Abstract
Breslow thickness and Clark level are prognostic factors for human cutaneous melanomas. Breslow thickness is measured with an ocular micrometer from the top of the granular layer of the epidermis to the deepest invasive cell across the broad base of the tumor, while Clark level is based on the anatomical level of invasion through the layers of the dermis. Because of the anatomical differences between humans and dogs, we evaluated the tumor thickness and a modified Clark level in 77 canine primary cutaneous melanocytic tumors. Tumor thickness (using both a traditional and a more convenient system) and modified Clark level were measured and associated with histological diagnosis and clinical outcome. Tumor thickness was a prognostic factor, being greater in animals with shorter overall survival and disease-free time. Cutoffs of 0.95 cm and 0.75 cm defined a higher hazard for an unfavorable outcome and to develop recurrence/metastasis, respectively. Because of an excellent agreement between the 2 methods, it was concluded that tumor thickness could be measured with a ruler when an ocular micrometer is not available. Modified Clark level was not found to be relevant for prognosis. However, we suggest that both tumor thickness and a modified Clark level can be valid additional parameters when histological diagnosis is uncertain. Further studies, including a wider sample population, would be worthwhile to confirm the prognostic significance of these 2 parameters.
Breslow thickness is the most important independent prognostic morphologic factor in human cutaneous melanocytic tumors, and it is measured, with an ocular micrometer, from the top of the granular layer of the epidermis (or, when the surface is ulcerated, from the base of the ulcer) to the deepest invasive cell across the broad base of the tumor. 5,17 The American Joint Committee on Cancer (AJCC) established a cutoff of 1 mm: thinner melanomas usually have a very good prognosis compared to the thicker ones, with a disease-free survival close to 100%. 3,5 Since most melanocytic tumors in dogs are confined to the dermis with no involvement of the epidermis, a study of 65 dogs evaluated cutaneous melanocytic tumor thickness from the more superficial to the deepest layer of tumor cells 11 and demonstrated a greater thickness of tumors with malignant behavior (average thickness of 0.73 cm in tumors with benign behavior vs average thickness of 1.42 cm in tumors with malignant behavior); however, the authors of the study could not identify a cutoff with prognostic value.
In 1969, Clark et al 2 introduced a classification system based on the anatomical level of invasion through the layers of the dermis in human cutaneous melanomas (Clark level). These authors demonstrated that the level of invasion was associated with the frequency of metastasis and death, 14 although at present, the utility of the Clark level is still debated. 12,15,17
To our knowledge, no studies investigating the prognostic value of tumor thickness and the applicability of the Clark level in canine cutaneous melanocytic tumors have been performed.
Therefore, the aim of our study was to examine the applicability and usefulness of assessing tumor thickness and the Clark level in canine cutaneous melanocytic tumors and their association with survival and hazard of death. Furthermore, we evaluated a more convenient system for measuring the tumor thickness that can be adopted in any laboratory, since not all laboratories have an ocular micrometer for measurements.
Materials and Methods
Selection of Samples and Histology
We retrospectively examined 77 formalin-fixed, paraffin embedded samples of primary cutaneous melanocytic tumors from dogs, selected from the archives of the Department of Veterinary Medicine at University of Perugia (Italy), sent to the Veterinary Pathology Service from 2009 to 2016. Selected samples were reexamined by 4 operators (C.B., I.P., S.S., and S.R.) to evaluate histological features that have potential prognostic value in canine melanocytic tumors according to the recent literature. 19 When a discrepancy emerged, the pathologists (C.B., I.P., and S.S.) reviewed the cases and came to a final consensus. The initial histological diagnosis was confirmed in all cases after reexamination. The melanocytic origin of nonpigmented spindle cell tumors was confirmed by immunohistochemistry for Melan A and PNL2. The volume of each tumor was estimated using the major diameter (measured on the formalin-fixed sample) and calculating the volume of a sphere, as previously described. 9
The evaluation included the following: predominant cell type (polygonal/epithelioid, spindle/fibromatous, mixed, dendritic, balloon type); degree of pigmentation, evaluated as intensity of pigmentation (absent/mild/marked) and percentage of pigmented cells (0%, ≤50%, and >50%); presence of necrosis; presence of ulceration; presence of intraepithelial neoplastic cells; cellular pleomorphism (mild/moderate/marked); nuclear atypia according to Spangler and Kass,
21
subsequently stratified into 2 groups (<20% and ≥20%) as previously described;
19
mitotic count evaluated on 10 contiguous high-power fields (field number of the ocular of 22mm and a 40× objective),
13
starting from hotspots and avoiding areas with necrosis or severe inflammation; vascular invasion; excision margins: sufficient (when the tumor does not reach the boundaries of the histological section) or not sufficient.
Since most melanocytic neoplasms in dogs are exclusively intradermal, with no epidermal component, tumor thickness instead of Breslow thickness was considered. 11 An ocular micrometer was used to calculate the thickness of the tumor on the histological section, from the most superficial cell layer to the deepest point of invasion; in ulcerated tumors, the thickness was measured from the base of the ulcer. However, to have a convenient system that could be used in any histology laboratory, we also measured the depth of the tumor with a ruler that was applied on the surface of the glass slide, perpendicularly to the epidermis (Fig. 1).
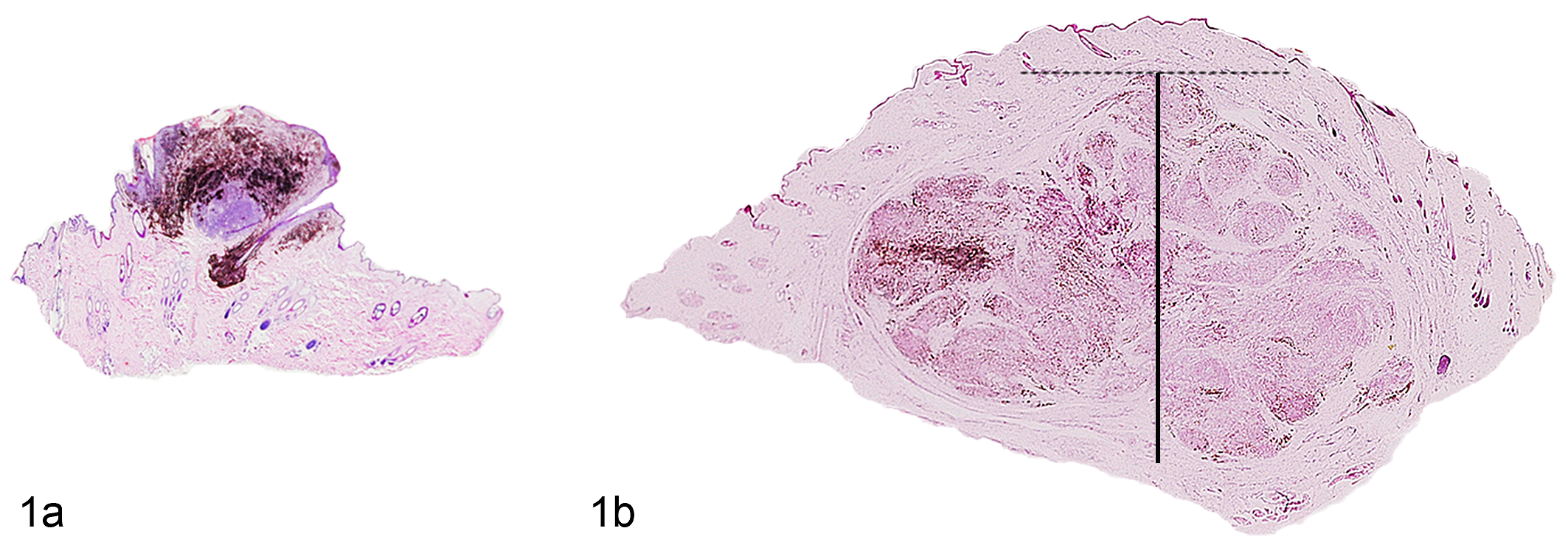
Melanocytic tumors, skin, dog. Representative differences of tumor thickness between (a) melanocytoma, with lower tumor thickness, and (b) melanoma, with greater tumor thickness. Hematoxylin eosin. The lines show how to apply the ruler (continuous line) on the glass surface, perpendicular to the epidermis and starting from the top of the lesion (dashed line).
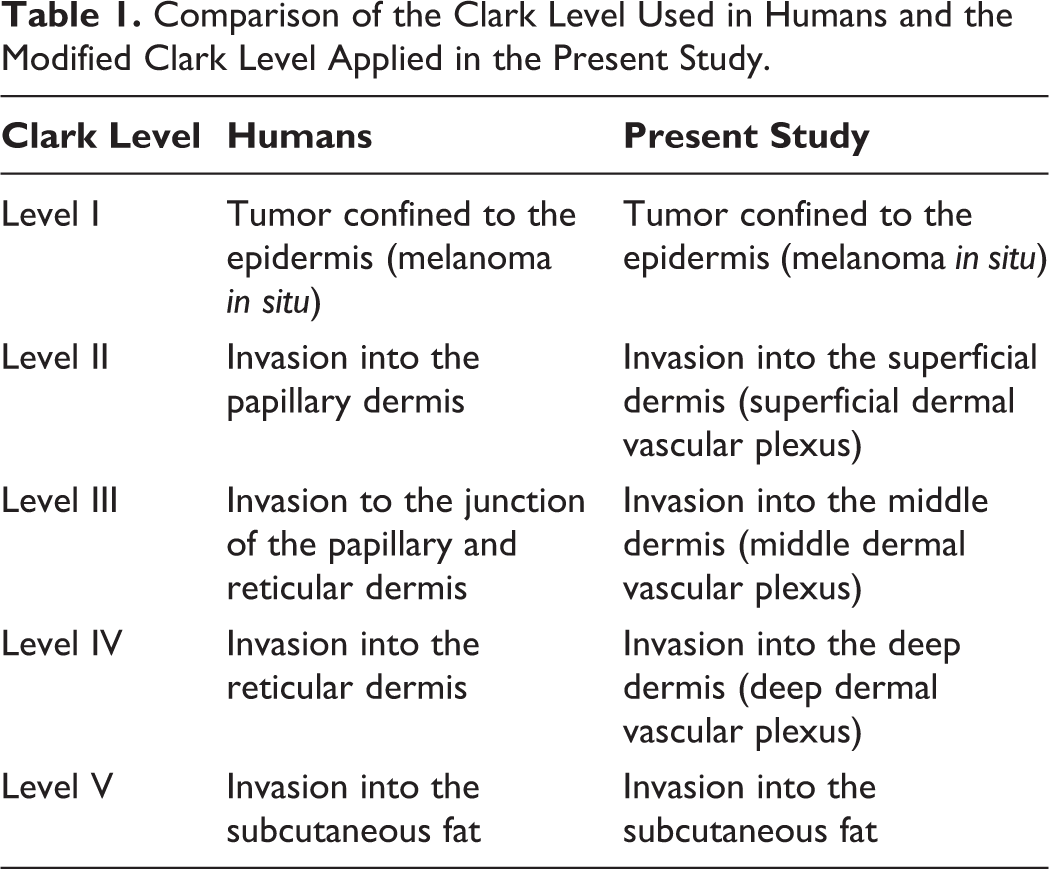
Even though its utility is still controversial in human medicine, 5,17 we investigated the usefulness of the Clark level in cutaneous canine melanocytic tumors: since in the dog the papillary dermis is not present, we elaborated a modified Clark level. Thus, to evaluate our samples, we considered the 3 levels in which the dermis is traditionally divided in animals, 4 and we translated it, maintaining the 5 reference levels established in humans as shown in Table 1 and Fig. 2. Furthermore, we stratified cases into 2 groups (I–III level and IV–V level), which were analyzed as a dichotomous variable.
Comparison of the Clark Level Used in Humans and the Modified Clark Level Applied in the Present Study.
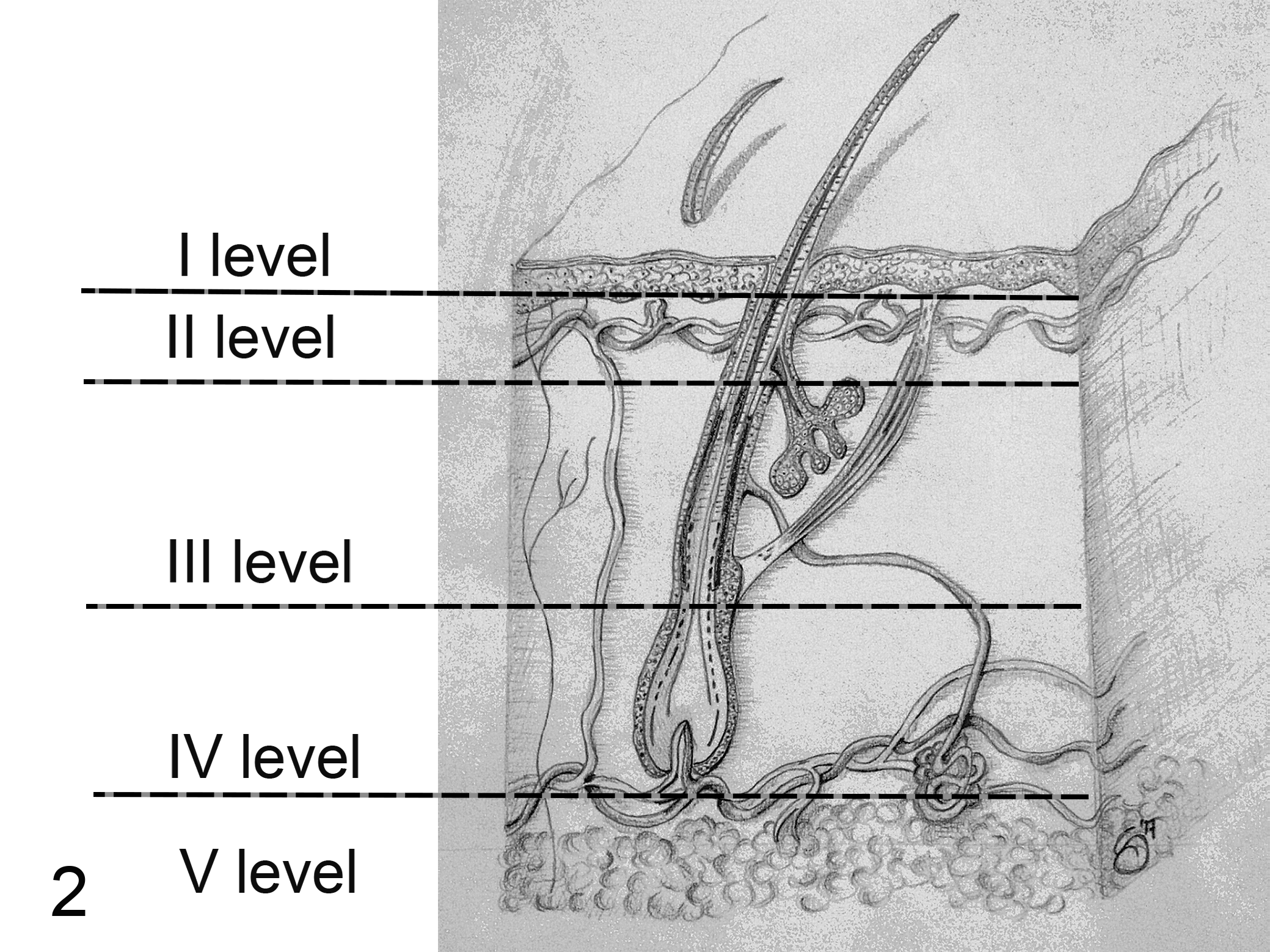
Skin, dog, drawing. Scheme of the modified Clark level. I level: tumor is confined to epidermis; II level: tumor invades the superficial dermis (between the epidermis and the end of sebaceous gland ducts); III level: tumor invades the middle dermis (between the end of sebaceous gland ducts and the insertion of arrector pili muscle); IV level: tumor invades the deep dermis (between the insertion of arrector pili muscle and the subcutis); V level: tumor invades the subcutaneous fat.
After the case selection, we retrospectively asked the clinicians for the follow-up by telephonic interview, investigating whether the dogs were alive, were in therapy, or had presented local recurrence, regional lymph node metastasis, or distant metastasis and the cause of death. Only dogs that did not receive chemotherapy were included in the study. We considered the clinical outcome “unfavorable” when the patient died of melanoma or “favorable” when it was alive or dead because of causes not related to the tumor.
Statistical Analysis
Descriptive statistics were used for clinical data. Values were expressed as number and percentage or median with range. As the assumptions of normality were not met, we used the Mann-Whitney U test and median with interquartile range (IQR) to analyze and present continuous variables. One-sample χ2 test was used to evaluate the distribution of predominant cell type within each diagnosis. Categorical data were compared using χ2 test of independence or Fisher’s exact test and z test. For multiple comparisons, Bonferroni-adjusted P values are shown. Receiver operating characteristic (ROC) curve analysis and Youden Index were performed to identify a cutoff for tumor thickness according to histological diagnosis, clinical outcome (died because of the tumor, or alive or dead because of other causes), and presence of recurrence/metastasis.
We used Kaplan-Meier curves and log-rank test to compare overall survival according to diagnosis. Prognostic significance of the variables was evaluated by the univariate Cox proportional hazard model (Cox regression). Variables with P < .05 on the univariate analysis were entered into the multivariable model using the backward selection method. Results were expressed as hazard ratio (HR) with corresponding 95% confidence interval (CI) and P values (from Wald statistics).
To explore the difference between the 2 methods of tumor thickness evaluation, Bland-Altman plot of the difference against mean and the intraclass correlation coefficient (ICC) test were also applied.
Statistical analysis was performed using SPSS 23.0 (SPSS, Inc, an IBM Company, Chicago, IL), GraphPad version 5.0 (GraphPad Software, San Diego, CA), and the software R (R version 3.4.3; R Foundation for Statistical Computing, Vienna, Austria). 16 P values ≤.05 were considered statistically significant.
The row data analyzed in this study are not available as supplemental materials.
Results
Sample Population
The study included cutaneous melanocytic tumors from 77 dogs, including 42 melanomas and 35 melanocytomas. Based on incomplete signalment information, most of the dogs were mixed breed (23/72, 31.9%), followed by German Shepherd (10/72, 13.9%), Rottweiler (5/72, 6.9%), Labrador and Pinscher (each were 4/72, 5.56%), and others (26/72, 36.1%). The median age at the time of diagnosis was 10 years (range, 1–15 years; IQR, 7–11 years), and dogs with cutaneous melanocytoma were usually younger than those with melanoma (P = .037). Data regarding clinical and macroscopic features according to diagnosis are summarized in Supplemental Table S1.
Histological Data
Histological diagnosis was significantly associated with several of the features investigated, such as the mitotic count, nuclear atypia, nuclear atypia stratified into 2 groups (<20% or ≥20%), cellular pleomorphism, presence of ulceration, presence of necrosis, and percentage of pigmented cells, as well as with clinical outcome and the presence of recurrence/metastasis (Suppl. Table S2).
Tumor thickness was significantly associated with histological diagnosis, being greater in melanomas than in melanocytomas. This was true independently of the measurement method used (P ≤ .001 and P = .001, for measurements obtained by ocular micrometer and by a ruler respectively; Mann-Whitney U test; Table 2).
Associations Between Tumor Thickness (Measured With an Ocular Micrometer and a Ruler) and Histological Diagnosis, Clinical Outcome, and Presence of Recurrence/Metastasis in Canine Melanocytic Tumors.a

Values in bold are statistically significant (a = .05).
aMann-Whitney U test.
Abbreviation: IQR, interquartile range.
Using the ruler, the tumor thickness cutoff to distinguish cutaneous melanomas from melanocytomas as defined by histopathology was 0.45 cm (ROC curve analysis, area under the curve [AUC] = 0.824; 95% CI, 0.719–0.928; P < .001); this cutoff had a sensitivity of 87% and a specificity of 64%.
In addition, tumor thickness was associated with clinical outcome and presence of recurrence/metastasis, being greater in animals that died of melanoma or that developed recurrence/metastasis. This was true independently of the measurement method used (Table 2). It is worth noticing that neither the tumor thickness nor the presence of metastasis/recurrence had any association with the completeness of the excision margins (P = .187 and P = .285, respectively).
Agreement between the 2 methods for evaluation of tumor thickness was excellent: 10 ICC was 0.987 (95% CI, 0.972–0.994; P < .001), and the Bland-Altman analysis of measurement with the ocular micrometer vs measurement with the ruler showed a bias of –0.02067 ± 0.1169 (95% limits of agreement = –0.2498 to 0.2085; Fig. 3).
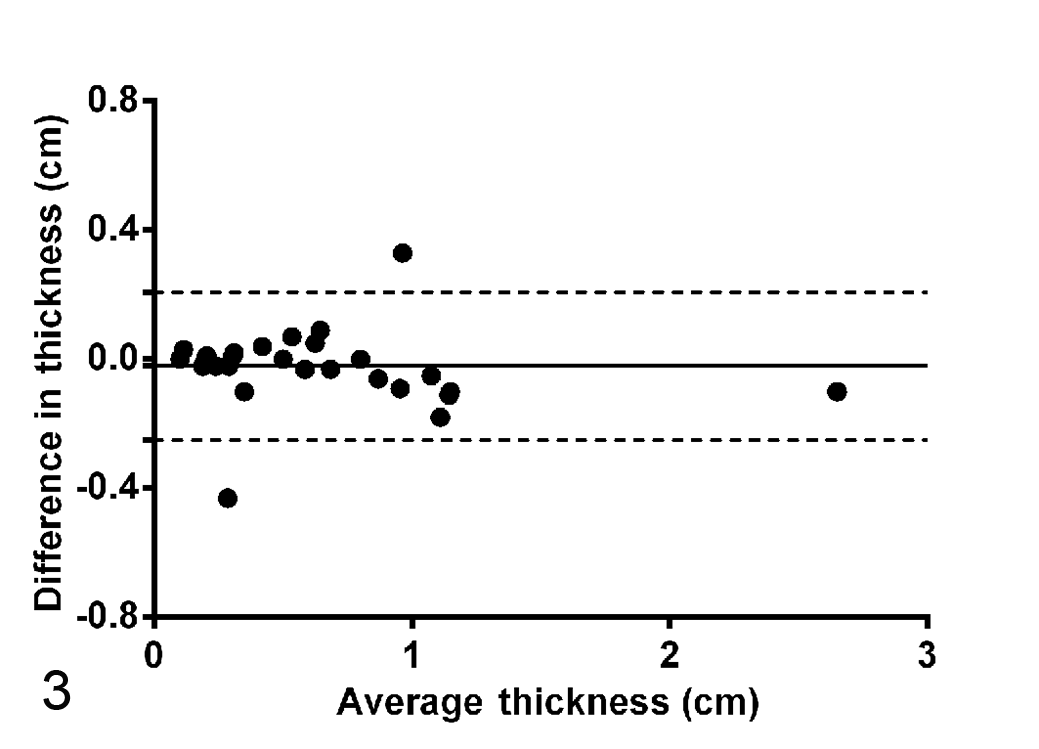
Bland-Altman analysis. Comparing measurement of tumor thickness with the ocular micrometer vs measurement with the ruler. Plot of the difference against mean, show.
Modified Clark level was significantly associated with the histological diagnosis when classified as a 5-level variable (P = .001) or when classified as a dichotomous variable (P < .001). Cutaneous melanocytomas were more frequently modified Clark level <IV (21/34, 61.8%) than melanomas (4/30, 13.3%). However, the Clark level was not significantly associated with clinical outcome or with the presence of recurrence/metastasis (P = .670 and P = 1, respectively; Table 3).
Associations of Modified Clark Level (5-Level or Dichotomous) With Histologic Diagnosis, Clinical Outcome, and Presence of Recurrence/Metastasis in Canine Melanocytic Tumors.
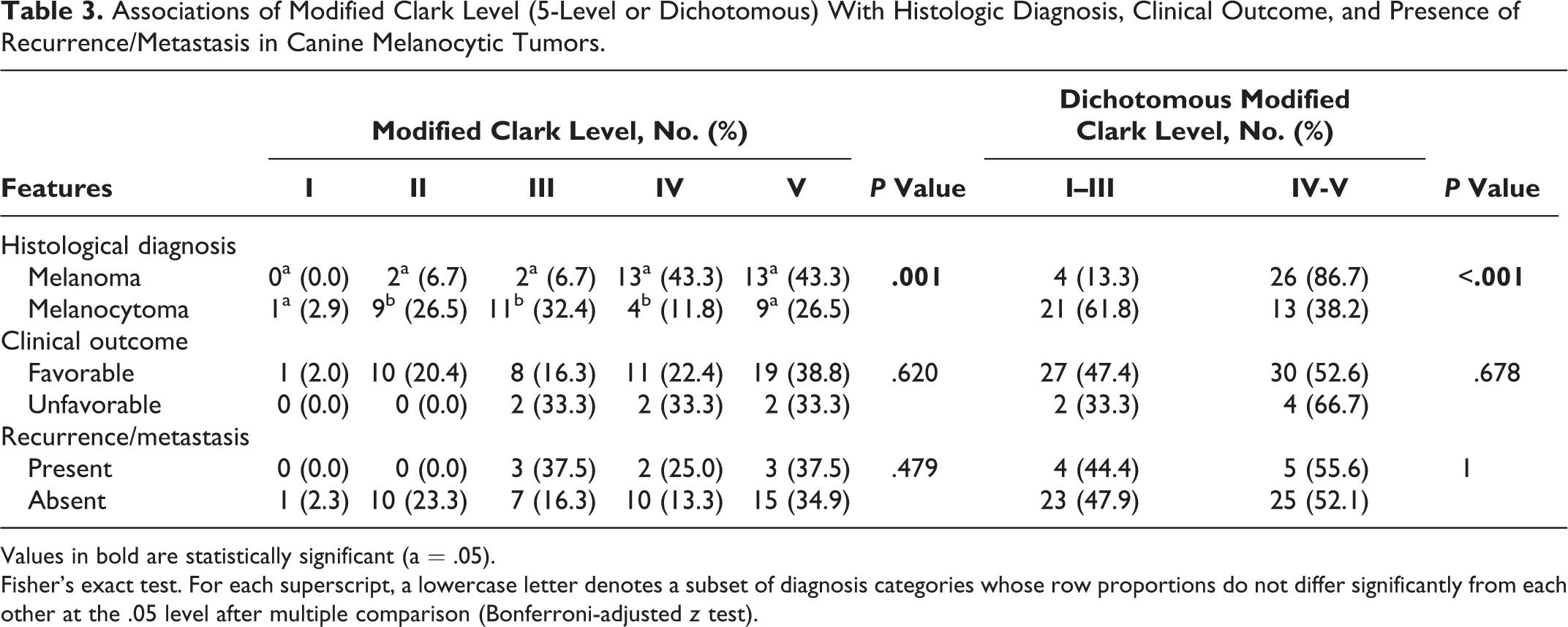
Values in bold are statistically significant (a = .05).
Fisher’s exact test. For each superscript, a lowercase letter denotes a subset of diagnosis categories whose row proportions do not differ significantly from each other at the .05 level after multiple comparison (Bonferroni-adjusted z test).
Survival Analysis: Overall Survival
Follow-up at the end of the study ranged from 207 to 2823 days, with a median follow-up of 982 days (IQR, 694–1273). In 11 of 77 cases (14.3%), dogs were lost to follow-up. Seven dogs died because of the tumor (9.1%), and all had an original diagnosis of melanoma. No dogs with melanocytoma died. Median survival time was not reached for cutaneous melanoma or for melanocytoma. Kaplan-Meier curves and log-rank test showed a significant difference in survival time according to histological diagnosis (P = .011; Fig. 4a); indeed, the estimated 1-year survival probability was 77.0% ± 8.0% and 100% ± 0% for dogs with cutaneous melanomas and cutaneous melanocytomas, respectively.
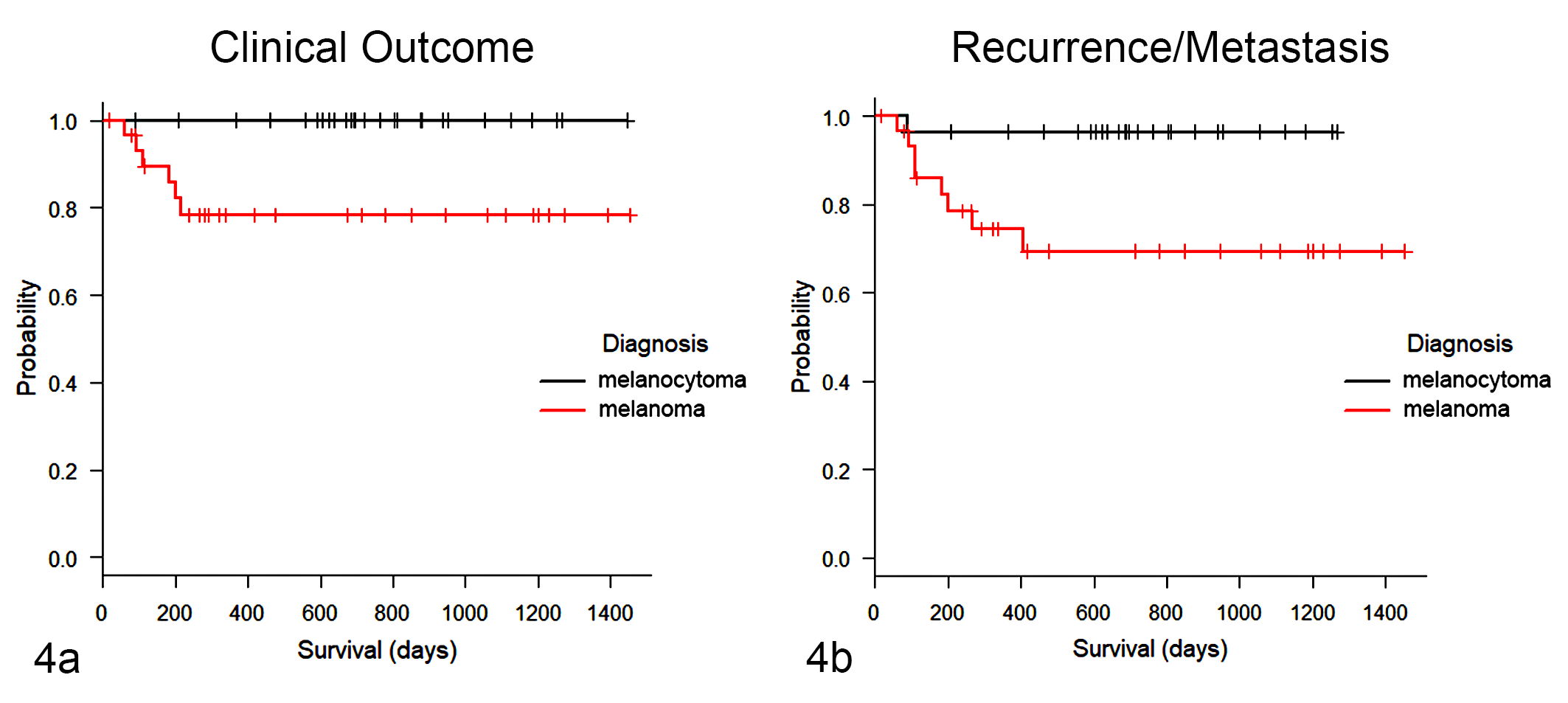
Kaplan-Meier curves according to histological diagnosis. (a) The overall survival time in dogs diagnosed with melanoma is significantly lower than in those with melanocytoma (P = .011). (b) The disease-free time in dogs diagnosed with melanoma is significantly lower than that in dogs with melanocytoma (P = .012).
Univariate Cox proportional regression analysis showed that tumor thickness, mitotic count, nuclear atypia, and nuclear atypia ≥20% were prognostic factors for overall survival (Table 4), while 5-level modified Clark level and modified Clark level classified as a dichotomous variable were not prognostic factors (overall P = .670 and P = .864, respectively). Multivariable analysis results are shown in Supplemental Table S3.
Relationship Between Histologic Factors and Overall Survival in Dogs With Cutaneous Melanocytic Neoplasms, Based on Univariate Cox Proportional Regression Analysis.
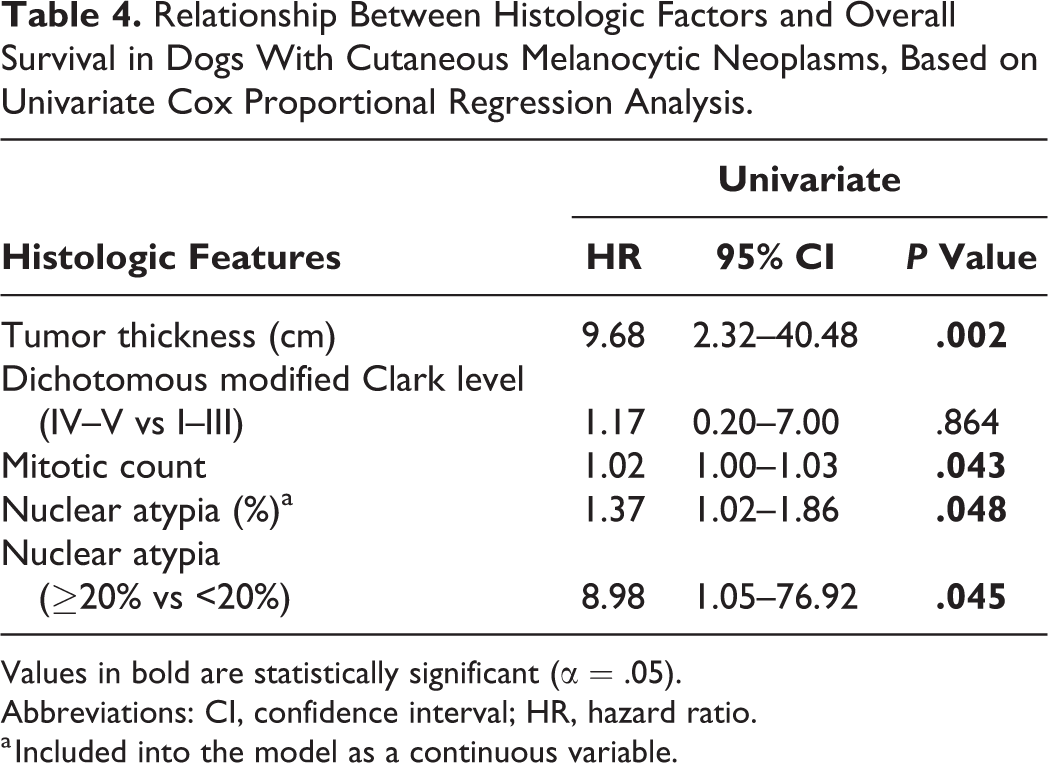
Values in bold are statistically significant (α = .05).
Abbreviations: CI, confidence interval; HR, hazard ratio.
a Included into the model as a continuous variable.
Using a ruler, a tumor thickness cutoff of 0.95 cm discriminated between favorable and unfavorable (tumor-related death) clinical outcome (sensitivity = 100%, specificity = 86%; ROC curve analysis, AUC = 0.886; 95% CI, 0.795–0.977; P = .005). Dogs with tumor thickness >0.95 cm had a shorter overall survival than those with tumor thickness ≤0.95 cm (P < .001; Fig. 5a,b). In particular, the 1-year estimated survival probability was 45.0% ± 18.8% for dogs with tumor thickness >0.95 cm and 100% ± 0% for those in which it was ≤0.95 cm, since no animals died in this group.
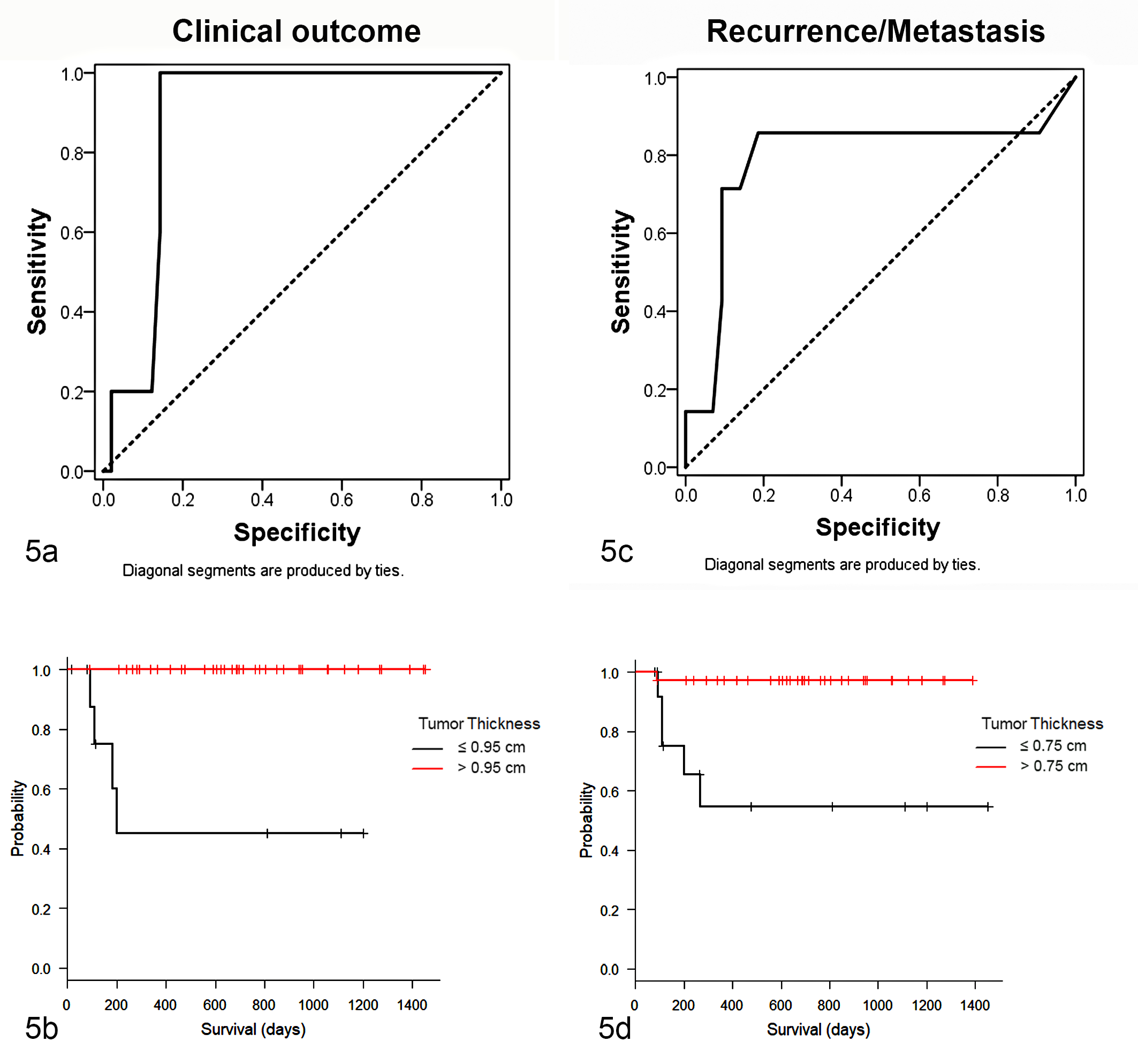
Receiver operating characteristic (ROC) and Kaplan-Meier curves according to tumor thickness cutoff values identified. In both cases, the sensitivity and specificity of the cutoffs were high. (a) ROC curve analysis depending on clinical outcome (ie, died because of the tumor, or alive or dead because of other causes; cutoff = 0.95 cm). (b) Kaplan-Meier curves show a shorter overall survival time in dogs with tumor thickness >0.95 cm (P < .001). (c) ROC curve analysis depending on presence of recurrence/metastasis (cutoff = 0.75 cm). (d) Kaplan-Meier curves show a shorter disease-free time in dogs with tumor thickness >0.75 cm (P < .001).
Survival Analysis: Disease-Free Interval
At the end of the study, 11 of 77 dogs had developed recurrence or metastasis (14.3%); 10 of the 11 animals with recurrence/metastasis had cutaneous melanoma, while 1 had cutaneous melanocytomas. Median disease-free time was not reached for cutaneous melanoma or melanocytoma. Kaplan-Meier curves and log-rank test showed a significant difference in disease-free time according to histological diagnosis (P = .012; Fig. 4b); indeed, the 1-year estimated probability of not developing recurrence or metastasis was 74.0% ± 9% and 96.0% ± 4%, respectively, for cutaneous melanomas and cutaneous melanocytomas.
Univariate Cox proportional regression analysis showed that tumor thickness, nuclear atypia, and nuclear atypia ≥20% were prognostic factors for disease-free time (Table 5), while 5-level modified Clark level and modified Clark level classified as a dichotomous variable were not prognostic factors (overall P = .354 and P = .993, respectively). Multivariable analysis results are shown in Supplemental Table S4.
Relationship Between Histologic Factors and Disease-Free Time in Dogs With Cutaneous Melanocytic Neoplasms, Based on Univariate Cox Proportional Regression Analysis.
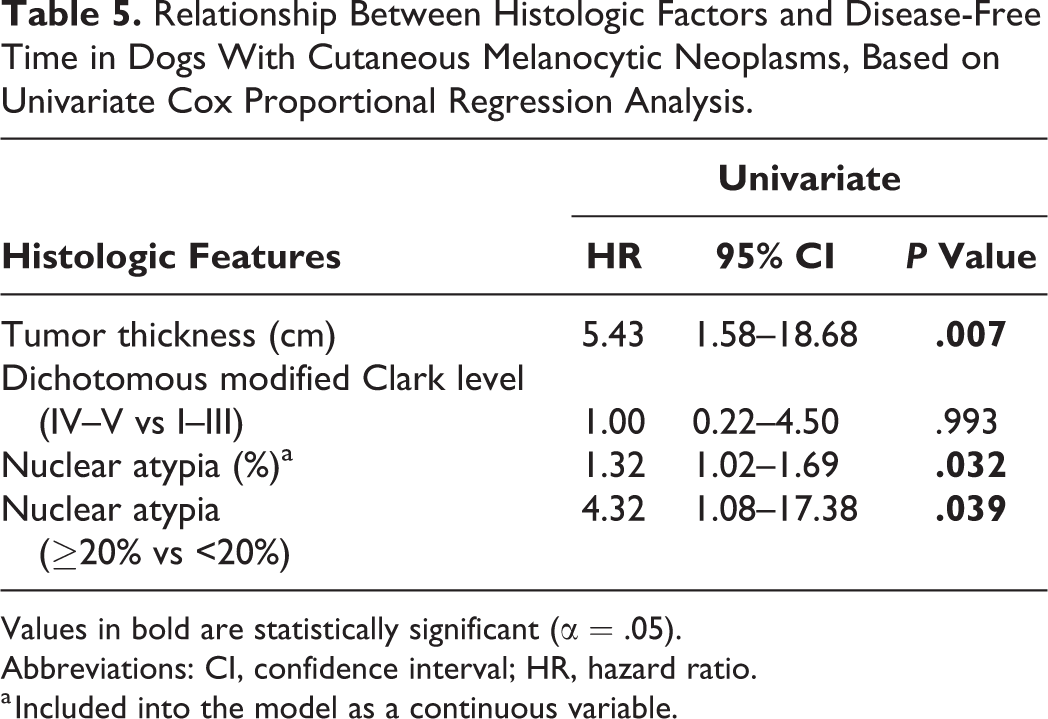
Values in bold are statistically significant (α = .05).
Abbreviations: CI, confidence interval; HR, hazard ratio.
a Included into the model as a continuous variable.
Using a ruler, a tumor thickness cutoff of 0.75 cm discriminated the possibility of having or not having recurrence/metastasis (sensitivity = 86%, specificity = 81%; ROC curve analysis, AUC = 0.886; 95% CI, 0.795–0.977; P = .005). Dogs with tumor thickness >0.75 cm had a shorter disease-free time than those with tumor thickness ≤0.75 (P < .001; Fig. 5c,d). In particular, the 1-year estimated probability of not developing recurrence/metastasis was 54.7% ± 15.4% and 97.2% ± 2.7% for dogs with tumor thickness >0.75 cm and ≤0.75 cm, respectively.
Discussion
In humans, Breslow thickness is used as a primary parameter for staging cutaneous melanomas, and the final prognosis is better defined using stage modifiers, such as ulceration, mitotic count, Clark level, and anatomic site. 3,6,18 Considering the great biological variability of canine melanocytic tumors, 1 it would be advisable to define a similar system for use in dogs to predict prognosis.
In humans, Breslow thickness is an independent prognostic morphologic factor in the assessment of cutaneous melanocytic tumors, and it is normally measured with an ocular micrometer from the granular cell layer. 5,17 However, since most of the melanocytic tumors in dogs are within the dermis, the evaluation of tumor thickness seems to be more appropriate. 11 We demonstrated an association between the histological diagnosis and tumor thickness: cutaneous melanocytomas were significantly thinner than melanomas. This association was demonstrated for both measurement methods adopted in this study—namely, the ocular micrometer and the ruler. Indeed, through the Bland-Altman plot and ICC, we showed that the agreement between these 2 methods was excellent, so we can suggest that either can be used.
By ROC curve analysis, we found that a cutoff of 0.45 cm using the ruler could assist in distinguishing cutaneous melanomas and melanocytomas in challenging diagnostic situations. However, it is important to keep in mind that sensitivity (86%) and specificity (64%) associated with this value are not optimal with the possibility of false-positive results.
Moreover, tumor thickness was greater in animals that died of melanoma, as previously shown, 11 and in those that developed recurrence/metastasis.
Tumor thickness was associated with overall survival time, disease-free time, hazard of death, and hazard of developing recurrence/metastasis, thus demonstrating its prognostic importance. In particular, in univariate analysis, it has been shown that dogs with greater tumor thickness have nearly a 10 times higher hazard of death and more than a 5 times higher hazard to develop recurrence/metastasis than dogs with thinner tumors.
Since multiple variables were associated with the prognosis, we carried out multivariate analysis to determine their influence on survival. 22 Unfortunately, the assumptions for performing multivariate analysis were not met since the number of “events” (tumor-related death or recurrence/metastasis) and the sample size in our study were limited. Consequently, the results should be taken as a preliminary attempt suggesting that the tumor thickness might be an independent prognostic factor, but it needs further confirmation.
An optimal tumor thickness cutoff of 0.95 cm and 0.75 cm, measured with the ruler, could be advantageously used to discriminate a favorable from an unfavorable (tumor-related death) outcome and the possibility of having recurrence/metastasis, respectively. These cutoff values are both associated with high sensitivity and specificity, albeit with some risk of false-positive and false-negative outcomes. Patients with tumor thickness >0.95 cm and >0.75 cm had, respectively, a shorter overall survival and shorter disease-free time than those with tumor thickness ≤0.95 cm and ≤0.75 cm. The cutoff value associated with the outcome (0.95 cm) seems to be in contrast with previously reported results; 11 however, Lacroux and colleagues 11 used a different approach, calculating the median thickness of tumors with benign behavior (0.73 cm) that were generally thinner than tumors with malignant behavior (1.43 cm), but they did not calculate a prognostic cutoff.
Modified Clark level was significantly associated with the histological diagnosis, being higher in melanomas than in melanocytomas; this was particularly true when the Clark level was classified as a dichotomous variable (I–III level and IV–V level). Therefore, even though in humans the importance of the Clark level is still debated and partially overtaken by the Breslow thickness, in dogs it could be a useful feature to evaluate in routine diagnostics, since the invasion to the deep dermis (IV level) could support a histological diagnosis of malignancy in ambiguous cases. Nonetheless, a modified Clark level was not associated with clinical outcome or presence of recurrence/metastasis and did not show a prognostic significance in our study.
Some of our results confirm previous reports, 19 since histological features such as mitotic count, nuclear atypia, nuclear atypia stratified into 2 groups (<20% or ≥20%), cellular pleomorphism, presence of ulceration, presence of necrosis, and percentage of pigmented cells were associated with histological diagnosis, and the histological diagnosis was associated with clinical outcome and the presence of recurrence metastasis. Also, overall survival time and disease-free time were shorter in cutaneous melanomas than in melanocytomas. However, by univariate analysis, only tumor thickness, mitotic count, and nuclear atypia had prognostic value in canine cutaneous melanocytic tumors. This is likely a consequence of the limitations in our study, since not only the time of follow-up but also the sample size and the number of “events” (death caused by the tumor) influenced the survival analysis.
Some limitations should be taken into account. 20,22 First, since we performed a retrospective cohort study, the presence of potential confounders or biases cannot be completely ruled out. In addition, the sample size was relatively limited and, as often happens in veterinary medicine, signalment and follow-up information were sometimes incomplete. Finally, the outcomes of interest (tumor-related death or recurrence/metastasis) were not numerous due to the relatively benign clinical behavior of canine cutaneous melanomas. These limitations could have affected the results, making it more difficult to demonstrate or confirm the prognostic significance of some variables.
In conclusion, our study revealed that tumor thickness is a prognostic factor in univariate analysis, and its evaluation could be advantageous in the assessment of canine cutaneous melanocytic tumors. Thus, we would suggest taking it into account together with the other prognostic factors for achieving a greater accuracy. Dogs with thicker tumors have shorter overall survival and disease-free time, and a cutoff of 0.95 cm and 0.75 cm, measured with the ruler, can be used to define a higher hazard of an unfavorable outcome and developing recurrence/metastasis, respectively. In addition, tumor thickness was associated with histological diagnosis, and the cutoff of 0.45 cm could be used for supporting the diagnosis of malignancy in difficult cases. We also demonstrated that the tumor thickness can be easily measured in every laboratory with a ruler when the ocular micrometer is not available.
Despite not having a prognostic significance, we suggest the use of the modified Clark level (especially when dichotomized) in the assessment of histological diagnosis, particularly in borderline cases, since it is significantly associated with the histological diagnosis and reflects the degree of tumor invasion. Further studies, including a wider sample population, are essential for confirming the prognostic value of tumor thickness and clarifying the usefulness of the modified Clark level in canine cutaneous melanocytic tumors.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818798094 - Tumor Thickness and Modified Clark Level in Canine Cutaneous Melanocytic Tumors
Supplemental Material, DS1_VET_10.1177_0300985818798094 for Tumor Thickness and Modified Clark Level in Canine Cutaneous Melanocytic Tumors by Serenella Silvestri, Ilaria Porcellato, Luca Mechelli, Laura Menchetti, Sofia Rapastella, and Chiara Brachelente in Veterinary Pathology
Footnotes
Acknowledgements
We are grateful to Luca Stefanelli for technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
