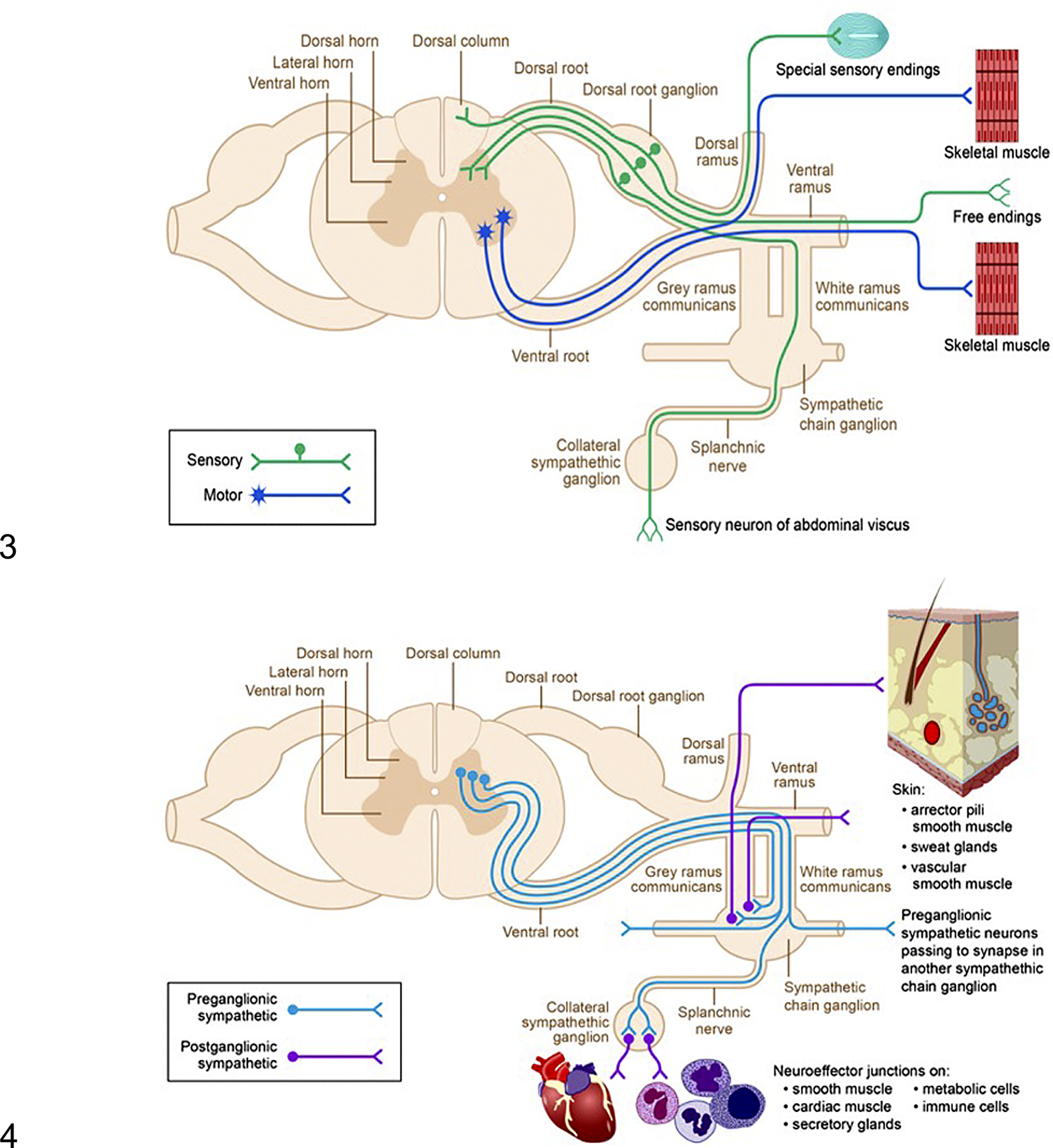
The neural circuitry of the somatic peripheral nervous system. Sensory nerve fibers (green) originate from peripheral receptors, extend to sensory neurons that reside in dorsal root ganglia, and then travel through dorsal spinal nerve roots to enter the dorsal (or posterior) column of the spinal cord. Motor nerve fibers (blue) originate from motor neurons in the ventral horn gray matter of the spinal cord, leave via the ventral spinal nerve roots, and then synapse on effector organs in the periphery.
The 2 conventional components of the autonomic nervous system are the parasympathetic and sympathetic divisions. 61,63,113,130,191 These divisions utilize a 2-neuron circuit, where axons of preganglionic neurons in the CNS synapse with postganglionic neurons in peripheral ganglia (Fig. 4). In the brain, preganglionic neurons for the parasympathetic and sympathetic divisions are localized in brain nuclei associated with the cranial nerves. In the spinal cord, parasympathetic fibers arise from preganglionic neurons in gray matter of the sacral portion of the spinal cord, while sympathetic fibers project from preganglionic neurons in the gray matter zona intermedia in the thoracolumbar spinal cord; a recent report suggests that many autonomic fibers extending from the sacral spinal cord support a sympathetic function. 56 Postganglionic sympathetic neurons are confined to ganglia typically located at a distance from the organs they control (Fig. 4), while postganglionic parasympathetic neurons form ganglia inside the walls of their target organs. The enteric division of the autonomic system, which has a parasympathetic-like function, consists of a complex web-like mesh of linked ganglia within the walls of the digestive tract. 68 The enteric nervous system can function independently of CNS control.
Axons in the PNS are ensheathed by different amounts of myelin produced by myelinating Schwann cells. Myelin is a multilayered insulating cuff containing 70% lipid (cholesterol, galactosphingolipids such as cerebrosides, and phospholipids) and 30% of numerous myelin-specific proteins including myelin basic protein (MBP), myelin-associated glycoprotein (MAG), protein 0 (P0), protein 1 and 2 (P1, P2) and peripheral membrane protein 22 (P22), and proteolipid protein (PLP). The protein profiles of CNS and PNS myelin overlap but are not identical; some proteins are enriched in CNS myelin (eg, PLP), some in PNS myelin (eg, P0), and some occur in both CNS and PNS myelin (eg, MBP). 204 Recently, ectopic PNS-derived Schwann cells have been reported (in horses and cattle) to enter and proliferate within the CNS as a nonneoplastic, congenital developmental defect. 153 One Schwann cell envelops a single axon, which contrasts to the arrangement in the CNS where one oligodendrocyte myelinates up to 50 axons. An individual Schwann cell forms a unit called an “internode” that covers on average about 1 to 1.5 mm of an axon’s length. The nodes of Ranvier are narrow gaps of unmyelinated axon that exist between 2 adjacent internodes (Schwann cells). A proportional relationship exists between the axon diameter and myelin thickness. Larger axons have thicker myelin sheaths, while very small “unmyelinated” axons are covered by a single layer of Schwann cell membrane. All nerve fibers are encompassed by a basement membrane that is produced by the Schwann cell. However, regenerated peripheral nerves fibers have decreased conduction velocity because the thickest axons are thinner than normal, their myelin sheaths are relatively thin, and the length of internodes is shorter (which results in more nodes of Ranvier than usual). Perineurial cells comprise the other significant cellular contribution to the peripheral nerve sheath. Like Schwann cells, they are neural-crest derived and may actually represent modified Schwann cell progenitors. 123,180 They are often involved with reactive lesions of the peripheral nerve, including traumatic injury.
The blood-nerve barrier (BNB) is a highly selective, semipermeable barricade that limits passive diffusion while facilitating active transport of key molecules. The BNB is composed of endoneurial vascular endothelium and a multilayered sheath of perineurial connective tissue. 185,186,252 The BNB includes tight junctions but is not as tight as the CNS blood-brain barrier (BBB), especially in DRGs, 207 due to absence of the rich bed of astrocyte foot processes that add an additional layer to the BBB; therefore, the BNB is relatively more susceptible to penetration and damage by xenobiotics and infectious agents than is the BBB. 188 For this reason, some authors suggest that the term “blood-nerve interface” should be used in place of the BNB since a porous interface more accurately reflects the dynamic regulation of vascular permeability in ganglia and nerves. 158,252 The epineurium contains lymphatic vessels, but nerve cores do not possess lymphatic capillaries. 2,247 The lack of lymphatic drainage in the endoneurial space and the presence of myelinating Schwann cells, which can act as facultative antigen-presenting cells and phagocytes, further bolsters the degree of protection afforded by the BNB.
When evaluating the PNS, a key factor in the assessment is to be able to discriminate unusual but normal structures from lesions. 175 For example, regularly spaced, chevron-shaped interruptions in the myelin sheath—termed myelin (Schmidt-Lanterman) incisures—may be observed along the internode in paraffin-embedded nerve sections (Fig. 2). These incisures arise as small foci of cytoplasm left behind during myelination, and they are postulated to regulate structural stability during myelin formation and modulate signal transduction. 235 Other structures observed in association with nerves or nerve termini in routine paraffin sections include receptors (laminar [or Pacinian] corpuscles, muscle spindles); ectopic neurons; axon hillocks of neurons (Fig. 1); and variable Schwann cellularity and myelin density between somatic and autonomic nerves. Intracytoplasmic inclusions in Schwann cells (eg, mu [or Elzholz] and pi [or Reich] granules) have been described by light microscopy and/or transmission electron microscopy in nerves embedded in hard plastic resin. 65,146 Renaut bodies appear as large, loosely whorled, elongated connective tissue accumulations in somatic and occasionally autonomic nerves of humans, dogs, and horses, seemingly as a response to sustained mechanical trauma (eg, nerve entrapment) or pressure. 54,69,229,251 Certain artifactual changes (eg, DRG neuronal vacuolation) and incidental background changes (eg, DRG neuronal autophagy) also must be appreciated as being “within normal limits.” 175 Pathologists unfamiliar with the normal anatomy of ganglia and nerves should consult atlases of PNS pathology 115,170,175,213 to ensure that they do not inadvertently diagnose normal features or artifacts as evidence of damage to PNS neurons, axons, or myelin.
Basic PNS Physiology
The somatic (or sensorimotor) PNS is responsible for gathering sensory input and directing effector organs that undertake voluntary functions. Somatic activities are mediated by higher brain centers in the cerebral cortex and cerebellum. The autonomic nervous system controls effector organs that perform involuntary homeostatic functions, with the slowly responding parasympathetic elements (including enteric nervous system) regulating basal (“resting”) activities and the rapidly reacting sympathetic components mediating reactions to immediate (“fight or flight”) or prolonged challenges. 67,191 The parasympathetic and sympathetic divisions typically (but not exclusively) serve opposing or compensatory functions for tissues that are innervated by both components, but an important fact to remember is that the parasympathetic division serves sites only of the head, viscera, and external genitalia while the sympathetic division is dispersed to nearly all tissues of the body. Autonomic activities are mediated by brain centers concentrated in the hypothalamus and brain stem.
Nerve fibers in the PNS are sorted into A (with 2 subtypes), B, and C classes based on their conduction velocities and diameters (Table 1). Except for their size, the structure and composition of large-diameter and small-diameter axons is similar. 243 Large-diameter fibers have bigger axons, thicker myelin sheaths, conduct nerve impulses more quickly, and typically serve somatic functions. Axons in the PNS have an 8-nm-thick axolemma (cytoplasmic membrane) that surrounds a cylinder of axoplasm (cytoplasm) containing many organelles, including cytoskeletal proteins (“neurofibrils”), mitochondria, and smooth endoplasmic reticulum. The collective term “neurofibrils” includes both neurofilaments, which are responsible for maintaining the axon diameter, and microtubules, which are tasked with controlling the radial growth and stability of axons and intracellular transport. Neurofilaments (NFPs) are neuron-specific, 10-nm-diameter intermediate filaments composed of 3 peptide subunits with low (68 kDa), medium (150 kDa), and high (200 kDa) molecular weights. Microtubules are composed of tubulin protein dimers arranged in hollow-core cylinders that measure 25 nm in diameter and are 10 to 20 µm long; microtubules subunits may be phosphorylated as they are exported into the axon, thereby speeding axonal transport. Another neuron-specific intermediate protein, peripherin, is a 58 kDa component of large-diameter axons, and it is upregulated during nerve regeneration. The axons lack protein-synthesizing machinery and thus rely on constant access to the cell’s Nissl substance (ie, dense aggregates of rough endoplasmic reticulum), which is found in the neuronal perikaryon and dendrites. The impact of this dependence is that separating an axon from the neuronal cell body prevents raw materials for maintenance and renewal from reaching the distal extremities of the axon. A corollary of this vulnerability is that larger and longer axons, which have the greatest metabolic demands, tend to be more susceptible to damage.
Physiological Attributes of Nerve Fibers in the Peripheral Nervous System.

Delivery of material from the cell body to the far reaches of the PNS and back is accomplished via 2 axonal transport mechanisms. Anterograde or proximodistal axonal transport (from the cell body to the axon terminus) has both rapid and slow components. Rapid anterograde transport carries critical metabolic substrates and neurotransmitters at rates up to 400 mm/day using microtubules and kinesin. Slow anterograde transport bears structural proteins like microtubules and neurofilaments at rates of 1 to 4 mm/day utilizing dynamin, a microtubule-associated motor protein, as well as actin, clathrin, and others. Retrograde or distoproximal axonal transport (from the axon terminus to the cell body) moves materials back to the cell body for recycling or degradation at rates up to 200 mm/day using dynein (microtubule-associated protein 1C). Common substances slated for recycling include neurotrophic substances, such as nerve growth factor (NGF, a key factor for sensory and sympathetic neuron development) and ciliary neurotrophic factor (CNTF, a motor neuron survival factor); these molecules also appear to function as “retrophins,” providing support for neuronal processes. Retrograde axonal transport contributes to neural invasion by viruses (eg, enterovirus, herpesvirus, rhabdovirus) from the terminal nerve endings to the cell body.
The neurochemistry of the PNS is specific to the function of the pathways. 219 Somatic nerve fibers utilize acetylcholine as their neurotransmitter, and the effector organs they control (eg, skeletal muscles) use nicotinic-type cholinergic receptors. In the autonomic system, preganglionic neurons from both the parasympathetic and sympathetic divisions employ acetylcholine as their neurotransmitter, and the postganglionic neurons they stimulate harbor nicotinic-type cholinergic receptors. However, postganglionic neurons and target organs for these 2 autonomic divisions differ in their neurochemistry. The postganglionic parasympathetic cells still use acetylcholine as their neurotransmitter, but their target organs have muscarinic-type cholinergic receptors. The postganglionic sympathetic neurons utilize norepinephrine as their neurotransmitter, and their effector organs use α-type and β-type adrenergic receptors. The enteric circuits signal via a number of peptides including calcitonin gene-related peptide (CGRP), neuropeptide Y, substance P, and vasoactive intestinal polypeptide (VIP), to name only a few.
Clearance of neural debris from damaged nerves is undertaken by both resident (Schwann cells) and infiltrating (macrophages) elements, and chemical signaling plays an important role in this process. Schwann cells and their inherent functional plasticity are critical to both nerve fiber degeneration and repair. When confronted by nerve fiber damage, Schwann cells produce phosopholipase A2, which is capable of breaking down myelin within hours of nerve injury, as well as several cytokines (eg, interleukin-1α, tumor necrosis factor-α, monocyte chemotactic protein-1) that recruit leukocytes into the damaged nerve trunk. 142 Schwann cells also undergo a functional shift so that they can process and present myelin antigens to incoming lymphocytes; 78,151 this capability has been proposed as a mechanism for fomenting nerve-centered autoimmune diseases. Sometime after initial Schwann cell activation, circulating monocytes arrive at sites of nerve damage via the bloodstream and differentiate into macrophages. Both macrophages and Schwann cells are capable of phagocytosis, and thus contribute to the removal of nerve fiber debris. 142 Finally, with the loss of viable axons nearby, the Schwann cells will adopt a “repair” phenotype that is characterized by upregulated trophic factor expression and formation of solid Schwann cell columns (termed “bands of Büngner”) within the intact basal lamina of each injured nerve fiber to help guide axonal regeneration. 99,100 Pericytes have the capacity to secrete neurotrophic factors in vitro, 218 suggesting that they also might contribute to the nerve repair process.
Effective Sampling and Processing of the PNS
Appropriate collection and processing practices are critical to ensure that the morphology of cells and tissues in the PNS are suitable for histopathologic evaluation. Because the distinction between the PNS and CNS is purely conceptual, a purely peripheral neuropathy (ie, nerve lesions limited solely to PNS cells and nerve fibers) is rare. For this reason, an effective neuropathological assessment requires the practitioner to sample and evaluate multiple sites in both the CNS and PNS when investigating PNS diseases.
Recommended (“best”) practices have been defined for the collection, processing, and evaluation of PNS tissues for experimental studies, 18,19,36 and these same procedures should be suitable for diagnostic studies. In general, multiple spinal cord divisions (cervical and lumbar at minimum), ganglia (cervical and lumbar DRGs and the trigeminal [cranial nerve V] ganglion at minimum), and nerves (sciatic, tibial, and fibular) should be collected and preserved for potential analysis. For a complete PNS assessment, additional spinal cord levels (thoracic and sacral), ganglia (thoracic DRGs and sympathetic ganglia), and nerves (from the distal hind limb and the forelimb) also should be harvested. The rationale for such extensive sampling is that the animal is only available once for necropsy, so anything not gathered up front will be lost forever. The reason for sampling multiple nerves is that peripheral neuropathies develop in a length-dependent fashion, with lesions occurring first in the most distal branches. The common practice of examining only a mid-level sciatic nerve section and no DRGs is likely to miss all but the most advanced peripheral neuropathies.
The ideal means for preserving lipid-rich nervous system tissues is whole body perfusion with an aldehyde-based fixative followed by immersion postfixation with a lipid-stabilizing agent. 17,19,106 Perfusion fixation permits more rapid penetration of fixative into the vicinity of deeply buried ganglia and nerves. However, perfusion is often impractical due to its labor-intensive nature and low throughput. Accordingly, immersion fixation (of isolated specimens or—for rodents and small non-rodents—the basal skull, portions of limbs, or segments of vertebral column) is the standard practice for diagnostic and most experimental studies. Isolated ganglia usually are placed in cassettes while nerve segments typically are applied to thick paper (ie, card stock) to maintain a linear orientation during fixation; the natural “stickness” of the epineurial sheath allows the specimen to adhere to the card without the need for any other means of attachment. Suitable fixatives for PNS tissues by perfusion or immersion include conventional neutral-buffered 10% formalin (NBF, which contains about 1% methanol as a stabilizer to reduce formaldehyde cross-linking); 4% methanol-free formaldehyde (ie, 4% paraformaldehyde); and methanol-free mixtures of formaldehyde (2%) and glutaraldehyde (2.5%). Formaldehyde is chosen for most purposes since it penetrates more quickly, but glutaraldehyde is important for fixing neural tissues since it provides better cross-linking, especially of myelin components. 112 The chemistry of the fixative solution must be carefully adjusted so that the solution will preserve delicate neural tissues without inducing artifacts; the osmolarity should be mildly hypertonic (400–600 mOsmol), and the pH should approximate the physiologic range (7.2–7.5). 62 Postfixation of the PNS usually involves immersion of very small (1 mm3) blocks in 1% osmium tetroxide (“osmication”) for 2 hours. Osmium is categorized as an acute hazardous waste, so its use and disposal is regulated heavily in accordance with federal and state guidelines. In general, most laboratories use NBF for most applications involving the PNS. Since formaldehyde is cross-linked or oxidized (to formic acid) over time, NBF should be replaced if either a precipitate develops or the pH falls below 6.5—which typically transpires approximately 3 months after it is reconstituted from concentrated (37%) stock solutions. 40
Tissue trimming can make or break a PNS histopathologic analysis. When evaluating postmortem specimens, spinal cord and nerves generally should be examined in both cross and longitudinal (for nerve) or oblique (for spinal cord 15 ) orientations since nerve fiber damage often is most evident when long expanses of fibers may be viewed. Ganglia are assessed in longitudinal orientation since this plane reveals a greater number of ganglionic neurons and supporting glial cells. The 2 most common mistakes made in trimming the nervous system when investigating a peripheral neuropathy are to not include the spinal cord (Figs. 5, 6) and to sample too few ganglia. 121
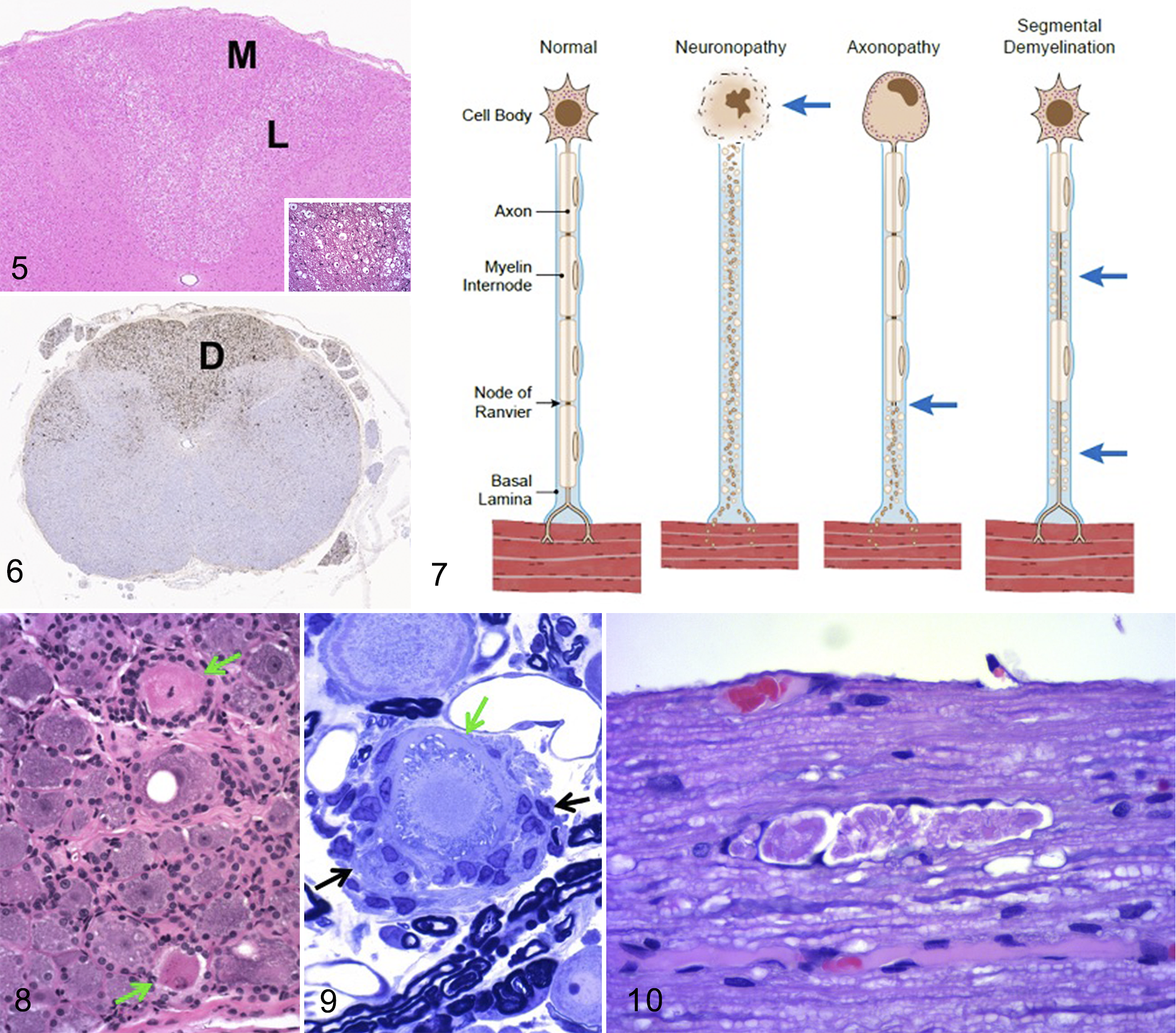
Degeneration in the spinal cord dorsal (posterior) funiculus, juvenile cynomolgus monkey.
For routine screening, PNS tissues usually are processed routinely into paraffin, after which a conventional 5-µm-thick hematoxylin and eosin (H&E)-stained section is prepared. Special procedures to highlight axons (eg, Bielschowsky or Bodian’s silver stains, anti-NFP, anti-PGP9.5) or myelin (Luxol fast blue, anti-MBP, anti-CNPase [2′,3′-cyclic nucleotide 3′-phosphodiesterase]) may be performed on serial 5-µm-thick sections to explore the primary site of PNS damage. In spinal cord, the same axonal and myelin biomarkers may be used to showcase damage, but additional immunohistochemical markers for astrocytes (anti-glial fibrillary acidic protein [GFAP]) or microglia (anti-ionized calcium-binding adaptor molecule 1 [Iba1]) also can reveal sites of injury (Fig. 6). A complete PNS assessment requires that one or more nerves in cross section be osmicated and embedded in hard plastic resin (such as araldite or epon) to permit sectioning at 1 µm. 19,106 Such plastic-embedded, toluidine blue-stained sections provide much greater resolution of myelin structure, especially for small-diameter fibers, than can be obtained in paraffin sections, so this preparation is essential for distinguishing whether PNS diseases initially affect axons or myelin. However, most special neurohistological procedures (eg, immunohistochemistry [IHC]) cannot be performed easily, if at all, on resin-embedded sections.
The ability to diagnose PNS diseases depends on the quality of the samples available for evaluation. 13 Inappropriate handling during collection and processing results in many artifacts that may complicate interpretation. The most common examples are nerve fiber distortion resulting from traumatic removal or excessive tissue stretching and vacuolation or myelin disruption due to inadequate fixation. 175 Handling artifacts may be mitigated by harvesting delicate or hard-to-find PNS tissues (eg, distal nerve branches, sympathetic ganglia) after carcasses or limbs have been fully fixed and more time is available for a methodical dissection. Artifacts also are controlled by careful selection of the fixation and processing protocols, especially for large-diameter nerves of larger animals. 66
Quantitative techniques may be important for diagnosing subtle neuropathies. Common options include stereological measurements of ganglion neuron numbers 28 and assessment of intraepidermal nerve fiber density. 135,137 These techniques require very precise harvest, handling, fixation, cutting, and staining procedures and should be thoroughly practiced before being used on important diagnostic material.
Comparative Pathology of the PNS
Effective neuropathologic evaluation of the PNS requires ready access to references for multiple aspects of PNS biology during health and disease. 20 Key resources include texts detailing the anatomy and functions of major PNS structures, 46,113,177,239 the tissue targets and mechanisms of action for known classes of etiologic agents that impact the PNS, 16,46,52,201,210,239,242 and at least one atlas illustrating recognized incidental background findings and disease-related lesions in PNS ganglia and nerves. 111,115,170,175,213 Many agents have been demonstrated to attack the PNS (Table 2). This comparative pathology review will introduce major concepts relevant to several key categories of PNS disease that may be encountered regularly in diagnostic practice or under controlled experimental conditions.
Selected Nonneoplastic Peripheral Nervous System Diseases.
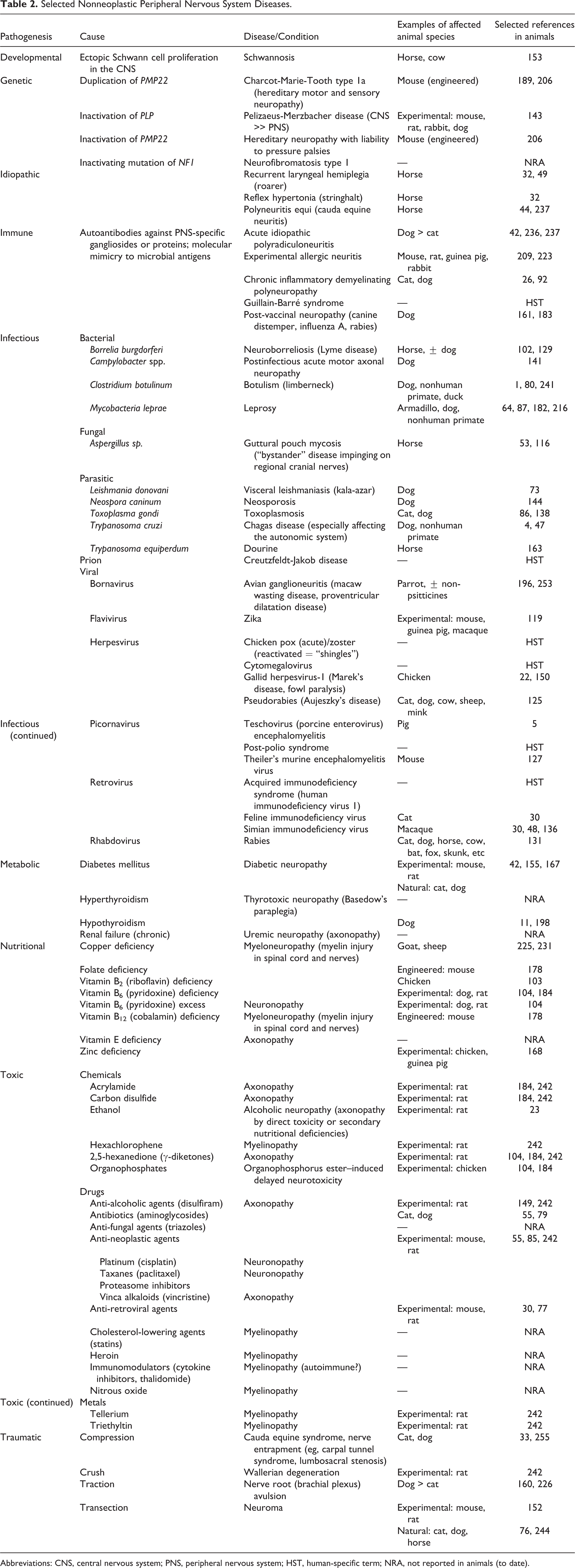
Abbreviations: CNS, central nervous system; PNS, peripheral nervous system; HST, human-specific term; NRA, not reported in animals (to date).
Lesions in the PNS generally present as either a peripheral neuropathy (ie, disease of nerve fibers) or a ganglionopathy (ie, disease of ganglionic neurons [“neuronopathy”]; Fig. 7). 103,175,239 In general, PNS syndromes in animals and humans present as peripheral neuropathies associated with damage to either axons (“axonopathies”) or myelin (“myelinopathies”) rather than neuronopathies; axonopathies are the most common form of PNS disease in all species. Neuroaxonal dystrophy is a distinctive axonopathy that is visible in tissue sections as swollen axons (“spheroids”) rather than the disintegrating fibers of conventional axonopathies; such axonal swelling typically results from disrupted anterograde or retrograde transport leading to intra-axoplasmic logjams of cytoskeletal elements, tubulovesicular profiles, and distorted organelles. 173,175,242 Peripheral neuropathy may affect one (“mononeuropathy”) or more (“polyneuropathy”) cranial, spinal, and/or autonomic (parasympathetic and/or sympathetic) nerves; in most instances, peripheral neuropathies in animals and humans present as polyneuropathies. Similarly, ganglionopathy may impact neuronal cell bodies in cranial, dorsal root, and/or autonomic ganglia, and lesions tend to develop in multiple ganglia. Neuropathies may be reversible or permanent given the regenerative capacity of axons and Schwann cells in the PNS, 71 while ganglionopathies are generally considered to be permanent since severely damaged neurons lack the ability to regenerate, and thus die.
In addition to lesions centered on neurons and nerve fibers (axons and myelin), other PNS cell types also exhibit characteristic changes to PNS perturbation. 175 For example, the satellite glial cells that normally surround ganglionic neurons increase in number to support degenerating neurons (ie, “satellitosis”) and form dense nodules at sites once filled by necrotic neurons (ie, “residual [Nageotte] nodules”). Furthermore, IHC using markers specific for lymphocytes (eg, CD3 for T-cells) and satellite glial cells (eg, glutamine synthetase) may be useful in differentiating resident from infiltrating reactive cells. 19 Nonneural PNS elements in ganglia and nerves, such as endothelial cells and fibrous connective tissue layers, also must be assessed since primary damage to these constituents may lead to ischemia, edema, and eventually fibrosis within ganglia or nerves and may inflict secondary damage on nearby neural components. 185
Chemically Induced Neurotoxicity in the PNS
Historically, most neurotoxicant-induced peripheral neuropathies were discovered first in humans who were accidentally exposed to bulk chemicals (for agricultural or industrial use) 184 or purposely treated with drugs 55,245 (Table 2). Mechanistic understanding of such neurotoxic conditions typically was obtained via subsequent investigative toxicology studies conducted in animals. Lesions induced in the PNS by neurotoxic agents are morphologically similar to those resulting from some metabolic diseases and nutritional deficiencies.
The hallmark lesions of neuronopathy in the PNS are degeneration and ultimately necrosis of the neuronal cell bodies in sensory ganglia (Figs. 8, 9). The sensory nature of peripheral neuropathies resulting from a primary neuronopathy can best be appreciated in the spinal nerve roots, where the dorsal (sensory) root is affected and the ventral (motor) one is spared. A classic example of neuronopathy in DRGs results from exposure to very high doses of pyridoxine (vitamin B6). 104 The site of structural lesions as well as the severity of neurological dysfunction depend on the neurotoxicant dose and the timing of neuropathological evaluation. 122 The pathogenesis of pyridoxine neurotoxicity is not known. Destruction of the ganglionic neuron results in an inability to sustain its axon. Therefore, the entire nerve fiber degenerates, starting with the axon but later encompassing the myelin sheath (since Schwann cells depend on axon-derived trophic factors for survival; Fig. 7). This change often is best appreciated in the dorsal (posterior) funiculus of the spinal cord, which carries sensory nerve fibers originating in DRGs cranially (Figs. 5, 6). Sublethal injury to neurons may allow cells to recover. In this event, neuronal processes in the PNS may undergo regeneration, 105 but projecting fibers within the spinal cord will not.
The most prominent lesion of primary axonopathy in the PNS is axonal degeneration and ultimately fragmentation. Well characterized examples of neurotoxic axonopathy in the PNS include exposure to anti-neoplastic agents (eg, vincristine) 6,55 and organophosphates (ie, organophosphorus ester–induced delayed neurotoxicity [OPIDN] 108,120 ). Axonal damage first occurs in the most distal portions of nerves, and in particular in large-diameter, thickly myelinated axons that have the highest metabolic requirements and most distant cell body-based support machinery. The initial lesion is swelling and discoloration (either pallor or increased staining intensity), which proceeds in due course to fragmentation of the axon. The adjacent myelin sheath is broken down as a secondary response (Fig. 7). The nerve fiber debris is degraded by Schwann cells (which can function as phagocytes when needed) or infiltrating macrophages (ie, blood-borne professional phagocytes) within “digestion chambers” (Fig. 10). If the basal lamina surrounding the PNS nerve fiber remains intact, subsequent proliferation of Schwann cells forms a solid column of cells (termed the “band of Büngner”) that can serve as a guide through which sprouting axons can grow during regeneration (Fig. 10). While fiber tracts in the CNS may be affected by the axonopathy, regeneration of spinal cord nerve fibers does not occur.
The major lesion of primary myelinopathy in the PNS is segmental demyelination. Dietary riboflavin (vitamin B2) deficiency in young, rapidly growing chickens is a useful surrogate for experimental studies of toxicant-induced myelin damage. 107 Riboflavin is a key factor in lipid metabolism and mitochondrial energy synthesis, which are essential Schwann cell functions in producing and maintaining myelin sheaths. Affected Schwann cells become enlarged and engulf myelin early during the disease (Fig. 11), while in late stages badly damaged Schwann cells die and the resulting internode is lost (Fig. 7). Axons have little or no myelin and over time may become atrophied due to the loss of Schwann cell-derived trophic factors, but they nevertheless can conduct nerve impulses albeit at a slower rate. Schwann cells may be replenished if the insult is removed, but conduction velocity may remain slow since commonly more than one new Schwann cell is needed to cover the denuded axons. 21
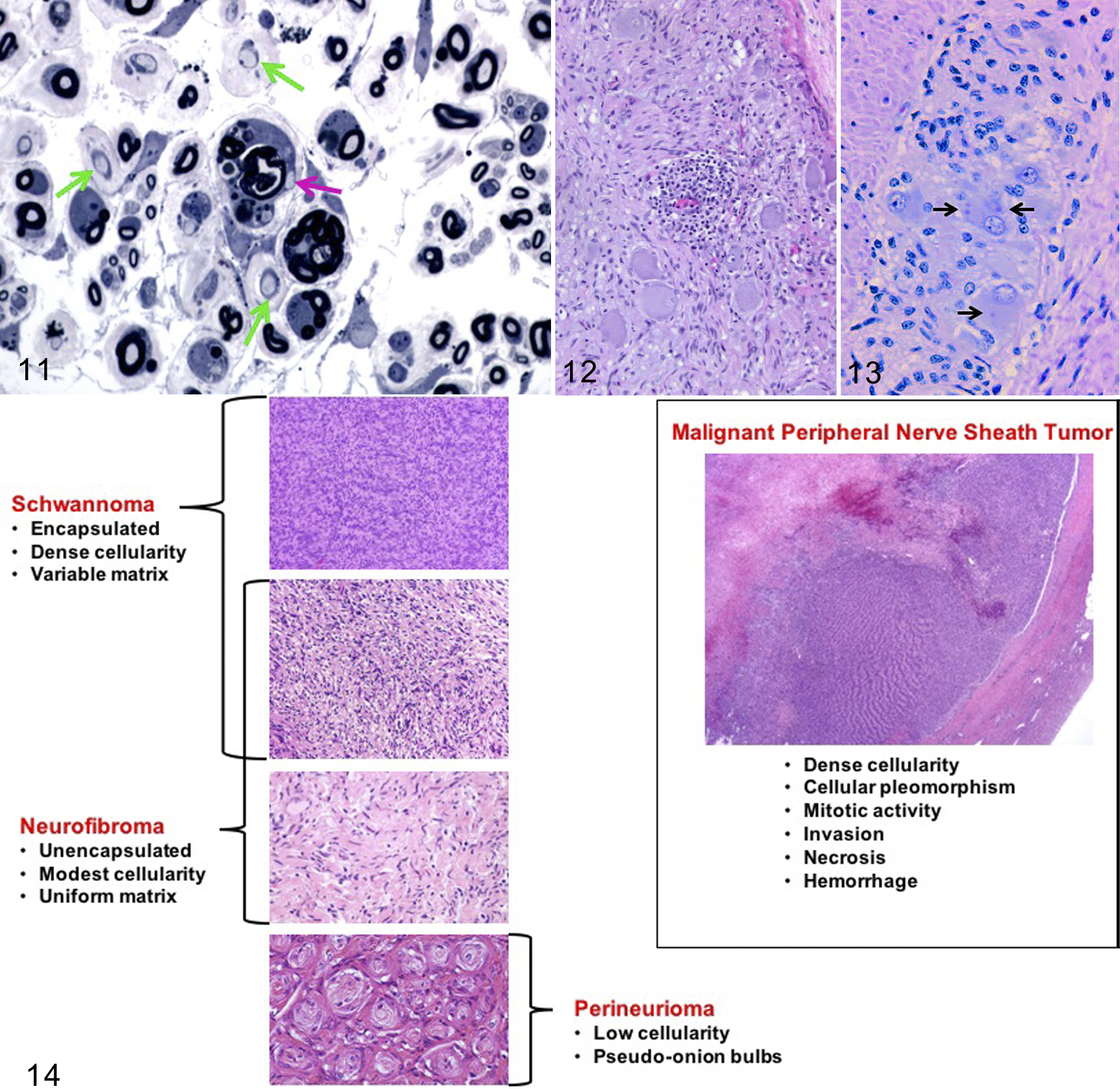
Primary segmental demyelination (myelinopathy) caused by riboflavin deficiency, sciatic nerve, 14-day-old chick. Schwann cells are enlarged and contain intracytoplasmic myelin-free axon profiles and nearby fragments of blue-stained degraded myelin (pink arrow). Other axons show more advanced stages of demyelination (green arrows). The wide separation of nerve fibers indicates endoneurial edema. Aldehyde perfusion followed by osmication, plastic resin, toluidine blue and safranin. [Image reproduced from Jortner,
103
by permission of Sage Publications.]
End-stage lesions in peripheral neuropathy typically present as advanced nerve fiber degeneration. In such cases, the primary target site (axon or myelin) cannot be discerned using H&E or special neurohistological stains.
Metabolic Diseases of the PNS
Peripheral neuropathies secondary to systemic metabolic diseases are an important medical complication in humans and, to a lesser extent, in companion animals. The most important of the metabolic-related PNS diseases is diabetic neuropathy (DN). Diabetes mellitus–associated peripheral neuropathy in humans, which affects up to 60% to 70% of patients, 27 results in progressive degeneration of nerves. The most prominent early effect of DN in humans is hyperalgesia due to damage to free sensory endings and the most distal portions of nerves, while later in disease weakness and numbness reflect the migration of nerve fiber lesions to proximal nerve trunks. 167 In contrast to humans, animals with diabetes mellitus seldom exhibit clinical signs of DN, and when they do the spectrum differs from the signs and symptoms reported by human patients. Clinical signs in cats include plantigrade/palmigrade stance and gait, difficulty jumping, poor postural reactions, ataxia/paresis, muscle atrophy especially in the pelvic limbs, and irritability when touching or manipulating the paws. 156 Clinical signs of DN are observed rarely in dogs with diabetes mellitus but include slowly progressive paraparesis and hind limb ataxia. 42 Ultrastructural PNS lesions reported in cats with diabetes mellitus include splitting and ballooning of myelin sheaths, fewer completely demyelinated fibers, some remyelination, cytoplasmic glycogen in Schwann cells, axonal shrinkage and degeneration in the most severely affected animals, and endoneurial fibrosis and microvasculopathy. 57,155 –157 This constellation of lesions suggests that demyelination predominates over axonal changes in cats, 154 while axonal changes may be the initial change in dogs. 24,42 Proposed causes and pathogenic mechanisms for DN are numerous and extensively investigated in animal models 72,95,167 where conflicting findings can be found with different species, strains, and methods of diabetes mellitus induction. For example, hyperglycemia can act through the polyol pathway to cause accumulations of sorbitol and fructose in nerves of humans and rodents with diabetes mellitus yet, while the polyol pathway is altered in cats with diabetes mellitus, no sorbitol accumulation is present in their peripheral nerves. 156 Other possible pathogenic mechanisms include glycosylation of myelin and axonal structural proteins, nerve growth factor deficiencies, impaired vascular supply or mitochondrial abnormalities that lead to oxidative stress, and glutamate excitotoxicity. 27,42,59 Rodent models of diabetes mellitus produce subclinical peripheral neuropathy that is quantified by changed behavioral responses to thermal or tactile stimuli, nerve conduction changes, and, in some cases, morphologically reduced intraepidermal nerve fiber density similar to that seen in humans. 167,169 Rodent variants of diabetes mellitus can be divided into spontaneous/genetic and chemically induced models. The morphologic changes observed in the PNS can be rare and highly variable depending on background strain and age, dosing regimens of diabetes-inducing chemicals such as streptozotocin, and type I versus type II diabetes models where contributions from other elements of the metabolic syndrome can influence the latter. 95,167 In addition, precise correlations between morphologic changes, like reduced intraepidermal nerve fiber density, and changes in behavioral testing of sensory perception can be lacking in nonclinical studies with rodent models of diabetic neuropathy, often leading to frustrating failures in subsequent human clinical trials. 59,169
Another metabolic-related PNS disease results from hypothyroidism. Two types of peripheral neuropathy are seen in hypothyroid humans: a sensorimotor polyneuropathy, and an entrapment syndrome in the wrist caused by deposition of mucopolysaccharides in the perineurium and endoneurium of the median nerves. Reported morphologic lesions in case reports range from none to segmental demyelination and axonal degeneration, accumulations of glycogen in Schwann cells, and endoneurial fibrosis. 51,166 Hypothyroid dogs have lower motor neuron disease manifesting as generalized weakness, vestibular deficits, megaesophagus, and laryngeal paralysis; 42,97 morphologic lesions in such cases include axonal degeneration and depletion of large myelinated fibers, demyelination, and remyelination. 42 Proposed mechanisms relate to decreased T4 stimulation of mitochondria leading to decreased ATP-dependent Na+/K+ pump activity, defective axonal transport, and finally axonal degeneration. 166 Accumulation of glycogen in Schwann cells also has been proposed to induce segmental demyelination. 51
Peripheral neuropathy associated with hyperthyroidism, termed Basedow’s paraplegia in humans, was initially reported in the late 1880s 35 although linkage of the hormone imbalance with the PNS syndrome has been questioned. 181,222 Reported light microscopic changes include thinly myelinated large-diameter nerve fibers and axonal loss, while ultrastructural lesions include myelin splitting and degeneration, axonal aggregates of microtubules or neurofilaments, and mitochondrial changes in both axons and Schwann cells. 171 Similar neuropathic changes have not been reported in companion animals with hyperthyroidism.
Renal failure can induce a distal, symmetrical, mixed sensorimotor neuropathy in humans. 7 Initial symptoms include pain, numbness, and tingling sensations in the feet and legs that can be followed by weakness, muscle atrophy, and reduced reflexes in advanced disease. 10,195 Sural nerve biopsies show extensive loss of myelinated fibers without inflammation or vasculopathy. 195 The pathogenesis is unknown, but proposed mechanisms include deficiencies in essential nutrients such as biotin, thiamine, and zinc; accumulations of uremia-associated toxic molecules (so-called “middle molecules” not removed by hemodialysis); and secondary hyperparathyroidism with excess circulating parathyroid hormone. 194 In cells, elevated parathyroid hormone increases myoinositol, a neurotoxin in rats, and is associated with nerve conduction deficits in experimentally induced uremia in dogs. 74 While peripheral neuropathy has been demonstrated in animal models of induced renal failure, reports of similar lesions in domestic animals with clinical cases of chronic kidney disease have not been reported.
Paraneoplastic Diseases of the PNS
The juxtaposition of metabolic and paraneoplastic neuropathies acknowledges the current hypothesis that many paraneoplastic conditions reflect imbalances in systemic chemistry (eg, circulating levels of hormones) or an immune attack against antigens that are expressed by both neoplastic cells and PNS tissues. Paraneoplastic syndromes are nonmetastatic complications that impact sites distant from a primary tumor, and while they may affect any part of the CNS or PNS, the neurological signs and structural changes most often reflect PNS alterations. Paraneoplastic neuropathic syndromes are rare in humans. They may present prior to identification of the associated malignancy, and they often result from immunologic reactions to shared epitopes between the tumor cell proteins and those in the nervous system. 42,75,128,148 They are divided into “classical syndromes,” with a high likelihood of paraneoplastic etiology, and “nonclassical syndromes,” which only rarely are associated with cancer. 75,128 Discrimination of definite versus possible paraneoplastic neuropathic syndromes relies on detection of an identifiable neoplasm, demonstration of onconeural antibodies, and/or remission of neurologic symptoms after tumor removal/treatment. 128,148 Paraneoplastic neuropathic syndromes have been associated with particular types of spontaneous neoplasia in both humans and animals, including (as reported in cats and dogs) many hematopoietic tumors (lymphocytic leukemia and lymphoma), hormone-producing tumors (eg, insulinoma), some carcinomas (hepatocellular and biliary), thymoma, and rarely other tumor types (eg, osteosarcoma). 25,34,42,96,139,190,221,246 The typical clinical presentation in veterinary practice is paraparesis to tetraparesis with lower motor neurons signs of hyporeflexia, hypotonia, and muscle atrophy. 42,96 Morphologic lesions seen in nerves include myelin vacuolization, demyelination and remyelination, axonal degeneration and loss, and increased numbers of Schwann cells. 25,34 Resolution of clinical signs has been reported in animals following tumor treatment or removal. 139,246 Reproducible animals models are a major need for future studies to investigate the pathogenic mechanisms of these rare diseases.
Inflammatory and Infectious Diseases in the PNS
Inflammation plays an important role in both naturally occurring and experimental PNS disease. Immune-mediated damage to the PNS has many causes and several fundamental mechanisms.
Immune-Mediated PNS Disease
One major set of inflammation-associated neuropathies results from immune-mediated attack on nerves. Examples of such diseases include Guillain-Barré syndrome (GBS) in humans; experimental allergic neuritis (EAN) in mice, rats, guinea pigs, and rabbits; 209,223 post-vaccinal polyneuropathy in dogs and humans; 131,183 and possibly acute idiopathic polyradiculoneuritis (APN) in dogs. These acute conditions result in PNS segmental demyelination stemming from an anti-myelin T-cell-mediated response and usually present as a widespread polyneuropathy characterized chiefly by lower motor neuron disease that first affects hind limb nerves but eventually ascends to include forelimb nerves and often cranial nerves. 197 In dogs with APN, ventral spinal nerve roots are affected preferentially. 42 The severity of inflammation is highly variable and may include lymphocytes, plasma cells, macrophages, and neutrophils. Most affected individuals achieve partial or complete recovery, although a small proportion of patients may relapse.
Multiple factors have been implicated in immune-mediated polyneuropathies. Most cases of GBS and APN are preceded by exposure to foreign antigens, chiefly via acute infections (mainly gastrointestinal or upper respiratory) or recent vaccinations. 86,256 In particular, molecular mimicry to Campylobacter spp. antigens has been posited in the pathogenesis of both APN and GBS based on the fact that some bacterial lipooligosaccharides are similar to GM1 ganglioside found in nerve. 141,257 In EAN, antigens used as immunogens to incite clinical disease include whole nerve homogenates and myelin-specific proteins such as myelin protein 0 (P0), myelin protein 2 (P2), and/or myelin protein of 22 kDa (PMP22), 223 but autoantibodies to these constituents are not present consistently in GBS patients. 134
Chronic inflammatory demyelinating polyneuropathy (CIDP) is a heterogeneous group of diseases in humans, clinically distinct from GBS by virtue of its protracted, relapsing, and/or progressive clinical course. 117 Clinical disease may manifest as deficits that are primarily motor, sensory, or both. 92 Spontaneous CIDP-like diseases have been described in dogs and cats, 26 including multifocal involvement of spinal nerves and spinal roots. Histologic features are primarily paranodal demyelination/remyelination, “onion bulbs” (concentric layers of Schwann cell processes and collagen encircling an axon as a result of cyclic segmental demyelination and remyelination), and varying mononuclear cell infiltration within the endoneurium. Human comorbidities include diabetes mellitus and inflammatory bowel disease. 117 Some rodent variants of EAN have been used to model CIDP, 110,203 and these studies have suggested that autoreactive CD4+ T cells are directing an antibody-mediated response against myelin P0. 114
Infectious PNS Diseases
The second major mechanism of inflammation-associated neuropathies is innate and acquired immune responses resulting from microbial infection of PNS ganglia and/or nerve. Many different pathogens have been identified as causes (Table 2). Most etiologic agents in animals result from bacterial, 164,215,230 protozoal, 43,140,163,199 or viral 8,12,30,38,88,118,133,220 infections. Neuropathies have also been demonstrated recently in humans with prion diseases 9,249 but to date have not been reported in prion diseases of animals (eg, bovine spongiform encephalopathy in cattle, chronic wasting disease in cervids, scrapie in sheep). The microbes may reach the PNS by direct inoculation leading to PNS uptake and retrograde transport (eg, a bite from a rabid animal) or by hematogenous delivery (eg, human immunodeficiency virus [HIV], leprosy). 12,30,220 These pathogens, especially viruses, produce a heterogeneous and broad spectrum of disease that ranges from asymptomatic infection to polyneuropathy (or less often polyganglionopathy) up to fatal CNS complications with or without concurrent PNS lesions. Depending on the disease, inflammatory changes occur in multiple ganglia, cranial nerves, and spinal nerves; 43,163 however, many such changes, such as neuritis in cutaneous mycobacteriosis 45 and trigeminal ganglionitis in rabies, occur inconsistently. Primary changes associated with PNS infection may be complicated by neurotoxic lesions associated with treating the infection, as seen in patients with HIV-induced distal symmetric polyneuropathy who develop sensory neuron lesions in DRGs following treatment with a nucleoside analog reverse transcriptase inhibitor. 30 For this reason, sample collection in both diagnostic practice and experimental studies should be far-reaching.
The inflammatory reaction to microbial diseases (regardless of cause) generally includes numerous lymphocytes, variable numbers of plasma cells and macrophages, and fewer neutrophils (Fig. 12). While the pathogenesis of neurotropic viral infections commonly involves nerves, morphologic evidence of PNS inflammation may be minimal or absent for host-adapted pathogens during the latent phase (Fig. 13). In contrast, in viral infection in aberrant hosts such as dogs exposed to pseudorabies virus through feral swine, trigeminal ganglioneuritis may be pronounced and may have a more extensive neutrophilic influx. 38 Evidence of infection—bacteria, 31 protozoal amastigotes, or viral inclusions—may be observed in affected PNS tissues. Antemortem biopsies may reveal only degenerative changes in nerve fibers. 41
The PNS is especially vulnerable to viral infection by virtue of its direct contact with a diverse range of tissue types spanning nearly the entire exterior and interior of the body, including many exposed nerve endings that are not enclosed by a blood-nerve barrier. Retrograde viral transport through sensory nerve fibers carries virus to the neuronal cell bodies in PNS sensory ganglia serving cranial nerves, spinal nerves, and/or autonomic nerves. 162,205 In contrast, retrograde invasion of motor neurons at neuromuscular junctions transports virus more directly to the CNS, and is of prime importance for CNS viral infections such as rabies and poliovirus.
Damage to infected PNS tissues is postulated to occur by 2 basic mechanisms. In some cases, damage is thought to represent indirect parenchymal injury, where the PNS elements are trapped in an intense antipathogen reaction. 227 In other cases, PNS components are thought to be the primary targets as the immune system responds to foreign antigens expressed by the infected PNS cells. This latter pathogenesis also has been proposed to explain ganglionic and nerve lesions that develop in test animals following intrathecal administration of viral gene therapy vectors bearing recombinant human transgenes. 83,89
Bystander PNS Diseases
Peripheral nerves may be involved indirectly as nearby inflammatory lesions impinge on local nerve trunks. Sometimes the clinical manifestations of such peripheral nerve involvement are responsible for significant morbidity. For example, horses with guttural pouch mycosis may develop laryngeal hemiplegia due to vagal nerve (CN X) involvement, dysphagia due to glossopharyngeal nerve (CN IX) and/or spinal accessory nerve (CN XI) lesions, facial paralysis due to facial nerve (CN VII) damage, or lingual paralysis due to hypoglossal nerve (CN XII) injury. 116 Inflammation of peripheral nerves is sporadically reported in other diseases, such as horses with Halicephalobus gingivalis infections. 101
Idiopathic PNS Diseases
Polyneuritis equi (“cauda equina neuritis”) is a slowly progressive syndrome primarily involving spinal nerve roots at the terminus of the spinal cord. 44 Chronic clinical manifestations are sensory and motor deficits involving the tail and perineum, with urinary and fecal incontinence. Peripheral nerves in addition to the cauda equina can be affected including cranial nerves (especially CN V, CN VII, and CN VIII). Microscopically, the primary changes are granulomatous inflammation, axonal degeneration, and demyelination. The inflammatory reaction also may include T cells, plasma cells, and even neutrophilic microabscesses. While definitive antemortem diagnosis can be difficult, inflammatory infiltrates have been noted in terminal intramuscular nerve branches, together with neurogenic muscle atrophy. 3
Equine recurrent laryngeal hemiplegia results from denervation atrophy of laryngeal muscle, mainly on the left side, secondary to degeneration of the recurrent laryngeal nerve. 32 The paralysis leads to an inability to adduct the arytenoid cartilage and vocal folds, which results in a partial airway obstruction. No clinical difficulty is experienced at rest, but affected horses exhibit inspiratory stridor (“roaring”) during intense exertion. The fundamental lesion (axon vs myelin) and distribution (mononeuropathy [unilateral or bilateral] of the laryngeal nerve or polyneuropathy) have yet to be defined. 49
Equine reflex hypertonia (stringhalt) is a neuromuscular condition in aged horses characterized by an involuntary, exaggerated upward jerking motion (hyperflexion) of the distal hind limb. The underlying cause appears to be a distal axonopathy of myelinated fibers. 32 The gait abnormality often is unilateral in the idiopathic (classical) condition but typically is bilateral in the acquired (sporadic or Australian) variant, which is associated with seasonal ingestion of the toxic plant Hypochaeris radicata (false dandelion or flatweed). Axonal lesions may be visible in other nerves in the acquired form.
Neoplastic Diseases of the PNS
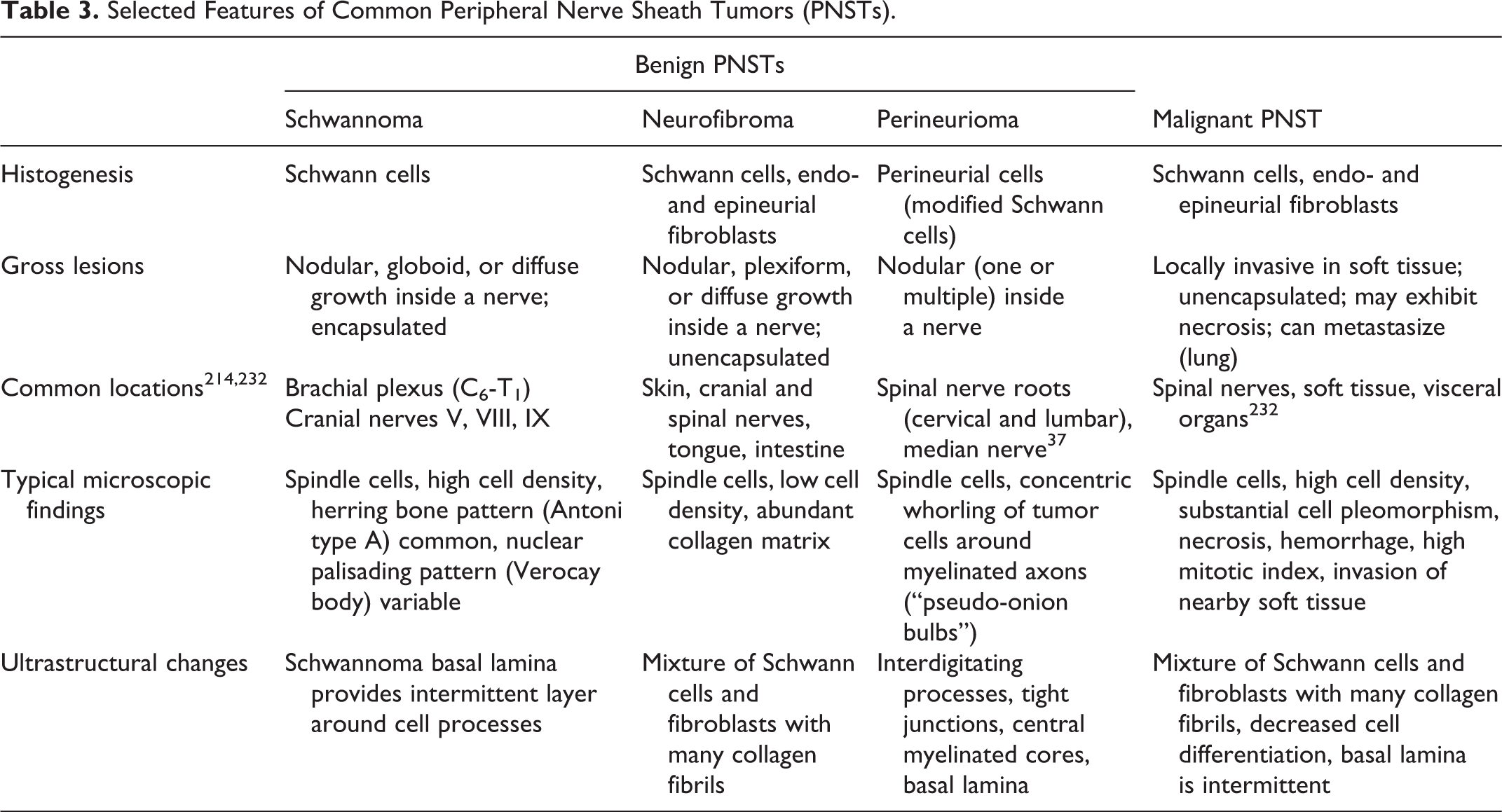
Tumors of the PNS represent a substantial diagnostic challenge in veterinary pathology since they are relatively common in animals and display not only significant morphologic similarity among themselves, but also to other tumors that are not of PNS origin. Additionally, nonneoplastic, proliferative PNS lesions are seen occasionally that may mimic histologic features of these tumors. For these reasons, a diagnostic flow chart (Fig. 14) of key histopathologic similarities and differences is an indispensable guide in differentiating among various diagnostic options.
Nomenclature for PNS tumors in animals has been adapted largely from terminology used in medical pathology, which often has been misused or applied in a cavalier manner in veterinary pathology. The ensuing confusion commonly leads to a diagnosis of “peripheral nerve sheath tumor” (PNST) as a “garbage can” diagnosis that lumps benign and malignant lesions into a single group while providing no useful mechanistic or prognostic information that might yield more refined morphological and/or molecular classification criteria. When properly classified, many of the PNS neoplasms and their histologic subtypes that are seen in humans also are found in domestic animals, though their incidences differ among species. However, many of the specific nerve sheath tumors described in humans are associated with particular germline or somatic mutations. While mutations have been reported in individual cases in domestic animals, we generally lack this knowledge in veterinary medicine—all the more reason to accurately classify these tumors! 145,159,165
Neoplasms arising from the PNS are classified into several categories based on their cell of origin and biological behavior (Table 3). Diagnostic criteria needed to distinguish different PNS tumor types are based on the microscopic pattern within the neoplastic foci and, if available, the spectrum of molecular markers expressed by the cancer cells. Tumors arising from the PNS all exhibit endoneurial spread, which is distinct from the perineurial growth pattern of nonneural tumors that are only invading an ensnared nerve. 29 It is crucial to emphasize that there are no specific IHC markers for PNS-derived tumors, and they must be evaluated in conjunction with relevant histologic and clinicopathologic data. However, there are some useful markers that may be employed to distinguish nerve sheath tumors from other mesenchymal tumors (Table 4).
Selected Features of Common Peripheral Nerve Sheath Tumors (PNSTs).
Selected Immunohistochemical Markers for Common Neoplasms of the Peripheral Nervous Systema.

Abbreviations: PNST, peripheral nerve sheath tumor; GFAP, glial fibrillary acidic protein; NF200, neurofilament protein 200 kD; SMA, smooth muscle actin; Var, variable immunoreactivity; %, the percentage of immunoreactive cells; —, no expression.
a Table adapted from Higgins et al (pp. 882–883), 81 by permission of John Wiley & Sons.
Schwannomas
These benign tumors originate from Schwann cells. These tumors most often are well-circumscribed, encapsulated masses that are eccentrically located with respect to the nerve of origin. While they often are attached to a nerve, the lack of a visible attachment should not deter a diagnosis of nerve sheath tumor. 192 Schwannomas have been reported in dogs, cattle, and horses and have been described less often in cats, goats, and pigs. 81 The affected PNS organs vary with the species. Dogs tend to have unilateral schwannomas affecting an intradural spinal nerve root or a nerve branch feeding the brachial plexus. Old cattle often develop multicentric tumors localized to the autonomic ganglia and plexuses. 81 Humans exhibit a predilection for sensory nerve involvement that has not be investigated in domestic animals.
Multiple histological subtypes have been identified in humans 192 and also have been described in animals, though the patterns occur less consistently in schwannomas of animals. The most common subtype in humans is the “conventional” schwannoma, defined by intermingled regions of dense cellularity (Antoni type A pattern, often arranged in a storiform [spiraling] pattern) and cell-poor regions (Antoni type B pattern) sometimes with focal areas of nuclear palisading (Verocay bodies). These features, while occasionally identified in animal schwannomas, are nonetheless not a defining feature of schwannomas in animals. The most common histologic subtype in animals is the cellular schwannoma, which is relatively uncommon in humans. The densely cellular arrangement comprises all or nearly all (90%) of the mass, and nuclear palisading is largely absent. Despite the high cellular density, nuclei lack significant pleomorphism, spontaneous necrosis, and mitotic figures—all features indicating that the tumor will exhibit benign biological behavior. Plexiform schwannomas are multinodular masses, and they are seen in animals. 211 The nodules (several to several dozen) are interconnected grossly and generally well-circumscribed but may lack clear encapsulation. Similar to cellular Schwannomas, these tumors consist chiefly of Antoni type A pattern and lack Verocay bodies.
Schwannomas occasionally exhibit unusual findings that may confound the diagnostic pathologist. One such microscopic feature, termed “ancient change,” is characterized by pronounced karyomegaly with remarkable nuclear atypia and folding. 93 Despite the striking nuclear abnormalities, these tumors are otherwise histologically unremarkable, and the presence of ancient change bears no impact on prognosis. Another unusual variant is the melanocytic Schwannoma, worthy of discussion as a differential diagnosis for other pigmented tumors of neural crest lineage such as melanocytoma or melanoma. 176 These schwannomas are darkly pigmented because the fusiform tumor cells are making melanin. Finally, in the dog, schwannomas at 2 anatomic locations warrant specific discussion. The first is the brachial plexus (and less commonly the pelvic plexus). Here, spinal nerve roots may be infiltrated by this tumor with secondary compression of the spinal cord resulting in significant clinical signs, including severe pain. 124 Tumors at this site commonly involve multiple nerve roots. Second, involvement of the trigeminal (CN V) is commonly reported. Grossly, these tumors are more yellow and softer than meningiomas, which are the primary differential diagnosis at this location. 233
Neurofibromas
These benign nerve sheath tumors harbor a mixture of Schwann cells, perineurial cells, and fibroblastic cells, and thus are more heterogeneous in appearance than the relatively hypercellular schwannoma. While Schwann cells may be difficult to identify within the tumor, this entity is now known to arise from Schwann cells. 224 In contrast to schwannomas, however, neurofibromas generally are of low cellularity, unencapsulated, and have a uniform matrix. Nuclear appearance is also characteristic, with “bent” or comma-shaped, hyperchromatic nuclei. These cells are most often arrayed in a mucin-rich matrix, with abundant, variably sized, wavy collagen fibers. In humans, this tumor often affects dozens of locations along multiple nerves—a condition known as von Recklinghausen disease—due to inactivating mutations of the neurofibromatosis type 1 (NF1) tumor suppressor gene, which encodes the protein neurofibromin. 254 Neurofibromin acts to inhibit the activity of the Ras oncogene, which promotes tumor formation by moderating the mitogen-activated protein kinase (MAPK) pathway. 109 What role, if any, increased MAPK signaling plays in veterinary neurofibromas remains unexplored.
Neurofibromas are most often reported in the dermis of dogs and horses, where 3 histologic subtypes analogous to their human orthologs have been identified. 126,211 These variants are localized, diffuse, and plexiform. 81 Localized cutaneous neurofibroma in humans is believed to arise not from nerves but from skin-derived precursors. 126 These tumors often affect the dermis and underlying subcutis and are either nodular or polypoid in appearance. As these arise from skin-derived precursors, typically no discrete connection exists to associated nerves; however, localized tumors also may arise in an intraneural location where they form a slow-growing, mucin-rich matrix that surrounds and expands along individual nerve fibers. 192 Diffuse neurofibromas appear similar histologically to localized tumors but exist in large, thick, cellular lesions. Plexiform neurofibroma is an uncommon lesion in both humans and veterinary medicine. These neoplasms tend to associate with large-caliber nerves in humans, although they have also been reported in the subcutis, large intestine, and tongue in dogs. 211 Diffuse and plexiform neurofibromas are often associated with mutations in NF1 and have been described in veterinary medicine as infiltrative into adjacent muscle, suggesting that they could represent a heightened risk for malignant transformation. 224
Perineuriomas
The term perineurioma (variant spelling: perineuroma) should be reserved for a tumor comprised exclusively of perineurial cells. This tumor is rarely reported in veterinary medicine, and only in dogs to date. 82 These tumors may arise at an intraneural site or within soft tissue. In humans, they have been variably associated with deletion at chromosome 22q, affecting the NF2 gene. 208 Their very characteristic microscopic appearance features concentric whorls (“pseudo-onion bulbs”) comprised of proliferating perineurial cells, individual nerve fibers, and endoneurial capillaries. 132 The appearance of individual perineurial cells often resembles that of a fibroblast, with a fusiform, hyperchromatic nucleus and minimal visible cytoplasm. Diagnosis in humans utilizes ancillary tests including expression of epithelial membrane antigen. 132 Unfortunately, antibodies against epithelial membrane antigen have been unsuccessful in the dog. Claudin 1 (a component of tight junctions) has been described as a potentially useful marker for discriminating this tumor in dogs, although this is not specific for perineurial differentiation. 98
Hybrid Tumors
The nerve sheath of the PNS is a heterogeneous microenvironment, with multiple Schwann cell precursor-derived elements. Therefore, the existence of tumors exhibiting features of multiple nerve sheath tumor types should not be surprising. These so-called “hybrid tumors” are benign, but owing to their confusing cellular appearance they may represent an increased diagnostic challenge. 240 The most common hybrid tumor is the schwannoma/perineurioma combination. These tumors largely keep the storiform pattern of the schwannoma but also exhibit the whorling pattern of the perineurioma. 90 Schwann cells are distinguished by their plumper, tapered nuclei, and pale eosinophilic but visible cytoplasm, while perineurial cells have smaller, more slender nuclei. In humans, these tumors often have complex expression patterns of Schwann and perineurial differentiation, which speaks to the plasticity of the Schwann cell. 90
Intratumoral Metaplasia
Metaplastic changes are also encountered occasionally in both human and animal nerve sheath tumors, most often in schwannomas. 50,193 Typically, metaplastic tissue exhibits mesenchymal differentiation—cartilage matrix is most common, although sometimes osseous and rarely rhabdoid phenotypes also may be seen. A fibro-fatty matrix may be visualized, and care should be exercised to determine if this represents infiltration into the subcutaneous tissue or true adipocytic metaplasia. Epithelial (glandular) metaplasia within schwannomas has been described in humans; 200,234 to our knowledge, this finding has not been described in animals. When present, intratumoral metaplasia in nerve sheath tumors should prompt a careful examination for evidence of malignant transformation. In particular, the combination of pleomorphic cells of both nerve sheath and skeletal muscle lineages (referred to as a “Triton tumor”) generally defines an aggressive malignancy in humans, although the significance of this finding is still unknown in veterinary medicine. 91,228
Malignant Peripheral Nerve Sheath Tumors (MPNSTs)
Similar to their benign counterparts, MPNSTs have suffered from historically poor, or poorly specific, morphological criteria. The identification of genetic lesions in humans helps in part to predict biological behavior, but ultimately the diagnostic classification remains based on light microscopy with some assistance provided by the tumor location. Nerve sheath tumors involving a major nerve in humans are considered malignant nearly by default. Commonly reported sites of MPNSTs in animals include spinal nerves of the dog 81 and the eyelids of dogs and particularly cats. 84,248
MPNSTs are invariably hypercellular, which is a useful criterion in humans as they typically arise from conventional Schwannomas. 192 In contrast, most benign schwannomas in animals are of the cellular subtype (ie, composed of hypercellular Antoni type A regions), so cellular density is somewhat less useful in diagnosing MPNSTs of animals. Instead, the 2 most reliable indicators of malignancy are elevated mitotic index and spontaneous necrosis within the neoplasm. Benign nerve sheath tumors typically have a low mitotic index, even in densely cellular areas. 172,179,202 While no established criteria exist in veterinary medicine, human MPNSTs routinely exceed 4 mitotic figures per 10 high-power fields. 179 Nuclear morphology is a helpful ancillary feature, as the anisokaryosis index is typically upwards of 3-fold in malignant tumors, with karyomegaly standing out in contrast to the normally tapered Schwann cell nucleus. Necrosis and hemorrhage are also increasingly associated with malignancy. As with all sarcomas, these features may be geographically variable, so care should be exercised to thoroughly section the tumor when grossing the sample.
Immunohistochemical Features of Nerve Sheath Tumors
In addition to histologic features on H&E, IHC is frequently employed as a means to differentiate tumors of the PNS (Table 4). Arguably the most common marker used for discrimination of Schwann cell tumors is laminin. 70,212 Schwann cells secrete several proteins that contribute to the formation of an adjacent basement membrane. Thus, either collagen IV or laminin should be present. Unfortunately, the interpretation of this IHC method must be done with care as this is not a “positive or negative” readout. Rather, the marker should reveal delicate tendrils of the basement membrane surrounding individual cells. This determination is made most readily in cross sections of the tumor displaying multiple transverse profiles of axons.
As both Schwann and perineurial cells are neural-crest derived, they also commonly express the SRY-related homeobox transcription factor SOX10. 60 While expression of this marker is often heterogeneous, in our (KDW) experience about 40% to 50% of tumor cells in animals are positive for this marker. As this is a transcription factor, specific immunoreactivity is also more easily evaluated as it is confined to the nucleus. Of note, SOX10 also labels melanocytes, so the use of SOX10 in conjunction with laminin or collagen IV is required to differentiate melanocytic Schwannomas from melanocytic tumors.
Nerve sheath tumors are also often immunoreactive to S100 and glial fibrillary acidic protein (GFAP), although the diagnostic utility of the former marker remains in doubt in veterinary medicine. 70,212 GFAP, while expressed in a minority of tumors (roughly 30% to 40%), is nonetheless a helpful marker to identify nerve sheath tumors in animals. Personally, we (KDW) and others have used GFAP to identify intestinal nerve sheath tumors that are negative for CD117 and smooth muscle actin (SMA). 147,187 In tumors with weak or negative GFAP immunoreactivity, the addition of S100 may be useful in suggesting that the mass originates from peripheral nerve cells. The precise distribution, intensity, and frequency of S100 immunoreactivity in nerve sheath tumors of animals is more variable compared to human tumors. Nonetheless, this marker may aid in distinguishing suspected nerve sheath tumors from some non-melanocytic or fibroblastic mimics.
Conclusion
The ability to efficiently and effectively collect, process, and evaluate the ganglia and nerves of the PNS is an underdeveloped skill for many pathologists who do not regularly encounter the joys of neuropathology. This gap in understanding is unfortunate given the number of conditions in which the PNS is affected, and sometimes substantially so. As shown in this article, the basic principles of PNS preparation and analysis are similar to those utilized for other organs and systems with the exception of some special techniques (eg, perfusion fixation, resin embedding of nerves) that may be employed to better show defects in PNS myelin. The authors trust that the range of PNS changes highlighted in this review as well as the list of references to other, more detailed PNS resources will aid comparative and diagnostic pathologists become not only familiar but also comfortable with their abilities as PNS neuropathologists in their future projects.
Footnotes
Authors’ Note
This article summarizes and extends the content provided during a half-day concurrent scientific session discussing the “Peripheral Nervous System” held at the 2019 Annual Meeting of the American College of Veterinary Pathologists (ACVP), San Antonio, TX, on November 10, 2019.
The views expressed in this article are those of the authors and do not necessarily represent the policies, positions, or opinions of their respective employers and agencies.
Acknowledgements
The authors appreciate the support originally provided to formulate this course by the Annual Symposium Committee of the American College of Veterinary Pathologists (ACVP), which selected this session as a symposium for presentation at the 2019 annual meeting; the superb schematic illustrations in Figures 3, 4, and 7 designed by Mr Tim Vojt (Columbus, OH); permission to publish ![]() by Dr William Valentine; and assistance with finalizing the figure plates by Mr Tim Lanigan (Frederick, MD).
by Dr William Valentine; and assistance with finalizing the figure plates by Mr Tim Lanigan (Frederick, MD).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Where warranted for experimental studies, all procedures used to prepare images of animal specimens were performed in accordance with regulations and established guidelines for humane treatment of research animals and were reviewed and approved in advance by an Institutional Animal Care and Use Committee.
