Abstract

As a diagnostic veterinary histopathologist I often find myself, more frequently than in the past, being asked to give a second opinion on a case.
Differently from the cases I had so far, I believe the following is worth sharing. A 9-year-old neutered female beagle was found bearing a 15-cm-diameter hepatic nodular mass which was biopsied (fine needle biopsy) and diagnosed as a well-differentiated hepatocellular carcinoma by a commercial lab. The animal was then referred couple of days later to the Small Animal Teaching Hospital (University of Liverpool) for surgical resection (lobectomy) and the neoplasm submitted for histopathological examination to our diagnostic service (Veterinary Laboratory Services–University of Liverpool). The pathologist who received the lobectomy sample (a colleague of mine) was not aware of the results of the initial biopsy, and a diagnosis of hepatoma was made. I was involved at this point, when the clinician dealing with the case here on campus asked me to independently review both samples (the initial biopsy from the commercial lab after requesting the slides and blocks, and the excised tumor diagnosed by my colleague) hoping in an agreement (either way) which could help the clinical decision making. I was then approaching a third (not a second) opinion on that neoplasm.
When I received such a request, I was already foreseeing the scenario in my mind, with a bit of disappointment: a very common problem related to the morphological “grey area” existing between “benign” hepatocellular tumors and “well differentiated” carcinomas, 1 in relation to which, I would probably be able to offer very little help. I was wrong.
Reading the biopsy report from the first pathologist, there was mention of 20 mitoses per 10 HPFs, while the second pathologist reported none. I then approached the slides, with a sort of bad feeling, confident that someone must have seen too many or too few mitotic figures. I was wrong again. Comparing the 2 biopsies (Figs. 1 and 2), despite being able to notice a better technical quality of the second sample (with a degree of pride), I could clearly see in both a trabecular architecture in which the hepatocytes looked relatively well differentiated. However it was evident that in the first biopsy there were large numbers of mitotic figures (Fig. 1) while I was not able to find a single one in the entire slide from the second biopsy (Fig. 2). Although I was happy that none of the pathologists were proven wrong, I was now facing a mystery: the same neoplasm was exhibiting a strikingly different mitotic activity in 2 different samples taken at different times; interestingly (and against the principle of tumor progression), the lowest mitotic count was in the most recent sample. The first hypothesis was an easy one to make: possibly the neoplasm was exhibiting areas with different degrees of differentiation, and the 2 samples were representative of these 2 different areas. If this was the explanation, since I still had the whole sample in formalin, I should have been able to find some areas of increased mitotic activity, but after sampling an additional 20 different sections of the tumor, the picture was always the same, without any mitotic figures evident—I was starting to believe that the neoplasm really was behaving differently at different times.
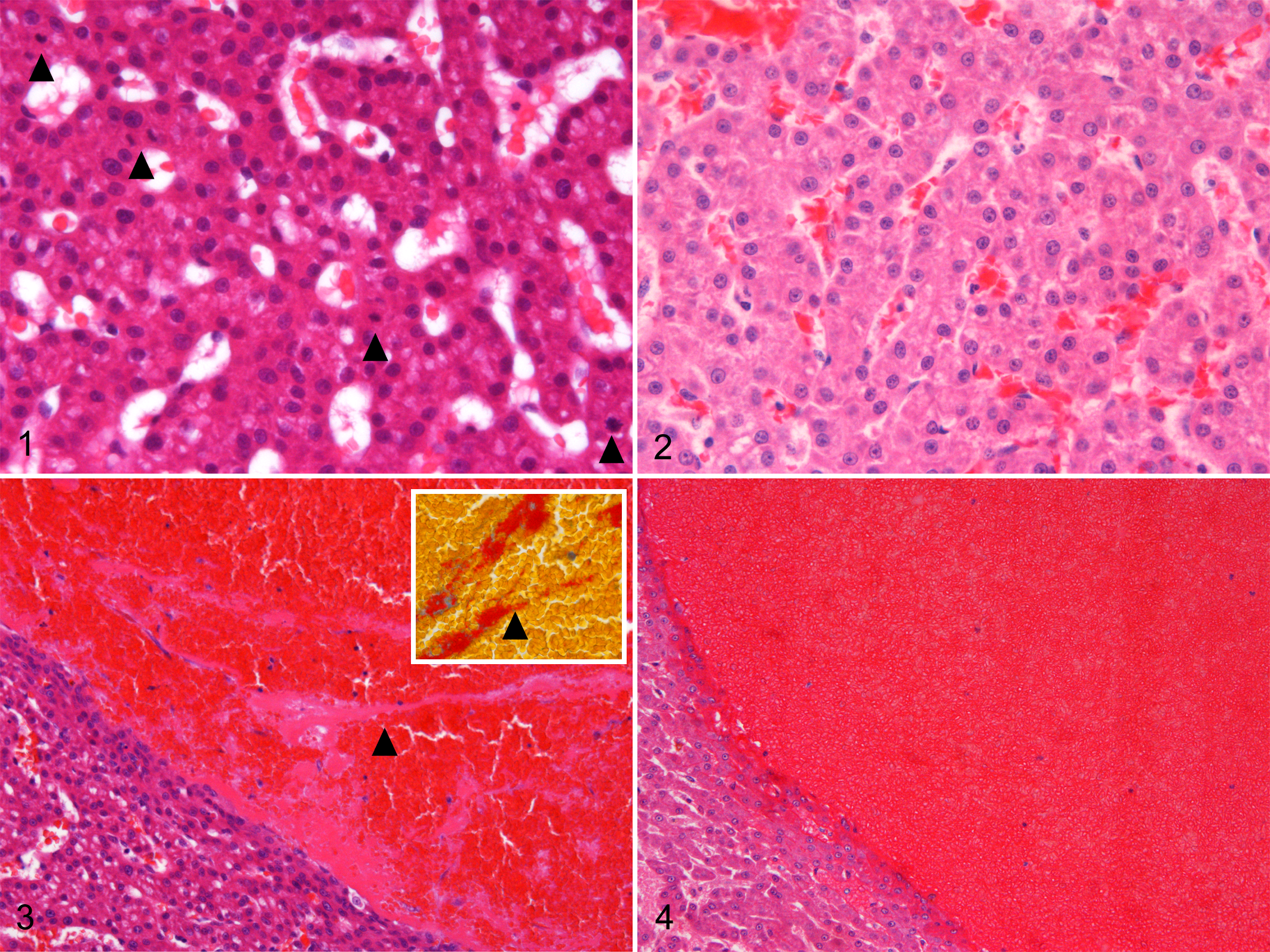
The first sample (fine needle biopsy). There is proliferation of well-differentiated hepatocytes, and large numbers of mitotic figures are evident (arrowheads). Hematoxylin and eosin (HE). Hepatoma, liver, dog.
At this point I had a breakthrough coming from the years I spent in biomedical research and in particular from some work on liver regeneration after hepatectomy in rats with which I was involved. At that time, we were observing that in certain circumstances (never clarified) the portions of the lobes we were resecting out of the liver (which we were aiming to use as an internal control for a baseline of proliferation) were instead occasionally exhibiting a significant number of mitotic figures, especially if the resected portion was not fixed immediately in formalin (data not shown). This was making a kind of a sense. I always thought that if the response of the liver to resection is to start proliferating, it is possible that even in small resected portions as long as there is ATP available (in particularly favorable conditions) it could do so, even without blood flow, as long as there was energy available. Going back to our biopsy, what if that biopsy was left for a certain amount of time in a plastic tube, without formalin, and started to proliferate as a reaction to the “resection” and possibly due to the new environment within the plastic tube? What if the second biopsy didn’t do so since it was immediately fixed? It is, as an example, recently demonstrated that cultured hepatocytes compared to cells in situ profoundly change their metabolism and phenotype just due to the change in oxygen tension. 2
To even remotely substantiate this, I needed at least the proof that the first biopsy was indeed left for a reasonable amount of time unfixed. The obvious way was to ask the Surgeon, but it is well known how much the human brain forgets unnecessary things, so not a good idea.
The forensic pathology approach once again helped me here: Looking carefully at the largest vessels present in the samples there was a striking difference. In the first biopsy it was clear that strands of fibrin were accumulating, which were nicely Martius scarlet blue positive (Fig. 3), while just blood was evident in the second biopsy (Fig. 4). With a big smile on my face, I started to write one of the most controversial comments to a second (third) opinion I ever made, happy that possibly mysteries of biology and not discrepancies between pathologists, this time, were to be blamed. Beware of “undead” biopsies!
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
