Abstract
Lymphoma is the most common intestinal neoplasm in horses, but its clinical-pathological features are poorly characterized. Primary intestinal lymphoma was diagnosed in 20 horses on biopsy samples and further confirmed by postmortem examination in 16 cases. Lymphoma was found in the small intestine in 12 of 20 (60%), in the colon in 5 of 20 (25%), and in both small and large intestines in 3 of 20 (15%) cases. Gross findings included thickening of the intestinal wall (45%), mural nodules or masses (30%), and both thickening and nodules (10%). Cases were classified according to the human World Health Organization classification as enteropathy-associated T-cell lymphoma (EATL) type 1 (40%), EATL type 2 (45%), and T-cell-rich large B-cell lymphoma (TCRLBCL) (15%). With respect to histologic grade, 70% of cases were grade 1 and 30% were grade 2. Of EATLs, the infiltrate was mucosal only (12%), mucosal and submucosal (53%), or transmural (35%). EATL1 was submucosal to transmural (2/8 and 6/8), EATL2 was mucosal to submucosal (3/9 and 6/9), and TCRLBCL was always transmural. Epitheliotropism was present in most EATLs and characterized by single-cell infiltrates within the epithelium in EATL1 and intraepithelial clusters or plaques in EATL2. Median survival was 25 days for EATL1, 90 days for EATL2, and 187.5 days for TCRLBCL; differences were not statistically significant. Of the EATLs, grade 1 had a median survival of 60 days and grade 2 had a median survival of 25 days; differences were not statistically significant.
Lymphoma is the most common intestinal neoplasm in horses, and intestinal lymphoma is the third most frequent type following multicentric and cutaneous. 3,17 The small intestine is the most commonly affected segment, but multiple segments of both large and small intestines can be simultaneously infiltrated. 13,17
The pathological features of equine intestinal lymphoma are poorly characterized, and descriptions are limited to a small number of case series 3,13,17 or single case reports. 6,7,12,14 Nineteen horses with alimentary lymphoma were described by Taylor et al 17 in a retrospective review of 34 intestinal tumors, and T-cell lymphoma predominated, although only a small proportion of cases were confirmed with immunohistochemistry. 17 Twenty-four additional cases were classified by Durham et al 3 in a large survey of equine lymphomas, and T-cell-rich large B-cell lymphoma (TCRLBCL) was the most common histologic subtype both systemically and in the intestine. An older study of 31 cases of lymphoma in horses found only 1 case that was infiltrating the intestine, which displayed a T-cell phenotype. 4
Enteropathy-associated T-cell lymphoma (EATL) is a type of intestinal lymphoma described in the human World Health Organization (WHO) lymphoma classification. 15 EATLs are classified into 2 distinct subtypes, EATL types 1 and type 2, which show distinct clinical and morphological features. 15 As it became apparent that the 2 subtypes were distinct clinical entities, the human classification system was updated and EATL1 and EATL2 were changed to EATL and monomorphic epitheliotropic intestinal lymphoma (MEITL), respectively. 16 These 2 entities are also recognized in dogs and cats and share similar histopathological features with their human counterparts. 5,8 In humans, EATL is strongly associated with inflammatory bowel disease, 15 but this connection has not been established in other animal species.
The goal of this study was to describe the morphological and immunohistochemical features of equine primary intestinal lymphoma in accordance with the human WHO classification, 15 as well as to report the clinical features and outcomes of the cases.
Materials and Methods
Case Selection
Tissues from 20 horses (Nos. 1–20) with a diagnosis of primary intestinal lymphoma (2006–2014) were retrieved from the archive of the Anatomic Pathology section of the Faculty of Veterinary and Agricultural Sciences of the University of Melbourne. Formalin-fixed, paraffin-embedded (FFPE) tissues from intestinal biopsies (
Histopathology and WHO Classification
All samples were fixed in formalin, routinely processed, and embedded in paraffin. Sections were stained with hematoxylin and eosin (HE). All cases were reexamined by 2 pathologists (B.B., A.W.S.), and the diagnosis of intestinal lymphoma was confirmed based on the presence of dense populations of neoplastic lymphocytes with abnormal cell size and/or invasion beyond the mucosa. To reach a conclusive diagnosis and for the histologic classification, both biopsies and postmortem samples, when available, were evaluated.
For each case, the following histologic parameters were assessed: (1) depth of the lymphocytic infiltrate (mucosal, submucosal, or transmural); (2) lymphocyte size as small (nuclear diameter <2 red blood cells [RBCs]) or large (nuclear diameter >2.0 RBCs), defined based on the majority of the neoplastic cell population; (3) presence of epitheliotropism, including single cells, nests (clusters of >5 lymphocytes infiltrating the epithelium), or plaques (>5 adjacent epithelial cells overrun by clustered lymphocytes); 5 (4) presence of eosinophils (0 = none, 1 = <2%, 2 = 2%–5%, 3 = >5%) in a representative field, as previously described; 1 and (5) mean mitoses calculated by averaging the mitotic figures counted in 3 high-power fields (400×, with a field of view area of 0.237mm 3 ) in regions with the best fixation and least amount of artifact. Cases were finally classified according to the human WHO classification scheme with consideration of immunophenotype. 15 To allow comparison of intestinal lymphoma characteristics across species, the terminology of EATL (enteropathy-associated T-cell lymphoma) was applied in accordance with the WHO classification in domestic animals 19,20 and was further classified into EATL1 and EATL2 according to the human classification scheme, as previously applied to other domestic animal species. 15 Histological grade was assigned as follows: grade 1 (low) with 0 to 5 mitoses/400× field, grade 2 (intermediate) with 6 to 10 mitoses/400× field, and grade 3 (high) with >10 mitoses/400× field. 20
Immunohistochemistry
Immunohistochemistry (IHC) was performed to determine immunophenotype and proliferation index. Tissues were immunohistochemically labeled using a rabbit polyclonal antibody to human CD3 (Dako Flex, Sydney, Australia) and a mouse monoclonal antibody to human PAX5 (clone 24/Pax-5; BD Biosciences, Sydney, Australia)) following heat-induced epitope retrieval submersion in citrate buffer (pH 6.0). Immunolabeling for Ki-67 was performed with a mouse monoclonal antibody (MIB-1 clone; Dakocytomation, Carpinteria, CA). A streptavidin-biotin-peroxidase complex detection method was used. The Ki-67 index was determined by counting the number of Ki-67–positive cells per 1000 neoplastic cells, assisted by a digital cell counter (ImageJ, Bethesda, MD).
Positive immunohistochemical controls for all antibodies consisted of a normal equine lymph node (CD3 and PAX5) and a normal equine intestine (Ki-67) incubated with primary antibodies as described. For negative controls, primary antibodies were replaced with nonimmune sera.
Follow-up
Survival times and other follow-up information were obtained through the medical records database of the University of Melbourne Veterinary Hospital or via telephone interviews with the referring veterinarians or owners.
The end point considered for each horse with lymphoma was time to death, calculated as the time elapsed from the date of diagnosis to the date of death, or to the date of last clinical information for horses that were alive at the closing date of the study. 21 The patient status was recorded as alive, dead/euthanized because of lymphoma-unrelated causes (NLR), or dead because of lymphoma-related causes (LR). Cases in which the patients died or were euthanized for unrelated causes (including euthanasia due to diagnosis/suspicion of neoplasia) were censored.
Statistical Analysis
Survival estimates are presented as medians. Survival curves were estimated by the Kaplan-Meier method with differences in WHO subtypes and histologic grades assessed by the log-rank test. Ki-67 index analysis between groups was conducted by 1-way analysis of variance (ANOVA). Analysis of the correlation between Ki-67 and survival was conducted by the Spearman test. Statistical analyses were performed using GraphPad 8.0 for Windows (GraphPad Software, La Jolla, CA). Significance was set at
Results
Cases and Clinical Data
Signalment and clinical data are summarized in Supplemental Table S1.
The age of the horses ranged from 1 to 27 years (mean ± standard deviation 13 ± 7.0 years); the age was not known for 1 case. Thirteen horses were geldings and 7 were female; there were 8 Thoroughbreds, 3 Quarter Horses, 3 Arabians, 2 Warmbloods, and 4 ponies. Breeds and sex reflected the distribution of the hospital database population admitted for intestinal disease.
Major clinical findings at presentation included colic (14/20, 50%), weight loss (7/20, 30%), diarrhea (5/20, 25%), anorexia (4/20, 20%), and fever (4/20, 10%).
Hypoalbuminemia was found in 10% (3/20) of cases. At initial clinical evaluation, disease was attributed to the small intestine (10/20), large intestine (9/20), or could not be localized (1/20). Of the 10 cases that underwent exploratory laparotomy for colic, 7 had masses or thickening of the intestinal wall, and 1 had multiple small intestinal diverticulae that resulted in partial obstruction. One horse had a large colon displacement, and 1 had a cecocecal intussusception.
Of the 20 biopsies, 17 were collected during exploratory laparotomy and 2 were surgical resections, while in 1 case, a rectal mucosal biopsy was taken. Biopsies were collected from the large intestine in 5 cases, from the small intestine in 12 cases, and from both the small and large intestine in 3 cases.
Lymphoma was found in the small intestine in 12 of 20 (60%) cases, in the colon in 5 of 20 (25%) cases, and in both large and small intestines in 3 of 20 (15%) cases. In 16 of 20 horses, a full necropsy was performed. Gross findings, including those observed during surgery or exploratory laparotomy in the 4 of 20 cases in which a necropsy was not performed, included intestinal wall thickening (9/20, 45%), nodular masses in the intestinal wall (6/20, 30%), mucosal reddening (4/20, 20%) (Fig. 5), mesenteric lymph node enlargement (3/20, 15%), mucosal erosion and ulceration (3/20, 15%), and mural diverticulae (1/20, 5%). According to histologic subtypes (as classified below), EATL had thickening of the intestinal wall (9/17) and/or mural masses (6/17), while all TCRLBCL presented as mural masses (3/3).
Histopathology and Immunohistochemistry
Microscopic features, classification, and immunophenotypes are summarized in Table 1. Seventeen cases were T-cell lymphomas and 3 cases were B-cell lymphomas, based on CD3 and PAX5 expression in the predominant malignant cell population. Cases were classified according to human WHO classification system as follows: 17 of 20 (75%) EATLs and 3 of 20 (15%) TCRLBCLs.
Microscopic Features of 20 Equine Intestinal Lymphomas.
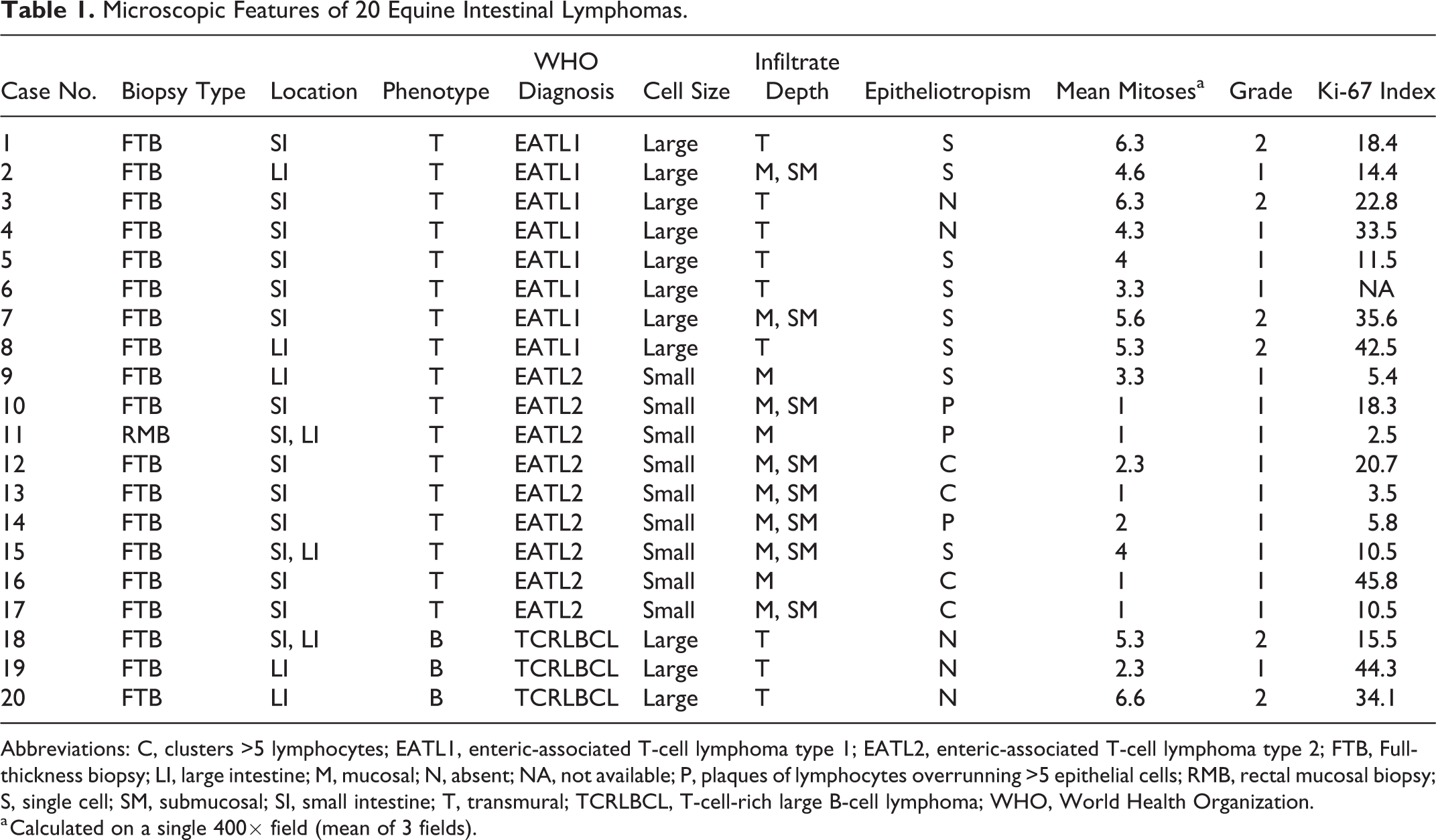
Abbreviations: C, clusters >5 lymphocytes; EATL1, enteric-associated T-cell lymphoma type 1; EATL2, enteric-associated T-cell lymphoma type 2; FTB, Full-thickness biopsy; LI, large intestine; M, mucosal; N, absent; NA, not available; P, plaques of lymphocytes overrunning >5 epithelial cells; RMB, rectal mucosal biopsy; S, single cell; SM, submucosal; SI, small intestine; T, transmural; TCRLBCL, T-cell-rich large B-cell lymphoma; WHO, World Health Organization.
a Calculated on a single 400× field (mean of 3 fields).
EATL cases were characterized by infiltration of small (9/17, 53%) or large (8/17, 47%) CD3-positive lymphocytes. Infiltration of the intestinal wall was mucosal only (2/17, 11%), mucosal and submucosal (9/17, 52%), or transmural (6/17, 35%). Epitheliotropism was present in 15 of 17 (88%) EATLs.
Based on the features described above, EATL cases were further categorized as EATL1 (8/17, 47%) or EATL2 (9/17, 53%). Of the 8 EATL1 cases, 5 were small intestinal and 3 were large intestinal. Of the 9 EATL2 cases, 6 were small intestinal, 1 was large intestinal, and 2 affected small and large intestine. Cases classified as EATL1 were characterized by infiltration of large lymphocytes, extending transmurally in the majority of cases (6/8) or reaching the submucosa (2/8) (Figs. 1–4). EATL2 cases were characterized by infiltration of small lymphocytes, either confined to the mucosa (3/9) or extending into both the mucosa and submucosa (6/9) (Figs. 5–9).
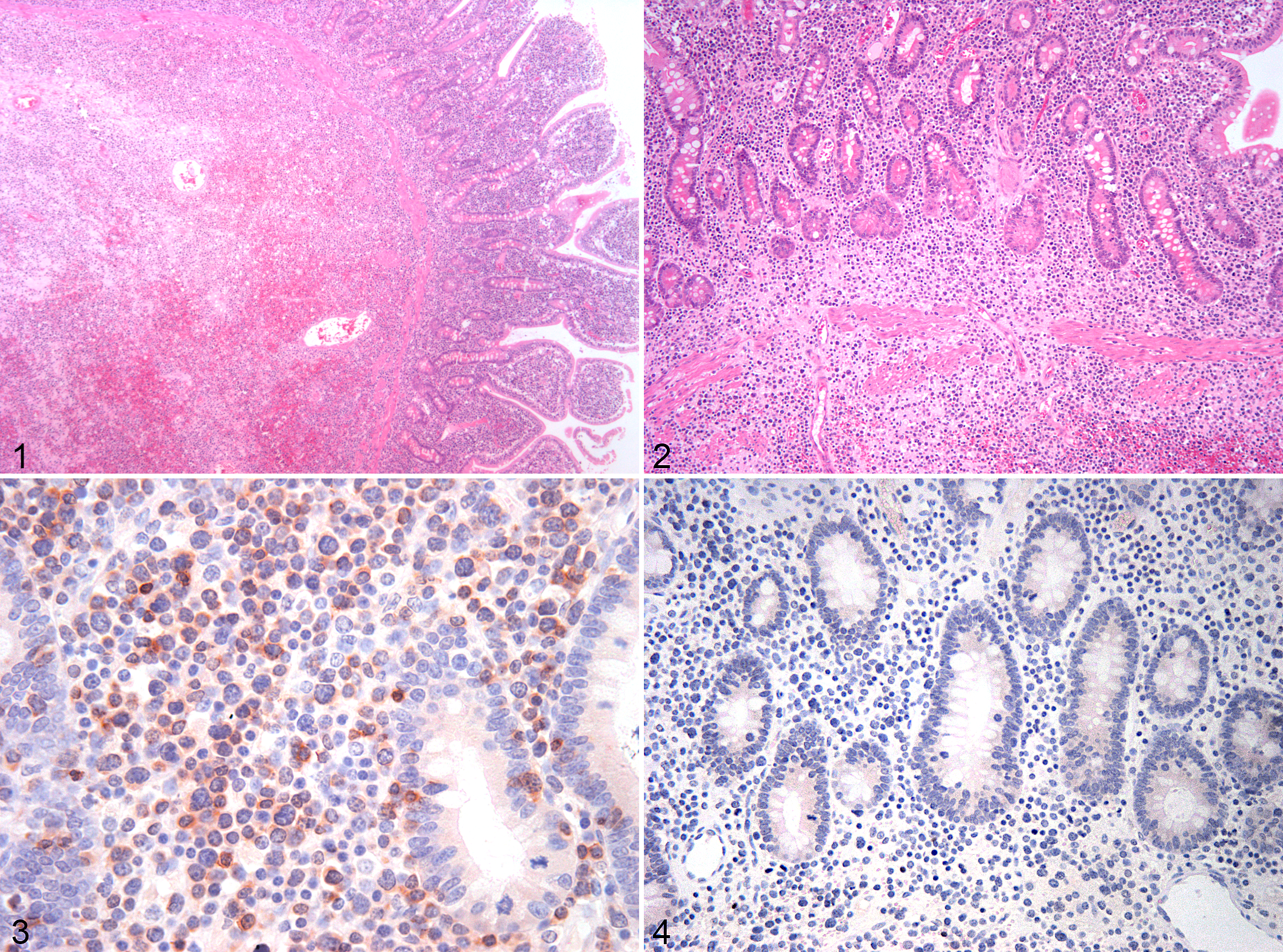
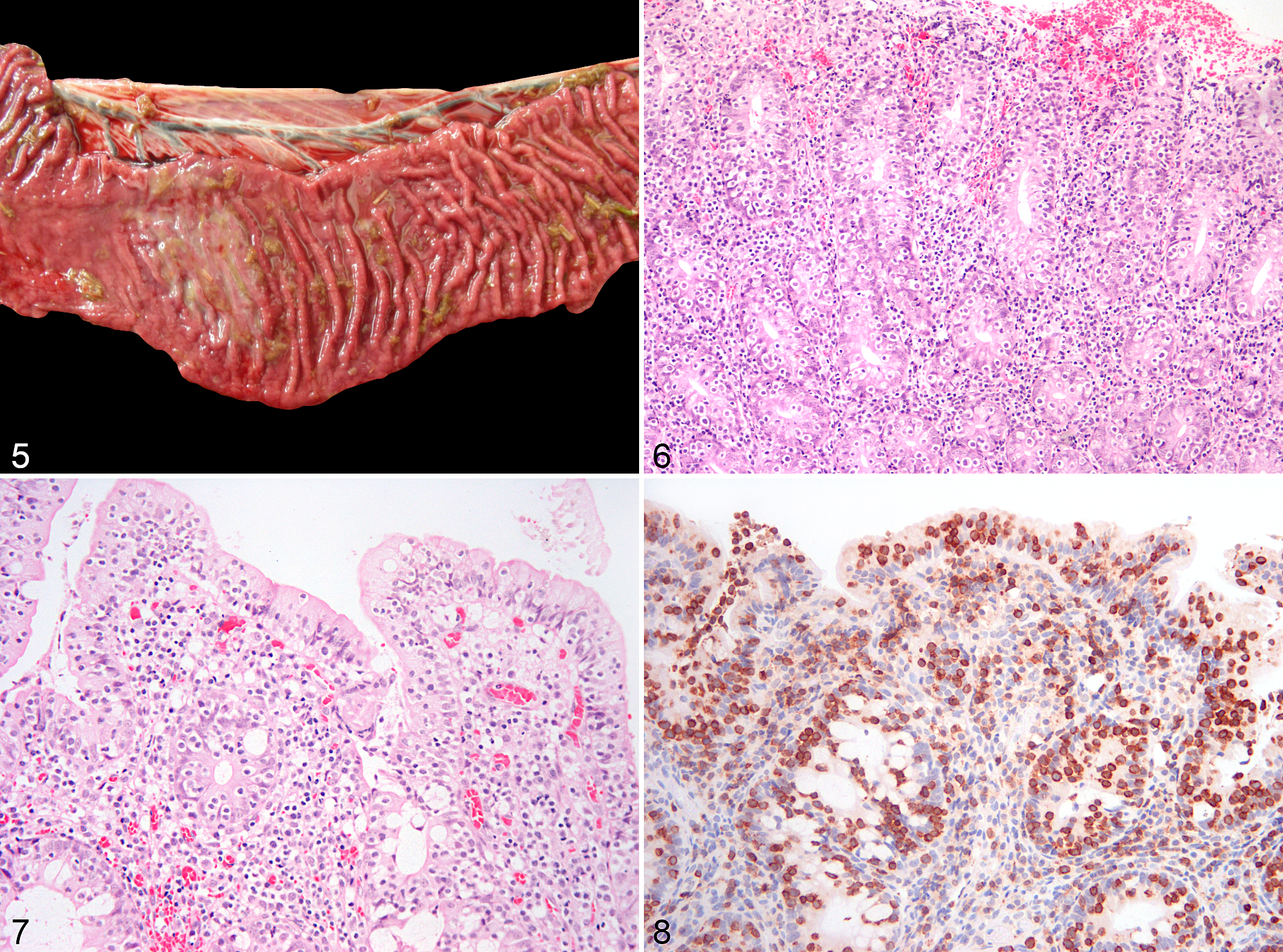
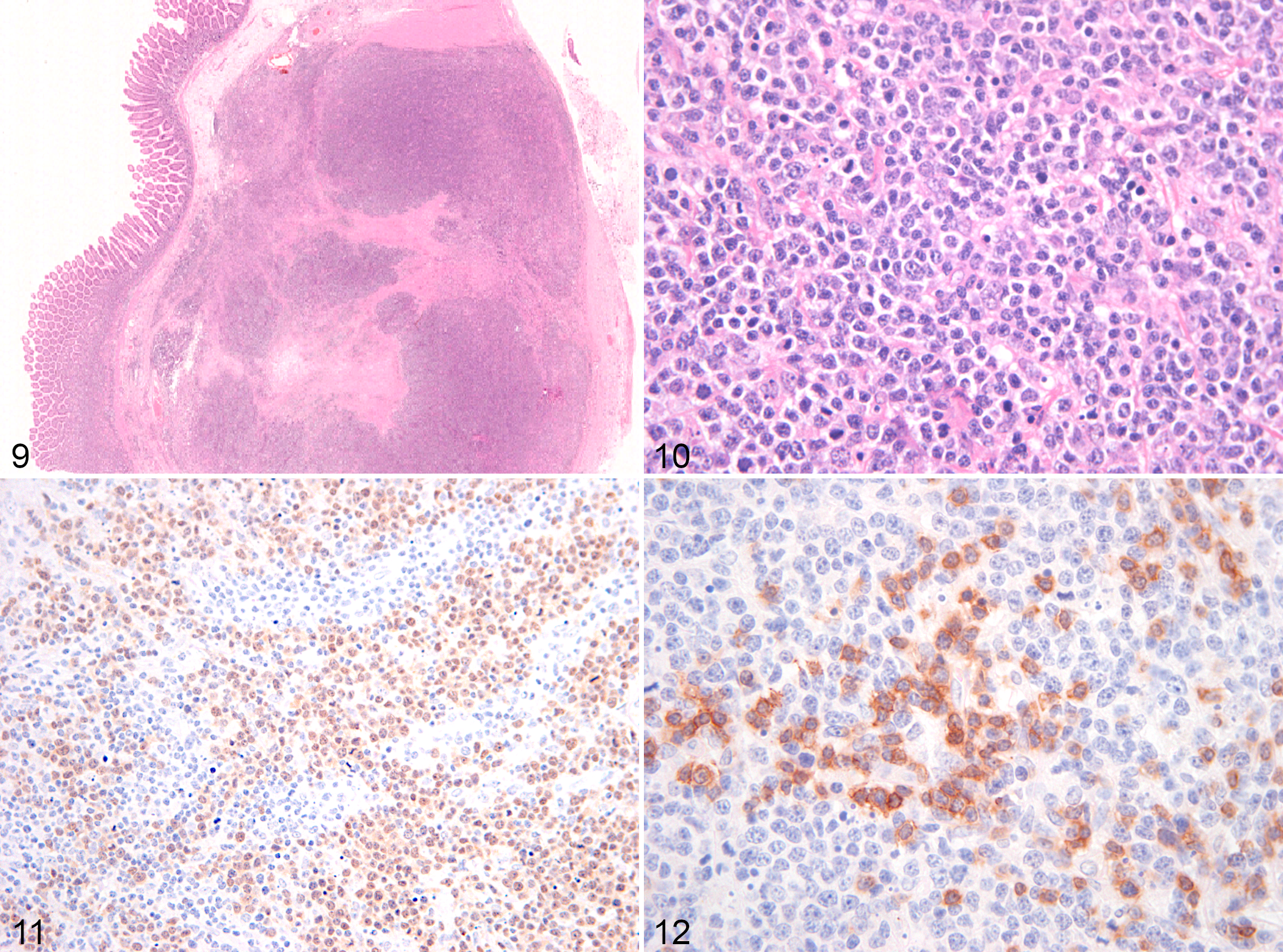
Of the EATLs, epitheliotropism (which was best visualized with CD3 labeling) was observed equally in the surface epithelium and in the crypts (Figs. 6–8). Single-cell infiltrates were observed in 8 of 17 cases, clusters in 4 of 17, and plaques in 3 of 17 cases. All EATL1 cases had only single-cell infiltrates, while EATL2 cases displayed all distribution patterns, with a predominance of clusters or plaques.
TCRLBCLs were characterized by transmural infiltration of a mixed population of medium to large atypical lymphocytes that were PAX5 positive, admixed with a population of small CD3-positive lymphocytes. CD3-positive lymphocytes represented 30% to 50% of the total lymphocyte population in the infiltrated areas. The PAX5-positive lymphocytic population had scant amphophilic to eosinophilic cytoplasm, large vesicular nuclei that were rarely binucleated, and a central prominent nucleolus (Figs. 9–13). Epitheliotropism was not present. Severe necrosis was also present in all cases. Of the 3 TCRLBCL cases, 2 were large intestinal, and 1 affected small and large intestine. The neoplastic infiltrate within the wall was invariably transmural.
A concurrent eosinophilic infiltrate was observed in 7 cases (35%), of which 6 were EATL2 and 1 was EATL1. Scattered plasma cells were visible in 8 cases (40%): 4 EATL1 and 4 EATL2.
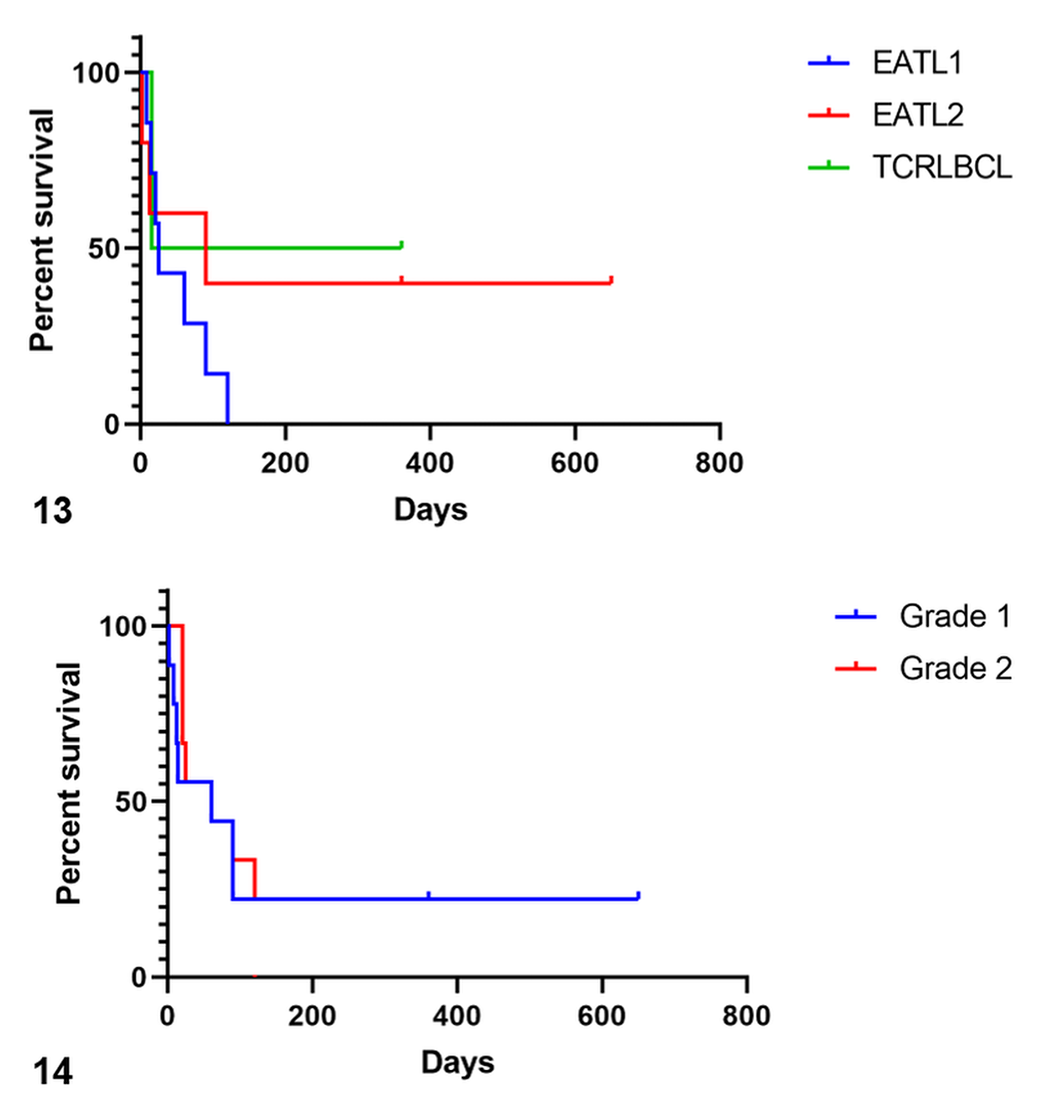
The average number of mitoses per 400× field was 4 for EATL1, 1.5 for EATL2, and 4.7 for TCRLBCL. Based on the current grading system for lymphoma in dogs, 20 14 cases were grade 1 and 6 cases were grade 2. Grade distribution according to WHO subtypes is presented in Table 2.
Grade Distribution of 20 Equine Intestinal Lymphomas According to World Health Organization Classification Subtypes.
Abbreviations: EATL1, enteric-associated T-cell lymphoma type 1; EATL2, enteric-associated T-cell lymphoma type 2; TCRLBCL, T-cell-rich large B-cell lymphoma.
Ki-67 index was calculated in 19 of 20 cases. No reactivity was observed in 1 case possibly due to prolonged formalin fixation. The Ki-67 index was 20.8% ± 14.3% positive nuclei (mean ± standard deviation range: 2.5% to 45.8%). The percentage of cells labeled for Ki-67 (mean ± standard deviation) was 25.5% ± 11.7% in EATL1, 13.6% ± 13.6% in EATL2, and 31.3% ± 14.6% in TCRLBCL. The Ki-67 indices were not significantly different between the 3 WHO groups (
Mesenteric lymph nodes were available for examination in 15 cases at the time of biopsy, and infiltration was identified for 1 TCRLBCL (horse No. 18). At necropsy, lymph node effacement was observed in 5 of 16 cases, including 2 EATL1s, 1 EATL2, and 2 TCRLBCLs.
Treatment and Outcome
Follow-up information was available in 19 of 20 cases (Suppl. Table S1). Chemotherapy was not used in any of the cases. Six cases were managed with corticosteroids and 5 by surgical resection of grossly affected intestine where a discrete tumor was present and accessible at laparotomy. Other treatments were supportive and are not detailed here.
Of the 20 cases, 14 died or were euthanized for lymphoma-related causes due to progression of the disease and/or worsening of the clinical signs. Three horses were alive at the end of the study with a survival time of 360 and 650 days. Three cases were euthanized at the time of diagnosis for reasons not related to the patient’s clinical condition (1 had a cervical vertebral fracture and 1 died of cardiovascular collapse).
Median overall survival was 60 days (range, 2 to 650 days). Median survival times in relation to WHO subtypes were 25 days for EATL1, 90 days for EATL2, and 187.5 days for TCRLBCL (Fig. 14); these differences were not statistically significant (
Discussion
In this study, intestinal lymphoma occurred predominantly in middle-aged to older horses except 1 case in a 1-year-old pony. Intestinal lymphomas in horses are more commonly reported in the small intestine but have been also described in the large intestine. In our case series, a small number of cases were found in the colon, with or without small intestinal involvement. The majority were of T-cell origin as previously found, 17 However, this contrasts with another report with an equal proportion of B- and T-cell neoplasms in the gastrointestinal tract because 30% were diagnosed as TCRLBCL. 3 The discrepancy may reflect the selection criteria: since TCRLBCL tends to progress to a multicentric disease, our selection of cases that were restricted to the intestine may have excluded most TCRLBCLs. Likewise, in the previous study, samples mostly consisted of the tissue of interest, which may have resulted in a diagnosis of intestinal TCRLBCL instead of multicentric disease. 3
EATLs are well characterized in humans and are also recognized in dogs, 8 cats, 5,11 and horses. 3 EATLs can be further classified into 2 distinct entities, type 1 (EATL1) and type 2 (EATL2), both originating from epithelial-homing lymphocytes. 15 In humans, EATL1 is characterized by infiltration of the small intestine with pleomorphic medium to large lymphocytes often associated with necrosis and inflammation. This entity was renamed as EATL in the 2017 WHO classification. 5 In dogs and cats, EATL1 is often associated with transmural infiltration, and in cats, this entity is often composed of large granular lymphocytes (LGLs). 5,11 By contrast, EATL2 is characterized by infiltration with small lymphocytes and marked epitheliotropism. In both dogs and cats, EATL2 is largely restricted to the mucosa and is characterized by distinct patterns of epitheliotropsim. 5,11 EATL2 has been recently reclassified in humans as MEITL. 16 In humans, in addition to the histologic and immunohistochemical features, the main difference between the 2 conditions is the association of EATL, and not MEITL, with celiac disease. Both EATL1 and EATL2 were identified in our case series and occurred mainly in the small intestine.
In addition to cell size and pattern of infiltration of the intestinal wall, epitheliotropism is an important feature in the diagnosis of EATL. In particular, nests of intraepithelial lymphocytes (IELs) in the epithelium are considered an essential feature in the diagnosis of EATL2 in dogs and cats. 2,5,11 IELs were observed in 88% of equine EATLs and were equally present in the villous epithelium and crypts. CD3 immunolabeling aided in the identification of epitheliotropism as previously emphasized. 11 Although IELs were visible in both EATL subtypes, the presence of clusters and plaques was only seen in EATL2 as expected. Intraepithelial lymphocytes were also observed in the majority of EATL1 but exclusively as scattered, single-cell infiltrates and never as aggregates. In the absence of epitheliotropism, the extension of the infiltrate beyond the mucosa was essential to reach a conclusive diagnosis of lymphoma. Although the number and distribution of IELs have not been defined in normal equine intestine, the presence of increased numbers of IELs was more readily recognizable in the crypt epithelium than in the villous/surface epithelium in our cases, as reported for feline lymphomas. 11
Horses with EATL had a median survival time of 90 days. According to the histologic subtypes, horses with EATL1 and EATL2 had median survival times of 25 and 90 days, respectively, but the difference was not statistically significant. A major limitation of this study is the small number of cases examined; therefore, a larger case series is needed to predict the outcome in these cases.
TCRLBCL is reported as the most frequent form of lymphoma in horses, frequently presenting as multicentric and cutaneous forms. 3,4,10,17 In our case series, only 3 (15%) of the cases were TCRLBCL. The histologic and immunohistochemical features of TCRLBCL parallel those of previous published cases. 3,4,17
Although systemic or nodal dissemination is expected in most equine lymphomas, 17 we only found evidence of mesenteric lymph node infiltration at the time of diagnosis in 1 TCRLBCL. Lymph node infiltration was reassessed at necropsy and was found in all 3 TCRLBCLs and in 3 of 17 EATLs. This may suggest that EATL has a slower tendency to metastasize and evolve systemically compared to TCRLBCL, although the low case numbers and initial exclusion of cases with systemic involvement may have biased this estimate. TCRLBCL is reported to have a slow, indolent course in dogs and cats. 18,19 A similar behavior has been reported in cutaneous forms in horses, 10 while in humans, it is considered an aggressive lymphoma. Of the 3 cases of TCRLBCL in our series, 1 was still alive at the end of the study after surgical resection of the tumor, and the other 2 were euthanized shortly after the diagnosis.
Gross findings were observed at necropsy and/or exploratory laparotomy in the majority of cases and included intestinal wall thickening, single or multiple mural masses, mucosal erosion or ulceration, and mesenteric lymph node enlargement. These findings partially parallel previous reports of alimentary lymphoma in horses. 17 Multifocal to diffuse mural thickening was seen in the majority of cases at necropsy, but it was a less commonly reported surgical finding, where masses were more commonly visualized. This may be influenced by ease of detecting masses or by presence of mural edema during surgical procedures. In 1 horse, outpouchings of the intestinal wall (pseudodiverticula) were observed during surgery. This has been reported previously in 3 cases of T-cell lymphoma and is likely to be the result of weakening of the muscularis mucosae due to the neoplastic infiltration. 7 From our findings, it appears that EATLs presented as either masses or intestinal wall thickening, while all TCRLBCLs presented as discrete masses within the intestinal wall.
Clinical signs associated with intestinal lymphoma in horses are reported as chronic diarrhea, weight loss, anorexia, and protein-losing enteropathy. 9,17 Although similar signs were seen in our series, nearly half of the cases presented with clinical signs consistent with intestinal obstruction. We speculate that in the early stages of disease, neoplastic infiltration of the intestinal wall might cause dysmotility and colic. Routine biopsy of the intestine in surgical colics might identify lymphoma at an earlier stage in selected cases.
Conclusion
The current study confirms that lymphoma in horses has histologic features consistent with EATL1 and EATL2 similar to those seen in dogs, cats, and humans. Primary intestinal forms had a predominant T-cell origin, with EATL being the most common diagnosis in contrast to previous studies in which TCRLBCL was the most frequent.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Bacci_et_al - Equine Intestinal Lymphoma: Clinical-Pathological Features, Immunophenotype, and Survival
Supplemental Material, Combined_supplemental_materials-Bacci_et_al for Equine Intestinal Lymphoma: Clinical-Pathological Features, Immunophenotype, and Survival by Barbara Bacci, Andrew William Stent and Elizabeth Ann Walmsley in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
