Abstract
Bovine babesiosis, caused by Babesia divergens, is in general a rare disease in Europe. Nonetheless, local outbreaks can cause severe economic damage, and postmortem identification represents a diagnostic challenge. During a recent outbreak in May 2018 in northern Germany, 21 animals of a herd of 150 cattle died within 40 days having had clinical signs of fever and hemoglobinuria. Gross examination of 4 of the 21 deceased animals revealed a tick infestation, jaundice, and dark brown staining of urine and kidneys. Histologically, there were iron-positive deposits, hyperplasia of the red pulp of the spleen, and centrilobular necrosis of hepatocytes. In several locations, small basophilic granules suggestive of intraerythrocytic parasites were visible in hematoxylin-eosin- and Giemsa-stained sections. Peripheral blood smears from a living cow from the herd and polymerase chain reaction (PCR) of feeding ticks revealed B. divergens infection. In situ hybridization (ISH) was applied on formalin-fixed, paraffin-embedded (FFPE) tissue of the necropsied cattle to confirm babesiosis in these animals postmortem. Digoxigenin-labeled DNA probes were generated based on a specific nucleotide sequence for B. divergens, obtained by PCR and sequencing of DNA isolates from infected Ixodes ricinus ticks from deceased cattle. ISH using these probes allowed postmortem diagnosis of B. divergens infection in routinely fixed FFPE tissues.
Keywords
Babesiosis is a tick-borne infection caused by apicomplexan protozoal parasites of the genus Babesia. 29 Babesia are distributed worldwide, and cattle can be infected with different species of this parasite. 14,39 Bovine babesiosis in Central Europe is usually caused by infection of naïve, adult cattle with Babesia divergens. 16 B. divergens predominantly cycles between the European castor bean tick (Ixodes ricinus) and cattle (Bos taurus). 9,64 The distribution of B. divergens in Europe reaches from the Mediterranean sea up to Norway, mainly along the southern and western coasts, 52 and in Finland. 38 Due to the fact that I. ricinus exhibits only a narrow host-pathogen spatial mobility, 46 the incidence of ticks infected with Babesia spp. can show high local variability between 0% and 100%. 5 Most investigations in Europe showed that <1% in questing and feeding I. ricinus were infected with B. divergens. 11,26,28,38,48,49,57,59,72 Prevalence of B. divergens infections in pastured cattle is also highly variable 8,21,27,75 ; consequently, knowledge about local endemic pastures and herds is required to suspect babesiosis. Peaks of clinical babesiosis in cattle align with the activity of ticks in spring (May, June) and autumn (September). 16 I. ricinus ticks show low host specifity, 36 and B. divergens infections are reported in several other host species including birds, 19,26 domesticated and wild small ruminants, 47,74 and humans. 10,17,23,31,45 Most fatal cases in humans occur in immunocompromised or splenectomized patients. In Central Europe, about 2% to 4% of the human population is seropositive for B. divergens. 31,63
Pathogens transmitted by tick saliva are known to use specific properties of vector feeding behavior to evade host defense mechanisms. 35 Together with different anticoagulative and vasodilatory molecules, feeding ticks secrete excessive water from their feed back into the wound of their host. 35 Via this route, infected ticks transmit sporozoites of B. divergens from their salivary glands into the host. 25 These infect erythrocytes by attachment on and invagination into their membrane. 41 Within the erythrocyte, merogony (asexual replication) with the formation of multiple pear-shaped merozoites from ring-form trophozoites takes place. 33 The paired form of merozoites represents the classical picture of B. divergens infestation. 39 However, also the tetrad form, called “Maltese cross,” can be observed among other developmental stages within one blood smear. 37 High numbers of merozoites lead to lysis of the host cell and infection of further erythrocytes. 29 After feeding, most parasites are disintegrated, 25 but some merozoites transform into gametocytes within the tick’s mid-gut lumen, form gametes, are fertilized (gamogony; sexual replication), and form a zygote. 33 The zygote infects the mid-gut epithelium of the feeding tick and forms an intracellular parasitic vacuole with excessive production of kinetes. 33 Kinetes are transported via the hemolymph into the ticks ovaries and salivary glands where they can survive until the next feeding. The locally formed sporozoites can infect the offspring transovarially or the next host during feeding. 2 Babesia infection interferes with the tick’s health; thus, ticks developed different defense mechanisms within mid-gut, hemolymph, salivary glands, and ovaries against the pathogens, resulting in a co-evolution of host and pathogen. 25
Calves younger than 9 to 12 months exhibit an equal susceptibility for B. divergens infection compared with adult cattle. However, calves show only mild clinical signs and are able to develop specific immunity after infection. 16,76 Little is known about the underlying mechanism that results in this inverse age resistance. 76 There is stable immunity within the herds in endemic regions. Production of specific antibodies is insufficient for protection; rather, antibody-dependent cell-mediated immunity seems to mount a protective response. 76 Similarly, successful vaccination of cattle against B. divergens is associated with significant mononuclear cell proliferation. 70
About 7 days post infection, adult, naïve cattle are febrile and show reduced general condition, ruminal paresis, diarrhea, anal spasms, anemia, jaundice, and hemoglobinuria. Heart rate is elevated (120 beats/min) with a reduced respiratory rate. Hematology shows low hematocrit, erythropenia with anisocytosis, and lymphocytosis (up to 80% of leukocytes). 16
Macroscopic findings are due to hemolysis and anemia in clinically affected cattle. This includes dark brown staining of kidneys and urine, as well as enlarged liver and spleen. 55 Histologically, hemoglobin accumulation can be identified as eosinophilic granules (iron-positive) within the tubular epithelium and as casts within the tubular lumen, resulting in the macroscopic findings of dark brown discoloration of renal parenchyma and urine. Within the liver, the extent of centrolobular degeneration and necrosis depends on the severity of hypoxic stress (anemia). 55
For routine diagnostics, polymerase chain reaction (PCR) and staining of fresh peripheral blood smears are mainly used. Hematological staining for parasites include Giemsa, May-Grünwald stains, and other Romanowsky-type stains. 44 Further established diagnostic methods include enzyme-linked immunosorbent assay, indirect immunofluorescence antibody test, indirect hemagglutination test, next-generation sequencing, 7,16,34 and reverse line blot hybridization 8 of blood and serum samples or ticks.
Currently, no reliable, specific feasible method for the morphological postmortem diagnosis of babesiosis is available. The aim of the present study was to establish a robust method to detect Babesia species in formalin-fixed, paraffin-embedded (FFPE) tissues and to describe the pathogen distribution and morphology.
Materials and Methods
Preparation of Blood Smears
Capillary and venous blood samples were taken prior to necropsy from one animal with suspected babesiosis. Capillary blood was obtained by puncture of cutaneous vessels in the ear and collection of the first drop on a glass microscope slide. The “push” (wedge) method 1 was used to prepare thin smears of capillary blood. Slides were air-dried and Giemsa stained, according to established protocol. 50
Blood and Urine Analysis
Liver and kidney function tests as well as minerals and electrolytes (Supplemental Table S1) were analyzed photometrically (Abx Pentra 400). Hematological parameters were determined using a hematology analyzer (Celltac α MEK-6450, Nihon Kohden Corporation). Leukocyte differentiation and urine sediment analysis were performed microscopically.
Necropsy, Tissue Preparation, and Light Microscopy
Four animals with suspected babesiosis were necropsied between 4 and 12 hours after death. Samples of brain, spinal cord, liver, spleen, blood clots, intestine, fat, musculature, lymphoid and endocrine organs, urinary and genital tract, mammary gland, respiratory tract, and heart were fixed in 4% buffered formaldehyde solution for 24 hours before embedding in paraffin wax for routine histology, Giemsa staining, and in situ hybridization. For light microscopic examination, 2 to 3 µm tissue sections were routinely stained with Giemsa or hematoxylin and eosin. 50
Polymerase Chain Reaction
PCR and subsequent sequencing of the amplicon was performed on a pool of ticks from 2 affected animals. Ticks were minced with a sterile razor blade, and DNA was isolated using the DNeasy blood and tissue kit (Qiagen) according to the manufacturer’s protocol for tissues. A partial 18S ribosomal DNA region was amplified by PCR using universal primers PIRO-A (5′-AATACCCAATCCTGACACAGGG-3′) and PIRO-B (5′-TTAAATACGAATGCCCCCAAC-3′) known to amplify fragments from several cattle and deer piroplasms including B. divergens and B. microti. 3 PCR was performed with HOT FIREPol Blend Master Mix (Solis BioDyne), 200 nM final concentration of forward and reverse primers each, and 100 ng of DNA in a 50 µl reaction volume. PCR cycling conditions were as follows: 15 minutes activation/initial denaturation at 95 °C, 35 cycles of 20 seconds denaturation at 95 °C, 30 seconds annealing at 55 °C, and 1 minutes 72 °C extension, followed by a 5 minutes 72 °C final elongation. The obtained amplicons were purified and sequenced using the primers for amplification (LGC Genomics). Sequences were checked manually, assembled, and analyzed by similarity search of the GenBank database using BLAST (http://www.ncbi.nlm.nih.gov/BLAST/).
In Situ Hybridization (ISH)
The DNA probe for ISH was designed in accordance with the sequenced PCR product (407 base pairs [bp] and 364 bp without primer), obtained by usage of the PIRO-A/PIRO-B primers and checked for specificity against the GenBank database using BLAST (http://www.ncbi.nlm.nih.gov/BLAST/). Sense (Bab-fwd) and anti-sense (Bab-rev) digoxigenin (DIG)-labeled DNA probes of 42 nucleotides (sense: Dig-5′ GTCTTGAATACTTCAGCATGGAATAATAGAGTAGGACTTTGG 3′; anti-sense: Dig-5′ CCAAAGTCCTACTCTATTATTCCATGCTGAAGTATTCAAGAC 3′) were used to detect B. divergens in FFPE tissue sections as previously described. 6,20,24,54,77 Briefly, Roti-Histol (Carl Roth) and decreasing graded ethanol were used to deparaffinize and hydrate tissue sections, followed by washing steps in diethylpyrocarbonate-treated water. Afterwards, proteolytic digestion using 1 μg/ml proteinase K (Roche Diagnostics;), postfixation, acetylation, prehybridization, DNA denaturation at 99 °C for 10 minutes, and hybridization overnight at 52 °C in a moist chamber with a probe concentration of 1000 ng/ml were performed. An alkaline phosphatase (AP)-labelled anti-DIG-antibody (1:200; Roche Diagnostics) was used for the detection of probes. Nitroblue tetrazoliumchloride (NBT; Sigma-Aldrich Chemie) and 5-bromo-4-chloro-3-indolyl phosphate (BCIP, X-Phosphate) served as substrates. ISH results were verified by real-time PCR and conventional nested PCR/sequencing (see Supplemental Materials).
Results
Case History
A herd of German Red Holstein and Maine Anjou beef cattle (herd 1), composed of cows (>3 years), their calves, and a bull, had been moved onto a new pasture (pasture 1). Ten days later, several animals started to show reduced general condition and hemoglobinuria. Within the next 3 days, 7 animals died. The herd was moved to another pasture (pasture 2) and brought together with a second herd (herd 2). In the following weeks, a further 6 cows of herd 1 and 8 cows of the formerly unaffected herd 2 died. In both herds, calves were not affected. Altogether, 21 of 150 adult cattle died within 40 days (see Supplemental Materials).
Marginal grazing areas of the pastures were surrounded by shrubs and trees. In pasture 1, a group of trees represented a favorite resting place for the cattle (personal communication with the owner). May 2018 was very dry in Lower Saxony (Hannover, May: Rainfall total: 10 l/m2 [18% of expected average]; sunshine: 300.4 h [138% of expected average]; https://www.wetterkontor.de/de/wetter/deutschland/monatswerte-station.asp) and warm with daily peaks of up to 33 °C (Hannover, May: mean temperature: 16.9 °C [+3.5 °C of expected average]).
Pathology
Four out of 21 dead animals, including the cow hospitalized in the clinic (see below), were submitted for necropsy. The macroscopic findings were similar in all cattle. Animals were in a good body condition, but all animals were moderately infested with ticks (Ixodes ricinus) of different developmental stages, especially in the axillary and inguinal regions as well as along the ventral abdomen (Fig. 1).
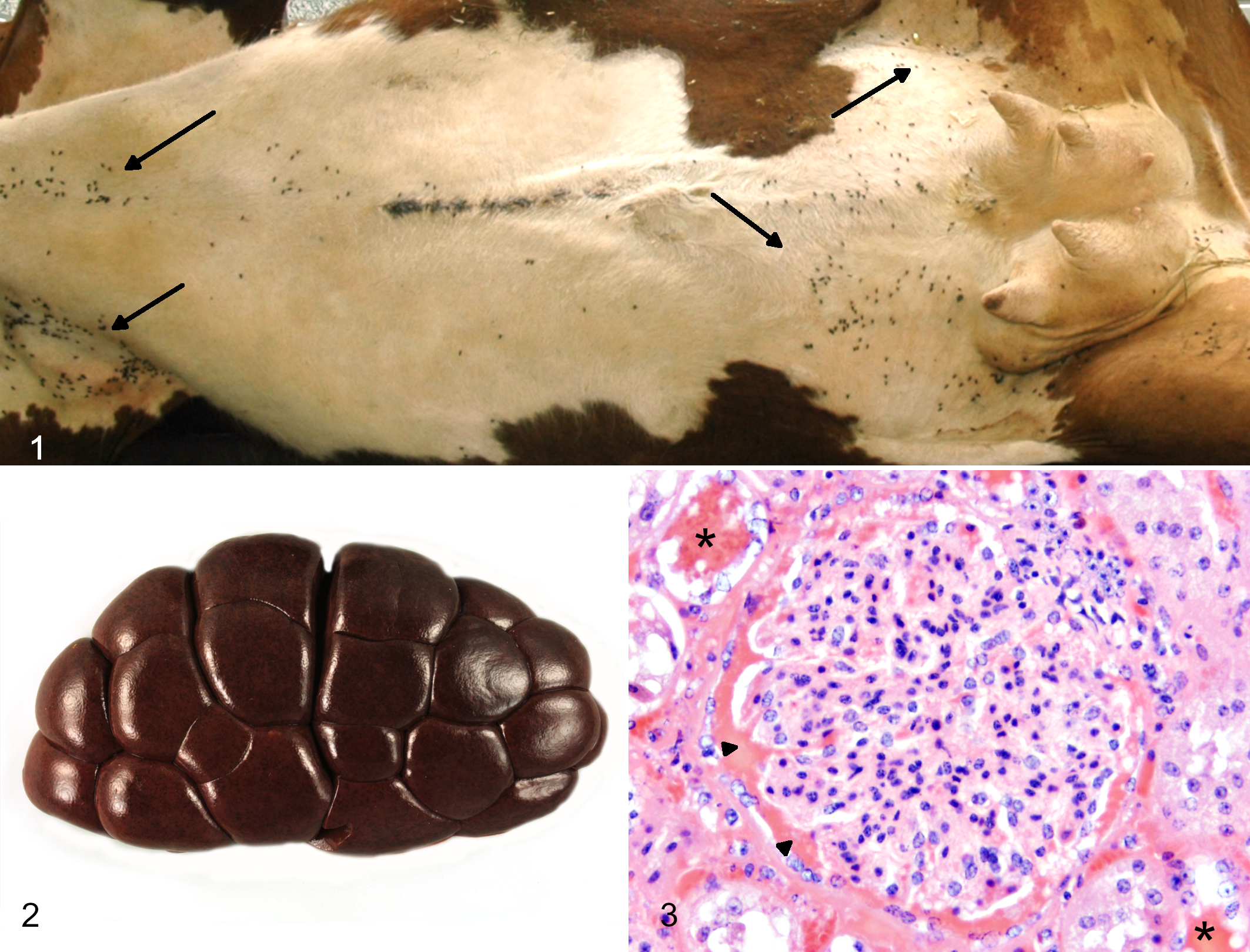
Tick infestation (Ixodes ricinus), cow. There are numerous feeding ticks in axillary, sternal, abdominal, and inguinal regions (arrows).
Grossly, the animals exhibited mild, multifocal, subcutaneous edema and mild jaundice. The blood was dark-red to brown. The urine was dark brown and contained finely granular, black material. Both kidneys were diffusely dark brown with finely stippled, black discoloration in the cortex (hemoglobinuric nephrosis; Fig. 2), which was identified histologically as hyaline hemoglobin casts (Fig. 3). The spleens were increased in size and histologically showed hyperplasia of the red pulp. Livers were slightly increased in size, with rounded edges and mild diffuse light-brown discoloration.
Hematological Findings and Blood Smears
An 11-year-old cow (from herd 1) of the breed German Red Holstein was admitted to the Clinic for Cattle of the University of Veterinary Medicine Hannover, with a history of 1-day illness and calving 4 weeks ago. Fifteen cows within the same herd had died during the past 2 weeks. Detailed epidemiological and serological data of the herds as well as prevalence of Babesia in collected ticks from the pastures were obtained until March 2020 and will be published elsewhere (Springer A, Höltershinken M, Lienhart F, et al, unpublished data).
The cow showed tick infestation (>30 ticks), spontaneous passing of dark brown urine (hemoglobinuria), reduced feed uptake, depression, and reluctance to move. On clinical examination, there was elevated body temperature (39.4-39.8 °C), increased heart rate (140 per minute with a pounding beat), and normal respiratory rate (36 breaths per minute). The rumen was hypo-mobile and mucous membranes were pale and yellow (anemia and jaundice). Blood analysis of this cow revealed hemolysis, anemia, leukocytopenia, increased liver enzymes, and elevated urea and total protein.
Giemsa-stained blood smears of blood samples taken by the local practitioner were unremarkable. From the cow hospitalized in the Clinic for Cattle of the University of Veterinary Medicine Hannover, Giemsa-stained blood smears of capillary blood revealed intraerythrocytic parasites (37-39.3% parasitemia; Fig. 4). They were morphologically identified as B. divergens. Intraerythrocytic parasites mainly showed the marginal ring stage (trophozoites) and the paired figure (merozoites). Extraerythrocytic merozoites showed a rounded or pear-shaped morphology.
Babesia divergens, peripheral blood smear. Erythrocytes with ring stage trophozoites (arrows) and paired merozoites (asterisks). Giemsa.
Histopathology
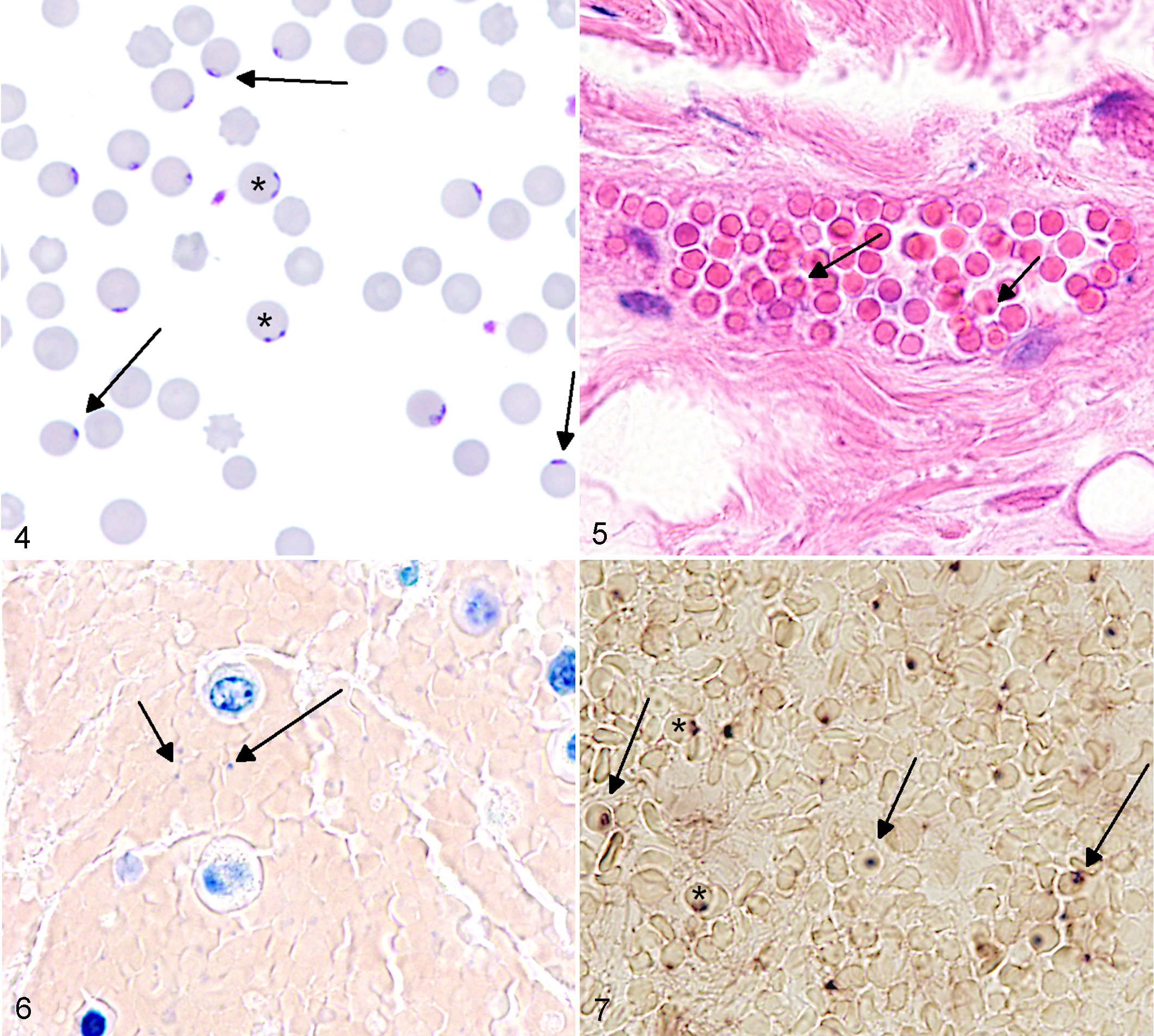
Histopathological changes were predominantly found in the kidneys and were characterized by marked intratubular hemoglobin casts, vacuolization and degeneration of tubular epithelial cells, and iron-positive pigment deposition within tubular lumina and epithelial cells. Livers showed mild, centrilobular necrosis of hepatocytes, and the other areas had hepatocellular lipidosis and canalicular cholestasis. Mild, multifocal hemorrhage was observed in dermal and subcutaneous adipose tissue, as well as subendocardial tissue, and in a single animal within the cerebral cortex. Hemosiderosis was present in spleen, lymph nodes, and pulmonary alveolar histiocytes. Occasionally, merozoite-like structures were visible within the systemic circulation and specifically within the erythrocytes of the choroid plexus, rete mirabile of the pituitary gland, renal glomeruli, and vessels of the tongue, adipose tissue, and musculature. The presumed parasitic structures were weakly basophilic, approximately 1 µm diameter, and round, and they were either not associated with cells or were intracellular at the margin of erythrocytes (Fig. 5). In animals with severe anemia or autolytic changes including clostridial overgrowth, blood vessels of the tongue were the most frequent sites for detection. Basophilic structures matching the size and morphology of Babesia spp. were present within the spleen but were questionable due to non-specific staining.
Giemsa staining of FFPE histologic sections revealed few small positive granules within blood vessels and erythrocytes in the kidney and small foci of hemorrhage within adipose subcutaneous tissue (Fig. 6). These round granules had a diameter of approximately 1 µm and were suggestive of merozoites. Similar positively stained structures were observed in the spleen. However, the morphology based on hematoxylin-eosin and Giemsa staining remained inconclusive. Similarly, staining of FFPE tissue of ticks collected from affected cattle revealed no definite morphological proof of B. divergens.
Polymerase Chain Reaction
A partial (407 bp) Babesia 18S ribosomal rDNA sequence was amplified from DNA of pooled ticks collected from 2 affected cows. Direct sequencing of the amplicon revealed 100% identity to B. divergens sequences (eg, KY296360) in GenBank.
In Situ Hybridization
ISH using sense and anti-sense DNA probes with FFPE tissues revealed a positive signal in numerous erythrocytes within blood clots (Fig. 7) as well as in blood vessels of various organs and especially in spleen, kidney, and liver. Positive signals were seen on the inner margins of erythrocytes or free within the blood vessels and matched the size of the structures suspected to be Babesia in hematoxylin-eosin- and Giemsa-stained blood smears and tissue sections. Morphologically, they predominantly matched the marginal ring form of the parasite. The results of ISH were confirmed by real-time and seminested conventional PCR and sequencing (see Supplemental Materials).
Discussion
Babesia divergens is dependent on the distribution and feeding success of host ticks. 29 Climate change may influence the distribution of ticks and incidence of tick-borne diseases in Europe. 22 Increase of temperatures in Northern Europe and decreased rainfall in Southern Europe may have shifted the ticks’ habitat from south to north. 13,56 In the described case, pastures were located in a nature reserve in Northern Germany (Lower Saxony) with a large variety of herbs and grasses, providing an advantageous environment for ticks and their wildlife hosts. 60 Surveillance of ticks and tick-borne diseases in Germany is mainly concentrated to the southern and middle parts of the country, 19,31,53,59,69 but since babesiosis is also endemic in parts of Norway, 51 it can be assumed that there are endemic regions in Northern Germany, too. Questing behavior of I. ricinus, which involves climbing up low vegetation and waiting for possible host contact, increases with temperature, until low humidity forces the tick back down into the ground microhabitat. 67 On hot days, trees and shrubs on the pastures create a microclimate with lower solar radiation, increased humidity, and slower air movement, 71 favorable for ticks and their vertebrate hosts. 60 Control strategies include reduction of vectors selection of native and also nonnative plants with low water consumption to maintain a dry habitat (xeriscaping), mowing, introduction of tick pathogens, rodent control, and restricted access for other wildlife hosts; 65 however, these are not practicable in nature reserves because they modify the landscape and fauna (https://www.nlwkn.niedersachsen.de/naturschutz/schutzgebiete/einzelnen_naturschutzgebiete/die-naturschutzgebiete-niedersachsens-45243.html). Application of antiparasitic drugs and substances on animals and pastures on European farms is generally restricted by (EC) No 528/2012 66 and further defined by the guidelines of different food associations (eg, Bioland 40 ). Vaccination against babesiosis is possible, but is quite costly and not approved in all countries. 18 Imidocarb is currently the only substance approved by the authorities of the European Union, 76 but it is not always available in all member countries (https://www.vetpharm.uzh.ch/reloader.htm?tak/06000000/00062138.01?inhalt_c.htm). The current rarity of clinical babesiosis in Central and Northern Europe corresponds to decreased awareness of the disease among farmers and veterinarians, potentially resulting in delayed diagnosis and treatment with subsequent increased disease-related costs and mortality rates. 75 Postmortem diagnosis of suspected cases can help identify endemic pastures and gives farmers the opportunity to adapt pasture management for protection of the remaining herd.
The present report describes a local outbreak including clinical findings, diagnostic tools, and morphological findings of babesiosis in cattle and the usefulness of molecular methods to detect the causative pathogen on FFPE tissues.
Cytology of peripheral blood remains the quickest way of diagnosing of Babesia in vivo. 16 To obtain a reliable blood smear, it is important to use peripheral, capillary blood (preferably the first drop) of febrile animals. 16 Preparation of blood smears should take place immediately after sampling on site. Compared with capillary blood, the concentration of Babesia is much lower in venous blood samples. 12 Cooling of the sample at 4 °C for transport or storage significantly reduces parasitic load within 24 hours and, more troublesome, changes the morphology of B. divergens. The latter forms appear as shrunken fragments within or detached from the erythrocytes and are difficult to identify as specific pathogens. 12 Improper sampling (eg, sampling of nonfebrile or dying animals, thick drops or smears on the slides, collection of venous blood), handling (cooling, long storage of blood samples), and transportation (ie, submission of whole blood instead of air-dried blood smears) can lead to misdiagnosis even after correct handling and staining in the laboratory. 12,16,30
Morphologic diagnosis of Babesia in FFPE tissue is extremely challenging and in most instances not possible. As shown in the present study, nonspecific staining methods like HE and Giemsa, commonly used in routine diagnostics, might support a suspected diagnosis, but do not allow a conclusive diagnosis.
Postmortem changes of temperature, autolysis, and putrefaction are partially responsible for destroying pathogen integrity. 12 Similarly, a shift from the “large” to the “small” phenotype is described as a postmortem change of B. canis in Romanowsky-stained cytological preparations from dogs. 30 With increasing autolysis, merozoites tend to separate from erythrocytes and exhibit a rounded morphology. 30 This was also seen in the FFPE tissue of the present case (see Figs. 5–7). The altered postmortem morphology and detachment of Babesia from erythrocytes represent major obstacles for a final diagnosis and is especially pronounced in animals with severe anemia or progressive autolytic changes. Another set of diagnostic challenges are other pathological findings with similar morphological presentation in affected erythrocytes. Howell-Jolly bodies are micronuclei or nuclear DNA remnants in erythrocytes, frequently seen in hemolytic anemia, and appear as basophilic small (∼ 1 μm) dots on erythrocytes. 4 Heinz bodies are another frequent change in erythrocyte morphology in hemolytic anemia 32 and are eosinophilic granules of precipitated or denatured hemoglobin in damaged erythrocytes that are normally phagocytosed within the spleen. 32 Presumably due to their delicate appearance, the morphology of Howell-Jolly and Heinz bodies in FFPE sections have yet to be described. Nonetheless, it cannot be ruled out that some of the basophilic granules within the erythrocytes, seen in hematoxylin-eosin- and Giemsa-stained sections, are Howell-Jolly bodies. Therefore, molecular tools to identify the pathogen are needed for a reliable diagnosis.
The ISH method developed in this study allows for specific determination of the identity of the observed intra- and extraerythrocytic structures. Routine PCR is mainly used for antemortem detection of Babesia spp. using blood, 58 and can be used for postmortem diagnosis by using fresh or frozen tissues from infected mammals or ticks. However, FFPE tissue is the most commonly available material in postmortem diagnostics and remains the best way to inexpensively archive tissues. Nonetheless, PCR on FFPE tissue requires special protocols since the DNA is highly fragmented, forming crosslinks and breaks that interfere with polymerization during the process. 15 As such, success of PCR from FFPE material is highly variable. 43 For B. odocoilei, which infects cervids, it has been shown that PCR can be performed on FFPE tissue with variable success depending on the amplicon length. 42 In contrast, techniques like immunohistochemistry, ISH, and fluorescence in situ hybridization (FISH) preserve organ and pathogen morphology and allow localization of the pathogen within the host.
Immunohistochemical detection in FFPE material has been successfully employed for B. microti and the B. duncani strain using both human and animal tissues. 68 Staining included detection of either the whole parasite, the parasitic wall, or a granular staining in erythrocytes 68 with a roundish phenotype, 30 similar to our findings. In Beagle dogs experimentally infected with B. gibsoni, it was possible to detect the parasites with a probe matching a 516 bp DNA fragment of the heat-shock protein 70 (BgHsp70) gene via ISH. 73 FISH is available for the detection of other Babesia spp. (B. duncani, B. microti) in fresh human 61 and hamster blood smears. 62
The ISH probe developed in this study is the first established for postmortem diagnosis of Babesia in FFPE tissues from cattle. Intraerythrocytic parasites were detectable in various organs that are commonly collected during routine postmortem diagnostic investigations. A method to detect Babesia in FFPE tissue sections not only allows postmortem verification of bovine babesiosis but also facilitates retrospective studies on archived tissue samples.
In conclusion, although clinical babesiosis is generally rare in Central and Northern Europe, a local outbreak can present a diagnostic challenge and requires a reliable diagnostic toolbox. Suboptimal sampling and storage of blood samples can lead to false-negative diagnoses, whereas optimal cytological preparations allow timely and reliable antemortem diagnostics. Although routine postmortem findings remain nonspecific and may only suggest babesiosis, a final, definitive diagnosis requires state-of-the-art molecular tools. ISH represents a new tool for specific postmortem diagnosis of Babesia in cattle on histologic sections of FFPE tissues.
Supplemental Material
Combined_supplemental_materials-Hulskotter_et_al - Bovine Babesiosis Diagnosed in Formalin-Fixed, Paraffin-Embedded Tissues by Using In Situ Hybridization
Combined_supplemental_materials-Hulskotter_et_al for Bovine Babesiosis Diagnosed in Formalin-Fixed, Paraffin-Embedded Tissues by Using In Situ Hybridization by Kirsten Hülskötter, Vanessa M. Pfankuche, Lydia van Dyck, Martin Höltershinken, Andrea Springer, Fabienne Lienhart, Sandra Ermel, Jürgen Rehage, Martina Hoedemarker, Christina Strube, Jörg Hirzmann, Christian Bauer, Wolfgang Baumgärtner, Annika Lehmbecker and Peter Wohlsein in Veterinary Pathology
Footnotes
Acknowledgements
The authors would like to thank Danuta Waschke, Caroline Schütz, Julia Baskas, Siegfried Jelitto, Dunja Hoffmann, Angela Karl, Iris Grewe, Antje Lecht, and Nicole Kusserow for their excellent technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
