Abstract
Prolonged exposure to water, known as immersion foot syndrome in humans, is a phenomenon first described in soldiers during World War I and characterized by dermal ischemic necrosis. In this report, we describe the pathologic findings of a condition resembling immersion foot syndrome in 5 horses and 1 donkey with prolonged floodwater exposure during Hurricane Harvey. At necropsy, all animals had dermal defects ventral to a sharply demarcated “water line” along the lateral trunk. In 5 animals, histologic examination revealed moderate to severe perivascular dermatitis with vasculitis and coagulative necrosis consistent with ischemia. The severity of the lesions progressed from ventral trunk to distal limbs and became more pronounced in the chronic cases. The pathophysiology of immersion foot syndrome is multifactorial and results from changes in the dermal microvasculature leading to thrombosis and ischemia. Prompt recognition of this disease may lead to appropriate patient management and decreased morbidity.
Hurricane Harvey made landfall as a category 4 hurricane on the Texas coast on August 25, 2017. In the following days, the storm stalled over southeast Texas, leading to the largest rainfall of any recorded US hurricane. 4 Rainfall estimates were as high as 70 inches across southeast Texas. 4 The economic cost exceeded $125 billion, while the livestock loss was an estimated $93 million. 6 Many animals, particularly horses and livestock, were trapped in floodwaters and developed severe skin problems in the lower extremities.
Damage to the skin associated with prolonged exposure to water is a phenomenon rarely reported in humans. Most reports of prolonged water exposure stem from the military, and most cases are restricted to the lower extremities. 1,13,15 The term immersion foot syndrome has been used to describe a variety of injuries from prolonged immersion or moisture. 1 Four immersion foot syndromes have been described in military personnel, depending on the water temperature: trench foot, immersion foot, tropical immersion foot, and warm water immersion foot. 1 Trench foot was first described in World War I, although the condition was recognized as far back as the Napoleonic campaigns. 1,2
There is a paucity of recent reports of immersion foot syndrome in humans. Rare cases of immersion foot and trench foot have been documented in homeless populations. 10,13,20 Immersion foot syndrome has also been reported in elderly patients, in a hiker with prolonged exposure to wet conditions, and with Pseudomonas aeruginosa infection in a child with a foot cast. 9,15,17 Various flood-related injuries have been described in horses, including integumentary injuries, 11 but immersion foot syndrome has not been reported in the veterinary literature. Here we describe a series of a cases resembling immersion foot syndrome in 6 equids exposed to floodwaters following Hurricane Harvey.
The Texas A&M University Veterinary Medical Teaching Hospital admitted 52 horses rescued from across southeastern Texas. Of those, 8 were submitted for necropsy. Six cases were included in this series, including 5 horses and 1 miniature donkey. All 6 animals were admitted between September 1 and September 5, 2017. Owners were identified for only 1 case (case No. 1). Full necropsies were performed on all 6 cases. In addition to routinely collected tissues, several sections of skin were sampled and fixed in 10% neutral buffered formalin for 24 to 48 hours. Sections were then processed, paraffin embedded, trimmed to 4 μm, and stained with hematoxylin and eosin (HE) for microscopic examination.
Five of the cases were adult animals between ages 2 and 20 years, and 1 was a 1-month-old foal (case No. 3). All six cases had severe concurrent disease, with respiratory disease reported in four cases (case Nos. 2, 3, 5, 6). All were euthanized except case No. 4, which died following colonic rupture. The last case (case No. 6) failed to improve after nearly a month in the hospital and was euthanized. A summary of the cases is provided in Table 1.
Signalment, Pathological Changes, and Concurrent Disease Processes in Equids With Immersion Foot Syndrome.

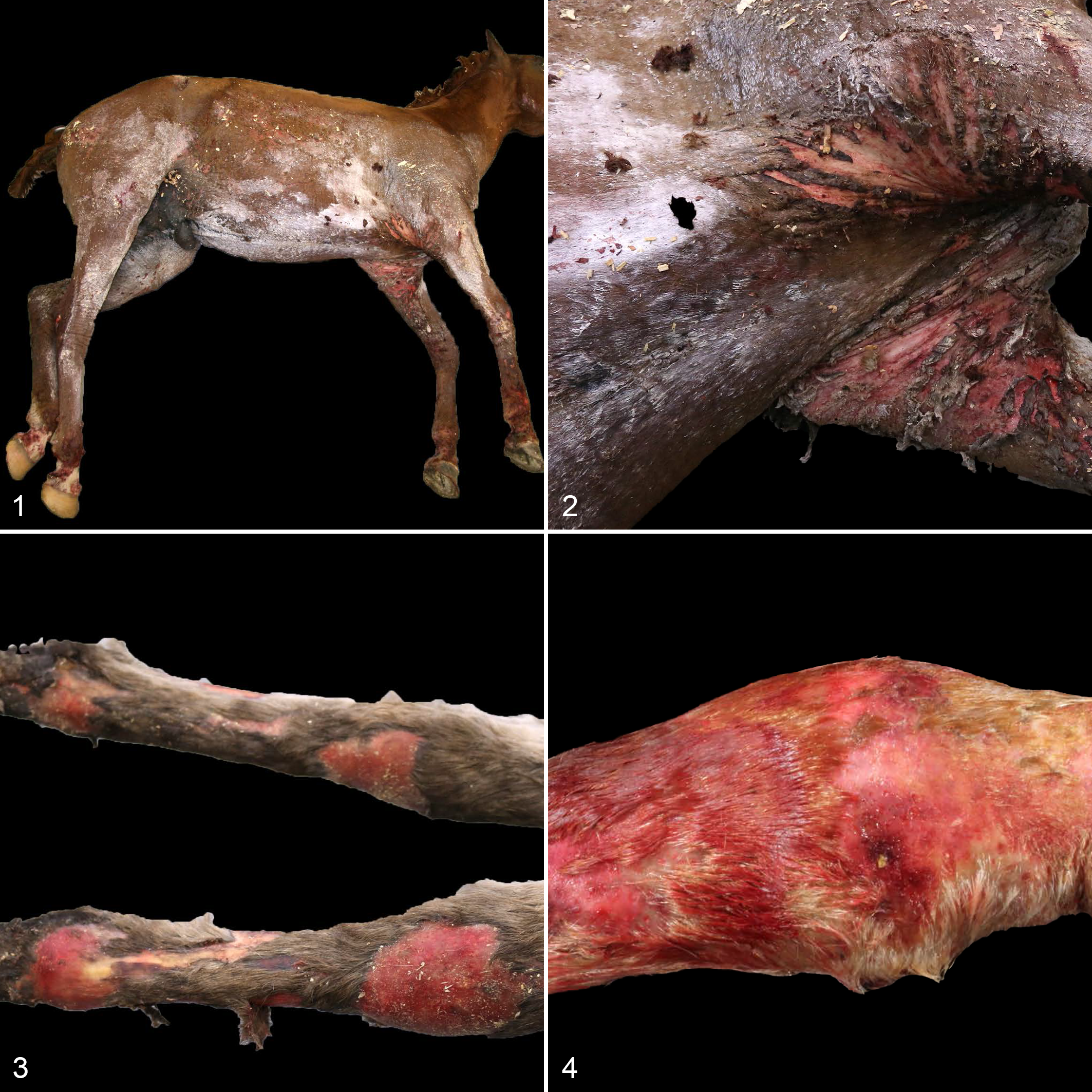
Grossly, all 6 animals had skin defects that progressed in severity from the dorsal trunk to the distal limbs. Five of 6 animals (case Nos. 1–5) had well-demarcated “water lines” along the lateral aspect of the trunk (Fig. 1), and the majority of the skin distal to this line was cracked and fissured with severe ulceration and sloughing (Fig. 2). Below this water line on the ventrum and limbs were numerous small, linear to arborizing abrasions and varying degrees of large, multifocal to coalescing areas of epidermal sloughing, crusting, erythema, and ulcerations (Figs. 3, 4). The hair coat of the last case (case No. 6), including the dorsum, was multifocally alopecic. This was clinically attributed to resolved dermatophilosis. In addition, 2 cases (case Nos. 1, 2) had laminitis, with severe rotation of the pedal bone in all 4 feet in case No. 2. Lung lobe consolidation was present in case Nos. 2, 3, and 5. The nasal cavity of case No. 6 was dark red and edematous. Gastrointestinal lesions were present in 3 cases (case Nos. 1, 4, 6). Case No. 1 had a severe enterotyphlocolitis, and Salmonella enterica was cultured from a fresh colon sample obtained at necropsy. Case No. 4 had a small colonic rupture secondary to a feed impaction. Case No. 6 had a severe, chronic gastric ulcer.
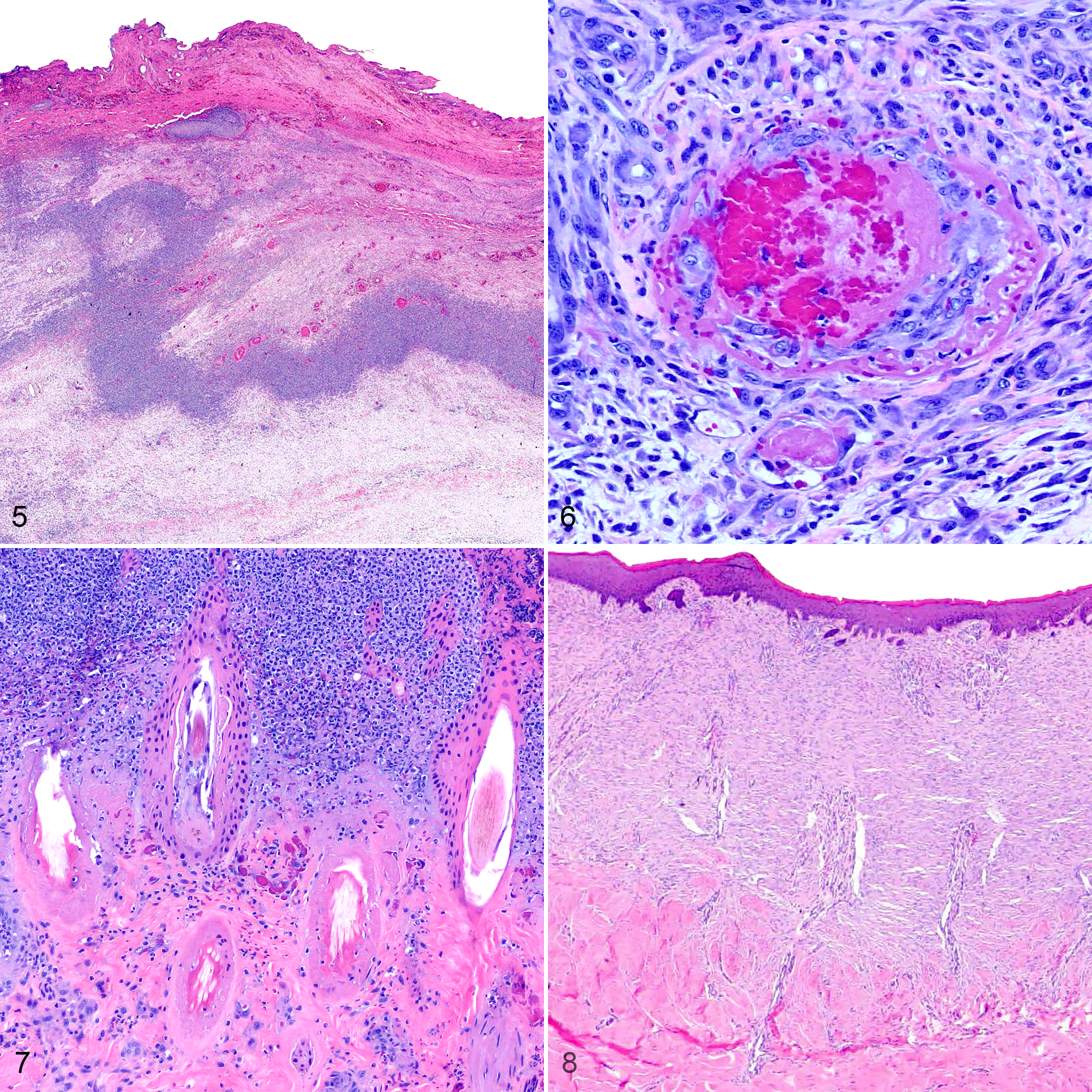
Histopathologic examination mirrored gross findings. Examination of the distal limbs in case Nos. 1 to 5 revealed varying degrees of necrotizing vasculitis, lymphoplasmacytic and histiocytic perivascular dermatitis, and epidermal, dermal, and subcutaneous coagulative necrosis (Fig. 5). Deep dermal and subcutaneous vasculitis was accompanied by thrombosis and fibrinoid necrosis (Fig. 6). Ischemic hair follicles were notable in case No. 3 and characterized by smudgy eosinophilic perifollicular collagen (Fig. 7). Case Nos. 1 to 5 had thick serocellular crusts containing necrotic debris and coccoid bacterial colonies. Case No. 6 had severe, extensive granulation tissue with diffuse adnexal loss in the skin of the distal limbs (Fig. 8). Severe suppurative bronchitis and bronchiolitis was present in case Nos. 3 and 5, and suppurative bronchopneumonia was present in case No. 2. These respiratory changes were attributed to floodwater aspiration. Bacterial culture was not performed in these cases.
In this case series, both the gross and histologic lesions were consistent with the ischemic changes described in human cases of immersion foot syndrome. Gross lesions in human reports include erythema, skin blebbing, and desquamation with subcutaneous edema and venous dilation. 7,12 In 1 case, this venous dilation ended abruptly at the demarcation zone of water immersion, 7 which correlates with a reflex venous dilation discussed below. The histopathologic changes included epidermal and dermal degeneration and necrosis with moderate to heavy leukocyte infiltration and dilated blood vessels. 7 Ulceration preceded the formation of inflamed granulation tissue, 7 and examination of chronic immersion foot cases revealed widespread fibrosis. 12 These late-stage changes resemble those of case No. 6.
The process of immersion foot has been divided into 3 stages: prehyperemic (preinflammatory), hyperemic (inflammatory), and posthyperemic (postinflammatory). 1,19 The histologic changes of all of these stages are suspected to result from direct injury to the dermal microvasculature. Histologically, the prehyperemic stage is characterized by edema, thrombosis, and vasoconstriction. 1 The hyperemic stage follows, characterized by exacerbated tissue injury, including increases in vascular permeability, subepidermal vesiculation, and reflex vasodilation. 1 Reflex or cold-induced vasodilation is an axonally driven attempt to reperfuse vasoconstricted peripheral tissues. 5,19 This vasodilation is followed by vasoconstriction in a cyclic process known as “the hunting reaction.” 5 This phasic dilation is thought to last between 5 and 30 minutes. 5,12 Continued hyperemia in immersion foot syndrome may be due to disturbances in reflex vasodilation. 19 The posthyperemic stage is histologically characterized by fibrin deposition in vessel walls, dermal atrophy, and perivascular and perineural fibrosis. 1,12 In this report, all but case No. 6 displayed varying degrees of the histologic changes associated with the hyperemic and posthyperemic stages.
While the pathogenesis of immersion foot syndrome is poorly understood, the direct effect of cold temperature on tissues has been proposed as a causal factor. Colder temperatures increase blood viscosity as well as initiate vasoconstriction in an attempt to maintain core body temperature. 1 Changes in temperature are also believed to contribute to restricted tissue perfusion, leading to an accumulation of catabolic metabolites and subsequent ischemia. 19 Direct endothelial injury can occur in chilled tissues as a result of cellular membrane damage or the crystallization of intracellular water. 3
Water itself has been described as an irritant and can incite inflammation independent of bacterial burden. 13,18 The stratum corneum is a protective barrier to foreign substances but can become more susceptible to injury when excessively hydrated. 1,18 Cracked skin is thought to be the result of denatured keratin and loss of corneal lipids, leading to decreased skin pliability. Weak irritants, including water, have been shown to incite these changes. 18 Mechanical breaks in the stratum corneum facilitate entry of bacteria and other foreign contaminants. In 2005, Hurricane Katrina floodwater was found to have fecal coliforms levels in excess of water quality standards. 14 It is likely that the floodwater from Hurricane Harvey had similar contaminants, thus contributing to secondary bacterial infections in these cases.
Differential diagnoses for dermal vascular injury include fungal infections, frostbite, and thermal burns. In this report, sections stained with Grocott’s methenamine silver (GMS) stain were examined in all 6 cases. In case No. 1, fungal hyphae were scattered within the superficial crusts as well as embedded among the deep dermal and subcutaneous inflammation. These deeper hyphae were not considered the primary source of the inflammation but rather a contributing factor to the immersion-induced changes, although a primary fungal dermatitis cannot be excluded. In addition, given the predominant distribution of inflammatory cells along the periphery of necrotic tissue, the leukocytic inflammation in these cases was assumed reactive to the severe necrosis and not in response to an infectious agent. The bacteria embedded in the crusts and the superficial fungal hyphae in case No. 1 were considered secondary to ulceration. Fungal hyphae were not evident in the examined sections for case Nos. 2 to 6.
The pathophysiology of frostbite is frequently described in parallel with immersion foot. 1,7,12 Frostbite leads to necrotizing vasculitis, thrombosis, and subsequent dermal necrosis, 10 much like immersion foot. Thermal burns also produce extensive coagulative necrosis, and vascular changes become increasingly severe as reflected by the depth of involvement. 8,10 Immersion foot syndrome is thought to manifest after at least 24 to 48 hours of exposure. 1 Due to the lack of history provided, the precise duration of immersion is not known in these horses. Given the timeline of clinical presentation, we estimate continual and progressive water immersion for at least 4 days. The clinical history and pathologic findings in this case series are most compatible with immersion foot syndrome.
In summary, this case series demonstrates a condition clinically and pathologically similar to immersion foot syndrome in humans. Since 1995, there has been an increase in the number and intensity of hurricanes in the Atlantic that may be associated with an increase in anthropogenic climate change. 16 As increasingly intense hurricanes and heavier precipitation continue to cause flooding in susceptible areas worldwide, the risk of morbidity secondary to prolonged water immersion becomes an emerging veterinary and medical concern. While exact mechanisms remain unclear, understanding the pathogenesis and its consequences on the dermal microvasculature may warrant new or alternative therapeutic strategies. Our hope is that prompt recognition of this disease will lead to appropriate medical intervention and decreased morbidity and mortality. These findings demonstrate the continued need for veterinary intervention in disasters involving companion animals and livestock.
Footnotes
Acknowledgements
We thank Drs. Brian Porter and Erin Edwards in the Pathobiology Department for their diagnostic support and Drs. Michelle Coleman, Kati Glass, and Canaan Whitfield in the Texas A&M Large Animal Hospital for their clinical diagnoses. We also thank Joshua Freeman and Alen Merdzo for their assistance in these necropsies and the Pathobiology Histology Laboratory for the histology slides.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
