Abstract
MicroRNAs (miRNAs) are a group of small noncoding RNAs that act as regulators of posttranslational gene/protein expression and are known to play a key role in physiological and pathological processes. The objective of our study was to compare expression of miR-21 in renal tissue from dogs affected with chronic kidney disease (CKD) caused by X-linked hereditary nephropathy (XLHN), a disease equivalent to human Alport syndrome, to that from unaffected dogs. Additionally, we sought to characterize changes in relative mRNA expression of various genes associated with miR-21 function. miRNA was isolated from kidney tissue collected from both affected dogs and unaffected, age-matched littermates at defined milestones of disease progression, including end-stage renal disease (ESRD). Additionally, autopsy samples from affected dogs at ESRD and corresponding unaffected dogs were evaluated. Samples were scored based on histological changes, and relative expression of miR-21 and kidney disease-related genes was determined using quantitative real-time polymerase chain reaction. In affected dogs, significant upregulation of kidney miR-21 was first detected at the milestone corresponding with increased serum creatinine. Furthermore, miR-21 expression correlated significantly with urine protein: urine creatinine ratio, serum creatinine concentration, glomerular filtration rate, and histologic lesions (glomerular damage, tubular damage, chronic inflammation, and fibrosis). At end-stage disease, COL1A1, TGFB1 and its receptor, TGFB2, and Serpine1 were upregulated, while PPARA, PPARGC1A, ACADM, SOD1, and EGF were downregulated. In conclusion, miR-21 is abnormally upregulated in the kidneys of dogs with CKD caused by XLHN, which may play an important pathologic role in the progression of disease by dysregulating multiple pathways.
Keywords
Small, noncoding RNAs known as microRNAs (miRNAs) play a significant role in a variety of physiologic and disease processes. 3 MicroRNAs act as negative regulators of posttranslational gene/protein expression by binding to the 3’-untranslated regions of specific mRNAs, typically resulting in either suppression of protein translation or degradation of the target mRNA. 3 The processing of miRNA is tightly regulated, and miRNAs are expressed in both a tissue-specific and cell type–specific manner. Alterations in miRNA expression profiles in tissue and body fluids have been observed with many diseases, including chronic kidney disease (CKD) in humans and animals, 7,47 and dysregulation of miRNAs may contribute to pathogenesis. Due to the important roles of miRNAs in the regulation of gene expression and pathogenesis of different diseases, some miRNAs have been considered as therapeutic targets for a variety of diseases. 10,45
In dogs, CKD is a common cause of morbidity and mortality. 1 While there are many different causes of CKD in dogs, the mechanisms leading to end-stage renal disease (ESRD) are thought to be similar. One rare, although well-characterized form of CKD in dogs is X-linked hereditary nephropathy (XLHN). Affected dogs have abnormal type IV collagen present in their glomerular basement membrane, and affected male dogs demonstrate rapid progression to ESRD, typically by 1 year of age. 15 This disease is equivalent to Alport syndrome (AS) in people, and these dogs serve as a large animal model for investigating the pathogenesis of AS disease development. 13,42,43 Furthermore, these dogs serve as a good model of CKD due to their predictable nature, thorough monitoring of disease progression, and potential similarities in pathogenesis leading to ESRD. 2,35
In CKD, interstitial fibrosis is a characteristic finding with progressive renal injury, regardless of cause. 37,41 The TGF-β1 signaling pathway is a key promoter of CKD progression and fibrosis. 5,37 TGF-β activation exerts pro-fibrotic effects in a number of ways, including recruitment and activation of inflammatory cells, stimulation of extracellular matrix synthesis, proliferation of fibroblasts, and increased tubular epithelial cell apoptosis. 6,37 While several miRNA genes have been identified as targets regulated by TGF-β and important modulators of renal fibrosis, one of particular interest is miR-21. 12,37,41,47,49 TGF-β promotes miR-21 production by both increasing transcription of miR-21 through activation of the Smad complex (Smad2/3/4) and enhancing posttranslational processing of pre-miR-21. 37,41 In turn, miR-21 promotes TGF-β expression by inhibiting Smad7, an inhibitor of the TGF-β/Smad2/3 pathway, thereby creating a positive feedback loop. 37 Additionally, miR-21 promotes proliferation and inhibits apoptosis of fibroblasts through alteration of several metabolic pathways. 12,41 Further supporting the role of miR-21 in progression of CKD, miR-21 silencing (using anti-miR-21 oligonucleotides or genetic knockout) ameliorated development of kidney fibrosis in several different murine models of CKD, including a model of AS. 8,23,41 Therefore, further investigation of miR-21 and its function in CKD development is essential.
The role of miRNAs in renal fibrosis, particularly miR-21, has been evaluated extensively in various rodent models of CKD. 8,23,49 –51 To date, there are few studies that have focused on characterizing miRNA expression profiles in dogs. 4,20,29,38,40 Regarding kidney tissue, miRNA expression profiling has been evaluated in both healthy and diseased dogs. In kidney tissue of healthy dogs, expression profiles were found to differ between the renal cortex and medulla. 29 More recently, miRNA expression profiles generated from both urinary exosomes and kidney tissue collected by laser-microdissection from clinically healthy dogs and dogs with kidney disease were created and compared. In dogs with kidney disease, miR-21a was increased in the tubulointerstitium but not the glomeruli. 28
The role of miR-21 in both humans with AS and dogs with XLHN is currently unknown. Therefore, the objective of our study was to evaluate the expression of miR-21 and associated genes in renal tissue collected over the course of disease progression in dogs affected with XLHN. Results obtained can provide further insight into the pathogenesis of progression of CKD in dogs and particularly the role of miR-21 in this process. The results might also be applicable to AS progression in humans.
Materials and Methods
Animals
Dogs were from a colony maintained at Texas A&M University in which the causative mutation of the disease in the affected males is a naturally occurring, 10 base pair deletion in the gene encoding the α5 chain of type IV collagen. 15 Development and progression of XLHN in these dogs has been described. 35 No treatments were administered to the dogs used in the study. The study protocol was reviewed and approved by the Texas A&M University Institutional Animal Care and Use Committee.
Sample Collection
Blood and midstream voided urine were collected for determination of physiologic data, including serum creatinine (sCr) and urine protein: creatinine ratio (UPC) (Vitros 250, Johnson & Johnson Co, Rochester, NY). Glomerular filtration rate (GFR) was also measured as previously described for each dog using either renal scintigraphy 39 or iohexol clearance 33 throughout the advancement of disease. To monitor disease progression, milestones were set and defined by the following criteria: milestone 1 (MS 1) = presence of microalbuminuria for 2 consecutive weeks, milestone 2 (MS 2) = UPC ≥2 for 2 consecutive weeks, milestone 3 (MS 3) = sCr ≥1.2 mg/dL, milestone 4 (MS 4) = sCr ≥2.4 mg/dL, and milestone 5 (MS 5) = sCr ≥5 mg/dL or clinical signs of uremia for 2 or more consecutive days (ie, anorexia, dehydration, vomiting). Testing for microalbuminuria was performed only until milestone 1 was reached using a semi-quantitative test (E.R.D. HealthScreen Canine Urine Test strips, Heska, Loveland, CO).
Ultrasound-guided needle biopsies from alternating kidneys were obtained from both affected dogs and unaffected, age-matched littermates to serve as a control at each defined milestone during disease progression as previously described. 13,33,39 Biopsy cores were placed into either formalin or RNAlater (Thermo Fisher Scientific, Waltham, MA). Samples in RNA later were stored at –80°C for a range of 4 to 10 years until RNA isolation. In all, biopsy samples obtained from 11 affected and 10 unaffected dogs were evaluated.
When affected dogs reached advanced disease as defined by serum creatinine ≥5 mg/dL or had clinically significant disease (as described previously), they were humanely euthanized under anesthesia following biopsy collection. Additional kidney tissue was collected for evaluation at autopsy and was snap frozen in liquid nitrogen and stored at –80°C for a range of 4 to 10 years until RNA isolation. Autopsy samples from a total of 27 affected and 5 unaffected dogs were evaluated.
Light Microscopy Evaluation
For light microscopy, formalin-fixed, paraffin-embedded biopsies were sectioned at 3 µm and stained with hematoxylin and eosin, Masson’s trichrome, and periodic acid-Schiff. Sections were scored for glomerular and tubulointerstitial damage as previously published and as outlined in Supplemental Tables S1 and S2. 39 Four histopathologic changes were compared to relative miR-21 expression: glomerular damage, tubular damage, chronic inflammation, and interstitial fibrosis. A glomerular damage score was determined for each dog by assessing the overall percentage of abnormal glomeruli that involved any of the following features: segmental sclerosis, global sclerosis, synechia, obsolescence, fetal glomeruli, crescents, Bowman’s capsule dilation, and glomerular atrophy. An average tubular damage score was determined using the following features: tubular dilation, loss of brush border, tubular atrophy, tubular epithelial cell degeneration/regeneration, and tubular single cell necrosis. Chronic inflammation was defined as consisting of lymphocytes, plasma cells, and/or macrophages Interstitial fibrosis was defined as increased extracellular collagenous matrix with an increase in fibroblasts/myofibroblasts. Chronic inflammation and interstitial fibrosis were graded on a scale of 0 to 3 depending on distribution or severity, respectively.
RNA Isolation, Reverse Transcription, and Quantitative Polymerase Chain Reaction
All samples were homogenized in RLT Buffer (Qiagen, Valencia, CA) using a Bead Ruptor Mill Homogenizer (Omni International, Kennesaw, GA). Total RNA was isolated using the mirVana miRNA Isolation Kit (ThermoFisher Scientific, Waltham, MA) following the manufacturer’s protocol. Total RNA concentration was determined using the NanoDrop 2000 UV-Vis Spectrophotometer (ThermoFisher Scientific, Waltham, MA). RNA quality was assessed by the Agilent RNA 6000 Nano Kit on the Agilent 2100 Bioanalyzer (Agilent, Santa Clara, CA) according to the manufacturer’s protocol. Samples with an RNA Integrity Number greater than 7 or that had adequate 28 S to 18 S peaks on analysis were considered adequate for further evaluation.
The amount of miRNA present in each sample was determined using the specific commercially available TaqMan microRNA Assay for miR-21 and miR-16, which served as an endogenous control (ThermoFisher Scientific, Waltham, MA, miR-21 assay ID 000397; miR-16 assay ID 000391) following the manufacturer’s protocol. Briefly, reverse transcription was performed using 5 ng of total RNA in a 15 µl reaction volume. Then, 3 µl of the reverse transcription product, along with appropriate non-template negative controls, were amplified using the TaqMan microRNA reaction in a total volume of 20 µl.
Genes associated with miR-21 expression or fibrosis development were evaluated using 2-step real-time quantitative polymerase chain reaction (RT-qPCR). Briefly, total RNA was reverse transcribed using RNA to cDNA EcoDry Premix (ClonTech, Mountain View, CA). The reverse transcription product along with appropriate non-template negative controls were used in the specific commercially available TaqMan gene expression assays (ThermoFisher Scientific, Waltham, MA) following the manufacturer’s protocol with18S serving as an endogenous control gene. The specific TaqMan assays used for all RT-qPCR reactions are listed in Supplemental Table S3.
Gene expression data were analyzed using the Delta-Delta-Ct (ddCt) method, and expression in affected dogs is shown as a relative fold change compared to age-matched unaffected controls. There were some unaffected dogs for which we did not have the corresponding affected littermate’s tissue available for comparison. For those unaffected dogs, data corresponding to that biopsy were distributed into the different milestones in a manner that provided a reasonable number of control dogs to span the age of the affected dogs within each milestone. Data were first normalized to the endogenous control gene (miR-16). Then, the expression level was calculated as a relative fold change compared to the mean fold change of the unaffected control group at each milestone. The mean fold change of the unaffected control group was arbitrarily set as 1.
Statistical Analysis
Statistical analysis of the clinical and histopathologic data comparing affected versus unaffected dogs was performed using JMP 12.0 software. Normality was assessed using a Shapiro-Wilks goodness-of-fit test on the residuals of sCr, UPC, GFR, and the histopathological scores (glomerular damage, average tubular damage, average chronic inflammation, and average fibrosis). As all the data were nonparametric, a Mann-Whitney test was performed to determine statistical significance between the 2 groups in each category at each milestone, with significance set at a P value of <.05.
Analysis of miR-21 expression with clinical and histological data was performed using GraphPad Prism 6 (GraphPad Software, San Diego, CA). In affected dogs, for comparison of relative miR-21 expression with UPC, sCr, and GFR, Pearson correlation was applied; for comparison of relative miR-21 expression with histopathological scores, Spearman correlation was applied. Correlation coefficients were calculated and presented as r value. For comparison of relative RNA expression of miR-21 among each stage of disease in the affected dogs, analysis was performed using SAS 9.1.4 (SAS Institute Inc, Cary, NC). We applied the 2-way ANOVA model with random effect nested in the dog, using genotype, stage (categorized), and their interaction as covariates; P value <.05 post-Bonferroni adjustment was considered statistically significant. For comparison of relative mRNA expression of miR-21–associated genes between affected and unaffected dogs, a Mann-Whitney test was performed using GraphPad Prism 6 (GraphPad Software, San Diego, CA). Statistical significance was defined as P value <.05.
Results
Clinical and Histopathologic Progression of Disease in Affected Dogs
The first clinical indication of disease in the affected dogs in this study was the onset of microalbuminuria, which occurred between 11 and 21 weeks of age (average 14 weeks). This progressively worsened to overt proteinuria followed by rapid advancement to ESRD between 26 to 46 weeks. Table 1 summarizes the clinicopathologic parameters of affected versus unaffected dogs throughout disease development. The UPC and sCr were significantly elevated in affected compared to unaffected dogs starting at milestone 2 (17–25 weeks of age) and milestone 3 (21–29 weeks of age), respectively. The GFR was not significantly different between the 2 groups until milestone 4 (21–40 weeks).
Comparison of Clinical Parameters (Median, Range) at Each Milestone of Disease in Dogs With X-Linked Hereditary Nephropathy Versus Unaffected, Age-Matched Littermates.a
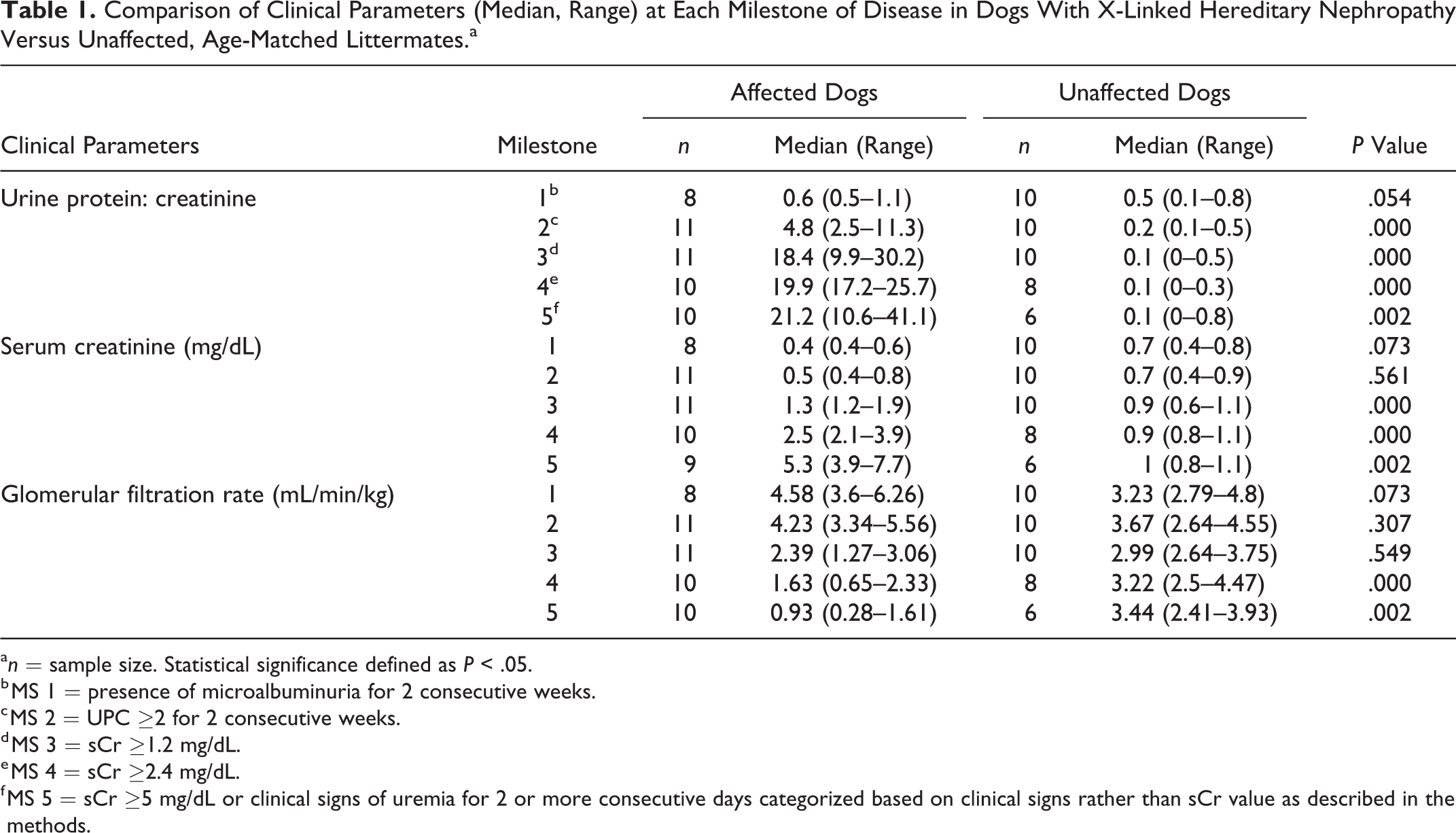
a n = sample size. Statistical significance defined as P < .05.
b MS 1 = presence of microalbuminuria for 2 consecutive weeks.
c MS 2 = UPC ≥2 for 2 consecutive weeks.
d MS 3 = sCr ≥1.2 mg/dL.
e MS 4 = sCr ≥2.4 mg/dL.
f MS 5 = sCr ≥5 mg/dL or clinical signs of uremia for 2 or more consecutive days categorized based on clinical signs rather than sCr value as described in the methods.
Histopathologic scores at each disease milestone in affected versus unaffected dogs are described in Table 2. Significant developments of all 4 categories of histologic lesions were not evident in kidney biopsies from affected dogs until milestone 3, when the dogs were mildly azotemic.
Comparison of Histological Scores (Median, Range) for Various Features on Light Microscopy as Observed on Evaluation of Kidney Biopsies Taken at Each Milestone of Disease in Dogs With X-Linked Hereditary Nephropathy Versus Unaffected, Age-Matched Littermates.a
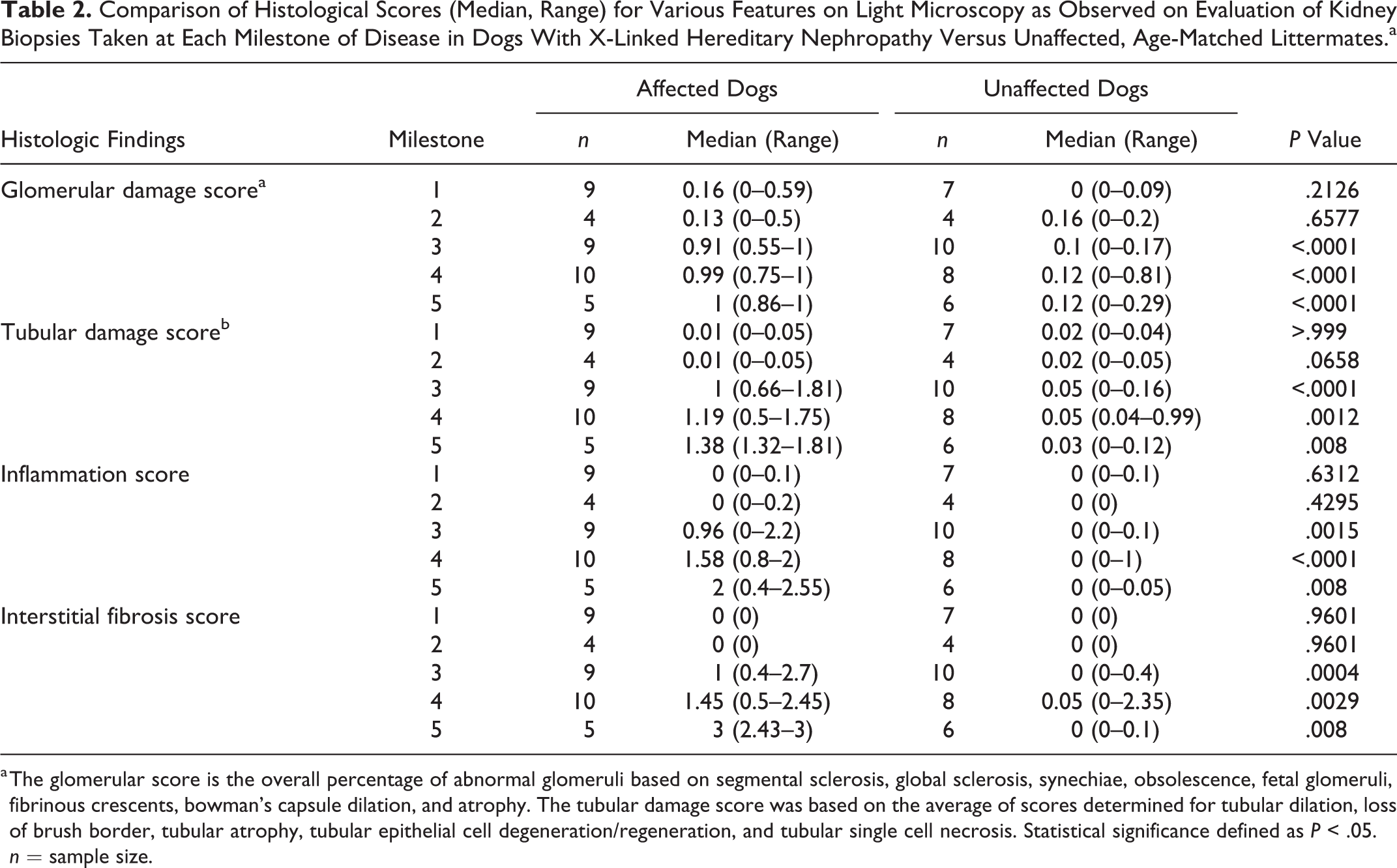
a The glomerular score is the overall percentage of abnormal glomeruli based on segmental sclerosis, global sclerosis, synechiae, obsolescence, fetal glomeruli, fibrinous crescents, bowman’s capsule dilation, and atrophy. The tubular damage score was based on the average of scores determined for tubular dilation, loss of brush border, tubular atrophy, tubular epithelial cell degeneration/regeneration, and tubular single cell necrosis. Statistical significance defined as P < .05. n = sample size.
miR-21 Expression During Disease Progression
Based on measurements performed on RNA isolated from biopsy samples, relative expression of miR-21 did not significantly increase in affected dogs until milestone 3 as compared with unaffected, age-matched control dogs (Fig. 1). Relative miR-21 expression in unaffected versus affected dogs at each milestone are shown in Supplemental Figures S1-S5. While there was little change in miR-21 expression between milestones 3 and 4, miR-21 expression significantly increased in the last milestone of disease (MS 5 or ESRD) compared to both milestones 3 and 4.
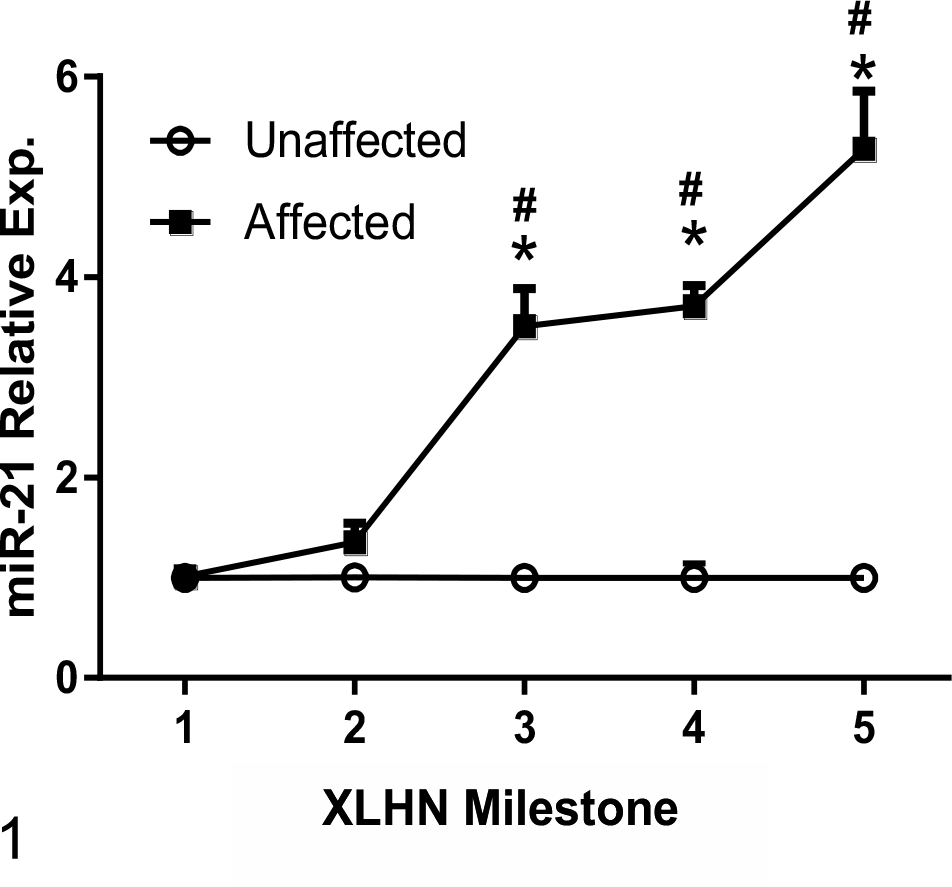
Relative renal miR-21 expression, as determined by RT-qPCR using RNA isolated from kidney biopsies sampled at different milestones during progression of disease. The data are presented as the values in dogs affected with X-linked hereditary nephropathy (XLHN) relative to the values in age-matched unaffected dogs. Relative renal miR-21 expression significantly increases from milestone (MS) 3 to end stage renal disease (MS 5). Exp. = expression. *P < .05 versus unaffected dogs and versus milestone 1 and milestone 2 affected dogs. **P < .05 versus milestone 3 and milestone 4 affected dogs.
In affected dogs, alterations in expression of renal miR-21 correlated with clinical markers of kidney function (Figs. 2–4). There was a positive correlation with UPC and sCr (r = .798, P < .0001 and r = .737, P < .0001, respectively) and a negative correlation with GFR (r = –.859, P < .0001). Additionally, a positive correlation was observed between miR-21 expression and histologic changes, indicating that increased miR-21 was associated with more severe pathological damage: glomerular damage (r = .6952, P < .0001), tubular damage (r = .799, P < .0001), chronic inflammation (r = .627, P = .0004), and interstitial fibrosis (r = .798, P < .0001) (Figs. 5–8).
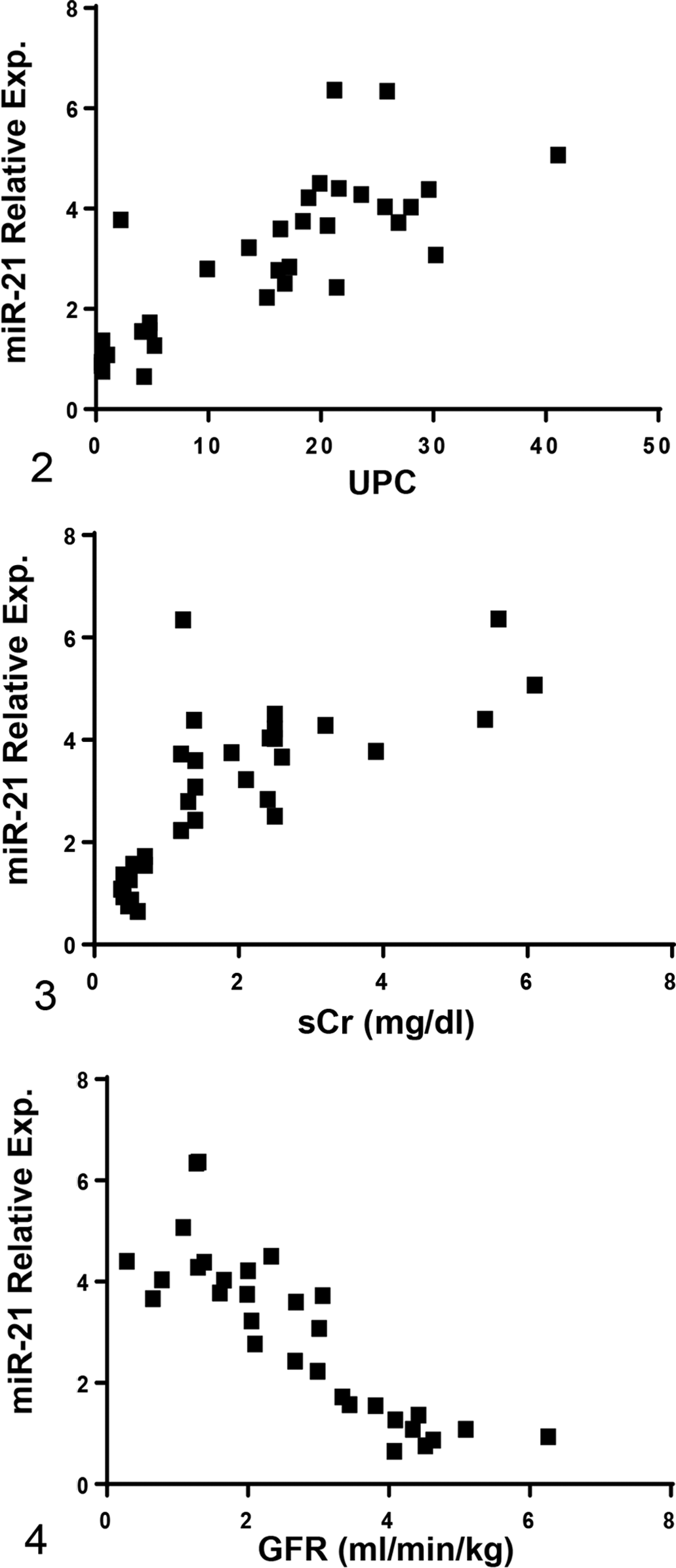
Relative renal miR-21 expression, measured by RT-qPCR using RNA isolated from kidney biopsies, positively correlates with urine protein: creatinine ratio (UPC) and serum creatinine concentration (sCr) and negatively correlates with glomerular filtration rate (GFR). The miR-21 relative expression data are presented as the values in dogs affected with X-linked hereditary nephropathy (XLHN) relative to the values in age-matched unaffected dogs. Exp. = expression. P < .05.
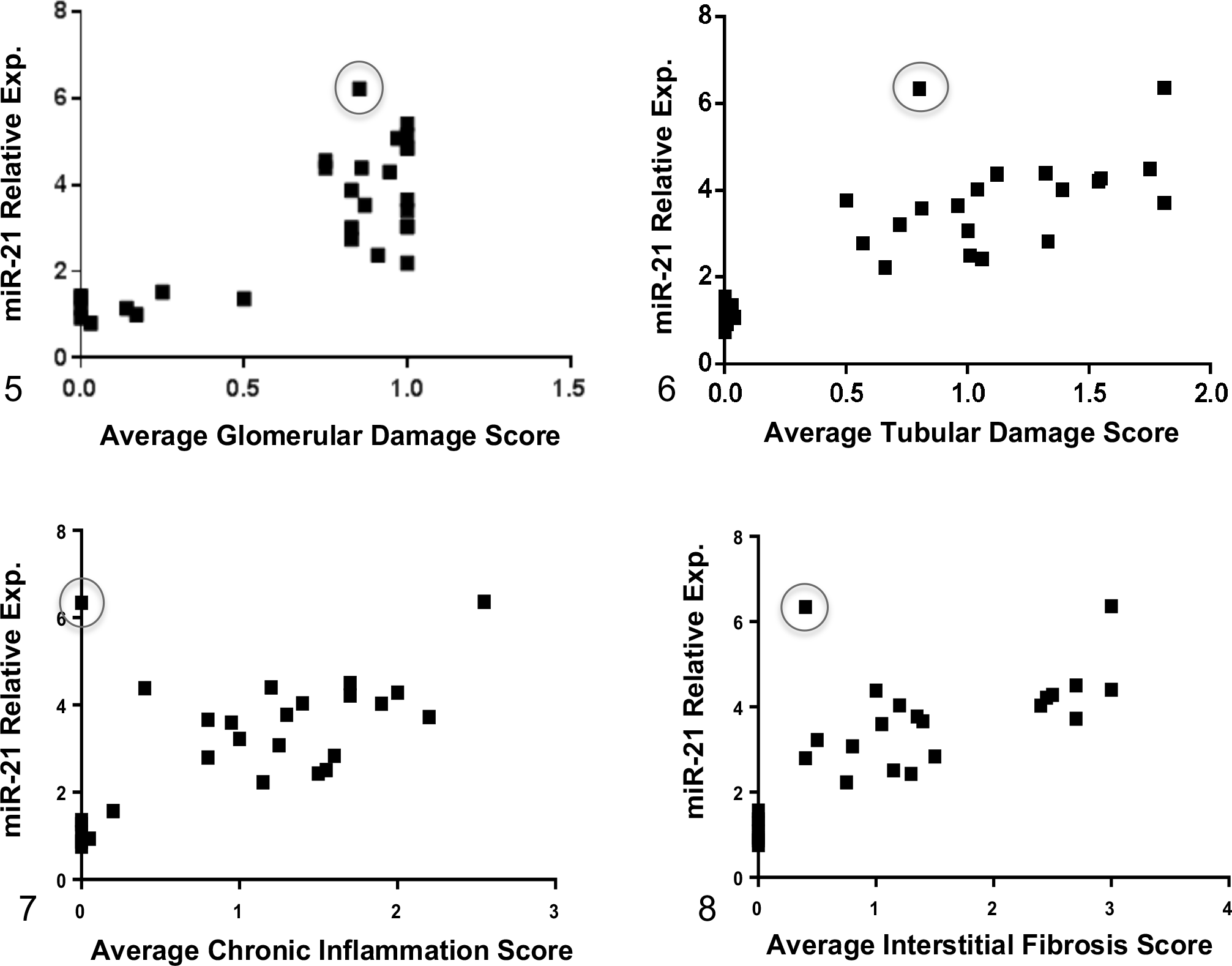
Relative renal miR-21 expression, measured by RT-qPCR using RNA isolated from kidney biopsies, positively correlates with average histopathological scores based on light microscopy, including glomerular damage, tubular damage, chronic inflammation, and interstitial fibrosis. The miR-21 relative expression data are presented as the values in dogs affected with X-linked hereditary nephropathy (XLHN) relative to the values in age-matched unaffected dogs. The circles indicate a sample from an affected male that demonstrated high relative expression of miR-21 but relatively low average histopathological scores. Exp. = expression. P < .05.
There was 1 biopsy sample where high expression of miR-21 (relative expression 6.34-fold) at milestone 3 was present with lower average histological scores (ie, minimal tubular damage and absence of chronic inflammation and only mild interstitial fibrosis observed on light microscopy) (circled on Figs. 5–8). To determine if the values from this dog changed the significance of the milestone 3 data in relation to other milestones, his information was removed and the data reevaluated. While there was a small increase in miR-21 expression between milestone 3 and 4, it was not statistically significant (data not shown). Individual Cq values along with relative miR-21 expression for both the unaffected dogs and the affected dogs at each milestone are shown in Supplemental Tables S4 and S5, respectively.
Measurement of miR-21 was also performed on RNA isolated from autopsy samples at ESRD. Autopsy samples have the advantage in that they are larger and therefore more representative of the kidney as a whole compared to biopsy samples. Analysis of the autopsy samples showed a significant, 6-fold increase in miR-21 in dogs at ESRD as compared with tissue from unaffected dogs similar in age (Fig. 9). These results support the histology findings based on biopsies, indicating that miR-21 expression is significantly increased in late-stage disease, when fibrosis has been shown to be the most extensive on light microscopy evaluation. 2
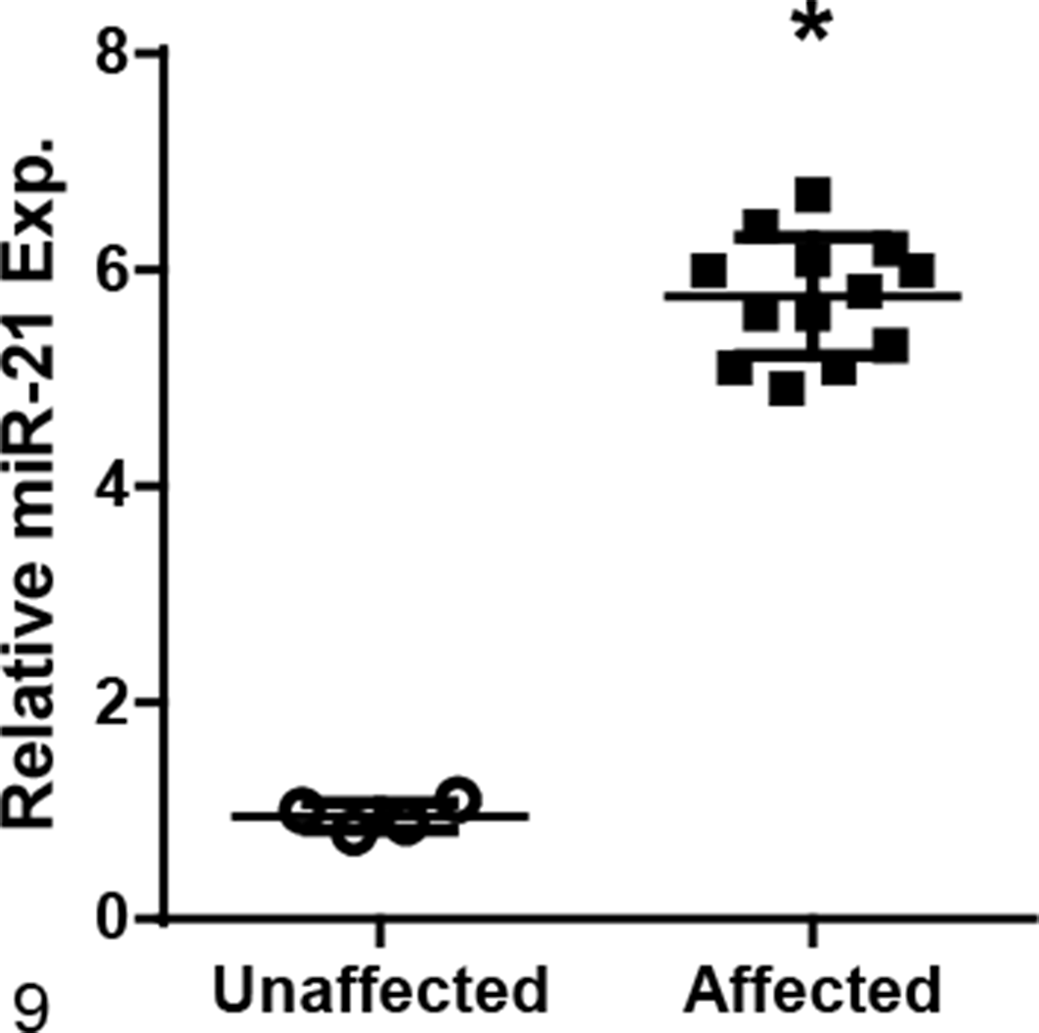
Renal miR-21 expression, measured by RT-qPCR on RNA isolated from tissue obtained at autopsy, is significantly increased at end-stage renal disease in dogs affected with X-linked hereditary nephropathy compared with age-matched unaffected dogs. Bars represent mean and standard deviation. Exp. = expression. *P < .05.
Dysregulation of Genes Associated With CKD and miR-21 Function
To gain further understanding of the developmental process of renal fibrosis in dogs with XLHN, relative mRNA expression in cortical tissue from the autopsy samples was measured for a number of key genes known to play a pivotal role in the progression of CKD and/or be involved in the miR-21 pathway of fibrosis development.
Among the 12 genes evaluated, mRNA levels of affected dogs were increased as compared to unaffected dogs for the following genes associated with fibrosis development: collagen 1 alpha 1 (COL1A1), transforming growth factor-beta 1 (TGFB1) and -beta 2 (TGFB2), transforming growth factor-beta receptor 1 (TGFBR1), and Serpine1 (Figs. 10–14). Of those genes, COL1A1, a prominent component of the extracellular matrix of fibrotic tissue, had a mean 30-fold increase in affected compared to unaffected dogs. Additionally, Serpine1, the gene that encodes for plasminogen activator inhibitor-1, had a mean 125-fold increase. In contrast, mRNA expression levels of genes related to the regulation of metabolic pathways in the mitochondria were either decreased in affected dogs or had no significant changes including: peroxisome proliferator-activated receptor alpha (PPARA), peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PPARGC1A), acyl-CoA dehydrogenase (ACADM), and carnitine palmitoyltransferase 1A (CPT1A) (Figs. 15–18). Of the 2 genes evaluated that encode for antioxidant enzymes, superoxide dismutase 1 (SOD1) was significantly decreased while MPV17 (a mitochondrial inner membrane protein) had no relative change in expression between affected and unaffected dogs (Figs. 19, 20). Additionally, relative mRNA expression of epithelial growth factor (EGF) was markedly decreased in affected dogs (Fig. 21).
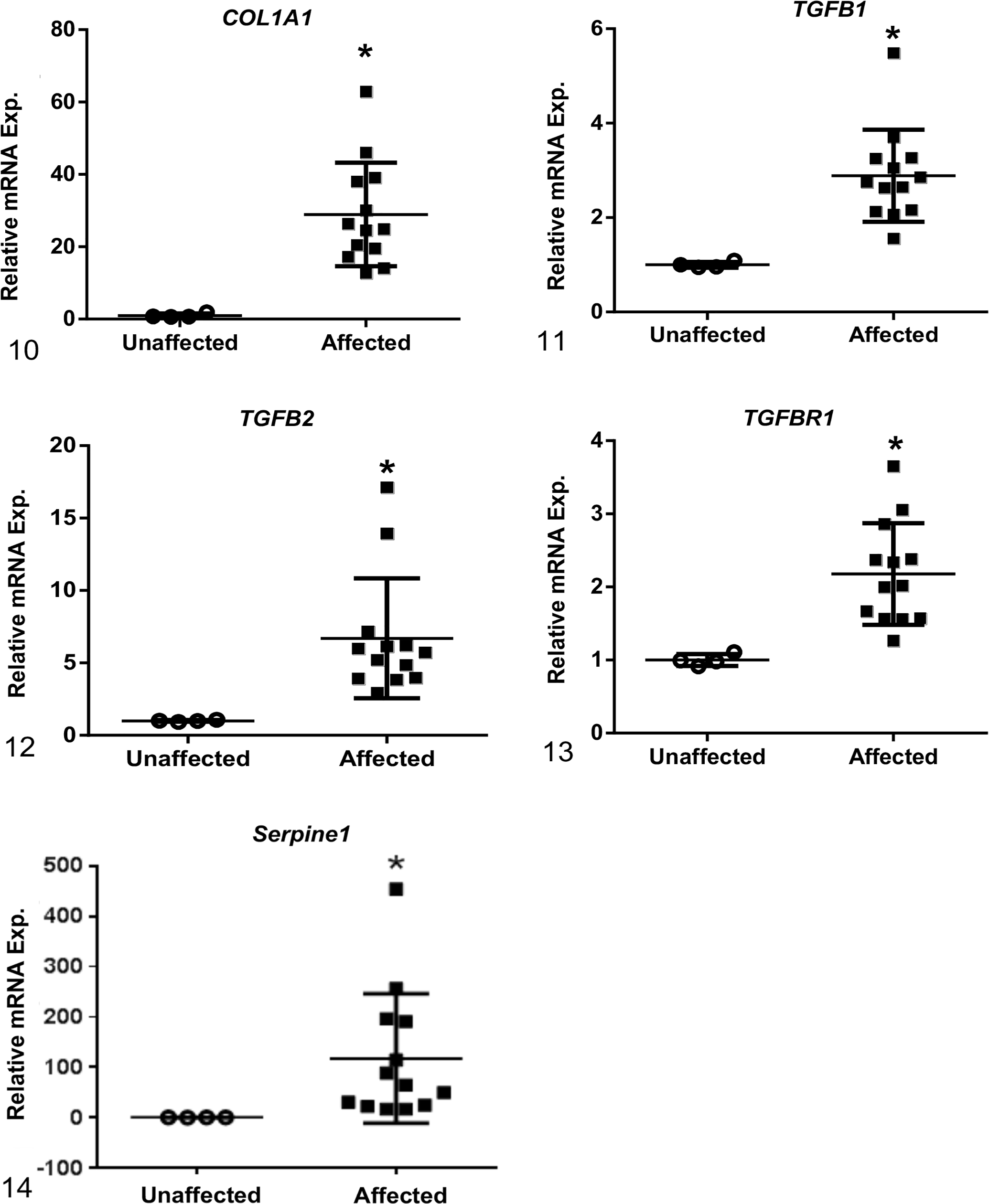
Relative mRNA expression levels of genes associated with fibrosis at end-stage renal disease. Based on RT-qPCR, mRNA expression of collagen 1α1 (COL1A1), transforming growth factor-β1 (TGF-B1) and -β2 (TGF-B2), transforming growth factor beta receptor 1 (TGFBR1), and Serpine1 were increased in renal tissue from dogs affected with X-linked hereditary nephropathy (XLHN) compared to unaffected dogs. Bars represent mean and standard deviation. Exp. = expression. *P < .05.
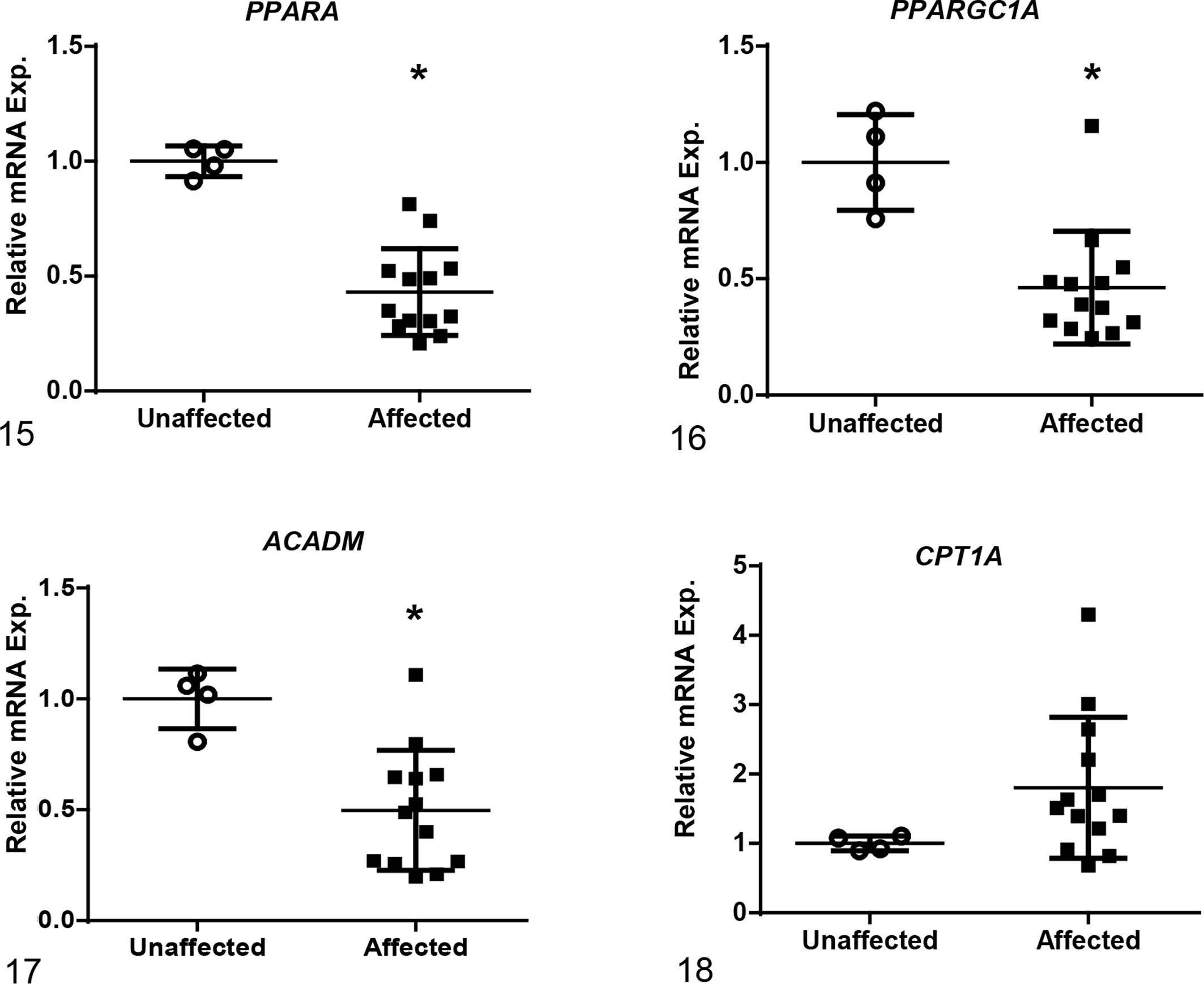
Relative mRNA expression based on RT-qPCR of genes associated with regulation of metabolic pathways in the mitochondria. Gene expression in affected dogs at end-stage renal disease is significantly decreased in renal tissue compared to age-matched control dogs for peroxisome proliferator-activated receptor alpha (PPARA), peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PPARGC1A), and acyl-CoA dehydrogenase (ACADM). Expression of carnitine palmitoyltransferase 1A (CPT1A) was unchanged when compared to unaffected dogs. Bars represent mean and standard deviation. Exp. = expression. *P < .05.
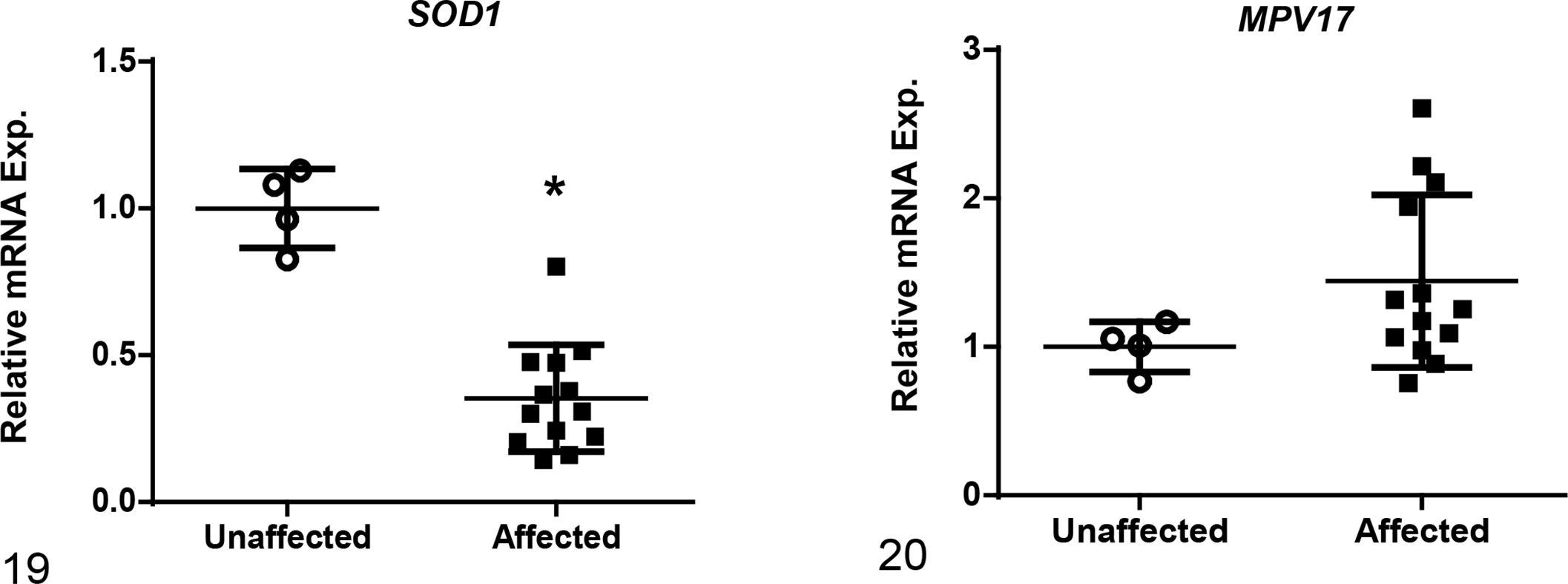
Relative mRNA expression based on RT-qPCR of genes that are responsible for minimizing the generation of reactive oxygen species. Gene expression of superoxide dismutase 1 (SOD1) was significantly decreased in renal tissue while MPV17 (a mitochondrial inner membrane protein) was unchanged in affected versus unaffected dogs at end-stage renal disease. Bars represent mean and standard deviation. Exp. = expression. *P < .05.
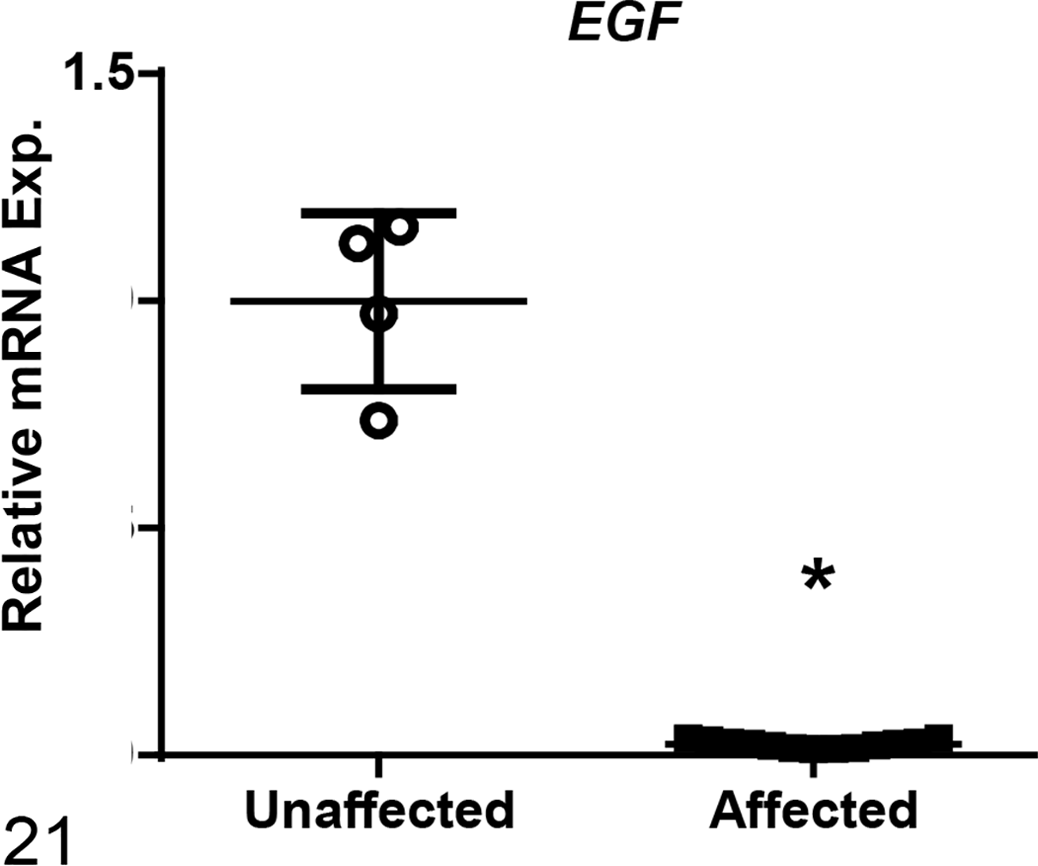
Relative mRNA expression of epithelial growth factor (EGF) was significantly decreased in renal tissue in affected dogs compared to unaffected dogs at end stage renal disease based on RT-qPCR analysis. Bars represent mean and standard deviation. Exp. = expression. *P < .05.
Discussion
Our primary objective was to compare the expression of miR-21 in serially collected renal tissue from unaffected dogs to dogs affected with CKD caused by XLHN. Additionally, we sought to explore the association of dysregulated miR-21 expression with clinical and histopathologic changes and the genes associated with miR-21 function to characterize their roles in the development of canine CKD. We found that miR-21 expression correlated with both the clinical parameters of renal function and histological changes, supporting our hypothesis that increased miR-21 expression plays a role in the pathogenesis of CKD. We were also able to characterize changes in relative mRNA expression of a number of genes associated with miR-21 function and/or CKD progression.
Over the course of disease, we determined that a significant increase in miR-21 expression in affected dogs occurred between milestone 2 and milestone 3, which corresponds with the first clear evidence of decreased GFR (based on sCr) and histologic changes (based on light microscopy). Expression further increased from milestone 4 to milestone 5. These findings support the involvement of miR-21 in the progression of kidney disease, including development of interstitial fibrosis. In affected dogs, the presence of interstitial fibrosis can be detected as early as milestone 1 based on immunofluorescent staining with fibronectin, although the amount of fibrosis is minimal. 13 The lack of increased miR-21 expression in milestones 1 and 2 as compared with unaffected dogs may indicate that either initiation of fibrosis might be directed by other factors not involving miR-21 or overall expression of miR-21 is not enough to be detected as a significant difference in expression levels in these kidney tissue samples despite a potential impact on development of fibrosis.
During progression of disease, we expected that miR-21 would show a progressive increase with each milestone. The lack of evident increase between milestones 3 and 4 is partially because one of the affected dogs demonstrated much higher miR-21 expression compared to the other dogs at milestone 3, as presented in the Results. This dog developed more severe clinical signs, including gastrointestinal abnormalities, during the progression of his disease compared with other dogs at a similar sCr. He reached his clinical endpoint based on clinical signs rather than sCr. While the exact cause for these clinical abnormalities were not confirmed, either an additional acute kidney injury or a secondary chronic disease unrelated to the kidneys may have contributed to increased miR-21 expression despite lower interstitial fibrosis and chronic inflammation scores. Alternatively, the samples used for RNA evaluation, while harvested at the same time, are different biopsy cores than those that were used for histologic evaluation. Thus, it is possible to have a discrepancy between the 2 samples, particularly for patchy changes like tubulointerstitial lesions. When data were reanalyzed excluding this value, there appeared to be a small increase in miR-21 expression in milestone 4 as compared with milestone 3. However, statistical significance remained unchanged, likely in part due to the small number of dogs represented in each group.
The source of miR-21 in kidney tissue might also help to explain, at least in part, some of the lack of statistical difference between relative miR-21 expression between milestones 3 and 4. A previous study using unilateral ureteral obstruction (UUO) as a model for inducing renal fibrosis in mice determined that enhanced expression of miR-21 originated primarily from tubular epithelial cells. 49 In the dogs, as disease worsens, functional kidney tissue is replaced by fibrosis, which results in fewer tubular epithelial cells to express miR-21. However, another study also using UUO as a model of fibrosis development in mice found that fibroblasts derived from the fibrotic kidney tissue had higher levels of miR-21 expression compared to control fibroblasts. 21 Additionally, TGF-β significantly induced expression of miR-21 in normal kidney fibroblasts. 21 The loss of normal renal tubular cells expressing miR-21 might be offset by increased production of miR-21 by fibroblasts, resulting in similar overall expression between milestones 3 and 4 in the dogs. Ultimately, there are likely multiple sources and factors that contribute to the production of miR-21 during progression of kidney disease that need to be further explored.
Additional studies could be conducted to help gain a more in-depth perspective of the role of miR-21 in the various stages of disease development. Evaluation of kidney tissue and miR-21 expression at time points between milestone 2 and milestone 3 may help pinpoint a more precise time point of elevation in miR-21 expression, further defining its role in disease development. Additionally, evaluation of the individual cell populations within the kidney in conjunction with changes in expression of miR-21 within each of these cell types is needed. Methodologies such as laser capture microdissection or in situ hybridization could be employed to more specifically explore gene expression in the different cell populations.
The various genes involved in CKD progression and fibrosis have been previously explored in both mice and dogs with various causes of CKD, including AS. 2,11,16,23,50 The involvement of TGF-β and its connection with miR-21 in the development of fibrosis is already well established, and elevated TGFB mRNA expression in kidney tissue, particularly at early milestones of disease, has previously been identified in dogs with XLHN. 2 TGF-β1 was also identified as the most activated upstream regulator when comparing XLHN dogs with rapid versus slow disease progression. 11 Our study mirrors those findings. COL1α1 is a known component of fibrosis and is indirectly increased by miR-21 through the TGF-β signaling pathway. Serpine1, the gene that encodes for plasminogen activator inhibitor-1, promotes fibrosis by inhibiting the activities of urokinase, tissue-type plasminogen activator, plasmin, and matrix metalloproteinases. 19 Additionally, plasminogen activator inhibitor-1 expression is induced by TGF-β through activation of the Smad pathway, and it has been shown to be specifically upregulated in the kidney in renal disease but is essentially undetectable in the normal kidney. 44 Given this relationship, plasminogen activator inhibitor-1 is also indirectly increased by miR-21 through upregulation by the TGF-β signaling pathway. The often markedly increased expression of these genes in affected dogs at ESRD corresponds with collagen accumulation and fibrosis development.
In addition to being a pivotal player in fibrogenesis, miR-21 contributes to the development of disease by dysregulating a number of metabolic pathways in both the mitochondria and peroxisomes of glomerular and tubular epithelial cells. These findings have been demonstrated in mice with diabetic nephropathy, CKD using a doxycycline-inducible transgenic system, and Alport mice using anti-miR-21 oligonucleotide therapy. 17,22,23,32 Tubular epithelial cells depend primarily on fatty acid oxidation (FAO) as a source of energy. 32 PPARα and PPARGC1A act together as key transcription factors to regulate expression of enzymes involved in FAO. In addition, PPARα has a number of important downstream effects, including stimulating healthy mitochondrial function, inhibiting NF-κB signaling to promote an anti-inflammatory environment, and inhibiting TGF-β signaling to diminish profibrotic responses. 23,34 ACADM is the gene that encodes for medium-chain acyl-CoA dehydrogenase, an enzyme that is also important for FAO. CPT1A is another enzyme that is essential for FAO by connecting carnitine to long-chain fatty acids so that they can cross the inner membrane of mitochondria and be metabolized. It is considered the rate limiting enzyme in FAO. Impaired fatty acid metabolism and detoxification contribute to kidney disease by reprogramming tubular epithelial cells into a profibrotic phenotype and causing higher levels of apoptosis, de-differentiation, and increased intracellular lipid accumulation. FAO suppression is controlled through regulation of PPARα and PPARGC1A, which are directly regulated by both TGF-β1 and miR-21 activity. 8,23,32 We identified decreased gene expression of PPARA, PPARGC1A, and ACADM in affected dogs compared to unaffected dogs, highlighting the potential importance of alterations in cellular metabolism, particularly FAO, in the advancement of kidney fibrosis in dogs with XLHN. Additionally, loss of renal tubular cells during disease progression might also contribute to lower expression of these metabolic-related genes. Further studies are needed to understand the role of FAO in disease progression and fibrosis development in the dog.
Generation of reactive oxygen species (ROS), particularly by kidney epithelial cells, stimulates additional inflammatory responses, which then contributes to further cell damage and fibrosis. In several mouse models of CKD, miR-21 was shown to control generation of ROS in the mitochondria by silencing key antioxidant enzymes, including MPV17 and SOD1. 8,23 Similarly, we found SOD1 to be significantly decreased in affected dogs compared to unaffected counterparts, and this decreased expression could be contributing to the pathogenesis of disease in affected dogs through ROS-mediated damage. In contrast, MPV17 did not show significant change between the 2 groups.
In human kidney tissue, in situ hybridization has been used to confirm that synthesis of EGF occurs primarily in the tubular compartments, particularly the thick ascending limb of the loop of Henle and the distal tubles. 30,31 In both acute and chronic kidney disease, EGF has been shown to play a role in regulating cell proliferation and tissue repair response after tubulointerstitial damage. 24,27,30,46 Moreover, EGF gene expression has been shown to be reduced in CKD. 31,36 Based on the markedly decreased levels of expression of EGF at ESRD in affected dogs, our findings support that the reduction of EGF reflects damage and loss of renal tubular cells as fibrosis ensues. The dysregulation of the EGFR pathway in mediating renal fibrosis in CKD has already been established and is in part due to increased expression of TGF-β. 9,26 Given the connection between miR-21 and TGF-β, one can surmise that changes in miR-21 expression may also influence EGF expression; however, to the authors’ knowledge, a direct correlation has not yet been established.
Our findings are overall concordant with those identified in homogenized kidney from Col4a3 -/- Alport mice, whereby expression of miR-21 increased with disease progression. 23 However, in Alport mice, upregulation of miR-21 preceded morphologic changes in tissue on light microscopy, with a 2-fold elevation observed prior to histologic changes. 23 There are several possible reasons for this discrepancy. First, in the dogs, kidney biopsies were collected for evaluation, while in the mice, whole kidney tissue was used. This procedural difference may have resulted in larger variation in both histologic evaluation and miR-21 expression within the canine samples, leading to decreased sensitivity. Second, biopsy samples in the dogs were taken at time points that were based on clinical testing, whereas mice were sampled at predetermined ages in weekly increments up to the point of minor histological changes. Therefore, while the first 2 kidney biopsies in the dog were taken at the point of mild to moderate dysfunction of the glomerular basement membrane (ie, microalbuminuria and overt proteinuria), changes resulting in increased miR-21 expression and histologic changes evident by light microscopy appear to have occurred between the onset of overt proteinuria and the onset of azotemia. Certainly, the timeframe between milestone 2 and milestone 3 is relatively large (average of 8 weeks), with a vast number of molecular changes occurring between these 2 times. This may help to explain, at least partially, the marked increase in renal miR-21 from milestone 2 to milestone 3. As previously mentioned, evaluation of kidney tissue at time points between milestone 2 and milestone 3 could help detect molecular changes that occur prior to substantial histologic changes in the affected dogs. Lastly, there could be differences in the molecular mechanism of disease occurring in the kidney tissue between the dogs and mice. Therefore, the mechanistic role of miR-21 in the development of fibrosis in canine kidney tissue still needs to be proven.
One prior study evaluated differences in miRNA expression levels between healthy dogs and dogs with kidney disease. 28 In this study, miR-21a was increased in the tubulointerstitium, and similar to our study, higher levels correlated with abnormal histopathologic findings. Additionally, urinary exosome-derived miR-21 significantly increased with renal dysfunction. While that study provided insight into miRNA levels in dogs with kidney disease, the dogs were already azotemic with obvious renal lesions identified on light microscopy evaluation of autopsy tissue, and only one time point was evaluated. Additionally, the cause of CKD was not considered, and the dogs used in both groups for the comparison of miR-21 expression varied in age. In our study, the cause of CKD and progression of the disease is known and well characterized by longitudinal sampling done at the level of the individual over the entire course of disease. Therefore, serial evaluation of miR-21 expression from early disease through ESRD was possible, providing a more comprehensive analysis of miR-21 expression during advancement of disease. For instance, this experimental design allowed examination of the temporal relationship between changes in miR-21 expression and development of changes in the kidney during disease progression. In turn, this temporal relationship can help to establish a causal effect between miR-21 expression and progression of disease. This, in addition to the comparison with unaffected age-matched littermates, allows for increased confidence in corroborating that changes observed in miR-21 expression can be attributed to CKD and not from varying physiological states due to extra-renal disease or age.
This study is relevant to understanding the pathogenesis of AS. The progression of canine XLHN parallels the progression of disease in human AS patients. To our knowledge, this study is the first to demonstrate increased renal miR-21 expression in a model of AS other than mice. Our findings provide more confidence in the pathological role of dysregulated miR-21 expression in Alport syndrome across species.
In both animals and humans, CKD is a prevalent yet insidious disease. Because of the limitations of traditional diagnostic tools, CKD is often diagnosed only after significant damage to the kidneys has occurred. Moreover, while traditional biomarkers like sCr can provide an estimate of GFR and changes in kidney function during disease progression, evaluation of kidney tissue is the gold standard for determining the extent of renal damage. Irrespective of etiology, renal fibrosis is a key characteristic finding in CKD and has been shown to best correlate with renal function. 1,18 Given the correlation of miR-21 with both clinical markers and pathologic abnormalities, as identified in this study, miR-21 may be a promising biomarker for monitoring disease progression as it relates to fibrosis development. Additionally, because of their stability in bodily fluids, mature circulating miRNAs are good candidates for noninvasive biomarkers as compared to techniques such as renal biopsy. 14,48 Evaluation of miR-21 in serum and urine of dogs with XLHN along with comparison of these values to conventional clinical and histological evidence of disease can provide additional insight regarding the use of miR-21 as a noninvasive biomarker for evaluating progression of CKD and therefore should be explored. Furthermore, an understanding of the role of miR-21 in CKD development can provide a basis for the generation of novel therapeutic options. For example, based on observations in Alport mice, a single-stranded, chemically modified oligonucleotide (RG-012) that binds to and inhibits the function of miR-21 is being developed for treatment of AS in humans. 25
In conclusion, we found that miR-21 is upregulated in the kidneys of dogs with CKD caused by XLHN. Increased expression of miR-21 might play an important pathologic role in the progression of XLHN by dysregulating multiple pathways. To gain a better understanding of the role of miR-21 in the pathogenesis of CKD, further evaluation of miR-21 expression in dogs with CKD due to a variety of causes is warranted.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818806050 - Abnormal Expression of miR-21 in Kidney Tissue of Dogs With X-Linked Hereditary Nephropathy: A Canine Model of Chronic Kidney Disease
Supplemental Material, DS1_VET_10.1177_0300985818806050 for Abnormal Expression of miR-21 in Kidney Tissue of Dogs With X-Linked Hereditary Nephropathy: A Canine Model of Chronic Kidney Disease by Sabrina D. Clark, Wenping Song, Rachel Cianciolo, George Lees, Mary Nabity, and Shiguang Liu in Veterinary Pathology
Footnotes
Acknowledgements
We acknowledge the assistance of Mary Sanders (Texas A&M University), whose technical expertise contributed to sample collection and pathologic evaluations, and Kiera Pope (Texas A&M University), for her assistance with statistical analysis on the clinical and histological data. Additionally, we acknowledge Xiwen Ma (Sanofi) for his assistance on statistical advice and analysis. Fellowship funding for Dr Clark is provided in part by the Zoetis-Morris Animal Foundation Fellowship (Grant ID No. D14CA-904). Zoetis and the Morris Animal Foundation had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding support was provided in part by Sanofi and the National Institutes of Health (DK57676 and DK64273).
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
