Abstract
The changes associated with condemned lungs in cattle with chronic pleural lesions of the caudal lobes were characterized by histology and immunohistochemistry (IHC). Fibroproliferative pleural lesions were microscopically confirmed. Occasionally, the pleural lesions also included adipose, chondroid, and osseous metaplasia that were covered by mesothelial cells, mostly in the absence of inflammation. Other lungs also showed fibrosis in the subpleural interstitium and interlobular septa. In both condemned and noncondemned lungs, immunoreactivity to Wilms tumor 1 (WT1) was normally observed on surface mesothelial cells but not on the submesothelial fibroblasts and myofibroblasts. Conversely, the myofibroblasts beneath the pleura, but not the mesothelial cells, showed immunoreactivity to alpha smooth muscle actin and calponin. However, in the lungs with myofibroblastic foci in the pleura, the proliferated cells maintained WT1 immunoreactivity similar to those of some metaplastic cells. These findings may reflect the plasticity of mesothelial cells in vivo.
Keywords
Bovine respiratory disease complex (BRDC) is the leading cause of morbidity and mortality in feedlot cattle. 7 The typical pattern of BRDC-related pneumonia is fibrinosuppurative bronchopneumonia. 16 There is a trend for cases of bronchopneumonia in feedlot cattle to have longer courses, and chronic lesions are more frequently observed than 30 years ago. 7 Therefore, chronic bronchopneumonia and its sequelae could be responsible for many lung condemnations in abattoirs. Nonetheless, in 2 consecutive surveys in abattoirs in Monterrey, Mexico (2014–2016), pleural lesions not related to BRDC were the main cause of lung condemnation. Herein, we characterized the pleural and parenchymal changes.
This study was performed in 2016 in a government-certified abattoir (TIF 356, SAGARPA-SENASICA) in Monterrey, Mexico. This abattoir specializes in cattle fattened in pens. All procedures adhered to the official norms (Official Mexican Norm NOM-009-ZOO-1994 Sanitary Processes of Meat and NOM-033-ZOO-1995 Humane Slaughter of Wild and Domestic Animals).
Previously, an official veterinarian had confirmed that the animals were in good health based on antemortem inspection. The veterinary inspector was unaware of the study and had full decision-making power in condemning or approving the reviewed lungs. The condemned lungs were used as our study material. A representative survey of 389 bovines, including 57 condemned lungs, was statistically convenient (Epitools 17 ). The lungs were carefully examined, palpated, recorded, and photographed. Based on the gross appearance, the lesions were divided into 2 main groups: chronic bronchopneumonia and chronic pleural lesions. Other causes of condemnation were excluded. Ten noncondemned lungs from fattened cattle were included as controls.
From all cases and controls, representative samples were obtained from the right lung, including 4 samples from the caudal lobe (2 from the dorsal surface and 2 from the ventral edge), 1 from the middle lobe, and 2 each from the cranial and caudal parts of the cranial lobe. In addition to the systematic sampling procedure, samples were collected from small, hard, and depressed circular areas with a range between 3- and 15-mm diameter, recognized in several lungs. All samples were collected at a depth that included the subjacent parenchyma. In a subsequent sampling in lungs condemned with pleural lesions (8/100 = 0.08), central samples in caudal and cranial lobes were also included. These samples were fixed in 10% neutral buffered formalin, processed routinely, sectioned (4 µm), and stained with hematoxylin and eosin (HE) and Masson’s trichrome. Additional sections were labeled by immunohistochemistry (IHC) employing the 2-step system EnVision™ FLEX on an automated platform (Autostainer Link 48) according to the manufacturer’s instructions (Agilent Technologies Dako, Comercial Biomédico JR, Monterrey, Nuevo Leon, Mexico). The primary antibodies included mouse anti-human Wilms tumor protein (WT1, clone 6F-H2 prediluted; Dako), which reacts with the amino-terminal amino acids of all the complete isoforms of WT1 and was intended as a marker of mesothelial cells; mouse anti-human alpha smooth muscle actin (α-SMA; clone 1A4 prediluted; Dako), which is specific to amino-terminal amino acids of human α-SMA with a cross-reaction in cattle and was used as a marker of myofibroblasts and smooth muscle cells; and mouse anti-human calponin (CALP; clone CALP 1:100 dilution; Bio Care Medical, Pacheco, California, USA), which reacts with the isoform 34kD and was intended as a marker of smooth muscle cells, myoepithelium, and myofibroblasts. The IHC procedure included a streptavidin-biotin complex with diaminobenzidine as the substrate (Dako; Comercial Biomédico), followed by slight counterstaining by hematoxylin. The negative controls were lymphoid tissue in all 3 assays, and the positive controls were fallopian tube, colon, and mammary gland tissue. Selected lung samples were fixed in 2.5% buffered glutaraldehyde and subsequently in osmium tetroxide, embedded in Epon, and sectioned on an ultramicrotome to obtain semithin (≈0.8-µm) sections. These sections were stained with toluidine blue and routinely mounted with a coverslip for observation under a light microscope.
Of 389 cattle inspected during the period of the study, there were 57 condemned lungs (57/389, 14.6%; 95% confidence interval [CI], 11.5%–18.5%). Among the condemned lungs, the prevalence of typical bronchopneumonia (7/57, 12.3%; 95% CI, 6.1%–23.2%) was lower than the prevalence of pleural lesions (50/57, 87.7%; 95% CI, 76.7%–93.9%). Most lungs with pleural lesions lacked cranioventral consolidation of lung tissue, with a few (8/50) exhibiting minimal consolidation. Chi-square (uncorrected) analysis showed highly significant differences (P < .0001) between the groups, 17 indicating that grossly, the chronic caudal pleural lesions and chronic bronchopneumonia were found as separate lesions. Subsequently, the study exclusively focused on the lungs that were condemned because of pleural lesions.
Lungs condemned for pleuritis were partially collapsed and showed combinations of 3 distinctive lesions: locally extensive villous excrescences formed by proliferation of pleural connective tissue, depressed foci of pleural metaplasia, and extensive fibrosis of the pleura and subjacent interlobular septa. The gross, histologic, and IHC findings in each of these are described as follows. First, all 50 lungs condemned for pleuritis had soft hyperemic veils of pleural villous excrescences at the edges of the caudal lobes, or more thickened plaques that were irregularly attached to the dorsal surface but not over the cranial lobes (Figs. 1 and 2). The caudal pleural lesions were not identified in lungs condemned for bronchopneumonia or other reasons. These caudal pleural lesions were located only on the visceral pleura and not on the parietal pleura. Microscopically, these villous excrescences corresponded to extensive deposits of collagen accompanied by newly formed blood vessels and infiltrating lymphocytes and plasma cells, and were covered by mesothelial cells (Fig. 1, inset). Whereas the controls had WT1 immunoreactivity only in mesothelial cells (Fig. 3), WT1 immunoreactivity in the villous lesions of condemned lungs was present in surface mesothelial cells and in cells within the proliferative tissue (Fig. 4). α-SMA immunoreactivity in control lungs was present in small blood vessels and rare subpleural myofibroblasts but not in surface mesothelial cells, whereas α-SMA immunoreactivity was present in numerous cells within the villous lesions of condemned lungs (Fig. 5). Similar but less intense calponin immunoreactivity was present.
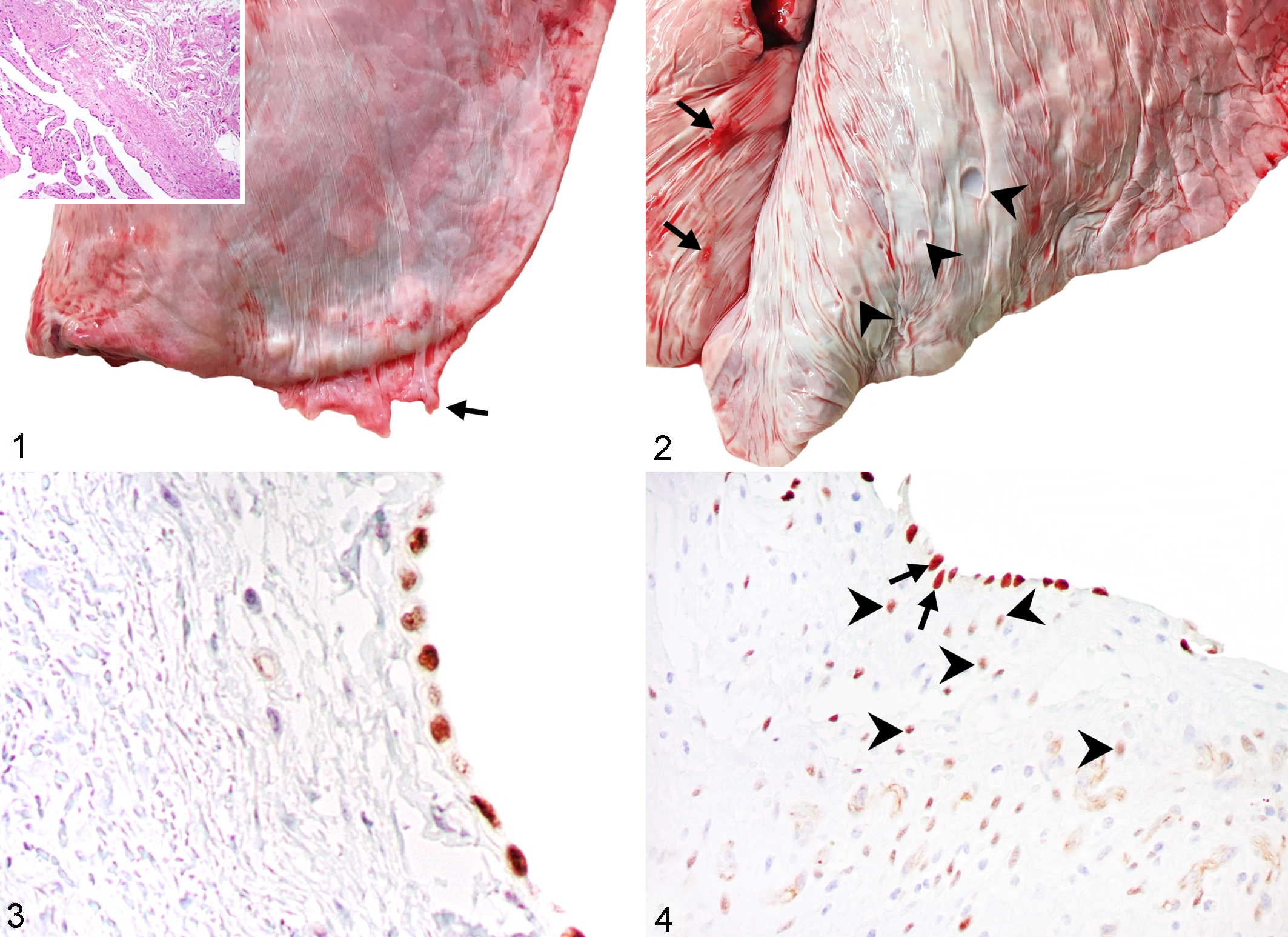
Chronic caudal pleural lesions, lung, cattle.
Pleural myofibroblastic foci and metaplasia, lung pleura, cattle.
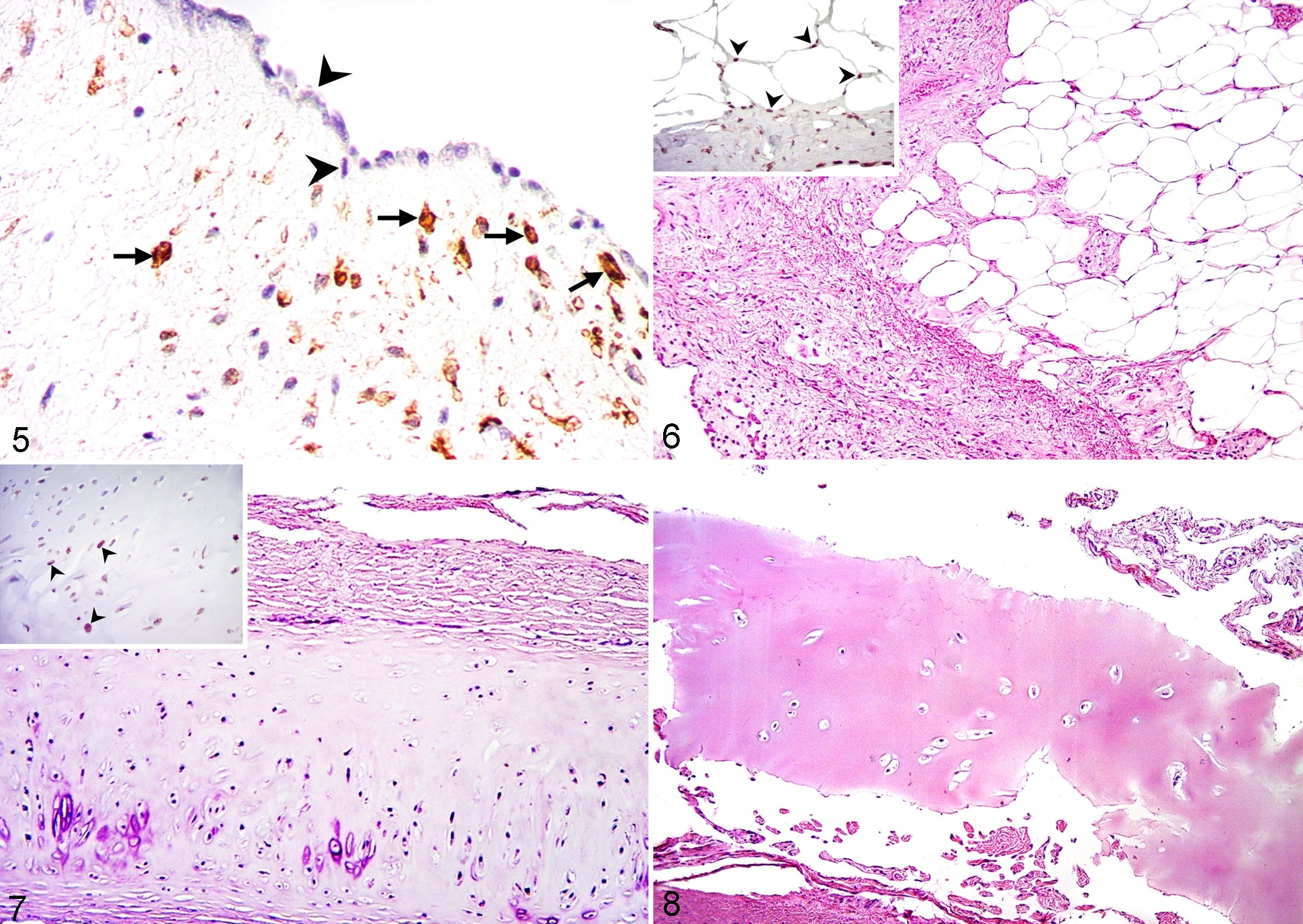
Second, several lungs had hard and brittle circular depressions on the pleura (Fig. 2). The depressions were present in 31 of the 50 lungs condemned for pleuritis (62%; 95% CI, 48.1%–74.1%). Microscopically, these corresponded to zones of pleural metaplasia that were subjacent to the above-described proliferative lesions. They always contained adipose tissue (100%) (Fig. 6), and some (14/31, 45.1%; 95% CI, 29.1%–62.2%) also contained hyaline cartilage (Fig. 7) and bone tissue (2/31, 6.4%; 95% CI, 1.7%–20.7%) (Fig. 8), but there were minimal inflammatory changes. In these lesions, immunoreactivity to WT1 occurred in the surface mesothelium and occasionally in the metaplastic tissue (Figs. 6 and 7 insets).
Third, extensive fibrosis of the pleura and subjacent interlobular septa was observed microscopically in 9 of the 50 lungs condemned for pleuritis (18%; 95% CI, 9.7%–30.8%). These pleural lesions were focally extensive and extensively distorted the lung tissue (Figs. 6 and Supplemental Fig. S1). These lesions did not involve the alveoli or alveolar septa of the lung parenchyma, but the epithelium of bronchioles frequently showed squamous metaplasia (Fig. S2). The subsequent sampling in lungs condemned because of pleuritis confirmed that the deeper lung parenchyma was not affected. Moreover, numerous degranulated mast cells were present (Figs. S3 and S4). Within the lesions of pleural and interlobular fibrosis, calponin immunolabeling was present in proliferated and hypertrophied myoblastic cells in the tunica intima of small veins and highlighted the arterial and venous smooth muscle hypertrophy (Figs. S5–S8).
In this study, pleural lesions were the main cause of lung condemnation. All affected lungs were from apparently healthy animals approved for slaughter. The findings of pleural metaplasia and subjacent chronic interstitial fibrosis were unexpected. Identical pleural lesions have been observed in routinely slaughtered beef cattle, 4 but pleural metaplasia was not reported.
Mesothelial cells have the potential to differentiate into myofibroblasts. 1,10,13,15 The in vitro plasticity of mesothelial cells is broad and includes specialized cell lineages, such as adipocytes, chondrocytes, and osteoblasts. 1,11,13,15 The clonal proliferation of the mesothelial-derived myofibroblasts is influenced in a restrictive manner by WT1. 9,10,15,18 WT1 maintains the epithelial phenotype in the pleura, and its downregulation favors myofibroblastic transdifferentiation. Transdifferentiation is a prerequisite for mesothelial cells to traffic into the subjacent lung parenchyma. 9,10,15,18 The WT1 immunoreactivity on the mesothelial surface and the positive reaction in subpleural myofibroblastic foci identified here are similar to that described in human idiopathic pulmonary fibrosis (IPF). 9,18 Furthermore, the WT1 immunoreactivity in the periphery of metaplastic tissues is compatible with their mesothelial origin, suggesting the potential of mesothelial cells to differentiate in adipose and cartilage tissues in vivo. Previous studies have also shown peritoneal cartilaginous metaplasia in human patients after surgical procedures in the abdomen. 5
Endothelial cells can also undergo myofibroblastic transdifferentiation, as occurs in lung fibrosis and pulmonary hypertension. 6,8 In fact, pulmonary hypertension and muscularization of small arteries and veins commonly occur in hypertensive lung disease and have been reproduced in animal models of lung fibrosis. 8
Alternatively, a recent morphometric analysis identified that myofibroblastic foci in fibrotic lung disease forms a reticulum complex that extends from the pleura to the subjacent parenchyma. 3 Therefore, episodes of insidious damage related to peripheral (subpleural) tractional forces could explain the caudal pattern of the lung lesions in this study, similar to changes in basal lobes in some fibrotic lung diseases in humans, and might reflect the naturally thick pleura in cattle and humans. 12,14
In our study, inflammatory cells were uncommon, except for degranulating mast cells. Their significance in these lesions of cattle is not known, although mast cells contribute to abnormal remodeling in pulmonary fibrosis. 2
This study describes proliferative, fibrotic, and metaplastic lesions in the caudal pleura of cattle, which are a frequent cause of lung condemnation. We propose that the metaplastic tissues originated from transdifferentiated mesothelial cells.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819837719 - Fat, Cartilage, and Bone Metaplasia in Lungs of Cattle With Caudal Pleural Lesions and Subjacent Interstitial Fibrosis
Supplemental Material, DS1_VET_10.1177_0300985819837719 for Fat, Cartilage, and Bone Metaplasia in Lungs of Cattle With Caudal Pleural Lesions and Subjacent Interstitial Fibrosis by Cecilia Ramírez-Hernández, Luis Jorge García-Márquez, Horacio Decanini-Arcaute, Julio Martínez-Burnes and Rafael Ramírez-Romero in Veterinary Pathology
Footnotes
Acknowledgments
We express our sincere gratitude to the personnel of the Anatomía Patológica Department, Hospital Universitario, Universidad Autónoma de Nuevo León, for their great support in performing all of the techniques employed herein.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: SEP, México; PRODEP, Research Network on Wildlife Diseases. Project: 2013; extension 2016-2017.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
