Abstract
CD30 is a transmembrane glycoprotein of the tumor necrosis factor receptor superfamily included in the diagnostic algorithm of human cutaneous, anaplastic large cell and Hodgkin lymphomas and represents an optimal therapeutic target for CD30+ tumors. Similar diagnostic and therapeutic approaches are largely missing for feline lymphomas. Cross-reactivity of the antihuman CD30 receptor clone Ber-H2 was investigated in feline lymphomas. Comparative analysis of feline and human CD30 identified 61% identity of the amino acid sequence, with 100% identity of the main sequence of the epitope targeted by the antibody (RKQCEPDYYL). CD30 expression in normal feline tissues was restricted to rare lymphoid cells in perifollicular and interfollicular lymph node areas and in the thymic medulla. In feline lymphoma, CD30 was expressed in 4 of 33 (13%) T-cell lymphomas, 3 of 22 (14%) B-cell lymphomas, and 5 of 7 (71%) mixed-cell lymphomas, showing diffuse (1/5) or multifocal (4/5) positivity restricted to neoplastic multinucleated lymphoid cells and binucleated cells consistent with Reed-Sternberg-like cells. Based on the human classification system, cell morphology, expression of multiple markers (mixed cell components), and CD30 positivity, these cases were considered most consistent with classical Hodgkin-like lymphoma (HLL). The other 2 mixed-cell lymphomas were CD30 negative and thus most consistent with either T-cell-rich large B-cell lymphoma (TCRLBCL) or nodular lymphocyte-predominant Hodgkin lymphoma (NLPHL). These findings provide multiple data supporting the cross-reactivity of the Ber-H2 anti-CD30 clone in feline tissues and give evidence of the usefulness of CD30 in the diagnostic evaluation of feline lymphoma.
CD30 is a transmembrane cytokine receptor of the tumor necrosis factor (TNF) receptor superfamily characterized by an extracellular domain, a transmembrane region, and an intracytoplasmic domain with kinase activity. 6,9,17,27 CD30 has different and somewhat antithetical functions, comprising promotion of cell proliferation and survival (including cancer cells) or suppression of replication leading to apoptosis. Functions vary, depending on the context, the target cells, and the different downstream signaling pathways. 6,17,27
CD30 has been extensively studied in human lymphomas, due to its diagnostic, therapeutic, and prognostic significance. 6,17 CD30 is expressed by normal subpopulations of activated B and T lymphocytes mostly in perifollicular areas of lymphoid organs and human Hodgkin cells and Reed-Sternberg cells of classical Hodgkin lymphoma. 6,18,26,29,30,32
CD30 expression is consistently included in the diagnostic workup of human lymphomas for the specific identification of 3 groups of lymphoid neoplasms comprising classical Hodgkin lymphoma (HL), anaplastic large cell lymphoma (ALCL), and primary cutaneous CD30+ T-cell lymphoproliferative disorders. 6,18,21,24,26,32 Other lymphoma types, such as diffuse large B-cell lymphoma (DLBCL), primary mediastinal B-cell lymphoma, peripheral T-cell lymphoma, mycosis fungoides, and adult T-cell leukemia/lymphoma, variably express CD30 with variable diagnostic and prognostic implications. 6,18 –20,29,35
Recently, studies regarding CD30 expression in human lymphomas have increased exponentially owing to its relevance as a therapeutic target due to its unique tissue distribution pattern. Specifically, CD30 is not expressed in most normal tissues outside of lymphoid organs and has minimal to no expression in resting monocytes and nonactivated lymphocytes, making this an ideal therapeutic target for CD30-positive lymphoid proliferations. Thus, selected anti-CD30 treatments have been extensively developed. Particularly impressive antitumor activity in both HL and ALCL has been shown with brentuximab vedotin (BV) T, a chimeric IgG1 anti-CD30 antibody conjugated by a protease-cleavable linker to the microtubule-disrupting agent monomethyl auristatin E. 6,11,18,23,26,29
In veterinary medicine, CD30 expression has been demonstrated in Reed-Sternberg-like cells of a single case of canine pulmonary lymphomatoid granulomatosis, and variable weak positivity has been observed in canine testicular Sertoli and germ cells and in canine seminomas. 15,25,49
Despite the establishment of several classifications systems for lymphomas, knowledge regarding prognosis and possible therapeutic molecular targets for feline lymphoma subtypes is still limited. 39 Despite their frequency, few successful and manageable therapeutic approaches are available for feline lymphomas. 38 In cats, relevant entities that morphologically parallel the human counterparts are represented by DLBCL, which is considered one of the most common feline lymphoma types; T-cell rich large B-cell lymphoma (TCRLBCL); and ALCL. 38 In addition, cats seem to develop Hodgkin-like lymphoma (HLL) more frequently than other domestic mammals. 26,36,37,40,42,47,48 Based on the established importance of CD30 in human lymphoma and the need for diagnostic, prognostic, and therapeutic targets for feline lymphoma, we investigated the cross-reactivity and the expression pattern of the anti-CD30 Ber-H2 antibody clone, recognizing the CD30 extracellular domain, in feline normal tissues and selected lymphomas. This clone was selected due to its demonstrated cross-reactivity in the dog. 26 The epitope recognized by the anti-CD30 clone Ber-H2 antibody in humans is located between amino acid residues 112 and 412 that constitute most of the extracytoplasmic domain, close to the N-terminus of the molecule. 8,31
Materials and Methods
CD30 Identity and Distribution in Normal Feline Tissues
The anti-CD30 clone Ber-H2 antibody was used to assess CD30 expression in normal feline tissues and selected feline lymphomas. To support anti-CD30 clone Ber-H2 antibody cross-reactivity in the cat, amino acid sequence identity of human (Uniprot P28908) and feline (XP_023113904.1) CD30, and specifically the Ber-H2 target epitopes, was evaluated via BLAST (National Institutes of Health, Bethesda, MD), Jalview (University of Dundee, Nethergate, United Kingdom), and Phyre2 (Structural Bioinformatics Group, Imperial College, London, UK). 2,8,12
Three-dimensional models of feline CD30 protein were created using MPI Bioinformatics toolkit (Dept. of Protein Evolution, Max Planck Institute for Developmental Biology, Tübingen, DE) and MODELLER (Departments of Biopharmaceutical Sciences and Pharmaceutical Chemistry, and California Institute for Quantitative Biomedical Research, Mission Bay Byers Hall, University of California San Francisco, San Francisco, USA) software. 50
Human and feline CD30 amino acid sequences were also scanned for conserved domains with ScanProsite software (ExPASy, Swiss institute of Bioinformatics, Lausanne, CH).
Following sequence evaluation, the CD30 expression pattern was analyzed in normal feline tissues using a tissue microarray (TMA) block that included the following organs: skin, liver, stomach, jejunum, peripheral lymph node, spleen, kidney, central nervous system, thymus, and pancreas.
Case Selection and Diagnosis
A retrospective study was performed on surgical biopsies and samples collected from necropsies of feline lymphoma cases, submitted to the diagnostic histopathology service of the Istituto Zooprofilattico Sperimentale delle Venezie of Padua, from 2008 to 2016. Samples were fixed in 10% neutral-buffered formalin, routinely processed, and stained with hematoxylin and eosin (HE). Only the cases that reported complete data about signalment, tumor site, and clinical course were included in the study.
Immunohistochemistry
Immunohistochemistry was performed on feline lymphomas included in the study for classification purposes (B cell, T cell, or mixed lymphoma) and to assess CD30 expression in feline lymphomas.
Immunohistochemistry with antihuman CD3 (polyclonal CD3-∊; Dako, Glostrup, Denmark), CD20 (polyclonal; Thermo Scientific, Waltham, US), CD79a (clone HM57; Dako, Glostrup, Denmark), and CD30 (clone Ber-H2; Dako, Glostrup, Denmark) antibodies was performed using an automated immunostainer (BenchMark ULTRA; Ventana, Medical Systems, Tucson, USA). Detailed information about the panel of primary antibody dilutions, source, retrieval, and detection systems is listed in Table 1. Cross-reactivity of antihuman CD3, CD79a, and CD20 with feline tissues has been previously demonstrated, 1,7,32 and these antibodies have been extensively used in feline tissues for diagnostic purposes.
Panel of Primary Antibodies Applied to Feline Lymphomas.
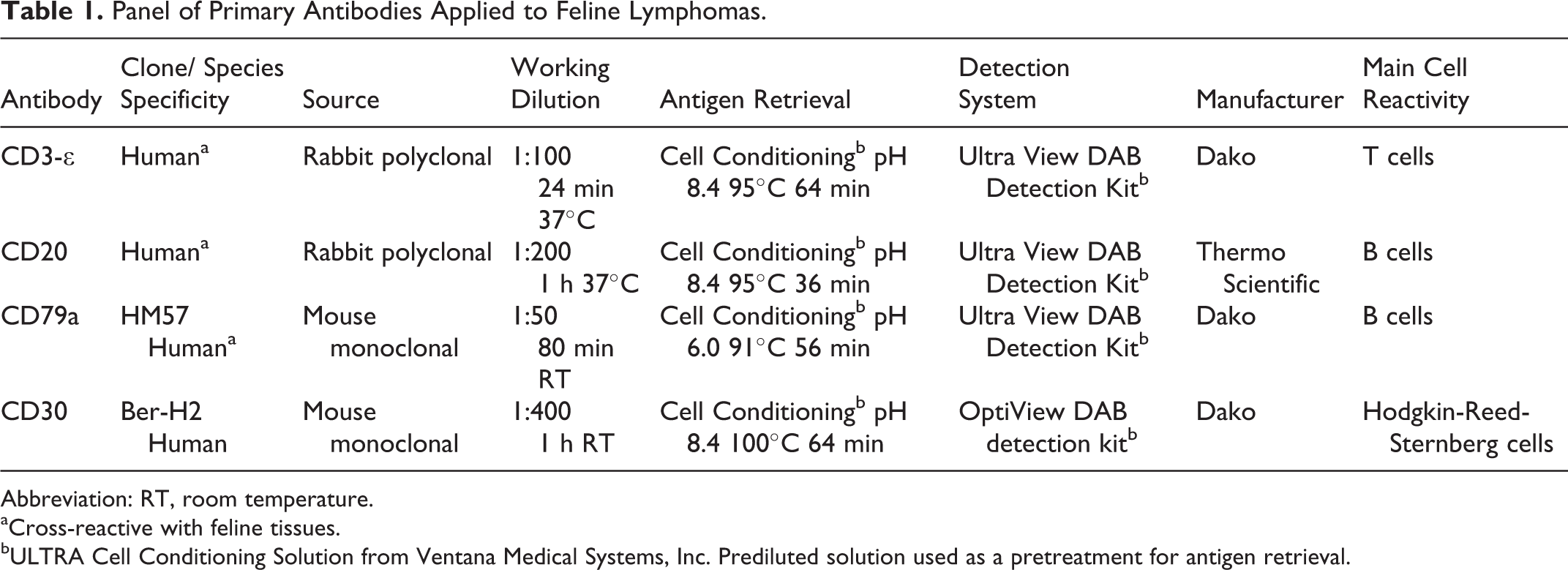
Abbreviation: RT, room temperature.
aCross-reactive with feline tissues.
bULTRA Cell Conditioning Solution from Ventana Medical Systems, Inc. Prediluted solution used as a pretreatment for antigen retrieval.
Briefly, paraffin sections (4 µm thick) were mounted onto poly-L-lysine–coated slides and dewaxed in an aqueous-based detergent solution (EZ Prep Concentrate, Ventana Medical Systems, Tucson, USA), and they underwent heat-induced antigen retrieval (Table 1). A phosphate buffer with casein and goat immunoglobulin was incubated for 4 minutes to mask the nonspecific binding sites. Sections were counterstained with Mayer’s hematoxylin. Human and feline reactive lymph nodes were used as positive controls. Negative controls were obtained by incubating replicate sections of the positive control tissues with the antimouse IgG1 (Code X0931; Dako, Glostrup, Denmark) diluted to the same mouse IgG concentration as the primary antibody.
Definitive Diagnosis
Lymphomas were classified on the basis of their anatomical location, cell type, grade, and phenotype, with the consensus of 3 pathologists (A.C., C.Z., P.R.) applying the modified World Health Organization (WHO) classification for lymphomas. 32,36,42
Results
CD30 Identity
The Ber-H2 antibody recognizes a human epitope between amino acids 112 and 412. 8,9 Alignment of the human and feline protein sequences demonstrated 100% identity in this region (Suppl. Fig. S1).
Antibody-epitope affinity of Ber-H2 was tested by Dong et al 8 by fragmenting the extracellular portion of CD30 into 117 random, partially overlapping peptides that were systematically tested against anti-CD30 Ber-H2 antibody. Three peptides demonstrated the highest affinity for Ber-H2 (Table 2) and were considered the most effective target epitopes for this antibody. 8 Selected peptides recognize a common, relatively conserved, formol-resistant epitope (RKQCEPDYYL) located in the amino acid range 239-253 aa and 237-241 aa on human and feline CD30, respectively (Table 2 and Suppl. Table S1). 8,9 Based on epitope 3-dimensional reconstruction and pocket detection analysis, these peptides seem to be located in pockets of CD30 that may increase interaction affinity with the ligand (Suppl. Table S1).
CD30 Epitopes Targeted by Ber-H2 as Reported by Dong et al 8 and Identity with Feline CD30.

Abbreviation: AA, amino acid.
Three-dimensional reconstruction of the monomeric unit of feline CD30 showed no evidence of steric impedance to the antibody binding to the protein (Suppl. Table S2).
CD30 Distribution in Feline Tissues
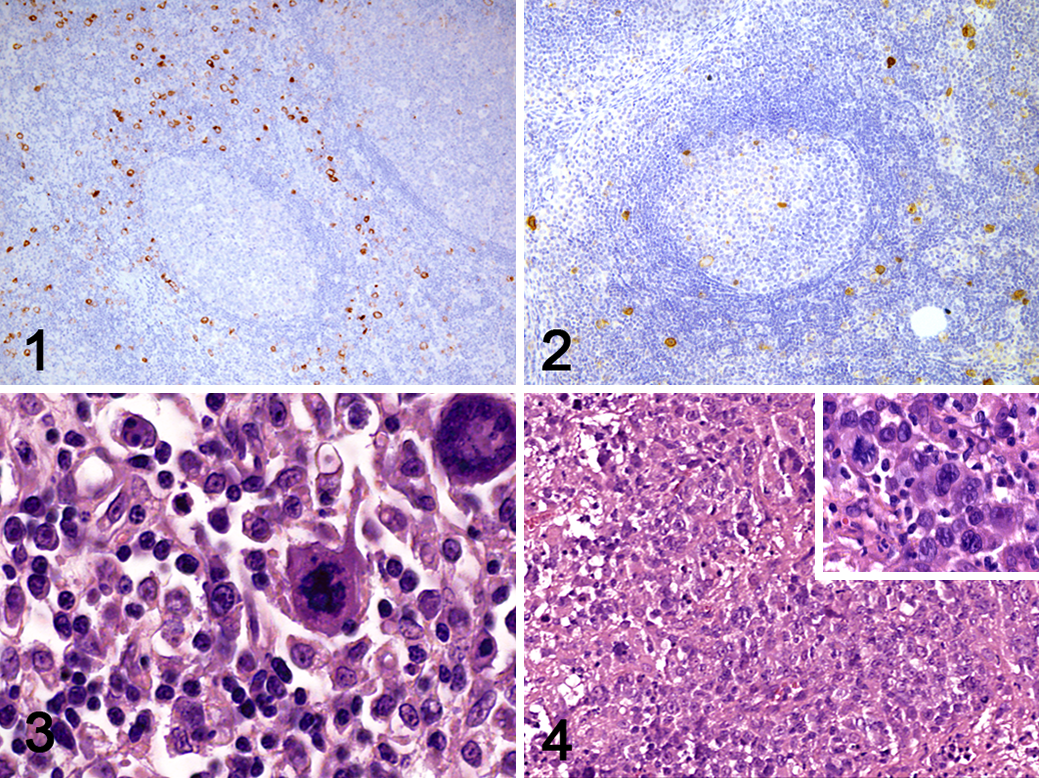
All feline tissues included in the TMA were CD30 negative with the exclusion of scattered cells in lymphoid organs. Specifically, scattered CD30 expression was detected in a small population of extrafollicular lymphoid cells and some large lymphoid cells characterized by a central nucleolus. These cells were mainly found surrounding secondary lymphoid follicles and, to a minor degree, at the edge of germinal centers in human (Fig. 1) and feline (Fig. 2) lymph nodes. Nonspecific and/or background immunolabeling was not observed in TMA samples.
Classification of Lymphomas
Sixty cases of lymphoma were included in this study. Cats represented in the study included 57 domestic shorthaired (DSH), 2 Siamese, and 1 Singapura, with a mean age of 9.7 years (range, 1–16 years). Thirty-three cats were male (3 intact) and 27 were female (2 intact). Based on CD3, CD20, and CD79a immunohistochemistry, 32 cases were T-cell lymphomas (53%), 20 cases were B-cell lymphomas (33%), 7 cases showed a mixed population of B and T lymphocytes (12%), and 1 case was null-cell type (2%). Two T-cell lymphomas were classified as ALCL, based on the severe pleomorphism, frequent multinucleation, and “Hallmark cells” with C-shaped nuclei (Fig. 3). The mixed-cell lymphomas, characterized by a proliferation of small lymphocytes with scattered large and sometimes binucleated cells, were not definitively classified due to morphological overlap between HLL and TCRLBCL (Fig. 4). Five intestinal lymphomas were classified as large granular lymphoma (LGL), on the basis of the presence of granular cytoplasm and immunophenotype (4 T cell and 1 null cell).
CD30 Expression in Feline Lymphomas
CD30 was expressed in 20% of cases, varying from a diffuse (8/60) to multifocal (4/60) pattern. According to the immunophenotype, CD30 was expressed in 13% of T-cell lymphomas, 14% of B-cell lymphomas, and 71% of mixed-cell lymphomas (Suppl. Table S3). Both cases of ALCL showed diffuse membranous and cytoplasmic CD30 expression, often in association with a cytoplasmic dot-like immunolabeling thought to represent the Golgi apparatus (Fig. 5a–d). Multinucleated neoplastic cells showed strong CD30 positivity (Fig. 5e).
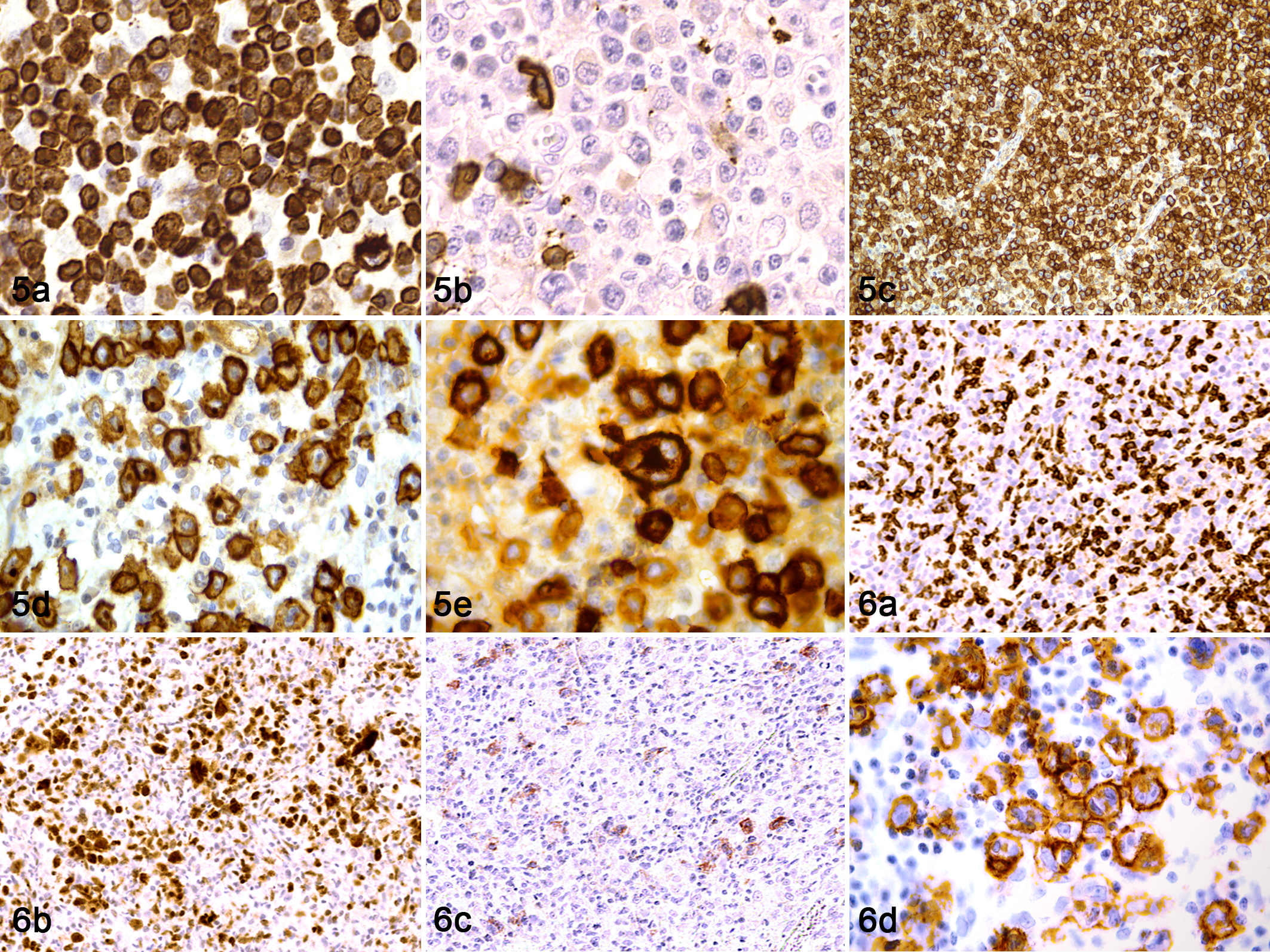
Five of 7 mixed-cell lymphomas expressed CD30, showing diffuse (1/5) or multifocal (4/5) positivity restricted to neoplastic multinucleated lymphoid cells and binucleated cells consistent with Reed-Sternberg-like cells (Fig. 6a–d).
Based on the human classification system, cell morphology, expression of multiple markers (mixed-cell components), and CD30 positivity, these cases were considered most consistent with classical HLL. The other 2 mixed-cell lymphomas were CD30 negative and thus most consistent with either TCRLBCL or nodular lymphocyte-predominant Hodgkin lymphoma (NLPHL). Detailed data of the cases are listed in Supplemental Table S3.
Discussion
The aims of this work were multiple. The first aim was to analyze the effective feline protein cross-reactivity of the antihuman CD30 Ber-H2 primary antibody that had been previously successfully tested in dogs. 26 The Ber-H2 monoclonal antibody binds to a formol-resistant epitope of the extracellular domain of the human CD30 molecule, allowing its utilization in routine diagnostics and in retrospective studies. 5,17,31 Although the overall amino acid identity of human and feline CD30 was 61%, the identity between anti-CD30 Ber-H2 epitopes and the target complementary sequences was 100%. From a functional point of view, the presence of TNFR/NGFR cysteine-rich domains, located in similar positions in both human and feline CD30 sequences, suggests that CD30 finds cytokines and has a similar function in both species. Moreover, cysteine-rich domains are involved in multimerization and signal transduction of TNFR superfamily members, 4 and these functions are likely to be conserved in orthologous molecules.
Although digital reconstruction of secondary and ternary structures of the feline CD30 molecule did not show any sterically relevant difference in Ber-H2 binding sites between the human and feline proteins, bioinformatics-based predictions have limits that have to be taken into account. One of the major limits is that 3-dimensional reconstruction is based on tridimensional models available for molecules sharing similar structures that may cause biases in the model generation. In the present case, the immunolabeling pattern in feline lymphomas and previous results obtained in dogs support the hypothesis that there is no steric impediment to the anti-CD30 Ber-H2 antibody binding complementary sequences in the cat or dog. Several molecules with major roles in immune function such as CD3, CD79a, and CD20 are characterized by having high identity between mammalian species. Human antibodies raised against these molecules have demonstrated formol resistance and effective cross-reactivity, allowing for their extensive application to veterinary studies in several species, including the characterization of lymphomas in cats. 1,7,28,29,33,34
Likewise, CD30 expression in normal feline tissues was observed in interfollicular areas of feline lymphoid tissues and in the thymic medulla, paralleling descriptions of CD30 expression in human medicine. 5,10,17 These results are compatible with CD30 restriction to activated CD4 and CD8 T cells of the thymic medulla, perifollicular and interfollicular T-cell areas of peripheral lymph nodes, and a minority of stimulated B-cell immunoblasts located at the edge of germinal centers and in extrafollicular lymph node regions. 6 The immunolabeling pattern of CD30 in feline lymphomas was characterized by membranous and dot-like cytoplasmic expression with a more prevalent and intense immunolabeling in large cells (immunoblasts, HLL cells, and Reed-Sternberg-like cells), paralleling the CD30 cell expression pattern described for human counterparts. 3,34,35
Ideally, a Western blot comparative analysis using fresh feline and human tissues expressing CD30 would have completed the interspecies cross-reactivity evaluation. However, CD30 is scarcely expressed in normal tissues, and no fresh tissues from feline and human lymphomas could be obtained, so Western blot analysis was not possible in this retrospective study.
The second aim of the present study was to evaluate the usefulness of CD30 in feline lymphoma classification. CD30 is considered one of the most specific markers for the diagnosis of ALCL and all forms of classical HL in people. 6,17,43,44,46 In addition, CD30 has been found in a subset of DLBCL and peripheral T-cell lymphomas. 6,17,41 Although no conclusions can be drawn due to the low number of specific feline lymphoma types included in the current caseload, we believe this study provides promising preliminary results for the inclusion of CD30 in the diagnostic workup of lymphoproliferative disorders, particularly for classification of ALCL and HLL. Notably, while the diagnosis of human systemic and cutaneous ALCL is based on morphology and CD30 expression, no specific markers assisting in the diagnosis of ALCL and HLL are available in veterinary pathology, and their diagnosis is based only on morphological features. 36,42,46 In humans, THRLBCL, LGL, and natural killer (NK), hepatosplenic, T-zone, and angioimmunoblastic lymphomas and T-cell prolymphocytic leukemia/lymphomas are mostly CD30 negative. 19 As a parallel, in our caseload, feline TCRLBCL, LGL, and T-zone lymphomas did not have detectable CD30 expression. In veterinary medicine, feline HLL can be misdiagnosed as TCRLBCL, and differentiation of these 2 entities can be difficult due to the lack of specific markers. In humans, CD30 is considered one of the best diagnostic tools to differentiate these 2 entities since all classical HL subtypes express CD30 while nodular lymphocyte-predominant HL and THRLBCL do not express CD30. 11,45 Their diagnostic differentiation has clinical and therapeutic relevance since HL and THRLBCL bear very different prognoses in humans and likely in cats. Feline HLL has been reported to be less aggressive than non-Hodgkin lymphoma, although the number of reported cases is limited and extensive epidemiological analysis of survival data would be necessary to validate these findings. 40
In addition, in our caseload, CD30 was diffusely expressed by subpopulations of T-cell (13%) and B-cell (14%) lymphomas, specifically DLBCL. In our view, this finding partially resembled the distribution described for human lymphomas where CD30 is variably expressed by up to 20% of DLBCL and 30% of T-cell malignancies. 4,6,7,17 –19,33,36,42 Although CD30 was variably identified in subsets of human DLBCL and peripheral T-cell lymphoma not otherwise specified, the diagnostic and prognostic role of CD30 in these cases is still a matter of debate. 14,16,18,24
Major limitations of this study were the low number of specific feline lymphoma entities and the lack of adequate follow-up to draw clinical or prognostic conclusions. Regarding the possible prognostic role of CD30 in feline lymphomas, this aim should be considered minor for a retrospective immunohistochemical investigation, while the main purpose was to evaluate the cross-reactivity and applicability of CD30 expression to the feline lymphoma diagnostic workup. The prognostic relevance of CD30 in humans is supported by the identification of HL and of a subgroup of human cutaneous lymphomas specifically termed CD30-positive lymphoproliferative disorders that are characterized by a favorable to excellent prognosis in comparison to CD30-negative cutaneous lymphomas.
Prospective studies on more numerous and focused caseloads are needed to confirm the usefulness of CD30 in the diagnosis of selected feline lymphoproliferative disorders and in the analysis of its possible prognostic relevance. Nonetheless, our findings suggest that CD30 could be a therapeutic target for CD30-positive feline lymphomas as it is for human beings. In humans, CD30 is considered a useful molecular target for several reasons, including its absence in most normal tissues outside of the immune system. 6,13,17,22,25 Thus, multiple treatments directed to CD30 have been developed in people, such as naked monoclonal antibodies, radiolabeled antibodies, bispecific antibodies, and antibody-drug conjugates. 21,25,27
In summary, our work attests to the cross-reactivity of the Ber-H2 anti-CD30 clone in feline tissues, provides preliminary evidence of the usefulness of CD30 in the diagnostic evaluation of specific feline lymphoma subtypes such as ALCL and HLL, and encourages future studies aimed to evaluate the role of CD30 as a therapeutic target for selected feline lymphomas.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819875745 - CD30 Cross-Reactivity and Expression in Feline Normal Tissues and Lymphomas
Supplemental Material, DS1_VET_10.1177_0300985819875745 for CD30 Cross-Reactivity and Expression in Feline Normal Tissues and Lymphomas by Antonio Carminato, Marco Tecilla, Paola Roccabianca, Claudia Zanardello, Erica Melchiotti, Katia Capello and Marta Vascellari in Veterinary Pathology
Footnotes
Acknowledgements
We thank the technical staff at the Histopathology Department of the Istituto Zooprofilattico Sperimentale delle Venezie of Padova (Italy) for their assistance with this research. Thanks to Mr. Alberto Masiero for the technical support in graphic design.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
