Abstract
Over a 3-year-period, 17 wild-caught opaleye (Girella nigricans) housed in a public display aquarium were found dead without premonitory signs. Grossly, 4 animals had pinpoint brown or black foci on coelomic adipose tissue. Histologically, liver, spleen, heart, and posterior kidney had mesomycetozoan granulomas in all cases; other organs were less commonly infected. Four opaleye had goiter; additional substantial lesions were not identified. Granulomas surrounded melanized debris, leukocytes, and mesomycetozoa represented by folded membranes (collapsed schizont walls), intact schizonts (50- to >200 µm in diameter with a multilaminate membrane), plasmodia (budding from schizonts or free in tissue), or rarely germinal tubes (budding from schizonts). Ichthyophonus was grown from fresh tissues in tissue explant broth cultures of the heart, liver, and/or spleen. Polymerase chain reaction using 18S ribosomal DNA primers amplified a 1730-bp region, and the DNA sequence was most similar to Ichthyophonus hoferi, which is often associated with freshwater aquaculture fish.
Ichthyophonus spp. are parasites with a broad host range, including marine and freshwater fishes from across the globe. They were originally classified as fungi, but molecular studies within the past several decades determined they are relatives of choanoflagellates and the rosette agent, and they have been reclassified as Mesomycetozoae. 8 While incomplete, our current understanding of Ichthyophonus phylogenetics suggests that the majority of disease-related mortality in wild, marine, and anadromous fishes is due to a single ubiquitous marine form, while a second “freshwater” type is commonly found in trout aquaculture. 2 Based on host provenance of original species descriptions, this freshwater form is likely the type species Ichthyophonus hoferi. Diagnosis of ichthyophoniasis can be made using histology, polymerase chain reaction (PCR), or in vitro culture of tissue explants, with the latter representing the most sensitive diagnostic method. 6
The pathogenicity of Ichthyophonus may depend on multiple factors, including host/environment dynamics. Three disease outcomes have been described: acute disease and mortality, chronic disease with decreased condition, and subclinical infection. 3
Despite the broad host range, Ichthyophonus spp. have not been described in animals from the family Kyphosidae, which includes fish native to the Atlantic, Indian, and Pacific Oceans, that typically live close to the shore. Opaleye (Girella nigricans) is an omnivorous marine kyphosid fish native to the eastern portion of the Pacific Ocean with a range extending from the coast of California to Baja Mexico. 1 The aim of this work is to describe the pathologic findings of ichthyophoniasis in captive opaleye.
A school of opaleye were wild caught off of the coast of California in 2010 and moved to a tank in a public display aquarium. In 2015, 25 opaleye were moved to a new exhibit. Prior to transfer, the school received a freshwater bath as part of standard biosecurity protocol. The bath was dechlorinated tap freshwater matched for the temperature and pH of the exhibit’s saltwater. Animals were netted, placed in the freshwater bath for 3 to 5 minutes, and then renetted and placed in the new exhibit. The opaleye were moved to a mixed-species exhibit with common murre (Uria aalge), puffins (Fratercula spp.), kelp bass (Paralabrax clathratus), and a sheephead wrasse (Semicossyphus pulcher). No new fish had been added since 2015. Animals were fed a variety of flash-frozen fish (never fresh) that were thawed before feeding. These included Pacific smelt (Osmerus sp.), Columbia River smelt (Thaleichthys sp.), and capelin (Mallotus villosus). Water was sourced from Mission Bay of the Pacific Ocean (open water system). It was treated to control particulates and microbes prior to addition to the exhibit. Water quality in the exhibit was monitored and managed to maintain a constant temperature of 14°C along with controlled levels of CO2, nitrogen cycle products, and dissolved oxygen.
Opaleye that died were examined within 24 hours postmortem. A subset of tissues was fixed in 10% neutral buffered formalin and processed routinely for histology with paraffin embedding, sectioning at 5 μm, and hematoxylin and eosin staining. Gomori methenamine silver stain and periodic acid–Schiff (PAS) reaction were performed in a subset of cases.
Infection status was determined by explant culture of heart, liver, and/or spleen. Tissues were aseptically removed and placed in a 5-ml culture tube containing 3 ml Tris-buffered (pH 7.8) Eagle’s minimum essential medium (MEM), supplemented with 5% fetal bovine serum, 100 IU/mL penicillin, 100 μg/mL streptomycin, and 100 μg/mL gentamicin. Tissue explant cultures were incubated at 15°C and examined microscopically (40× magnification) for the presence of Ichthyophonus life stages. Each culture was examined twice, after 7 and 14 days of incubation, and scored as positive when Ichthyophonus life stages were detected in the broth medium. 3
PCR was performed on Ichthyophonus cultured from tissue explants. Methods were used as previously described. 2 Briefly, forward and reverse primers (NS1 5′-GTAGTCATATGCTTGTCTC-3′ and NS8 deg 5′-TCCGCAGGTTCACCWACGGA-3′) from White et al. 9 were used for PCR amplification of a region approximately 1770 base pairs long of the 18S ribosomal RNA (rRNA) gene. The amplicon 199-2 from opaleye heart DNA was directly sequenced in the forward and reverse directions using the above primers and 7 other designed primers. Sequences were generated with a BigDye Terminator v1.1 Cycle Sequencing Kit and 3130 Genetic Analyzer (Applied Biosystems, Foster City, CA). The sequence reads were aligned and ambiguous base calls manually edited using the Sequencher program (Gene Codes Corp., Ann Arbor, MI). The new contig was aligned with Ichthyophonus rDNA sequences from GenBank as well as sequences from isolates in the author’s library (J.G., data not shown). Aemoebidium sequences from GenBank were included as an outgroup. The alignment was trimmed to 1597 base pairs and a phylogeny was constructed using Maximum Likelihood methods in the program MEGA7.
Mortalities occurred in 68% (17/25) of the captive opaleye from 2015 to 2019. All animals were found dead and diagnosed with presumptive ichthyophoniasis by histopathology. Ten were male, 6 were female, and 1 was unknown sex. At necropsy, 6 animals had evidence of scavenging and/or trauma (case Nos. 1, 7, 10, 14, 15, 17), 5 animals had no gross lesions (case Nos. 2, 6, 8, 9, 12), 4 animals had a mass ventral to the opercula that was confirmed histologically as goiter (case Nos. 4, 11, 13, 16), and 4 animals had pinpoint dark brown/black spots on the coelomic adipose tissue (Fig. 1) that were confirmed histologically as mesomycetozoan granulomas (case Nos. 5, 13, 15, 16).
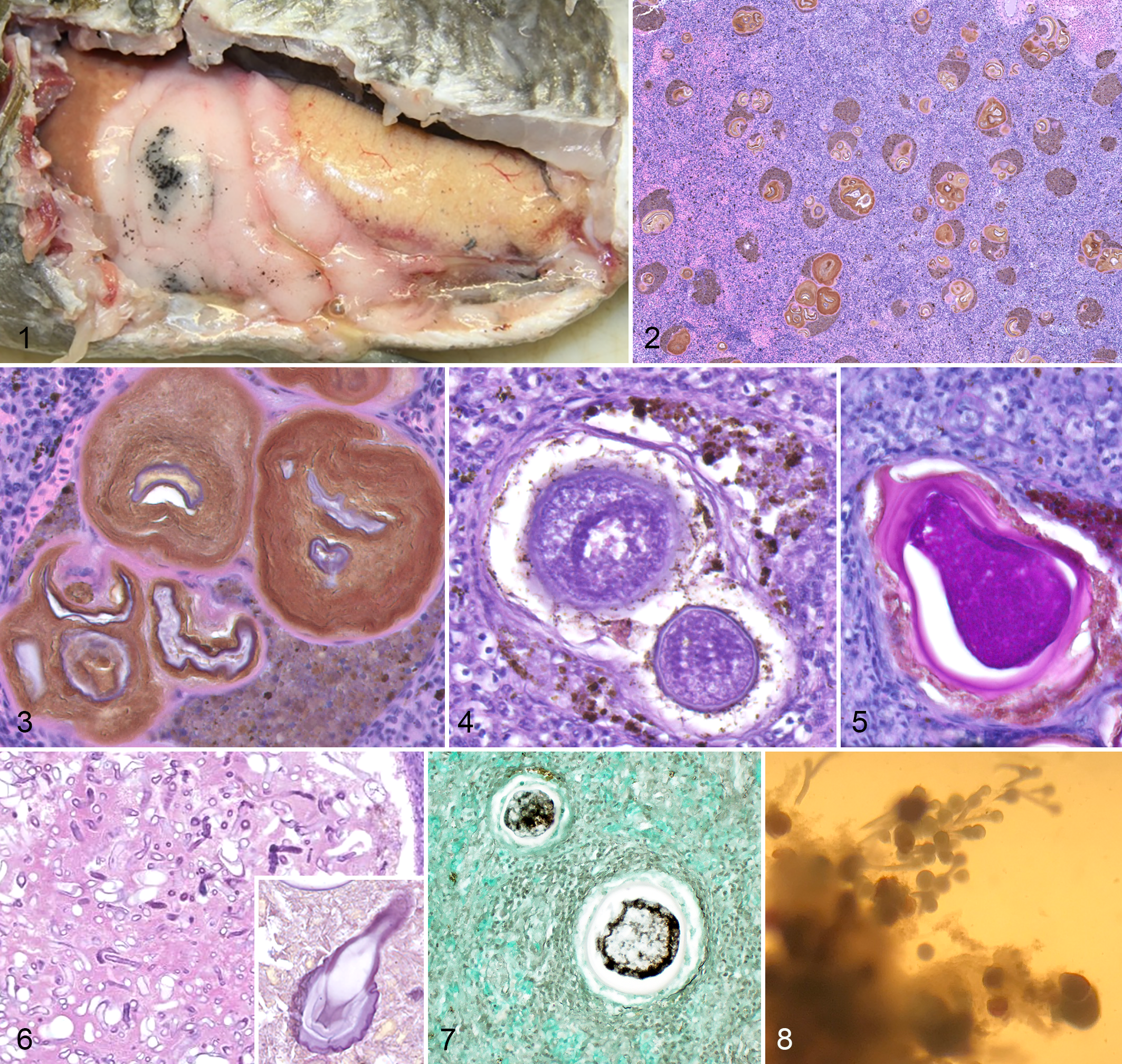
Liver, spleen, heart, and posterior kidney were examined histologically in 94% (16/17), 94% (16/17), 65% (11/17), and 47% (8/17) of mortalities, respectively. In all cases for which these organs were examined, mesomycetozoans encapsulated by a host cellular granuloma were uniformly observed in all 4 organs. Histologically, the remaining organs most commonly infected with mesomycetozoan granulomas were the stomach (88%; 14/16), intestines (86%; 12/14), coelomic adipose (86%; 12/14), anterior kidney (80%; 4/5), gills (50%; 7/14), gonad (43%; 7/16), thyroid (33%; 2/6), swim bladder (30%; 3/10), pancreas (22%; 2/9), and body wall (14%; 2/14). Mesomycetozoan granulomas occupied large portions of affected organs and were multifocal to coalescing (Fig. 2). They ranged markedly in size from macroscopic (up to 2 mm in diameter) to microscopic (40 to 200 µm in diameter). Granulomas were characterized by a thin wall composed of fibroblasts and collagen with variable numbers of internal macrophages. The wall surrounded highly melanized, acellular debris; various life stages of mesomycetozoan organisms; and rarely fragmented leukocytes. Mesomycetozoan organisms had one of several forms: intact schizonts, plasmodia, germ tubes, or empty, collapsed, folded membranes (schizont walls) (Fig. 3). Schizonts ranged from approximately 50 to >200 µm in diameter with a multilaminate membrane surrounding abundant, globular, basophilic merozoites (Fig. 4). Plasmodia were multinucleate, budded from the schizont, and sometimes had typical “flask-shape;” the membrane of schizonts and plasmodia was PAS positive (Fig. 5). Rarely, in some very large granulomas (>100 µm in diameter), schizonts also had budding germ tubes that were nonparalleled walled and nonseptate (Fig. 6). Plasmodia and merozoites were argyrophilic on Gomori methenamine silver stain (Fig. 7). Remaining histological examination was unremarkable, except in the 4 animals with goiter, in which the thyroid was markedly proliferative with numerous, hyperplastic, small, colloid-filled follicles. Tabular presentation of the histologic findings is available in Supplemental Table S1.
Tissue explant culture was performed on the liver and spleen of case No. 11 and the heart of case Nos. 13 to 15. In vitro growth and multiplication of Ichthyophonus occurred in all cultures as represented by schizonts and nonseptate germ tubes originating from parent schizonts (Fig. 8).
Ichthyophonus sp. 18S primers amplified a 1730–base pair region. When compared to reported sequences in GenBank, the sequence most closely aligned with a type from Mediterranean mullet (7 mismatches). A phylogeny based on the 18S sequence indicated the opaleye isolate was closely related to Ichthyophonus parasites isolated from freshwater aquaculture (Fig. 9).
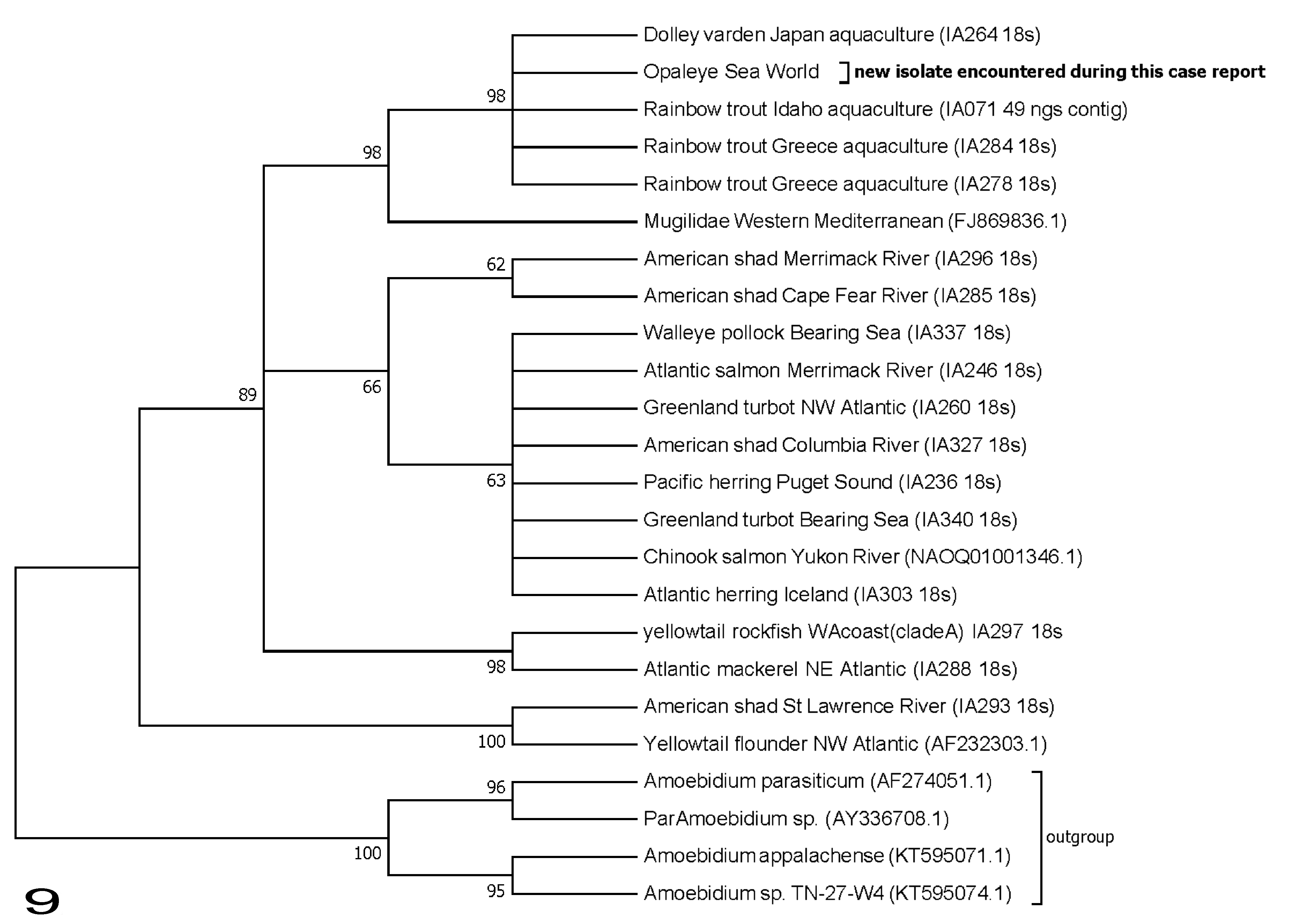
Phylogeny of Ichthyophonus spp. isolates from case No. 13 (opaleye) based on ribosomal DNA small subunit sequences (1591 base pairs). Gene tree inferred using maximum likelihood methods on a GTR-I model with 1000 bootstrap replicates in the program MEGA7 (Kumer et al. 2016). 7 Bootstrap support for clades (%) is indicated at tree nodes. Members of Amoebidiaceae included as outgroup. For Ichthyophonus isolates, host and collection location are indicated at branch tips. GenBank accession numbers or authors’ isolate identifiers are included in parentheses.
Ichthyophoniasis was associated with an ongoing, low-level rate of death in this population over a period of several years. The 3 previously characterized disease presentations of ichthyophoniasis are acute epizootic mortality, chronic morbidity without mortality, and subclinical infection. 3 The mortality in this group of opaleye was not considered epizootic due to substantial temporal spacing. The unique presentation of ichthyophoniasis in these opaleye (which does not fit into 1 of the 3 classic presentations) may be due to the nature of a closed, captive population, which would affect disease transmission and host survival.
Grossly, ichthyophoniasis sometimes causes small white nodules and pigmented ulcers in the skin, as well as small white nodules throughout infected internal viscera. 3 In opaleye, these lesions were not apparent. Nodules were present but were dark brown and most common in the contrastingly pale tan to white coelomic fat. In other organs (eg, liver, spleen), small dark brown foci could not be distinguished from melanomacrophage centers on gross examination.
Terminology used to describe life stages of ichthyophoniasis is variable due to previous classification of this organism as a fungus and then a parasite. Detailed consideration of the names for different life stages of Ichthyophonus suggests that the most appropriate terms are schizont, merozoite, plasmodium, and germ tube (previously called hypha). 5 In some animals in this series, definitively identifiable life stages were rarely present, and granulomas were centered on either necrotic cellular debris or folded membranes. These membranes represented the collapsed, acellular membranes of schizonts that had discharged all cellular material. The paucity of identifiable life stages made the histological diagnosis of ichthyophoniasis challenging in these cases. Ichthyophoniasis should be considered a differential diagnosis in opaleye with pigmented granulomas containing folded membranes. The most diagnostic tissues for histologic identification of ichthyophoniasis in opaleye were the heart, liver, and spleen, all of which were infected in all animals in which these organs were examined.
In low-level chronic infections, histology is likely an insensitive means to detect ichthyophoniasis. In some cases that are histologically negative for ichthyophoniasis, the organism can be more sensitively identified by tissue explant culture. 6 In opaleye in this case series, histologic examination of a full tissue set was a sensitive diagnostic means to detect infection in naturally infected captive opaleye. The distribution and number of organisms may differ by host species and method of infection (eg, natural vs experimental), and histopathology may not be as sensitive in other species. Tissue explant culture or PCR may have improved sensitivity in those cases.
It is unclear how these opaleye were exposed to Ichthyophonus. The isolate sequenced here is primarily found in freshwater trout aquaculture (Fig. 9) but has also been encountered in marine fish with mixed haplotype isolates 2 and in mullet (Mugilidae) from the Mediterranean Sea. 4 The opaleye in this study had no known exposure to living freshwater fish. Although they had access to some frozen freshwater fish (including river smelt, which was fed to puffins in the exhibit), freezing kills Ichthyophonus life stages, preventing transmission of infection. 3 The only known exposure these animals had to freshwater was during a freshwater bath in mid-2015. Interestingly, the first diagnosis of ichthyophoniasis in these animals was shortly after that freshwater bath in late 2015. It is possible these animals were naturally infected with Ichthyophonus sp. prior to capture and that the infections progressed to overt disease in captivity. Alternatively, they may have been infected in captivity. Because of the broad host range of Ichthyophonus, the parasite could be present in other marine fish within the aquarium.
Continued transmission of infection in the population may have occurred due to conspecific scavenging as there was evidence at necropsy that dead opaleye had been scavenged. Opaleye are omnivorous, and their diet changes depending on food availability in their environment. 1 Regardless of time of infection, it is clear that infections lasted weeks to months or possibly years before mortalities occurred.
Supplemental Material
Supplemental Material, Ichthyophonus_Suppl_Table_1 - Ichthyophonus sp. Infection in Opaleye (Girella nigricans)
Supplemental Material, Ichthyophonus_Suppl_Table_1 for Ichthyophonus sp. Infection in Opaleye (Girella nigricans) by Elise E. B. LaDouceur, Judy St Leger, Alexandria Mena, Ashley Mackenzie, Jacob Gregg, Maureen Purcell, William Batts and Paul Hershberger in Veterinary Pathology
Footnotes
Acknowledgements
We thank Drs. Salvatore Frasca Jr. and Kevin Snekvik for consultation on pursuing molecular diagnostics on these cases; Drs. Gardiner and Gasper for pathologic review of preliminary case material; Drs. Clarke, Schmitt, and Erlacher-Reid for clinical management and pathology submission of these animals; and Rachel Powers for assistance with PCR. Laboratory supplies were provided through the support of the Exxon Valdez Oil Spill Trustee Council (Project #19120111-E) and the US Geological Survey Fisheries Program, Ecosystems Mission Area. The use of trade, firm, or corporation names in this publication is for the information and convenience of the reader. Such use does not constitute an official endorsement or approval by the US government of any product or service to the exclusion of others that may be suitable. This is a SeaWorld Technical manuscript contribution number 2019-20.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclaimer
With the exception of the US Geological Survey, the views expressed in this manuscript are those of the author and do not reflect the official policy of the Department of the Army/Navy/Air Force, Department of Defense, or US government.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
