Abstract
A 5-month-old, female, Aberdeen Angus heifer presented to the veterinary medical teaching hospital for evaluation of slowly progressive hindlimb ataxia. The calf was clinically normal until 4 months of age, following routine pregnancy and delivery. Neurologic examination revealed marked symmetric spastic hindlimb paraparesis. Thoracolumbar radiographs and cerebrospinal fluid (CSF) analysis were unremarkable. A presumptive diagnosis of T3-L3 myelopathy was made, and neurologic status remained static for 3 months with broad-spectrum antibiotic and nonsteroidal anti-inflammatory therapy. Additional diagnostic tests were refused, and a necropsy was performed following euthanasia. A moderately well delineated, reddish-tan, soft mass 18 mm in diameter replaced 80% of the fourth lumbar spinal cord segment. Histologic examination revealed two distinct features: undifferentiated, primitive, polygonal-to-round cells with typical morphologic characteristics of primitive neuroectoderm; and interspersed areas containing myelinated axons and cells with neuronal differentiation. immunohistochemical examination confirmed the presence of primitive neuroepithelium and cells with neuronal differentiation.
The most current World Health Organization histologic classification defines neuroblastoma as an embryonal neuroepithelial neoplasm with limited neuronal differentiation. 13 It is one of the group of primitive neuroectodermal tumors of which medulloblastoma is the prototype, and includes ependymoblastoma, thoracolumbar spinal cord tumor of young dogs and pineoblastomas. The primitive neuroectodermal tumors (PNET) family of neoplasms, are composed of primitive neuroepithelial cells with a common embryonic morphologic appearance putatively originating from a multipotential stem cell. 9 Neuroblastoma in people occurs in infants and children, is rarely seen in persons more than 10 years of age, and is slightly more common in males. It is the most common cancer in infants and the third most common type of cancer in children. Sites of occurrence are equally divided between the adrenal glands, the sympathetic ganglia of the abdomen, and the sympathetic ganglia of the chest or neck or the parasympathetic ganglia of the pelvis. Primary spinal cord origin is possible. 2 Although rare, reports in animals exist, most commonly in young cattle and dogs with the adrenal gland most frequently the site of origin. 8, 10, 15– 19, 22
This current report describes the morphologic characteristics and immunohistochemical-staining pattern of a case of spinal cord neuroblastoma with areas of divergent neuronal differentiation in a calf.
A 5-month-old, Aberdeen Angus heifer calf was presented for evaluation of slowly progressive hindlimb ataxia of approximately 1 month in duration. The heifer, an embryo transfer calf was normal until 4 months of age. The only abnormalities noted on physical examination were marked symmetric, spastic paraparesis of the hindlimbs and partial-thickness decubital sores over the lateral aspects of both tarsi. Manual assistance was necessary for the calf to stand and nurse. There was no muscle atrophy, and fecal and urinary continence were normal. Deep pain and bilateral hindlimb patellar reflexes were also normal. Thoracolumbar radiographs and lumbosacral cerebrospinal fluid analysis were unremarkable. A presumptive diagnosis of T3-L3 myelopathy was made, and neurologic status remained static for 3 months with broad-spectrum antibiotic and nonsteroidal anti-inflammatory therapy. The owner declined further diagnostic tests and therapy, and the 8-month-old heifer was humanely killed. A gross postmortem examination was performed, and tissue samples were obtained, fixed in 10% buffered formalin, and processed routinely for histopathologic examination. Segments of spinal cord were examined with hematoxylin and eosin, Luxol Fast Blue–Cresyl Violet, Luxol Fast Blue–periodic acid Schiff (PAS), Cresyl Violet, PTAH, Bodian, Holmes, and Bielschowski. Additionally, unstained, paraffin-embedded sections from the mass were processed routinely for streptavidin–biotin peroxidase immunohistochemical evaluation for S-100, glial fibrillary acidic protein (GFAP), vimentin, synaptophysin (Biogenex, San Ramon, CA), neuron specific enolase (NSE), and neurofilament (NF) (Zymed Laboratories Inc., South San Francisco, CA) expression.
The gross findings at postmortem examination were primarily restricted to the spinal cord where a moderately well-delineated, reddish-tan, soft mass, 18 mm. in diameter at the widest point replaced approximately 80% of the spinal cord within the fourth lumbar cord segment (Figs. 1, 2). The mass was grossly visible within the posterior segment of the third lumbar spinal cord segment and extended caudal within the spinal cord to its terminus at L6. Other findings were a chronic traumatic fracture of the first coccygeal vertebra and mild esophagitis.
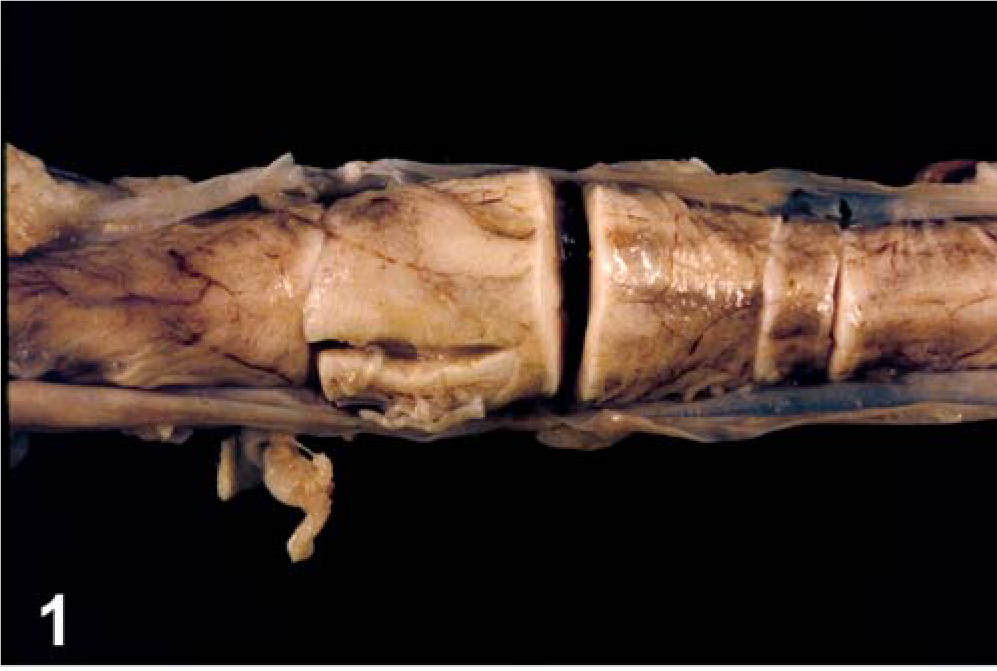
Spinal cord; calf. Pallor with swelling centered on fourth lumbar spinal cord segment.
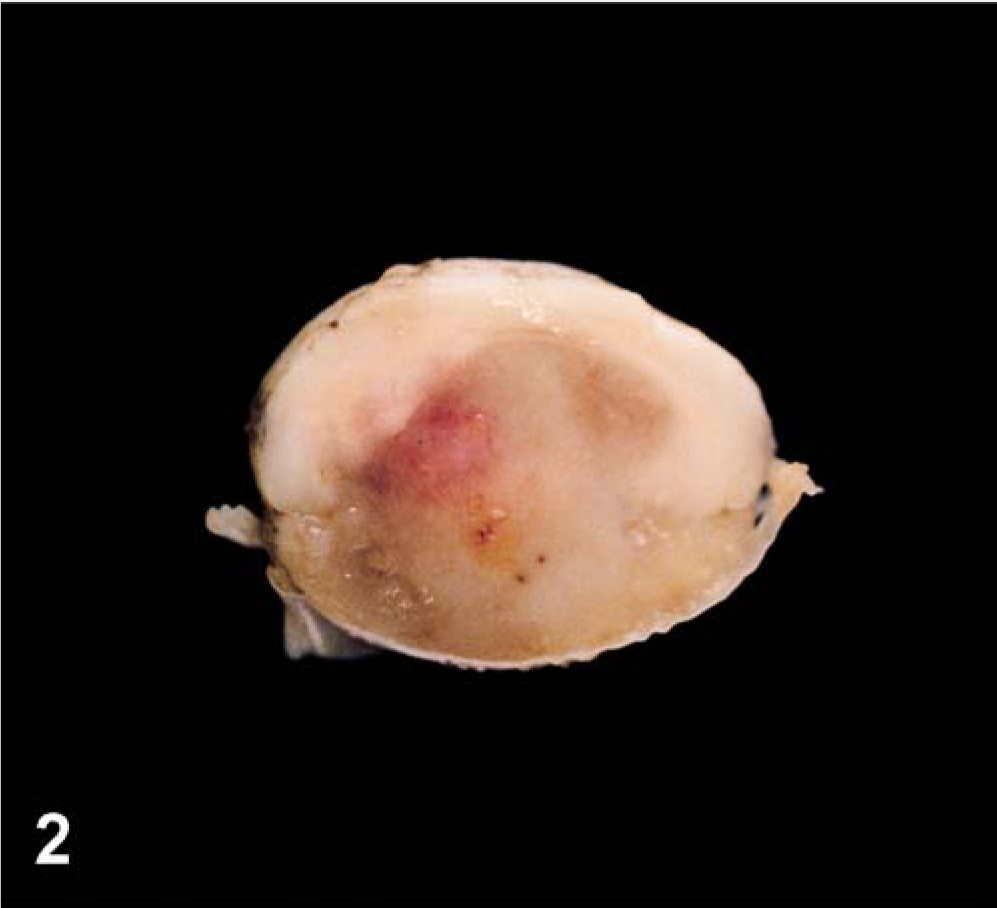
Spinal cord; calf. Transverse section through fourth lumbar spinal cord segment, illustrating moderately well-delineated ventral mass effacing most of spinal cord.
Microscopic examination revealed the mass consisted of moderately densely packed sheets of polygonal-to-round cells with moderate amounts of amorphous, fibrillar eosinophilic cytoplasm with distinct cell boundaries. These cells had moderately pleomorphic, variably sized, central or eccentric, round to slightly ovoid nuclei, with dense or finely stippled chromatin and multiple small prominent nucleoli (Fig. 3). As many as five mitotic figures were present in a high-powered field. A thin rim of PAS-positive material frequently encircled individual cells or small clusters of cells. The cells were in well-vascularized and occasionally cystic stroma with multiple expansive areas of necrosis. Neoplastic cells blended into adjacent areas of neuropil and infiltrated into the meninges, along proximal spinal nerve roots, and locally into the central canal. Histochemical stains illustrated the presence of neurons, myelin, and axons throughout the undifferentiated cell population.
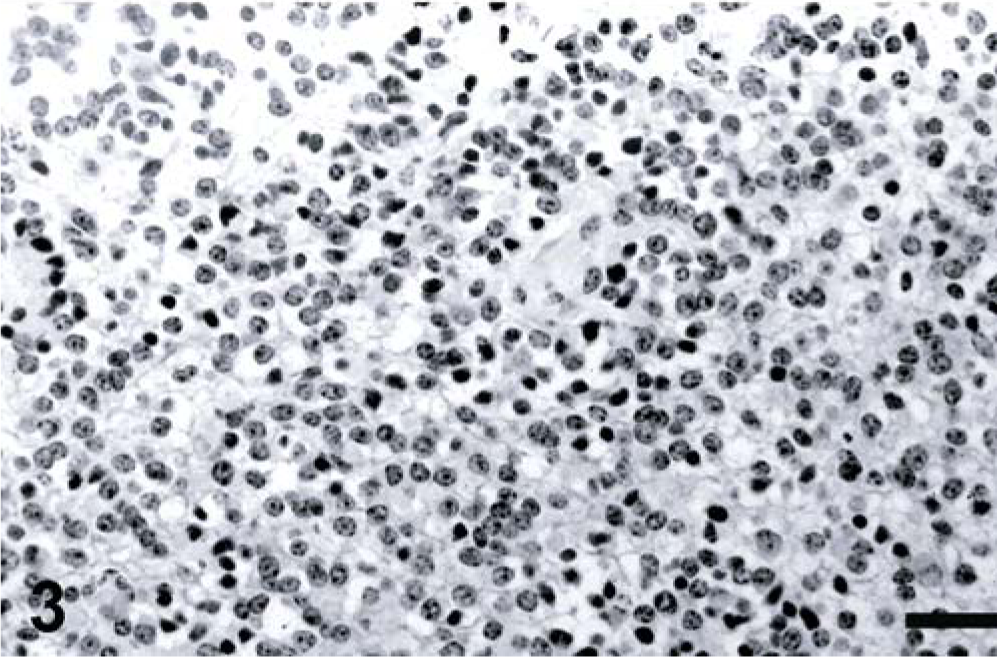
Spinal cord; calf. Histologic section from spinal cord mass with pleomorphic cellular features. HE stain. Bar = 100 µm.
Antigen expression of the neoplastic cells is shown in Table 1. The primitive neuroepithelium stained for the expression of vimentin, synaptophysin, NSE, and S-100. Expression of NF and GFAP was not present in the neoplastic cells (Figs. 4–9). Only those cells immediately adjacent to blood vessels within the tumor mass stained positively with antibody to GFAP and most likely these cells represent perivascular reactive astrocytes.
Immunohistochemical staining of tumor cells.

∗ ± = positive staining in some tumors, negative in others.
† ND = not done.
‡ Neoplastic cells did not stain. Staining occurred only in cells immediately adjacent to blood vessels.
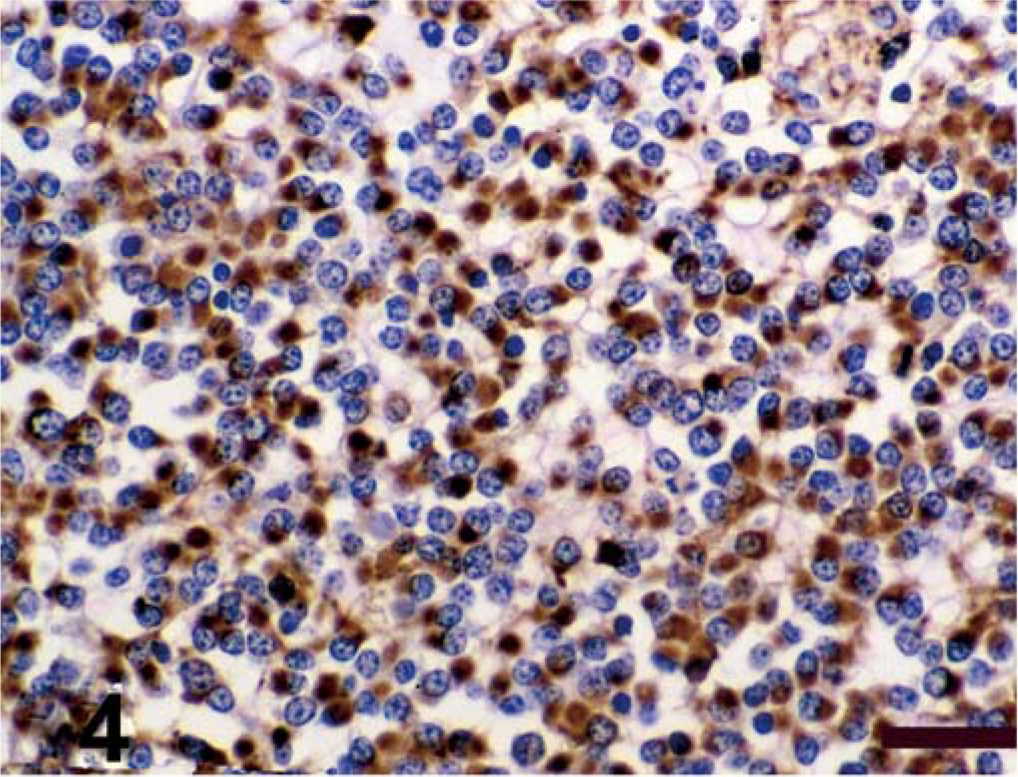
Spinal cord; calf. Antigen expression of spinal cord mass. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bars = 100 µm. Positive immunohistochemical staining for vimentin;
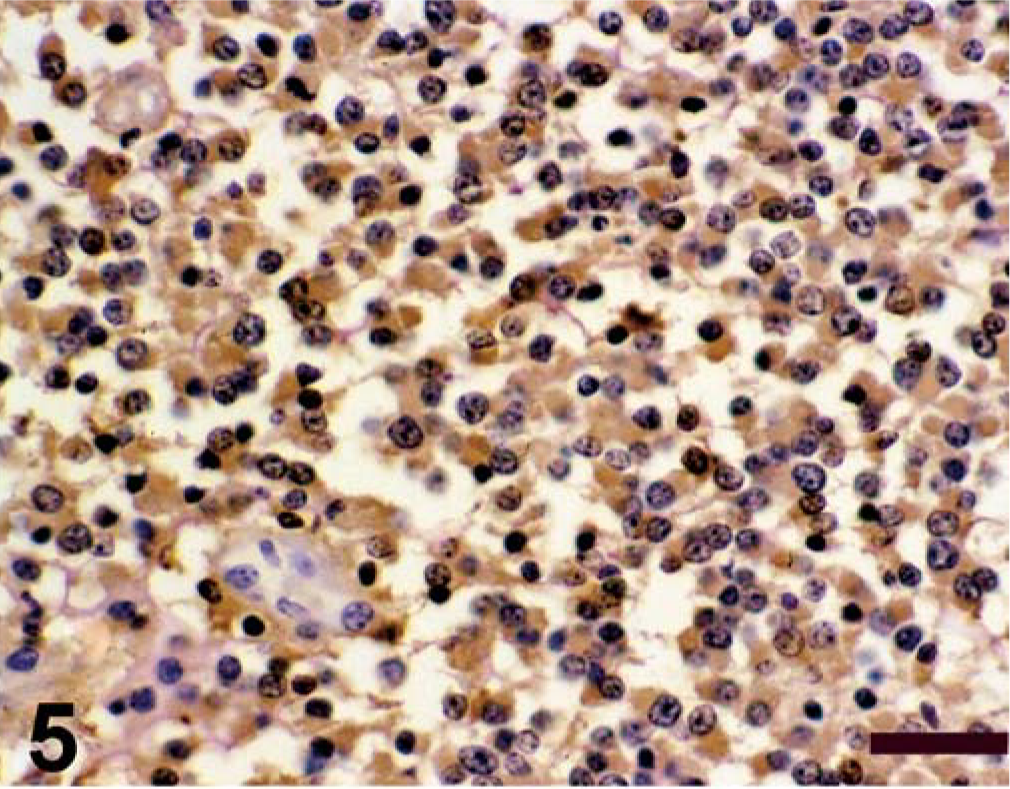
Spinal cord; calf. Antigen expression of spinal cord mass. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bars = 100 µm. Positive immunohistochemical staining for S-100;
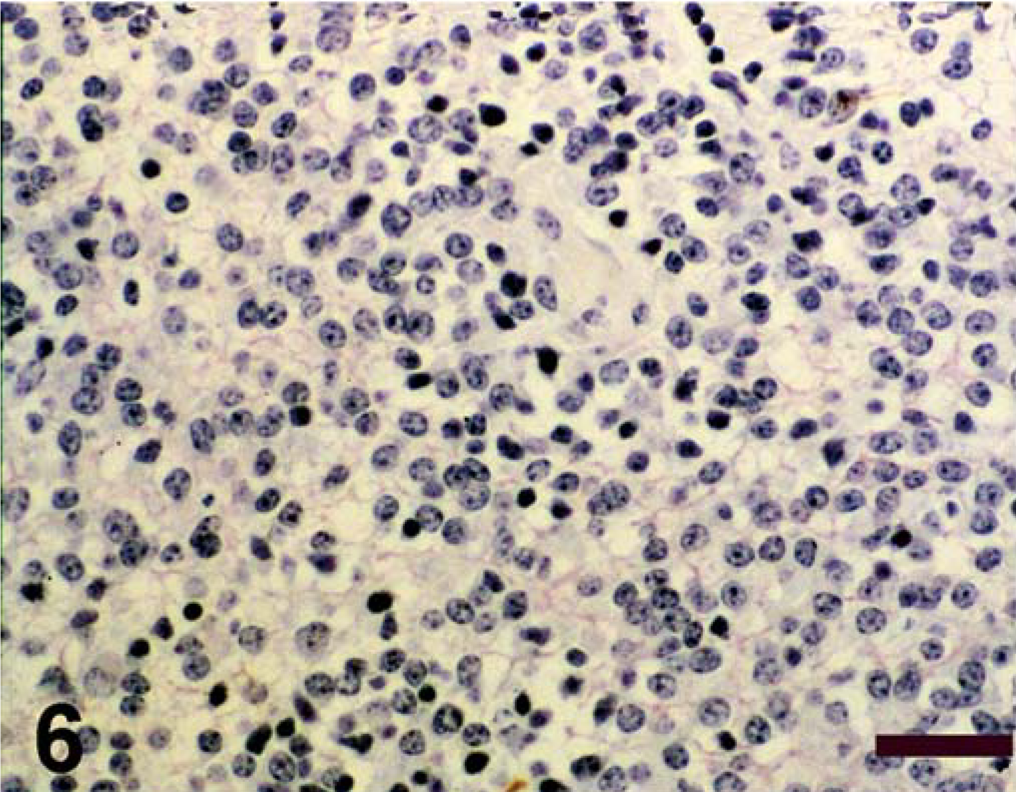
Spinal cord; calf. Antigen expression of spinal cord mass. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bars = 100 µm. Negative immunohistochemical staining for GFAP;
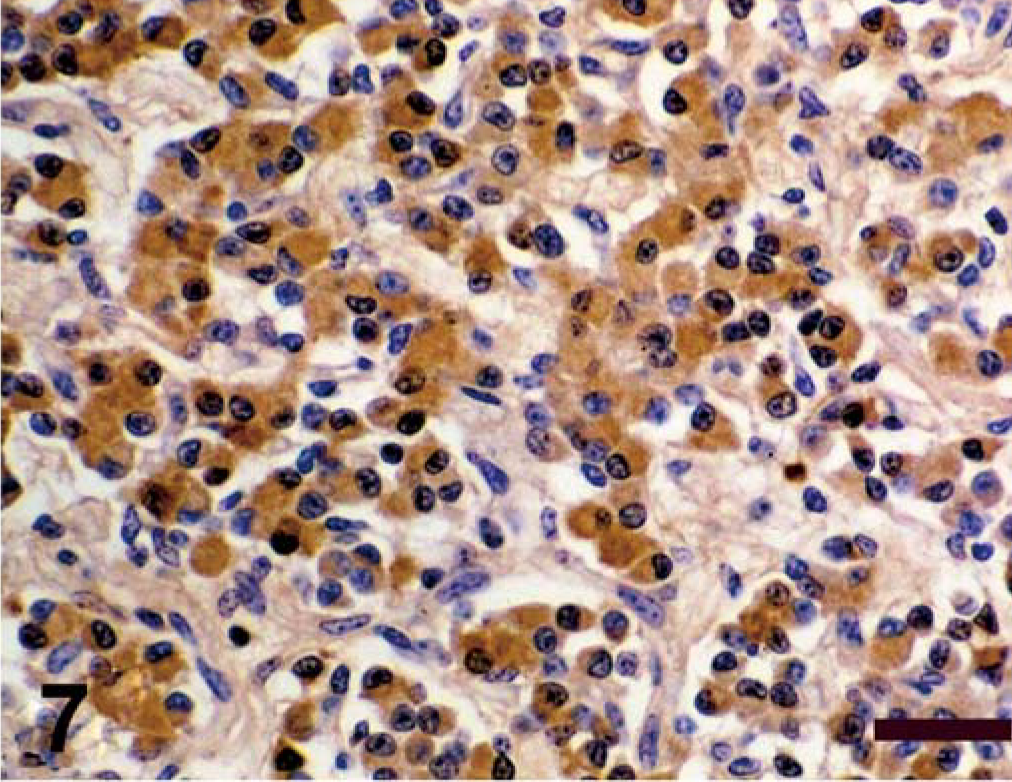
Spinal cord; calf. Antigen expression of spinal cord mass. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bars = 100 µm. Positive immunohistochemical staining for NSE;
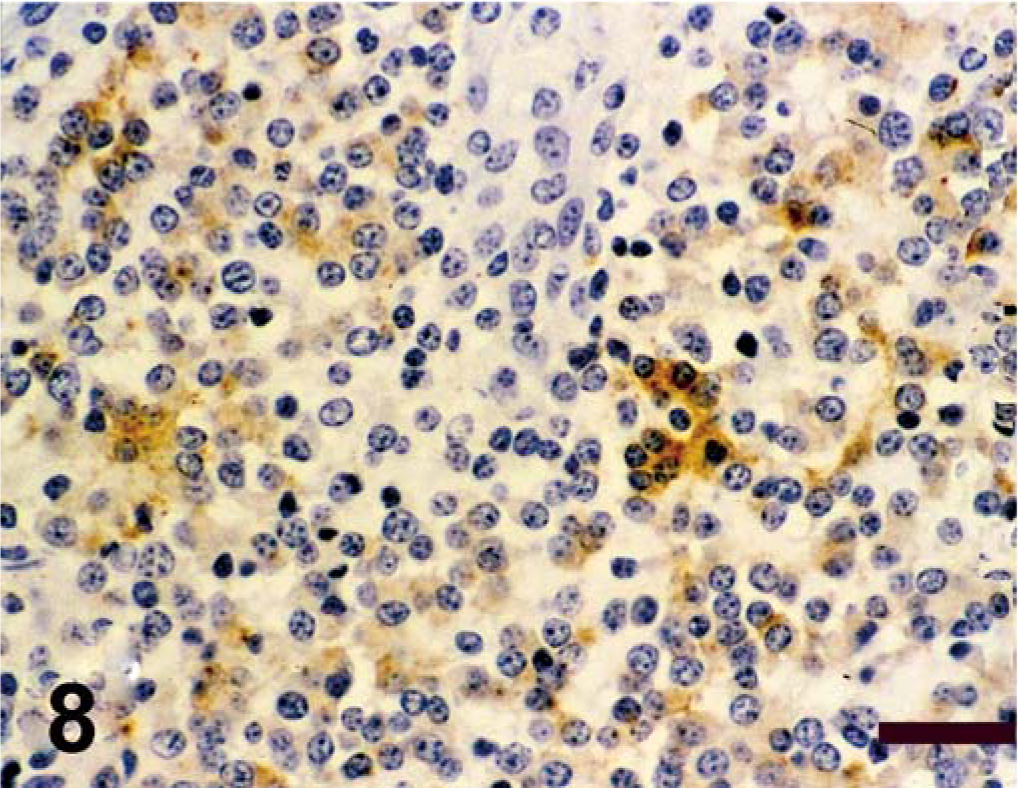
Spinal cord; calf. Antigen expression of spinal cord mass. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bars = 100 µm. Positive immunohistochemical staining for synaptophysin;
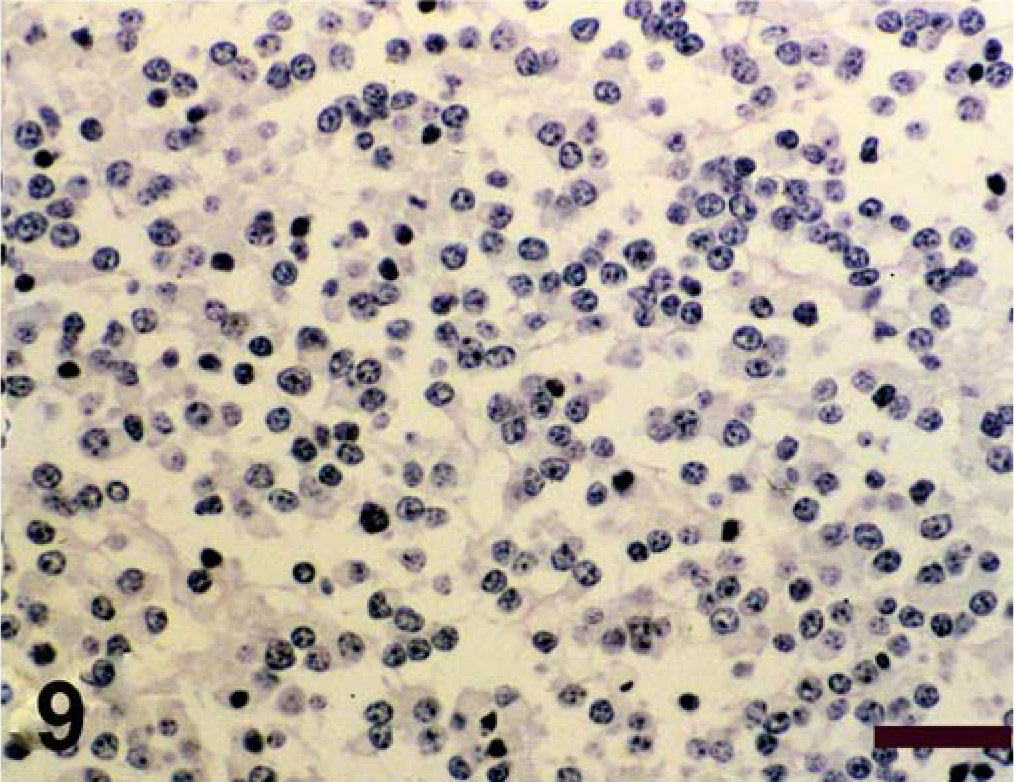
Spinal cord; calf. Antigen expression of spinal cord mass. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bars = 100 µm. Negative immunohistochemical staining for NF.
The diagnosis of neuroblastoma with neuronal differentiation was made based on the above morphologic characteristics and histochemical and immunohistochemical findings.
Aside from malignant lymphoma, neoplasia of the spinal canal or spinal cord in cattle is extremely rare. Reports describe the occurrence of tumors mostly of neural origin with diagnoses of schwannomas and neurofibrosarcoma, 11, 20 ganglioneuroma, 3 oligodendroglioma, 1 ganglioglioma, 21 and paraganglioma 12 and one report of nonneural origin, a lipoma. 5 Numerous breeds (Holstein, Hereford, Friesian, Red-and-White breed of East Flanders) and both sexes are affected. Age at diagnosis ranges from 4 months to 8 years.
Whereas neuroblastoma is the most common PNET in infants and children, 2 medulloblastomas are the most common PNET in animals, primarily of young cattle and puppies. 7 Neuroblastoma is rare in animals, most frequently reported in cattle and dogs with a wide age range. 8, 10, 15– 19, 22 In dogs, they can occasionally occur in the cerebrum, brain stem, and spinal cord. In cattle however, the adrenal medulla or sympathetic ganglia are the reported sites of occurrence. 14, 23
Evidence suggests that a neuroepithelial central nervous system (CNS) progenitor cell can differentiate to both neurons and astrocytes. This progenitor cell expresses nestin and vimentin; whereas synaptophysin, neurofilament proteins, and GFAP production are absent. As maturation along the neuronal cell line progresses, synaptophysin and neurofilament proteins replace nestin expression, vimentin can be variably expressed, and GFAP continues to be absent. In those cells maturing toward astrocytes, GFAP and not nestin is expressed; vimentin continues to be expressed, and neurofilament proteins are never expressed. 9 Although the antibodies used for this case (S-100, NSE, GFAP, NF, synaptophysin, and vimentin) do not necessarily exclusively stain for these cell types, they were used to help distinguish neuroepithelial progenitor cells, neuroblasts, glial cells, and neurons. 6
Very few reports of the immunohistochemical staining patterns of neuroblastomas exist in the veterinary literature. When performed on bovine samples, the results have been variable with some being NSE- or NF-positive and others negative; 10, 14, 19, 22 some synaptophysin-positive, others -negative; 14, 22 and some GFAP-, S-100-, and vimentin-negative. 19, 22 In one report where an adrenal neuroblastoma in a goat was evaluated, the findings are more consistent with much of the human literature, with NSE, synaptophysin, and vimentin all present, and NF and GFAP absent. 4 In three reports examining canine neuroblastomas, NSE was present in all, 8, 15, 16 synaptophysin and weak or rare NF-positive cells present were stained where evaluated, 8, 16 S-100 and GFAP were absent, 8, 15, 16 and vimentin was not stainable. 8 This variability either reflects the various stages of differentiation toward more mature neural elements in these examples, species differences in staining characteristics, or inappropriate diagnosis.
The current case demonstrates the occurrence of bovine neuroblastoma in the spinal cord, rather than adrenal gland or sympathetic or parasympathetic ganglia, and illustrates the ability of the cells of a bovine neuroblastoma to undergo neuronal maturation within a tumor with cells exhibiting mostly primitive morphology.
