Abstract
The large intestines of 21 raccoons (Procyon lotor; 11 wild caught, 10 laboratory confined) were examined for the presence of intestinal spirochetes. Light microscopy of sections stained with hematoxylin and eosin and Warthin-Starry stain showed the presence of spiral shaped organisms deep within the lumina of intestinal glands at the ileocolonic junction of 16 raccoons (76% prevalence). All laboratory-confined, grouphoused raccoons harbored the organisms, but only 6/11 (55% prevalence) live-trapped raccoons were positive for these spirochetes. The organisms were free in the glandular lumina, and there were no microscopic lesions. Two types of spirochetes were identified in the colonic glands: a slender spirochete 10-13 μm in length, 0.3 mm in diameter, and possessing long, thin tapered ends and a larger, regularly waved spiral organism (0.5 μm in diameter). The slender spirochete did not resemble any of the known spirochete genera and failed to grow on medium used to propagate oral treponemes and members of the genus Brachyspira.
Keywords
Raccoons (Procyon lotor) are highly adaptable nocturnal mammals that are found throughout North America and in some parts of Europe and Asia, where they were introduced in the early 1900s. 1 They share their immediate environment with humans and, therefore, are considered good monitors of infectious diseases and environmental contaminants for both humans and animals. 2 7 8
Spirochetes are helically coiled, motile, gram-negative bacteria that are characterized by the presence of periplasmic flagella located between the outer membrane and the protoplasmic cylinder. They are part of the normal intestinal flora and have been isolated from primates, pigs, dogs, rodents and birds. 6 9 Although many morphologic types have been described in the intestines, only members of the genus Brachyspira (formerly Serpulina) and Treponema succinifaciens have successfully been cultivated. 5 12 15 The majority of intestinal spirochetes are commensals, however some species of Brachyspira have pathogenic capability. B. hyodysenteriae is the agent of swine dysentery, B. intermedia and B. alvinipulli have been associated with production problems and wet litter in poultry, and B. pilosicoli is the agent of intestinal spirochetosis. 9–11, 16
Here we document light and electron microscopic descriptions of naturally occurring nonpathogenic spirochetes in the colonic mucosa of raccoons in Iowa.
During a 12-month period in 1999, 21 adult raccoons were examined at the National Animal Disease Center (NADC) in Ames, Iowa. Eleven raccoons were obtained for the purpose to document baseline pathologic data. Eleven raccoons were livetrapped (No. 207 traps, Tomahawk Live Trap Company, Tomahawk, Wisconsin), sedated with ketamine/xylazine, euthanatized, and necropsied. The remainder (n = 10) had been confined in the laboratory for approximately 10 months and were euthanatized at the time of termination of an experiment not related to this investigation.
Representative samples of all major organs including a section of colon obtained at the ileocolonic junction (Fig. 1) were immersion-fixed in 10% buffered formalin and routinely processed for histolopathology. Tissue sections were cut at 5 µm, stained with hematoxylin and eosin (HE), and examined by light microscopy. Selected sections of the ileocolon also were stained by Gram, periodic acid–Schiff (PAS), Gomori's methenamine silver (GMS), and Warthin–Starry stains to detect the presence of spirochetes. Only the results of the colon examination are reported here.
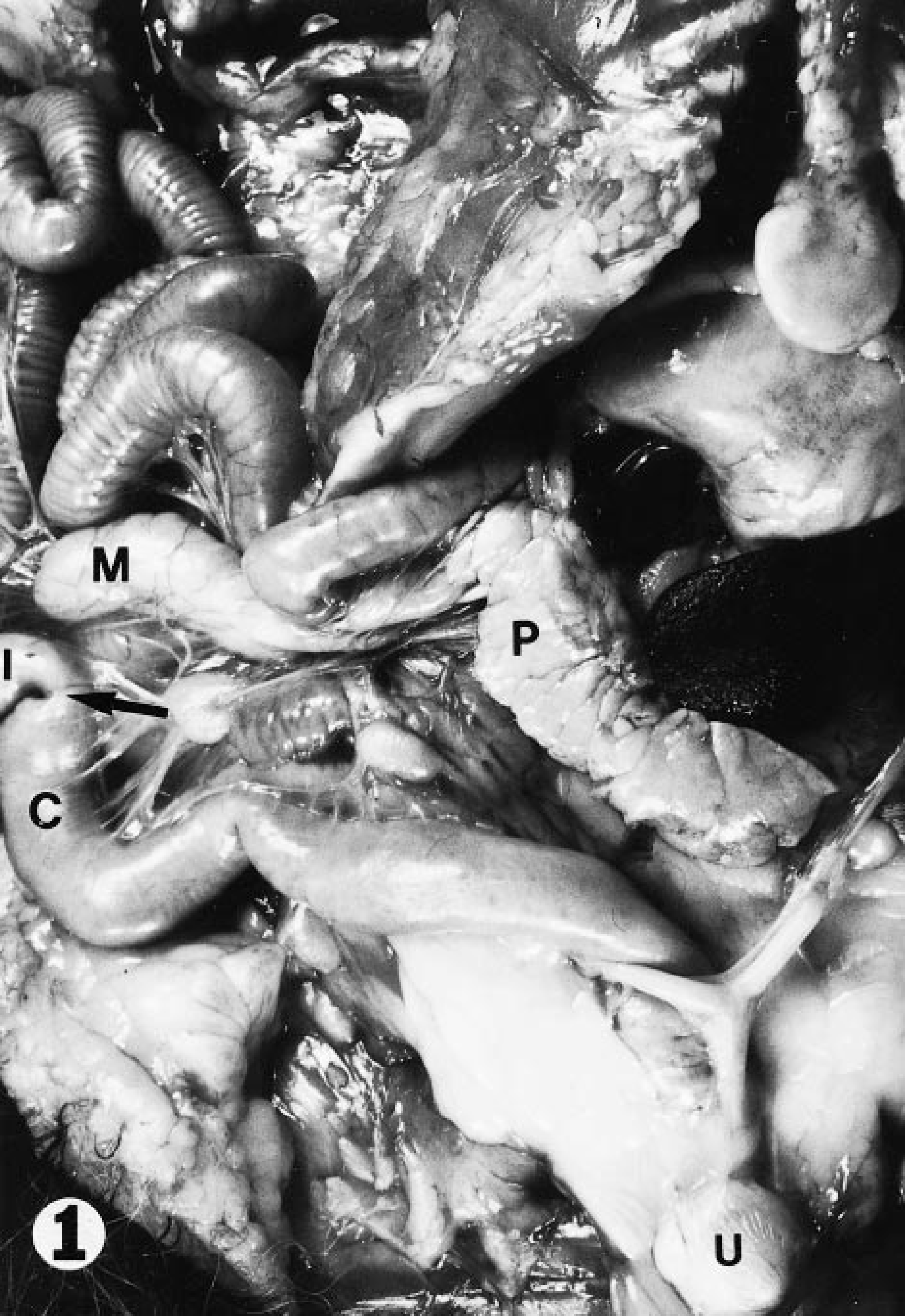
Internal abdominal organs; adult female raccoon. Note the absence of cecum and slight constriction (arrow) at the junction of the colon (c) and the ileum (I). M = mesenteric lymph node; P = pancreas; U = urinary bladder.
Selected samples of formalin-fixed colonic mucosa with microscopic evidence of spirochetes were postfixed in 2.5% glutaraldehyde in 0.1 M cacodylate buffer and then in 1.0% osmium tetroxide in 0.1 M cacodylate buffer and embedded in Eponate mixture (Ted Pella, Redding, CA). Thick sections (1 µm) were stained with toluidine blue and selected areas were thin sectioned, stained with saturated uranyl acetate and lead citrate, and examined with a Philips model 410 transmission electron microscope (FEI Company, Hillsboro, OR).
During necropsy 2- × 10-mm samples of fresh colonic mucosa were obtained from the ileocolonic junction and placed into 7 ml of anaerobic brain-heart infusion (BHI) broth for transport to a Coy anaerobic chamber (Laboratory Products Inc., Grass Lake, MI). The colonic samples were macerated with a scalpel blade on a sterile board and vortexed in 1 ml of BHI to release bacteria from the crypts. Aliquots (50 µl) of the suspension were examined with phase contrast microscopy for the presence of spirochetes and placed into 7 ml of BHI containing 10% serum, or plated onto trypticase soy agar containing 5% defibrinated bovine blood (TSB), or onto oral Treponema isolation agar containing 10% serum (OTI). 13 Antibiotics that were used to limit the growth of other fecal organisms included spectinomycin (400 µg/ml) or rifampin (25 µg/ml) alone or in combination with spiramycin (25 µg/ml), vancomycin (25 µg/ml), colistin (25 µg/ml), or polymixin B (5 µg/ml). Culture plates and broth were incubated at 37 C in an anaerobic Coy chamber and were examined for the presence of bacterial growth 24 hours after inoculation and then weekly for up to 21 days.
The remainder of the suspension was centrifuged at 300 × g for 10 minutes. The supernatant was centrifuged at 14,000 × g for 10 minutes and the resulting pellet was washed once with sterile phosphate-buffered saline (pH 7.4) and then resuspended in 200 µl of sterile distilled water. Aliquots (10 µl) were mixed with an equal volume of 2% phosphotungstic acid (pH 7.0), placed on grids, and examined under the transmission electron microscope.
Microscopic examination of HE-stained sections of colon showed numerous filamentous bacteria deep within the lumina of intestinal glands of 16 raccoons (76% positive), which included all 10 laboratory confined raccoons and six of the 11 (55%) live-trapped raccoons. The bacteria were predominantly present deep within the colonic crypts, which were mildly distended. The lining epithelium showed no degenerative changes, and there was no inflammation in the lamina propria. The individual organisms were weakly gram negative and did not stain with PAS and GMS. The bacteria stained positively with the Warthin–Starry technique and were filamentous and spiral (Fig. 2).
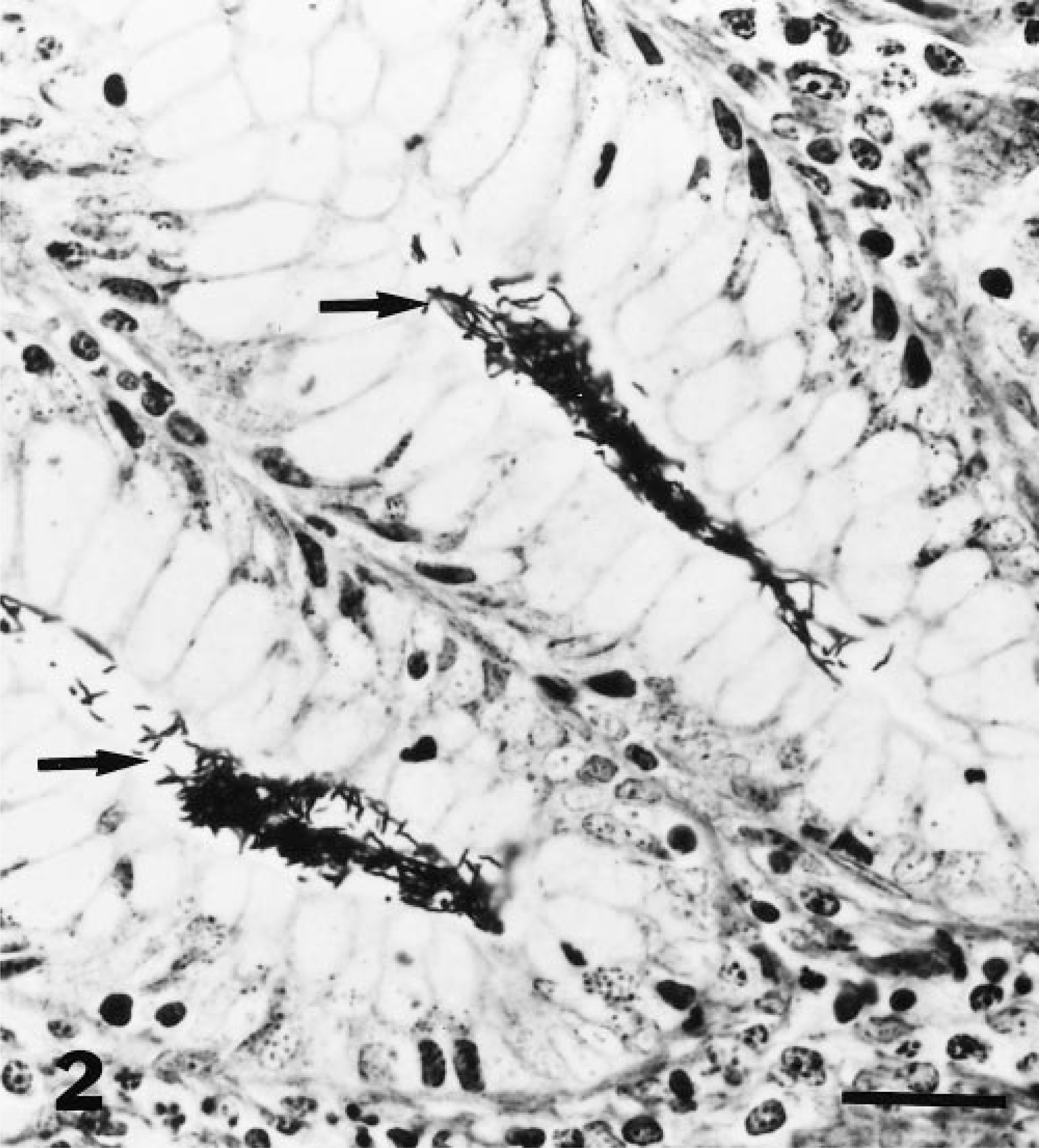
Colon; raccoon. Numerous silver-positive spiral organisms (arrows) are visible within the lumina of intestinal glands. Warthin–Starry stain. Bar = 50 µm.
Electron microscopic examination of formalin-fixed colonic mucosa revealed at least two different spirochetes. The majority of sections contained spiral-shaped, whiplike organisms < 0.3 µm in diameter with periplasmic flagella characteristic of spirochetes (Fig 3). Although the spirochetes were occasionally observed in close proximity to the mucosa, they were not found attached to the brush border of enterocytes. Some sections contained spiral-shaped organisms that were more regularly waved in longitudinal section and somewhat larger in diameter (0.45–0.50 µm). These organisms had a typical spirochete morphology and resembled members of the genus Treponema, but the presence of periplasmic flagella could not be confirmed.
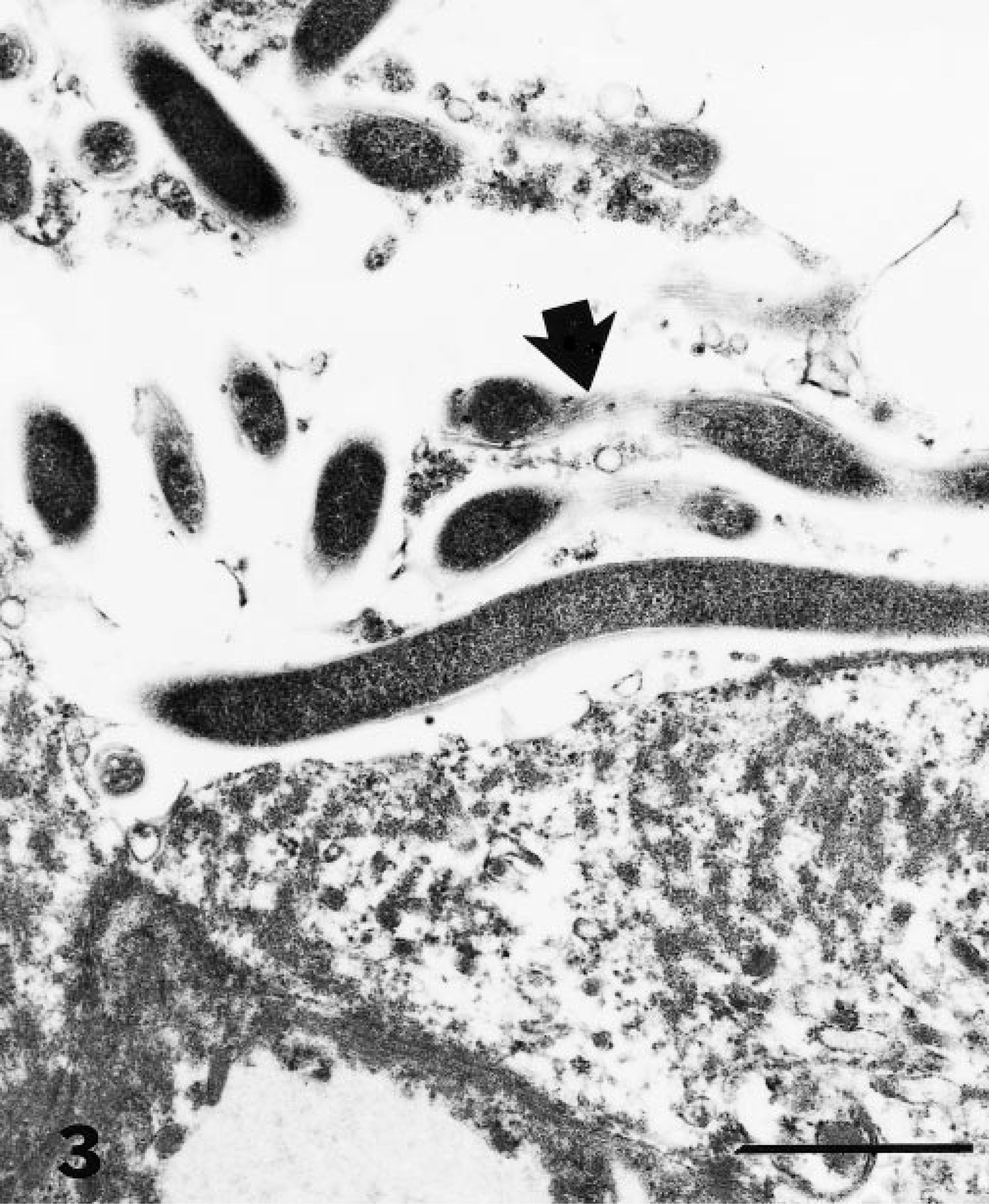
Electron micrograph. Colonic mucosa; raccoon. Many spiral-shaped organisms are present within the lumen of an intestinal gland. The organisms are identifiable as intestinal spirochetes by the presence of periplasmic flagella (arrow). Bar = 1 µm.
Phase contrast microscopy of colonic scrapings from 15 of the 21 raccoons also showed the presence of two distinct types of spirochetes. A slender whiplike organism with long irregular waves of variable amplitude approximately 10 µm in length was the only spirochete observed in seven sections examined (46.7%). A thicker, more regularly waved spirochete of approximately the same length was the sole spirochete observed in one of the sections (6.7%). Both spirochetes (the slender spirochete predominating) were observed in five sections (33.3%) and no spirochetes were observed in two sections (13.3%).
A slender whiplike spirochete also was observed in negatively stained colonic scrapings examined under the transmission electron microscope. These spirochetes were 10–13 µm in length and 0.3 µm in diameter at the widest margin and were characterized by extremely long tapered ends with small rounded tips. Starting at the spirochete tips and extending 1 µm towards the flagella crossover in the middle of the cell, the diameter of the spirochete cell remained < 0.1 µm. The exact number of periplasmic flagella per cell could not be determined because of the difficulty of visualizing insertion discs in the narrow tip region of the protoplasmic cylinder; however, at least five flagella were attached to each end of the cell (a total of 10 periplasmic flagella/cell). The thick spirochete was not observed in negatively stained colonic scrapings examined by electron microscopy.
Attempts to isolate the spirochetes using TSB for Brachyspira species or OTI for oral treponemes, with a number of antibiotic combinations, were unsuccessful. The spirochetes survived in anaerobic BHI containing 10% newborn calf serum, spectinomycin and rifampin for at least 4 hours, but the cultures were rapidly overgrown with motile rods after overnight incubation at 37 C.
Raccoons are unusual among mammals in that they do not possess a cecum. A slight constriction is the only grossly observable feature that indicates the location of the ileocolonic junction (Hamir, unpublished). Histologic examination of this region was performed as part of a study to determine baseline pathologic findings in raccoons in Iowa and revealed large numbers of spiral shaped organisms deep within the intestinal glands in 76% of the raccoons examined. The organisms show a distinct predilection for the ileocolonic junction; they were not observed in sections taken either proximally or distally from this area. Reduced numbers of spirochetes were observed by phase contrast microscopy in scrapings of distal sections of colon. However, these organisms may have been flushed from their preferred site.
Although it is difficult to definitively correlate histologic, ultrastructural, and phase contrast microscopy findings without concurrent culture of the organisms, the slender spirochete appeared to be the predominant organism in the ileocolonic crypts. The higher prevalence of spirochetes in the laboratory-confined raccoons may be related to grouphousing, which provides more opportunities for fecal–oral transmission.
As assessed by transmission electron microscopy, the slender spirochetes lying deep within the ileocolonic crypts initially resembled members of the genus Brachyspira in their diameter, shape and number of periplasmic flagella. Brachyspira species are relatively easy to cultivate by virtue of their tolerance to low levels of oxygen. 14 The raccoon spirochetes could not be cultivated on TSB medium or on OTI medium, which is more nutritionally rich and is designed to support the growth of fastidious, strictly anaerobic spirochetes. 12 Examination of negatively stained colonic scrapings showed that the slender spirochete had a unique morphology; no other cultivated spirochete has a similar structure. Although its dimensions were similar to those of members of the genus Brachyspira, the slender spirochete was unusual in having long tapered ends. Generally, spirochete ends are rounded, except for B. pilosicoli, which has pointed ends that are involved in the unique end-on attachment of this organism to the colonic mucosa. 16 B. pilosicoli-like spirochetes have been observed attached to the colonic epithelium of North American opossums. 17
Because of the absence of attachment, invasion, or histologic lesions, we concluded that these raccoon spirochetes are members of the autochthonous intestinal flora. They appear to have found a unique ecological niche within the crypts of the ileocolonic junction. They may have strict nutritional requirements that would present a challenge for in vitro cultivation. The taxonomic identity of these bacteria and their relationship to other species of spirochetes could be investigated by amplifying and sequencing 16S ribosomal DNA from the raccoon ileocolonic junction using spirochete-specific primers. Such techniques have previously been used to identify uncultivatable treponemes associated with gingivitis and papillomatous digital dermatitis. 3 4
Footnotes
Acknowledgements
We thank M. Church and J. Donald for technical assistance. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture.
