Abstract
Epizootic epitheliotropic disease virus (salmonid herpesvirus-3; EEDV) is responsible for the death of millions of hatchery-raised lake trout (Salvelinus namaycush) in the Laurentian Great Lakes Basin. However, little is known about its biology, pathology, tropism, and host interactions. In this study, the presence and disease progression of EEDV were evaluated following exposure of naïve juvenile lake trout to EEDV via bath immersion under controlled laboratory conditions (n = 84 infected; n = 44 control). Individual tissues (n = 10 per fish), collected over 6 weeks, were analyzed for viral load by quantitative polymerase chain reaction, gross and histopathologic changes, and virus cellular targets using in situ hybridization. Skin, fin, and ocular tissues were the earliest viral targets and yielded the highest viral loads throughout the course of infection. Early gross lesions included exophthalmia, ocular hemorrhage, fin congestion, and hyperemia of visceral blood vessels. Advanced disease was characterized by multifocal to coalescing erosions and ulcerations of the skin, and congestion of visceral organs. Microscopically, there was cellular degeneration and necrosis in the epidermis and spleen, and lymphohistiocytic perivasculitis of the dermis, omentum, and the epicardium. EEDV DNA was first detected by in situ hybridization in epithelial cells of the epidermis, with subsequent labeling in the epithelial lining of primary and secondary gill lamellae. During advanced disease, EEDV was detected in endothelial and dendritic cells as well as blood monocytes. This study characterized EEDV tissue tropism and associated pathologic features, to guide research aimed at understanding EEDV disease ecology and improving strategies for disease control.
Keywords
Epizootic epitheliotropic disease virus (EEDV; salmonid herpesvirus 3) has been implicated in multiple mortality events throughout the Midwestern United States over the past 3 decades 2,3,13 and continues to be a concern in Great Lakes fisheries management today. 24 EEDV is particularly pathogenic to hatchery-raised juvenile lake trout (Salvelinus namaycush) during natural mortality events, 2 and experimental challenges have also resulted in high mortality. 2,17 The clinical manifestations of EEDV in affected fish often includes ocular hemorrhage, corneal opacity, gill pallor, skin lesions ranging from pallor to erosions and ulcerations, water mold overgrowth, erosion and congestion of the fins, and erythema of the oral cavity, isthmus, and ventrum. 24 Detection of EEDV has also been reported from reproductive fluids and kidneys of mature spawning lake trout. 11,13
Unfortunately, specific information regarding pathology and host interactions remains largely unknown, in part due to the lack of an EEDV-susceptible cell line and inability for in vitro growth. This knowledge gap hampers the design and implementation of effective disease control strategies in the Laurentian Great Lakes Basin. A recently developed in vivo model of immersion challenge and virus replication has provided a crucial step in studying the pathogenesis of EEDV, 24 allowing for challenge dose control and examination of EEDV disease progression under controlled laboratory conditions. Elucidating the sequential microscopic alterations and disease progression following EEDV infection is of vital importance as this virus continues to threaten lake trout rehabilitation efforts throughout the Midwestern United States.
Previous histopathologic examination of EEDV-infected tissues has been limited to severely diseased fish collected during an epizootic episode. 2,24 In such severe cases, the primary skin lesions have often been obscured by the invasion of opportunistic and pathogenic bacteria and water mold into damaged skin, leading to difficulties in accurate assessment of underlying disease pathology. In order to alleviate the inherent challenges of attributing lesions of multifactorial compounded disease to EEDV during natural infections, this study was designed to characterize the progression of gross and histopathologic lesions and identify cellular targets after experimental exposure of naïve host fish to a moderately lethal dose of EEDV. Our objective is to provide researchers, diagnosticians, and fisheries managers with information regarding the early gross and microscopic tissue alterations associated with an EEDV infection.
Materials and Methods
Fish and Maintenance
EEDV experimental challenges were performed using juvenile (6 months post-hatch) Lake Superior strain lake trout supplied by the Marquette State Fish Hatchery (Marquette, MI). Prior to leaving the hatchery, all fish had been vaccinated against Renibacterium salmoninarum (oral; MSU-AAHL, East Lansing, MI) and Aeromonas salmonicida (bath immersion; AquaTactics Fish Health, Kirkland, WA). All experimental fish were randomly collected from a lot certified to be free of any reportable pathogens as per the American Fisheries Society–Fish Health Section (AFS-FHS) Blue Book. 1 Certification was achieved following normal clinical examination of 60 randomly collected fish. Additionally, the absence of EEDV in the lot was confirmed using quantitative polymerase chain reaction (qPCR) as detailed below. Experimental challenges were performed at the Michigan State University—Research Containment Facility (MSU-URCF, East Lansing, Michigan) in accordance with the MSU Institutional Animal Care and Use Committee guidelines and approval. On receipt, all fish were housed in a 680-L fiberglass aquarium and allowed to acclimate to standard laboratory conditions for a minimum of 1 month prior to experimental challenges. At all times, fish received continuous, oxygenated well water and were fed 1.0 mm sinking feed ad lib (BioOregon, Westbrook, ME). During the experimental studies, fish were housed in 42-L fiberglass aquaria receiving continuous, flow-through, oxygenated well water at a temperature of 9.0 ± 0.5 °C.
EEDV In Vivo Serial Passages and Stock Production
To prepare EEDV inoculum for later use in experimental challenges, a stock of infectious EEDV was produced from the skin of naturally infected lake trout collected during a natural outbreak. Skin was homogenized in a sterile phosphate-buffered saline solution (pH 7.5 ± 0.5; Sigma-Aldrich, St. Louis, MO) at a 1:3 (w/v) ratio, and clarified via low-speed centrifugation (1400 × g) for 20 minutes at 4 °C. The initial EEDV stock was then passed through multiple groups of naïve juvenile lake trout via either an intraperitoneal (IP) injection or an immersion bath challenge. Initial passages were performed via IP injection in order to best ensure equal virus exposure while later passages were performed via bath challenge to more closely mimic a natural route of infection. For the intraperitoneal challenges, fish were anesthetized using tricaine methanesulfonate (MS-222; Western Chemical, Ferndale, WA; 0.1 mg/mL) then injected with 300 to 400 µL of virus stock solution and allowed to recover from sedation prior to return to flow-through aquaria for the duration of the study. For the immersion bath challenges, fish were transferred to glass aquaria containing combined volumes of virus stock and water. Fish were held in these static aquaria at a constant water temperature (9 ± 0.5 °C) with continuous aeration for 1 hour, after which time they were transferred back to their flow-through aquaria for the duration of the study. Following virus exposure, fish were monitored daily for development of morbidity or mortality. On death or development of severe clinical disease (e.g., loss of equilibrium, difficulties respiring), the fish were euthanized with an overdose of MS-222 (0.25 mg/mL), and their skin was collected and processed as described above with the substitution of Earle’s salt-based minimal essential medium (MEM; Invitrogen, Thermo Fisher Scientific, Waltham, MA), supplemented with 12 mM Tris buffer (Sigma-Aldrich, St Louis, MO), penicillin (100 IU/mL; Invitrogen), streptomycin (100 μg/mL; Invitrogen), and amphotericin B (250 μg/mL; Invitrogen) rather than phosphate-buffered saline. Skin homogenates created from multiple individual fish within each passage were pooled together to create new batches of EEDV stock from which a new group of naïve lake trout were in turn infected. This process continued until an adequate volume of seventh passage stock was produced for use in all future studies. Viral exposure route and dose are presented in Table 1.
Mortality Data Following In Vivo Passage of Epizootic Epitheliotropic Disease Virus (EEDV) Stocks Through Juvenile Lake Trout (Salvelinus namaycush)a.
Abbreviations: IP, intraperitoneal injection; Imm, immersion bath challenge.
a Viral passage number represents stock batch used to infect specified group of fish. Dose received is presented in viral copies per fish (intraperitoneal injection; IP) or viral copies per mL of water (immersion bath; Imm). First and last mortalities are presented as the number of days post virus exposure. Viral titers calculated per mg of skin tissue collected from infected fish following mortality.
Experimental Challenges: Dose Range Determination
Following the challenges outlined in Table 1, our first experiment extrapolated on preliminary results in order to determine the dose range or viral load required to lead to infection and development of clinical EED. This study included a total of 60 fish and 6 treatment groups (5 viral doses and 1 negative control group). Viral doses and the negative control dose were prepared as described above using seventh-passage EEDV stock. Exact viral doses were determined using qPCR to test water samples and calculate viral copies per mL of water for each dose (Supplemental Table S1). The highest infection group was exposed to 1.0 × 106 viral copies/mL of water with subsequent doses being 1:10 dilutions made using sterile sample diluent (MEM) down to 2.1 × 101 viral copies/mL of water.
Immersion exposure was achieved as described above, after which fish were monitored daily for a period of 30 days for mortalities or development of clinical morbidity. Any moribund fish with severe clinical signs such as altered behavior, inability to maintain balance, gasping for air, or significantly pale gills was euthanized. Skin and gill tissues were collected from each fish immediately following death. These tissues were tested for the presence of EEDV using qPCR, and the viral loads in bath immersion water samples were used to calculate projected viral dose ranges and mortality percentage pairings based on the calculations of Reed and Muench (1938; Supplemental Table S2).
Experimental Challenges: Lesion Progression
Fish were assigned (haphazard selection from holding tank) into an EEDV group (n = 84) and a negative control group (n = 44). All fish were challenged via the immersion method described above. The EEDV group was exposed to the median lethal dose of EEDV that was calculated based on the previous challenges, while the negative control group was immersed in a sham suspension of MEM diluent. A single tank was used for each treatment group (EEDV or negative control). This challenge dose (LD50) was chosen in order to allow for monitoring of diseased fish over an extended period of time. After completion of the exposure period (1 hour), fish were transferred back to their experimental flow-through aquaria (one tank for infected fish and one for control fish) for the duration of the study where they were monitored daily for development of clinical disease signs.
Sampling occurred in parallel from the experimental and control groups on days 0, 1, 3, 6, 9, 12, 15, 18, 21, 28, 35, and 42 post infection (PI), at which time 7 EEDV-infected fish and 4 (days 0-28) or 2 (days 35, 42) negative control fish were haphazardly collected and euthanized in a manner that minimized the stress for both sampled and remaining fish. If present, moribund fish or mortalities were removed first (and counted among the 7 or 4 fish). On each sampling day, full external and internal examinations were performed on 6 of the 7 infected and 3 of the 4 negative control fish (both negative control fish on days 35 and 42) and portions of skin, fin, gill, eye, brain, spleen, heart, liver, intestine, anterior kidney, and posterior kidney were collected from these fish. Each tissue was divided for (1) detection of EEDV by PCR (frozen at −20 °C) and (2) microscopic examination (fixed in 10% neutral buffered formalin). One whole eye was collected for each assay, and the “intestine” sample consisted of a segment approximately 1 cm oral to the vent.
Virus Detection and Quantification
Without the use of a susceptible cell line, identification and quantification of EEDV in challenge fish, passage stocks, and experimental tissues were achieved using qPCR as described by Glenney et al. 11 Briefly, DNA extractions were performed using the Mag Bind Blood and Tissue DNA Kit (OMEGA Bio-tek, Inc, Norcross, GA), following the manufacturer’s instructions and with the addition of a filtering step using the E-Z 96 Lysate Clearance Plate (OMEGA Bio-tek, Inc). Approximately 10 mg of tissue was used for each extraction and eluted DNA was quantified using a Quant-iT DS DNA Assay Kit and a Qubit fluorometer (Life Technologies, Grand Island, NY). All PCR reactions were carried out in a Mastercycler ep realplex 2 S real-time PCR machine (Eppendorf, Hauppauge, NY) with a total reaction volume of 20 μL. Each reaction contained 10 µL SYBR Select Master Mix (2×; Life Technologies), 1.0 μM of forward and reverse primers, and 50 nmol total DNA template. Positive control standards for quantification were produced using known positive skin samples following the method outlined by Glenney et al. 11 Positive extraction controls consisted of known positive skin samples; MEM was used for the negative extraction control and nuclease free water was used for a PCR negative control. Viral loads (genome-equivalent copies per mg) were then calculated using resulting reaction copy number following qPCR and original tissue weights (mg) prior to digestion (Supplemental Table S3).
Statistical Analysis
Statistical analyses were performed in order to evaluate the relationships between the number of positive samples or the viral DNA load with respect to organ, days post viral exposure, and external versus internal tissue groups. These comparison analyses were generated using a generalized linear mixed model in SAS software, Version 9.4, of the SAS System (Copyright 2017 SAS Institute Inc). For viral loads, analyses were performed on log-transformed copies per mg tissue in order to increase normality of distribution. Statistical significance was determined based on a probability level of 1% or 5% as indicated below.
Histopathology and Lesion Scoring
After fixation, tissues were processed for routine paraffin embedding. Paraffin blocks were sectioned at 5 μm, and slides were routinely stained with hematoxylin and eosin (H&E) for histopathologic examination. 20 All slides were examined by an American College of Veterinary Pathologists board-certified pathologist (SF) who was blinded to the presence of gross lesions or results of EEDV detection in individual tissues (with the exception of the negative control slides from fish 2 and 3 on each sampling day—examined at a later date). All lesions were scored on a scale of 0 to 3: 0 = no lesion (normal), 1 = a mild lesion, 2 = a moderate lesion, and 3 = a severe lesion (Supplemental Table S4).
In Situ Hybridization
An EEDV-specific oligonucleotide probe was designed following a previously described algorithm, 7 using the computer program Oligo 6 and based on the glycoprotein gene sequence published in GenBank (JX886027.1). This oligonucleotide probe (5′-GCT CAA TTT ATC GTG CTC AAA TGG TTC ACT GGC CAG CTC CAT GTC CAT CG-3′) was labeled with digoxigenin at the 5′ end (IDT). This specific probe was developed to differentiate EEDV from the other 4 salmonid herpesviruses, and use of the Basic Local Alignment Search Tool (www.ncbi.nlm.nih.gov/blast.cgi) demonstrated no cross-reactivity with salmonid herpesvirus 1, 2, 4, or 5. The probe was purified by high-performance liquid chromatography (IDT).
In order to maximize the sensitivity and specificity of this in situ hybridization (ISH) assay, preliminary tests were performed in order to identify the optimal protocol and reagent concentrations as previously described. 18 Briefly, 5-μm-thick sections were cut from paraffin-embedded tissues previously collected and placed onto positively charged slides, which were then deparaffinized and fixed using the Discovery XT automated slide-processing system (Ventana Medical Systems, Inc, Tucson, AZ) as programed in the protocol for the RiboMap in situ hybridization reagent system (Ventana Medical Systems). Protease 3 (0.02 units mL−1 alkaline protease; Ventana Medical Systems) was used for 12 minutes at 37 °C for a proteolytic treatment followed by a mild cell conditioning step using the citrate buffer-based RiboCC reagent (Ventana Medical Systems) for 4 minutes at 95 °C. The slides were then denatured for 4 minutes at 37 °C, followed by hybridization for 1 hour at 37 °C with the antisense oligonucleotide probe for EEDV suspended in hybridization buffer (RiboHybe; Ventana Medical Systems). The concentration used for the EEDV probe was 1.59 ng mL−1 (1:10,000 dilution). Four stringency washing steps were performed at 42 °C using 0.1× RiboWash (equivalent to 0.1× saline sodium citrate; Ventana Medical Systems) for 4 minutes for the first 3 and for 8 minutes for the fourth washing step. After the stringency washes, the slides were incubated with a rabbit monoclonal antidigoxigenin antibody (Invitrogen Corporation, Frederick, MD) at a dilution of 1:10,000 for 32 minutes at 37 °C. Slides were then incubated in streptavidin-alkaline phosphatase conjugate (UMap anti-Rb AP; Ventana Medical Systems) for 16 minutes at 37 °C and the signal was detected automatically using the BlueMap nitroblue tetrazolium-BCIP (5-bromo-4-chloro-3-indolyl phosphate) substrate kit (Ventana Medical Systems) for 2 hours at 37 °C. The final step involved counterstaining the slides with nuclear fast red-equivalent reagent Red Counterstain II (Ventana Medical Systems) for 4 minutes before adding a coverslip. Skin and gill tissues collected from naïve lake trout raised in a biosecure containment facility were used as negative tissue controls while experimentally infected lake trout with qPCR confirmed EEDV-positive tissues were used as positive controls. EEDV positive tissues labeled with an oligoprobe against canine herpesvirus 15 as a negative reagent control had no ISH signal.
Results
In Vivo Serial Passages of EEDV
Consecutive rounds of intraperitoneal and immersion challenges of naïve juvenile lake trout with clarified tissue homogenate from previously EEDV-infected fish consistently produced morbidity and mortality, from which EEDV nucleic acid was recovered (Table 1). Additionally, clinical signs and lesions included multifocal skin erosions (Fig. 1), iridal reddening (possible congestion, hemorrhage or hyperemia; Fig. 2), fin erosion and congestion (Fig. 3), pale gills and visceral organs, renal congestion, and hyperemia of visceral blood vessels. Initiation of mortality ranged from 6 to 36 days following viral exposure, with the first mortalities occurring 2 to 3 weeks after viral exposure in the majority of trials. The cessation of mortalities occurred over a similar range, with 0 to 2 fish surviving in most trials approximately 1 month after initial infection (Table 1). Viral titers in EEDV stocks increased approximately 1000-fold from the initial stock to the sixth passage (Table 1).
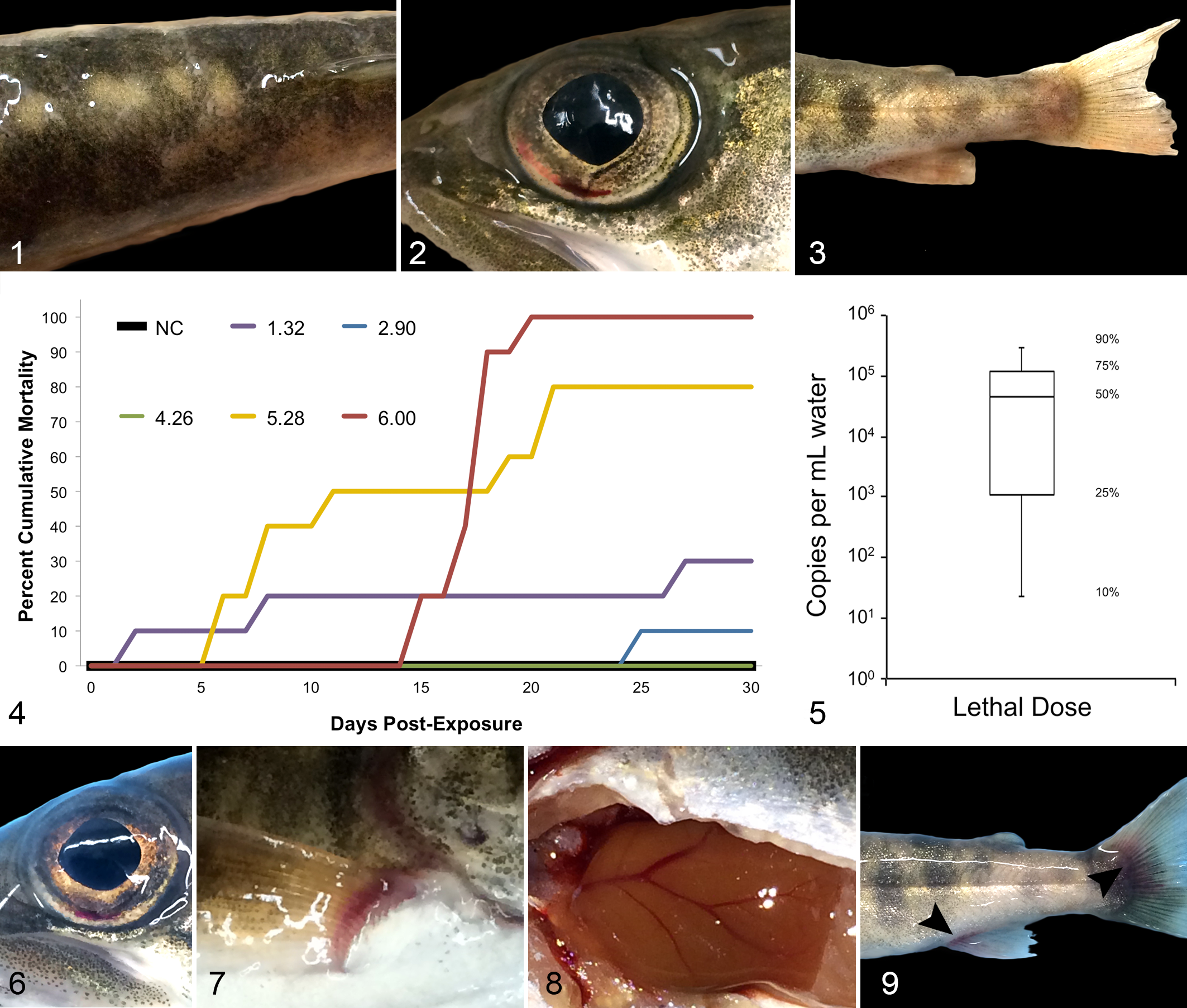
Epizootic epitheliotropic disease (EED; experimental infection via immersion bath exposure), juvenile lake trout.
Morbidity and Mortality Response to Varying Doses of EEDV
Five groups of fish were infected with EEDV at doses of 2.1 × 101 to 1.0 × 106 viral copies/mL of water, and monitored for a period of 35 days following viral exposure to the 10-fold dilutions of EEDV, after which time all survivors were euthanized. During that time, the negative control group had 2 mortalities (no evidence of EEDV infection) while mortalities in fish exposed to EEDV (presented as low dose to high dose, n = 10 per group) were 3, 1, 0, 8, and 10, respectively (Fig. 4). Clinical findings were similar to those detailed above. Using qPCR to test skin samples collected from all fish, EEDV was detected in all 10 fish infected with the high dose, one of the fish in the first dilution group, and none of the fish infected with the lower doses (Supplemental Table S1). Projected mortality percentages ranged from 2.3 × 101 (10%) to 3.8 × 105 (90%; Supplemental Table S2, Fig. 5). The median lethal dose (utilized for further studies) was calculated at 4.7 × 104 viral copies/mL water.
Clinical Disease and Gross Lesion Progression
Infected fish had mild exophthalmia and iridal reddening (possible congestion, hemorrhage, or hyperemia; Fig. 6) as early as 1 day PI, which might have been incidental due to euthanasia or fluid imbalance rather than a direct result of the virus targeting the eye (see qPCR results below). Other early signs of clinical disease (i.e., first observed in a minimum of 2 fish per day prior to Day 18 PI) included congestion at the base of pectoral and pelvic fins (Fig. 7) and along the isthmus, mild splenic pallor, and hyperemia or engorgement of enteric and hepatic blood vessels (Fig. 8). During later stages of disease (i.e., Days 21–42 PI), fish consistently had moderate to severe congestion of multiple fins, iridal reddening (possible congestion, hemorrhage or hyperemia), exophthalmia, multifocal to coalescing erosions, and ulcerations of the skin, fins, and caudal peduncle (Fig. 9), as well as congestion of visceral organs such as kidney, liver, and spleen. The percentage of fish with clinical disease increased on each sampling day from 33% on Day 6 to 100% on Days 15 to 42. Likewise, the total number of gross lesions each day increased from ≤6 on Days 0 to 12 (average of <1 per fish per day) to averaging >5 lesions per fish per day from Days 21 to 42. Mortalities occurred in the infected group on Day 28 PI (n = 4) while there were no mortalities or EEDV-attributable gross lesions in the negative control group.
Assessment of Viral Load in Tissues of Experimentally Infected Fish Using qPCR
The earliest detection of EEDV DNA was in a single fish on Day 9 PI. By Day 18 PI, viral DNA was detected in half of the fish sampled (3/6). As infection advanced, EEDV DNA was detected in multiple tissues from all fish sampled on Day 21 PI, through the end of the observation period, that is, Day 42 (Supplemental Table S3). There was no EEDV DNA detected in any of the tissues sampled from the negative control fish.
EEDV DNA was detected first in ocular tissues (Supplemental Table S3). And was detected in the skin and fins by Day 18 PI. From Day 21 through the end of the observation period, EEDV DNA was most prevalent in the eye, skin, fin, and gill tissue. There was no viral DNA detected in internal organs until Day 21 PI when brain, kidney, liver, and spleen tissues from multiple fish were EEDV-positive. The infection peaked by Day 28 PI, at which point the virus was detected in all tissues of all fish and 4 mortalities occurred. As infection subsided across the following 2 sampling events, EEDV was detected in some but not all internal organs of sampled fish. In contrast, EEDV remained at high levels in all external tissues, that is, eyes, skin, fins, and gills, through the end of the observation period.
When examining differences between tissue types across the entire study, eyes, skin, fins, and gills were most frequently EEDV-positive, yet the only statistically significant differences were between heart (n = 13) and eye, skin or fin samples (n = 25 each; P < .05). When individual days were examined, however, there were significant differences. On Day 18 PI, the number of positive skin and fin tissues (n = 2 each) was significantly greater than the number of positive gill, kidney, spleen, heart, liver, intestine, or brain tissues (n = 0 each; P < .01). On Day 21 PI, in pairwise comparisons, the number of positive eye, skin, fin, or gill tissues (n = 5 each) was significantly greater than the number of positive kidney (n = 3), spleen (n = 2), heart (n = 2), intestine (n = 3), or brain (n = 2) tissues (P < .01). On Day 35 PI, in pairwise comparisons, the number of positive eye, skin, fin, or gill tissues (n = 6 each) was significantly greater than the number of positive spleen (n = 3) or heart tissues (n = 4; P < .01). Finally, on Day 42 PI, the number of positive eye, skin, fin, or gill tissues (n = 6 each) was significantly greater than the number of positive kidney (n = 2), heart (n = 1), liver (n = 4), or intestine (n = 3) tissues. In addition to representing a larger number of positive samples throughout the study, the eye, skin, and fin also consistently contained the highest EEDV DNA loads, often 100- to 1000-fold higher than the amount of viral DNA detected in internal organs (Supplemental Table S3, Fig. 10). When first detected in the eye, fin, and skin (Days 9 and 18 PI), EEDV DNA loads ranged from 103 to 105 viral copies per mg host tissue (Supplemental Table S3). A similar range was observed on Day 21 PI, when EEDV was first detected in the gills and remaining internal tissues, while the viral loads in the eye, skin, and fin reached 107 to 108 copies per mg host tissue.
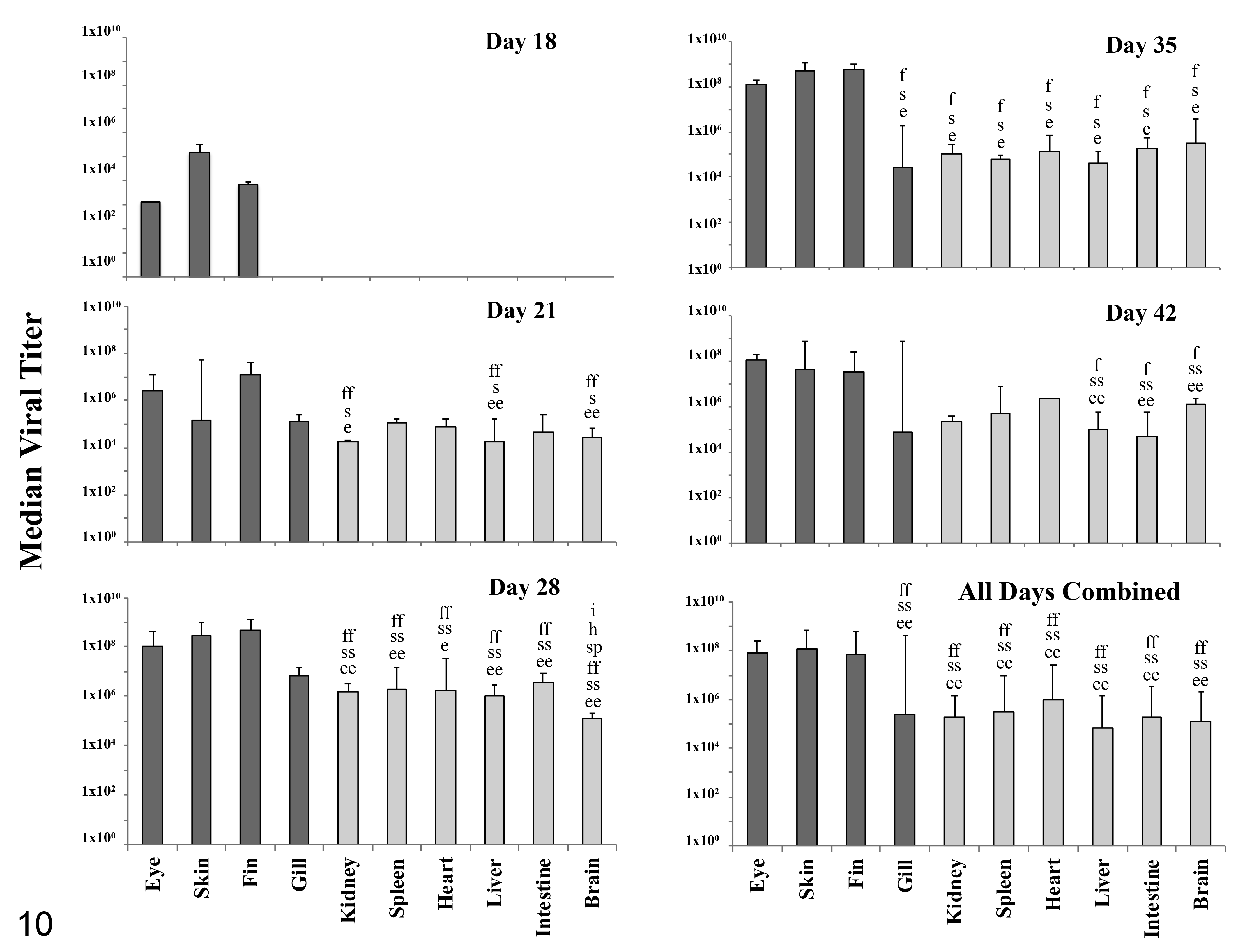
Median titer of epizootic epitheliotropic disease virus by tissue type and sampling day following experimental infection via bath immersion. Letters above bars indicate a statistically significant difference in viral titers compared to specified tissue; P < .05 designated by a single letter, P < .01 designated by a double letter. Letters correspond to tissues as follows: (e) eye, (s) skin, (f) fin, (g) gill, (k) kidney, (sp) spleen, (h) heart, (l) liver, (i) intestine, (b) brain. Error bars signify 1 standard deviation. External tissues are shaded dark gray while internal tissues are light grey.
At the apparent peak of infection, on Day 28 PI, viral loads in external tissues ranged from 106 to 109 copies per mg host tissue while the viral loads in internal tissues ranged from 104 to 107 copies per mg host tissue (Supplemental Table S3). While certain gill tissues contained a viral load equivalent to those of the eye, skin, and fin, as a whole, viral loads in gill tissues were more similar to viral loads of internal tissues.
During the final 2 sampling events, the number of positive tissues decreased, and the viral loads in internal tissues decreased, yet the viral loads in the eye, skin, and fin remained high (up to 109 viral copies per mg host tissue). By pairwise comparison, viral loads in the eye, skin, and fin tissues were significantly higher than in internal organs on Day 21 (kidney, liver, and brain; P < .05), Day 28 (kidney, spleen, heart, liver, intestine, and brain; P < .05), Day 35 (all other tissue types; P < .01), and Day 42 PI (liver, intestine, and brain; P < .05).
An additional statistical analysis was performed comparing the number of positive samples, as well as the average viral load, by sampling day, from all external tissues combined versus all internal organs combined on Days 21 to 42. On each of these 4 sampling days, the external tissues had a significantly higher viral load compared to internal organs (P < .01) and except for Day 28 when EEDV was detected in all 60 tissues tested, the external tissues also harbored the virus in a larger number of tissue samples than the internal organs (P < .01).
Microscopic Lesions and Scoring
A total of 754 tissues from 33 control fish and 67 infected fish were examined for microscopic lesions. The original study period was extended to account for slow disease progression and as such, fewer control fish were available on the final 2 sampling days (see supplemental tables). Additionally, samples were only collected for histopathologic analysis if fish were freshly euthanized—mortalities on day 28 were not included due to poor tissue quality. A small number of tissues (<5%) were unable to be appropriately examined due to preservation, preparation artifact, or sectioning angle that led to insufficient tissue of each organ available for analysis.
The primary microscopic lesions were in the skin and fins (Figs. 11–13). The earliest lesions occurred in the epidermis at Day 21 and consisted of individual epithelial cell degeneration and necrosis, and single acantholytic cells (Fig. 11a). In multiple areas, there were intracytoplasmic eosinophilic accumulations in a few epithelial cells as well as rare intraepithelial inflammatory cells (Fig. 11a). Lesions progressed in some areas to epithelial erosions with sloughing of degenerated epithelial cells. By Day 28, epithelial erosions were more severe with extensive intraepithelial inflammatory infiltrates and cellular degeneration (Fig. 12a) and in the most advanced lesions, epithelial cells were undergoing degeneration and necrosis throughout all layers of the epidermis. The most severe lesions occurred at Day 35 and consisted of massive epithelial ulceration and complete epidermal loss (Fig. 13a). Cutaneous lesions did not occur prior to Day 21, but were present in all fish examined at later time points except for a single fish on Day 35 and one fish on Day 42.
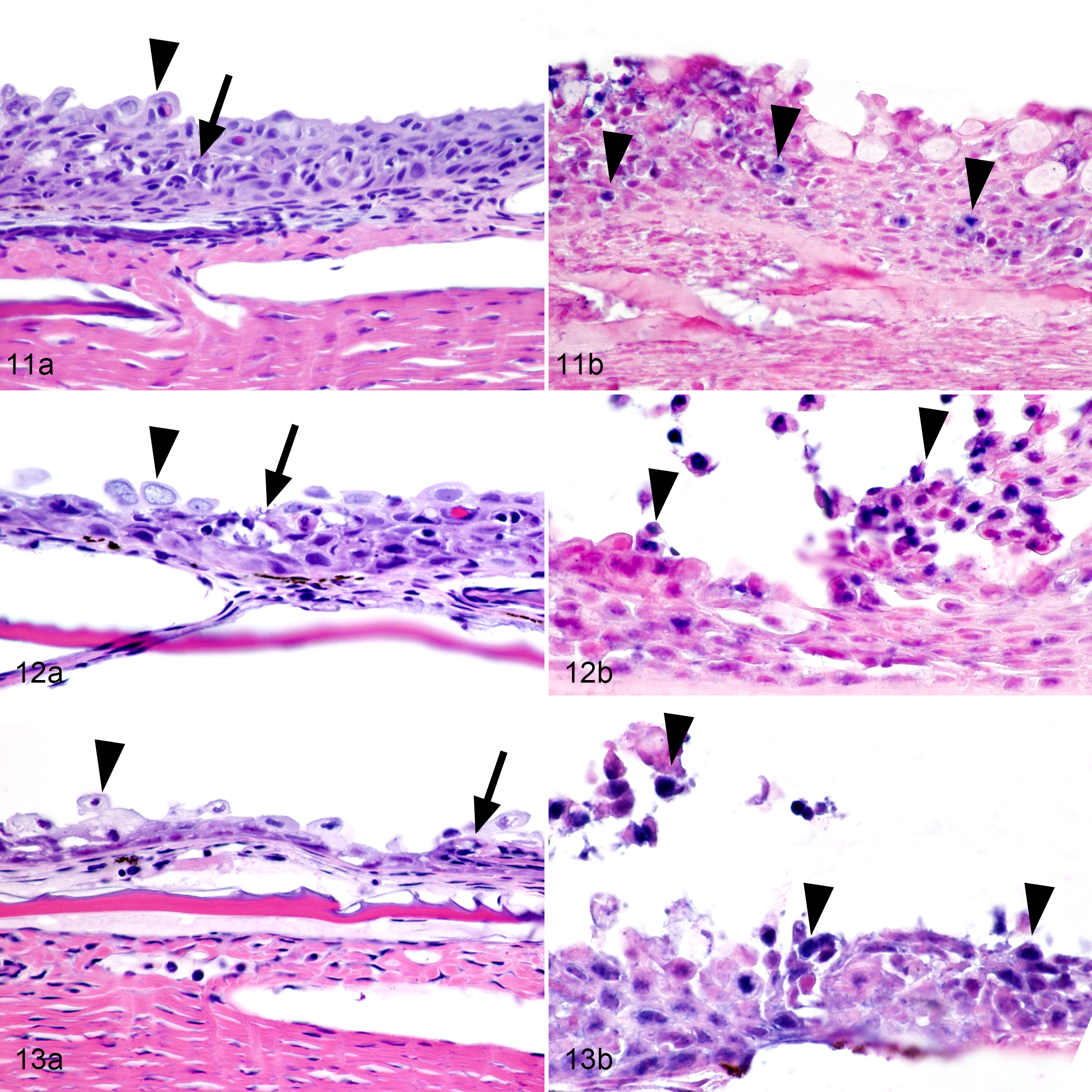
Epizootic epitheliotropic disease (EED; experimental infection via immersion bath exposure), juvenile lake trout, skin. Hematoxylin and eosin (11a, 12a, 13a) and in situ hybridization for EED virus (11b, 12b, 13b; blue labeling) with nuclear fast red counterstain.
In the gills, early lesions were characterized by mild thickening of the primary lamellae (epithelial cell hyperplasia), infiltration of mononuclear cells into the lamina propria, swelling of epithelial cells, congested blood vessels, and few degenerated epithelial cells (Fig. 14a). More advanced gill disease (Figs. 15a, 16a) was characterized by blunting and fusion of the secondary lamellae and massive infiltration of mononuclear cells. However, mild proliferative branchitis with focal to multifocal lymphohistiocytic inflammatory cell infiltrates was present in some control fish.
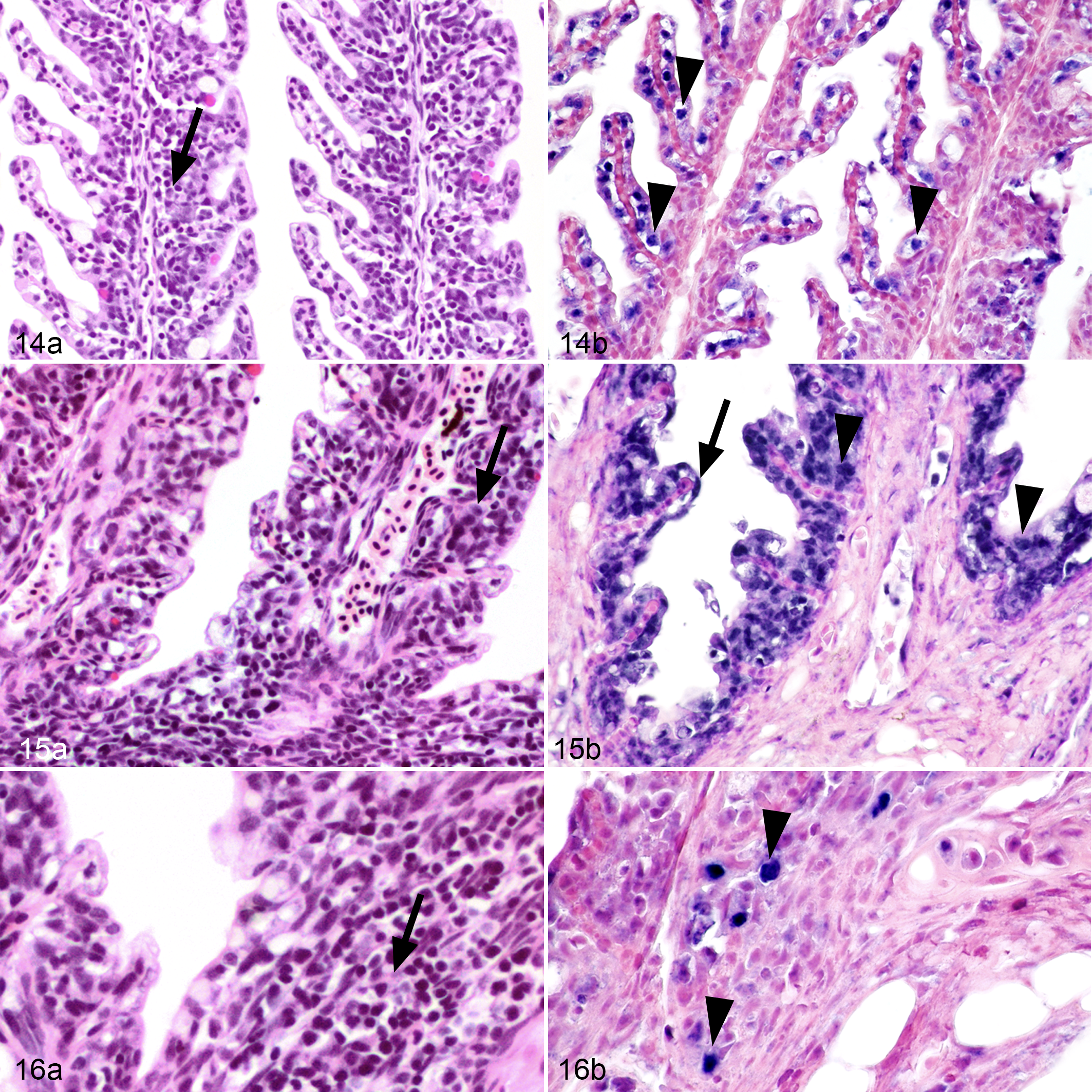
Epizootic epitheliotropic disease (EED; experimental infection via immersion bath exposure), juvenile lake trout; gills. Hematoxylin and eosin (14a, 15a, 16a) and in situ hybridization for EED virus (14b, 15b, 16b; blue labeling) with nuclear fast red counterstaining.
Hepatocellular vacuolation was nearly ubiquitous among control fish throughout the study, and among infected fish during the early weeks of the experiment, yet was present in only 30% of infected fish examined during the last 3 sampling weeks. There was a mild, patchy proliferative branchitis in the gills of many fish in both groups throughout the study. This included blunting and fusion of the secondary lamellae, with epithelial cell hyperplasia filling the spaces between the lamellae, and infiltration of mononuclear cells. These lesions were not obviously associated with either parasites or bacteria. There was no evidence of an increasing severity over time, and there were no differences in distribution and severity of branchitis between fish in the negative control group and the EEDV group.
Also at 28 days post exposure, both examined fish had severe lymphoid necrosis of the spleen (Fig. 17) and lymphohistiocytic perivasculitis affecting multiple organs, but this was most severe in the epicardium (Fig. 18), omentum (Fig. 19), and liver. Lymphohistiocytic perivasculitis was most severe in the omentum of one fish euthanized on Day 28 and there were rare intranuclear inclusion bodies in monocytes in the lumens of affected blood vessels. As early as Day 3 post exposure, areas of single cell necrosis were in the liver, including hepatocytes with deeply basophilic, shrunken, pyknotic nuclei and hypereosinophilic, contracted cytoplasm. Such changes occurred with increasing severity as evidenced by increasing numbers of foci of pyknotic cells and in more fish per sampling day. Beginning on Day 9, ≥66% of fish were affected by similar lesions with 100% of fish that were sampled over the final 3 weeks with at least mild changes. Starting at Day 18, single cell necrosis focally expanded to small foci of hepatocellular necrosis and there was also lymphohistiocytic perivascular inflammation affecting both hepatocellular arteries and veins. Lesions were most severe in fish collected on Days 21 and 28, but similar lesions were found in fish examined at later days.
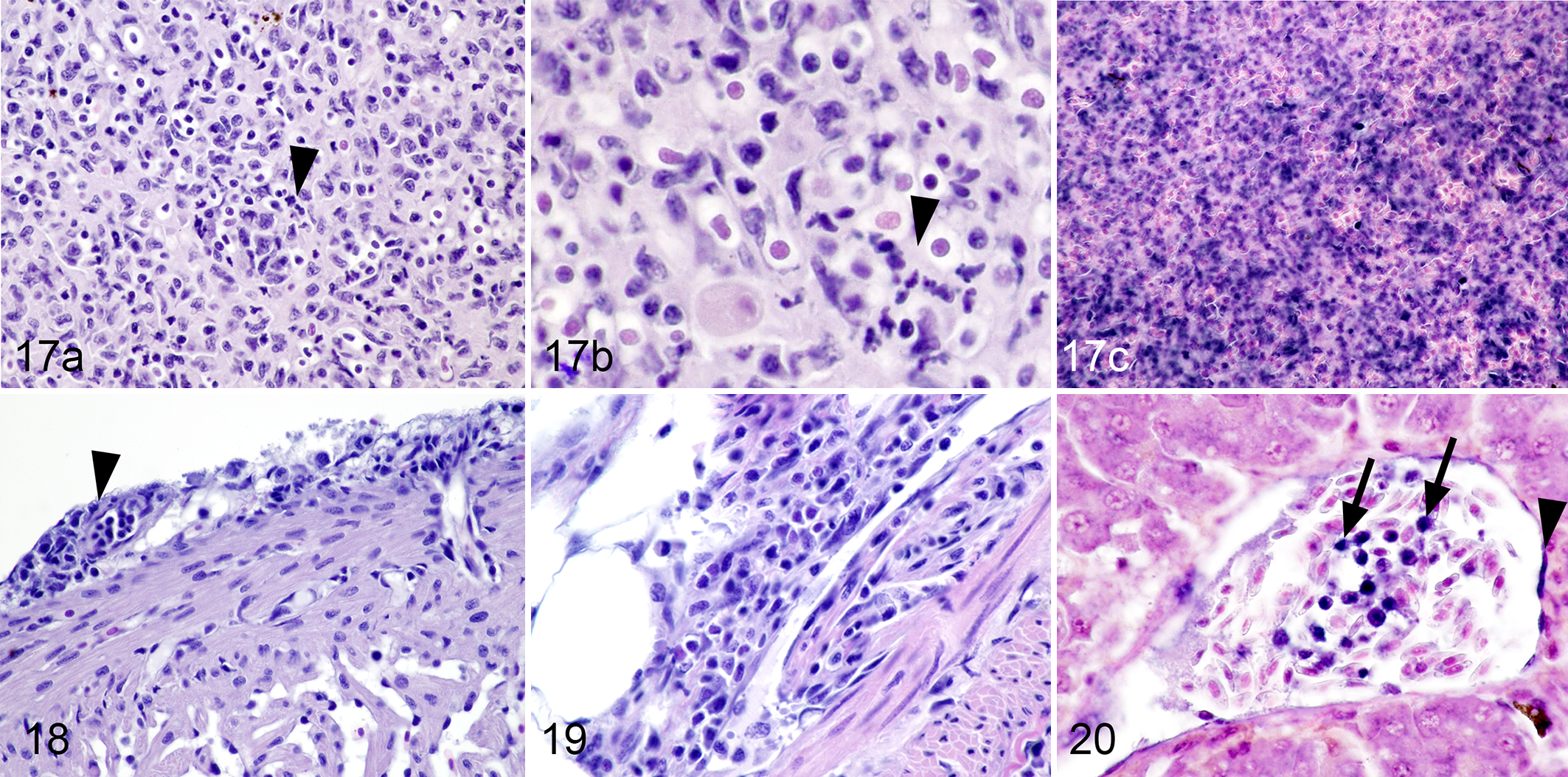
Epizootic epitheliotropic disease (EED; experimental infection via immersion bath exposure), juvenile lake trout; spleen (17), heart (18), omentum (19) and liver (20). Hematoxylin and eosin (17a, 17b, 18, 19) and in situ hybridization for EED virus (17c, 20; blue labeling) with nuclear fast red counterstaining.
Epithelial lesions in the skin and fins were not present until Day 21 PI, and represented the first evidence of viral disease in external tissues. While single cell necrosis in the liver was described at an earlier stage of the experiment, multi-organ involvement with lymphohistiocytic perivasculitis and lymphoid necrosis did not occur until Day 28 PI.
Visualization of EEDV in Infected Tissues Using ISH Assay
The ISH assay was performed on all tissues from fish that tested positive via qPCR and selected time-matched controls. Due to the inherent lower sensitivity of ISH compared to qPCR, as well as the challenges associated with processing and refacing blocks containing small tissue samples from young fish, in order to clearly demonstrate ISH positivity, some photomicrographs were obtained from different fields of view and/or different blocks for H&E and ISH analyses. Epithelial cells in the skin and gills, vascular endothelial cells, and monocytes in the spleen, within vascular lumina and within inflammatory lesions, were positive for EEDV by ISH in fish sampled on Days 28, 35, and 42 PI. The number of positive cells varied between days with the largest number of positive cells correlating to the most advanced stages of disease and the highest viral load based on qPCR. Positive labeling was not evident in any of the negative control tissues tested.
In skin tissue collected from fish in early stages of disease, positive ISH labeling confirmed the presence of EEDV in the nuclei of degenerating epithelial cells as well as in infiltrating lymphocytes and dendritic cells. In early skin lesions (Fig. 11b) viral nucleic acid was detected in the nuclei of individual necrotic epithelial cells. In advanced cutaneous lesions (Fig. 12b), viral nucleic acid was present in large numbers of nuclei of degenerate and necrotic epithelial cells that had commonly sloughed. In the most severe skin lesions (Fig. 13b), viral nucleic acid was detected in the nuclei of the vast majority of epithelial cells throughout all layers prior to epithelial loss.
In the gills, viral nucleic acid was detected in the nuclei of morphologically unremarkable epithelial cells (Fig. 14b) during early stages of infection. In more advanced gill disease (Figs. 15b, 16b), viral nucleic acid was detected in the nuclei of attenuated epithelial cells and nuclei of infiltrating mononuclear cells. The severity of these gill lesions did not correlate with the number of days PI.
During the later disease stages, lesions in internal organs most likely developed secondary to viremia, as is supported by the sudden presence of large amounts of virus in internal organs by qPCR. Viral nucleic acid was detected in the nuclei of large numbers of mononuclear cells in the spleen (Fig. 17c). A severe lymphohistiocytic perivasculitis occurred, most likely secondary to viral infection of endothelial cells (Fig. 20). Significant nuclear labeling of mononuclear cells in the vessels of different organs indicates likely development of viremia in later stages of disease.
Discussion
The present study is the first to unravel which lake trout tissues and cell types support the replication of EEDV. The described ISH protocol resulted in the visualization of EEDV DNA in formalin-fixed, paraffin-embedded tissues collected from infected lake trout. The combination of gross and microscopic lesions, qPCR detection, and ISH visualization sheds light on the spread of this virus within the tissues of its host, thereby improving our understanding of EEDV pathogenesis.
The challenges outlined in Table 1 served multiple purposes: to maintain virus infectivity, as some herpesviruses are known to lose their virulence following extensive frozen storage; 26 to serve as a guide to decide on the doses for future experimental challenges; to increase viral titers in available stocks; and to expand our knowledge about the disease course and associated clinical signs following exposure to EEDV. With these trials, we were able to successfully maintain an active and virulent stock of EEDV over the span of 12 months, increase the relative viral loads within each stock batch, and reproduce clinical EED consistent with natural outbreaks in a controlled laboratory environment.
Based on the results of the dose-dependent challenges, for future in vivo EEDV experiments to produce clinical morbidity and mortality in juvenile lake trout, we recommend an immersion challenge model as follows: virus dose at or above 4.7 × 104 copies/mL water, exposure of at least 1 hour, and constant maintenance during and after viral exposure of water temperature at 9 ± 1 °C. The use of these conditions with EEDV has demonstrated the ability to cause juvenile LS strain lake trout to develop clinical disease. Finally, we recommend a monitoring period of at least 60 days rather than the 30 used here, as this virus appears to be rather slow growing and may take additional time to develop an active infection.
Our data demonstrated that the EEDV incubation period is between 9 and 18 days following a water-borne infection at 9 ± 1 °C, significantly longer when compared to other viral diseases of fish. Many factors may affect the length of this initial incubation period including virus exposure dose, fish density and stress, water temperature, or prior health status. 4,21,24 The earliest gross lesions occurred in the eyes, skin, and fins with qPCR findings clearly pointing to the early involvement of the integument and ocular tissues in EEDV infection, underscoring the potential of the integument as a primary target for EEDV to enter its host. While exophthalmia and iridal reddening were described grossly, no ocular lesions occurred microscopically, which may suggest that at least a portion of these observed gross lesions were due to vessel congestion. These early ocular lesions were most likely unrelated to viral pathogenesis (e.g., euthanasia artifact) as only from day 18 and later could ocular lesions consistently be associated with viral presence. The gross skin lesions corresponded with the degenerative and necrotic epidermal lesions described microscopically and represent the first manifestation of EEDV in lake trout. While the experimental infection in this study took place via exposure to virus-laden water, it is plausible to consider skin-to-skin contact with infected fish as another potential source of infection. Skin is known to be a primary portal of entry for many aquatic viruses including koi herpesvirus and infectious salmon anemia virus. 5,25
Early cutaneous lesions were followed by viremia that resulted in severe lesions in internal organs and mortality at Day 28. The severity of gross lesions, histologic lesions, and mortalities also coincided with peak viral load around 28 days PI based on qPCR. These results demonstrate the prolonged and delayed development of clinical disease following exposure to EEDV. This is ecologically and epidemiologically important in terms of identifying potential viral sources following a mortality event. The extended incubation period in the present study suggests that exposure might occur up to a month prior to the onset of clinical disease.
In the early stages of infection, EEDV seems to target squamous and cuboidal cells of the epidermis, and at later stages, the virus becomes ubiquitously distributed throughout the layers of the skin epithelium, often leading to necrotic changes in infected cells and subsequent erosions and ulcerations. The fact that the integument and eye tissues maintained relatively high copy levels of EEDV DNA along with intense intranuclear ISH staining attests to these organs being a major site of virus replication throughout the disease course. As the epithelium erodes and ulcerates in advanced stages of disease, the sloughed epithelial cells—full of virus—may serve as a primary source of infection for cohabitating fish. In addition to the skin epithelial cells, intense staining was noticed in the nuclei of intraepidermal mononuclear cells, whose numbers also increased as the infection progressed. The intranuclear ISH staining of the mononuclear cells indicates that EEDV replicates in these mononuclear cells as well, a matter that may aid in the virus dissemination. We speculate that secondary infection of intraepidermal dendritic cells and infiltrating intracutaneous interstitial dendritic cells as well as monocytes may present the first stage of viremia as has been described with other herpesvirus infections in fish and other vertebrates. 6,12,14
Data acquired in the current study demonstrated that gills actually harbored significantly fewer EEDV copies during the early stages of the disease than the integument, despite their external location. Additionally, no progression of branchitis occurred in EEDV-infected fish throughout the course of infection and differences in microscopic lesions of infected and control fish were minimal. This is in contrast to trends with ictalurid herpesvirus 1, cyprinid herpesvirus 2, and anguillid herpesvirus 1, where host gills are preferentially and persistently infected. 16,23,27 The embryonic origin (ectoderm vs endoderm) of the gills in bony fish is currently debated. However, recent evidence using cell lineage tracers demonstrated that pharyngeal gills of gnathostomes (jawed vertebrates—cartilaginous and bony fishes) are endodermally derived. 10 In this study, however, similar to the skin, ISH staining was observed in the branchial epithelium. The virus was also detected in the mononuclear cells both infiltrating gill tissues, and inside blood vessels, suggestive of a possible cause of viremia. Detection of EEDV in endothelial cells lining blood vessels also supports EEDV replication in these cells similar to other herpesvirus infections. 8,26
Positive staining of mononuclear cells occurred in large numbers in the spleen of fish at advanced stages of the disease, suggesting that viral replication also occurs within the splenic hematopoietic cells. As with the epithelial and mononuclear cells of the skin and gills, this staining was intranuclear, lending support to active viral replication. These infected splenic cells may represent a large source from which virus can spread to other organs and tissues.
Following the initial detection of EEDV DNA in external tissues, it was approximately 2 weeks before any virus appeared in internal tissues. This apparent delayed spread of virus to visceral organs (e.g., kidney, spleen, and liver) after initial occurrence is evidence of EEDV first targeting and establishing an infection in external tissues, followed later by spread and development of systemic disease. This pattern of an initial infection site in external tissues followed by systemic spread has also been described in the herpesviruses of cyprinids, catfish, and eels. 9
Similar viral copy levels were detected (by qPCR) in brain, liver, kidney, heart, and intestine. However, EEDV could not be visualized by ISH in the parenchyma of these organs. These findings suggest that cells in these organs were not supportive of virus replication and the virus detected by qPCR originated from tissue fluids, circulating blood or blood cells. The presence of intense ISH labeling in mononuclear cells but not in other cells types within the visceral organs enforces our notion that EEDV replication occurs in integument epithelial cells, endothelial cells, and mononuclear cells (in skin, gills, circulating blood and spleen) only.
This study also provides evidence for the potential cause of death of EEDV-infected fish. During the last 3 weeks of the experiment, cutaneous lesions occurred in 80% to 100% of infected fish. Epithelial necrosis as reported here is known to lead to hypo-osmotic shock and death in fish infected with other viruses such as koi herpesvirus (cyprinid herpesvirus 3). 19 Additionally, by Day 28 PI, infected fish had developed lesions in multiple organs that were primarily centered on vessels. Intranuclear inclusion bodies in blood monocytes indicates transmission of virus from affected surface epithelium through infected histiocytic cells causing viremia and secondary infection of internal organs.
This study provides important information that directs pathogen detection. Kidney and spleen are commonly used for diagnosis of pathogenic aquatic viruses. However, as is also the case with some of the other aquatic herpesviruses such as koi herpesvirus, 9,19 these are less appropriate tissues for diagnosis of EEDV. When compared to external tissues (e.g., eye, skin, fin), the kidney and spleen carry consistently lower viral loads, that are also not detectible as early in, or as long throughout, the course of infection. While internal tissues collected and tested from a highly infected individual may have readily detectible levels of EEDV, in order to maximize chances at detecting a low level of virus infection, external tissues should be used for screening instead. Because of high virus copy number in both skin and fin samples from the current study, fin clips are a potentially promising, cost-effective, nonlethal method of sampling both captive and wild fish, to allow for continuous monitoring of the EEDV in a population.
Finally, when comparing the relatively minor mortality rates in the current study with the high viral titers, a concern arises that lake trout are able to not only survive an EEDV infection but such fish may in fact be able to act as a viral reservoir and subsequently pose a substantial risk to younger (i.e., more susceptible) populations of fish. While we have demonstrated that EEDV causes a lethal disease in experimentally challenged lake trout, we have also shown that fish are able to survive despite high viral loads and advanced lesions. This highlights the need for aggressive and persistent screening for EEDV in captive lake trout in order to rapidly identify and quarantine any potentially infected populations before the virus can spread throughout the hatchery. The data provided herein establishes that EEDV replication is supported by a myriad of cells of different embryonic origins such as skin epithelium, gill epithelium, endothelial cells, and circulating mononuclear cells underscoring its wide range of susceptible cell types. This information can be used to alter screening efforts of Great Lakes Basin lake trout populations as well as to focus future research into the location and establishment of latency as an explanation why there have been long periods in which this virus was not detected. Recent mortality events 24 have highlighted the fact that EEDV remains present and a threat within the Great Lakes basin, and this current study has provided key information regarding the host-virus interactions.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Shavalier_et_al - Disease Progression in Lake Trout (Salvelinus namaycush) Experimentally Infected With Epizootic Epitheliotropic Disease Virus (Salmonid Herpesvirus-3)
Supplemental Material, Combined_supplemental_materials-Shavalier_et_al for Disease Progression in Lake Trout (Salvelinus namaycush) Experimentally Infected With Epizootic Epitheliotropic Disease Virus (Salmonid Herpesvirus-3) by Megan Shavalier, Mohamed Faisal, Thomas P. Loch, Scott D. Fitzgerald, Tuddow Thaiwong and Matti Kiupel in Veterinary Pathology
Footnotes
Acknowledgements
The authors would like to acknowledge the technical contributions of the histopathology technicians at the Michigan State University Veterinary Diagnostic Laboratory. We would also like to thank the Michigan Department of Natural Resources, specifically Marquette State Fish Hatchery and staff for supplying the fish used in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Great Lakes Fishery Trust (Award # 2014.1455).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
