Abstract
This is a histopathologic and endocrinologic study of 6 calves diagnosed with cryptorchidism. Cases 1–3 were diagnosed as resembling testicular regression syndrome. In cases 1 and 2, the extracted tissue was a small, firm, gray-white mass, and there was lack of obvious testicular tissue in case 3. Histopathologically, the excised tissue in cases 1–3 was a fibrotic testicular remnant with inflammation, mineralization, hemosiderin-laden macrophages or lipofuscin-laden macrophages, and lack of germ cells and interstitial endocrine cells. These findings were compared with cases 4–6, which were diagnosed as testicular hypoplasia due to cryptorchidism. These cases had small but otherwise grossly unremarkable intra-abdominal testicular tissue and histologically had a few germ cells and sustentacular cells with arrested spermatogenesis and an increase in interstitial endocrine cells. Cases 1–3 had more severe degenerative changes compared with cases 4–6. In case 2, the average diameter of the seminiferous tubules was much smaller than in cases 4–6, and there were few tubule cross sections. Anti-Müllerian hormone (214 pg/ml) was detected in the plasma of case 2. Based on the macroscopic and histopathologic findings as well as endocrinologic profiles, the testicular degeneration in cases 1–3 was considered similar to that of testicular regression syndrome. In this condition, it is thought that a normally developing intra-abdominal testis undergoes degeneration due to heat or a vascular disorder such as torsion.
Keywords
Congenital anomalies involving the testis in animals include testicular hypoplasia, monorchidism, and cryptorchidism. 1,9,18,21,23,24,27,31,35 Testicular hypoplasia, in which the testis fails to grow to a normal size, is very common in bulls and can be associated with sex chromosome disorders of sex development and cryptorchidism. 9,18 Microscopic findings of hypoplasia include a total lack of germ cells, decrease in germ cell number, arrested spermatogenesis at certain stages, testicular sustentacular cell–only pattern, or intermixing of normal and abnormal tubules in the most severe cases. 9,18 In feline, canine, and equine monorchidism, there is a complete absence of 1 testis. 20,21,23,24,35 In previous studies, the testis was completely absent on one side, and dense fibrous tissue was observed in a stallion. 24 Cryptorchidism is the failure of 1 or both testes to descend into the scrotum, with an incidence of 0.2% in cattle. 1,31 Fattening bulls with cryptorchidism usually results in bulls with a stout body type that exhibits masculine characteristics in the neck and skull because of the testosterone produced by their functional abdominal testes. 27 The traditional endocrinologic approaches to evaluating the functionality of testes in the abdomen are the measurement of testosterone concentrations before and after human chorionic gonadotrophin stimulation and the measurement of plasma anti-Müllerian hormone (AMH), which is specifically produced by testicular sustentacular cells. 13
In humans, anorchidism or monorchidism is understood as testicular regression syndrome or vanishing testis and is a condition in which a normally developing testis is thought to have once existed in fetal life and has subsequently atrophied and disappeared. 2,10,17,25 In many reports, a fibrotic testicular remnant that has calcification, hemosiderin, and fibrosis is found at the terminus of the blood vessels (pampiniform plexus) and deferent duct. 2,5,6,8,9,25,30,38 The cause of testicular regression syndrome is thought to be late antenatal or perinatal vascular thrombosis, torsion, or endocrinopathy. 2,10,25,30 However, this condition has not been reported in any animal species.
Testicular degeneration in animals can result from heat, advancing age, chemicals, and so on. 9 Histopathologic findings in heat-induced testicular degeneration include reduction in spermatogenesis, reduction of tubular diameter, reduced number of testicular sustentacular cells, thickened hyaline basement membrane, granulomas, and mineralization. 9,19 Testicular torsion or ischemic necrosis is observed in the cryptorchid boar, stallion, and dog. 4,9,22
In this study, we conducted histopathologic and endocrinologic examinations in 6 cryptorchid calves, 3 with drastically small testicular remnants that were observed by laparoscopy and 3 cryptorchid calves. The degenerative changes in these calves were similar to testicular regression syndrome in humans and testicular hypoplasia, respectively.
Materials and Methods
Ethics
All clinical protocols were performed in compliance with the Institutional Review Board for Animal Experiments of the University of Miyazaki, Japan. All experimental procedures were approved by the Animal Care and Use Committee of the University of Miyazaki (approval No. 2017-027).
Case Presentation
Cases 1 to 3 were Japanese Black calves aged 7, 5, and 9 months, respectively. The retained testes (cases 1–2: unilateral; case 3: bilateral) were not found on the body surface by palpation or ultrasonography. Therefore, after the endocrinologic evaluation, the intra-abdominal testicular structures were identified and extracted by laparoscopy, as in a previous study. 12 For comparison, we used 3 unilaterally cryptorchid Japanese Black calves (cases 4–6, aged 7, 8, and 7 months, respectively), in which the testicular structure was revealed on the body surface by palpation and ultrasonography. After the descended (intrascrotal) testes were surgically removed, the intra-abdominal testes were also extracted by laparoscopy. The left-side intrascrotal testes of 5 normal 9-month-old Japanese Black male calves (calves 7–11) were surgically removed and used as controls.
Histopathologic Examination
The extracted intra-abdominal testicular structures of 6 cases and the scrotal testes of 5 control cases were fixed in 10% neutral buffered formalin and then routinely processed for paraffin embedding, sectioned at 2 μm, and stained with hematoxylin and eosin (HE). Samples from cases 1–3 were also stained with periodic acid-Schiff (PAS), Masson’s trichrome (MT), and Berlin blue. Histologically, the total number of the cross sections of seminiferous tubules was counted. In those cases with seminiferous tubules, their size was measured in 5 high-power fields.
Immunohistochemical Examination
Immunohistochemistry was performed using monoclonal antibodies against cytokeratin (clone AE1/AE3; Dako, Glostrup, Denmark) and vimentin (clone V9; Dako) and polyclonal antibodies against Iba1 (Wako, Osaka, Japan), inhibin-alpha (Sigma-Aldrich, St Louis, Missouri, USA), and AMH (MIS[C-20]: sc-6886; Santa Cruz Biotechnology, Santa Cruz, California, USA). Briefly, antigens were retrieved by incubation with citrate buffer (pH 6.0) at 105°C for 10 minutes. Endogenous peroxidase activity was suppressed by 3% hydrogen peroxide in methanol for 10 minutes at room temperature. After the reaction, the sections were incubated with blocking solution (Nacalai Tesque, Kyoto, Japan) at room temperature for 30 minutes to avoid nonspecific reactions. After incubation with the primary antibody at 4°C overnight, the sections were incubated with a horseradish peroxidase–labeled anti-mouse and anti-rabbit immunoglobulin (Envision Dual Link System-HRP; Dako) or anti-goat immunoglobulin G (Histofine Simple Stain MAX-PO [G]; Nichirei, Tokyo, Japan) at 37°C for 30 minutes and then reacted with 0.05% 3,3′-diaminobenzidine and 0.03% hydrogen peroxide in Tris-hydrochloric acid buffer. Afterward, the sections were counterstained with hematoxylin, dehydrated in a graded series of ethanol, cleared with 3 changes of xylene, and mounted using a mounting solution. Negative controls were performed by replacing the primary antibodies with phosphate-buffered saline, and positive controls were performed by using normal testis from calves 7–11.
Endocrinologic Examinations
Blood samples were collected by jugular venipuncture into vacuum tubes with ethylene diamine tetra-acetic acid just before extracting the descended testes in the control and more than 7 days after castrating the contralateral descended testis in cases 1–6. The sera were separated by centrifugation (1500 × g for 30 minutes at 4°C) and stored at –30°C until the hormonal assay was conducted. The plasma AMH concentrations were measured by an enzyme-linked immunosorbent assay (ELISA) (bovine AMH ELISA, AL-114; Ansh Labs, Webster, Texas, USA). The plasma testosterone concentrations were measured by an enzyme-linked fluorescent assay (VIDAS testosterone Ⅱ; Japan bioMerieux, Tokyo, Japan). The sensitivity and the intra-assay coefficients of variation were 11 pg/ml and <3% in the AMH assay and 0.05 ng/ml and <10% in the testosterone assay. 14 If the plasma AMH and testosterone were not detected, the value was defined as the lower limit of detection (sensitivity).
Statistical Analysis
All statistical analyses were conducted using JMP12 (SAS Institute Inc., Cary, North Carolina, USA). The Wilcoxon signed-rank test was used to compare the plasma AMH and testosterone concentrations between each group. A P value of less than .05 or .1 was considered significant or as showing a tendency toward significance, respectively. All data are shown as mean ± SD.
Results
Extracted Testicular Structures
In cases 1 and 2, the results showed unilateral cryptorchidism and blood vessels with a small (Table 1), firm, gray-white mass and fibrotic tissue at one end. Elements of the deferent duct, testicular artery, and pampiniform plexuses were obvious, as were testicular tissue and atrophied epididymis (Figs. 1, 2). The cut surfaces of both masses were brown in the central area (Fig. 1, inset). The peripheral area of the masses was occupied by numerous vessels that extended toward the central area. In case 3, which was bilaterally cryptorchid, testicular tissues were not clearly observed, but both blood vessels (pampiniform plexus) and deferent ducts were identified (Fig. 3). In cases 4–6 (cryptorchidism with testicular hypoplasia), remarkable gross changes in intra-abdominal testicular tissue were not observed, except for their small size (Table 1).
Histologic Structures Comprising Intra-abdominal Testicular Remnants or Testes on the Affected Side.a

Abbreviations: –, not observed; +, observed; n/d, analysis was not done; n/r, not recorded.
a Difference in testicular structures was indicated.
b Total number and average size (μm) of cross sections of seminiferous tubules. By comparison, the normal controls (calves 7–11) had >100 cross sections of seminiferous tubules with average diameters of 174, 171, 146, 146 and 143 μm.
c The results indicate both intra-abdominal remnants.
d Testicular tissues were not clearly observed.
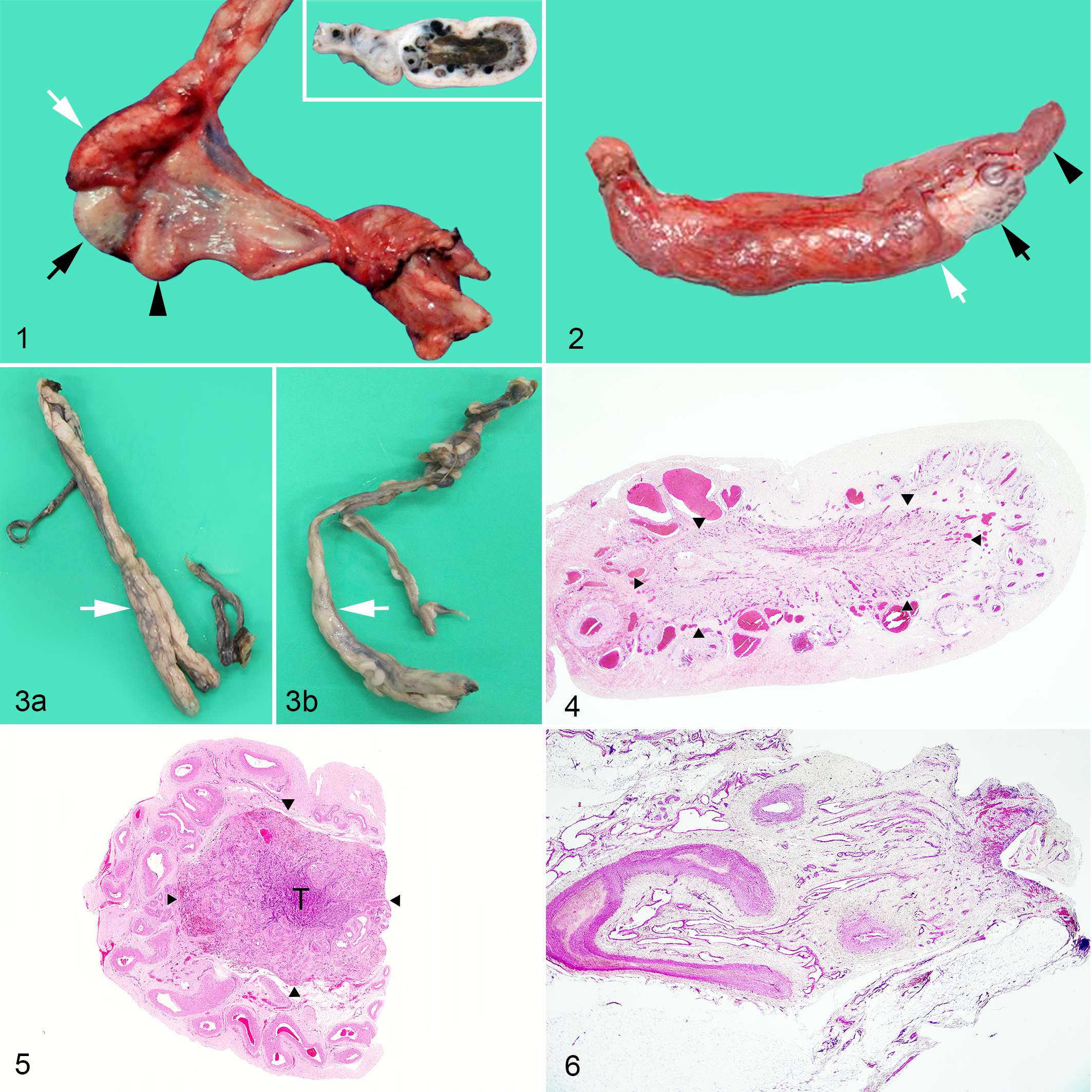
Histopathologic Findings
Cases 1–3
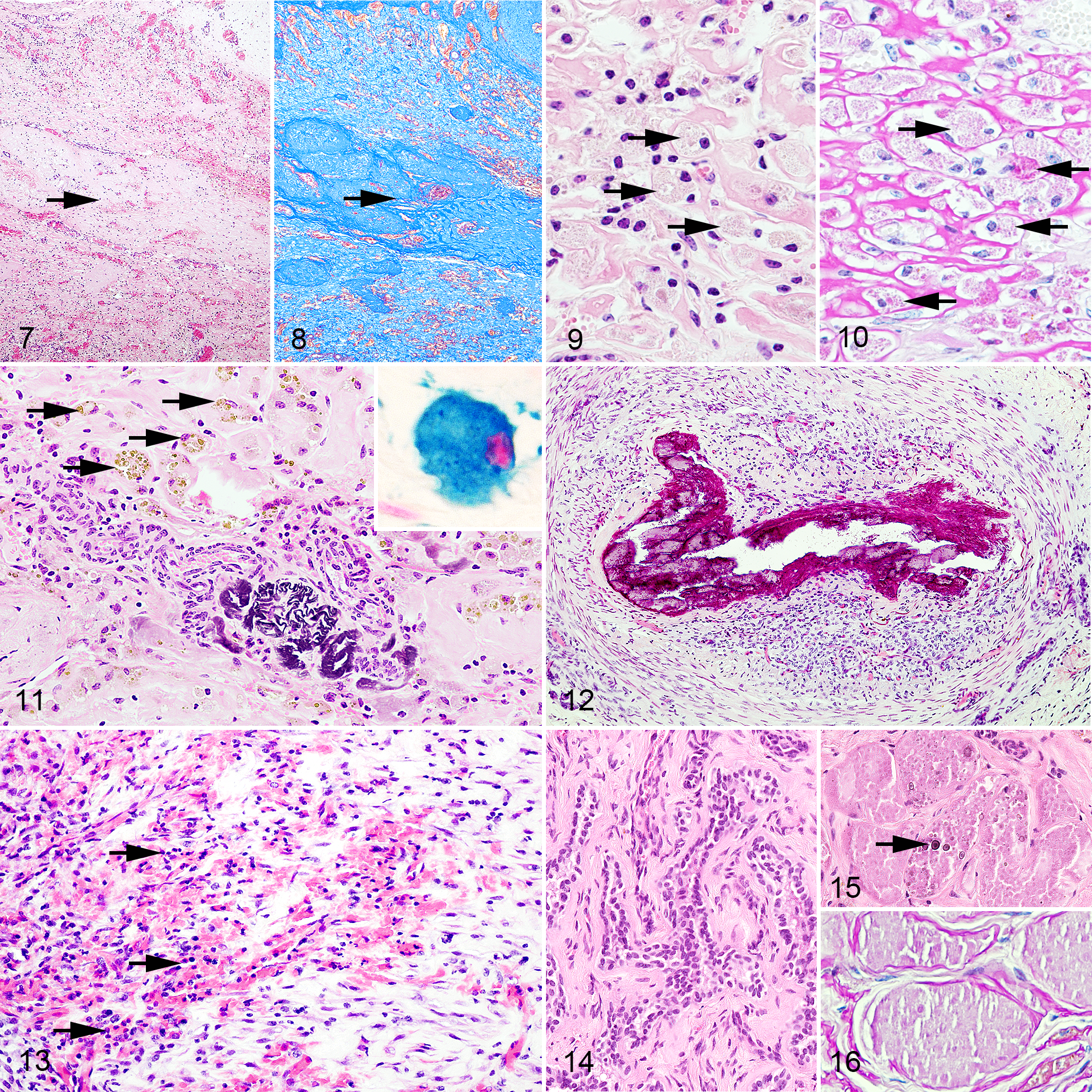
The testicular nodules were composed of fibrovascular tissue (Figs. 4–6). Remarkable fibrosis was observed as blue staining with MT (Figs. 7, 8); some macrophages contained lipofuscin, which appeared as yellow-brown pigment granules that reacted with PAS (Figs. 9, 10), and a few macrophages contained hemosiderin that stained with Berlin blue (Fig. 11, inset) and demonstrated mineralization (Figs. 11, 12) and inflammation (Fig. 13) in the central area. In the peripheral area, there were numerous blood-filled arteries. In addition, case 2 had many tubular structures consisting of cuboidal cells arranged in a meshlike arrangement (Fig. 14), and a few different tubules were formed by degenerate or necrotic cells characterized by clusters of eosinophilic amorphous substances lacking nuclei and were lined by thick basement membranes (Fig. 15). The basement membrane of the tubules was identified with PAS (Fig. 16). Detailed findings are presented as Supplemental Materials.
Cases 4–6
The epididymis, deferent duct, and pampiniform plexus were identified in cases 5 and 6. There were many cross sections of seminiferous tubules in the intra-abdominal testes in all cases (Fig. 17). A few germ cells were observed in the lumen of seminiferous tubules with arrested spermatogenesis (Fig. 18; Table 2). In addition, an increase in interstitial endocrine cells and in the remaining testicular sustentacular cells was found (Fig. 18). On the other hand, degenerative findings, such as mineralization, hemosiderin-laden macrophages, lipofuscin-laden macrophages, and inflammation, were not observed.
Histopathologic Findings in the Intra-abdominal Testicular Tissue of Calves With Testicular Degeneration Resembling Testicular Regression Syndrome (Cases 1–3) and Cryptorchid Calves (Cases 4–6).a
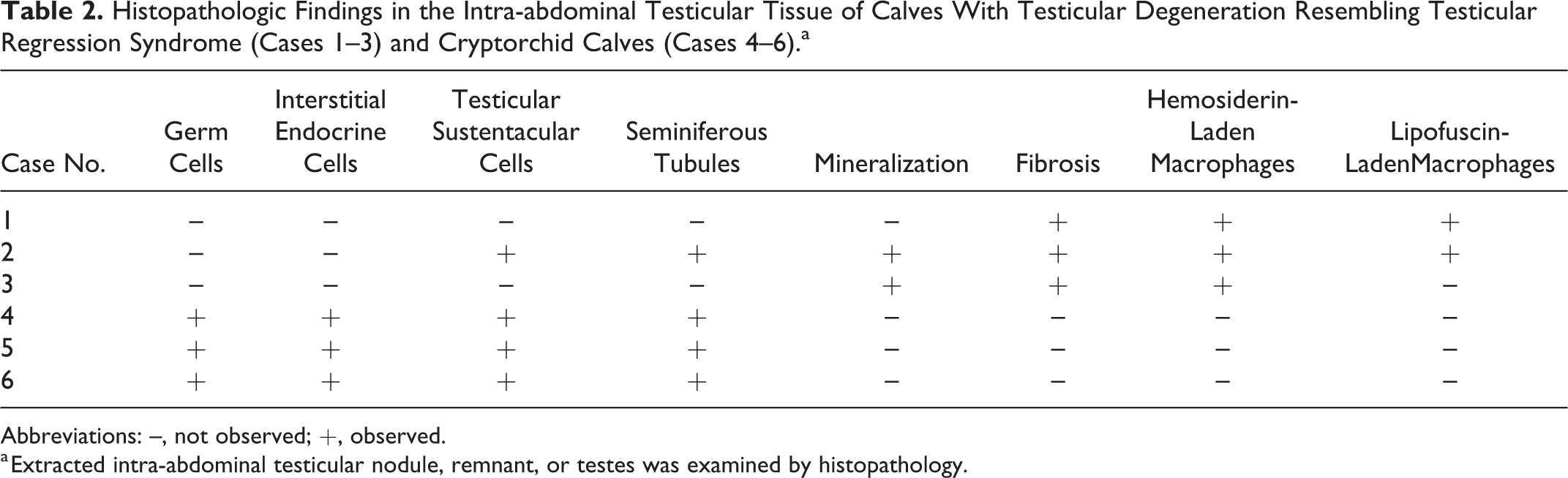
Abbreviations: –, not observed; +, observed.
a Extracted intra-abdominal testicular nodule, remnant, or testes was examined by histopathology.
Cases 7–11
The testes, epididymis, deferent duct, and pampiniform plexus were identified in all cases. No significant findings were observed in those structures (Fig. 19).
Morphometric and Immunohistochemical Findings
The total number of seminiferous tubules as well as their average cross-sectional areas were smaller in case 2 as compared with cases 4–6 and the control cases (Table 1).
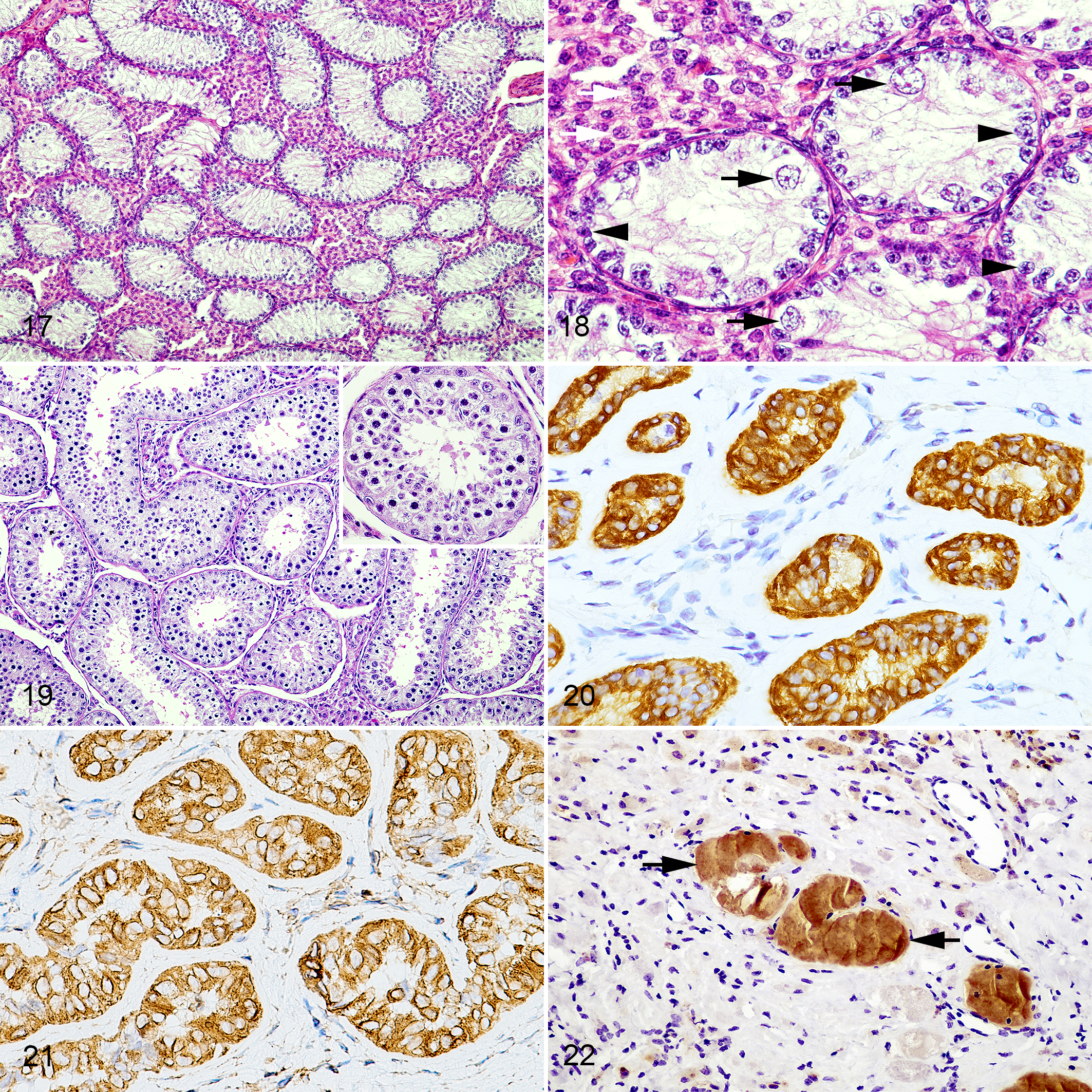
In case 2, the tubular structures were positively immunolabeled for cytokeratin and vimentin but negative for inhibin-alpha and AMH (Figs. 20, 21). The degenerate or necrotizing tubular structures were positive for AMH but negative for cytokeratin, vimentin, and inhibin-alpha (Fig. 22). In cases 1 and 3, tissues that could be identified as part of the testicular component were not immunolabeled for cytokeratin, inhibin-alpha, or AMH.
In cases 4, 6, and 7–11, cytokeratin and vimentin but not inhibin-alpha or AMH were strongly expressed in the rete testis. The testicular sustentacular cells were immunolabeled for vimentin, inhibin-alpha, and AMH, whereas the interstitial endocrine cells were immunolabeled for vimentin. On the other hand, the epididymal duct and deferent duct were immunolabeled for cytokeratin.
In cases 1–3, the hemosiderin-laden macrophages of testicular remnants were labeled for vimentin and Iba1.
Endocrinologic Findings and Statistical Analysis
Plasma testosterone concentrations were not detected in cases 1–3; thus, for analysis purposes, the value was defined as the lower limit of detection (0.05 ng/ml; Fig. 23). In the controls (cases 7–11), the plasma testosterone concentration (1.41 ± 0.79 ng/ml) was higher than in cases 1–3 and cases 4–6 (0.06 ± 0.02 ng/ml; P < .05).
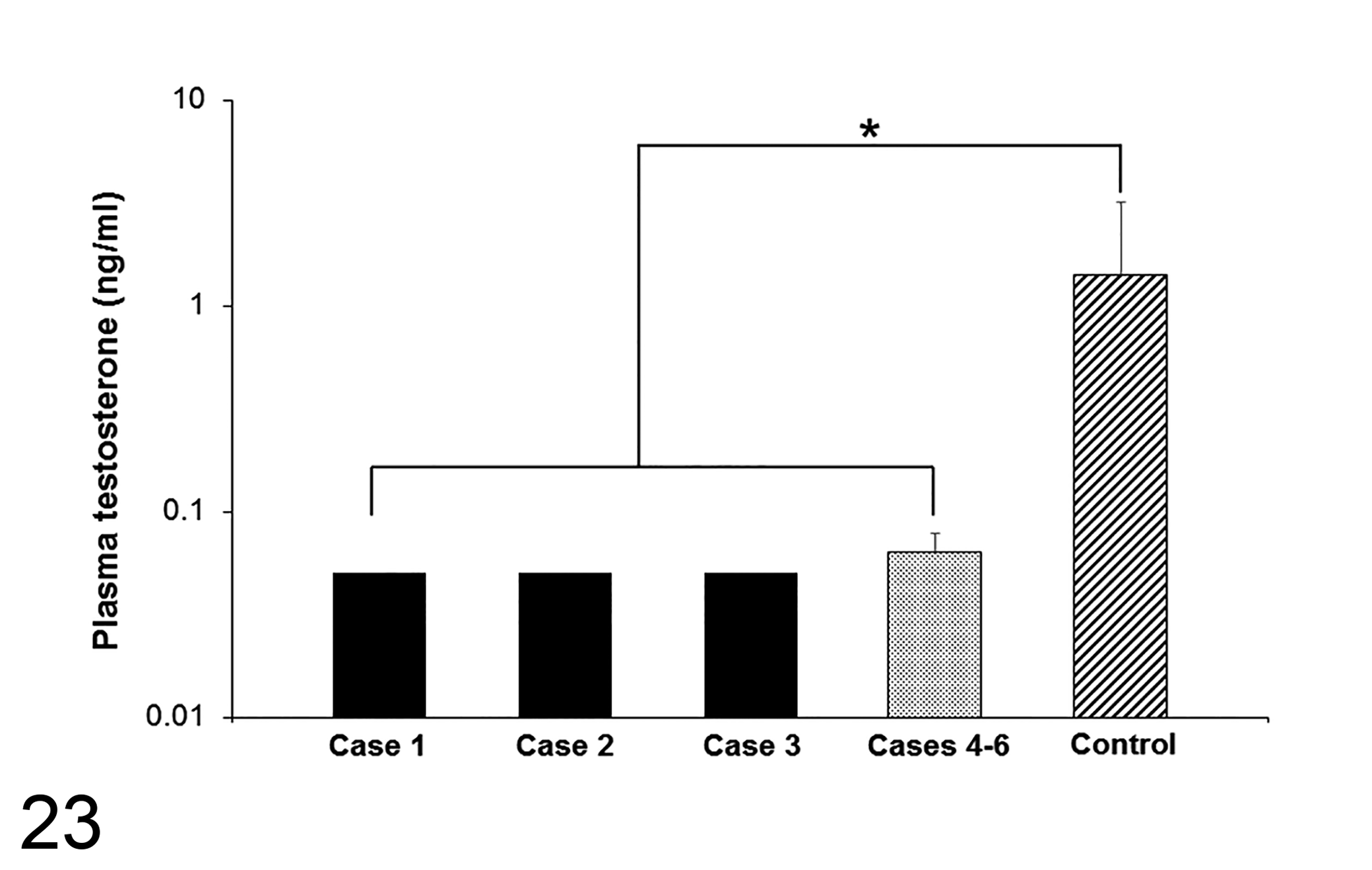
Comparison of plasma testosterone concentrations. Concentrations of control cases (cases 7–11) were significantly higher than in both cases 1–3 and cases 4–6. *P < .05.
Plasma AMH was detected at a concentration of 214 pg/ml in case 2, but it was not detected in cases 1 and 3; therefore, for purposes of analysis, the value was defined as the lower limit of detection (11 pg/ml). The plasma AMH concentration in the control (43 207 ± 26 322 pg/ml) was higher than in cases 1–3 and lower than in cases 4–6 (176 749 ± 29 366 pg/ml; both P < .05; Fig. 24). In addition, the plasma AMH concentration in cases 4–6 tended to be higher than in the control (P = .08).
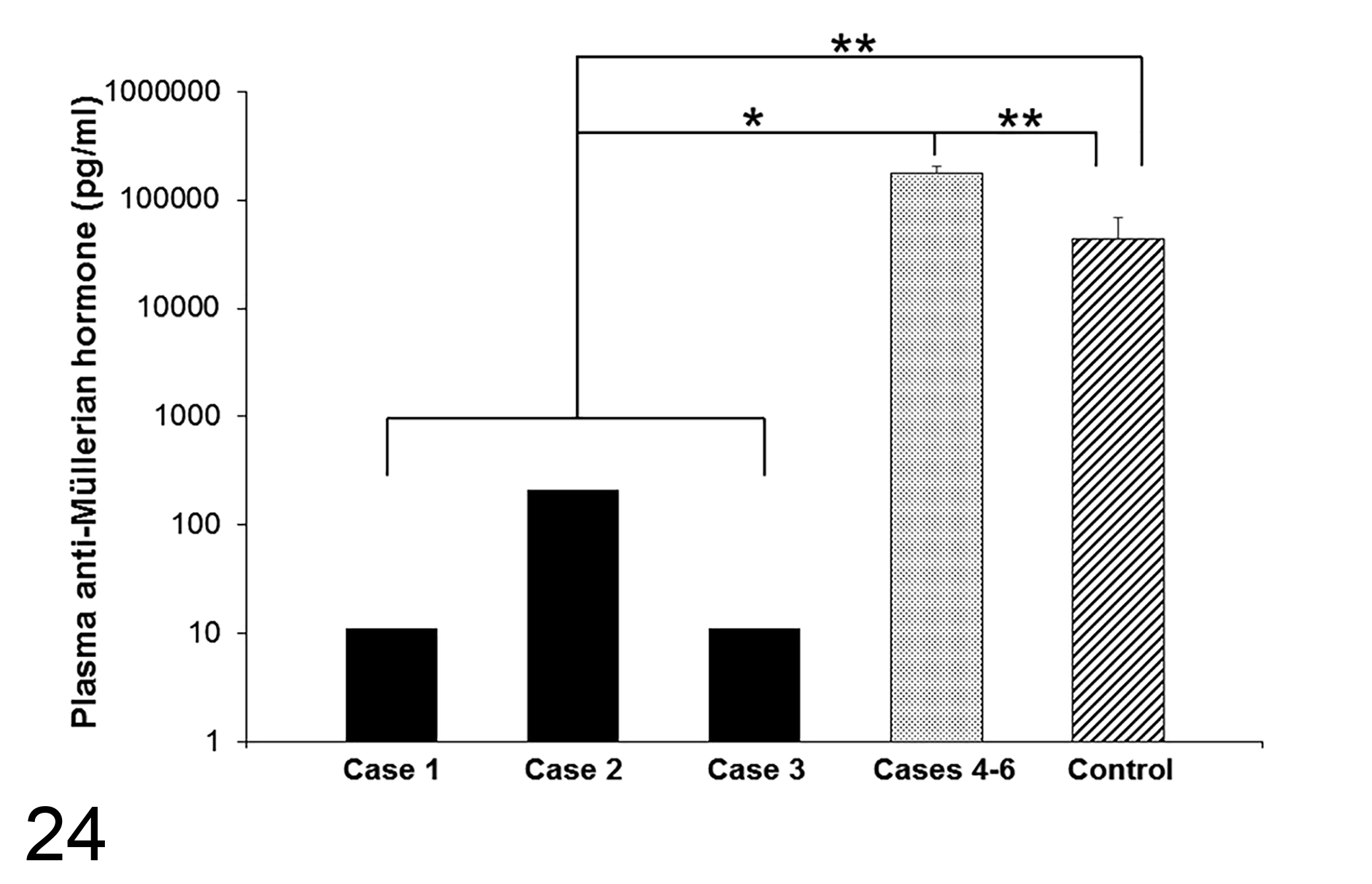
Comparison of plasma anti-Müllerian hormone concentrations. The concentrations of the controls (cases 7–11) were significantly higher than in cases 1–3 and lower than in cases 4–6. Cases 4–6 tended to be higher than controls (P = .08). *P < 0.1; **P < .05.
Discussion
Several congenital anomalies of testis have been reported in animals, such as testicular hypoplasia, cryptorchidism, and monorchidism. 1,9,18,21,23,24,27,31,35 Testicular hypoplasia is a frequent diagnosis on gross examination and has various causes, including cryptorchidism, extra X chromosome, or bovine viral diarrhea virus infection. 3,9,18 The histologic characteristics include absence or decreased number of germ cells, spermatogenic arrest, and undeveloped seminiferous tubules. 9,18 In animals, cryptorchidism is a common cause of testicular hypoplasia, as the retained testicular tissue fails to develop and resembles immature testis. Over time, the tissue degenerates and remains small, in which marked fibrosis, calcified remnants of seminiferous tubules, and lipofuscin-containing macrophages are found. 9 Monorchidism in stallions and dogs showed dense fibrous tissue with no signs of testicular tissue. 20,24,35 In cases 4–6, seminiferous tubules contained sustentacular cells, a few germ cells, and arrested spermatogenesis, as well as increased numbers of interstitial endocrine cells but an absence of degenerative changes. Accordingly, cases 4–6 were diagnosed with testicular hypoplasia due to cryptorchidism. In contrast, the fibrotic testicular nodules of cases 1 and 2 were much smaller than in cases 4–11, and no nodule was identified for case 3. Histologically, cases 1–3 had a lack of or marked decrease in seminiferous tubules, interstitial endocrine cells, and testicular sustentacular cells and had fibrosis, inflammation, hemosiderin-laden macrophages, and lipofuscin-laden macrophages. Because these findings were clearly different from the typical testicular hypoplasia, this diagnosis was unsuitable for these cases.
Testicular regression syndrome is a condition in humans in which a normally developing testis is thought to have once existed in fetal life and subsequently atrophied and disappeared. 2,10,17,25 In cases 1–3, a testicular nodule or remnant was observed in vascularized fibrous nodules with hemosiderin-laden macrophages, mineralization, and fibrosis. These histologic findings are characteristic of fibrotic testicular remnant in testicular regression syndrome. 5,8,25,28,29,34,38
Cases 1–3 had evidence of a chronic cellular response to tissue damage, including hemosiderin-laden macrophages, lipofuscin-laden macrophages, some inflammatory cells, foci of mineralization in vessel walls (particularly in case 3), as well as degenerated tubules (case 2). In humans, hemosiderin deposition and dystrophic mineralization were common in fibrotic testicular remnants. 2,5,6,8,9,25,30,38 Even in the absence of viable testicular tissue, normal deferent duct and epididymis were present in some cases. 25,30 The pathogenesis of testicular regression syndrome is thought to be the result of late antenatal or perinatal vascular thrombosis and torsion. 2,10,25,30 In stallions and puppies, cases of intra-abdominal testicular torsion, admitted for cryptorchidectomy, were also reported. 4,22 In calves, testicular remnants after Burdizzo castration (with interruption of the blood supply) showed decreased testicular sustentacular cells and spermatogonia within shadow tubules, interstitial fibrosis, and the absence of interstitial endocrine cells. 33 In cases 1–3 of the present study, we did not observe apparent testicular torsion. However, the histologic findings supported the hypothesis of a vascular disorder leading to such lesions seen in the testis. Considering the ages of cases 1–3, a developmental problem such as testicular torsion or other vascular disease might have occurred in fetal life or shortly after birth and have atrophied and disappeared.
In general, adult testes of domestic mammals are present in the scrotum to maintain lower temperature than the body temperature. Heat-induced testicular degeneration in animals can occur as a consequence of different factors, including fever, high environmental temperature, orchitis, periorchitis, scrotal dermatitis, edema and hematomas of the scrotum, and severe systemic disease. 19 Cases 1–3 were found to have intra-abdominal testicular structures. Therefore, those cases were affected by the animal’s body temperature in addition to the proposed vascular disorder, and these factors together might result in the observed gross and histopathologic findings. On the other hand, cases 4–6 were mainly affected by heat-induced degeneration due to cryptorchidism without vascular abnormalities.
The diameter of seminiferous tubules in bulls varies from 171 µm in normal testes to 118 µm in bilateral testicular hypoplasia. 16 A mean tubule diameter, which was effective in distinguishing hypoplastic from normal testes in young postpubertal bulls, was considered to be about 150 μm. 16 The seminiferous tubules in cases of complete hypoplasia were reported to be small. 18 The average size of the cross sections of seminiferous tubules in cases 4–6 was consistent with these reports. On the other hand, the average size and the number of the cross sections of seminiferous tubules in case 2 were drastically smaller than in the other cases, resulting in a remarkably small fibrotic testicular remnant.
Rete testis was immunolabeled for cytokeratin and vimentin in cases 4 and 6 (testicular hypoplasia) and control calves 7–11, and coexpression of cytokeratin and vimentin in rete testis was previously reported in humans and dogs. 7,37 In case 2 (testicular regression syndrome), the remaining tubular structure with a meshlike arrangement was immunolabeled for cytokeratin and vimentin, supporting the idea that the tubular meshlike structure was rete testis. The testicular sustentacular cells in cases 4–11 (testicular hypoplasia and controls) were immunopositive for vimentin, inhibin-alpha, and AMH and immunonegative for cytokeratin. In case 2, the degenerated tubules were positive for AMH. In bulls, it was found that testicular sustentacular cells were immunolabeled with vimentin, inhibin-alpha, and AMH, 11,32,36 and AMH was secreted only by immature testicular sustentacular cells in males. 36 In the present study, case 2 had immunolabeling for AMH and detection of AMH in plasma, indicating that the observed degenerated tubules might have been seminiferous tubules containing remnant AMH-producing sustentacular cells.
In this study, plasma testosterone is not detected in cases 1–3, whereas it was detected in the other cases. Testosterone is mainly synthesized and secreted by interstitial endocrine cells in the testes. After castration, plasma testosterone was not detected in cattle. 15 In cases 1–3, interstitial endocrine cells were not detected in the extracted intra-abdominal tissues, consistent with the lack of detectable plasma testosterone.
The plasma AMH concentration was lower in case 2 than in cases 4–6, although intra-abdominal testis was observed in all cases. Although cases 4–6 and controls were close in age, the plasma AMH levels of cases 4–6 were higher than those of controls. One reason might be that the volume of testicular tissue was smaller and the number of testicular sustentacular cells was lower in case 2 than in cases 4–6. In male calves, immature testicular sustentacular cells proliferate until 20 weeks of age, after which these cells begin to mature. 26 Mature testicular sustentacular cells lose the synthesis and secretion of AMH. 29 In normal male calves, plasma AMH levels decrease after 2 months of age. 15 Based on this knowledge, there may have been a failure of maturation of the testicular sustentacular cells in the intra-abdominal testis in cases 4–6.
In conclusion, the present study describes the degenerative changes in 3 cryptorchid testes in Japanese Black calves that differed from the usual cryptorchid cases. These testicular remnants were markedly small and fibrotic, and histologic findings included mineralization, fibrosis, hemosiderin-laden macrophages, lipofuscin-laden macrophages, decreased number and diameter of seminiferous tubules, lack of interstitial endocrine cells, and few germ cells or testicular sustentacular cells. The microscopic findings are similar to testicular regression syndrome in humans, although there is a lack of clinical evidence in this study to support the reduction in testicular size over time. Plasma testosterone was not detected in these 3 cases, and plasma AMH was detected in only 1 case. Considering the gross and microscopic findings and endocrinologic profiles, the pathogenesis of degenerative changes in cryptorchid testes may be caused by body heat and a vascular disorder such as torsion.
Supplemental material
Supplemental Material, Combined_supplemental_materials-Fuke_et_al - Severe Degenerative Changes in Cryptorchid Testes in Japanese Black Cattle
Supplemental Material, Combined_supplemental_materials-Fuke_et_al for Severe Degenerative Changes in Cryptorchid Testes in Japanese Black Cattle by Naoyuki Fuke, Go Kitahara, Soma Ito, Nguyen Van Diep, Angeline Ping Ping Teh, Uda Zahli Izzati, Ohnmar Myint, Takuya Hirai, Shidow Torisu, Yasuyuki Kaneko, Hiroyuki Sato, Yuichi Hidaka, Takeshi Osawa and Ryoji Yamaguchi in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
