Abstract
Feline neovascular vitreoretinopathy (FNV) is a newly recognized rare condition affecting kittens and young domestic cats. This study investigated the clinical and pathologic findings in 22 cats with FNV. In affected cats, ophthalmoscopy of the fundus (when visible) revealed avascular peripheral retinae and epiretinal vascular membranes. Frequent nonspecific clinical findings were buphthalmos (n = 21), medically uncontrollable glaucoma (n = 22), and lenticular abnormalities (n = 13). Anterior segment dysgenesis (ASD) was detected clinically in affected cats (n = 6). The fellow eye was affected in 11 of 18 cats to a variable degree or appeared clinically normal in 7 of 18 cats. The globes were examined histologically and using immunohistochemistry for vimentin, glial fibrillary acidic protein (GFAP), synaptophysin, neurofilament, laminin, factor VIII–related antigen (FVIII-RA), and smooth muscle actin (SMA). Histologically, diagnostic features included laminin-positive epiretinal vascular membranes affecting the central retina, with an avascular peripheral retina and gliosis. Enucleated globes exhibited multiple additional abnormalities, including corneal disease (n = 15), anterior segment dysgenesis (n = 21), lymphoplasmacytic anterior uveitis (n = 19), peripheral anterior synechiae (n = 20), retinal degeneration (n = 22), and retinal detachment (n = 19). Gliotic retinae labeled strongly for GFAP and vimentin with reduced expression of synaptophysin and neurofilament, consistent with degeneration or lack of differentiation. While an avascular peripheral retina and epiretinal fibrovascular membranes are also salient features of retinopathy of prematurity, there is no evidence to support hyperoxic damage in cats with FNV. The cause remains unknown.
Keywords
Feline glaucoma is most often acquired and caused by uveitis, trauma, or neoplasia. Primary glaucoma is less frequently encountered in cats. 5,11 Congenital glaucoma is rare in cats and may result from primary genetic or complex developmental abnormalities. 8,11 A subset of globes from kittens and young domestic cats clinically diagnosed with glaucoma that exhibited distinctive histopathological features, including epiretinal vascularized membranes and avascular, gliotic peripheral retinae, was identified within the archives and database of the Comparative Ocular Pathology Laboratory of Wisconsin (COPLOW).
The objectives of this study were to describe the clinical and pathologic ocular findings in kittens and young cats diagnosed with a condition that we have termed feline neovascular vitreoretinopathy (FNV).
Materials and Methods
The COPLOW holds an archive of approximately 11 000 formalin-fixed, paraffin-embedded feline ocular tissues. A search of the database was performed for cases diagnosed with “feline neovascular vitreoretinopathy.” A diagnosis of FNV was made based on histopathological features that included an avascular peripheral retina with gliosis and epiretinal neovascularization of the central retina. Tissue submission forms were reviewed and included history, signalment, laterality, and clinical presentation. Referring veterinary ophthalmologists were contacted by telephone or email, and patient medical records were obtained when available. Based on submission forms and available medical records, included animals had no known history of trauma or systemic, infectious, or neoplastic intraocular disease.
Binomial test was used to examine whether proportions of single dichotomous variables (ie, frequency of diagnosis as a function of laterality and sex) equaled those of the presumed population value. Significance was set at P < .05. Statistical analysis was performed using IBM SPSS Statistics for Windows (version 24.0; SPSS, Inc, an IBM Company, Chicago, IL).
Histopathology
Histological sections were routinely prepared from archived formalin-fixed, paraffin-embedded tissues from 23 globes from 22 cats and stained with hematoxylin and eosin. Slides were reviewed by an experienced veterinary ocular pathologist board certified by the American College of Veterinary Pathologists (R.R.D.). Histologic features systematically assessed and documented, as appropriate, for each case included corneal abnormalities, presence of cells or protein in the anterior chamber, preiridal fibrovascular membranes, anterior segment dysgenesis, angle recession, peripheral anterior synechiae, ectropion uveae, uveitis, lenticular abnormalities, hyphema or vitreal hemorrhage, retinal detachment, retinal atrophy, optic nerve cupping, and gliosis. True retinal detachment was distinguished from artifact based on morphological changes in the retinal pigment epithelium, degeneration of photoreceptor inner and outer segments, and the presence of debris or cells in the subretinal space. 2
Immunohistochemistry
Immunohistochemical staining was performed on selected, representative, affected globes and included labeling for glial fibrillary acidic protein (GFAP) (n = 7), neurofilament (n = 4), synaptophysin (n = 4), vimentin (n = 4), laminin (n = 9), factor VIII–related antigen (FVIII-RA) (n = 7), and smooth muscle actin (SMA) (n = 7). Immunohistochemical labeling for GFAP, neurofilament, synaptophysin, and vimentin was also performed on 3 control globes: 1 normal globe from a 12-year-old female spayed domestic longhaired cat, a globe with retinal detachment not associated with glaucoma from a 6-month-old female spayed domestic shorthaired cat, and a globe exhibiting primary congenital glaucoma from a 7-month-old male neutered domestic shorthaired cat.
Immunohistochemical labeling was performed using standard immunoperoxidase techniques on an automated immunostainer (Lab Vision 720-2D Autostainer; Thermo Fisher Scientific, Fremont, CA), with polyclonal or monoclonal antibodies (Table 1), after deparaffinization and heat-induced antigen retrieval, performed using citrate buffer (pH 6.0) for 20 minutes (Lab Vision PT module; Thermo Fisher Scientific). Diaminobenzidine (DAB Peroxidase Substrate Kit; Vector Laboratories, Burlingame, CA) was used as chromogen and Mayer’s hematoxylin as counterstain. Laminin immunohistochemistry was performed as previously described. 19
Antibodies Used for Immunohistochemistry in This Study.
Factor VIII-RA immunohistochemistry was performed using standard techniques on an automated stainer (Discovery; Ventana Medical Systems, Tucson, AZ). Heat-induced epitope retrieval with citrate buffer (pH 6.0) for 30 minutes was used. The sections were incubated in prediluted primary antibody. A prediluted, universal biotinylated secondary antibody and a DAB MAP detection kit (Ventana Medical Systems) were used to detect the immunoreactive complexes.
SMA immunohistochemistry was performed as previously described using standard techniques on an automated immunostainer (BioGenex i6000, San Ramon, CA). 19 Heat-induced epitope retrieval with citrate buffer (pH 6.0) was performed in a Decloaker (BIOCARE Medical, Walnut Creek, CA). Epitope retrieval was followed by blocking with Power Block (BioGenex blocking serum) and avidin and biotin block (Signet, Dedham, MA). Slides were incubated with prediluted primary antibody. The BioGenex Supersensitive Multilink kit was used for detection (Nova Red Chromogen; Vector Laboratories). Incubation time was 30 minutes for all antibodies.
Negative controls for each section omitting the primary antibody were incorporated in each run. Positive controls included histological sections of small intestine for vimentin, spinal cord or brain for GFAP, neurofilament and synaptophysin, lung or kidney for laminin, skin for FVIII-RA, and colon for SMA.
Results
Study Population
Twenty-three globes from 22 cats diagnosed with FNV were retrieved in a database search. Of these 23 globes, 1 globe of a bilaterally affected cat sustained severe ocular trauma and was excluded from the study; 22 globes from 22 cats met all inclusion criteria. The median age at histologic diagnosis of FNV was 6 months (range, 4–58 months). Thirteen cats were female (9 spayed, 4 intact), 8 cats were male (5 neutered and 3 intact), and sex was not reported for 1 cat. Although females were more frequent, the proportion of females of 0.62 was not greater than the expected of 0.50 (P = .383, 2-sided binomial test). Nineteen cats were domestic shorthair, 2 were Persian, and 1 was a British shorthair. Of the enucleated globes, 16 were left and 5 were right eyes (laterality was not specified for 1). The proportion of left eyes of 0.73 was significantly greater than the expected 0.50 (P = .027, 2-sided binomial test).
Clinical Findings
The ocular disease had been present prior to 1 year of age or since adoption for 18 of the 19 cats, in which the onset or duration was described in the clinical history. In 9 cats, the clinician noted that a congenital disease was suspected.
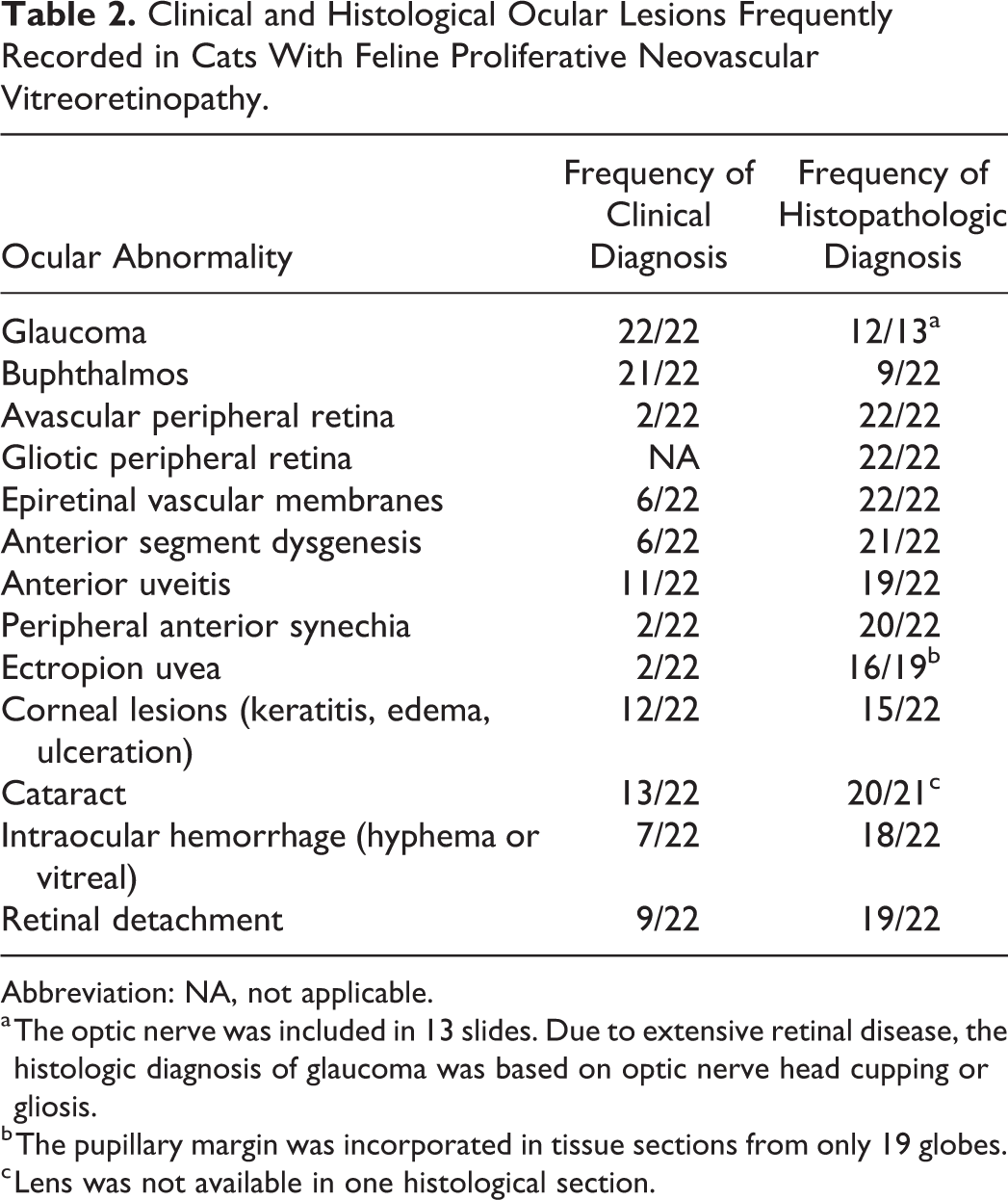
A summary of common clinical and histopathological findings is presented in Table 2. All cats were examined by board-certified veterinary ophthalmologists, who had made a clinical diagnosis of glaucoma in 22 of 22 cats. Of these affected eyes, 21 of 22 were described as buphthalmic. Detailed medical records and follow-up information were available for 13 of 22 cats. All 13 cats with available data had abnormally high intraocular pressures as measured by applanation or rebound tonometry. Intraocular pressure values were 46 ± 3.2 mm Hg (mean ± SEM) with a range of 24 to 63 mm Hg. Aside from buphthalmos, 1 or more additional secondary signs associated with glaucoma were described in 21 of 22 cats and included corneal disease (eg, edema, exposure keratitis; n = 13), lens luxation (n = 10), cataract (n = 13), uveitis (n = 11), and blindness of the affected eye (n = 14). Corneal and lenticular disease as well as intraocular inflammation at times precluded a complete examination of the fundus (Fig. 1).
Clinical and Histological Ocular Lesions Frequently Recorded in Cats With Feline Proliferative Neovascular Vitreoretinopathy.
Abbreviation: NA, not applicable.
a The optic nerve was included in 13 slides. Due to extensive retinal disease, the histologic diagnosis of glaucoma was based on optic nerve head cupping or gliosis.
b The pupillary margin was incorporated in tissue sections from only 19 globes.
c Lens was not available in one histological section.
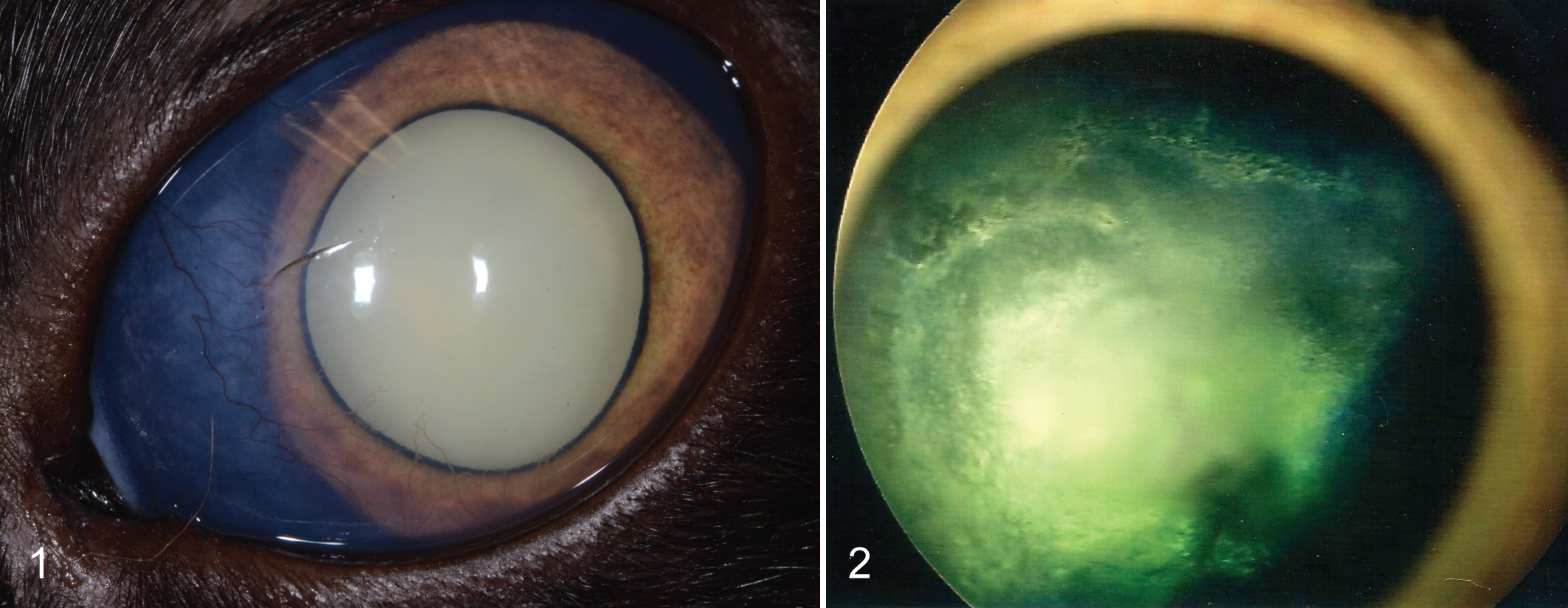
Additional clinical diagnoses were retinal detachment in 9 cats, retinal dysplasia in 3 cats, persistent fetal vasculature (suspected) in 6 cats, lenticonus in 2 cats, and microphakia in 1 cat. An avascular retina was clinically described in only 2 cats (Fig. 2).
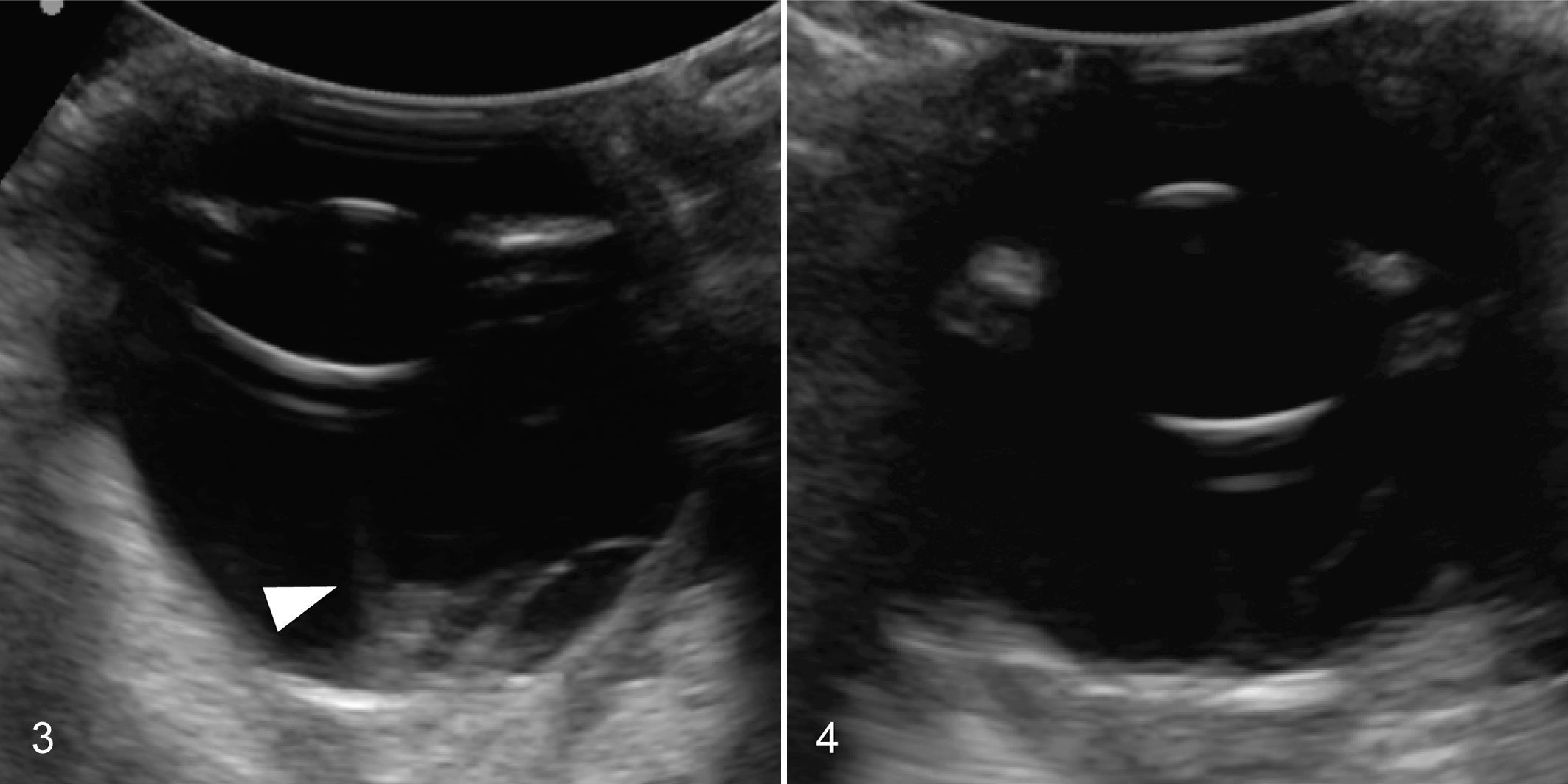
The status of the fellow eye was reported for 18 of 22 cats: the fellow eye was reported to be normal in 7 of 18 and affected in 11 of 18 to a variable degree (Figs. 3, 4), with lesions that ranged from retinal dysplasia (n = 3) to excessive retinal vascularity (n = 6), glaucoma (n = 3), and blindness (n = 2). General physical examination findings were reported for 13 cats and were unremarkable. Serum chemistry analysis and complete blood count were performed in 7 cats and were unremarkable. Serologic testing for Toxoplasma gondii IgG and IgM (n = 7 cats), feline immunodeficiency virus (FIV) and feline leukemia virus (FeLV) status (n = 8 cats), and Cryptococcus sp. antigen (n = 1 cat) were all negative. Feline coronavirus serology was performed in 2 cats, one of which was seropositive.
Histopathological Findings
Corneal lesions were present in 15 of 22 globes. Minimally affected corneas exhibited fibrosis, vascularization, and increased neutrophilic inflammatory infiltrates (n = 3/22) while more severely affected globes showed varying degrees of ulcerative (n = 9/22) and suppurative keratitis (n = 8/22).
Intraocular hemorrhage was noted in 18 of 22 eyes, including hyphema in 8 of 22 eyes and vitreal hemorrhage in 18 of 22 eyes (Fig. 5). In 3 of 22 eyes, hemosiderin and hematoidin were present within the epiretinal vascular proliferations.
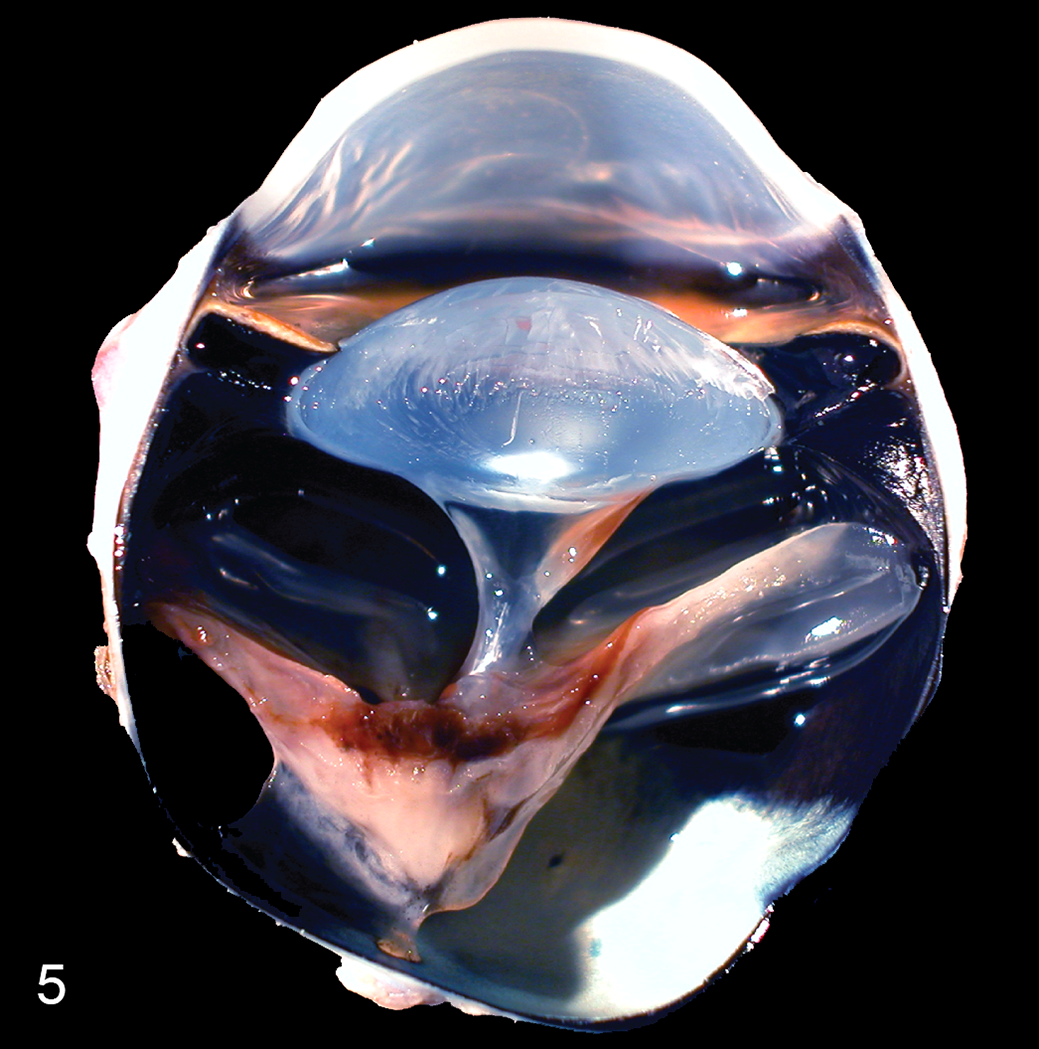
Neovascular vitreoretinopathy, cat. Section of a formalin-fixed globe. There is complete retinal detachment and epiretinal pigment deposition corresponding to hemorrhage and vasculature seen histologically (not shown). Vitreal traction bands extend from the retina to the lens.
The anterior segment was markedly affected in all globes. Anterior segment dysgenesis was identified in 21 of 22 cases. Diagnostic features of anterior segment dysgenesis (ASD) included an ill-formed iridocorneal angle substituted by mesenchymal tissue, with absence or collapse of the ciliary cleft, and an ill-defined or absent trabecular meshwork (Figs. 6, 7). In some instances, ASD was accompanied by an iris-like band of mesenchymal tissue attached to the arborizing termination of Descemet’s membrane resembling goniodysgenesis (Fig. 6). In ASD, the corneal stroma and limbal sclera often blended with each other and coincided with an absence or displacement of the scleral venous plexus (Fig. 6). In all 22 globes, a preiridal fibrovascular membrane traversed the anterior face of the iris, and in 20 of 22 globes, this preiridal fibrovascular membrane crossed the iridocorneal angle, creating peripheral anterior synechiae. Ectropion uveae were identified in 16 of the 19 eyes in which the pupil was included in the histologic sections. Other less frequent lesions included recession of the iridocorneal angle (n = 2), descemetization of the anterior iris (n = 3), ciliary body epithelial cysts (n = 4), retrocorneal membrane formation (n = 3), and persistent pupillary membranes (n = 2). Varying degrees of lymphoplasmacytic uveitis were noted in 19 of 22 globes, and 2 of 19 globes with uveitis were also diagnosed with a suppurative endophthalmitis.
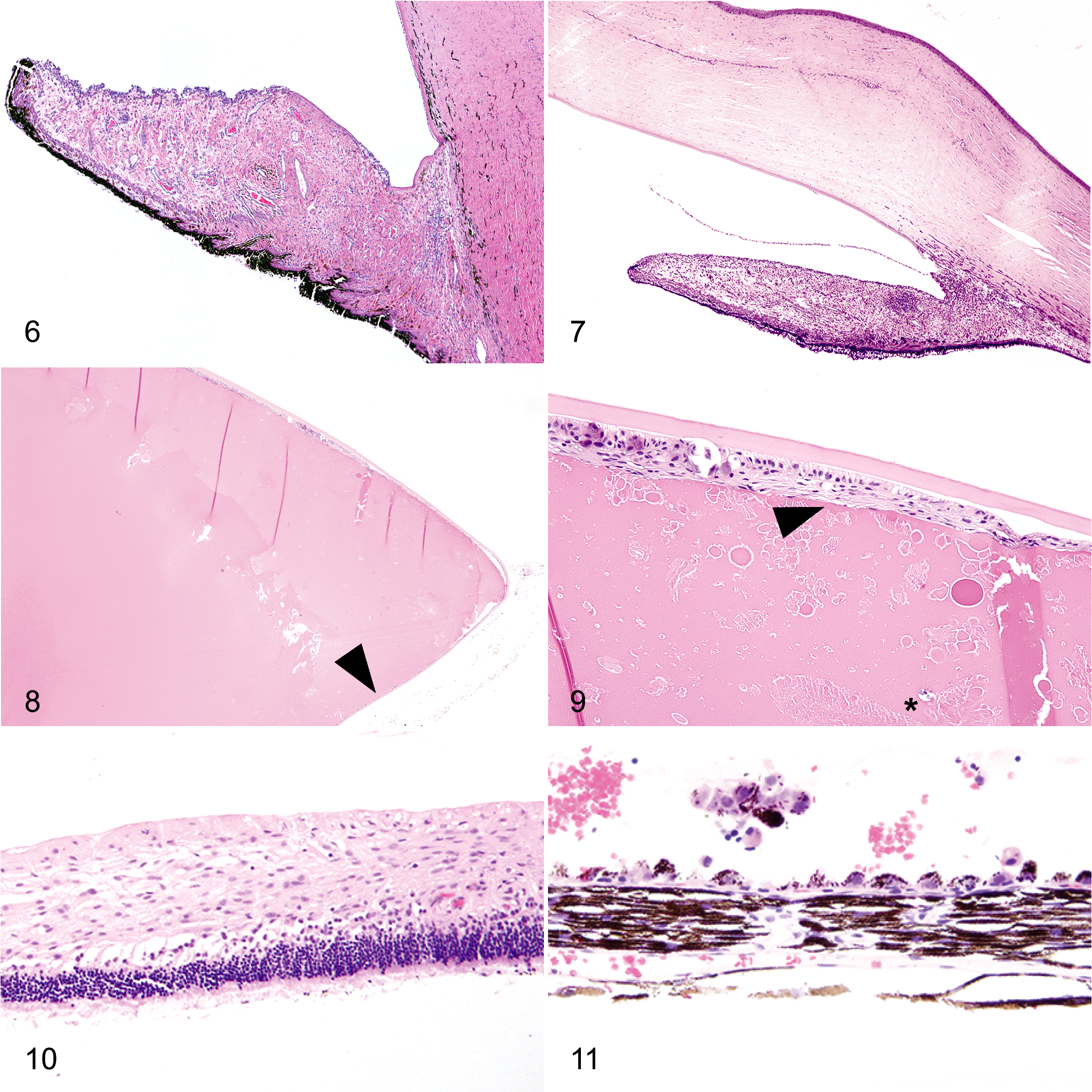
The lens was present in sections from 21 of 22 globes and was affected in 20 globes to a variable degree. Lens luxation was present in 7 of 21 globes. Cataract, identified in 20 of 21 globes, ranged in degree from localized posterior subcapsular cataract to hypermature, resorbing cataract, with formation of Morgagnian globules, mineralization, spindle cell metaplasia, and posterior migration of lens epithelial cells (Figs. 8, 9).
Retinal gliosis was noted in all 22 cats to varying degrees (Fig. 10). Complete or partial retinal detachment was noted in 19 of 22 globes (Figs. 10, 11). Other less frequently observed retinal abnormalities included retinoschisis (n = 3), retinal tears (n = 2), and retinal dysplasia (n = 1).
Optic nerves were included in sections from 13 of 22 globes and presented with varying degrees of gliosis and loss of prelaminar tissue (ie, optic nerve head cupping). The density of retinal ganglion cell somas in the inner retina was subjectively assessed to be reduced in 21 of 22 retinae, together with varying degrees of degeneration of other retinal layers (Fig. 10), supporting the clinical diagnosis of glaucoma.
Histomorphologic Diagnostic Features
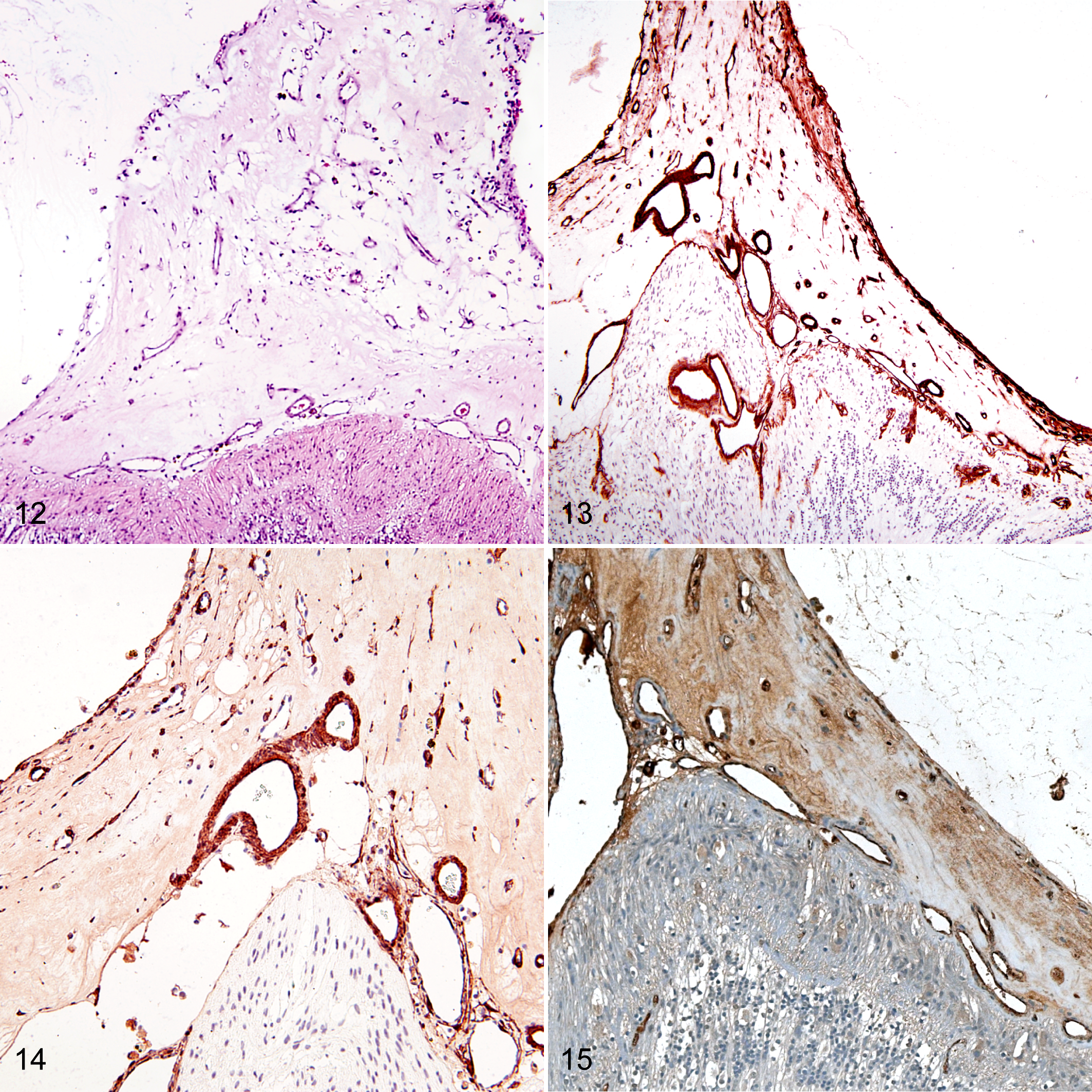
In all 22 eyes, epiretinal vascular membranes were observed internal to the neurosensory retina but still subtending a membrane continuous with the inner limiting membrane (Fig. 12). The extent of these vascular membranes varied from fine epiretinal capillary vessels to dense vascular tufts. The peripheral inner retina was avascular in all cases (Fig. 10). In all eyes, retinal vascularization seemed diminished throughout the entire retina, and in 1 eye, no retinal vessels were recognized histologically.
Immunohistochemical Findings
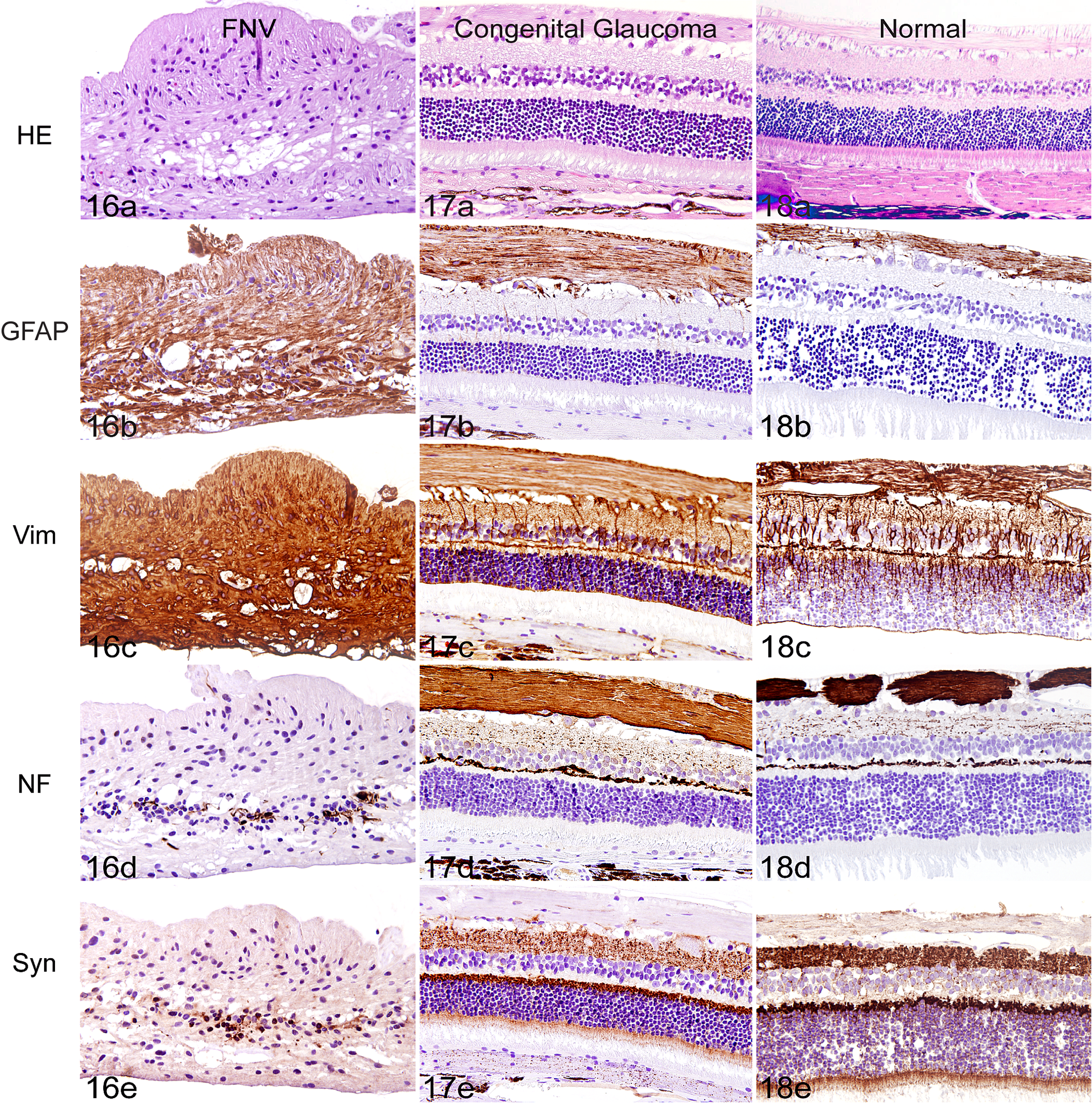
Epiretinal vascular membranes subtended a laminin-positive basal lamina that was continuous with the inner limiting membrane of the retina (7/7 sections). Laminin was also expressed in the adjacent fibrovascular tissue, which appeared contiguous with the retina and the surrounding vessels (Fig. 13). Smooth muscle actin was expressed in 7 of 7 epiretinal membranes, within pericytes of vessels and scattered spindle cells in the stroma of the epiretinal membrane (Fig. 14). FVIII-RA was expressed in endothelial cells within the epiretinal membranes (7/7 sections), as well as within vessels of the inner retina in areas that were not devoid of retinal vasculature (Fig. 15). There was intense positive immunolabeling of GFAP (7/7 sections) and vimentin (4/4 sections) throughout the retina, consistent with retinal gliosis (Fig. 16b,c). Immunolabeling of vimentin and GFAP in feline FNV was considerably more prominent than that seen in control sections from an eye with primary congenital glaucoma (Fig. 17b,c), from a normal feline eye (Fig. 18b,c), or in sections from a globe with retinal detachment (data not shown). The organized radiating structure of Müller cells occasionally seen in healthy feline retinae labeled with GFAP and vimentin 10 was occasionally noted in control eyes (Figs. 17c, 18c), but this was not identified in FNV cases where both GFAP- and vimentin-positive glial cells disrupted the lamellar organization of the retina. Neurofilament expression in affected globes (4/4 sections) was absent in the nerve fiber layer and inner plexiform layer, as well as diminished in the outer plexiform layer, compared to the strong labeling of the nerve fiber and outer plexiform layers in normal retina and retina affected by primary glaucoma (Figs. 16d, 17d, 18d). Synaptophysin labeling was reduced in the inner plexiform and nerve fiber layers in all cases (4/4 sections) when compared to controls. FNV affected retinae that exhibited a markedly disorganized lamellar structure had diminished synaptophysin labeling throughout the entire retina (Figs. 16e, 17e, 18e).
Discussion
Proliferative vitreoretinopathy is characterized by proliferation and migration of cells to form fibrocellular membranes in the vitreous and adjacent to the retina, and it is often diagnosed in people with ischemic retinal disease and rhematogenous retinal detachment. 9 Epiretinal membranes often have contractile properties, which contribute to further retinal detachment. 9 SMA-positive spindle cells seen in epiretinal membranes of affected globes in this series support a myofibroblastic cellular phenotype, which may contribute to this contractile nature. The identification of FVIII-RA-positive cells in epiretinal membranes in this series of feline globes confirmed the presence of vascular endothelial cells in these membranes.
Cases were included in this series based on the presence of these epiretinal vascularized membranes and an avascular peripheral retina, which were considered cardinal features. However, most of the cases presented in this series also exhibited ASD, as well as profound retinal gliosis. Histologically, ASD resembled that recognized in canines 13 and in New Zealand white rabbits 6,17 with early onset glaucoma. In humans, ASD is associated with congenital or primary glaucoma and was previously attributed to a developmental arrest in the third trimester of pregnancy, involving tissues derived from the neural crest and retention of primordial tissue in the iris and iridocorneal angle. 15 Specific genetic mutations are responsible for at least some forms of ASD, but the precise molecular pathogenesis responsible for these complex developmental disorders has not been completely elucidated. 16 Sporadic mentions of ASD in cats exist in the literature, 11 and at the time of writing, 45 cases of feline ASD not associated with FNV were recorded in the COPLOW database. Although ASD is not diagnostic of feline FNV, its prevalence in this series raises a possibility of a common pathogenesis for these conditions.
The cats presented in this series were younger than 6 months or had a longstanding history of ocular disease, supporting a developmental or congenital ocular syndrome. In affected cats, the left eye was more commonly or severely affected than the right eye (16 vs 5), and although females were slightly overrepresented (13 vs 8), these differences were statistically significant only for laterality. Even though this observation is based on a relatively small study population, it is noteworthy that in people, congenital ocular abnormalities, including persistent fetal vasculature, Peter’s anomaly, and unilateral congenital cataract, have been reported to occur frequently in the left eye and in females. 12 In people, left-sided and female predisposition has also been noted for Duane syndrome, a rare congenital eye movement disorder, and has been proposed to result from increased risk of thrombosis in females and thoracic asymmetry that increases sensitivity of the left side to vascular insults. 12
A history of glaucoma was reported on submission forms from all the cases in this series. In the 13 cats for which intraocular pressure (IOP) values were reported, values were above the normal reference values reported previously for both applanation and rebound tonometry (18.4 ± 0.6 mm Hg and 20.74 ± 0.5 mm Hg, respectively). 11 Glaucoma in young cats, as seen in this series, is commonly associated with marked buphthalmos, corneal edema, and exposure keratitis or ulceration. 11 The presence of intraocular inflammation is not uncommon in patients exhibiting chronic glaucoma, particularly with secondary buphthalmos and subsequent corneal disease. Both patients who exhibited suppurative endophthalmitis had a suppurative ulcerative keratitis, to which severe intraocular disease could be attributed.
In this series of feline patients, glaucoma could be of primary origin (ie, due to ASD) or secondary to extensive peripheral anterior synechiae and/or intraocular inflammation. The formation of preiridal fibrovascular membranes is commonly seen in eyes with retinal detachment, intraocular inflammation, and intraocular hemorrhage, as noted in this series. 19 Preiridal fibrovascular membranes occur due to an increase in proangiogenic factors in the eye and often result in distortion of the pupillary margin (entropion or ectropion uveae) and peripheral anterior synechiae and subsequent glaucoma. 19
Persistent fetal vasculature was suspected clinically in some cats in this series. However, in contrast to dogs, persistent ocular fetal vasculature is extremely rare in cats and has been described in only 2 cats in the veterinary literature. 1 Vitreal fibrovascular tissue and occasional persistent pupillary membranes may occur in FNV and in cats with persistent fetal vasculature; nonetheless, the FNV cases have marked fundus and anterior segment abnormalities not seen with persistent fetal vasculature, and they lack vasculature consistent with a persistent tunica vasculosa lentis.
In human infants, retinopathy of prematurity (ROP), also termed retrolental fibroplasia, results in an avascular peripheral retina, retinal detachment, and preretinal fibrovascular proliferations secondary to hyperoxic damage. 14 Experimental feline models of oxygen-induced retinopathy (OIR) exhibit similar changes to those seen in ROP, with an avascular peripheral retina and formation of preretinal vessels growing into the vitreous. 7 While the cases presented in this series exhibit similar retinal pathology to ROP and feline OIR, they are unlikely to have been exposed to hyperoxic conditions. In addition, the cases in this series have profound anterior segment disease uncommonly seen in ROP. Furthermore, in ROP, neovascular proliferation is usually peripheral rather than centrally located, as in cats in this series. Preretinal vasoproliferation contributes to retinal detachment secondary to the formation of traction bands and cicatrization in some patients with ROP, as noted in many cases in this series, but not in feline experimental OIR. Indeed, vasoproliferative retinopathy seen in kittens with experimental OIR more closely resembles that seen in people with ischemic vasoproliferative retinopathies than that seen in ROP, but in feline OIR, these proliferations regress without leaving traction bands, as seen in humans with proliferative vitreoretinopathy. 7 In the present series, retinal detachment was diagnosed histologically more frequently than clinically (19/22 vs 9/22 cats). Histological retinal detachment was distinguished from artifact based on pathological changes concomitant with true retinal detachment. 2 Discrepancies in the frequency of diagnosis of clinical and histopathological lesions seen in Table 2, including but not limited to retinal detachment, likely reflect pronounced corneal and/or lenticular disease that limits thorough clinical evaluation of intraocular structures. Inconsistency in details provided in medical records by many different clinicians is acknowledged as a common limitation of retrospective studies, and it is conceivable that clinically appreciable lesions were omitted from medical records or submission forms that accompanied enucleated globes.
Retinal gliosis seen in felines in this series was often profound, at times resembling that seen in human massive retinal gliosis. 3 Massive retinal gliosis is a nonneoplastic proliferation of retinal glial cells in the human retina attributed to a range of factors, including retinal injury, congenital retinal disease, inflammation, retinal vascular disorders, and intraocular neoplasia, 18 some of which were identified in this series of feline FNV. An increase in GFAP expression by Müller glia occurs following experimental retinal detachment in cats, 4 and GFAP and vimentin are upregulated in retinal gliosis attributed to Müller glial cells secondary to retinal disease or trauma. 10 However, gliosis in FNV exceeded that expected following retinal detachment alone and also occurred in areas where the retina was attached and in 1 of 22 globes that did not present with retinal detachment clinically or histologically. As synaptophysin and neurofilament stain normal neuronal structures, their decreased labeling in this series is consistent with either degeneration or perhaps even lack of differentiation of the retina.
Conclusions
Feline neovascular vitreoretinopathy appears to be an uncommon congenital or developmental syndrome with characteristic fundus and anterior segment pathology. Although the globes submitted in this series had been enucleated for the management of chronic, blinding glaucoma and buphthalmos, the coexisting milder lesions reported in the fellow eye of 11 of 18 cases may indicate that FNV can occur in a more subtle form that might be under- or misdiagnosed. Feline neovascular vitreoretinopathy should be considered as a differential diagnosis in young cats with anterior uveitis, elevated IOP, retinal detachment, leukocoria, and/or suspected persistence of fetal vasculature. Similarities to ROP and experimental OIR in cats suggest that an early ischemic event or developmental aberration may contribute to the pathogenesis of this condition.
Footnotes
Acknowledgements
We thank Dr. Barbara Easton, Dr. Carmen Colitz, and Dr. Andrew Greller for providing clinical images shown in this case series.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: GJM’s authorship was supported by unrestricted funds to the Department of Ophthalmology and Visual Sciences, University of Wisconsin-Madison from Research to Prevent Blindness.
