Abstract
Chondrodystrophy results in predictable and progressive biochemical and structural changes to the intervertebral disc, resulting in early onset degeneration and dystrophic mineralization of the disc. Accelerated degeneration and mineralization of the intervertebral disc are common in multiple dog breeds and can result in compromised function, herniation, pain, and a variety of neurological sequelae. A mutation responsible for chondrodystrophy in dogs has been identified as an aberrant fibroblast growth factor 4 (FGF4) retrogene insertion on chromosome 12 (CFA12) and is associated with short stature of the Nova Scotia Duck Tolling Retriever. Segregation of the CFA12 FGF4 retrogene in this dog breed provides an opportunity to examine the effect of retrogene presence on radiographic and histologic appearance of chondrodystrophic disc degeneration within a single breed. Here we found that in the intervertebral discs isolated from 2 dogs with the CFA12 FGF4 genotype, the nucleus pulposus was largely replaced by cartilaginous tissue, and physaliferous notochordal cells were rarely if ever identified. These findings are in contrast to the normal histologic findings in 2 breed-matched dogs lacking the mutation. The findings are consistent with premature chondroid degeneration of the intervertebral disc and suggest that the presence of the CFA12 FGF4 retrogene is sufficient to cause the chondrodystrophic phenotype.
The intervertebral disc (IVD) is a highly specialized cartilaginous joint or symphysis uniting 2 adjacent vertebrae and facilitating limited spinal movement. The IVD comprises 3 interdependent structures: the centrally located nucleus pulposus (NP), the surrounding annulus fibrosus (AF), and the cranial and caudal cartilaginous endplates marking the transition between the IVD and the adjacent vertebral bodies. 17 The NP is entrapped and encapsulated within the annulus fibrosus, a structure comprising concentric layers of fibrocartilage with alternating orientation of collagen molecules within each successive layer. The NP is the core of the IVD, a unique gelatinous tissue type, and a developmental remnant of the embryonic notochord. The notochord essentially disappears in the osseous sites (vertebrae) but persists inside the AF, where it is believed that the entrapped notochord cells synthesize the matrix of the NP. 15,16,30 In young animals, the matrix of the NP is translucent and rich in proteoglycan molecules and water. 21 Braund et al 5 have referred to this appearance of the NP as “mucoid.”
NP-associated proteoglycans consist of numerous glycosaminoglycan (GAG) sidechains covalently bound to a central protein core. In a healthy IVD, the presence of abundant GAG molecules within the NP osmotically maintains a high water content and facilitates the physiologic compressibility of the IVD. The cellularity of the immature NP is generally sparse but variable and includes clusters of small “fibrocyte”-like cells and large vacuolated physaliferous cells (also referred to as notochordal cells) arranged as individual cells or in clusters. 5,21
Chondrodystrophy is a term first introduced by Hansen 13 to describe dogs with premature degeneration of the nucleus pulposus of the intervertebral discs and shortened long bones. Although age-related degeneration of the intervertebral disc occurs in all dog breeds, premature degeneration is a distinct feature of chondrodystrophic dogs. Chondrodystrophic degeneration of the IVD occurs at an early age in predisposed dog breeds and results in an altered ratio of GAG to collagen in the NP (decreased GAG and increased collagen). 21 During degeneration, the NP undergoes organizational and biochemical changes, which result in alteration in the mechanical properties of the disc and eventually lead to structural failure. 10,28,30 Degeneration of the NP and the surrounding AF compromises the function of the IVD and may culminate in disc herniation, which can be associated with clinical manifestations, including pain, paresis, and/or paralysis. Nonchondrodystrophoid dogs are reported to lose their notochordal cells relatively late in life, around 5 years of age, 13 while in chondrodystrophic dogs, this change is accelerated. In chondrodystrophic dog breeds, the collagen composition of the NP increases rapidly with maturity and by 11 months of age averages approximately 25%, while in nonchondrodystrophic breeds, the collagen content generally remains <5% for life. 21 As a result of progressively decreasing proteoglycan content in chondrodystrophic breeds, the NP throughout the vertebral column is largely replaced by dry, gray-white fibrous tissue and cartilage by 12 months of age. 21 Mineralization of degenerate IVD often occurs and may be apparent radiographically.
The genetic variant that is responsible for chondrodystrophy in dogs was identified as a fibroblast growth factor 4 (FGF4) retrogene insertion on canine chromosome 12. This locus was defined by first identifying a genomic region associated with short stature in the Nova Scotia Duck Tolling Retriever (NSDTR) dogs followed by across-breed mapping of cases affected by disc herniation. 7 The NSDTR is a medium-sized dog bred for hunting and often mistaken as a small Golden Retriever. Retrogenes are DNA copies of processed RNA lacking introns (complementary DNA [cDNA]) that are inserted into the chromosomal DNA at a new location, resulting in additional copies of the parent gene. Although many retrogenes are considered nonexpressing pseudogenes, the chondrodystrophy-associated CFA12 FGF4 retrogene is known to be transcriptionally active, and a 20-fold higher expression of FGF4 has been demonstrated in discs from chondrodystrophic neonatal puppies. 7 Interestingly, the “shortened limb phenotype” has also been associated with another FGF4 retrogene insertion, in this case on canine chromosome 18, but this variant has not been described as a major contributor to intervertebral disc degeneration. 24 Long bone morphometric changes may be additive when both retrogenes are present in the same breed, and chondrodystrophic breeds with profound limb shortening such as Dachshunds, Basset Hounds, and Corgis frequently carry both CFA12 and CFA18 FGF4 retrogenes.
Most historical histopathological characterizations of intervertebral discs from chondrodystrophic dogs have been reported in breeds with a high CFA12 FGF4 retrogene frequency, and comparison to “normal” discs has been done using nonchondrodystrophic control breeds such as Greyhounds. 5 Based on breed standard conformation, the NSDTR breed would not be morphologically classified as chondrodystrophic. However, in this CFA12 FGF4 retrogene segregating breed, homozygous affected dogs may have a more classical chondrodystrophic limb phenotype, while unaffected animals have a more normal conformation. Segregation within the NSDTR breed provides an opportunity to compare histopathological characteristics of intervertebral discs in the presence or absence of the CFA12 FGF4 retrogene against a common breed background.
Materials and Methods
Animals, Pathology, and Postmortem Imaging
The animals used in this study were NSDTRs that died of natural causes or were euthanized at the request of their owners for medical reasons unrelated to IVD disease (Supplemental Table 1). Collection of samples for DNA analysis was approved by the University of California, Davis (UC Davis), Animal Care and Use Committee.
Animals were either euthanized and the entire animal was shipped (dog Nos. 1 and 2) or the spine was initially isolated via on-site necropsy (dog Nos. 3 and 4) and subsequently shipped to the UC Davis Veterinary Medical Teaching Hospital. DNA samples were obtained from dog Nos. 1 and 2 from skeletal muscle tissue prior to formalin fixation while DNA was extracted from buccal swabs obtained from dog Nos. 3 and 4. Samples were collected postmortem, chilled, and shipped with the vertebral column and spinal cord segment within 48 to 72 hours. A targeted necropsy was performed at UC Davis on dog Nos. 1 and 2, and CT or radiographic imaging of the entire vertebral column was available for these 2 cases.
Vertebral columns from all 4 animals were incised sagittally (hemisectioned) using a band saw and the cut surfaces of dog Nos. 1 and 2 were digitally photographed. Hemisectioned spines were immersed in 10% buffered formalin for 48 hours, and approximately 1.5- to 2-mm-thick parasagittal sections of the vertebral bodies and associated IVD were obtained using a band saw. These sections were further fixed in 10% buffered formalin for 8 hours, then decalcified in 15% formic acid for an additional 48 to 72 hours. Subsequent to decalcification, discs were isolated by cutting through the vertebral bodies, leaving the endplates intact. For each case, 2 to 4 representative IVD and adjacent vertebral bodies from the cervical, thoracic, and lumbar vertebral column regions were trimmed into embedding cassettes. Tissues were routinely processed and sectioned into 5-µm-thick formalin-fixed, paraffin-embedded (FFPE) sections, adhered to positively charged glass slides, and stained with hematoxylin and eosin (HE), Alcian Blue (American MasterTech, St Lodi, CA), or Picro-Sirius red (Picro-Sirius Red Stain Kit; American MasterTech) using standard protocols or the manufacturer’s instructions. Alcian blue stains mostly GAG while Picro-Sirius red stains collagen, with a higher affinity for collagen type I. All of the stained FFPE spinal sections were examined and digitally photographed by a single pathologist (B.G.M.). A single representative HE-stained section from each case was digitally scanned using an Olympus VS120 scanner. For all 4 cases, the available vertebral column sections and associated IVD were scored using 2 different rubrics, a histological grading scheme for canine intervertebral disc degeneration 2 and a chondrification index. 5
Genotyping
DNA was extracted from skeletal muscle tissue at the time of necropsy (dog Nos. 1 and 2) or from buccal swabs (dog Nos. 3 and 4) using a commercial kit (Gentra Puregene DNA purification extraction kit; Qiagen, Valencia, CA). Genotyping for the CFA12 and CFA18 FGF4 retrogenes was performed by a 3-primer polymerase chain reaction (PCR) reaction (HotStarTaq PCR Kit; Qiagen) as previously described. 7 A 654-bp amplicon is expected if the CFA12 FGF4 retrogene on chromosome 12 is present, and a 333-bp amplicon is expected from the normal allele. A 168-bp amplicon is expected if the CFA18 FGF4 retrogene on chromosome 18 is present, and a 388-bp amplicon is expected from the normal allele. PCR products were visualized on a 2% agarose gel using ethidium bromide, and heterozygous controls were included for both assays.
Results
Genotyping
Signalment and genotyping of cases are presented in Table 1. DNA samples were genotyped for the FGF4 retrogene on CFA12 to determine the presence or absence of the chondrodystrophy mutation (Fig. 1). Samples 1 and 2 (ages 31 months and 10 weeks, respectively) were determined to be homozygous for the FGF4 retrogene on CFA12, represented by a single 654-bp band. Samples 3 and 4 (ages 10 weeks and 7 weeks, respectively) were homozygous for the normal allele represented by a single band at 333 bp. All cases were also homozygous for the normal allele on CFA18 (data not shown).
Intervertebral Disc Histologic Lesion Scores for 2 Nova Scotia Duck Tolling Retriever Dogs With Intervertebral Disc Disease and 2 Controls.a
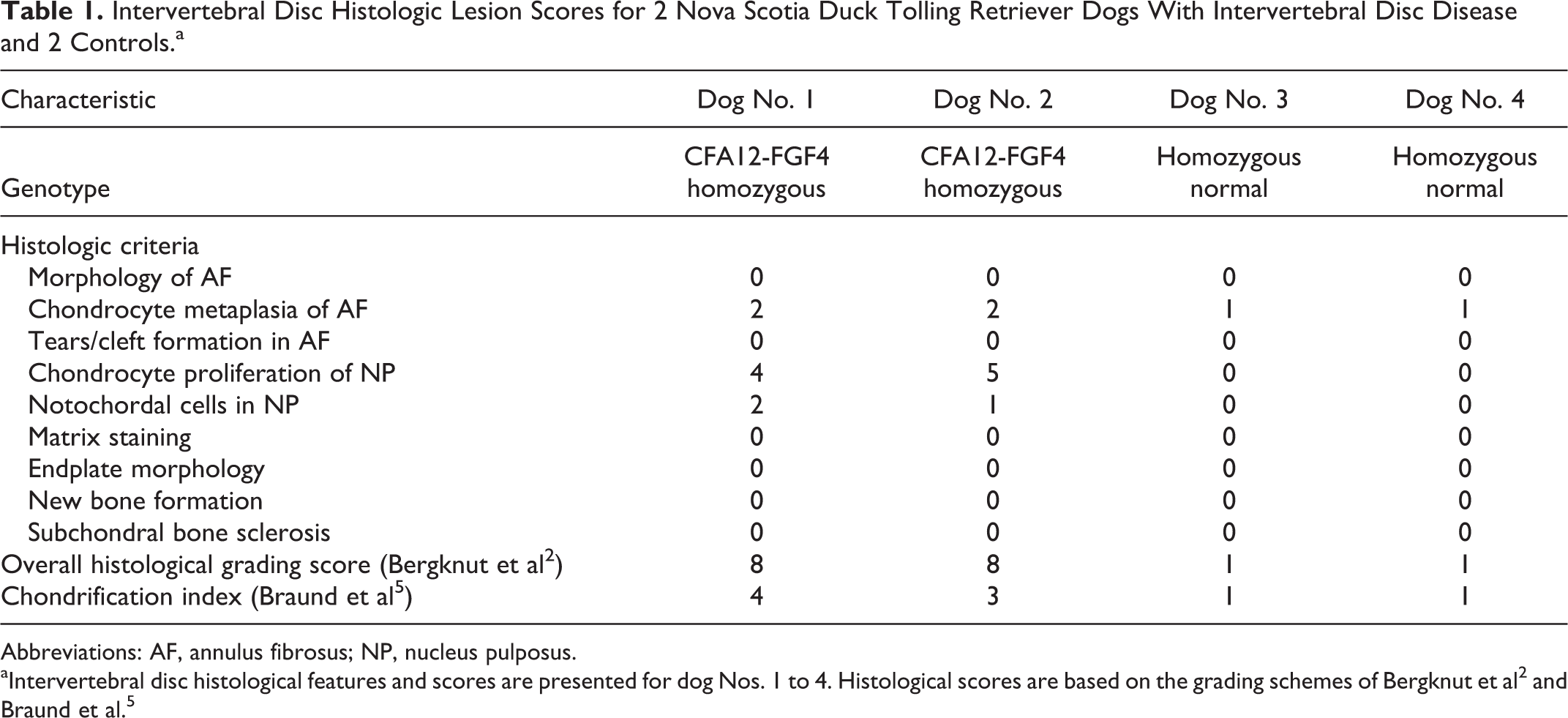
Abbreviations: AF, annulus fibrosus; NP, nucleus pulposus.
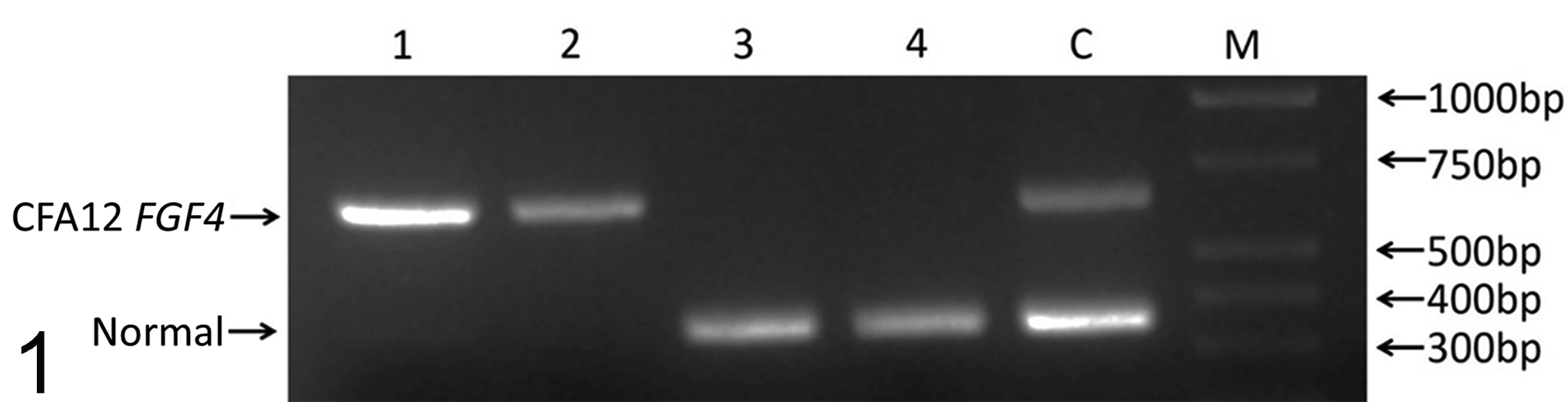
A polymerase chain reaction–based genotyping assay distinguishes between normal and CFA12 FGF4 retrogene-carrying animals. Lanes 1 to 4, dog Nos. 1-4; C, control heterozygote; M, DNA ladder marker. Dog Nos. 1 and 2 were homozygous for the CFA12 FGF4 retrogene on chromosome 12, and dog Nos. 3 and 4 were homozygous normal.
Radiography and Gross Pathology
On gross examination of unfixed, sectioned vertebral columns isolated from dog Nos. 1 and 2, the NP of the IVD was glistening, opaque, and white; the structures were grossly reminiscent of adipose tissue. For dog No. 2, the discs throughout the length of the vertebral column bulged on sectioning. In dog No. 1, only scattered discs bulged on sectioning. Vertebral endplates and physes were grossly evident in the adjacent vertebral bodies of dog No. 2 (10-week-old dog) but were not evident for dog No. 1 (31-month-old dog with closed vertebral physes). For multiple thoracic vertebrae in dog Nos. 2 and 4, the cranial and caudal physes of the vertebral body were often joined by an additional physis within the sagittal plane (Figs. 4, 8). This anatomical feature was evident both grossly and histologically. There were no gross abnormalities of the appendicular skeleton of dog Nos. 1 and 2.
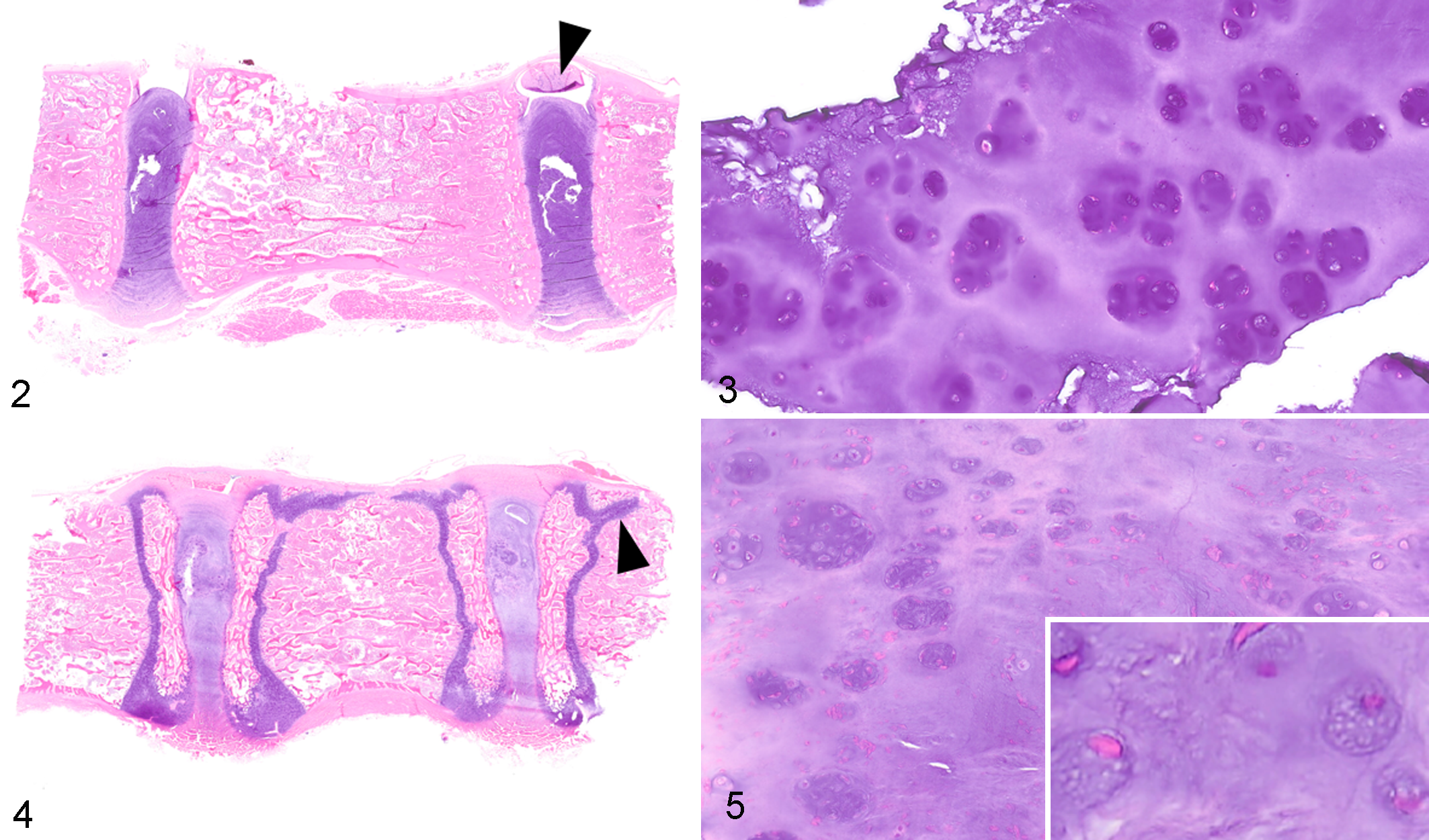
Intervertebral disc (IVD) chondrodystrophy, adjacent vertebral bodies and intervertebral discs, dog.
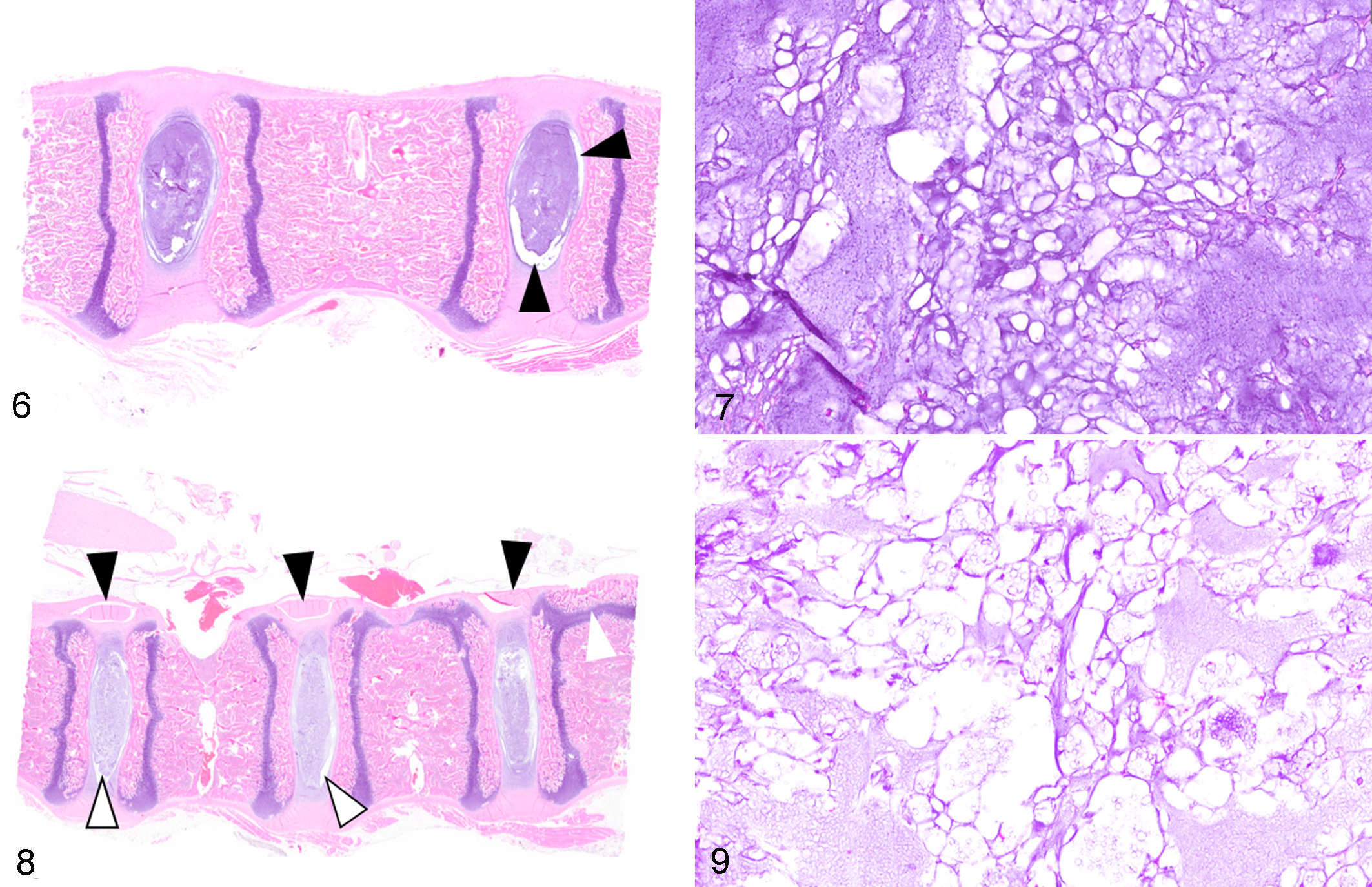
Normal control tissue, adjacent vertebral bodies and intervertebral discs, dog.
The long bones of the forelimbs of dog No. 1 had a normal radiographic appearance with no evidence of radial angulation in a frontal or sagittal plane, no evidence of carpal or elbow joint subluxation, and no evidence of carpal or elbow osteoarthritis. The computed tomography (CT) appearance of the forelimbs was also normal, without evidence of angulation or osteoarthritis. On the CT of the spine, intervertebral disc mineralization was visible at T1–T2 and T12–13 and was faintly visible at T7–T8 and T8–T9. The long bones of the forelimbs of dog No. 2 had a normal radiographic appearance: premature physeal closure, radial angulation in a frontal or sagittal plane, retained cartilaginous cores, and carpal or elbow joint subluxation were not observed.
Histopathology
Features and scores of the 2 grading schemes for dog Nos. 1 to 4 are tabulated in Table 1. Histological changes were similar for dog Nos. 1 and 2. In all examined discs, the NP was largely replaced by cartilage and was continuous with the surrounding AF (Figs. 2–5). The cells within the NP consisted predominantly of chondrocytes, arranged as individual cells or aggregated into clusters of 8 or more cells (clones), and associated with dark purple chondroid matrix. Normal physaliferous notochordal cells were either not identified in the examined discs or were rarely identified, consistent with a chondrification index of 4 (Suppl. File S1). 5 In Alcian blue–stained sections, the matrix of the NP stained diffusely dark blue while the matrix remained essentially unstained with Picro-Sirius red (components of the Bergknut histological grading scheme, Suppl. File S2). In the AF, approximately half of the mesenchymal cells exhibited chondrocyte metaplasia, but tears and clefts within the AF were not identified. The adjacent endplates of the vertebral bodies were considered histologically normal for all examined sections. Mineralization of the IVD was not detected in any of these demineralized sections. In dog No. 2, no overt morphologic changes were identified within the physis or metaphysis of the distal radius (data not shown).
In all examined sections of control dog Nos. 3 and 4, the NP was relatively circumscribed by a discontinuous cleft separating the NP from the surrounding AF and the cranial/caudal endplates of the vertebral bodies (Figs. 6, 8). In dog Nos. 3 and 4, the NP comprised numerous, variably preserved physaliferous notochordal cells with clear to vacuolated cytoplasm and round, polygonal to stellate morphology (Figs. 7, 9) consistent with a chondrification index of 1 (considered normal, Suppl. File S1). 5 These cells formed central clusters amid a loose, basophilic, often fragmented matrix. As with dog Nos. 1 and 2, in Alcian blue–stained sections, the matrix of the NP stained diffusely dark blue with no Picro-Sirius red staining. The majority of the mesenchymal cells of the AF had a normal spindloid, fibroblast-like appearance (lack of chondrocyte metaplasia), and vertebral endplates were histologically normal.
Discussion
We have described and compared the gross, radiographic, and histologic features of IVD degeneration in a group of young dogs from a single breed with known presence and absence of the CFA12 FGF4 retrogene and absence of the CFA18 FGF4 retrogene. Although the 2 affected individuals were of different ages, the histological changes in the IVD were similar in both animals. Neither dog had replacement of the NP with scar tissue, changes in the morphology of the AF, changes in the endplate morphology, or other degenerative features in the AF like tears and clefts, suggesting that some of the features of intervertebral disc degeneration may require multiple years to manifest. The chondrodystrophic degenerative phenotype is evident in the IVD as early as 10 weeks of age in dogs homozygous for the CFA12 FGF4 retrogene but is absent in breed-matched dogs lacking the mutation. These degenerative changes are similar to the detailed descriptions of degenerative “chondroid metamorphosis” in several chondrodystrophic dog breeds originally documented by Hansen 13 and those described in the Beagle and Dachshund breeds by Braund et al. 5 Based on known genotyping data, 1,7 the dog breeds studied by Hansen 13 and Braund et al 5 (particularly Dachshunds and Beagles) would have had a high likelihood of carrying the CFA12 FGF4 retrogene due to the limited segregation of the allele within those breeds (allele frequencies of 0.99 and 1.0, respectively, based on UC Davis data). While Dachshunds also have a very high allele frequency for CFA18 FGF4 retrogene (0.99), Beagles do not appear to carry the CFA18 FGF4 retrogene. 1,7 The current parallel findings in CFA12-homozygous/CFA18-normal, breed-controlled NSDTR dogs, together with previous data from CFA12-heterozygous dogs with intervertebral disc disease (IVDD), 1,7 suggest that the presence of the CFA12 FGF4 retrogene alone is sufficient to cause the chondrodystrophic phenotype, resulting in premature chondroid degeneration of the intervertebral disc.
Hansen’s pioneering studies 12,13 of canine IVDD established the currently used classification of IVDD. Type I IVDD is an acute extrusion of disc material, predominantly in younger, chondrodystrophic, small breed dogs, while type II disease is a more chronic protrusion of discs seen predominantly in older larger breed (nonchondrodystrophic) dogs. However, defining specific populations of dogs within IVDD studies can be challenging. More recent studies have suggested that type I and type II disease may have a common underlying pathology, 14,18 although it is likely that specific genetic mechanisms can vary. In addition, studies have historically been confounded by the difficulty in accurately defining what constitutes a chondrodystrophic dog. Publications evaluating clinical differences between chondrodystrophic and nonchondrodystrophic dogs do not have consensus on which breeds fall into which category 3,4,11,14,18,26 or even how the classification into the 2 categories should be performed. 20,25,31 Classification based on gross morphological traits is problematic since a short-limbed phenotype may be associated with the CFA18 FGF4 retrogene or the CFA12 FGF4 retrogene, and only the latter appears to also result in the chondrodystrophic phenotype with concurrent degeneration of the IVD. 1,7 The presence of the CFA12 and CFA18 retrogenes is variable among many small dog breeds, and many breeds segregate both CFA12 and CFA18 FGF4 retrogenes. 1,7 Also, a single copy of the CFA12 FGF4 retrogene appears to be sufficient to cause IVD degeneration, but effects on limb length appear to be related to gene dose, making “longer legged” heterozygous animals more challenging to define as chondrodystrophic. 7 Defining CFA12 FGF4 retrogene status in future studies is likely to clarify some of these issues, but the heterogeneity of IVDD phenotypes makes it likely that additional genetic factors are contributing to disease presentation.
It is well documented that anatomic location of disease, number of clinically affected discs, and age of onset differ between type I and type II IVDD. 11,13 However, disease presentation within the “chondrodystrophic” breeds can also vary in the age of onset, with French bulldogs having an earlier onset and commonly affected breeds such as Dachshunds having a later onset. 1,22 This likely reflects additional genetic modifiers that compound the CFA12 FGF4 retrogene-related pathology or genetic selection pressures within breeds (eg, Dachshunds) that are essentially fixed for the CFA12 FGF4 retrogene. In addition, premature chondroid degeneration and mineralization of IVDs can be seen, albeit less frequently, in nonchondrodystrophic breeds 8,9 and specifically in those that do not carry the CFA12 FGF4 retrogene. 1 IVDD in these animals may result from alternative mechanisms affecting the same FGF-related biology of the IVD (in the absence of limb length alterations) or may represent novel pathway alterations resulting in a common phenotype.
Disproportionate dwarf dog breeds, like the Dachshund, were selectively bred to achieve that specific phenotypic outcome. The associated premature degeneration of the IVD can be considered an “off-target” effect of that selective breeding process. In the FGF4-associated chondrodystrophic canine disc, the types of cells and matrix integrity of the NP become compromised at a young age. For dogs with chondrodystrophy, the biologically unique physaliferous notochordal cells are rapidly replaced by cells with a chondrocytic phenotype. Bergknut et al 2 found that for their IVD histological grading scheme, the average histological score was negatively correlated with IVD GAG content. That is, the GAG content of the NP decreases as the histological score increases (as the disc degeneration becomes more severe). Further study of the notochordal cells and the events that trigger their transformation into or replacement by chondrocytic cells may provide useful insights into disc degeneration and potential means of managing the damaged disc. 17 The molecular mechanism(s) connecting the genotype with this pathologic phenotype of IVD chondrodystrophy awaits resolution.
Currently, the molecular, physiologic, and pathologic connection between the FGF4 genotype and short body stature (dwarf phenotype) or abnormal development of the IVD is incompletely understood. Fibroblast growth factors are involved in a multitude of developmental processes, including proliferation, survival, migration, and differentiation. 6,19 FGF4 provides mitogenic and morphogenic signals to regulate normal limb development, 27 and activation of the FGF3 receptor (FGFR3) is critical for inhibition of chondrocyte hypertrophic differentiation and proliferation during postnatal endochondral ossification. 6 An activating mutation in FGFR3 is responsible for >95% of achondroplasia cases, the most common form of dwarfism in humans, 29 and FGF4 signals through several FGF receptors, including FGFR3, 23 providing a possible mechanism for growth plate abnormalities in dogs with chondrodystrophy and chondrodysplasia.
Interestingly, careful histological examination of 3 sections of the distal radial physis of dog No. 2 did not reveal any overt morphologic changes of the physis or metaphysis. The cranial and caudal physes of the vertebral bodies were also histologically indistinguishable between affected (dog Nos. 1 and 2) and unaffected animals (dog Nos. 3 and 4). In contrast to our findings, Braund et al 5 identified morphologic differences between newborn chondrodystrophic dogs (Beagles) and newborn nonchondrodystrophic dogs (Greyhounds) within the distal femoral physes. Specifically, Braund et al 5 identified poor cellular delineation in the germinal and proliferative zones of the physis, reduced tendency to form chondrocyte columns in the proliferative zone, narrow and irregular chondrocytes in the maturation zone, and retarded trabecular formation within the zone of ossification. None of these pathologic features were identified in the physes of the dogs studied here. This histological discrepancy may be the result of a difference in the age of the dogs or anatomic location. Perhaps the impact of the FGF4 retrogene insertion on dwarfism is to alter the rate or timing of normal endochondral ossification.
Canine IVDD is a complex and likely multifactorial disease. The current study is limited by small numbers and a lack of gene dosage-related data for dogs heterozygous for the CFA12 FGF4 retrogene. However, documenting histopathologically defined IVDD in CFA18 FGF4-negative, CFA12 FGF4 retrogene-carrying dogs compared to breed-matched controls further supports the pivotal role for the CFA12 FGF4 retrogene in the pathogenesis of IVDD in dogs. As discussed above, insertion of the CFA12 FGF4 retrogene is unlikely to be the only mechanism for premature degeneration of the IVD in dogs, and disease modification through additional genetic factors is possible. However, the mounting evidence for CFA12 FGF4 retrogene effects on intervertebral disc degeneration supports the further investigation of similar mechanisms across other species and selective breeding to reduce disease incidence in segregating dog breeds.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819868731 - Pathologic Features of the Intervertebral Disc in Young Nova Scotia Duck Tolling Retrievers Confirms Chondrodystrophy Degenerative Phenotype Associated With Genotype
Supplemental Material, DS1_VET_10.1177_0300985819868731 for Pathologic Features of the Intervertebral Disc in Young Nova Scotia Duck Tolling Retrievers Confirms Chondrodystrophy Degenerative Phenotype Associated With Genotype by Brian G. Murphy, Peter Dickinson, Denis J. Marcellin-Little, Kevin Batcher, Stephen Raverty and Danika Bannasch in Veterinary Pathology
Footnotes
Acknowledgements
We thank the owners of the dogs used in this study. We are grateful for the efforts of Matt Nichols, Dr Mai Mok, and Dr Devinn Sinnott for their assistance with the necropsy of dog Nos. 1 and 3. We are also grateful for Tanya Garcia for grossly sectioning the radius of dog No. 2 and for the histotechnology expertise of the UC Davis Veterinary Histopathology Laboratory.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this research was provided through the Maxine Adler Endowed Chair funds and other faculty discretionary funds.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
