Abstract
Ophidiomycosis (snake fungal disease) is caused by the fungus Ophidiomyces ophiodiicola. As ophidiomycosis is difficult to study in free-ranging snakes, a reliable experimental model is needed to investigate transmission, pathogenesis, morbidity, and mortality, and the effects of brumation and temperature on disease development. Our objective was to develop such a model via subcutaneous injection of O. ophiodiicola conidia in red cornsnakes (Pantherophis guttatus). The model was used to evaluate transmission and the effects of brumation and temperature in co-housed inoculated and noninoculated snakes. All 23 inoculated snakes developed lesions consistent with ophidiomycosis, including heterophilic and granulomatous dermatitis, cellulitis, and myositis, and embolic fungal granulomas throughout the liver and the coelomic connective tissue in 21/23 (91%). In the inoculated snakes, 21% of skin swabs, 37% of exuvia, and all liver samples tested positive by qPCR (quantitative polymerase chain reaction) for O. ophiodiicola. A post brumation skin swab from 1/12 noninoculated snakes that brumated in contact with inoculated snakes tested positive by qPCR, suggesting possible contact transmission. That snake had microscopic skin lesions consistent with ophidiomycosis, but no visible fungal elements. Of the 23 inoculated snakes, 20 (87%) died over the 70-day experiment, with ophidiomycosis considered the primary cause of death; 12 (52%) of the inoculated snakes died during brumation. Overall, this experimental model of ophidiomycosis reproduced skin lesions analogous to those of many natural cases, and internal lesions similar to the most severe natural cases. The study provides tentative experimental evidence for horizontal transmission in brumation, and offers a tool for future studies of this widespread snake disease.
Over the past decade, ophidiomycosis (snake fungal disease), caused by the fungus Ophidiomyces ophiodiicola, has been documented in captive and free-ranging snakes worldwide. 26,38 Ophidiomycosis was first described in 2011, and commonly reported lesions include facial swelling, granulomatous dermatitis, emaciation, and death. 26,35 Since then, ophidiomycosis has been documented in over 30 snake species in North America and Europe. 14,26,38 Furthermore, O. ophiodiicola is thought to be the most common cause of skin lesions in free-ranging snakes in the eastern United States. 7,38 At northern latitudes, and depending on snake species, the prevalence and severity of ophidiomycosis is generally highest immediately after emergence from brumation (ie, a hibernation-like state in ectotherms), a time of severe energy restriction and cohabitation. 17,24,26,40 However, the relative effects of brumation, environmental temperature, and nutritional status on the pathogenesis of ophidiomycosis, including aspects of transmission, are unknown. 23,26
Ophidiomyces ophiodiicola was demonstrated as the causative agent of fungal disease in snakes through experimental studies using 2 different models. 2,27 The first study introduced the fungus into the nasolabial pits of cottonmouths (Agkistrodon piscivorus) using a catheter. 2 The second applied fungus to abraded and nonabraded skin of red cornsnakes (Pantherophis guttatus) using a bandage. 27 Both approaches resulted in lesions analogous to those seen in mild natural ophidiomycosis cases. However, the methods applied in these studies prevented exact quantification of the amount of fungus actively participating in the infection, as conidia could have been lost to the environment or trapped within the bandage, and there was apparent clearing of the fungus in many snakes.
Developing a reliable experimental model of ophidiomycosis could facilitate direct, experimental investigation of the relative impacts of host and environmental factors in disease progression. Such studies can complement and inform work with free-ranging snakes, where natural conditions can be studied but confounding factors or small sample sizes may limit statistical power. We sought to experimentally replicate lesions of naturally acquired ophidiomycosis by subcutaneous inoculation with a known dose of O. ophiodiicola in juvenile red cornsnakes and to examine potential transmission dynamics and the effects of temperature and brumation on pathogenesis.
Materials and Methods
Experimental Animals
Forty-two captive bred, 3- to 6-month-old (4.5–17.0 g, young-of-the-year) red cornsnakes were obtained from 2 established breeding colonies in southern Ontario, Canada. On arrival, a general health assessment of each snake was performed by passive observation of behavior and external physical examination using manual restraint. Each snake was weighed, and we swabbed the skin along the dorsal, lateral, and ventral surfaces of the body, including the head, using a dry polyester-tipped applicator. Swabs were stored dry in individual cryovials at −20 °C. Snakes were initially housed individually in 46.9 × 21.5 × 21.5 cm polysulfonate vent rack cages (Allentown Cage and Equipment) in a biosafety level 2 facility at the University of Guelph. Each enclosure contained a halved plastic container for cover, a clay water dish, and paper lining. Snakes were provided with freshwater ad libitum and a ∼2.5 g neonatal mouse (Tails and Scales) once per week, except during prebrumation and brumation periods. Snakes were acclimated to the laboratory environment for 3 weeks prior to inoculation, during which time they were maintained at 22 °C to 28 °C on a 12-hour light and 12-hour dark cycle (Supplemental Fig. S1). This study was approved by the University of Guelph Animal Care Committee (Animal Use Protocol 3812) and conducted in accordance with applicable Canadian Council on Animal Care guidelines.
Fungus
The fungus stock consisted of O. ophiodiicola isolate UAMH 11863 from the University of Alberta Microfungus and Herbarium Centre for Global Microfungal Biodiversity (Toronto, Ontario, Canada) grown on potato dextrose agar (PDA) substrate. This sample was collected from a skin lesion of an eastern foxsnake (Pantherophis vulpinus, 2015 001) from Point Pelee National Park, Leamington, Ontario, and submitted by IDEXX Laboratories.
Conidia for the inoculum were cultured at 26 °C for 22 to 28 days on irradiated Sabourand’s agar or PDA plates and harvested by flooding the plates with 7 mL of phosphate-buffered saline with 1% Tween 20 (PBST). A sterilized glass stick was used to remove fungus from the plate. Approximately 3 mL of fluid was removed from each plate using a sterile plastic transfer pipette. Another 5 mL of PBST was added to this fluid, which was then vortexed for 2 minutes, filtered through a sintered glass wool plug, and centrifuged at 4000 × g for 10 minutes. The resulting conidia pellets were washed once with 1 mL of saline, vortexed for 5 seconds, and centrifuged at 4000 × g for 5 minutes. Conidia were enumerated using a hemocytometer and resuspended in 0.2 mL of sterile saline to approximately 105 conidia/mL. This inoculum dosage is similar to the number of conidia used in previous infection studies. 2,27
Experimental Design
Snakes were assigned to 1 of 3 groups to include a range of weights and ages within each: a fungus-inoculated group (n = 23; hereafter referred to as “inoculated”); snakes which were housed with inoculates during brumation, but not injected with any material (n = 12; hereafter referred to as “contact”); and sham-inoculated controls (n = 7; hereafter referred to as “control”). At 0 days post-inoculation (dpi), inoculated snakes were injected subcutaneously with 0.2 mL of conidia solution with a 25 g needle, equally divided between 2 locations: right lateral cranial body wall (immediately caudal to the head) and right lateral mid-body wall. Control snakes were sham-inoculated with sterile saline by the same method. Initially, control, contact, and inoculated treatment groups were housed on separate sides of the same room, and husbandry and handling of treatment groups was always completed in the order listed. Throughout the experiment, all snakes were monitored at least once daily for clinical signs, including the development of skin lesions and lethargy. All gross skin lesions were measured, described, and recorded. Lethargy was recorded when snakes were subjectively slow to react to handling in comparison to other snakes or were unresponsive. Snakes were also weighed and skin-swabbed weekly, except during the brumation period (21–49 dpi), during which time disturbance of snakes was minimized.
Environmental conditions from 1 to 21 dpi included 1 week with a 12-hour light and 12-hour dark cycle, weekly feedings, and ambient temperatures maintained at 26 °C to 28 °C with 10-cm-wide electrical heat tape at 30 °C running underneath enclosures. Two weeks prior to brumation, feedings and heat tape usage were discontinued, and the light cycle and ambient temperatures gradually decreased (by 1–2 hours per day and 1–2 °C per day) to a 4-hour light and 20-hour dark cycle and 10 °C, respectively. Temperatures were monitored using a digital thermometer placed on the shelving in each room and an analogue thermometer placed directly on the heat tape.
In addition, 7 days prior to initiation of brumation, snakes were individually placed into 2 cm of fresh water for 5 minutes each to stimulate defecation and thus clear the gastrointestinal tract.
The brumation period was from 21 to 49 dpi, during which time snakes were grouped together to simulate a communal hibernaculum. Inoculated and contact snakes were housed in 6 treatment groups of 3 inoculates and 2 contact snakes each, and control snakes were housed in 2 treatment groups, each with 3 or 4 snakes. Ambient brumation temperatures were between 4 °C and 13 °C and the daily light cycle was 1 hour of light and 23 hours of dark. Snakes were visually monitored during this time but not handled or fed.
Snakes were roused from brumation starting at 49 dpi by returning them to individual housing, gradually increasing the temperature by 1 °C to 2 °C per day, increasing the light cycle by 1 to 2 hours per day to 12 hours each of light and dark, and resuming weekly feedings. Half of the remaining snakes across both treatment groups were placed into a “spring room” in which the temperature was gradually increased to a maximum ambient temperature of 19 °C with heat tape at 24 °C, while the other half was placed into a “summer room,” in which the temperature was increased to a maximum of 29 °C with heat tape at 32 °C.
All snakes that survived to the predetermined endpoint of 70 dpi were anesthetized with an overdose of ketamine (80 mg/kg) administered via intramuscular injection into the dorso-cranial epaxial muscles. Snakes were weighed and swabbed after 15 minutes to allow for snakes to reach a deep plane of anesthesia, which was determined by absent jaw tone, absent righting reflex, and no response to tail pinching. Death was assured by severing of the spinal cord just caudal to the head.
Pathology
Postmortem examination was performed within 60 minutes of finding a snake dead and within 15 minutes of euthanasia at the endpoint. There were up to 12 hours between visual evaluations of the snakes for clinical signs and mortality. Gross findings were documented, and representative samples of fresh liver and skin were collected and stored at −20 °C. The remainder of each carcass was preserved in 10% neutral-buffered formalin.
After fixation, each carcass was serially cross-sectioned at 3-mm intervals from the rostrum to cloaca and embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin (HE), as well as Gomori’s methenamine silver stain (GMS) for tissues from all inoculates, allowing microscopic examination of all major organs at multiple levels for each snake. Histopathology of the skin was scored following the grading scheme previously described. 30 Briefly, points were assigned for degree of inflammation, extent of necrosis, ulceration, and serocellular crusting, and summed to determine a final histologic grade of I, II, or III corresponding to mild, moderate, and severe skin lesions, respectively. Liver histopathology was assessed by analyzing the most affected and intact section from each snake that comprised at least 30% of the diameter of the coelom, to ensure accurate representation of as maximal a section of liver as possible. The percentage of the analyzed liver section affected was measured using the software ImageJ. 37
All snakes were subjectively assessed microscopically for poor nutritional condition by evaluation of adipose stores (pericardial and visceral) and degree of muscular and pancreatic atrophy. Muscle atrophy was identified when there were angular and variably sized muscle fibers and pancreatic atrophy when there were reduced zymogen granules. Comorbidities with other infectious agents other than O. ophiodiicola were not assessed beyond microscopic examination for consistent lesions.
Polymerase Chain Reaction (PCR) Test
A commercially available kit (DNeasy Plant Mini Kit, Qiagen) was used following the manufacturer’s instructions to extract nucleic acid (DNA) from samples including skin swabs, fresh-frozen skin, and fresh-frozen liver. The internal transcribed spacer between the 18S and 5.8S ribosomal RNA gene specific for O. ophiodiicola was amplified using a TaqMan qPCR assay as described in Allender et al 3 using primers and probe from IDT (Coralville) and master mix from Roche. All histology processing and staining and qPCR (quantitative PCR) tests were performed at the Animal Health Laboratory (Guelph, Ontario, Canada).
Corticosterone Levels in Exuvia
All shed skin samples collected from control, contact, and inoculated snakes (both pre- and post-inoculation) of at least 0.002 g in weight were extracted for corticosterone analysis based on the protocol previously described, 10 with minor modifications. In brief, skin samples were cut into 0.5 cm pieces and weighed (up to 0.02 g) into glass vials. Samples were washed briefly by adding 4 mL of 100% methanol and vortexing for 10 seconds. The wash methanol was removed and the sample left for 30 minutes at room temperature for any alcohol residue to evaporate. The samples were then extracted by adding 100% methanol at a ratio of 0.005 g/mL and mixing on a rotator for 24 hours. The extract was then transferred to a clean vial, evaporated under air, and stored at −20 °C until analyzed.
Quantification of corticosterone concentrations in the shed extracts was done by enzyme immunoassay (EIA) as previously described. 9 Extracts were reconstituted in EIA buffer for a final 20× concentration (or less if sufficient extract volume was not available; 2–15× concentration in 25% of samples). Corticosterone standards (Steraloids; Q1550) ranged from 39 to 10 000 pg/mL. Assay components were diluted as follows: goat anti-rabbit IgG polyclonal antibody (Sigma-Aldrich), 0.25 μg/well; corticosterone polyclonal antibody (CJM006; C. Munro, University of California, Davis, CA), 1:200 000; corticosterone-horseradish peroxidase conjugate (C. Munro, University of California, Davis, CA), 1:1 000,000. Results are presented as ng of corticosterone/g of shed skin.
Statistical Analysis
All statistical analyses were performed in R, 33 with survival analysis done using the survival package 41 and figures produced using the package ggplot2. 44 Risk difference was calculated to compare the development of gross lesions between inoculated and control snakes. A simple linear regression was used to assess for an association between percentage of liver affected by granulomas and severity of microscopic lesions, as well as liver Ct values. For categorical variables, Kaplan-Meier survival functions were compared using the log rank test, and for continuous variables, analyses were performed using a Cox proportional hazards model, to identify correlations with survival time. A one-way analysis of variance was used to compare shed rates and corticosterone levels in exuvia among different experimental groups and nutritional conditions. A simple linear regression was used to assess for an association between weight and shed rate. For all statistical analyses, a significance level of 5% was used (ie, α = 0.05).
Results
Gross Pathology
Of the 23 inoculated snakes, gross skin lesions developed in 11 (48%; 95% confidence interval [CI95%]: 27.41–68.24, P = .029). Lesions included skin and scale discoloration (n = 2; 9%), thickened scales (n = 3; 13%; Fig. 1), dysecdysis (n = 5; 22%), vesicles (n = 3; 13%; Fig. 2), ulcers (n = 2; 9%), and serocellular crusts (n = 6; 26%; Fig. 3; Table 1). The earliest death (snake I.01) occurred at 3 dpi and this snake did not exhibit gross skin lesions. The earliest skin lesion (snake I.06) was noted at 7 dpi and consisted of a small number of discolored, raised scales, and mild swelling at the cranial inoculation site. Throughout the experiment, skin lesions progressed from scale discoloration to vesicle formation and later, to serocellular crusting.
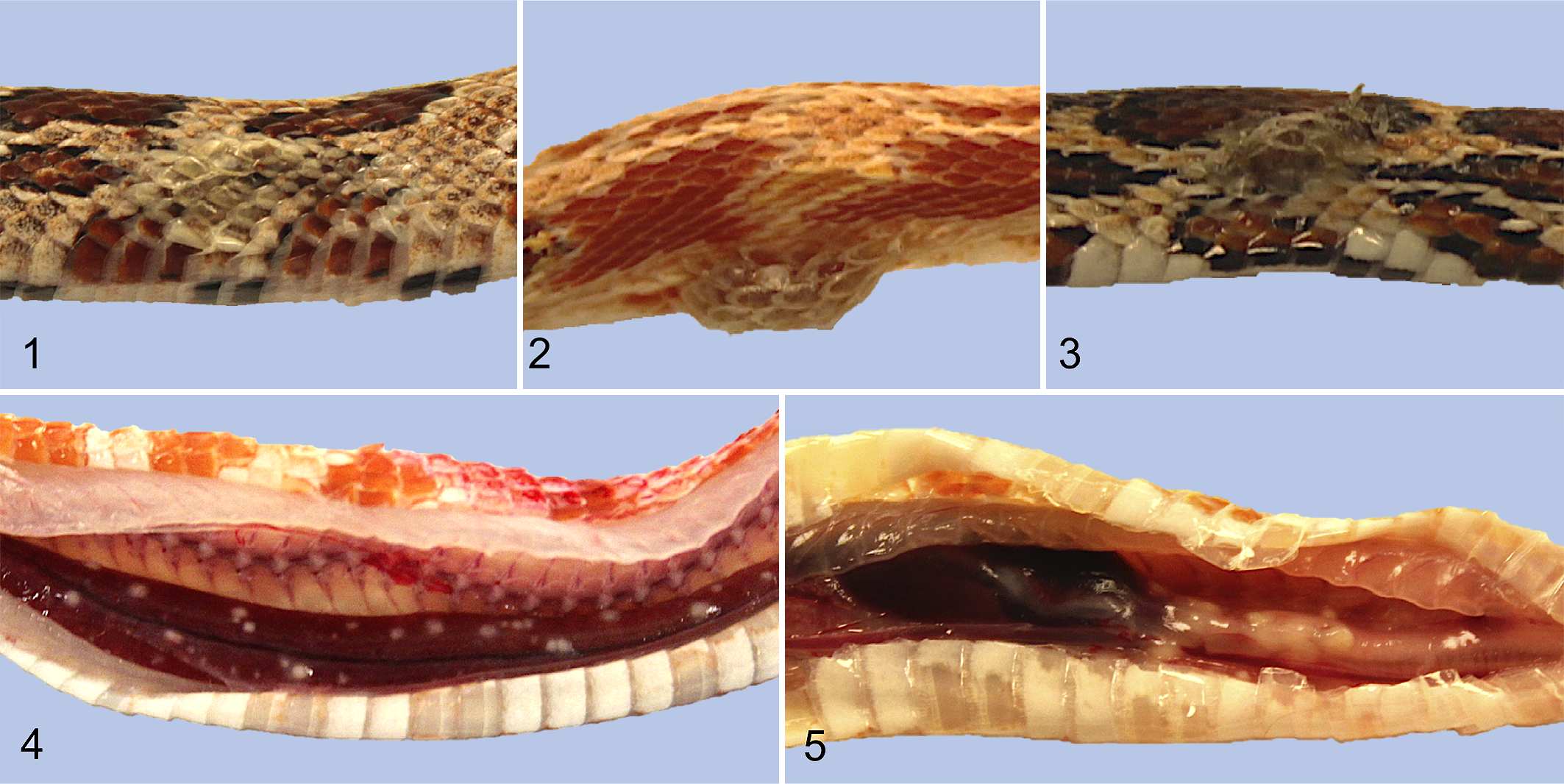
Experimentally induced Ophidiomyces ophiodiicola infection via subcutaneous injection, red cornsnakes (Pantherophis guttatus).
Summary of Gross and Microscopic Lesions of Carcasses From Cornsnakes (Pantherophis guttatus) Experimentally Infected With Ophidiomyces ophiodiicola.
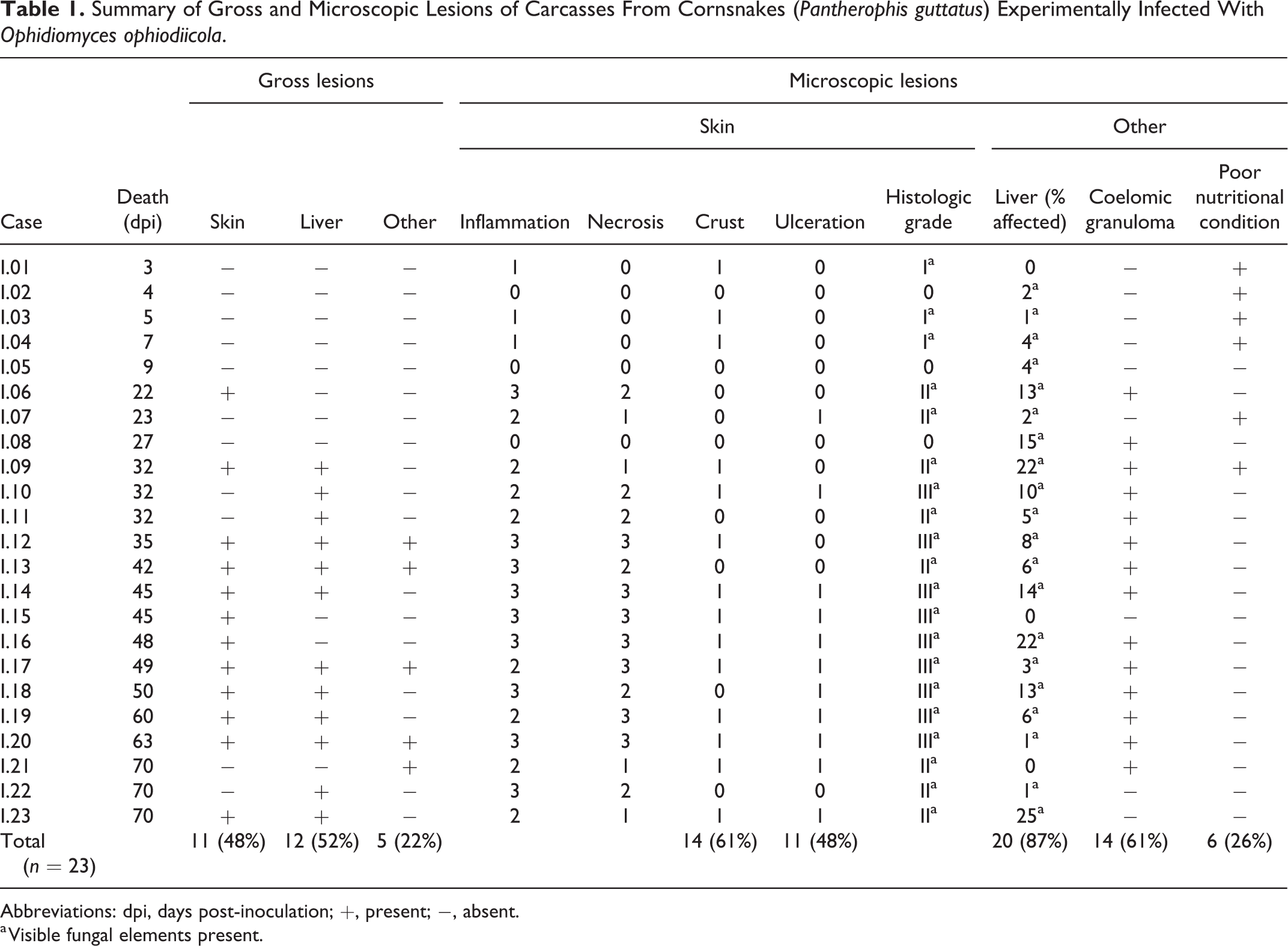
Abbreviations: dpi, days post-inoculation; +, present; −, absent.
a Visible fungal elements present.
Gross internal lesions were observed in 13/23 of the inoculated snakes (57%; CI95% = 31.76–72.50, P = .010). Of these, 12 had hepatic granulomas (52%; CI95% = 31.76–72.50, P = .029; Fig. 4). Five (22%) had granulomas within the intracoelomic connective tissue. These granulomas were primarily along the trachea and adjacent to the heart base (Fig. 5) but were less commonly throughout the remainder of the coelomic cavity. Granulomas were discrete, nodular, solid, white, and chalky, and ranged from <1 mm to 3 mm in diameter. None of the contact or control snakes developed gross lesions consistent with ophidiomycosis.
Histopathology
All 23 of the inoculated snakes developed microscopic lesions that contained visible fungal elements, either in the skin or visceral organs (Table 1). Fungal hyphae were amphophilic, 4 to 6 µm wide, septate, and had undulating walls and acute angle branching consistent with O. ophiodiicola. Conidia were rectangular and approximately 3 to 8 × 3 μm. The fungal elements stained positively with GMS. None of the control snakes developed notable microscopic lesions and none had visible fungal elements (Fig. 6).
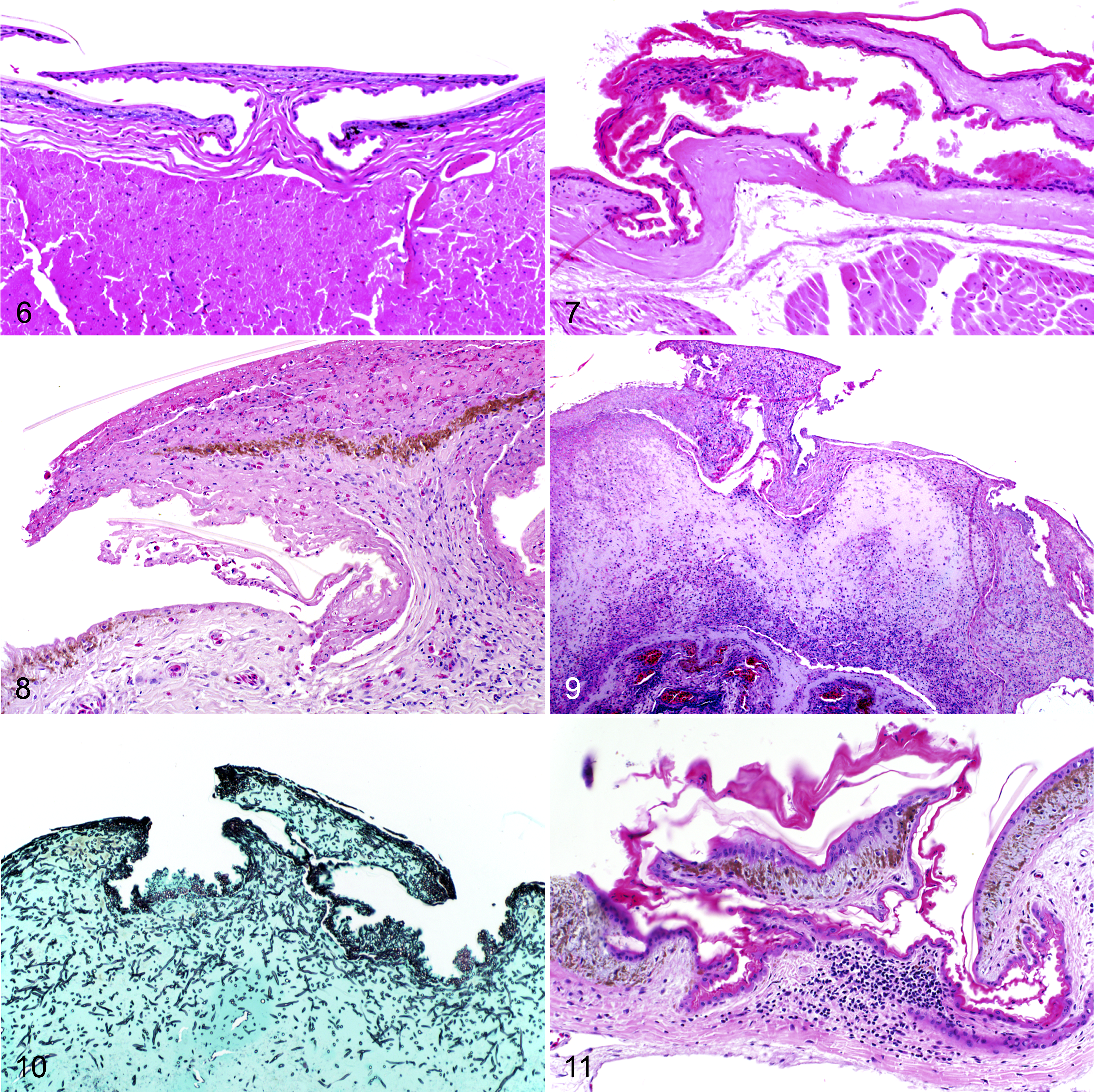
Experimentally induced Ophidiomyces ophiodiicola infection via subcutaneous injection, red cornsnakes (Pantherophis guttatus).
Of the 23 inoculated snakes, 20 (87%) developed microscopic skin lesions consistent with O. ophiodiicola infection. Of these, 3 displayed mild inflammation, with only a few scattered heterophils within the dermis; 8 had moderate inflammation, with heterophils, macrophages, and lymphocytes both in the epidermis and dermis; and 9 had severe inflammation, with a similar population of inflammatory cells expanding into the underlying musculature, and varying degrees of associated necrosis. The degree of necrosis was mild in 4/23 inoculated snakes, moderate in 5 with coalescing areas that affected less than a single scale width, and severe in 7 with coalescing foci of necrosis affecting an area greater than one scale width. The skin lesions had serocellular crusting in 14/23 (61%) of snakes, and 11 (48%) had epidermal ulceration. Collectively, 3 snakes (13%) had a histologic grade of I (Fig. 7), 8 (35%) had a grade of II (Fig. 8), and 9 (39%) had a grade of III (Fig. 9). The skin lesions had intralesional fungus consistent with O. ophiodiicola in 20/23 (87%) of snakes (Fig. 10). The majority of these lesions were limited to the sites of inoculation but 2 snakes (I.06 and I.10) had skin lesions on contralateral body walls, within the same body segment (ie, the same histologic section). Snake I.06 had a grade II skin lesion on one side and a grade I lesion on the opposite, both with intralesional fungi. In snake I.10, there was a grade III lesion on one side and multiple granulomas with intralesional fungus within the superficial and deep dermis on the opposite body wall.
One of the contact snakes developed skin lesions consistent with ophidiomycosis and tested positive using qPCR on a skin swab at one time point (described below), but no fungus was visualized in 20 histologic sections stained with GMS. In 3 sections taken at least 3 mm apart at the level of the trachea and esophagus, there was mild serocellular crusting affecting 1 to 2 scales, with a few lymphocytes and plasma cells within the superficial dermis (grade I skin lesions; Fig. 11).
Microscopically, 21/23 (91%) experimentally inoculated snakes had internal lesions consistent with O. ophiodiicola infection. Most snakes with internal lesions (20/21; 95%) had multiple, randomly scattered, well-circumscribed granulomas throughout the liver (Fig. 12). These granulomas effaced an average of 9% (range 1% to 25%) of the liver parenchyma and consisted of central necrosis that often contained fungal hyphae surrounded by a rim of macrophages, heterophils, and lymphocytes. Subcapsular hepatic granulomas often protruded into the coelomic cavity. The percentage of liver affected by granulomatous inflammation was not significantly associated with the severity of microscopic skin lesions (F = 0.622, r 2 = 0.029, P = .439; linear regression).
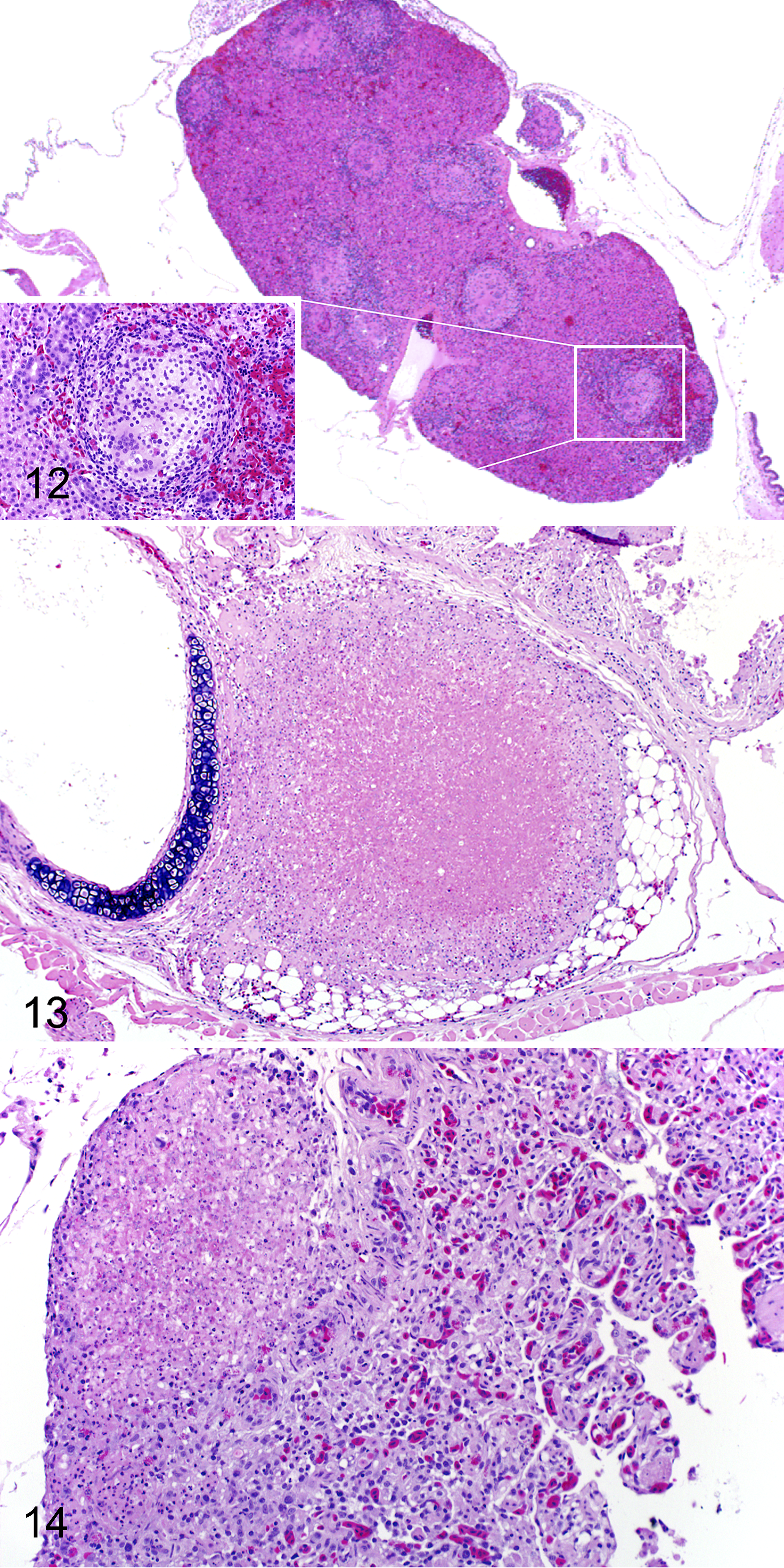
Experimentally induced Ophidiomyces ophiodiicola infection via subcutaneous injection, red cornsnakes (Pantherophis guttatus).
Fourteen of the snakes with internal lesions attributed to O. ophiodiicola infection (61%) had granulomas within the intracoelomic connective tissue. These granulomas were similar to those described in the liver and were located at various levels throughout the coelomic cavity (ie, peritracheally in the cranial coelom; Fig. 13, and in the caudal coelom within the mesentery and around the kidney). Granulomas were rarely observed in other organs, including the lung (snakes I.12 and I.14) and thyroid gland (snake I.09). Pulmonary granulomas were at the base of the faveolar septa, adjacent to the pleura (Fig. 14).
Six of the 23 inoculated snakes (26%), 3/12 contact snakes (25%), and 5/7 control snakes (71%) were deemed to be in poor nutritional condition at the time of postmortem based on microscopic examination. Four inoculated snakes (snakes I.02, I.04, I.18, I.20) had mild, segmental, heterophilic enteritis, and 2 snakes (snakes I.04 and I.07) had mild, heterophilic pneumonia, all with no apparent cause. Two contact snakes, which had diffusely dull skin at postmortem, had mild to moderate numbers of eosinophils within the epidermis of approximately 75% of scale hinge regions. Both of these snakes had a prominent stratum intermedium, which sometimes contained clefting and was overlain by a second β-layer. These changes were consistent with oncoming ecdysis. 28 None of the control snakes had heterophilic or eosinophilic inflammation in any tissue. There was mild variation in the lymphoid tissue of the thymus and spleen between snakes, primarily with regard to the prominence of the cortex; however, subjectively there was no discernable association with infection status or experimental time period.
Survival
Snakes that were still alive at the experimental endpoint (70 dpi) and required censoring from the population included 3 inoculated snakes (13% survival during the experimental period), 10 contact snakes (83% survival), and 5 control snakes (71% survival). The mortality rate among inoculated snakes over the course of the 70-day experimental period was 87%. The mean survival time for inoculated snakes, after censoring, was 35 days (N [n] = 23 [20], 95% CI = 27–50, range = 3–63 days; Fig. 15). Mean survival time could not be calculated for contact and control snakes because the survival function never reached 0.5 for either group. Ophidiomycosis was considered to be the primary cause of mortality in all inoculated snakes, as all had lesions consistent with ophidiomycosis at the time of death and no gross or microscopic lesions consistent with another disease process.
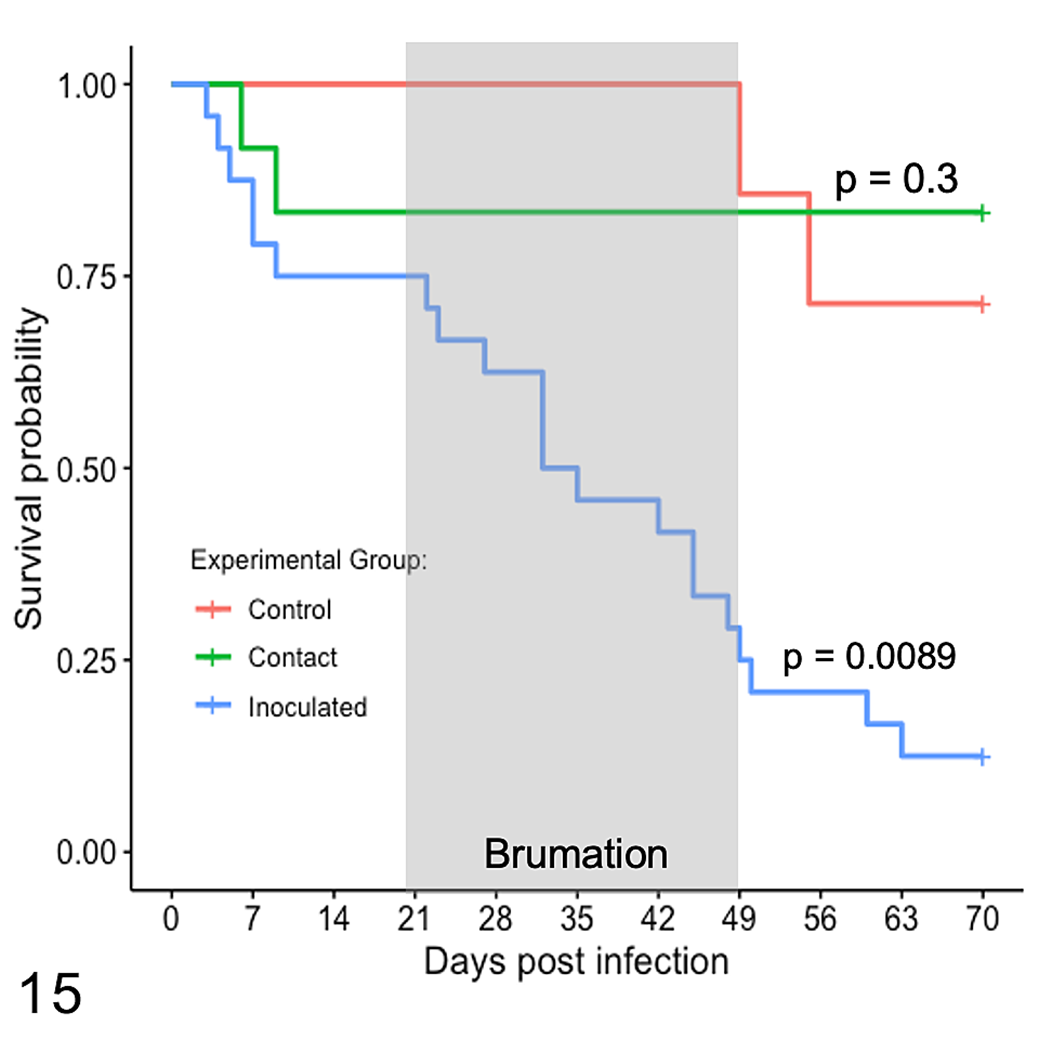
Kaplan-Meier survival curves for 23 juvenile Pantherophis guttatus subcutaneously inoculated with Ophidiomyces ophiodiicola (inoculated), 12 contact snakes (contact) housed with inoculated snakes for 4 weeks of simulated brumation for days 21 to 49 post-infection and 7 control snakes (control) inoculated with sterile saline and housed separately. Survival functions were compared to the control snakes using the log rank test.
Based on the Kaplan-Meier survival functions, survival rates were significantly different between inoculated and control snakes (P = .0089) and between inoculated and contact snakes (P = .0004). Survival rates were similar between contact and control snakes (P = .300).
Ophidiomycosis was not influenced by body weight in causing mortality, in either additive or multiplicative analyses, and body weight alone had no impact on survival (P = .768). Body weight at the time of inoculation as a continuous variable in univariate analyses met the proportional hazards assumption, but violated it when used as an additive term. However, for snakes in the lowest 25% percentile of weight at inoculation (ie, <7.05 g, n = 11), the Kaplan-Meier survival function was significantly different (P = .016) for those with a “poor” nutritional condition compared to the others. The mean survival time for snakes in poor nutritional condition, after censoring, was 23 days (N[n] = 11 (9), CI95% = 9 to infinity, range: 7–54 days) and the mean survival time for the others could not be determined as the survival function never was below 0.5.
We also found differences among the Kaplan-Meier survival functions for the 4 histologic skin grades (P < .0001), although there was no clear pattern of association between grade and survival. The mean survival time for snakes was 7 days for inoculated snakes with grade I lesions, 9 days for snakes with grade 0 lesions, 30 days for snakes with grade II lesions, and 49 days for snakes with grade III lesions. The percentage of liver microscopically affected by granulomatous inflammation microscopically as a continuous variable in univariate analysis met the proportional hazards assumption and did not have a statistically significant effect on survival (P = .7).
During brumation (40% of the duration of the experimental trial), 12 of the 23 (52%) inoculated snakes died of ophidiomycosis. During that time, one control snake died of emaciation and zero contact snakes died.
At the start of the post-brumation period, there were 6 surviving inoculated snakes, 10 contact snakes, and 6 control snakes, which were divided evenly between the “spring” and “summer” rooms. All 3 of the inoculated snakes and 1 of the control snakes in the “spring” room died, while no snakes in the “summer” room died. A log-rank test could not be performed on the Kaplan-Meier survival curves because the assumptions were failed.
qPCR Test Results
At least one sample from each O. ophiodiicola–inoculated snake tested positive for O. ophiodiicola by qPCR at some point during the experimental trial (Table 2), but results were inconsistent among sample types and individuals. Serial skin swab samples from 10 inoculated snakes consistently tested negative, including swabs from 2 snakes that survived for the full 70 days and were swabbed 7 times. Cycle threshold (Ct) values for positive skin swab samples ranged from 31.6 to 39.9. Snakes frequently alternated between having positive and negative skin swabs at different time points throughout the experimental trial. The qPCR sensitivity for skin swabs from inoculated snakes was 21% (n = 77, 95% CI = 12.37–31.54) and the false negative rate was 79%. All swabs of the cages (n = 8) and lining papers of inoculated snakes at 49 dpi (ie, immediately after brumation) tested negative by qPCR.
Summary of Quantitative PCR Results From Cornsnakes (Pantherophis guttatus) Experimentally Infected With Ophidiomyces ophiodiicola or in Contact With Infected Snakes.a
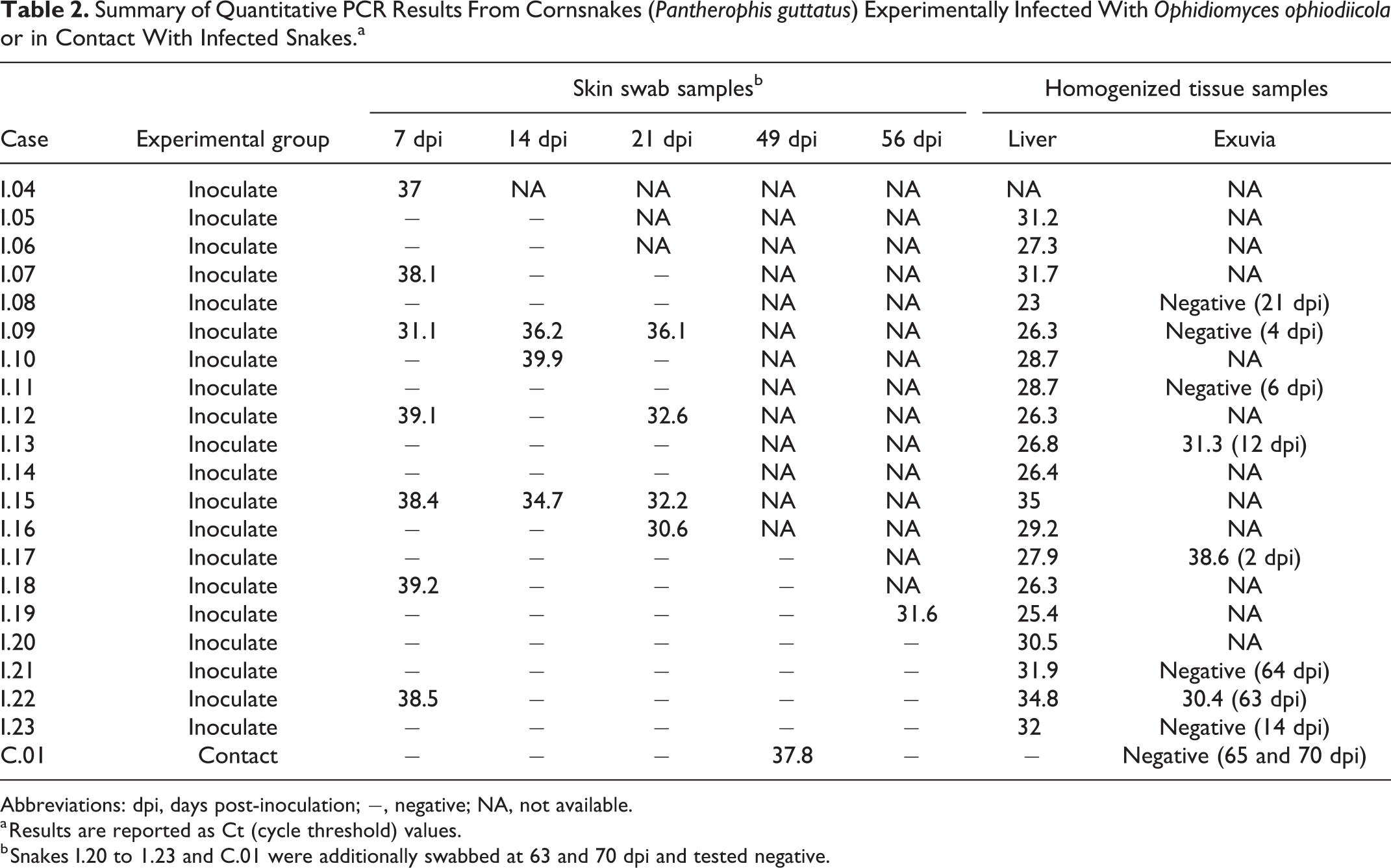
Abbreviations: dpi, days post-inoculation; −, negative; NA, not available.
a Results are reported as Ct (cycle threshold) values.
b Snakes I.20 to 1.23 and C.01 were additionally swabbed at 63 and 70 dpi and tested negative.
All homogenized liver samples tested from inoculated snakes were positive for O. ophiodiicola with Ct values ranging from 23.0 to 34.8 (n = 19). There was no association between liver Ct values and the percentage of livers affected by granulomatous inflammation, as determined histologically (P = .075; linear regression). Three of 8 homogenized exuvia samples from inoculated snakes tested positive with a sensitivity of 37% (n = 8, 95% CI = 8.52–75.51) and a false negative rate of 63%. Homogenized exuvia yielded a positive qPCR result for 2 snakes that never had a positive qPCR on a skin swab sample despite skin swabbing within 0 to 7 days of skin shedding.
A skin swab sample from a single contact snake tested positive on 49 dpi, the first time point after the brumation period, with a Ct value of 37.8. Skin swabs from this snake at the 3 remaining time points tested negative, as did the homogenized liver sample and 2 exuvia samples collected on 65 and 70 dpi.
Ecdysis Frequency and Cortisol Levels
Among all snakes, there were 47 ecdysis events during the acclimation and post-inoculation periods. Fourteen snakes did not shed skin during the study (13 weeks) while 10 snakes shed 2 to 3 times in that period. During the post-inoculation period, the mean ecdysis frequency was 0.015 sheds/day for inoculated snakes, 0.006 sheds/day for contact snakes, and 0.012 sheds/day for control snakes. Ecdysis frequency was similar among the experimental groups (F = 1.038, P = .363). Ecdysis frequency decreased with weight (F = 4.921, r 2 = 0.088, P = .032) and with poor nutritional condition at the time of inoculation (F = 4.734, P = .035).
The mean corticosterone concentration in exuvia was 3.69 ng/g from all shed events (n = 47, range = 0.78–22.54 ng/g, SD = 4.24). All exuvia were representative of preinoculation circulating corticosterone levels, as no snake shed more than once after inoculation. Corticosterone levels were similar across weights (F = 0.951, r 2 = −0.001, P = .335), between “poor” and “adequate” nutritional conditions (F = 2.203, P = .146), and across experimental groups (F = 0.913, P = .443).
Discussion
This study demonstrates that subcutaneous injection of O. ophiodiicola in juvenile red cornsnakes causes ophidiomycosis with skin lesions analogous to those observed in natural infections and previous experimental challenges, as well as internal lesions consistent with severe natural infections. 2,26,27,32 Inoculated snakes displayed varying degrees of heterophilic and granulomatous dermatitis, cellulitis, and myositis, as well as granulomas throughout the liver and coelomic connective tissue. One uninoculated snake in contact with infected snakes tested positive for O. ophiodiicola by qPCR and developed characteristic skin lesions, tentatively supporting the hypothesis of contact transmission during brumation.
In general, dermatomycoses in reptiles are initiated through skin contact with fungal spores or conidia, which germinate to colonize the keratinized layers and eventually penetrate into the stratum spinosum and stratum germinativum. 19 This triggers a heterophilic response, leading to necrosis and granuloma formation. 19,27 In addition to causing skin lesions over the head and body, ophidiomycosis often involves the nasolabial pits, especially in vipers, which have been proposed as another common point of entry for O. ophiodiicola. 2 Based on this premise, fungus was introduced topically in previous experimental models of ophidiomycosis. 2,27 Allender et al 2 used 105 colony forming units in aqueous solution per dose introduced to the surface of nasolabial pits, and Lorch et al 27 used bandages soaked in 104-5 conidia adhered to the skin, which was sometimes abraded prior to application. The drawback of topical application in these models is that the quantity of applied conidia that actively participate in the infection is unknown, as some may be lost to the environment 2 or are retained within the bandage. 27 The lesions that followed nasolabial inoculation were analogous to, but less severe than, those seen with naturally acquired ophidiomycosis and contained highly variable quantities of fungus. In addition, some snakes appeared to clear the infection. 2 Topical application of fungi via bandages also produced comparable skin lesions to natural ophidiomycosis, but some snakes appeared to clear infection through ecdysis, leading to multiple inoculation attempts to achieve infection, complicating the interpretation of study results. 27 Our use of subcutaneous injection guaranteed delivery of known quantities of fungal conidia to the tissues of each snake, and made it unlikely that snakes would clear the infection through ecdysis. Also, this ensured that inoculated snakes were infected for the brumation phase of the experiment, to assess for horizontal transmission to contact snakes.
The systemic spread of O. ophiodiicola fungus in the experimentally inoculated snakes in the present study is distinct from prior experimental infection results, but has been reported in severe natural cases. 2,27,32 Using subcutaneous injection as the mode of infection promoted systemic dissemination of the fungus, as it allowed the fungus to bypass the epithelial barrier and gain access to internal tissues. Although direct introduction of large numbers of conidia into the subcutaneous tissues is unlikely to occur naturally, fungus could be introduced in natural infections via penetrating wounds, fungal infection of mucous membranes, or deep ulceration associated with dermal ophidiomycosis. In naturally acquired infections, ophidiomycosis granulomas have been observed in the liver, spleen, kidney, lung, air sacs, coelomic fat, brain, salivary glands, and ovaries, in addition to skin. 13,36,42 In one report of naturally acquired ophidiomycosis in free-ranging water and terrestrial snakes, 37% of these snakes had visceral fungal lesions, with the liver being most commonly affected by granulomas, and there were sometimes no associated cutaneous lesions. 32 In reports of disseminated O. ophiodiicola granulomas with dermal lesions, mucous membranes, such as the cloaca or conjunctiva, were often affected. 13,36,42 Discrepancies between reported prevalence of visceral lesions may be due to a combination of differences in susceptibility among species and perhaps a lack of thorough postmortem examination in cases focusing on skin lesions.
The random distribution of the fungal granulomas throughout the liver and coelomic cavity of the experimentally inoculated snakes, and lack of generalized coelomitis, suggests hematogenous spread from the injection sites, as opposed to direct introduction during inoculation or direct extension through the body wall. It is unlikely that the fungus moved along fascial planes, given the intact hepatic capsule serving as a barrier and the high number of parenchymal hepatic granulomas. In addition to visceral dissemination of the fungus in the present study, the fungus also spread within the skin. Possible explanations for this finding include hematogenous spread within the dermis, or skin contamination with fungus by contact with the environment, fomites, or other infected snakes.
Despite direct inoculation of cornsnakes with a consistent, known fungal dose in the present study, the severity of resulting lesions varied widely. The first snake to die after inoculation (3 dpi) had grade I skin lesions and no granulomas in the liver. Of the inoculated snakes that survived to the predetermined endpoint, all had histologic grade II skin lesions and 0% to 25% of the liver affected by fungal granulomas. Although thin body condition appeared to affect mortality, as discussed below, it did not seem to correlate with the severity of skin or internal ophidiomycosis lesions (Table 2). Lesions did not become noticeably more severe during brumation, although it is difficult to draw conclusions as all snakes were placed into brumation. The post-brumation temperature may affect skin lesion development, as all 3 inoculated snakes that survived to the endpoint in the warmer room had less severe skin lesions than those in the cooler room, but samples sizes were too small to achieve significance. This is consistent with findings of increased severity of ophidiomycosis lesions with lower air temperatures. 29 Stress levels, as deduced from exuvia corticosterone levels, and concomitant immunosuppression, also did not appear to have a role in lesion development. It is clear that, even in an experimental setting, it is difficult to untangle the individual factors affecting lesion development and further research is required.
The most important predictor of survival time in this experiment was inoculation with O. ophiodiicola, which drastically reduced survival. The mortality rate of 87% in inoculated snakes in the present study is higher than those in other experimental trials, 2,27 but similar to that reported in naturally infected, free-ranging eastern massasauga rattlesnakes. 1,5 The high mortality rate reported here is likely due to a combination of a high fungal dose injected subcutaneously leading to systemic dissemination and the use of juvenile snakes. Research on ophidiomycosis in this young age class is lacking, but reports in neonates indicate rapid disease development with high mortality. 11,39 Body weight at the time of inoculation was not significant when considered as a continuous variable. This is consistent with other studies in which there were no associations between O. ophiodiicola infection and body weight, sex, age-class, or body length in wild snakes. 12,25 However, snakes with low body weights at the time of inoculation had reduced survival times, across all experimental groups. This suggests that there is a threshold for which reduced body weight becomes important in terms of survival.
Survival time was not associated with microscopic lesion severity in skin and liver of snakes in the present study; however, additional research including larger sample sizes would help clarify these associations. Microscopic skin lesion severity may not have been a significant predictor of mortality considering that the skin barrier was bypassed by using subcutaneous injection as the inoculation method. Although liver disease may have contributed to morbidity and mortality, it was likely not the sole factor based on lack of association between the percentage of liver affected and mortality. The amount of fungus in the liver, as estimated by the Ct values from homogenized liver, also did not correlate with the percentage of the liver affected by granulomas. This could be due to varying degrees in host response to the fungus, or different distribution of fungus and granulomas in the liver as different samples were used for histopathology and qPCR. Overall, our findings indicate that skin and liver lesion severity is not a good predictor of mortality when juvenile cornsnakes are subcutaneously inoculated with O. ophiodiicola.
Temperature and brumation are suspected to be major drivers in the epidemiology of ophidiomycosis in free-ranging snakes in temperate climates. 12,25 Brumation is a prolonged period of low temperatures leading to decreased activity in ectotherms that can negatively impact immune function and metabolic rate. 15,22,34 In reptiles and other ectotherms, there is seasonal cyclicity in lymphoid tissue in both the thymus and the spleen, with abundance peaking in the spring and the autumn. 45,46 Seasonal cyclicity has also been observed in the bactericidal ability of plasma in snakes, and it is lowest in the fall. 29 Overall fungal pathogenicity can also be affected by lower environmental temperatures. 20,21 Snakes in temperate climates may attempt to thermoregulate through behavioral modifications such basking in the open at inappropriate times of year, perhaps to induce a fever response and decrease fungal proliferation. 40 Over half of the inoculated snakes died during the brumation period, and all of the inoculated snakes in the cooler “spring” room died in the post-brumation period, suggesting that lower temperatures and brumation had a negative impact on survival. Although there was a small amount of variation in the lymphoid tissue between snakes, there was no association with infection status or experimental time point (ie, pre-brumation, brumation, and post-brumation). We may not have seen the typical lymphoid cyclicity as the experimental brumation period was shorter than in most natural circumstances. Despite the potential role of temperature in pathogenesis, ophidiomycosis has been observed in captive snakes with no history of temperature change as well as in snake populations that remain active (ie, non-brumating) year-round. 16,26 Overall, a better understanding of the role of environmental factors such as temperature, which are linked to behavioral activities such as brumation, in pathogenesis of ophidiomycosis, is needed.
Quantitative PCR of skin swabs is a commonly used diagnostic tool for ophidiomycosis in snakes and allows for noninvasive sampling. 18 However, results from inoculated snakes in the present study were inconsistent across individuals and over time. The high false negative rate for qPCR tests of skin swabs from inoculated snakes in the present study (79%) is similar to false negative rate reported for skin swabs of wild snakes with clinical disease (73%). 18 Such false negative rates may be attributable to swabbing techniques and clearance of fungus on the skin surface after it invades deeper into the underlying tissue. 2,18 Fungal abundance in skin lesions of naturally infected snakes varies, with increased fungus in abnormal scales and reduced fungus in ulcerative lesions based on qPCR results. 8 This is the basis for the recommendation of aggressive swabbing of skin lesions and/or skin biopsies for diagnosis. 2,4,6,31 The false negative rate was lower for homogenized exuvia (63%) in the present study; this rate may have been less if whole shed skin samples were tested (partial sheds were tested due to concurrent testing for corticosterones). Exuvia may be a superior sample for qPCR, as O. ophiodiicola is often within the outer layers of the skin and snakes have been observed to clear infections through skin shedding. 27
Transmission ecology, including major routes, represents a knowledge gap in our understanding of the pathogenesis and epidemiology of ophidiomycosis. A single contact snake in the present study attained the classification of “Ophidiomyces present” during brumation as per published case definitions for ophidiomycosis. 8 A single skin swab from this snake tested positive at the first post-brumation sampling time point. This snake did not develop gross lesions but had grade I (ie, low grade) microscopic skin lesions with no visible fungal elements. Given the experimental methods and the lack of skin lesions in control and other contact snakes, we suspect that the microscopic skin lesions and concurrent positive skin swab result represents horizontal transmission. Microscopic lesions were present without fungal elements may suggest the fungi caused the tissue damage but were then cleared prior to death. Alternatively, the snake may have been superficially contaminated with O. ophiodiicola from the environment, but not actually infected, and the low grade lesion seen microscopically at the end of the experiment could have resulted from another process that was not observed in other contact or control snakes. Previous experimental studies have documented infected snakes with microscopic lesions that lacked fungal hyphae, perhaps representing disease recovery phase. 2 This finding of presumed contact transmission indicates that snakes can, at minimum, be exposed to O. ophiodiicola during brumation with infected snakes. Communal brumation sites have been proposed as important for disease transmission due to close contact of snakes and contamination of the shared environment. 25 Some snake species have been shown to have strong brumation site fidelity and are known to share a single burrow, and O. ophiodiicola DNA has been recovered from soil samples in areas with infected snakes. 25,43 However, we did not find environmental contamination of O. ophiodiicola DNA, which may partially reflect a lack of robust sampling efforts. It is possible that subcutaneous injection of O. ophiodiicola reduced the amount of fungus on the skin surface, although fungus was observed on the surface of some snakes (Fig. 10), which limited the amount of environmental and direct contact transmission. Horizontal transmission of O. ophiodiicola has not been otherwise experimentally confirmed, although evidence of transmission has been observed in free-ranging snakes held in captivity. 11,39
Results from previous experimental trials of ophidiomycosis suggest that ecdysis frequency may increase with O. ophiodiicola exposure in adult snakes; however, this was not the case in the present study. 27 This result may have been affected by brumation, which involved an extended period of energy restriction, or by use of juvenile snakes, which have less available energy reserves to dedicate to ecdysis, as exuvia frequency decreased with weight. Exuvia from these ecdysis events were tested for corticosterone concentrations to assess for preexisting physiological stress. The corticosterone levels in exuvia reflect circulating corticosterone 2 weeks prior to the previous shed, while the skin is differentiating, and will increase with stressful events. 10 Therefore, all of the exuvia collected in this study are reflective of the time spent at the supplier or the acclimation period. Although the range of corticosterone concentrations was wide, levels were similar between experimental groups, indicating that physiologic stress was not an important dissimilarity prior to inoculation.
In this study, we demonstrated that subcutaneous injection of O. ophiodiicola into cornsnakes can experimentally replicate skin and internal lesions analogous to those observed in natural infections. We used this model to show that brumation and cooler temperatures have a negative impact on survival of infected snakes, and that body weight does not play an important role in pathogenesis unless the snakes are in very poor nutritional condition. A single noninoculated snake co-housed with inoculated snakes during brumation developed a mild skin lesion consistent with ophidiomycosis, suggestive of horizontal transmission. Together, these findings imply that the cooler temperatures of brumation and close contact of snakes in hibernacula play an important role in disease severity and epidemiology in free-ranging snake populations. This information is key to developing effective mitigation strategies to help conserve snake populations that may be threatened by this disease. Further research is required to determine the relative importance of these factors and others such as host taxonomy, life history, habitat degradation, food availability, environmental conditions (eg, temperature), season, and stress, as well as inoculation route and dose on disease development. 4,17,23,24,26,27,29
Supplemental Material
Combined_supplemental_materials-McKenzie_et_al - Ophidiomycosis in Red Cornsnakes (Pantherophis guttatus): Potential Roles of Brumation and Temperature on Pathogenesis and Transmission
Combined_supplemental_materials-McKenzie_et_al for Ophidiomycosis in Red Cornsnakes (Pantherophis guttatus): Potential Roles of Brumation and Temperature on Pathogenesis and Transmission by Christina M. McKenzie, Paul T. Oesterle, Brian Stevens, Leonard Shirose, Gabriela F. Mastromonaco, Brandon N. Lillie, Christina M. Davy, Claire M. Jardine and Nicole M. Nemeth in Veterinary Pathology
Footnotes
Acknowledgements
We thank the Animal Health Laboratory, especially Hugh Cai and the histotechnology team; Tami Sauder for technological expertise and assistance; Joe Crowley for providing logistical advice; Jeff Hathaway, Alexandra Reid, and Cedric Larouche for snake husbandry advice; the biosecurity facility animal care staff, including Mary Fowler and team; Emily Brouwer and Emily Rätsep for pathology expertise; and Mauricio Seguel and Christopher Cleveland for survival analysis assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Ontario Ministry of Natural Resources and Forestry—Species at Risk Research Fund and the Canadian Foundation for Innovation.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
