Abstract
A 4.5-year-old cow showing neurological signs consistent with predictors of bovine spongiform encephalopathy (BSE) was investigated as a potential BSE-suspect case and proved to be negative. Macroscopic analysis revealed a tan neoplastic mass growing along the leptomeninges of the caudal brain and extending into the third (III) ventricle without significantly involving the neuroparenchyma. Pathological features (uniform round hyperchromatic neoplastic cells embedded in abundant myxoid matrix, microcysts, microvascular proliferation) and diffuse Olig2 expression were most consistent with diffuse high-grade leptomeningeal oligodendrogliomatosis. In line with former reports of extensive leptomeningeal involvement in bovine oligodendroglioma, this report suggests that bovine oligodendroglial tumors have a strong propensity to grow within the leptomeningeal space. In addition, it indicates that Olig2 is a useful marker to confirm glial lineage in formalin-fixed, paraffin-embedded bovine tissue.
Keywords
Gliomas are infiltrative intra-axial tumors and have been conventionally divided into astrocytomas and oligodendrogliomas based on their histomorphological resemblance to astrocytes and oligodendrocytes, respectively. In the human 2016 World Health Organization (WHO) classification scheme, specific genetic mutations have become relevant for glioma classification, 11 but these have not been determined yet in animals. Gliomas occur most commonly in humans, with an incidence rate of 4.67 to 5.73 per 100 000 persons, 13 and in dogs, with a prevalence of 0.9%, accounting for roughly one-third (37%) of primary canine intracranial neoplasms. 16 In other animals, gliomas occur sporadically. In cattle, glial tumors are considered to be rare among nervous system tumors, 4 with only 3 oligodendroglioma cases reported to date: 2 intraspinal 2,18 and 1 intracranial. 7 In general, oligodendrogliomas grow as soft to gelatinous, infiltrative but fairly demarcated intraparenchymal masses frequently invading the meninges and/or ventricles and further spreading via the cerebrospinal fluid (CSF). Rarely, oligodendroglioma-like tumors can extensively grow within the leptomeninges and subarachnoid space, a condition known in humans as diffuse leptomeningeal glioneuronal tumor, diffuse leptomeningeal oligodendrogliomatosis, or disseminated oligodendroglioma-like neoplasm, which is listed in the current WHO classification in the group of neuronal and mixed neuronal-glial tumors. 11 This rare subtype of tumors has been sporadically reported in dogs 3,8,10 but not in other animals. Here we describe a case of disseminated leptomeningeal oligodendrogliomatosis in a young adult cow.
The brain of a 4.5-year-old cow was sent to the Division of Neurological Sciences, Vetsuisse Faculty, University of Bern (Switzerland) as a bovine spongiform encephalopathy (BSE)–suspect case for neuropathological examination. Clinically, the animal manifested ataxia, uncontrolled tremors, and circling movements and eventually died following collapse. The animal was diagnosed BSE negative based on histopathology, immunohistochemistry, and Western blot. Macroscopically, the leptomeninges between cerebellum and occipital lobe were significantly distended by a contiguously growing, soft to gelatinous and tan to grayish mass extending into the third (III) ventricle, over the entire cerebellum and to the dorsal aspect of the brainstem and attached C1 spinal cord segment. The remaining spinal cord and other organs were not available for examination. Cervical spinal cord, brainstem, cerebellum, cerebral cortex, and thalamus were embedded in paraffin, cut into 4-μm-thick sections, and stained with hematoxylin-eosin (HE). Histopathology revealed an unencapsulated and highly cellular mass primarily and continuously growing within the subarachnoid space of the occipital and temporal cortex, medulla oblongata (Fig. 1), cerebellum (Fig. 2), and first segment of the cervical spinal cord. It compressed and multifocally invaded the underlying neuroparenchyma, frequently along Virchow-Robin spaces. The neoplasm invaded into the third (III) ventricle and superficial areas of the thalamus forming a nodular mass, with additional invasion into the fourth (IV) ventricle and central canal. Neoplastic cells were arranged in sheets, nests, and cords; embedded in abundant amorphous, pale myxoid matrix staining with Alcian blue; and supported by a delicate fibrovascular stroma. In addition, the mass was traversed by thick fibrovascular bands containing hypertrophic vessels. Rarely, cells arranged around few vessels resembling pseudorosettes (Fig. 3). Large areas of the neoplasm were necrotic and contained central mineralization foci (Fig. 3). Multifocally, microcysts contained pale eosinophilic to basophilic amorphous material (Figs. 4, 5), which was interpreted to be mucin based on Alcian blue staining. These microcysts focally mimicked rosettes (Fig. 5). Neoplastic cells appeared relatively uniform and were round and small with distinct cell borders and scant eosinophilic cytoplasm. Nuclei were predominantly round, hyperchromatic, and centrally to eccentrically placed and had 1 prominent nucleolus. In addition, reniform nuclei and rare binucleated cells were observed. Anisocytosis and anisokaryosis were moderate. Mitotic figures were frequent, with 4.24 ± 0.38 (mean ± SD) mitotic figures in 10 high-power fields (400×; 2.7 mm2). Multifocally, cells had pyknotic nuclei and intense eosinophilic cytoplasm. In addition, intra- and peritumoral festoon-like glomeruloid microvascular proliferation was observed (Fig. 6). Few intratumoral large arteries showed atherosclerotic changes with thickening of the media, loss of the elastica interna, and subintimal proliferation of spindloid cells. Reactive changes included the presence of axonal spheroids, edema, and astrocytic gliosis, which were most severe in areas of compression.
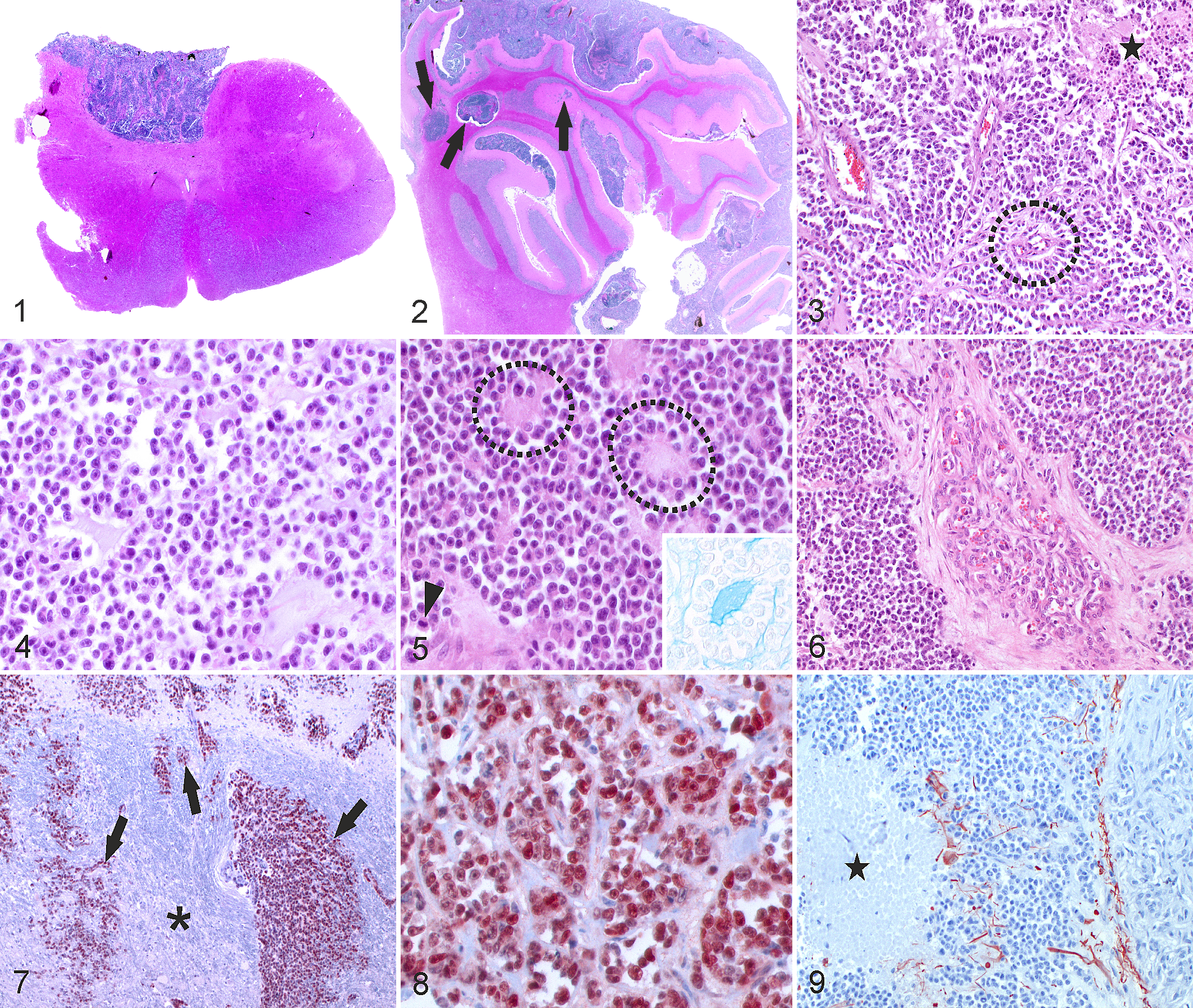
Selected tumor sections were immunohistochemically stained using antibodies listed in Table 1 according to the manufacturer’s instructions. Bovine intestinal tissue (MIB-1, cytokeratin, vimentin) and brain (Olig2, glial fibrillary acidic protein (GFAP), S100, synaptophysin, NeuN, neurofilament, β-III tubulin, CNPase) were used as positive controls. As negative control, primary antibodies were replaced with rabbit IgG (rabbit immunoglobulin, fraction [normal], 20 g/l; Dako, Glostrup, Denmark). Neoplastic cells extensively expressed nuclear Olig2 (>90% of the tumor) (Figs. 7, 8). They were negative for cytokeratin, GFAP, CNPase, vimentin, neurofilament, β-III tubulin, and synaptophysin, while normal structures present on the same slide stained with these antibodies. In contrast, both tumor tissue and brain of the cow were negative for MIB-1, NeuN, and the broad glial marker S100, while positive control bovine tissue was stained. Therefore, the negative S100, NeuN, and MIB-1 labeling was interpreted to be inconclusive, potentially due to prolonged fixation and formic acid treatment for prion inactivation due to clinical suspicion of BSE. The tumor contained few strongly GFAP-positive cells with long cytoplasmic processes interpreted to be reactive astrocytes (Fig. 9). Histopathological and immunohistochemical findings were most consistent with high-grade diffuse leptomeningeal oligodendrogliomatosis. A solid oligodendroglioma arising from the dorsal part of the thalamus growing into the III ventricle and spreading within the leptomeninges cannot be fully excluded, although the localization of the primary tumor and the very extensive leptomeningeal spread would be unusual.
Antibodies Used for Immunohistochemistry.a
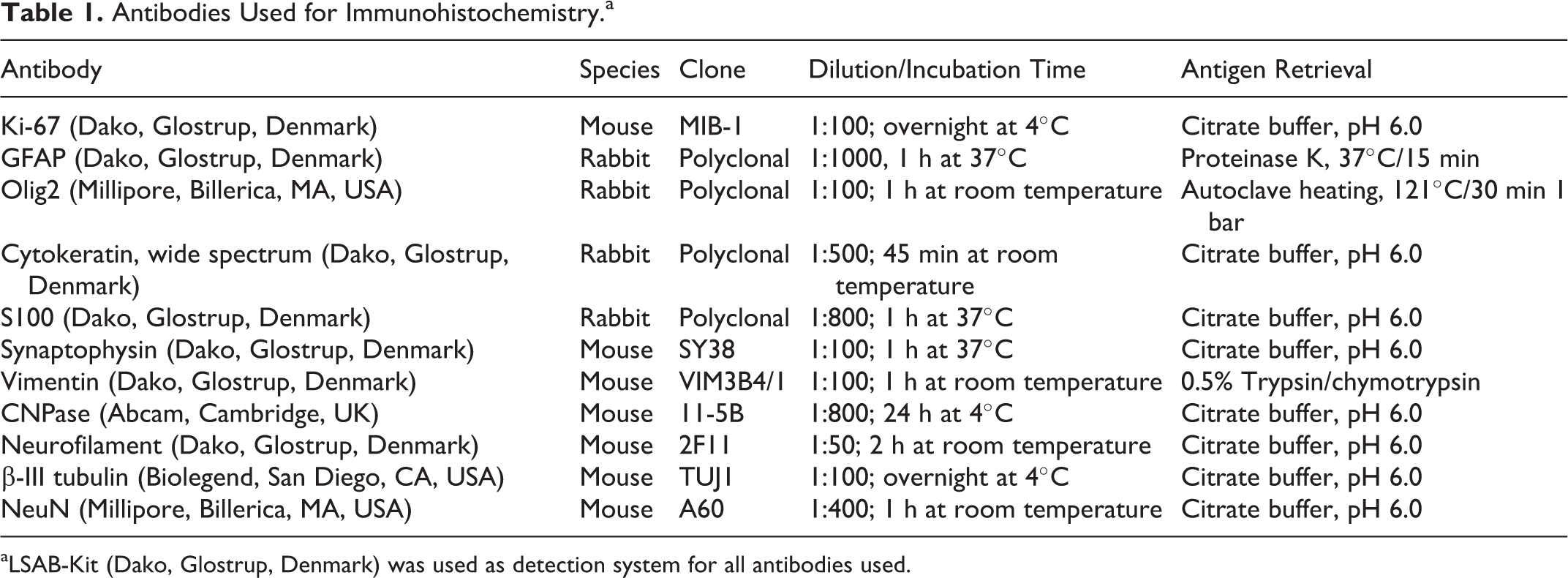
aLSAB-Kit (Dako, Glostrup, Denmark) was used as detection system for all antibodies used.
Oligodendroglial tumors are only sporadically reported in mammalian species other than human and dogs. Brachycephalic dog breeds appear to manifest a particular predisposition to develop oligodendrogliomas. 4,6 These are typically supratentorial, frequently involving the frontal or temporal lobes, although spinal localization has been rarely reported. 11 Oligodendrogliomas are prone to grow into the ventricular system and subarachnoid space and are either classified as WHO grade II or III (anaplastic) based on histopathological features. Criteria for malignancy include microvascular proliferation and necrosis, high cellularity, marked cellular atypia, and high mitotic activity. 11 In human patients, grade II oligodendrogliomas are more common, while in dogs, grade III oligodendroglioma is the most frequent type. 4,11 Several authors describe oligodendroglioma-like tumors with extensive leptomeningeal infiltration in humans as a particular tumor subtype. 1,11,14 Such tumors, described as diffuse leptomeningeal oligodendrogliomatosis, disseminated oligodendrogliomal-like leptomeningeal tumor, or diffuse leptomeningeal glioneuronal tumor, are frequently pediatric (median age of 5 years), usually low-grade (with few cases displaying anaplastic progression) tumors with or without intra-axial masses. A subset of tumors can feature neuronal differentiation with variable synaptophysin expression, hence their inclusion in the latest WHO classification in the neuronal and mixed neuronal-glial tumors. 11 Genetic mutations associated with these tumors include KIAA1549-BRAF fusion, 1p chromosomal arm deletions, and, in fewer cases, 1p/19q codeletions, which typically occur in oligodendrogliomas. 11 In veterinary medicine, 3 reports describe canine diffuse leptomeningeal oligodendrogliomatosis, but no associated genetic mutations are known. 3,8,10 Our diagnosis of high-grade (grade III) diffuse leptomeningeal oligodendrogliomatosis was supported by its morphological appearance and contiguous subarachnoid growth pattern without clearly discernible primary intra-axial mass, its extensive Olig2 immunoreactivity while lacking synaptophysin expression, and features of malignancy, including necrosis, microvascular proliferation, and high mitotic rate. Interestingly, 2 of 3 previously reported oligodendrogliomas in cattle occurred in young adult cows and featured a similarly prominent leptomeningeal infiltration without clear primary mass, similar to the case presented here. 7,18 Altogether, these reports may suggest that bovine oligodendroglial tumors have a strong preference to develop and grow contiguously in the subarachnoid space, which might be associated with specific yet unknown mutations or neuroectodermal progenitor cell heterotopies. 11
The transcription factor Olig2 represents a general glial rather than an exclusive oligodendroglial marker, being expressed in other neuroectodermal tumors such as astrocytomas, mixed gliomas, medulloblastomas, and, rarely, ependymomas. 9 However, its consistent expression in human as well as canine anaplastic oligodendrogliomas 6,9 makes it a fairly reliable marker of oligodendroglial tumors, particularly in the presence of morphological criteria. Nuclear positivity of bovine neoplastic cells in this report indicates that Olig2 is a robust antibody and might be employed for the diagnosis of bovine oligodendroglioma in FFPE tissue. Because of localization and some morphological features ([pseudo]rosettes, necrosis, microvascular proliferation), other neuroectodermal tumors including ependymoma, glioblastoma, medulloblastoma, pineoblastoma, and meningioma have to be considered. 4,11,17 Although the presence of pseudorosette- and rosette-like structures in HE-stained sections could be suggestive of a medulloblastoma, ependymoma, or pineoblastoma, these structures are very rare. In addition, pseudorosettes are a recognized feature in human oligodendrogliomas. 15 Further characteristics that favor an oligodendroglial tumor are the abundant interstitial mucinous matrix, frequent mucin-filled microcysts, and the monomorphous, predominantly round cell morphology. Alcian blue staining confirmed that the rosette-like structures were microcysts. Extensive nuclear expression of Olig2 in combination with lack of aneuronal marker and GFAP expression makes medulloblastoma, pineoblastoma, and ependymoma unlikely. Medulloblastoma and pineoblastoma are expected to show neuronal differentiation, 4,11 while ependymoma generally exhibits perivascular anuclear zones of GFAP-positive processes in pseudorosettes. 4,11 The absence of GFAP expression in neoplastic cells and cellular morphology was also used to rule out glioblastoma. 4,11 Grade II meningioma, in particular the chordoid variant, was also considered a differential diagnosis but was excluded by absence of vimentin expression. 4,11
In conclusion, although oligodendroglial tumors appear to be exceptionally rare in cattle, they have to be considered differential diagnoses in bovine neurological disease, including BSE, confirming former studies that observed neuroectodermal tumors in BSE-suspect cases. 5,12 The few reports of oligodendrogliomas in cattle may suggest that bovine oligodendroglial tumors are particularly prone to grow diffusely in the leptomeningeal space. In addition, this report shows that Olig2 expression can be detected by immunohistochemistry in bovine FFPE tissue. Further immunohistochemical characterization of ruminant oligodendroglial and other neuroectodermal tumor cases with Olig2 is desirable to assess its diagnostic validity as a glial lineage marker in this species.
Footnotes
Acknowledgements
We thank Camille Monney (Division of Neurological Sciences, Vetsuisse Faculty, University of Bern) as well as Manuela Bozzo, Erika Bürgi, and Bettina De Breuyn (Histology Laboratory, Institute for Pathology, Vetsuisse Faculty, University of Bern) for their support and excellent technical assistance. In addition, we thank the owner of the cow and the involved farm veterinarian for enabling this case report.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
